A Novel and Facile Method to Characterize the Suitability of Metallic Iron for Water Treatment
Abstract
1. Introduction
2. Materials and Methods
2.1. Solutions
2.2. Fe0 Materials
2.3. Experimental Procedure
2.3.1. Elemental Composition of Used Fe0 SW
2.3.2. Iron Dissolution
2.3.3. Analytical Method
2.3.4. Expression of Results
3. Results and Discussion
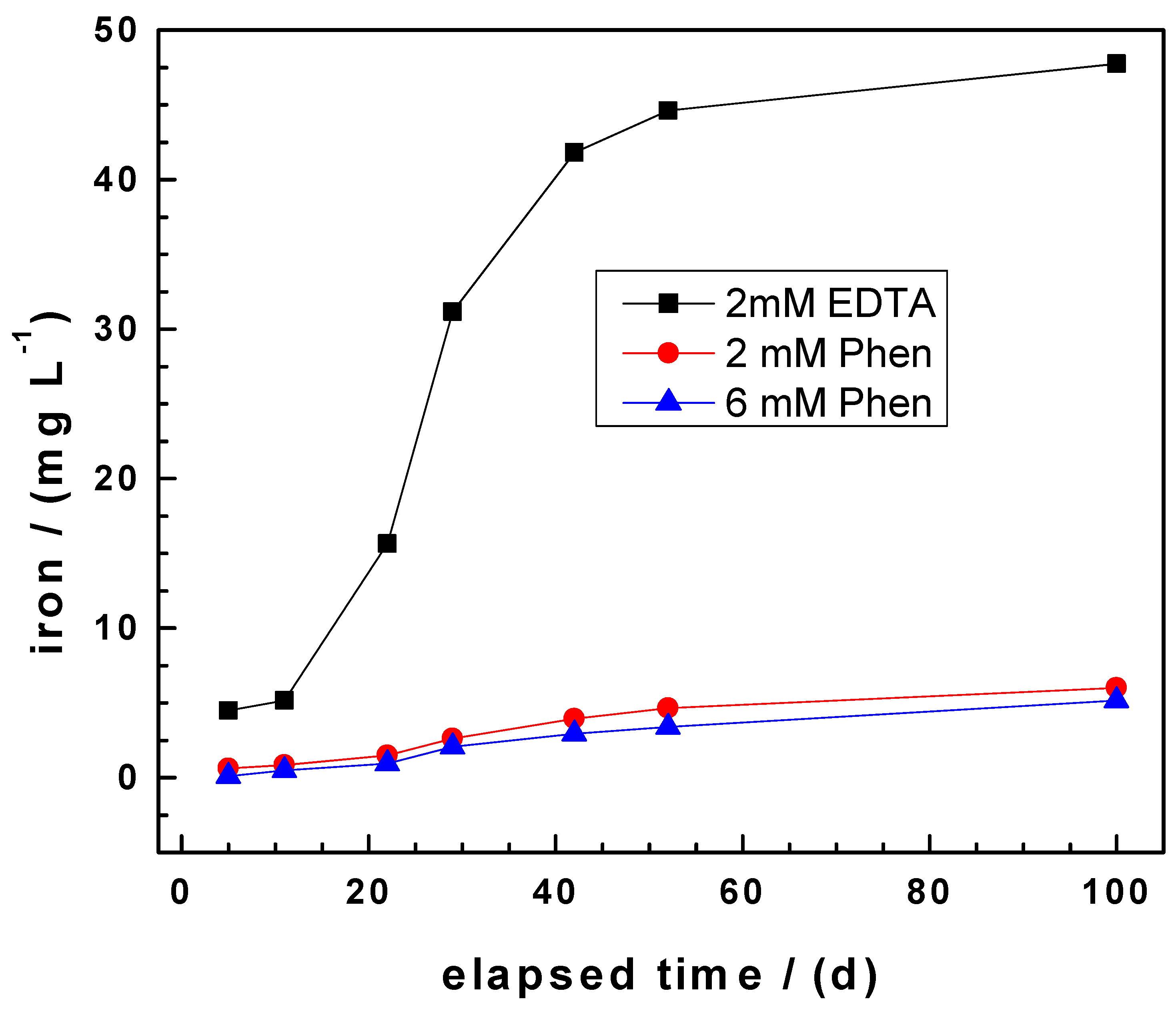
3.1. Appropriateness of the Experimental Approach
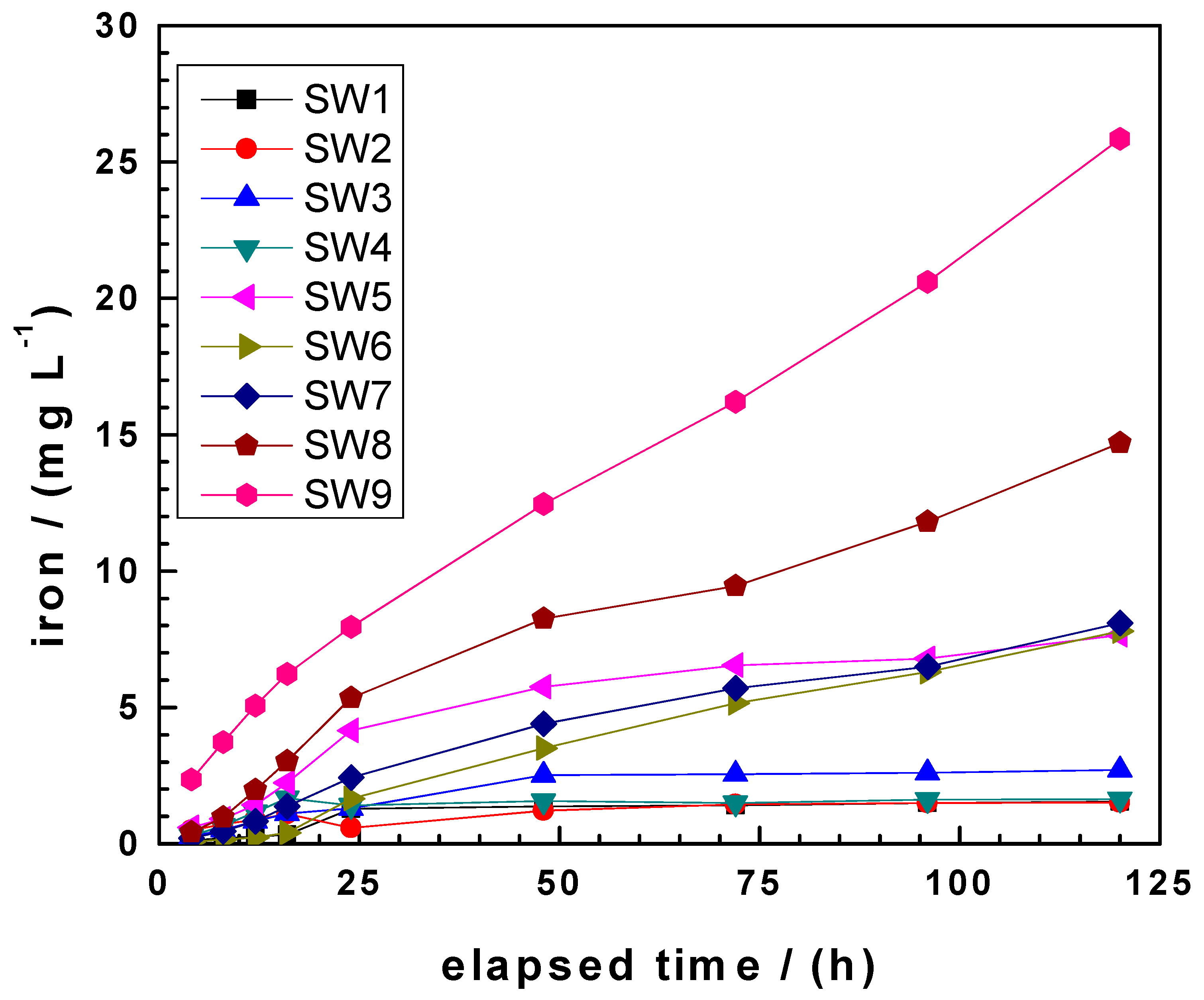
