Response of Submerged Aquatic Vegetation to Water Depth in a Large Shallow Lake after an Extreme Rainfall Event
Abstract
1. Introduction
2. Methods
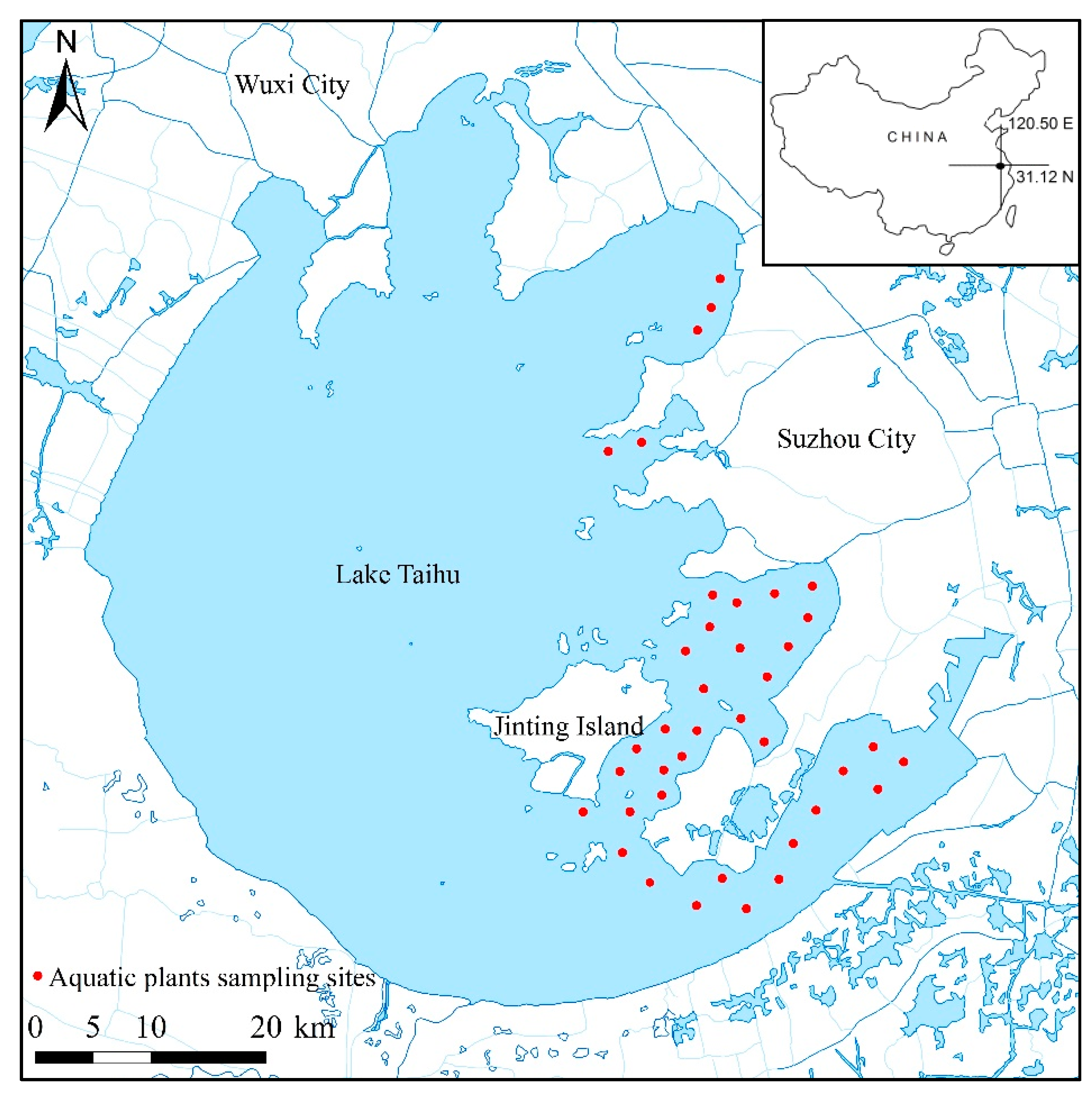
2.1. Aquatic Vegetation Biomass and Water Depth Survey
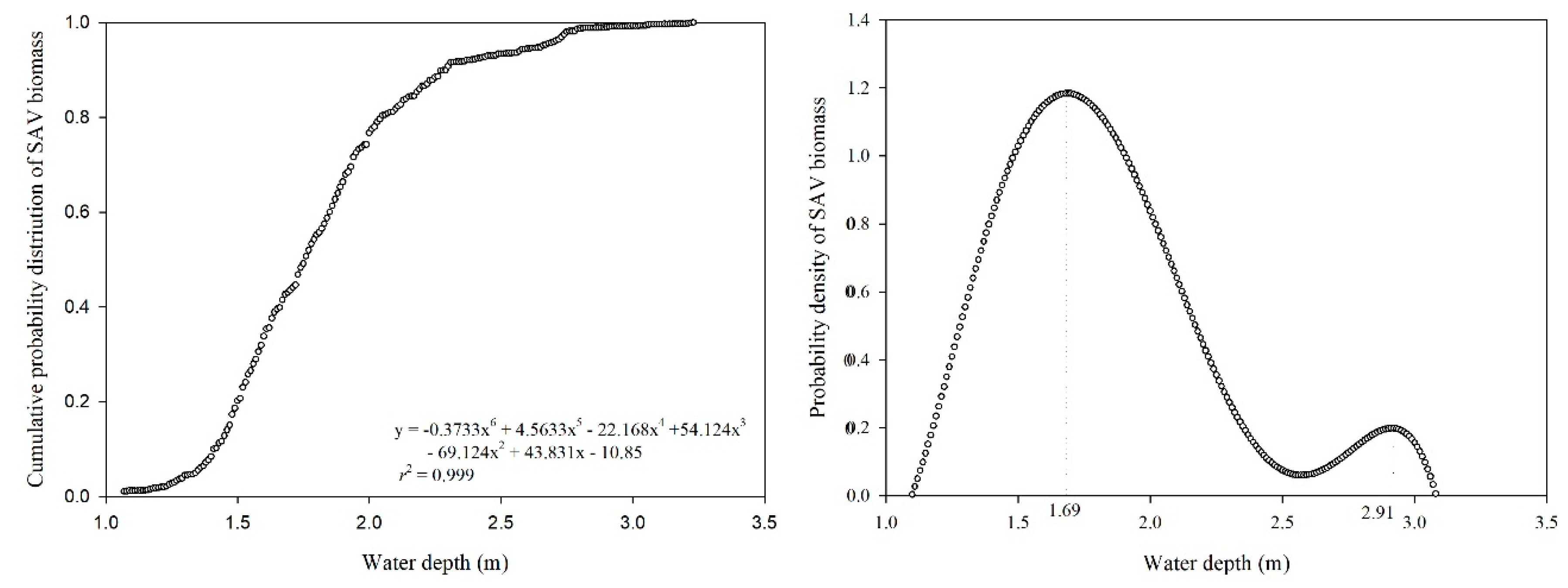
2.2. Calculation of Proper Water Depth for SAV
2.3. Frequency of Occurrence
2.4. Statistical Analysis
3. Results
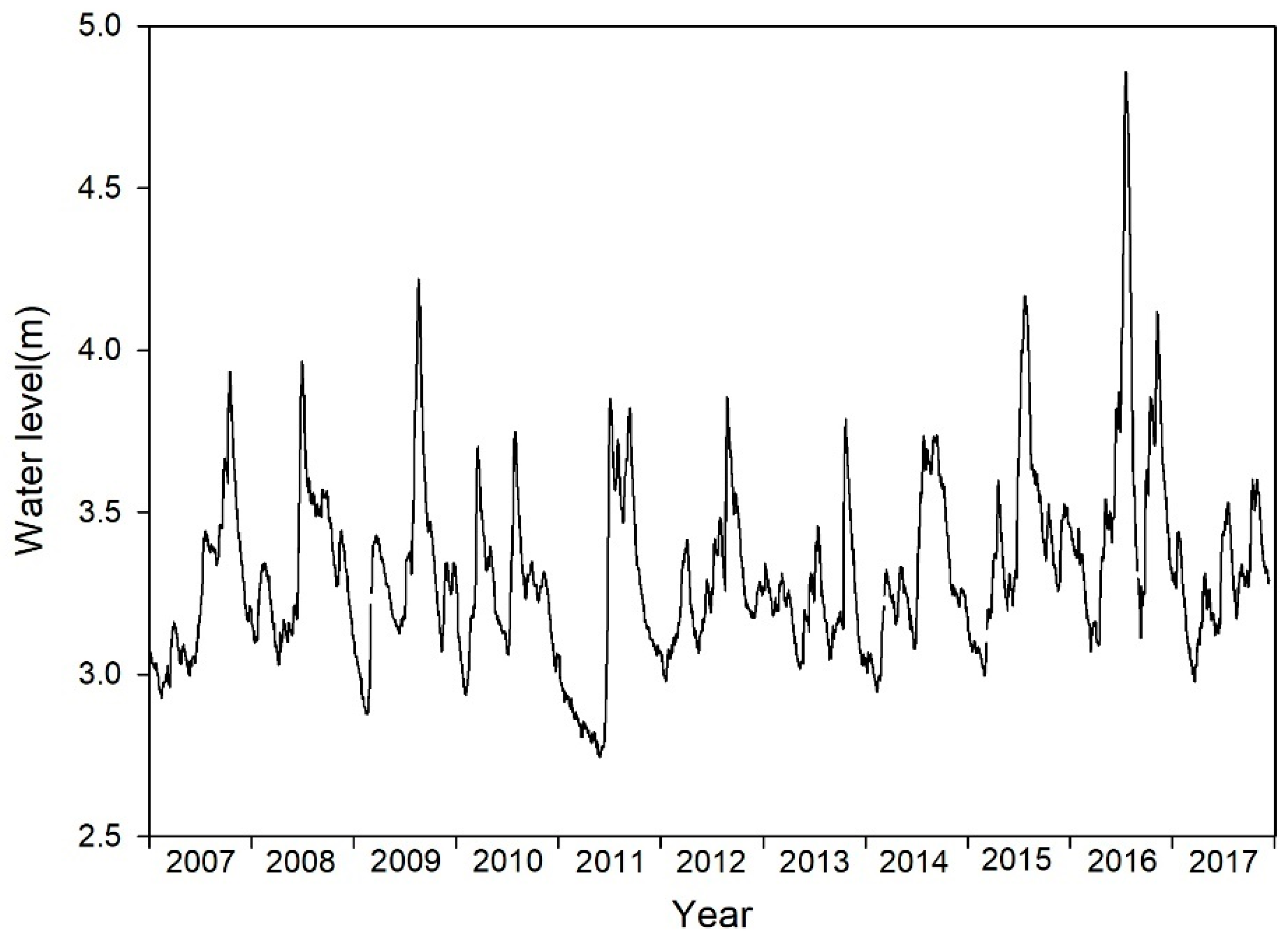
3.1. Variation in Water Levels
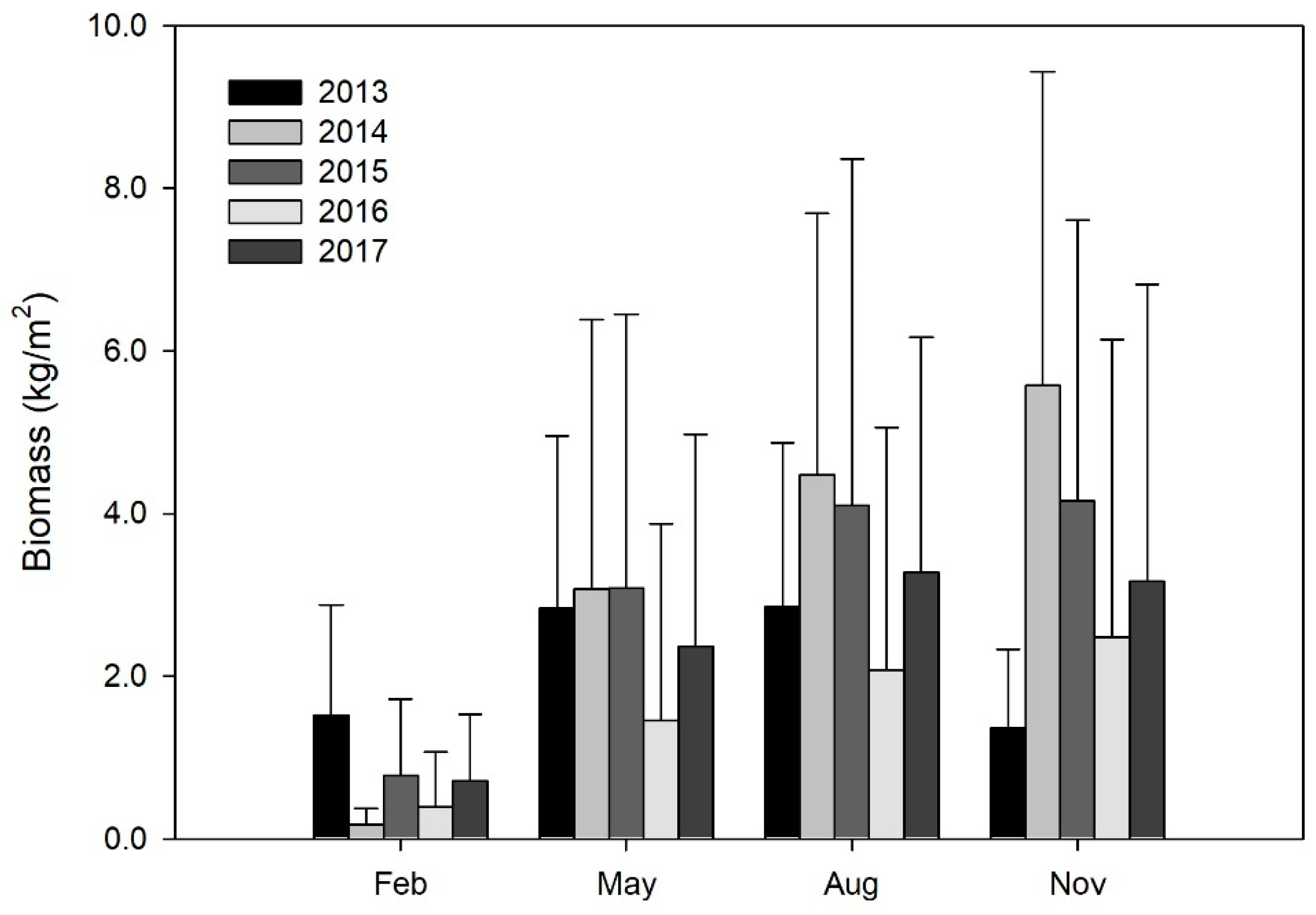
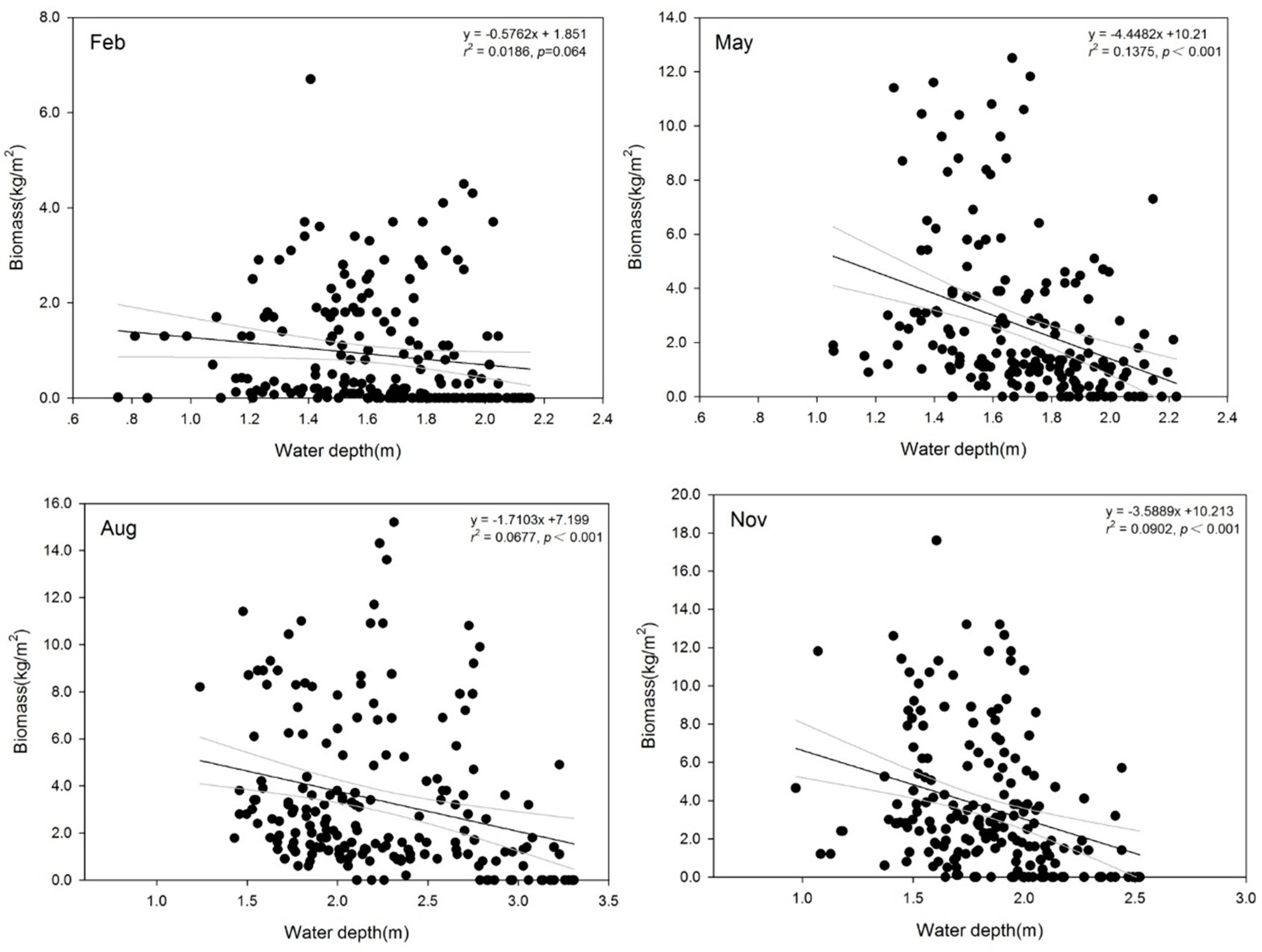
3.2. Variation in SAV Biomass
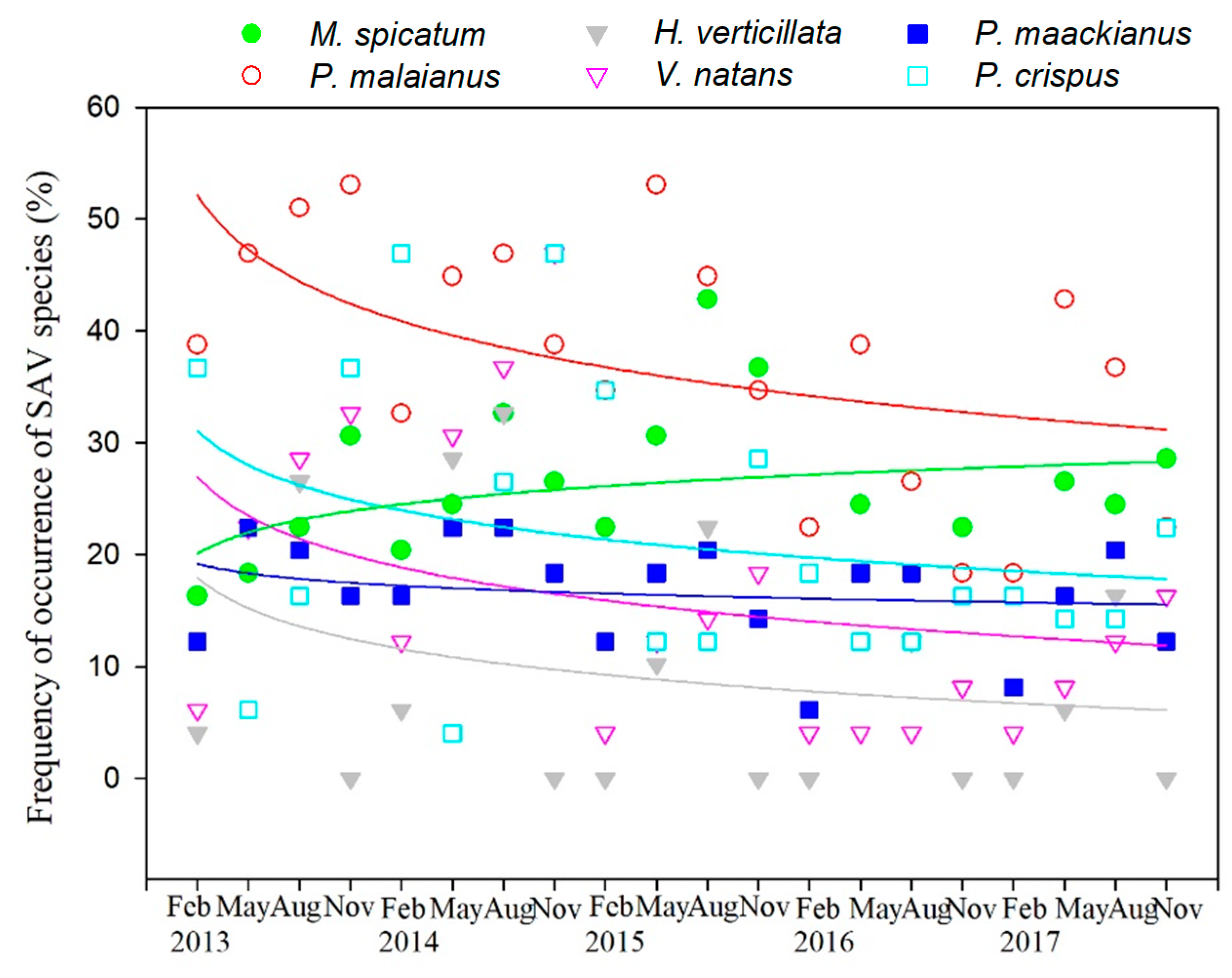
3.3. Variation in SAV Occurrence Frequency
4. Discussion
4.1. Effect of Water Depth on SAV Biomass
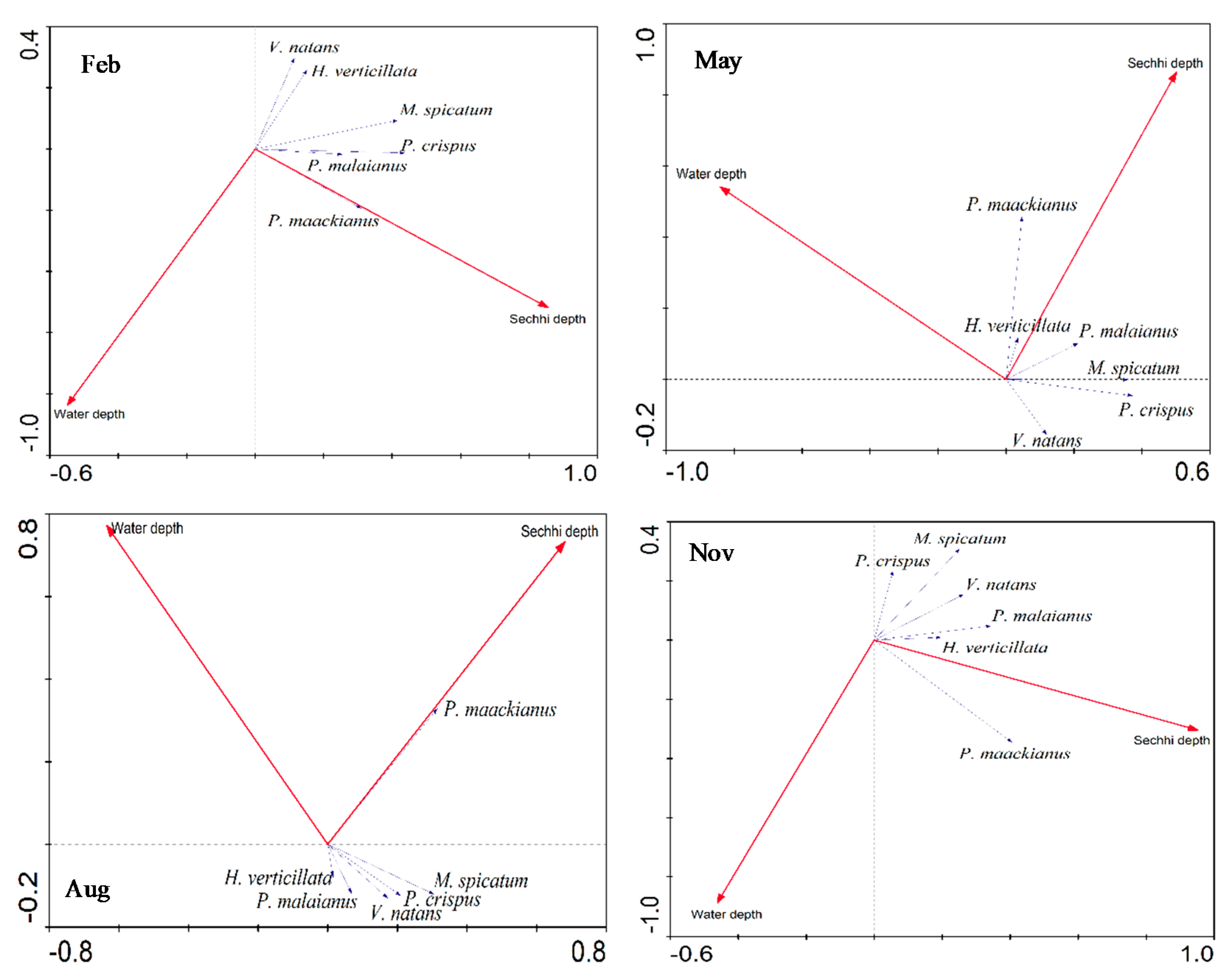
4.2. Effect of Water Depth on Different SAV Species
4.3. A New Method to Determine Proper Water Depth for SAV
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Carpenter, S.R.; Lodge, D.M. Effects of submersed macrophytes on ecosystem processes. Aquat. Bot. 1986, 36, 341–370. [Google Scholar] [CrossRef]
- Scheffer, M.; van den Berg, M.; Breukelaar, A.; Breukers, C.; Coops, H.; Doef, R.; Meijer, M.-L. Vegetated areas with clear water in turbid shallow lakes. Aquat. Bot. 1994, 49, 193–196. [Google Scholar] [CrossRef]
- Blindow, I.; Hargeby, A.; Andersson, G. Seasonal changes of mechanisms maintaining clear water in a shallow lake with abundant Chara vegetation. Aquat. Bot. 2002, 72, 315–334. [Google Scholar] [CrossRef]
- Havens, K.E.; Sharfstein, B.; Brady, M.A.; East, T.L.; Harwell, M.C.; Maki, R.P.; Rodusky, A.J. Recovery of submerged plants from high water stress in a large subtropical lake in Florida, USA. Aquat. Bot. 2004, 78, 67–82. [Google Scholar] [CrossRef]
