Potential of A Trait-Based Approach in the Characterization of An N-Contaminated Alluvial Aquifer
Abstract
1. Introduction
2. Materials and Methods
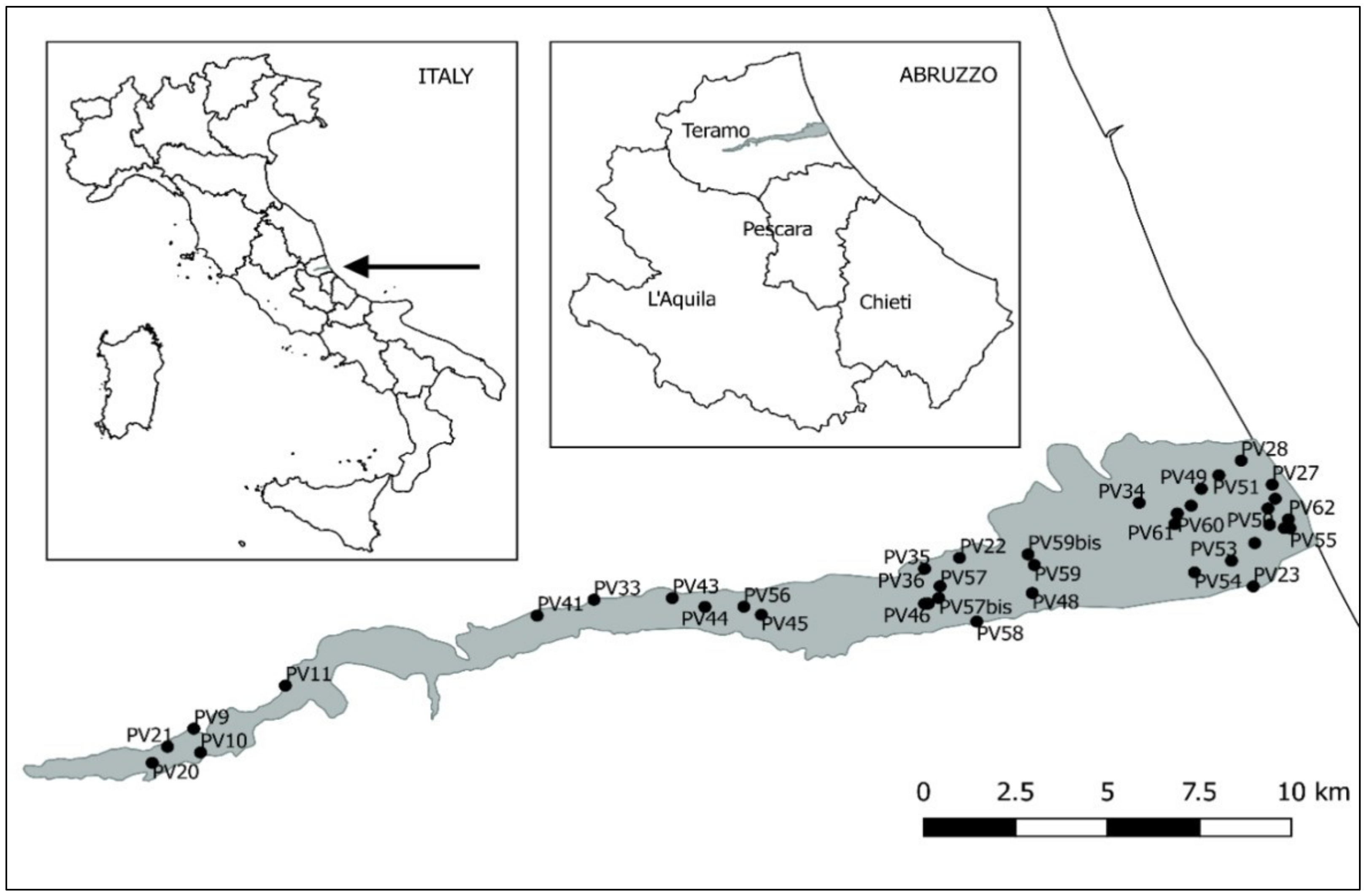
2.1. Study Area
2.2. Biological Survey
2.3. Environmental Survey
2.4. Data Analysis: Environmental Variables
2.5. Taxonomy-Based Approach
2.6. Trait-Based Approach
3. Results
3.1. Environmental Variables
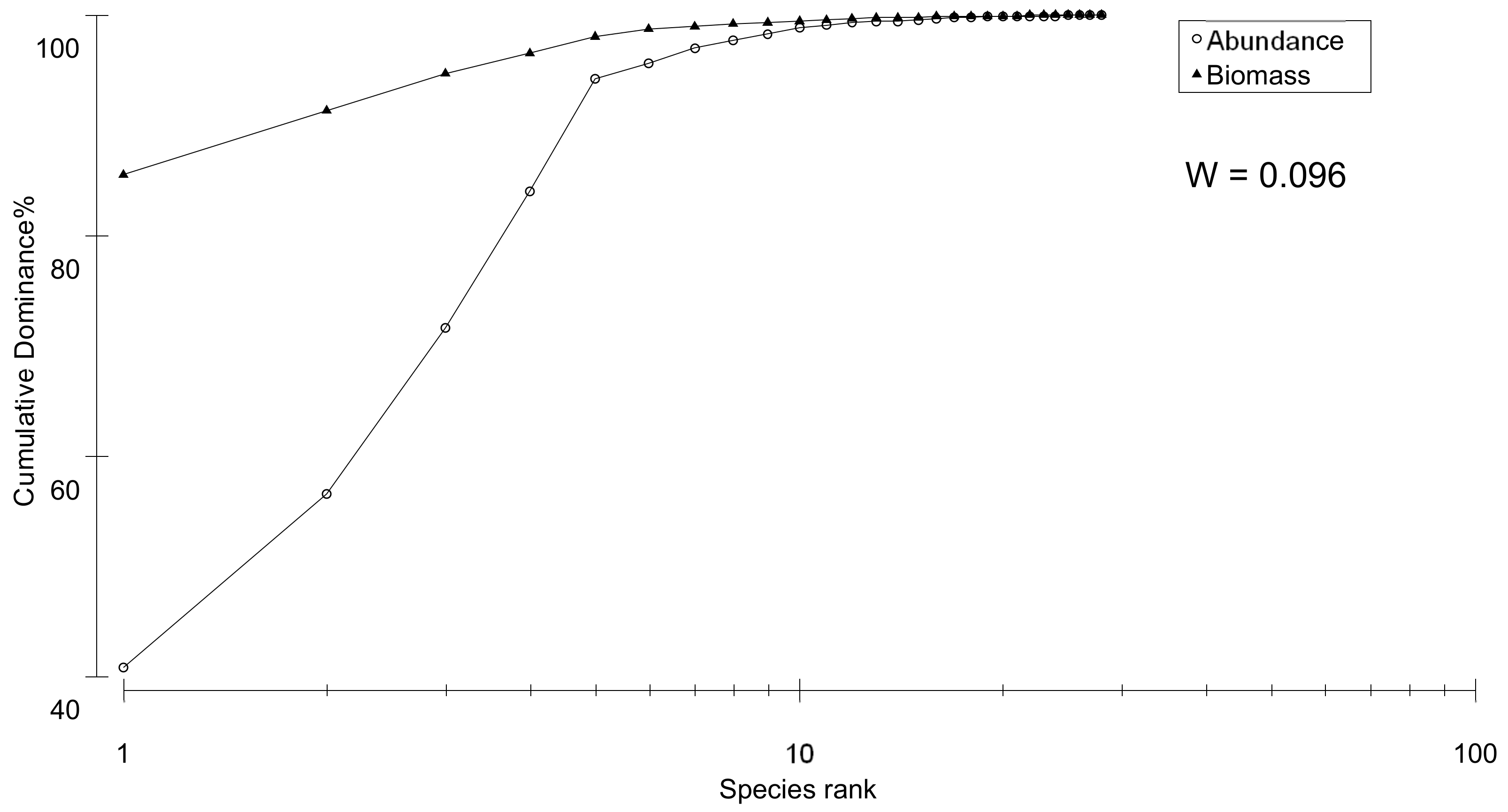
3.2. Taxonomy-Based Approach
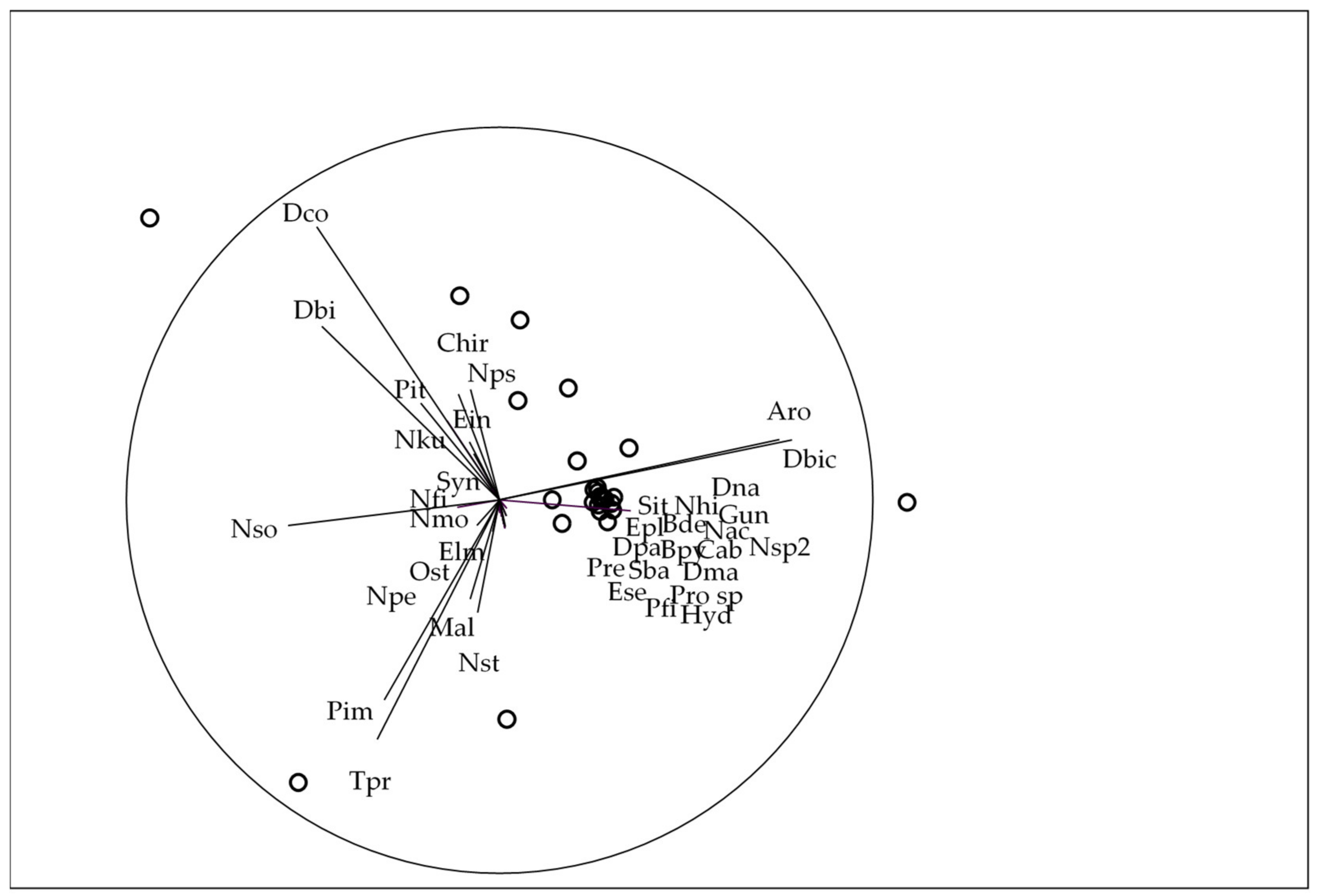
3.3. Trait-Based Approach
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Culp, J.M.; Armanini, D.G.; Dunbar, M.J.; Orlofske, J.; Poff, L.N.; Pollard, A.I.; Yates, A.G.; Hose, G.C. Incorporating traits in aquatic biomonitoring to enhance causal diagnosis and prediction. Integr. Environ. Assess. Manag. 2010, 7, 187–197. [Google Scholar] [CrossRef] [PubMed]
- Larras, F.; Coulaud, R.; Gautreau, E.; Billoir, E.; Rosebrery, J.; Usseglio-Polatera, P. Assessing anthropogenic pressures on streams: A random forest approach based on benthic diatom communities. Sci. Total Environ. 2017, 15, 1101–1112. [Google Scholar] [CrossRef] [PubMed]
- Van den Brink, P.J.; Alexander, A.; Dessrosiers, M.; Goedkoop, W.; Goethals, P.; Liess, M.; Dyer, S. Traits-based approaches in bioassessment and ecological risk assessment: Strengths, weaknesses, opportunities and threats. Integr. Environ. Assess. Manag. 2011, 7, 198–208. [Google Scholar] [CrossRef] [PubMed]
- McGill, B.J.; Enquist, B.J.; Weiher, E.; Westboy, M. Rebuilding community ecology from functional traits. Trends Ecol. Evol. 2006, 21, 178–185. [Google Scholar] [CrossRef]
- Petchey, O.L.; Gaston, K.J. Functional diversity: Back to basics and looking forward. Ecol. Lett. 2006, 9, 741–758. [Google Scholar] [CrossRef]
- Usseglio-Polatera, P.; Bournaud, M.; Richoux, P.; Tachet, H. Biological and ecological traits of benthic freshwater macroinvertebrates: Relationships and definition of groups with similar traits. Freshw. Biol. 2000, 43, 175–205. [Google Scholar] [CrossRef]
- Carter, J.L.; Resh, V.H. After site selection and before data analysis: Sampling, sorting, and laboratory procedures used in stream benthic macroinvertebrate monitoring programs by USA state agencies. J. N. Am. Benthol. Soc. 2001, 20, 658–682. [Google Scholar] [CrossRef]
- Jones, C.F. Taxonomic sufficiency: The influence of taxonomic resolution on freshwater bioassessments using benthic macroinvertebrates. Environ. Rev. 2008, 16, 45–69. [Google Scholar] [CrossRef]
- Gibert, J.; Culver, M.J.; Dole-Oliver, F.; Malard, F.; Christman, M.; Deharveng, L. Assessing and conserving groundwater biodiversity: Synthesis and perspectives. Freshw. Biol. 2009, 54, 930–941. [Google Scholar] [CrossRef]
