Efficient Degradation of Mordant Blue 9 Using the Fenton-Activated Persulfate System
Abstract
1. Introduction
2. Experimental
2.1. Reagents and Materials
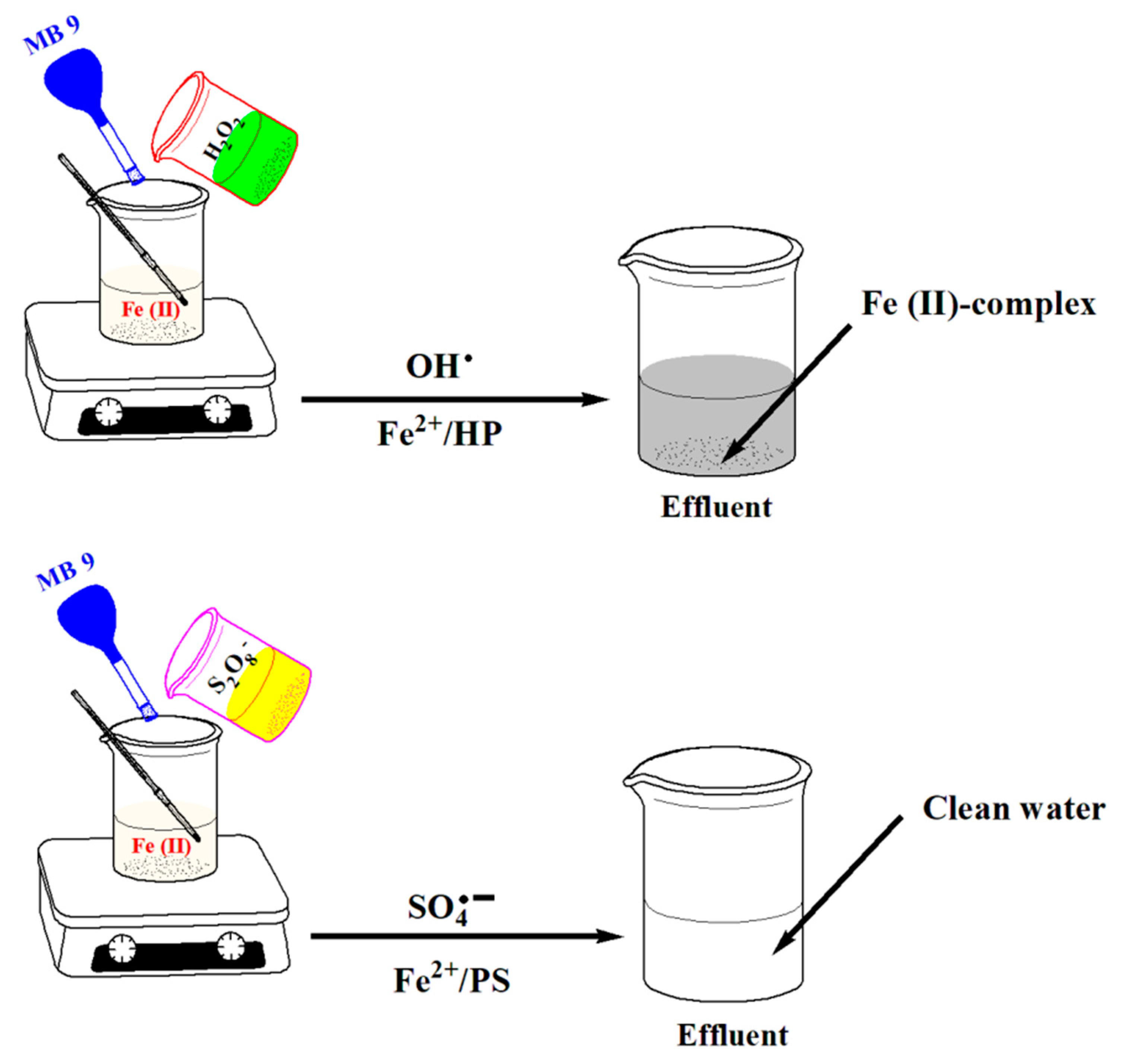
2.2. MB 9 Degradation Procedures
2.3. Analytical Methods
3. Results and Discussion
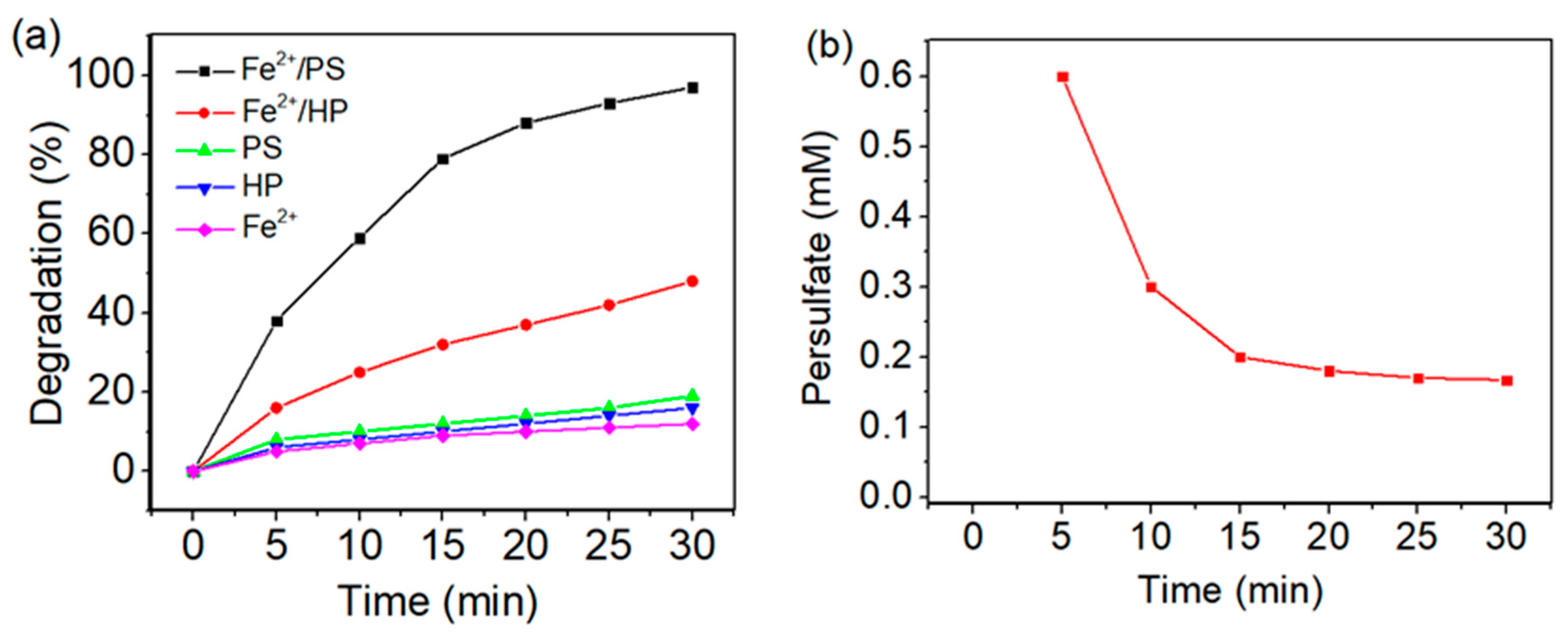
3.1. Comparative Studies on MB 9 Degradation Efficiency
3.2. Effect of Operational Parameters
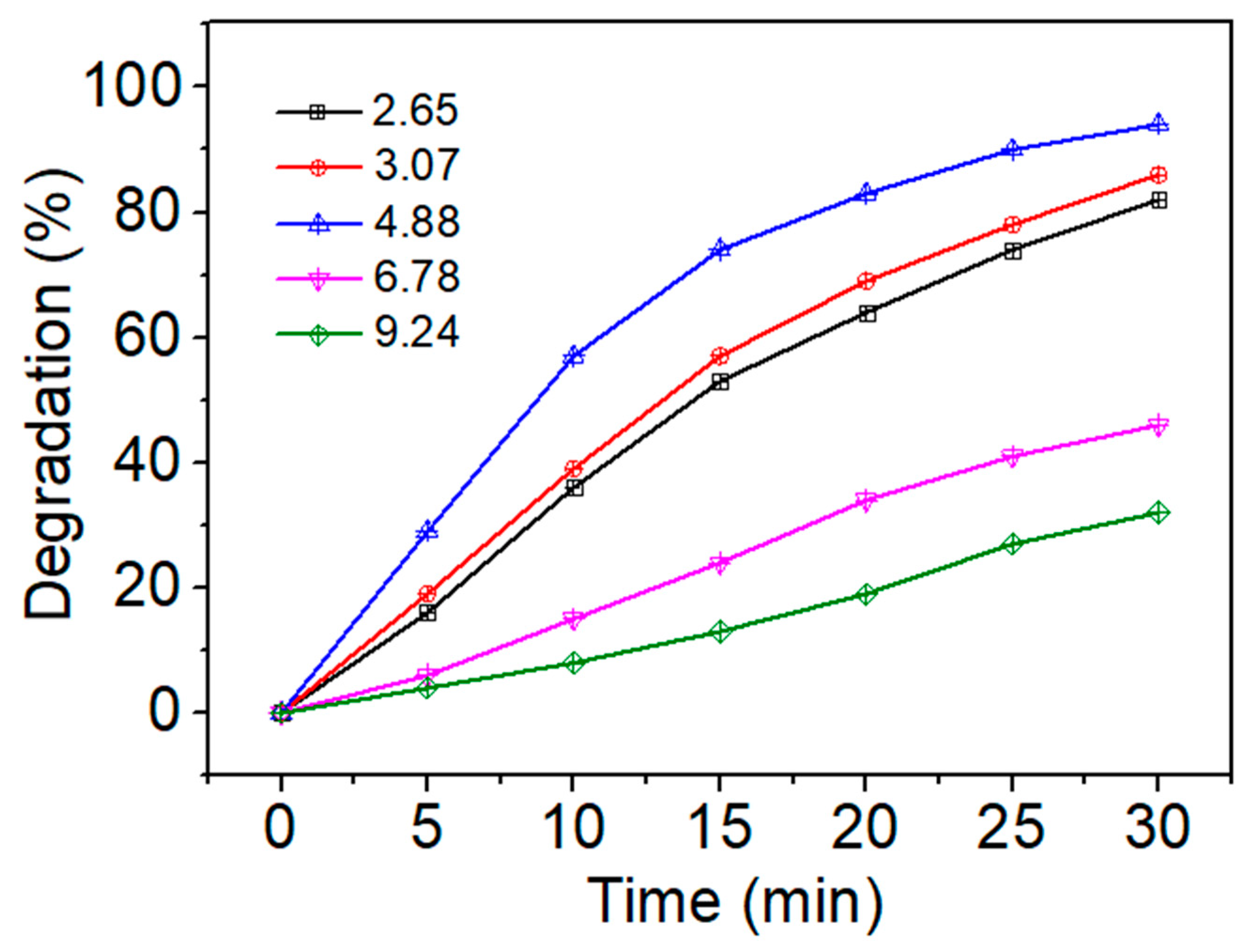
3.2.1. Effect of Initial pH
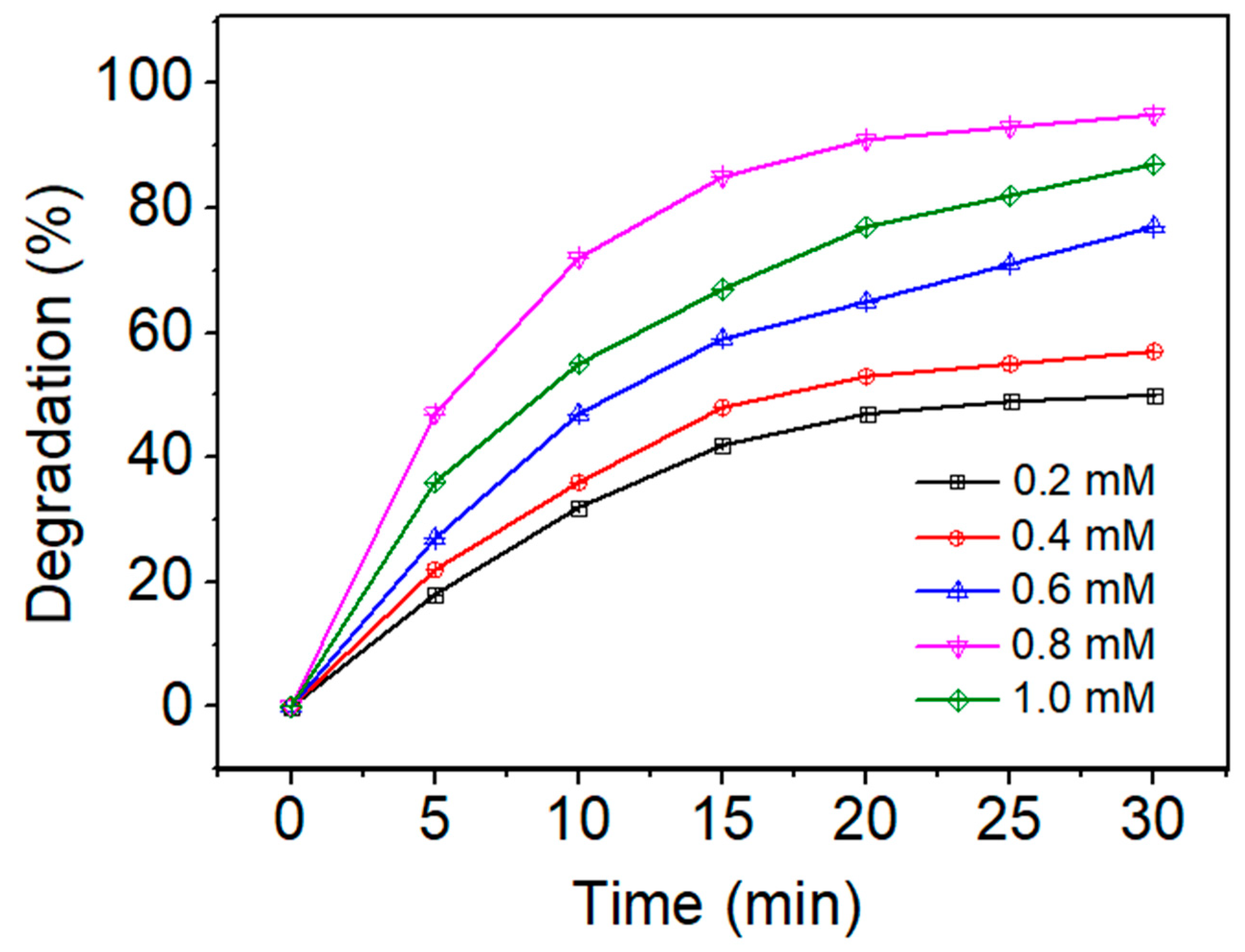
3.2.2. Effect of PS Concentration
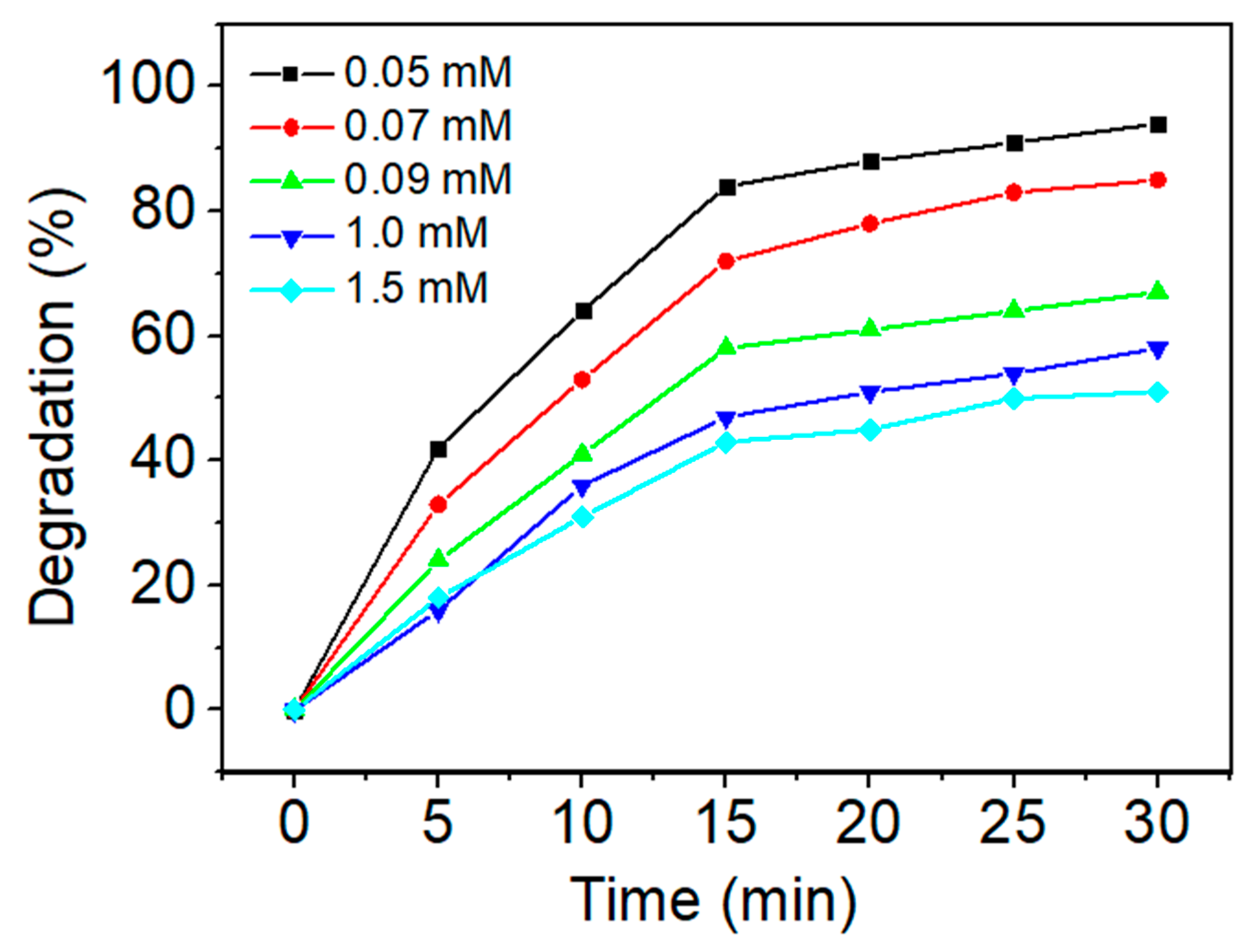
3.2.3. Effect of Fe2+ Concentration
3.2.4. Effect of MB 9 Concentrations
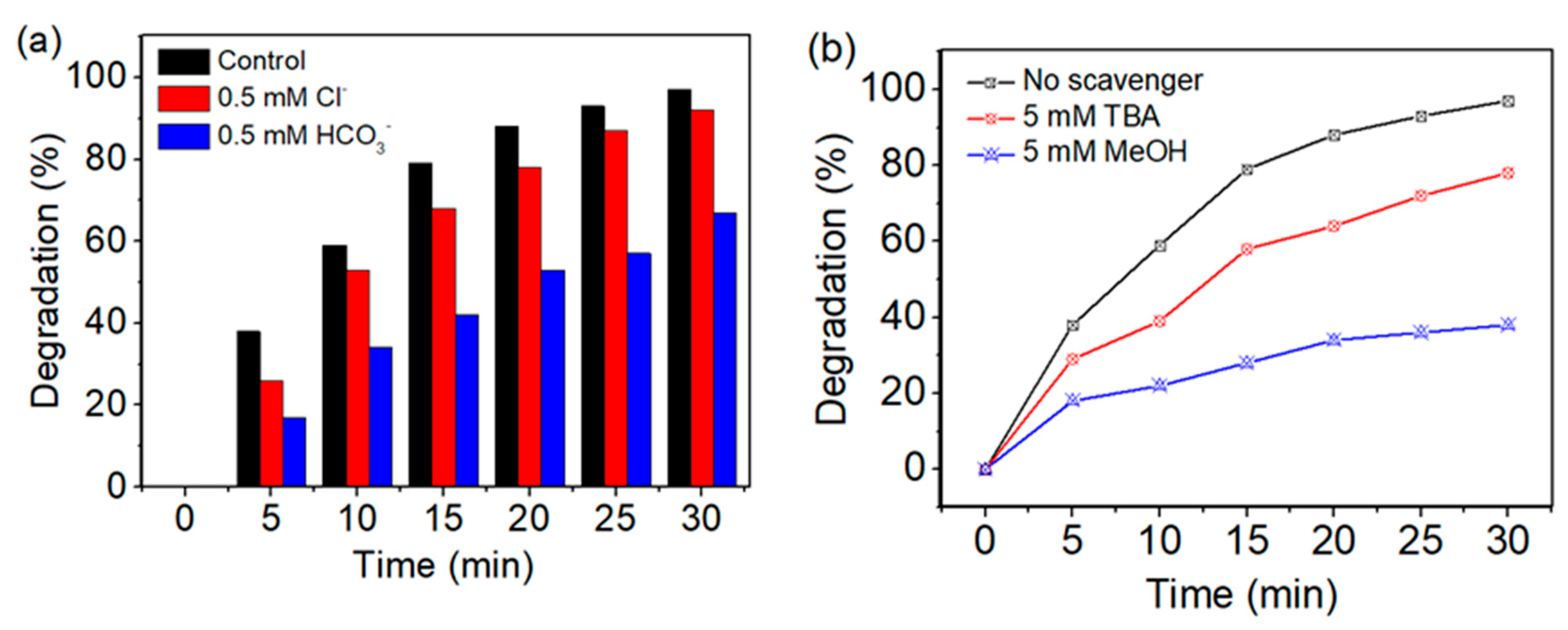
3.2.5. Effect of Inorganic Anions
3.2.6. Effect of Scavengers
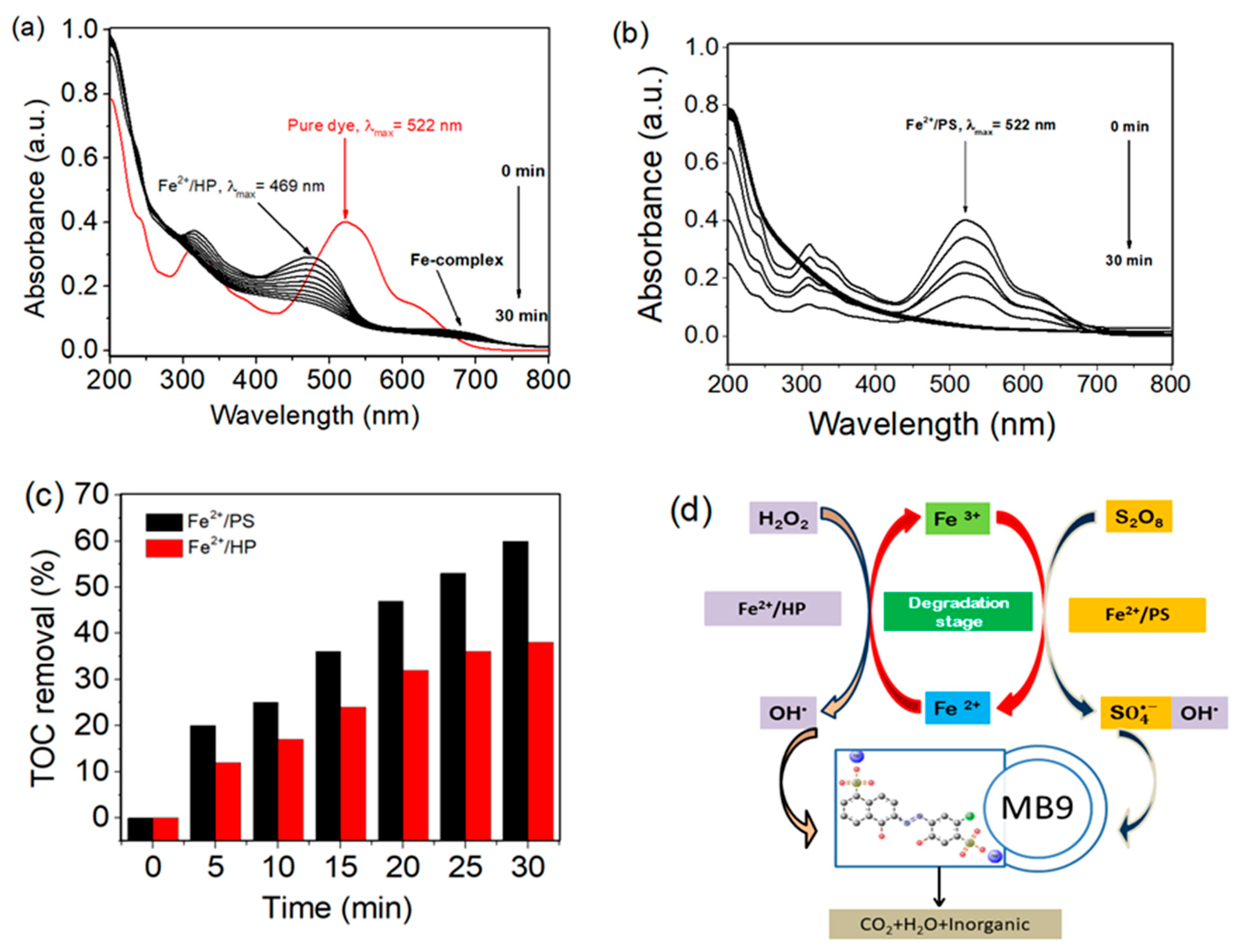
3.3. Degradation Pathway of MB 9
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Wu, L.; Xie, Q.; Lv, Y.; Wu, Z.; Liang, X.; Lu, M.; Nie, Y. Degradation of Methylene Blue via Dielectric Barrier Discharge Plasma Treatment. Water 2019, 11, 1818. [Google Scholar] [CrossRef]
- Angelakis, A.; Snyder, S. Wastewater treatment and reuse: Past, present, and future. Water 2015, 7, 4887–4895. [Google Scholar] [CrossRef]
- Telegin, F.Y.; Ran, J.H.; Morshed, M.; Pervez, M.N.; Sun, L.; Zhang, C.; Priazhinikova, V.G. Structure and Properties of Dyes in Coloration of Textiles: Application of Fragment Approach. Key Engineering Materials; Trans Tech Publications: Stafa-Zurich, Switzerland, 2016; pp. 261–266. [Google Scholar]
- El-Wakiel, N.A.; Rizk, H.F.; Ibrahim, S.A. Synthesis and characterization of metal complexes of azo dye based on 5-nitro-8-hydroxyquinoline and their applications in dyeing polyester fabrics. Appl. Organomet. Chem. 2017, 31, e3723. [Google Scholar] [CrossRef]
- Cai, Y.; David, S.; Pailthorpe, M. Dyeing of jute and jute/cotton blend fabrics with 2:1 pre-metallised dyes. Dye. Pigment. 2000, 45, 161–168. [Google Scholar] [CrossRef]
- Ding, Y.; Freeman, H.S. Mordant dye application on cotton: Optimisation and combination with natural dyes. Coloration Technol. 2017, 133, 369–375. [Google Scholar] [CrossRef]
- Edwards, L.C.; Freeman, H.S. Synthetic dyes based on environmental considerations. Part 3: Aquatic toxicity of iron-complexed azo dyes. Coloration Technol. 2005, 121, 265–270. [Google Scholar] [CrossRef]