3.2. Evidencing the Non-Linear Kinetic of Iron Corrosion
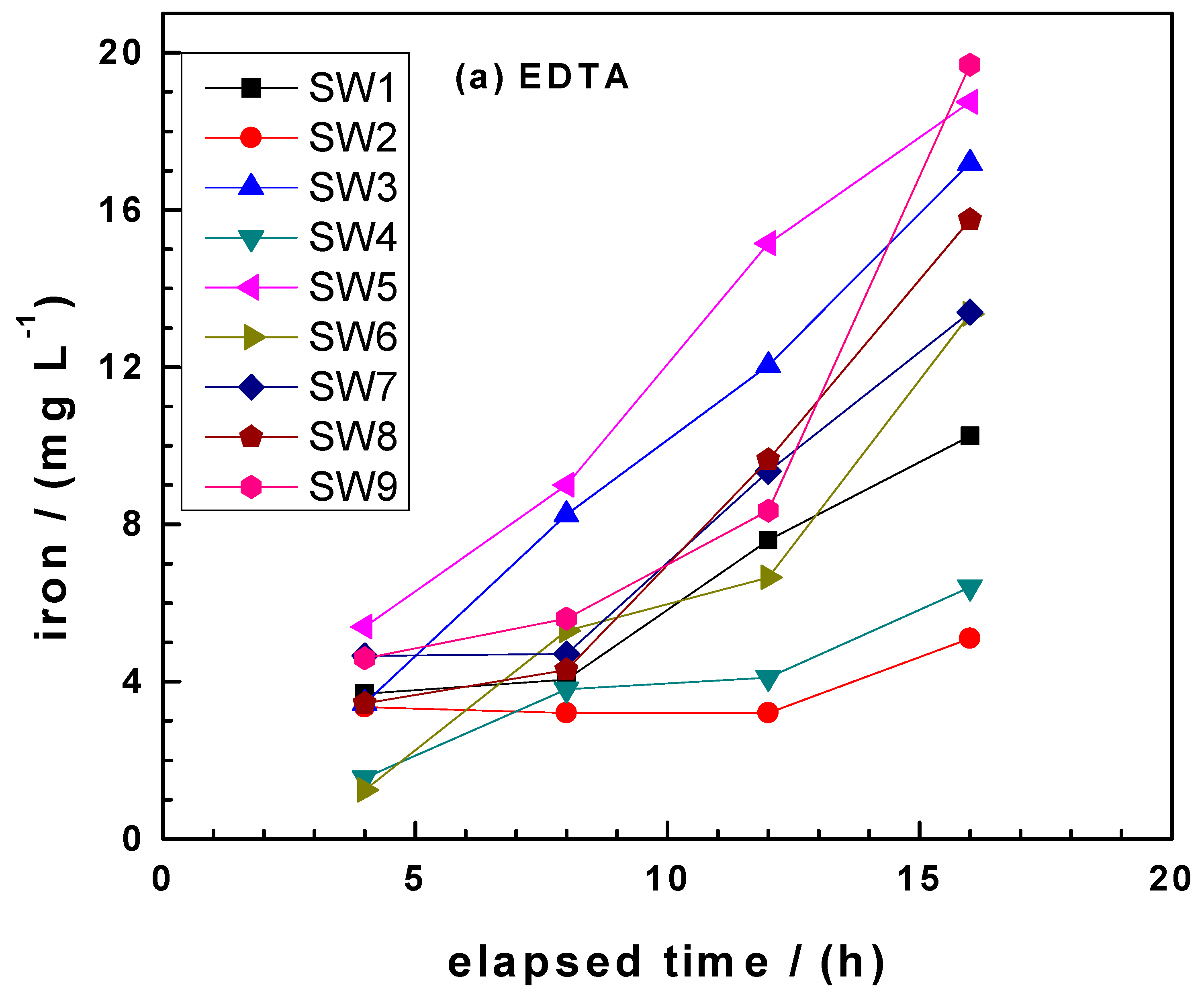
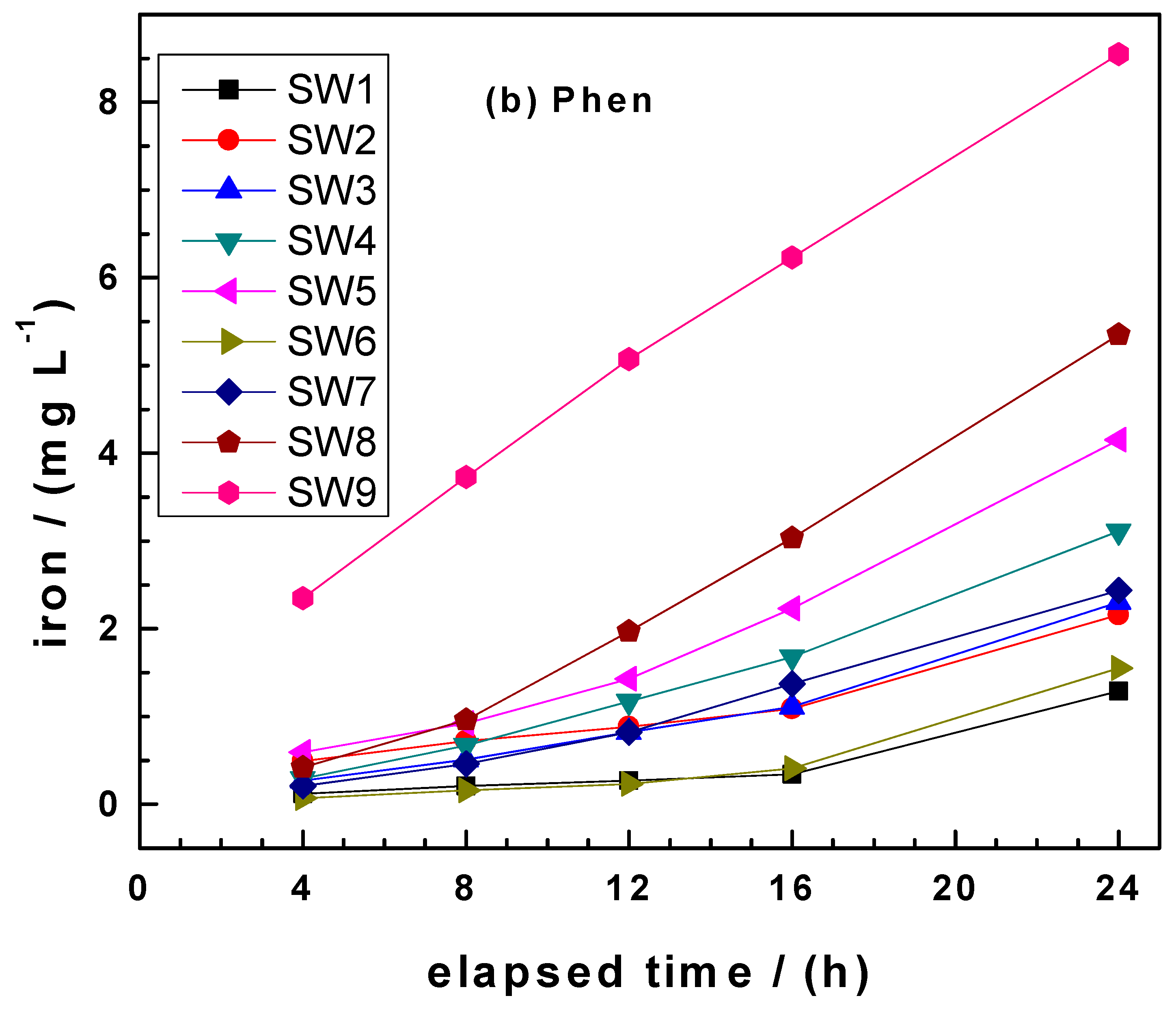
3.3. Comparing kEDTA and kPhen Values
- EDTA: SW2 < SW4 < SW1 < SW7 < SW6 < SW8 < SW3 < SW5 < SW9.