- Wang, P.; Zhang, Q.; Xu, Y.-S.; Yu, F.-H. Effects of water level fluctuation on the growth of submerged macrophyte communities. Flora 2016, 223, 83–89. [Google Scholar] [CrossRef]
- Ye, C.; Yu, H.-C.; Kong, H.-N.; Song, X.-F.; Zou, G.-Y.; Xu, Q.-J.; Liu, J. Community collocation of four submerged macrophytes on two kinds of sediments in Lake Taihu, China. Ecol. Eng. 2009, 35, 1656–1663. [Google Scholar] [CrossRef]
- Sousa, W.T.Z. Hydrilla verticillata (Hydrocharitaceae), a recent invader threatening Brazil’s freshwater environments: A review of the extent of the problem. Hydrobiologia 2011, 669, 1–20. [Google Scholar] [CrossRef]
- Zhang, X.; Liu, X.; Ding, Q. Morphological responses to water-level fluctuations of two submerged macrophytes, Myriophyllum spicatum and Hydrilla verticillata. J. Plant. Ecol. 2012, 6, 64–70. [Google Scholar] [CrossRef][Green Version]
- Grabas, G.P.; Rokitnicki-Wojcik, D. Characterizing daily water-level fluctuation intensity and water quality relationships with plant communities in Lake Ontario coastal wetlands. J. Great Lakes Res. 2015, 41, 136–144. [Google Scholar] [CrossRef]
- Grabas, G.P.; Fiorino, G.E.; Reinert, A. Vegetation species richness is associated with daily water-level fluctuations in Lake Ontario coastal wetlands. J. Great Lakes Res. 2019, 45, 805–810. [Google Scholar] [CrossRef]
- Havens, K.E. Submerged aquatic vegetation correlations with depth and light attenuating materials in a shallow subtropical lake. Hydrobiologia 2003, 493, 173–186. [Google Scholar] [CrossRef]