- Hong, E.; Yeneneh, A.M.; Sen, T.K.; Ang, H.M.; Kayaalp, A. A comprehensive review on rheological studies of sludge from various sections of municipal wastewater treatment plants for enhancement of process performance. Adv. Colloid Interface Sci. 2018, 257, 19–30. [Google Scholar] [CrossRef]
- Pervez, M.N.; Stylios, G.K. Investigating the synthesis and characterization of a novel “green” H2O2-assisted, water-soluble chitosan/polyvinyl alcohol nanofiber for environmental end uses. Nanomaterials 2018, 8, 395. [Google Scholar] [CrossRef]
- Clavijo, C.; Osma, J.F. Functionalized leather: A novel and effective hazardous solid waste adsorbent for the removal of the diazo dye congo red from aqueous solution. Water 2019, 11, 1906. [Google Scholar] [CrossRef]
- Gomez-Herrero, E.; Tobajas, M.; Polo, A.; Rodriguez, J.J.; Mohedano, A.F. Removal of imidazolium-based ionic liquid by coupling Fenton and biological oxidation. J. Hazard. Mater. 2019, 365, 289–296. [Google Scholar] [CrossRef]
- Vilardi, G.; Sebastiani, D.; Miliziano, S.; Verdone, N.; Di Palma, L. Heterogeneous nZVI-induced Fenton oxidation process to enhance biodegradability of excavation by-products. Chem. Eng. J. 2018, 335, 309–320. [Google Scholar] [CrossRef]
- Munoz, M.; De Pedro, Z.M.; Casas, J.A.; Rodriguez, J.J. Preparation of magnetite-based catalysts and their application in heterogeneous Fenton oxidation–a review. Appl. Catal. B Environ. 2015, 176, 249–265. [Google Scholar] [CrossRef]
- Olmez-Hanci, T.; Arslan-Alaton, I. Comparison of sulfate and hydroxyl radical based advanced oxidation of phenol. Chem. Eng. J. 2013, 224, 10–16. [Google Scholar] [CrossRef]
- Shushina, I.; Ran, J.H.; Telegin, F.Y. Destruction of azo dyes in chemical, biochemical and photochemical processes. In Proceedings of the Book of papers, Regional Conference “Innovations of Youth Science”; SP University of Technology and Design: St Petersburg, Russia, 23–27 April 2012. [Google Scholar]
- Shushina, I.; Ran, J.H.; Telegin, F.Y. The role of complex formation in inhibition of catalytic oxidative destruction of mordant dyes. In Proceedings of the Book of Papers, Regional Conference “Progress 2012”; Ivanovo State Academy of Textiles: Ivanovo, Russia, 30 May 2012. [Google Scholar]