- Phen: SW1 < SW6 < SW2 < SW3 < SW7 < SW4 < SW5 < SW8 < SW9.
3.4. Discussion
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Shannon, M.A.; Bohn, P.W.; Elimelech, M.; Georgiadis, J.G.; Marinas, B.J.; Mayes, A.M. Science and technology for water purification in the coming decades. Nature 2008, 452, 301–310. [Google Scholar] [CrossRef] [PubMed]
- Hussam, A. Contending with a development disaster: Sono filters remove arsenic from well water in Bangladesh. Innovations 2009, 4, 89–102. [Google Scholar] [CrossRef]
- Noubactep, C.; Schöner, A.; Woafo, P. Metallic iron filters for universal access to safe drinking water. Clean Soil Air Water 2009, 37, 930–937. [Google Scholar] [CrossRef]
- Noubactep, C. The fundamental mechanism of aqueous contaminant removal by metallic iron. Water SA 2010, 36, 663–670. [Google Scholar] [CrossRef]
- Banerji, T.; Chaudhari, S. A cost-effective technology for arsenic removal: Case study of zerovalent iron-based iit bombay arsenic filter in west bengal. In Water and Sanitation in the New Millennium; Nath, K., Sharma, V., Eds.; Springer: New Delhi, India, 2017. [Google Scholar]
- Noubactep, C. Editorial for the Special Issue: Filters in Drinking Water Treatment. Water 2019, 11, 522. [Google Scholar] [CrossRef]
- Gonzalez-Perez, A.; Persson, K.M.; Lipnizki, F. Functional channel membranes for drinking water production. Water 2018, 10, 859. [Google Scholar] [CrossRef]
- Hering, J.G.; Maag, S.; Schnoor, J.L. A call for synthesis of water research to achieve the sustainable development goals by 2030. Environ. Sci. Technol. 2016, 50, 6122–6123. [Google Scholar] [CrossRef]
- Noubactep, C.; Licha, T.; Scott, T.B.; Fall, M.; Sauter, M. Exploring the influence of operational parameters on the reactivity of elemental iron materials. J. Hazard. Mater. 2009, 172, 943–951. [Google Scholar] [CrossRef]
- Hu, R.; Gwenzi, G.; Sipowo, R.; Noubactep, C. Water treatment using metallic iron: A tutorial review. Processes 2019, 7, 622. [Google Scholar] [CrossRef]
- Ngai, T.K.K.; Murcott, S.; Shrestha, R.R.; Dangol, B.; Maharjan, M. Development and dissemination of Kanchan™ Arsenic Filter in rural Nepal. Water Sci. Technol. Water Supply 2006, 6, 137–146. [Google Scholar] [CrossRef]
- Naseri, E.; Ndé-Tchoupé, A.I.; Mwakabona, H.T.; Nanseu-Njiki, C.P.; Noubactep, C.; Njau, K.N.; Wydra, K.D. Making Fe0-based filters a universal solution for safe drinking water provision. Sustainability 2017, 9, 1224. [Google Scholar] [CrossRef]
- Polonski, M.; Pawluk, K.; Rybka, I. Optimization model for the design of multi-layered permeable reactive barriers. IOP Conf. Ser. Mater. Sci. Eng. 2017, 245, 072017. [Google Scholar] [CrossRef]
- Domga, R.; Togue-Kamga, F.; Noubactep, C.; Tchatchueng, J.B. Discussing porosity loss of Fe0 packed water filters at ground level. Chem. Eng. J. 2015, 263, 127–134. [Google Scholar] [CrossRef]
- Makota, S.; Nde-Tchoupe, A.I.; Mwakabona, H.T.; Tepong-Tsindé, R.; Noubactep, C.; Nassi, A.; Njau, K.N. Metallic iron for water treatment: Leaving the valley of confusion. Appl. Water Sci. 2017, 7, 4177–4196. [Google Scholar] [CrossRef]
- Pawluk, K.; Polonski, M.; Wrzesiński, G.; Lendo-Siwicka, M. Two-objective optimization for optimal design of the multilayered permeable reactive barriers. IOP Conf. Ser. Mater. Sci. Eng. 2019, 471, 112044. [Google Scholar] [CrossRef]
- Higgins, M.R.; Olson, T.M. Life-cycle case study comparison of permeable reactive barrier versus pump-and-treat remediation. Environ. Sci. Technol. 2009, 43, 9432–9438. [Google Scholar] [CrossRef]
- Nanseu-Njiki, C.P.; Gwenzi, W.; Pengou, M.; Rahman, M.A.; Noubactep, C. Fe0/H2O filtration systems for decentralized safe drinking water: Where to from here? Water 2019, 11, 429. [Google Scholar] [CrossRef]
- Mwakabona, H.T.; Ndé-Tchoupé, A.I.; Njau, K.N.; Noubactep, C.; Wydra, K.D. Metallic iron for safe drinking water provision: Considering a lost knowledge. Water Res. 2017, 117, 127–142. [Google Scholar] [CrossRef]
