A Feasibility Study of Wastewater Treatment Using Domestic Microalgae and Analysis of Biomass for Potential Applications
Abstract
:1. Introduction
2. Materials and Methods
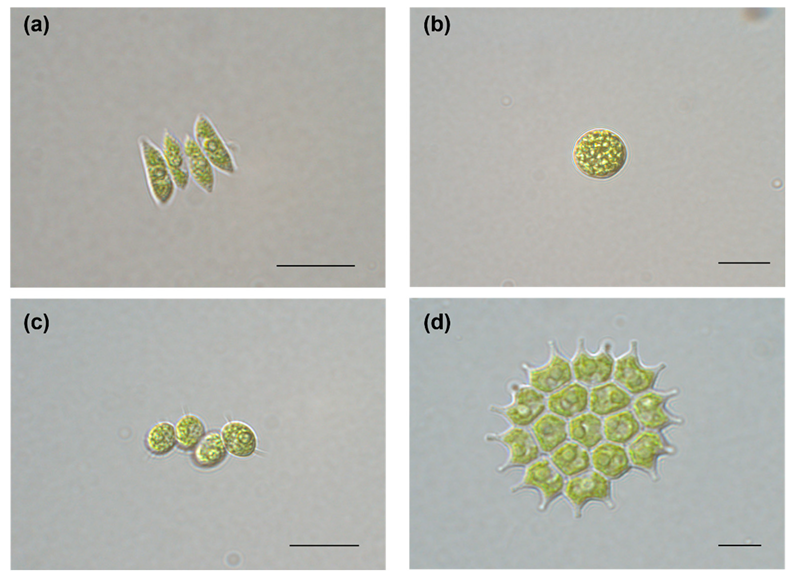
2.1. Selecting Candidate Strains for Wastewater Treatment
2.1.1. Screening Candidate Microalgae Using AW with KNUA Strains
2.1.2. Dominant Microalgae in Mass Cultivation in Chilgok
2.2. Wastewater Collection and Characterization
2.3. Measurement of Growth Rate
2.3.1. Growth Conditions
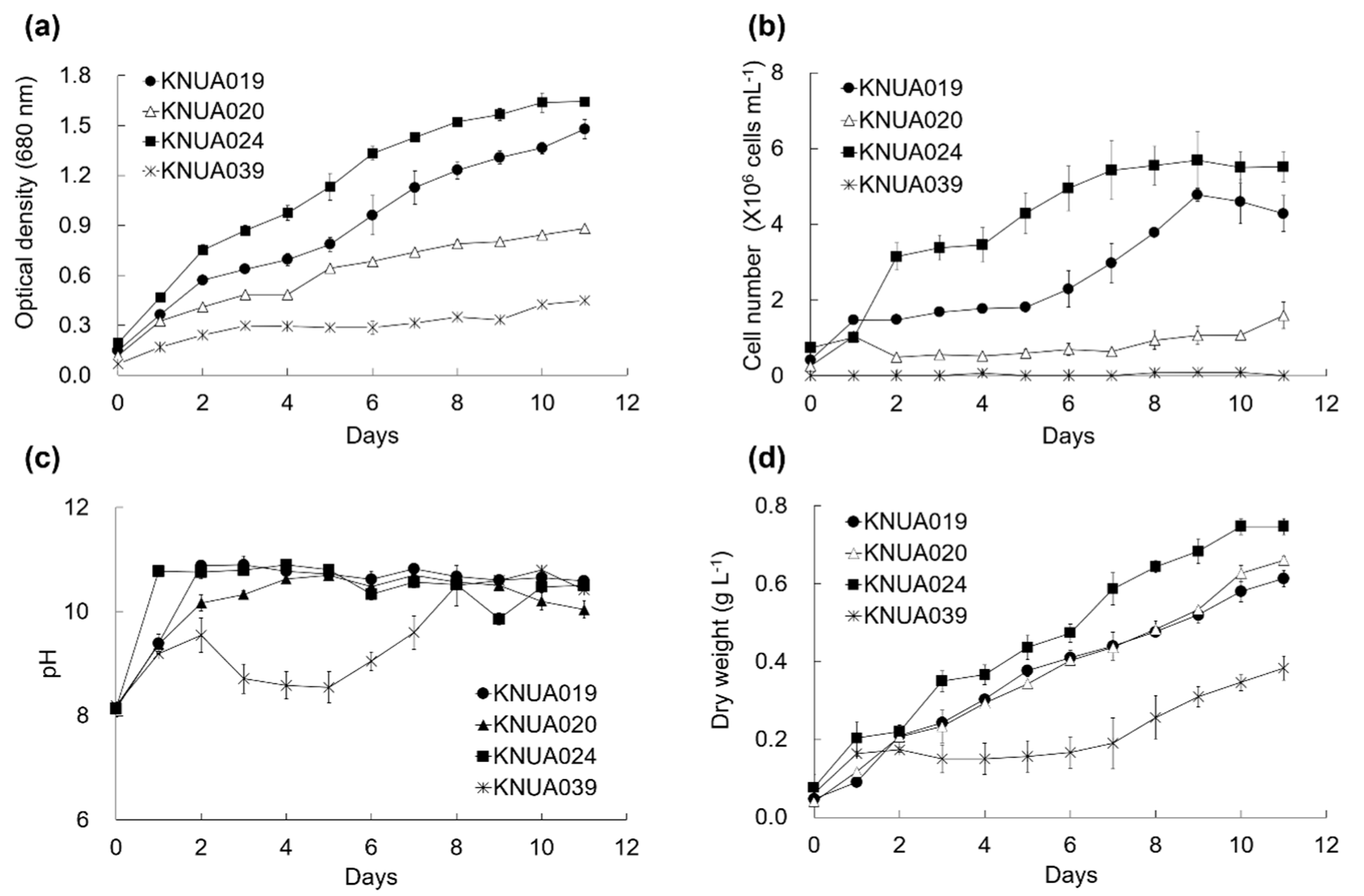
2.3.2. Measurement of Growth Rate and Biomass Productivity
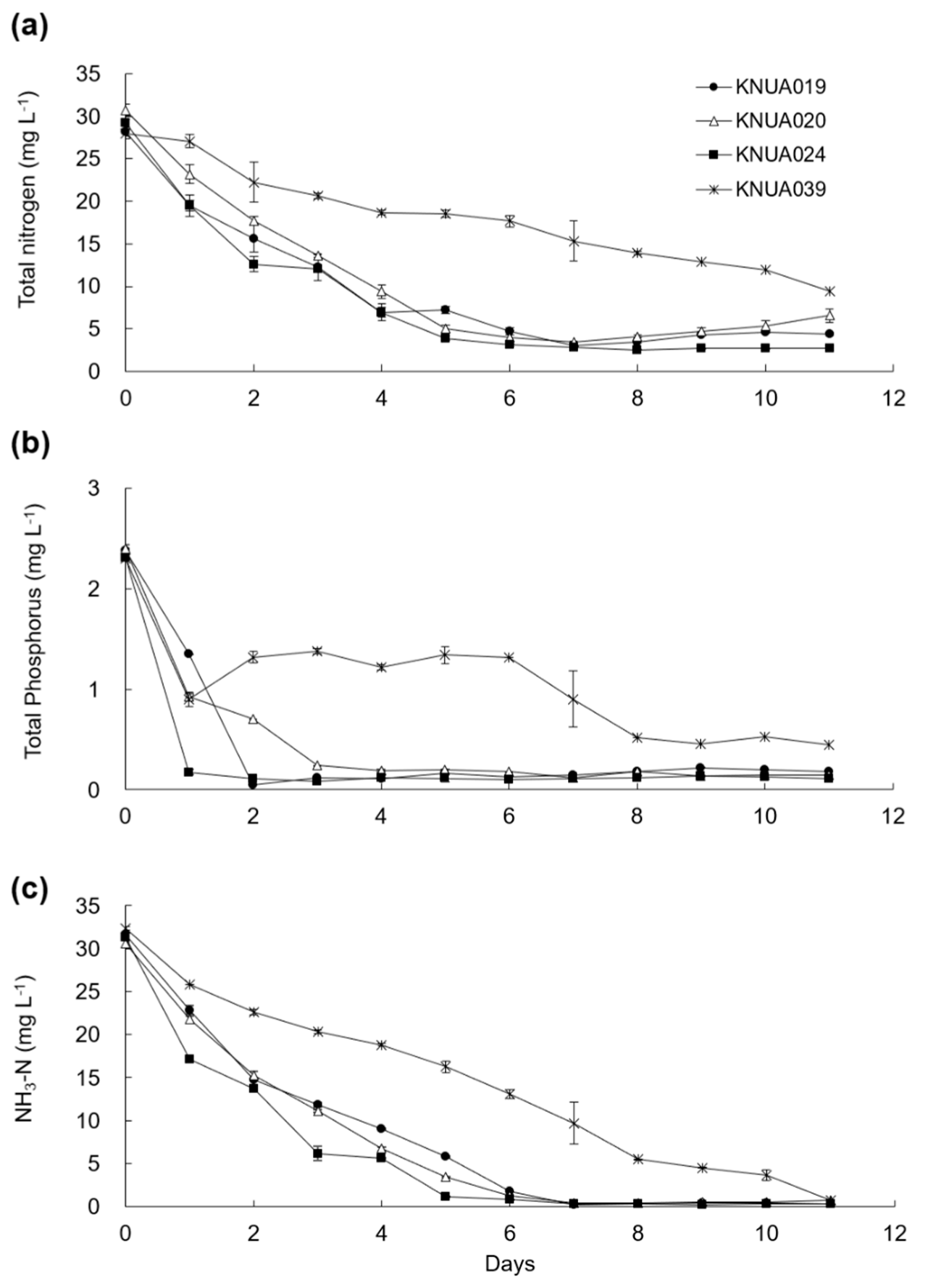
2.4. Measurement of Nutrient and Metal Removal
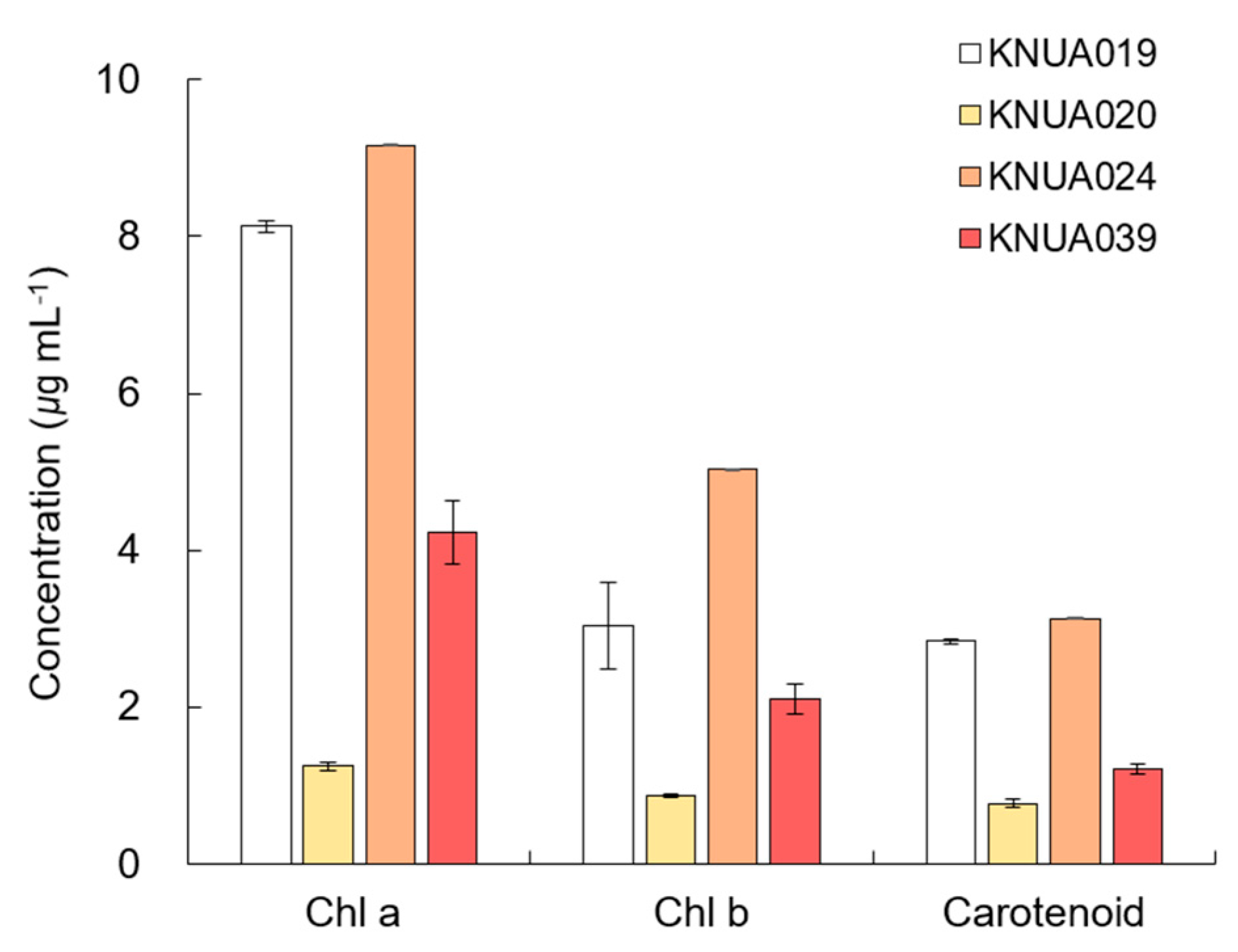
2.5. Measurement of Physicochemical Properties
2.6. Biochemical Composition of Biomass
2.7. Proximate and Ultimate Analysis
2.8. Analysis of Fatty Acid Methyl−Ester (FAME) Contents
2.9. Biodiesel Properties Based on FAME Profiles
3. Results
3.1. Selecting Candidate Strains for Wastewater Treatment
3.2. Wastewater Collection and Characterization