- Xu, W.; Hu, W.; Deng, J.; Zhu, J.; Zhou, N.; Liu, X. Impacts of water depth and substrate type on Vallisneria natans at wave-exposed and sheltered sites in a eutrophic large lake. Ecol. Eng. 2016, 97, 344–354. [Google Scholar] [CrossRef]
- Scheffer, M. Ecology of Shallow Lakes; Chapman and Hall: London, UK, 1998; p. 357. [Google Scholar]
- Luo, F.L.; Jiang, X.X.; Li, H.L.; Yu, F.H. Does hydrological fluctuation alter impacts of species richness on biomass in wetland plant communities? J. Plant. Ecol. 2016, 9, 434–441. [Google Scholar] [CrossRef]
- Zhao, G.; Gao, J.; Tian, P.; Tian, K.; Ni, G. Spatial–temporal characteristics of surface water quality in the Taihu Basin, China. Environ. Earth Sci. 2011, 64, 809–819. [Google Scholar] [CrossRef]
- Qin, B.; Gao, G.; Zhu, G.; Zhang, Y.; Song, Y.; Tang, X.; Xu, H.; Deng, J. Lake eutrophication and its ecosystem response. Chin. Sci. Bull. 2012, 58, 961–970. [Google Scholar] [CrossRef]
- Zhai, S.; Hu, W.; Zhu, Z. Ecological impacts of water transfers on Lake Taihu from the Yangtze River, China. Ecol. Eng. 2010, 36, 406–420. [Google Scholar] [CrossRef]
- Li, Q.; Hu, W.; Zhai, S. Integrative indicator for assessing the alert levels of algal bloom in lakes: Lake Taihu as a case study. Environ. Manag. 2016, 57, 237–250. [Google Scholar] [CrossRef]
- Li, K.-Y.; Liu, Z.-W.; Guan, B.-H. Effects of nutrient levels in surface water and sediment on the growth of the floating-leaved macrophyte Trapa maximowiczii: Implication for management of macrophytes in East Bay of Lake Taihu, China. Limnology 2009, 11, 95–101. [Google Scholar] [CrossRef]
- Dong, B.; Qin, B.; Gao, G.; Cai, X. Submerged macrophyte communities and the controlling factors in large, shallow Lake Taihu (China): Sediment distribution and water depth. J. Great Lakes Res. 2014, 40, 646–655. [Google Scholar] [CrossRef]