- Pabich, W.J.; Valiela, I.; Hemond, H.F. Relationship between DOC concentration and vadose zone thickness and depth below water table in groundwater of Cape Cod, U.S.A. Biogeochemistry 2001, 55, 247–268. [Google Scholar] [CrossRef]
- Shen, Y.; Chapelle, F.H.; Benner, R. Origins and bioavailability of dissolved organic matter in groundwater. Biogeochemistry 2015, 122, 61–78. [Google Scholar] [CrossRef]
- Boulton, A.J. River ecosystem health down under: Assessing ecological condition in riverine groundwater zones in Australia. Ecosyst. Health 2001, 6, 108–118. [Google Scholar] [CrossRef]
- Francois, C.M.; Mamillod Blondin, F.; Malard, F.; Fourel, F.; Lécuyer, C.; Douady, C.J.; Simon, L. Trophic ecology of groundwater species reveals specialization in a low-productivity environment. Funct. Ecol. 2016, 30, 262–273. [Google Scholar] [CrossRef]
- Hofmann, R.; Griebler, C. DOM and bacterial growth efficiency in oligotrophic groundwater: Absence of priming and co-limitation by organic carbon and phosphorus. Aquat. Microb. Ecol. 2018, 81, 55–71. [Google Scholar] [CrossRef]
- Stoch, F.; Galassi, D.M.P. Stygobiotic crustacean species richness: A question of numbers, a matter of scale. Hydrobiologia 2010, 653, 217–234. [Google Scholar] [CrossRef]
- Strona, G.; Fattorini, S.; Fiasca, B.; Di Lorenzo, T.; Di Cicco, M.; Lorenzetti, W.; Galassi, D.M.P. AQUALIFE software: A new tool for a standardized ecological assessment of groundwater dependent ecosystems. Water 2019. (accepted). [Google Scholar]
- Humphreys, W. Aquifers: The ultimate groundwater dependent ecosystem. Aust. J. Bot. 2006, 54, 115–132. [Google Scholar] [CrossRef]
- Galassi, D.M.P.; Huys, R.; Reid, J. Diversity ecology and evolution of groundwater copepods. Freshw. Biol. 2009, 54, 691–708. [Google Scholar] [CrossRef]
- Di Lorenzo, T.; Cipriani, D.; Fiasca, B.; Rusi, S.; Galassi, D.M.P. Groundwater drift monitoring as a tool to assess the spatial distribution of groundwater species into karst aquifers. Hydrobiologia 2018, 813, 137–156. [Google Scholar] [CrossRef]
- Malard, F.; Mathieu, J.; Reygrobellet, J.L.; Lafont, M. Biomonitoring groundwater contamination: Application to a karst area in Southern France. Aquat. Sci. 1996, 58, 158–187. [Google Scholar] [CrossRef]
- Di Lorenzo, T.; Galassi, D.M.P. Agricultural impact in Mediterranean alluvial aquifers: Do groundwater communities respond? Fundam. Appl. Limnol. 2013, 182, 271–282. [Google Scholar] [CrossRef]
- Di Lorenzo, T.; Cifoni, M.; Lombardo, P.; Fiasca, B.; Galassi, D.M.P. Ammonium threshold value for groundwater quality in the EU may not protect groundwater fauna: Evidence from an alluvial aquifer in Italy. Hydrobiologia 2015, 743, 139–150. [Google Scholar] [CrossRef]