- Ran, J.H.; Shushina, I.; Priazhnikova, V.; Telegin, F. Inhibition of Mordant Dyes Destruction in Fenton Reaction. Adv. Mater. Res. 2013, 821, 493–496. [Google Scholar] [CrossRef]
- Arellano, M.; Pazos, M.; Sanromán, M.Á. Sulfate Radicals-Based Technology as a Promising Strategy for Wastewater. Water 2019, 11, 1695. [Google Scholar] [CrossRef]
- Guerra-Rodríguez, S.; Rodríguez, E.; Singh, D.N.; Rodríguez-Chueca, J. Assessment of sulfate radical-based advanced oxidation processes for water and wastewater treatment: A Review. Water 2018, 10, 1828. [Google Scholar] [CrossRef]
- Wacławek, S.; Lutze, H.V.; Grübel, K.; Padil, V.V.; Černík, M.; Dionysiou, D.D. Chemistry of persulfates in water and wastewater treatment: A review. Chem. Eng. J. 2017, 330, 44–62. [Google Scholar] [CrossRef]
- Wu, B.; Gu, G.; Deng, S.; Liu, D.; Xiong, X. Efficient natural pyrrhotite activating persulfate for the degradation of O-isopropyl-N-ethyl thionocarbamate: Iron recycle mechanism and degradation pathway. Chemosphere 2019, 224, 120–127. [Google Scholar] [CrossRef]
- Ahn, Y.-Y.; Bae, H.; Kim, H.-I.; Kim, S.-H.; Kim, J.-H.; Lee, S.-G.; Lee, J. Surface-loaded metal nanoparticles for peroxymonosulfate activation: Efficiency and mechanism reconnaissance. Appl. Catal. B Environ. 2019, 241, 561–569. [Google Scholar] [CrossRef]
- Liang, C.; Su, H.-W. Identification of sulfate and hydroxyl radicals in thermally activated persulfate. Ind. Eng. Chem. Res. 2009, 48, 5558–5562. [Google Scholar] [CrossRef]
- Liu, X.; Yuan, B.; Zou, J.; Wu, L.; Dai, L.; Ma, H.; Li, K.; Ma, J. Cu (II)-enhanced degradation of acid orange 7 by Fe (II)-activated persulfate with hydroxylamine over a wide pH range. Chemosphere 2020, 238, 124533. [Google Scholar] [CrossRef] [PubMed]
- Rastogi, A.; Al-Abed, S.R.; Dionysiou, D.D. Sulfate radical-based ferrous–peroxymonosulfate oxidative system for PCBs degradation in aqueous and sediment systems. Appl. Catal. B Environ. 2009, 85, 171–179. [Google Scholar] [CrossRef]