3.3. Measurement of Growth Rate and Biomass Productivity
3.4. Nutrient Removal Efficiency
3.5. Chlorophyll and Total Carotenoid Contents
3.6. Biochemical Analysis
3.7. Proximate and Ultimate Analysis
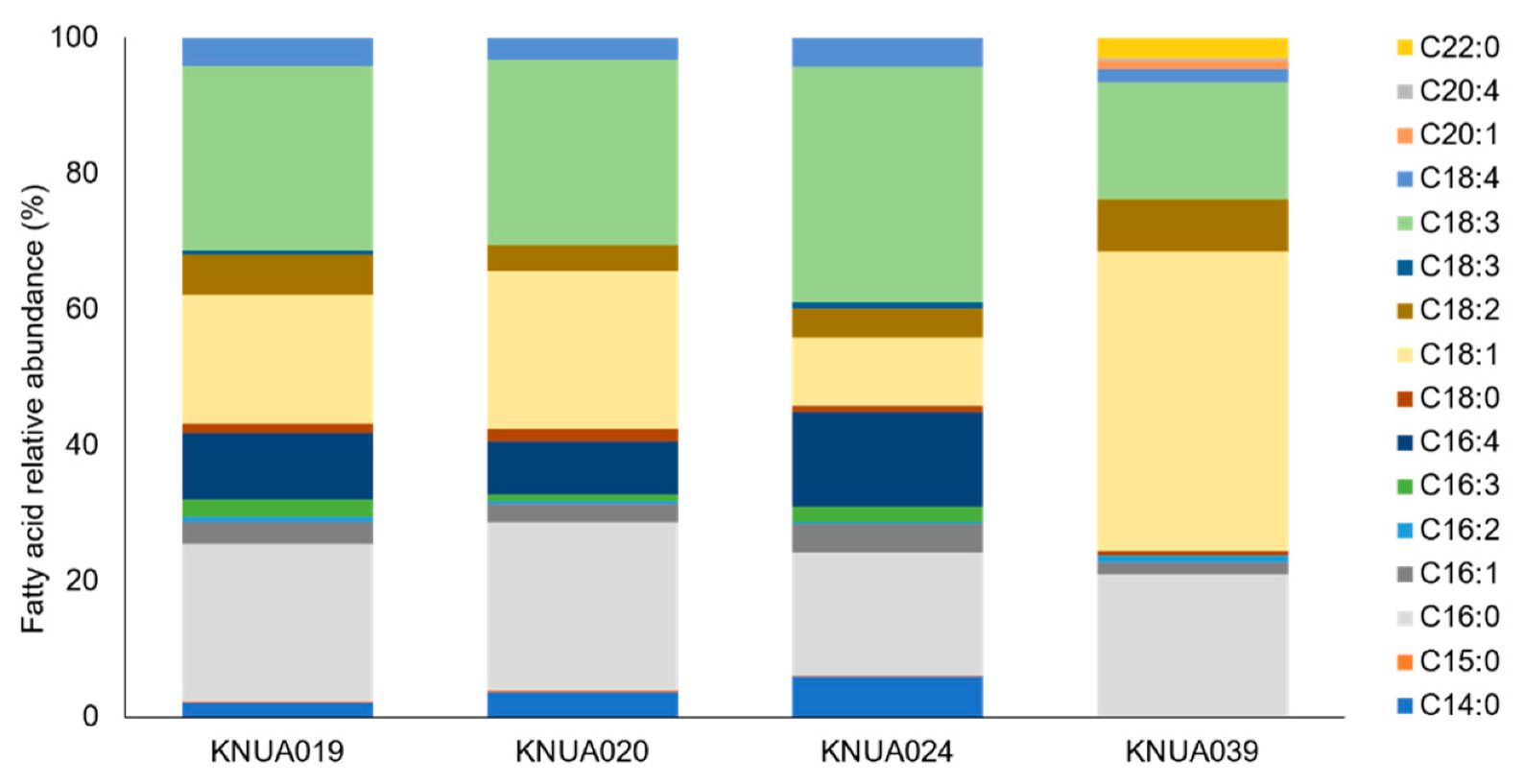
3.8. FAME Analysis with GC/MS
3.9. Biodiesel and Bio-Lubricant Properties Based on FAME Profiles
4. Discussion
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Zhu, B.; Chen, G.; Cao, X.; Wei, D. Molecular Characterization of CO2 Sequestration and Assimilation in Microalgae and Its Biotechnological Applications. Bioresour. Technol. 2017, 5, 199. [Google Scholar] [CrossRef] [PubMed]
- Bozbas, K. Biodiesel as an Alternative Motor Fuel: Production and Policies in the European Union. Renew. Sustain. Energy Rev. 2008, 12, 542–552. [Google Scholar] [CrossRef]
- Mondal, M.; Goswami, S.; Ghosh, A.; Oinam, G.; Tiwari, O.N.; Das, P.; Gayen, K.; Mandal, M.K.; Halder, G.N. Production of Biodiesel from Microalgae through Biological Carbon Capture: A Review. 3 Biotech. 2017, 7, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Slade, R.; Bauen, A. Micro-Algae Cultivation for Biofuels: Cost, Energy Balance, Environmental Impacts and Future Prospects. Biomass Bioenergy 2013, 53, 29–38. [Google Scholar] [CrossRef]