- Caschetto, M.; Barbieri, M.; Galassi, D.M.P.; Mastrorillo, L.; Rusi, S.; Stoch, F.; Di Cioccio, A.; Petitta, M. Human alteration of groundwater-surface water interactions (Sagittario River, Central Italy): Implication for flow regime, contaminant fate and invertebrate response. Environ. Earth Sci. 2014, 7, 1791–1807. [Google Scholar] [CrossRef]
- Reiss, J.; Perkins, D.M.; Fussmann, K.E.; Krause, S.; Canhoto, C.; Romeijn, P.; Roberton, A.L. Groundwater flooding: Ecosystem structure following an extreme recharge event. Sci. Total Environ. 2019, 625, 1252–1260. [Google Scholar] [CrossRef] [PubMed]
- Wilhelm, F.M.; Taylor, S.J.; Adams, G.L. Comparison of routine metabolic rates of the stygobite, Gammarus acherondytes (Amphipoda: Gammaridae) and the stygophile Gammarus troglophilus. Freshw. Biol. 2006, 51, 1162–1174. [Google Scholar] [CrossRef]
- Mezek, T.; Simčič, T.; Arts, M.T.; Bracelj, A. Effect of fasting on hypogean (Niphargus stygius) and epigean (Gammarus fossarum) amphipods: A laboratory study. Aquat. Ecol. 2010, 44, 397–408. [Google Scholar] [CrossRef]
- Descloux, S.; Datry, T.; Usseglio-Polatera, P. Trait-based structure of invertebrates along a gradient of sediment colmation: Benthos versus hyporheos responses. Sci. Total Environ. 2014, 466–467, 265–276. [Google Scholar] [CrossRef]
- Mathers, K.L.; Hill, M.J.; Wood, C.D.; Wood, P.J. The role of fine sediment characteristics and body size on the vertical movement of a freshwater amphipod. Freshw. Biol. 2018, 64, 152–163. [Google Scholar] [CrossRef]
- Sarkka, J.; Levonen, L.; Makela, J. Meiofauna of springs in Finland in relation to environmental factors. Hydrobiologia 1997, 347, 139–150. [Google Scholar] [CrossRef]
- Mauclaire, L.; Gibert, J.; Claret, C. Do bacteria and nutrients control faunal assemblages in alluvial aquifers? Arch. Hydrobiol. 2000, 148, 85–98. [Google Scholar] [CrossRef]
- Pacioglu, O.; Moldovan, O.T. Response of invertebrates from the hyporheic zone of chalk rivers to eutrophication and land use. Environ. Sci. Pollut. Res. 2016, 23, 4729–4740. [Google Scholar] [CrossRef] [PubMed]
- Boy-Roura, M.; Nolan, B.T.; Menció, A.; Mas-Pla, J. Regression model for aquifer vulnerability assessment of nitrate pollution in the Osona region (NE Spain). J. Hydrol. 2013, 505, 150–162. [Google Scholar] [CrossRef]
- Malard, F. Groundwater contamination and ecological monitoring in a Mediterranean karst ecosystem in Southern France. In Groundwater Ecology: A Tool for Management of Water Resources; Griebler, C., Danielopol, D., Gibert, J., Nachtnebel, H.P., Notenboom, J., Eds.; Official Publication of the European Communities: Luxembourg, 2001; pp. 183–194. [Google Scholar]