- Rao, Y.; Qu, L.; Yang, H.; Chu, W. Degradation of carbamazepine by Fe (II)-activated persulfate process. J. Hazard. Mater. 2014, 268, 23–32. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Wu, J.; Lu, X.; Xu, W.; Gong, Q.; Ding, J.; Dan, B.; Xie, P. Removal of acetaminophen in the Fe2+/persulfate system: Kinetic model and degradation pathways. Chem. Eng. J. 2019, 358, 1091–1100. [Google Scholar] [CrossRef]
- Shang, W.; Dong, Z.; Li, M.; Song, X.; Zhang, M.; Jiang, C.; Feiyun, S. Degradation of diatrizoate in water by Fe (II)-activated persulfate oxidation. Chem. Eng. J. 2019, 361, 1333–1344. [Google Scholar] [CrossRef]
- Zhu, J.-P.; Lin, Y.-L.; Zhang, T.-Y.; Cao, T.-C.; Xu, B.; Pan, Y.; Zhang, X.-T.; Gao, N.-Y. Modelling of iohexol degradation in a Fe (II)-activated persulfate system. Chem. Eng. J. 2019, 367, 86–93. [Google Scholar] [CrossRef]
- Kusic, H.; Peternel, I.; Ukic, S.; Koprivanac, N.; Bolanca, T.; Papic, S.; Bozic, A.L. Modeling of iron activated persulfate oxidation treating reactive azo dye in water matrix. Chem. Eng. J. 2011, 172, 109–121. [Google Scholar] [CrossRef]
- Rodriguez, S.; Vasquez, L.; Costa, D.; Romero, A.; Santos, A. Oxidation of Orange G by persulfate activated by Fe (II), Fe (III) and zero valent iron (ZVI). Chemosphere 2014, 101, 86–92. [Google Scholar] [CrossRef]
- Sellers, R.M. Spectrophotometric determination of hydrogen peroxide using potassium titanium (IV) oxalate. Analyst 1980, 105, 950–954. [Google Scholar] [CrossRef]
- Lutze, H.V.; Kerlin, N.; Schmidt, T.C. Sulfate radical-based water treatment in presence of chloride: Formation of chlorate, inter-conversion of sulfate radicals into hydroxyl radicals and influence of bicarbonate. Water Res. 2015, 72, 349–360. [Google Scholar] [CrossRef]
- Han, D.; Wan, J.; Ma, Y.; Wang, Y.; Li, Y.; Li, D.; Guan, Z. New insights into the role of organic chelating agents in Fe (II) activated persulfate processes. Chem. Eng. J. 2015, 269, 425–433. [Google Scholar] [CrossRef]
- Guan, R.; Yuan, X.; Wu, Z.; Wang, H.; Jiang, L.; Zhang, J.; Li, Y.; Zeng, G.; Mo, D. Accelerated tetracycline degradation by persulfate activated with heterogeneous magnetic NixFe3− xO4 catalysts. Chem. Eng. J. 2018, 350, 573–584. [Google Scholar] [CrossRef]
- Liang, C.; Bruell, C.J.; Marley, M.C.; Sperry, K.L. Persulfate oxidation for in situ remediation of TCE. I. Activated by ferrous ion with and without a persulfate–thiosulfate redox couple. Chemosphere 2004, 55, 1213–1223. [Google Scholar] [CrossRef] [PubMed]