- Ji, M.K.; Abou-Shanab, R.A.I.; Kim, S.H.; Salama, E.S.; Lee, S.H.; Kabra, A.N.; Lee, Y.S.; Hong, S.; Jeon, B.H. Cultivation of Microalgae Species in Tertiary Municipal Wastewater Supplemented with CO2 for Nutrient Removal and Biomass Production. Ecol. Eng. 2013, 58, 142–148. [Google Scholar] [CrossRef]
- Cheng, D.L.; Ngo, H.H.; Guo, W.S.; Chang, S.W.; Nguyen, D.D.; Kumar, S.M. Microalgae Biomass from Swine Wastewater and Its Conversion to Bioenergy. Bioresour. Technol. 2019, 275, 109–122. [Google Scholar] [CrossRef]
- Abou-Shanab, R.A.I.; Ji, M.-K.; Kim, H.-C.; Paeng, K.-J.; Jeon, B.-H. Microalgal Species Growing on Piggery Wastewater as a Valuable Candidate for Nutrient Removal and Biodiesel Production. J. Environ. Manag. 2013, 115, 257–264. [Google Scholar] [CrossRef]
- Wu, L.F.; Chen, P.C.; Huang, A.P.; Lee, C.M. The Feasibility of Biodiesel Production by Microalgae Using Industrial Wastewater. Bioresour. Technol. 2012, 113, 14–18. [Google Scholar] [CrossRef]