- Huang, W.; Chen, K.; Shi, X.; Ren, K.; Li, W. The contribution of seeds to the recruitment of a Nymphoides peltata population. Limnologica 2014, 44, 1–8. [Google Scholar] [CrossRef]
- Zhu, J.; Peng, Z.; Liu, X.; Deng, J.; Zhang, Y.; Hu, W. Response of aquatic plants and water quality to large-scale Nymphoides peltata harvest in a shallow lake. Water 2019, 11, 77. [Google Scholar] [CrossRef]
- Qin, B.; Zhu, G.; Gao, G.; Zhang, Y.; Li, W.; Paerl, H.W.; Carmichael, W.W. A drinking water crisis in Lake Taihu, China: Linkage to climatic variability and lake management. Environ. Manag. 2010, 45, 105–112. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Shi, X.; Yang, Z.; Yu, Y.; Shi, L.; Qin, B. Long-term dynamics and drivers of phytoplankton biomass in eutrophic Lake Taihu. Sci. Total Environ. 2018, 645, 876–886. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.; Zhang, Y.; Huang, T.; Yang, H.; Li, Y.; Zhang, Z.; He, M.; Hu, Z.; Song, T.; Zhu, A.X. Long-term variation of phytoplankton biomass and physiology in Taihu lake as observed via MODIS satellite. Water Res. 2019, 153, 187–199. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Gao, Y.; Li, Q.; Gao, J.; Zhai, S.; Zhou, Y.; Cheng, Y. Long-term and inter-monthly dynamics of aquatic vegetation and its relation with environmental factors in Taihu Lake, China. Sci. Total Environ. 2019, 651, 367–380. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Liu, X.; Qin, B.; Shi, K.; Deng, J.; Zhou, Y. Aquatic vegetation in response to increased eutrophication and degraded light climate in Eastern Lake Taihu: Implications for lake ecological restoration. Sci. Rep. 2016, 6, 23867. [Google Scholar] [CrossRef] [PubMed]