- Korbel, K.L.; Hose, G.C. A tiered framework for assessing groundwater ecosystem health. Hydrobiologia 2011, 661, 329–349. [Google Scholar] [CrossRef]
- Marmonier, P.; Maazouzi, C.; Baran, N.; Blanchet, S.; Ritter, A.; Saplairoles, M.; Dole-Olivier, M.J.; Galassi, D.M.; Eme, D.; Dolédec, S.; et al. Ecology-based evaluation of groundwater ecosystems under intensive agriculture: A combination of community analysis and sentinel exposure. Sci. Total Environ. 2018, 613–614, 1353–1366. [Google Scholar] [CrossRef] [PubMed]
- Regione Abruzzo. Piano di Tutela delle Acque. Relazione Generale e Allegati. 2010. Available online: http://www.regione.abruzzo.it/pianoTutelaacque/index.asp?modello=elaboratiPiano&servizio=lista&stileDiv=elaboratiPiano (accessed on 10 October 2019).
- Desiderio, G.; Nanni, T.; Rusi, S. La pianura del fiume Vomano (Abruzzo): Idrogeologia, antropizzazione e suoi effetti sul depauperamento della falda. Boll. Soc. Geol. Ital. 2003, 122, 421–434. [Google Scholar]
- EC (European Community). Directive 2000/60/EC of the European Parliament and of the Council of 23 October 2000 establishing a framework for Community action in the field of water policy. Off. J. 2000, L 327, 1–73. [Google Scholar]
- EC (European Community). Directive 2006/118/EC of the European Parliament and of the Council of 12 December 2006 on the protection of groundwater against pollution and deterioration. Off. J. 2006, L 327/19, 1–31. [Google Scholar]
- Cvetkov, L. Un fi let phréatobiologique. Bulletin de l’Institut de Zoologie et Musée Sofia 1968, 22, 215–219. [Google Scholar]
- Hancock, P.J.; Boulton, A.J. Sampling groundwater fauna: Efficiency of rapid assessment methods tested in bores in eastern Australia. Freshw. Biol. 2009, 54, 902–917. [Google Scholar] [CrossRef]
- Dussart, B.; Defaye, D. World Directory of Crustacea Copepoda of Inland Waters. II—Cyclopiformes; Backhuys Publishers: Leiden, The Netherlands, 2009; pp. 1–276. [Google Scholar]
- Boxshall, G.A.; Halsey, S.H. An Introduction to Copepod Diversity; The Ray Society: London, UK, 2004. [Google Scholar]
- Gibert, J.; Stanford, J.A.; Dole-Oliver, M.J.; Ward, J. Basic attributes of groundwater ecosystems and prospects for research. In Groundwater Ecology; Gibert, J., Danielopol, D., Stanford, J., Eds.; Academic Press: California, CA, USA, 1994; pp. 7–40. [Google Scholar]
- Reiss, J.; Schmid-Araya, J.M. Existing in plenty: Abundance, biomass and diversity of ciliates and meiofauna in small streams. Freshw. Biol. 2008, 53, 652–668. [Google Scholar] [CrossRef]
- Warwick, R.M.; Gee, J.M. Community structure of estuarine meiobenthos. Mar. Ecol. Prog. Ser. 1984, 18, 97–111. [Google Scholar] [CrossRef]