- Sebastian, S.; Nair, K.V.K. Total Removal of Coliforms and E. Coli from Domestic Sewage by High-Rate Pond Mass Culture of Scenedesmus Obliquus. Environ. Pollut. Ser. A Ecol. Biol. 1984, 34, 197–206. [Google Scholar] [CrossRef]
- Stemmler, K.; Massimi, R.; Kirkwood, A.E. Growth and Fatty Acid Characterization of Microalgae Isolated from Municipal Waste-Treatment Systems and the Potential Role of Algal-Associated Bacteria in Feedstock Production. PeerJ. 2016, 4, e1780. [Google Scholar] [CrossRef]
- Feng, Y.; Li, C.; Zhang, D. Lipid Production of Chlorella Vulgaris Cultured in Artificial Wastewater Medium. Bioresour. Technol. 2011, 102, 101–105. [Google Scholar] [CrossRef] [PubMed]
- Stanier, R.Y.; Deruelles, J.; Rippka, R.; Herdman, M.; Waterbury, J.B. Generic Assignments, Strain Histories and Properties of Pure Cultures of Cyanobacteria. Microbiol. 1979, 111, 1–61. [Google Scholar] [CrossRef] [Green Version]
- Hong, J.W.; Kim, O.H.; Jo, S.W.; Do, J.M.; Yoon, H.S. Microalgal Biomass Productivity and Dominant Species Transition in a Korean Mass Cultivation System. Algal Res. 2017, 26, 365–370. [Google Scholar] [CrossRef]