- Btatkeu-K, B.D.; Miyajima, K.; Noubactep, C.; Caré, S. Testing the suitability of metallic iron for environmental remediation: Discoloration of methylene blue in column studies. Chem. Eng. J. 2013, 215–216, 959–968. [Google Scholar] [CrossRef]
- Hu, R.; Cui, X.; Xiao, M.; Qiu, P.; Lufingo, M.; Gwenzi, W.; Noubactep, C. Characterizing the suitability of granular Fe0 for the water treatment industry. Processes 2019, 7, 652. [Google Scholar] [CrossRef]
- Li, J.; Dou, X.; Qin, H.; Sun, Y.; Yin, D.; Guan, X. Characterization methods of zerovalent iron for water treatment and remediation. Water Res. 2019, 148, 70–85. [Google Scholar] [CrossRef] [PubMed]
- Williamson, S.J.; Clark, L.A. Pressure required to cause cover cracking of concrete due to reinforcement corrosion. Mag. Concr. Res. 2000, 52, 455–467. [Google Scholar] [CrossRef]
- Caré, S.; Nguyen, Q.T.; L’Hostis, V.; Berthaud, Y. Mechanical properties of the rust layer induced by impressed current method in reinforced mortar. Cement Concr. Res. 2008, 38, 1079–1091. [Google Scholar] [CrossRef]
- Zhao, Y.; Yu, J.; Jin, W. Damage analysis and cracking model of reinforced concrete structures with rebar corrosion. Corros. Sci. 2011, 53, 3388–3397. [Google Scholar] [CrossRef]
- Pilling, N.B.; Bedworth, R.E. The oxidation of metals at high temperatures. J. Inst. Met. 1923, 29, 529–591. [Google Scholar]
- Caré, S.; Crane, R.; Calabrò, P.S.; Ghauch, A.; Temgoua, E.; Noubactep, C. Modeling the permeability loss of metallic iron water filtration systems. Clean Soil Air Water 2013, 41, 275–282. [Google Scholar] [CrossRef]
- Moraci, N.; Lelo, D.; Bilardi, S.; Calabrò, P.S. Modelling long-term hydraulic conductivity behaviour of zero valent iron column tests for permeable reactive barrier design. Can. Geotech. J. 2016, 53, 946–961. [Google Scholar] [CrossRef]
- Ndé-Tchoupé, A.I.; Lufingo, M.; Hu, R.; Gwenzi, W.; Ntwampe, S.K.O.; Noubactep, C.; Njau, K.N. Avoiding the use of exhausted drinking water filters: A filter-clock based on rusting iron. Water 2018, 10, 591. [Google Scholar] [CrossRef]
- Noubactep, C. Metallic iron for environmental remediation: Prospects and limitations. In A Handbook of Environmental Toxicology: Human Disorders and Ecotoxicology; D’Mello, J.P.F., Ed.; CAB International: Wallingford, UK, 2019; Chapter 36; pp. 531–544. [Google Scholar]
- Miyajima, K. Optimizing the design of metallic iron filters for water treatment. Freib. Online Geosci. 2012, 32, 1–60. [Google Scholar]
- Miyajima, K.; Noubactep, C. Characterizing the impact of sand addition on the efficiency of granular iron for water treatment. Chem. Eng. J. 2013, 262, 891–896. [Google Scholar] [CrossRef]
- Li, J.; Zhang, X.; Liu, M.; Pan, B.; Zhang, W.; Shi, W.; Guan, X. Enhanced reactivity and electron selectivity of sulfidated zerovalent iron toward chromate under aerobic conditions. Environ. Sci. Technol. 2018, 52, 2988–2997. [Google Scholar] [CrossRef] [PubMed]
- Gheju, M.; Balcu, I. Removal of chromium from Cr(VI) polluted wastewaters by reduction with scrap iron and subsequent precipitation of resulted cations. J. Hazard. Mater. 2011, 196, 131–138. [Google Scholar] [CrossRef] [PubMed]
- Gheju, M. Progress in understanding the mechanism of CrVI Removal in Fe0-based filtration systems. Water 2018, 10, 651. [Google Scholar] [CrossRef]
- Gheju, M.; Balcu, I. Sustaining the efficiency of the Fe0/H2O system for Cr(VI) removal by MnO2 amendment. Chemosphere 2019, 214, 389–398. [Google Scholar] [CrossRef] [PubMed]
- Sato, N. An overview on the passivity of metals. Corros. Sci. 1990, 31, 1–19. [Google Scholar] [CrossRef]
- Sato, N. Surface oxides affecting metallic corrosion. Corros. Rev. 2001, 19, 253–272. [Google Scholar] [CrossRef]
- Phukan, M. Characterizing the Fe0/sand system by the extent of dye discoloration. Freib. Online Geosci. 2015, 40, 1–70. [Google Scholar]
- Phukan, M.; Noubactep, C.; Licha, T. Characterizing the ion-selective nature of Fe0-based filters using azo dyes. Chem. Eng. J. 2015, 259, 481–491. [Google Scholar] [CrossRef]