- Kissoon, L.T.; Jacob, D.L.; Hanson, M.A.; Herwig, B.R.; Bowe, S.E.; Otte, M.L. Macrophytes in shallow lakes: Relationships with water, sediment and watershed characteristics. Aquat. Bot. 2013, 109, 39–48. [Google Scholar] [CrossRef]
- Gownaris, N.J.; Rountos, K.J.; Kaufman, L.; Kolding, J.; Lwiza, K.M.M.; Pikitch, E.K. The effects of water-level fluctuations on vegetation in a Lake Huron wetland. Wetlands 2008, 28, 487–501. [Google Scholar]
- Zhang, C.; Gao, X.; Wang, L.; Chen, X. Modelling the role of epiphyton and water level for submerged macrophyte development with a modified submerged aquatic vegetation model in a shallow reservoir in China. Ecol. Eng. 2015, 81, 123–132. [Google Scholar] [CrossRef]
- Wilcox, D.A.; Bateman, J.A. Photointerpretation analysis of plant communities in Lake Ontario wetlands following 65years of lake-level regulation. J. Great Lakes Res. 2018, 44, 1306–1313. [Google Scholar] [CrossRef]
- Istvánovics, V.; Honti, M.; Kovács, Á.; Osztoics, A. Distribution of submerged macrophytes along environmental gradients in large, shallow Lake Balaton (Hungary). Aquat. Bot. 2008, 88, 317–330. [Google Scholar] [CrossRef]
- Jarvis, J.C.; Moore, K.A. Influence of environmental factors on Vallisneria americana seed germination. Aquat. Bot. 2008, 88, 283–294. [Google Scholar] [CrossRef]
- Xiao, C.; Wang, X.; Xia, J.; Liu, G. The effect of temperature, water level and burial depth on seed germination of Myriophyllum spicatum and Potamogeton malaianus. Aquat. Bot. 2010, 92, 28–32. [Google Scholar] [CrossRef]