- Feller, R.J.; Warwick, R.M. Introduction to the study of meiofauna. In Energetics; Higgins, R.P., Thiel, H., Eds.; Smithsonian Institution Press: Washington, DC, USA, 1988; pp. 181–196. [Google Scholar]
- Hurlbert, S.H. Pseudoreplication and the design of ecological field experiments. Ecol. Monog. 1984, 54, 187–211. [Google Scholar] [CrossRef]
- Clarke, K.R.; Gorley, R.N. PRIMER v6: User Manual/Tutorial; PRIMER-E: Playmouth, UK, 2006. [Google Scholar]
- Clarke, K.R.; Green, R.H. Statistical design and analysis for a ‘biological effects’ study. Mar. Ecol. 1988, 46, 213–226. [Google Scholar] [CrossRef]
- Magurran, A.E.; McGill, B.J. Biological Diversity: Frontiers in Measurement and Assessment; University Press: Oxford, UK, 2011. [Google Scholar]
- Clarke, K.R.; Warwick, R.M. Change in Marine Communities: An Approach to Statistical Analysis and Interpretation, 2nd ed.; Playmouth Marine Laboratory: Playmouth, UK, 2001. [Google Scholar]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2013; Available online: http://www.R-project.org/ (accessed on 2 December 2019).
- Krupa, E.G. Population densities, sex ratios of adults, and occurrence of malformations in three species of cyclopoid copepods in waterbodies with different degrees of eutrophy and toxic pollution. J. Mar. Sci. Technol. 2005, 13, 226–237. [Google Scholar]
- Peschke, K.; Jonas, G.; Kohler, H.-R.; Karl, W.; Rita, T. Invertebrates as indicators for chemical stress in sewage-influenced stream systems: Toxic and endocrine effects in gammarids and reactions at the community level in two tributaries of Lake Constance, Schussen and Argen. Ecotoxicol. Environ. Saf. 2014, 106, 115–125. [Google Scholar] [CrossRef]
- Ozga, V.A.; da Silva de Castro, V.; da Silva Castiglioni, D. Population structure of two freshwater amphipods (Crustacea: Peracarida: Hyalellidae) from southern Brazil. Nauplius 2018, 26, e2018025. [Google Scholar] [CrossRef]
- Di Marzio, W.D.; Castaldo, D.; Di Lorenzo, T.; Di Cioccio, A.; Sáenz, M.E.; Galassi, D.M.P. Developmental endpoints of chronic exposure to suspected endocrine-disrupting chemicals on benthic and hyporheic freshwater copepods. Ecotoxicol. Environ. Saf. 2013, 96, 86–92. [Google Scholar] [CrossRef]
- Di Lorenzo, T.; Di Marzio, W.D.; Sáenz, M.E.; Baratti, M.; Dedonno, A.A. Iannucci, A.; Cannicci, S.; Messana, G.; Galassi, D.M. Sensitivity of hypogean and epigean freshwater copepods to agricultural pollutants. Environ. Sci. Pollut. Res. 2014, 21, 4643–4655. [Google Scholar] [CrossRef]