- Rice, E.W. Standard Methods for the Examination of Water and Wastewater, 22nd ed.; American Water Works Association: Washington, DC, USA, 2012. [Google Scholar]
- Levasseur, M.; Thompson, P.A.; Harrison, P.J. Physiological Acclimation of Marine Phytoplankton to Different Nitrogen Sources. J. Phycol. 1993, 29, 587–595. [Google Scholar] [CrossRef]
- Wellburn, A.R. The Spectral Determination of Chlorophylls a and b, as Well as Total Carotenoids, Using Various Solvents with Spectrophotometers of Different Resolution. J. Plant Physiol. 1994, 144, 307–313. [Google Scholar] [CrossRef]
- Mariotti, F.; Tomé, D.; Mirand, P.P. Converting Nitrogen into Protein—Beyond 6.25 and Jones’ Factors. Crit. Rev. Food Sci. Nutr. 2008, 48, 177–184. [Google Scholar] [CrossRef] [PubMed]
- Mishra, S.K.; Suh, W.I.; Farooq, W.; Moon, M.; Shrivastav, A.; Park, M.S.; Yang, J.W. Rapid Quantification of Microalgal Lipids in Aqueous Medium by a Simple Colorimetric Method. Bioresour. Technol. 2014, 155, 330–333. [Google Scholar] [CrossRef]