- Yin, L.; Zhang, R.; Xie, Z.; Wang, C.; Li, W. The effect of temperature, substrate, light, oxygen availability and burial depth on Ottelia alismoides seed germination. Aquat. Bot. 2013, 111, 50–53. [Google Scholar] [CrossRef]
- Hartleb, C.F.; Madsen, J.D.; Boylen, C.W. Environmental factors affecting seed germination in Myriophyllum spicatum L. Aquat. Bot. 1993, 45, 15–25. [Google Scholar] [CrossRef]
- Madsen, J.D.; Hartleb, C.F.; Boylen, C.W. Photosynthetic characteristics of Myriophyllum spicatum and six submersed aquatic macrophyte species native to Lake George, New York. Freshw. Biol. 1991, 26, 233–240. [Google Scholar] [CrossRef]
- Bini, L.M.; Thomaz, S.M.; Murphy, K.J.; Camargo, A.F.M. Aquatic macrophyte distribution in relation to water and sediment conditions in the Itaipu Reservoir, Brazil. Hydrobiologia 1999, 415, 147–154. [Google Scholar] [CrossRef]
- Van den Berg, M.S.; Joosse, W.; Coops, H. A statistical model predicting the occurrence and dynamics of submerged macrophytes in shallow lakes in the Netherlands. Hydrobiologia 2003, 506, 611–623. [Google Scholar] [CrossRef]
- Beklioglu, M.; Altinayar, G.; Tan, C.O. Water level control over submerged macrophyte development in five shallow lakes of Mediterranean Turkey. Arch. Hydrobiol. 2006, 166, 535–556. [Google Scholar] [CrossRef]
- Puijalon, S.; Léna, J.P.; Rivière, N.; Champagne, J.Y.; Rostan, J.C.; Bornette, G. Phenotypic plasticity in response to mechanical stress: Hydrodynamic performance and fitness of four aquatic plant species. New Phytol. 2008, 177, 907–917. [Google Scholar] [CrossRef]
- Coops, H.; Hosper, S.H. Water-level management as a tool for the restoration of shallow lakes in the Netherlands. Lake Reserv. Manag. 2002, 18, 293–298. [Google Scholar] [CrossRef]