- Phukan, M.; Noubactep, C.; Licha, T. Characterizing the ion-selective nature of Fe0-based filters using three azo dyes in batch systems. J. Environ. Chem. Eng. 2016, 4, 65–72. [Google Scholar] [CrossRef]
- Reardon, J.E. Anaerobic corrosion of granular iron: Measurement and interpretation of hydrogen evolution rates. Environ. Sci. Technol. 1995, 29, 2936–2945. [Google Scholar] [CrossRef]
- Landis, R.L.; Gillham, R.W.; Reardon, E.J.; Fagan, R.; Focht, R.M.; Vogan, J.L. An examination of zero-valent iron sources used in permeable reactive barriers. In Proceedings of the 3rd International Containment Technology Conference, Orlando, FL, USA, 10–13 June 2001; Florida State University: Tallahassee, FL, USA, 2001. 5p. [Google Scholar]
- Miehr, R.; Tratnyek, P.G.; Bandstra, Z.J.; Scherer, M.M.; Alowitz, J.M.; Bylaska, J.E. Diversity of contaminant reduction reactions by zerovalent iron: Role of the reductate. Environ. Sci. Technol. 2004, 38, 139–147. [Google Scholar] [CrossRef] [PubMed]
- Noubactep, C.; Meinrath, G.; Dietrich, P.; Sauter, M.; Merkel, B.J. Testing the suitability of zerovalent iron materials for reactive walls. Environ. Chem. 2005, 2, 71–76. [Google Scholar] [CrossRef]
- Reardon, E.J. Zerovalent irons: Styles of corrosion and inorganic control on hydrogen pressure buildup. Environ. Sci. Tchnol. 2005, 39, 7311–7317. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.; Yang, H.; Kim, J. Standardization of the reducing power of zero-valent iron using iodine. J. Environ. Sci. Health. A 2014, 49, 514–523. [Google Scholar] [CrossRef] [PubMed]
- Birke, V.; Schuett, C.; Burmeier, H.; Friedrich, H.-J. Impact of trace elements and impurities in technical zero-valent iron brands on reductive dechlorination of chlorinated ethenes in groundwater. In Permeable Reactive Barrier Sustainable Groundwater Remediation; Naidu, R., Birke, V., Eds.; CRC Press: Boca Raton, FL, USA, 2015; pp. 87–98. ISBN 978-1-4822-2447-4. [Google Scholar]
- Li, S.; Ding, Y.; Wang, W.; Lei, H. A facile method for determining the Fe0 content and reactivity of zero valent iron. Anal. Methods 2016, 8, 1239–1248. [Google Scholar] [CrossRef]
- Pierce, E.M.; Wellman, D.M.; Lodge, A.M.; Rodriguez, E.A. Experimental determination of the dissolution kinetics of zero-valent iron in the presence of organic complexants. Environ. Chem. 2007, 4, 260–270. [Google Scholar] [CrossRef]
- Hildebrant, B. Characterizing the reactivity of commercial steel wool for water treatment. Freib. Online Geosci. 2018, 53, 1–80. [Google Scholar]
- Hu, R.; Ndé-Tchoupé, A.I.; Lufingo, M.; Xiao, M.; Nassi, A.; Noubactep, C.; Njau, K.N. The impact of selected pre-treatment procedures on iron dissolution from metallic iron specimens used in water treatment. Sustainability 2019, 11, 671. [Google Scholar] [CrossRef]
- Ibanez, J.G.; Gonzalez, I.; Cardenas, M.A.J. The effect of complex formation upon the redox potentials of metallic ions: Cyclic voltammetry experiments. J. Chem. Educ. 1988, 65, 173–175. [Google Scholar] [CrossRef]
- Rizvi, M.A.; Syed, R.M.; Khan, B. Complexation effect on redox potential of iron(III)−iron(II) couple: A simple potentiometric experiment. J. Chem. Educ. 2011, 88, 220–222. [Google Scholar] [CrossRef]
- Rizvi, M.A. Complexation modulated redox behavior of transition metal systems. Russ. J. Gen. Chem. 2015, 85, 959–973. [Google Scholar] [CrossRef]
- Bashir, S.; Rizwan, M.S.; Salam, A.; Fu, Q.; Zhu, J.; Shaaban, M.; Hu, H. Cadmium immobilization potential of rice straw-derived biochar, zeolite and rock phosphate: Extraction techniques and adsorption mechanism. Bull. Environ. Contam. Toxicol. 2018, 100, 727–732. [Google Scholar] [CrossRef] [PubMed]
- Sakai, K. Determination of Trace Elements in Steel Using the Agilent 7900 ICP-MS. Agilent Publications 5991-6116EN. 2015. Available online: https://www.agilent.com/cs/library/applications/5991-6116EN-appnote_7900_ICP-MS_steel_analysis.pdf (accessed on 11 October 2018).