- Repubblica Italiana. Decreto Legislativo 16 Marzo 2009, n. 30: Attuazione della direttiva 2006/118/CE, relativa alla protezione delle acque sotterranee dall’inquinamento e dal deterioramento. Gazzetta Ufficiale 2009, 49, 1–45. [Google Scholar]
- Korbel, K.; Chartion, A.; Stephenson, S.; Greenfield, P.; Hose, G.C. Wells provide a distorted view of life in the aquifer: Implications for sampling, monitoring and assessment of groundwater ecosystems. Sci. Rep. 2017, 7, 40702. [Google Scholar] [CrossRef] [PubMed]
- Menció, A.; Korbel, K.L.; Hose, G.C. River–aquifer interactions and their relationship to stygofauna assemblages: A case study of the Gwydir River alluvial aquifer (New South Wales, Australia). Sci. Total Environ. 2014, 479–480, 292–305. [Google Scholar] [CrossRef] [PubMed]
- Dumas, P.; Bou, C.; Gibert, J. Groundwater macrocrustaceans as natural indicators of the Ariège alluvial aquifer. Int. Rev. Hydrobiol. 2001, 86, 619–633. [Google Scholar] [CrossRef]
- Hahn, H.J. The GW-Fauna-Index: A first approach to a quantitative ecological assessment of groundwater habitats. Limnologica 2006, 36, 119–137. [Google Scholar] [CrossRef]
- Galassi, D.M.P.; Stoch, F.; Fiasca, B.; Di Lorenzo, T.; Gattone, E. Groundwater biodiversity patterns in the Lessinian Massif of northern Italy. Freshw. Biol. 54, 830–847. [CrossRef]
- Brancelj, A.; Zibrat, U.; Jamnik, B. Differences between groundwater fauna in shallow and in deep intergranular aquifers as an indication of different characteristics of habitats and hydraulic connections. J. Limnol. 2016, 75, 248–261. [Google Scholar] [CrossRef]
- Iepure, S.; Rasines-Ladero, R.; Meffe, R.; Carreno, F.; Mostaza, D.; Sundberg, A.; Di Lorenzo, T.; Barroso, J.L. The role of groundwater crustaceans in disentangling aquifer type features—A case study of the Upper Tagus Basin, central Spain. Ecohydrology 2017, 10, e1876. [Google Scholar] [CrossRef]
- Shaw, P.J.A. Multivariate Statistics for the Environmental Sciences; Oxford University Press Inc.: Oxford, UK, 2003; pp. 1–231. [Google Scholar]
- Deharveng, L.; Stoch, F.; Gibert, J.; Bedos, A.; Galassi, D.M.P.; Zagmajster, M.; Brancelj, A.; Camacho, A.; Fiers, F.; Martin, P.; et al. Groundwater biodiversity in Europe. Freshw. Biol. 2009, 54, 709–726. [Google Scholar] [CrossRef]
- Mahi, A.; Di Lorenzo, T.; Haicha, B.; Belaidi, N.; Taleb, A. Environmental factors determining regional biodiversity patterns of groundwater fauna in semi-arid aquifers of northwest Algeria. Limnology 2019, 20, 309–320. [Google Scholar] [CrossRef]
- Stoch, F.; Artheau, M.; Brancelj, A.; Galassi, D.M.P.; Malard, F. Biodiversity indicators in European ground waters: Towards a predictive model of stygobiotic species richness. Freshw. Biol. 2009, 54, 745–755. [Google Scholar] [CrossRef]