- Stefánsson, A. Iron (III) hydrolysis and solubility at 25 C. Environ. Sci. Technol. 2007, 41, 6117–6123. [Google Scholar] [CrossRef] [PubMed]
- Li, R.; Jin, X.; Megharaj, M.; Naidu, R.; Chen, Z. Heterogeneous Fenton oxidation of 2, 4-dichlorophenol using iron-based nanoparticles and persulfate system. Chem. Eng. J. 2015, 264, 587–594. [Google Scholar] [CrossRef]
- Yan, J.; Lei, M.; Zhu, L.; Anjum, M.N.; Zou, J.; Tang, H. Degradation of sulfamonomethoxine with Fe3O4 magnetic nanoparticles as heterogeneous activator of persulfate. J. Hazard. Mater. 2011, 186, 1398–1404. [Google Scholar] [CrossRef]
- Dehghan, S.; Kakavandi, B.; Kalantary, R.R. Heterogeneous sonocatalytic degradation of amoxicillin using ZnO@ Fe3O4 magnetic nanocomposite: Influential factors, reusability and mechanisms. J. Mol. Liq. 2018, 264, 98–109. [Google Scholar] [CrossRef]
- Kakavandi, B.; Takdastan, A.; Jaafarzadeh, N.; Azizi, M.; Mirzaei, A.; Azari, A. Application of Fe3O4@ C catalyzing heterogeneous UV-Fenton system for tetracycline removal with a focus on optimization by a response surface method. J. Photochem. Photobiol. A Chem. 2016, 314, 178–188. [Google Scholar] [CrossRef]
- Long, A.; Lei, Y.; Zhang, H. Degradation of toluene by a selective ferrous ion activated persulfate oxidation process. Ind. Eng. Chem. Res. 2014, 53, 1033–1039. [Google Scholar] [CrossRef]
- Ayoub, G.; Ghauch, A. Assessment of bimetallic and trimetallic iron-based systems for persulfate activation: Application to sulfamethoxazole degradation. Chem. Eng. J. 2014, 256, 280–292. [Google Scholar] [CrossRef]
- Wu, J.; Zhang, H.; Qiu, J. Degradation of Acid Orange 7 in aqueous solution by a novel electro/Fe2+/peroxydisulfate process. J. Hazard. Mater. 2012, 215, 138–145. [Google Scholar] [CrossRef] [PubMed]
- Zha, S.; Cheng, Y.; Gao, Y.; Chen, Z.; Megharaj, M.; Naidu, R. Nanoscale zero-valent iron as a catalyst for heterogeneous Fenton oxidation of amoxicillin. Chem. Eng. J. 2014, 255, 141–148. [Google Scholar] [CrossRef]
- Wang, J.; Wang, S. Activation of persulfate (PS) and peroxymonosulfate (PMS) and application for the degradation of emerging contaminants. Chem. Eng. J. 2018, 334, 1502–1517. [Google Scholar] [CrossRef]