- Zhou, H.; He, Y.; Lan, Y.; Mao, J.; Chen, S. Influence of complex reagents on removal of chromium(VI) by zero-valent iron. Chemosphere 2008, 72, 870–874. [Google Scholar] [CrossRef] [PubMed]
- Elmagirbi, A.; Sulistyarti, H.; Atikah, A. Study of ascorbic acid as iron(III) reducing agent for spectrophotometric iron speciation. J. Pure App. Chem. Res. 2012, 1, 11–17. [Google Scholar] [CrossRef]
- Lufingo, M. Investigation of Metallic Iron for Water Defluoridation. Master’s Thesis, Nelson Mandela African Institution of Science and Technology, Arusha, Tanzania, 2019. [Google Scholar]
- Noubactep, C.; Caré, S. Dimensioning metallic iron beds for efficient contaminant removal. Chem. Eng. J. 2010, 163, 454–460. [Google Scholar] [CrossRef]
- Noubactep, C. Research on metallic iron for environmental remediation: Stopping growing sloppy science. Chemosphere 2016, 153, 528–530. [Google Scholar] [CrossRef] [PubMed]
- Tarvainen, V.; Ranta-Maunus, A.; Hanhijärvi, A.; Forsén, H. The effect of drying and storage conditions on case hardening of scots pine and norway spruce timber. Maderas Ciencia y tecnología 2006, 8, 3–14. [Google Scholar] [CrossRef]
- Fernando, W.J.N.; Ahmad, A.L.; Shukor, S.R.; Lok, Y.H. A model for constant temperature drying rates of case hardened slices of papaya and garlic. J. Food Eng. 2008, 88, 229–238. [Google Scholar] [CrossRef]
- Noubactep, C.; Caré, S.; Crane, R. Nanoscale metallic iron for environmental remediation: Prospects and limitations. Water Air Soil Pollut. 2012, 223, 1363–1382. [Google Scholar] [CrossRef]
- Huang, C.P.; Wang, H.W.; Chiu, P.C. Nitrate reduction by metallic iron. Water Res. 1998, 32, 2257–2264. [Google Scholar] [CrossRef]
- Hao, Z.-W.; Xu, X.-H.; Wand, D.-H. Reductive denitrification of nitrate by scrap iron filings. J. Zhejiang Univ. Sci. B 2005, 6, 182–186. [Google Scholar] [CrossRef] [PubMed]
- Noubactep, C. Untersuchungen Zur Passiven In. Situ-Immobilisierung von U (VI) aus Wasser. Ph.D. Thesis, Institut für Geologie Freiberg, Freiberg, Germany, 2003. [Google Scholar]
- Btatkeu-K, B.D.; Tchatchueng, J.B.; Noubactep, C.; Caré, S. Designing metallic iron based water filters: Light from methylene blue discoloration. J. Environ. Manag. 2016, 166, 567–573. [Google Scholar] [CrossRef] [PubMed]
- Matheson, L.J.; Tratnyek, P.G. Reductive dehalogenation of chlorinated methanes by iron metal. Environ. Sci. Technol. 1994, 28, 2045–2053. [Google Scholar] [CrossRef] [PubMed]
- Liang, L.; Yang, W.; Guan, X.; Li, J.; Xu, Z.; Wu, J.; Huang, Y.; Zhang, X. Kinetics and mechanisms of pH-dependent selenite removal by zero valent iron. Water Res. 2013, 47, 5846–5855. [Google Scholar] [CrossRef] [PubMed]
- Guan, X.; Sun, Y.; Qin, H.; Li, J.; Lo, I.M.C.; He, D.; Dong, H. The limitations of applying zero-valent iron technology in contaminants sequestration and the corresponding countermeasures: The development in zero-valent iron technology in the last two decades (1994–2014). Water Res. 2015, 75, 224–248. [Google Scholar] [CrossRef] [PubMed]
- Noubactep, C. Metallic iron for environmental remediation: A review of reviews. Water Res. 2015, 85, 114–123. [Google Scholar] [CrossRef] [PubMed]