- Camargo, J.A.; Alonso, A. Ecological and toxicological effects of inorganic nitrogen pollution in aquatic ecosystems: A global assessment. Environ. Int. 2006, 32, 831–849. [Google Scholar] [CrossRef]
- Di Lorenzo, T.; Di Marzio, W.D.; Fiasca, B.; Galassi, D.M.P.; Korbel, K.; Iepure, S.; Pereira, J.L.; Reboleira, A.S.; Schmidt, S.I.; Hose, G.C. Recommendations for ecotoxicity testing with stygobiotic species in the framework of groundwater environmental risk assessment. Sci. Total Environ. 2019, 681, 292–304. [Google Scholar] [CrossRef] [PubMed]
- Korbel, K.L.; Hose, G.C. Habitat, water quality, seasonality, or site? Identifying environmental correlates of the distribution of groundwater biota. Freshw. Sci. 2015, 34, 329–343. [Google Scholar] [CrossRef]
- Fattorini, S.; Lombardo, P.; Fiasca, B.; Di Cioccio, A.; Di Lorenzo, T.; Galassi, D.M.P. Earthquake-related changes in species spatial niche overlaps in spring communities. Sci. Rep. 2017, 7, 443. [Google Scholar] [CrossRef] [PubMed]
- Stumpp, C.; Hose, G.C. Groundwater amphipods alter aquifer sediment structure. Hydrol. Process. 2017, 31, 3452–3454. [Google Scholar] [CrossRef]
- Hose, G.C.; Stumpp, C. Architects of the underworld: Bioturbation by groundwater invertebrates influences aquifer hydraulic properties. Aquat. Sci. 2019, 81, 20. [Google Scholar] [CrossRef]
- Korbel, K.L.; Stephenson, S.; Hose, G.C. Sediment size influences habitat selection and use by groundwater macrofauna and meiofauna. Aquat. Sci. 2019, 81, 39. [Google Scholar] [CrossRef]
- Bork, J.; Berkhoff, S.; Bork, S.; Hahn, J.H. Using subsurface metazoan fauna to indicate groundwater–surface water interactions in the Nakdong River floodplain, South Korea. Hydrogeol. J. 2009, 17, 61–75. [Google Scholar] [CrossRef]
- Hahn, H.J.; Fuchs, A. Distribution patterns of groundwater communities across aquifer types in south-western Germany. Freshw. Biol. 2008, 54, 848–860. [Google Scholar] [CrossRef]
- Galassi, D.M.P. Groundwater copepods: Diversity patterns over ecological and evolutionary scales. Hydrobiologia 2001, 454–453, 227–253. [Google Scholar] [CrossRef]
- Migliore, L.; de Nicola Giudici, M. Toxicity of heavy metals to Asellus aquaticus (L.) (Crustacea, Isopoda). Hydrobiologia 1990, 203, 155–164. [Google Scholar] [CrossRef]
- Kadiene, U.E.; Bialais, C.; Ouddane, B.; Hwang, J.-S.; Souissi, S. Differences in lethal response between male and female calanoid copepods and life-cycle traits to cadmium toxicity. Ecotoxicology 2017, 26, 1227–1239. [Google Scholar] [CrossRef]
- Fisher, R.A. The Genetical Theory of Natural Selection; Clarendon Press: Oxford, UK, 1930. [Google Scholar]
- Leigh, E.G. Natural Selection and Mutability. Am. Nat. 1970, 104, 301–305. [Google Scholar] [CrossRef]