- Heidari, M.R.; Varma, R.S.; Ahmadian, M.; Pourkhosravani, M.; Asadzadeh, S.N.; Karimi, P.; Khatami, M. Photo-fenton like catalyst system: Activated carbon/CoFe2O4 nanocomposite for reactive dye removal from textile wastewater. Appl. Sci. 2019, 9, 963. [Google Scholar] [CrossRef]
- Wang, J.; Zhang, T.; Mei, Y.; Pan, B. Treatment of reverse-osmosis concentrate of printing and dyeing wastewater by electro-oxidation process with controlled oxidation-reduction potential (ORP). Chemosphere 2018, 201, 621–626. [Google Scholar] [CrossRef]
- Zazou, H.; Afanga, H.; Akhouairi, S.; Ouchtak, H.; Addi, A.A.; Akbour, R.A.; Assabbane, A.; Douch, J.; Elmchaouri, A.; Duplay, J. Treatment of textile industry wastewater by electrocoagulation coupled with electrochemical advanced oxidation process. J. Water Process Eng. 2019, 28, 214–221. [Google Scholar] [CrossRef]
- Anipsitakis, G.P.; Dionysiou, D.D.; Gonzalez, M.A. Cobalt-mediated activation of peroxymonosulfate and sulfate radical attack on phenolic compounds. Implications of chloride ions. Environ. Sci. Technol. 2006, 40, 1000–1007. [Google Scholar] [CrossRef]
- Sun, Y.; Zhao, J.; Zhang, B.-T.; Li, J.; Shi, Y.; Zhang, Y. Oxidative degradation of chloroxylenol in aqueous solution by thermally activated persulfate: Kinetics, mechanisms and toxicities. Chem. Eng. J. 2019, 368, 553–563. [Google Scholar] [CrossRef]
- Fang, G.-D.; Dionysiou, D.D.; Wang, Y.; Al-Abed, S.R.; Zhou, D.-M. Sulfate radical-based degradation of polychlorinated biphenyls: Effects of chloride ion and reaction kinetics. J. Hazard. Mater. 2012, 227, 394–401. [Google Scholar] [CrossRef]
- Wang, Y.R.; Chu, W. Degradation of a xanthene dye by Fe(II)-mediated activation of Oxone process. J. Hazard. Mater. 2011, 186, 1455–1461. [Google Scholar] [CrossRef]
- Rao, Y.; Shan, G.; Yang, X. Some recent advances in transition-metal-catalyzed ortho SP 2 CH functionalization using Ru, Rh, and Pd. Sci. China Chem. 2014, 57, 930–944. [Google Scholar] [CrossRef]
- Pan, X.; Yan, L.; Qu, R.; Wang, Z. Degradation of the UV-filter benzophenone-3 in aqueous solution using persulfate activated by heat, metal ions and light. Chemosphere 2018, 196, 95–104. [Google Scholar] [CrossRef] [PubMed]