- Mony, C.; Koschnick, T.J.; Haller, W.T.; Muller, S. Competition between two invasive Hydrocharitaceae (Hydrilla verticillata (L.f.) (Royle) and Egeria densa (Planch)) as influenced by sediment fertility and season. Aquat. Bot. 2007, 86, 236–242. [Google Scholar] [CrossRef]
- Zhu, G.; Li, W.; Zhang, M.; Ni, L.; Wang, S. Adaptation of submerged macrophytes to both water depth and flood intensity as revealed by their mechanical resistance. Hydrobiologia 2002, 696, 77–93. [Google Scholar] [CrossRef]
- Khanday, S.A.; Yousuf, A.R.; Reshi, Z.A.; Rashid, I.; Jehangir, A.; Romshoo, S.A. Management of Nymphoides peltatum using water level fluctuations in freshwater lakes of Kashmir Himalaya. Limnology 2016, 18, 219–231. [Google Scholar] [CrossRef]
- Paillisson, J.-M.; Marion, L. Water level fluctuations for managing excessive plant biomass in shallow lakes. Ecol. Eng. 2011, 37, 241–247. [Google Scholar] [CrossRef]
- Zhang, X.; Liu, X.; Wang, H. Developing water level regulation strategies for macrophytes restoration of a large river-disconnected lake, China. Ecol. Eng. 2014, 68, 25–31. [Google Scholar] [CrossRef]
- Zhang, X.; Wan, A.; Wang, H.; Zhu, L.; Yin, J.; Liu, Z.; Yu, D. The overgrowth of Zizania latifolia in a subtropical floodplain lake: Changes in its distribution and possible water level control measures. Ecol. Eng. 2016, 89, 114–120. [Google Scholar] [CrossRef]
- Liu, X.; Yang, Z.; Yuan, S.; Wang, H. A novel methodology for the assessment of water level requirements in shallow lakes. Ecol. Eng. 2017, 102, 31–38. [Google Scholar] [CrossRef]

| Dependent Variable | Source of Variation | Between Groups | Within Groups | F | Significance | ||