| Var. | Mean | SD | TV |
|---|---|---|---|
| Alt | 72.7 | 72.3 | |
| W.t. | 8.3 | 5.3 | |
| T | 16.9 | 1.7 | |
| EC | 1166 | 231 | |
| pH | 7.3 | 0.2 | |
| O2 | 4.7 | 1.6 | |
| POM | 0.73 | 2.32 | |
| TOC | 3.02 | 2.39 | |
| DOC | 2.18 | 1.70 | |
| NO2− | 10.25 | 11.19 | 0.5 |
| NO3− | 61.60 | 30.07 | 50 |
| SO42− | 103.75 | 51.97 | 250 |
| Cl− | 55.76 | 37.49 | |
| PO43− | 0.27 | 0.42 | |
| NH4+ | 0.23 | 0.50 | 0.5 |
| Ca2+ | 144.99 | 30.13 | |
| K+ | 5.22 | 2.312 | |
| Na+ | 60.17 | 33.31 | |
| DIC | 0.03 | 0.043 | 0.05 |
| TCE | 0.07 | 0.132 | 1.1 |
| CHL | 0.04 | 0.046 | 1.5 |
| THC | 25.7 | 4.480 | 350 |
| Country | Alluvial Aquifer | Area | T | SB | nSB | C | Co | NO3− | NO2− | NH4+ | REF |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Australia | Murray | 97,000 | 11 | 8 | 2 | 2.4 | 0.04 | [60] | |||
| Australia | Gwydir | 26,600 | 21 | >5 | 8 | 2 | 8.0 | [61] | |||
| France | Ariège | 250 | 10 | 10 | 0 | 10 | 0 | 90.0 | 31 | [62] | |
| France | Ariège and Hers | 540 | 36 | 14 | 22 | 4 | >50.0 | [35] | |||
| Germany | Rhine | 5000 | 35 | 16 | 22 | 35 | 23 | 4.2 | 0.33 | [63] | |
| Italy | Vibrata | 48 | 18 | 9 | 9 | 14 | 12 | 72.5 | [21] | ||
| Italy | Adige | 45 | 15 | 9 | 6 | 15 | 15 | 7.8 | 0.01 | 0.16 | [22] |
| Italy | Lessinia | 428 | 21 | 20 | 10 | 29.7 | [64] | ||||
| Italy | Vomano | 30 | 38 | 21 | 17 | 35 | 28 | 61.6 | 10.25 | 0.23 | (ts) |
| Slovenia | Brest | 6 | 38 | 22 | 16 | 23 | 15 | 2.5 | [65] | ||
| Spain | Tajo | 201 | 11 | 3 | 8 | 11 | 6 | 11.4 | 0.02 | 0.19 | [66] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Di Lorenzo, T.; Murolo, A.; Fiasca, B.; Tabilio Di Camillo, A.; Di Cicco, M.; Galassi, D.M.P. Potential of A Trait-Based Approach in the Characterization of An N-Contaminated Alluvial Aquifer. Water 2019, 11, 2553. https://doi.org/10.3390/w11122553
Di Lorenzo T, Murolo A, Fiasca B, Tabilio Di Camillo A, Di Cicco M, Galassi DMP. Potential of A Trait-Based Approach in the Characterization of An N-Contaminated Alluvial Aquifer. Water. 2019; 11(12):2553. https://doi.org/10.3390/w11122553
Chicago/Turabian StyleDi Lorenzo, Tiziana, Alessandro Murolo, Barbara Fiasca, Agostina Tabilio Di Camillo, Mattia Di Cicco, and Diana Maria Paola Galassi. 2019. "Potential of A Trait-Based Approach in the Characterization of An N-Contaminated Alluvial Aquifer" Water 11, no. 12: 2553. https://doi.org/10.3390/w11122553
APA StyleDi Lorenzo, T., Murolo, A., Fiasca, B., Tabilio Di Camillo, A., Di Cicco, M., & Galassi, D. M. P. (2019). Potential of A Trait-Based Approach in the Characterization of An N-Contaminated Alluvial Aquifer. Water, 11(12), 2553. https://doi.org/10.3390/w11122553