- Weng, C.-H.; Tao, H. Highly efficient persulfate oxidation process activated with Fe0 aggregate for decolorization of reactive azo dye Remazol Golden Yellow. Arab. J. Chem. 2015. [Google Scholar] [CrossRef]
- Nie, M.; Yang, Y.; Zhang, Z.; Yan, C.; Wang, X.; Li, H.; Dong, W. Degradation of chloramphenicol by thermally activated persulfate in aqueous solution. Chem. Eng. J. 2014, 246, 373–382. [Google Scholar] [CrossRef]
- Su, S.; Cao, C.; Zhao, Y.; Dionysiou, D.D. Efficient transformation and elimination of roxarsone and its metabolites by a new α-FeOOH@ GCA activating persulfate system under UV irradiation with subsequent As (V) recovery. Appl. Catal. B Environ. 2019, 245, 207–219. [Google Scholar] [CrossRef]
- Han, D.; Wan, J.; Ma, Y.; Wang, Y.; Huang, M.; Chen, Y.; Li, D.; Guan, Z.; Li, Y. Enhanced decolorization of Orange G in a Fe (II)-EDDS activated persulfate process by accelerating the regeneration of ferrous iron with hydroxylamine. Chem. Eng. J. 2014, 256, 316–323. [Google Scholar] [CrossRef]
- Lin, X.-m.; Ma, Y.-w.; Wang, Y.; Wan, J.-q.; Guan, Z.-y. Lithium iron phosphate (LiFePO 4) as an effective activator for degradation of organic dyes in water in the presence of persulfate. RSC Adv. 2015, 5, 94694–94701. [Google Scholar] [CrossRef]
- Yadav, A.; Sharma, A.; Sharma, R.K. Mesoporous iron gallate nanocomplex for adsorption and degradation of organic dyes. Colloids Surf. A Physicochem. Eng. Asp. 2019, 579, 123694. [Google Scholar] [CrossRef]
- Singh, S.; Pakshirajan, K. Enzyme activities and decolourization of single and mixed azo dyes by the white-rot fungus Phanerochaete chrysosporium. Int. Biodeterior. Biodegrad. 2010, 64, 146–150. [Google Scholar] [CrossRef]
- Pakshirajan, K.; Jaiswal, S.; Das, R.K. Biodecolourization of Azo Dyes Using Phanerochaete Chrysosporium: Effect of Culture Conditions and Enzyme Activities. J. Sci. Ind. Res. 2011, 70, 987–991. [Google Scholar]
- Pakshirajan, K.; Singh, S. Decolorization of synthetic wastewater containing azo dyes in a batch-operated rotating biological contactor reactor with the immobilized fungus Phanerochaete chrysosporium. Ind. Eng. Chem. Res. 2010, 49, 7484–7487. [Google Scholar] [CrossRef]