- Ebelle, T.C.; Makota, S.; Tepong-Tsindé, R.; Nassi, A.; Noubactep, C. Metallic iron and the dialogue of the deaf. Fresenius Environ. Bull. 2019, 28, 8331–8340. [Google Scholar]
- Hu, R.; Noubactep, C. Redirecting research on Fe0 for environmental remediation: The search for synergy. Int. J. Environ. Res. Public Health 2019, 16, 4465. [Google Scholar] [CrossRef]
- Noubactep, C. The operating mode of Fe0/H2O systems: Hidden truth or repeated nonsense? Fresenius Environ. Bull. 2019, 28, 8328–8330. [Google Scholar]
| Material Code | Diameter (mm) | Grade Number | Name | Elemental Composition | |||||
|---|---|---|---|---|---|---|---|---|---|
| Fe | Co | Cu | Pb | Ni | Cr | ||||
| SW1 | 0.025 | 0000 | Super Fine | 99.15 | 0.01 | 0.03 | 0.40 | 0.09 | 0.32 |
| SW2 | 0.035 | 000 | Extra Fine | 99.21 | 0.05 | 0.12 | n.d. | 0.16 | 0.47 |
| SW3 | 0.04 | 00 | Very Fine | 99.25 | 0.05 | 0.16 | n.d. | 0.11 | 0.43 |
| SW4 | 0.05 | 0 | Fine | 99.08 | 0.05 | 0.27 | n.d. | 0.11 | 0.49 |
| SW5 | 0.06 | 1 | Medium | 98.37 | 0.05 | 1.00 | n.d. | 0.11 | 0.45 |
| SW6 | 0.075 | 2 | Medium Coarse | 99.14 | 0.04 | 0.27 | n.d. | 0.10 | 0.45 |
| SW7 | 0.09 | 3 | Coarse | 98.69 | 0.05 | 0.40 | 0.40 | 0.14 | 0.33 |
| SW8 | 0.1 | 4 | Extra Coarse | 99.27 | 0.04 | 0.28 | n.d. | 0.10 | 0.30 |
| SW9 | 0.04 | 00 | Very Fine | 99.62 | 0.01 | 0.02 | n.d. | 0.03 | 0.32 |
| Sample | EDTA | O-Phen | ||||||
|---|---|---|---|---|---|---|---|---|
| kEDTA | b | N | R2 | kPhen | b | N | R2 | |
| (μg h−1) | (mg) | (-) | (-) | (μg h−1) | (mg) | (-) | (-) | |
| SW1 | 2.320 | 0.600 | 5 | 0.9256 | 0.072 | 0.055 | 5 | 0.9931 |
| SW2 | 0.525 | 2.400 | 5 | 0.5338 | 0.1960 | 0.3050 | 5 | 0.9957 |
| SW3 | 4.505 | (-)1.025 | 5 | 0.997 | 0.2830 | (-) 0.03 | 5 | 0.9974 |
| SW4 | 1.385 | 0.250 | 5 | 0.7809 | 0.4670 | (-) 0.215 | 5 | 0.9956 |
| SW5 | 4.620 | 0.525 | 5 | 0.988 | 0.5430 | (-) 0.065 | 5 | 0.9635 |
| SW6 | 3.765 | (-)2.775 | 5 | 0.9342 | 0.1090 | (-) 0.055 | 5 | 0.9539 |
| SW7 | 3.090 | 0.300 | 5 | 0.8992 | 0.3840 | (-) 0.245 | 5 | 0.9700 |
| SW8 | 4.225 | (-)2.275 | 5 | 0.9216 | 0.8840 | (-) 0.615 | 5 | 0.9808 |
| SW9 | 4.805 | (-)2.45 | 5 | 0.7985 | 1.2980 | 1.1000 | 5 | 0.9984 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lufingo, M.; Ndé-Tchoupé, A.I.; Hu, R.; Njau, K.N.; Noubactep, C. A Novel and Facile Method to Characterize the Suitability of Metallic Iron for Water Treatment. Water 2019, 11, 2465. https://doi.org/10.3390/w11122465
Lufingo M, Ndé-Tchoupé AI, Hu R, Njau KN, Noubactep C. A Novel and Facile Method to Characterize the Suitability of Metallic Iron for Water Treatment. Water. 2019; 11(12):2465. https://doi.org/10.3390/w11122465
Chicago/Turabian StyleLufingo, Mesia, Arnaud Igor Ndé-Tchoupé, Rui Hu, Karoli N. Njau, and Chicgoua Noubactep. 2019. "A Novel and Facile Method to Characterize the Suitability of Metallic Iron for Water Treatment" Water 11, no. 12: 2465. https://doi.org/10.3390/w11122465
APA StyleLufingo, M., Ndé-Tchoupé, A. I., Hu, R., Njau, K. N., & Noubactep, C. (2019). A Novel and Facile Method to Characterize the Suitability of Metallic Iron for Water Treatment. Water, 11(12), 2465. https://doi.org/10.3390/w11122465