|---|---|---|---|---|---|---|---|
| Sum of Squares | Sum of Squares | Mean Squares | df | ||||
| 2013 vs. 2014 | Biomass | 22.323 | 2443.072 | 8.226 | 297 | 2.714 | 0.101 |
| Water depth | 0.008 | 23.389 | 0.077 | 303 | 0.107 | 0.744 | |
| 2014 vs. 2015 | Biomass | 6.452 | 3792.313 | 12.641 | 300 | 0.51 | 0.476 |
| Water depth | 3.136 | 44.241 | 0.144 | 308 | 21.833 | 0.000 | |
| 2015 vs. 2016 | Biomass | 158.222 | 3053.607 | 9.914 | 308 | 15.959 | 0.000 |
| Water depth | 5.547 | 73.418 | 0.238 | 309 | 23.345 | 0.000 | |
| 2016 vs. 2017 | Biomass | 47.362 | 2424.001 | 7.87 | 308 | 6.018 | 0.015 |
| Water depth | 12.367 | 52.488 | 0.172 | 305 | 71.862 | 0.000 | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhu, J.; Deng, J.; Zhang, Y.; Peng, Z.; Hu, W. Response of Submerged Aquatic Vegetation to Water Depth in a Large Shallow Lake after an Extreme Rainfall Event. Water 2019, 11, 2412. https://doi.org/10.3390/w11112412
Zhu J, Deng J, Zhang Y, Peng Z, Hu W. Response of Submerged Aquatic Vegetation to Water Depth in a Large Shallow Lake after an Extreme Rainfall Event. Water. 2019; 11(11):2412. https://doi.org/10.3390/w11112412
Chicago/Turabian StyleZhu, Jinge, Jiancai Deng, Yihui Zhang, Zhaoliang Peng, and Weiping Hu. 2019. "Response of Submerged Aquatic Vegetation to Water Depth in a Large Shallow Lake after an Extreme Rainfall Event" Water 11, no. 11: 2412. https://doi.org/10.3390/w11112412
APA StyleZhu, J., Deng, J., Zhang, Y., Peng, Z., & Hu, W. (2019). Response of Submerged Aquatic Vegetation to Water Depth in a Large Shallow Lake after an Extreme Rainfall Event. Water, 11(11), 2412. https://doi.org/10.3390/w11112412