- Singh, S.; Pakshirajan, K.; Daverey, A. Screening and optimization of media constituents for decolourization of Mordant Blue-9 dye by Phanerochaete chrysosporium. Clean Technol. Environ. Policy 2010, 12, 313–323. [Google Scholar] [CrossRef]
- Šimek, M.; Mikulášek, P.; Kalenda, P.; Weidlich, T. Possibilities for removal of chlorinated dye Mordant Blue 9 from model waste water. Chem. Pap. 2016, 70, 470–476. [Google Scholar] [CrossRef]
- Tomić, N.a.M.; Dohčević-Mitrović, Z.D.; Paunović, N.M.; Mijin, D.a.Z.; Radić, N.D.; Grbić, B.k.V.; Aškrabić, S.M.; Babić, B.M.; Bajuk-Bogdanović, D.V. Nanocrystalline CeO2−δ as effective adsorbent of azo dyes. Langmuir 2014, 30, 11582–11590. [Google Scholar] [CrossRef] [PubMed][Green Version]

| Samples | Methods | Conc. of MB 9 | Removal Efficiency (%) | Ref |
|---|---|---|---|---|
| Fungal peroxidase enzymes | batch shake flasks | 10 mg L−1 | 97 | [62,63] |
| Fungal peroxidase enzymes | rotating biological contactor (RBC) reactor | 10 mg L−1 | 98 | [64] |
| Fungal peroxidase enzymes | media constituents | 10 mg L−1 | 100 | [65] |
| Ionic liquid | adsorption | 100 µmol | 98.28 | [66] |
| Fe2(SO4)3 | coagulation | 100 µmol | 28 | [66] |
| Fenton | oxidation | 100 µmol | 98 | [66] |
| CeO2 | adsorption | 200 mg L−1 | 91 | [67] |
| Fenton | oxidation | 0.05 mM | 48 | This work |
| Fenton-activated persulfate | oxidation | 0.05 mM | 97 | This work |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pervez, M.N.; Telegin, F.Y.; Cai, Y.; Xia, D.; Zarra, T.; Naddeo, V. Efficient Degradation of Mordant Blue 9 Using the Fenton-Activated Persulfate System. Water 2019, 11, 2532. https://doi.org/10.3390/w11122532
Pervez MN, Telegin FY, Cai Y, Xia D, Zarra T, Naddeo V. Efficient Degradation of Mordant Blue 9 Using the Fenton-Activated Persulfate System. Water. 2019; 11(12):2532. https://doi.org/10.3390/w11122532
Chicago/Turabian StylePervez, Md. Nahid, Felix Y. Telegin, Yingjie Cai, Dongsheng Xia, Tiziano Zarra, and Vincenzo Naddeo. 2019. "Efficient Degradation of Mordant Blue 9 Using the Fenton-Activated Persulfate System" Water 11, no. 12: 2532. https://doi.org/10.3390/w11122532
APA StylePervez, M. N., Telegin, F. Y., Cai, Y., Xia, D., Zarra, T., & Naddeo, V. (2019). Efficient Degradation of Mordant Blue 9 Using the Fenton-Activated Persulfate System. Water, 11(12), 2532. https://doi.org/10.3390/w11122532