- Bi, Z.; He, B.B. Characterization of Microalgae for the Purpose of Biofuel Production. Trans. ASABE 2013, 56, 1529–1539. [Google Scholar] [CrossRef]
- Given, P.H.; Weldon, D.; Zoeller, J.H. Calculation of Calorific Values of Coals from Ultimate Analyses: Theoretical Basis and Geochemical Implications. Fuel 1986, 65, 849–854. [Google Scholar] [CrossRef]
- Breuer, G.; Evers, W.A.C.; de Vree, J.H.; Kleinegris, D.M.M.; Martens, D.E.; Wijffels, R.H.; Lamers, P.P. Analysis of Fatty Acid Content and Composition in Microalgae. J. Vis. Exp. 2013, 80, e50628. [Google Scholar] [CrossRef]
- Ramos, M.J.; Fernández, C.M.; Casas, A.; Rodríguez, L.; Pérez, Á. Influence of Fatty Acid Composition of Raw Materials on Biodiesel Properties. Bioresour. Technol. 2009, 100, 261–268. [Google Scholar] [CrossRef] [PubMed]
- Francisco, É.C.; Neves, D.B.; Jacob-Lopes, E.; Franco, T.T. Microalgae as Feedstock for Biodiesel Production: Carbon Dioxide Sequestration, Lipid Production and Biofuel Quality. J. Chem. Technol. Biotechnol. 2010, 85, 395–403. [Google Scholar] [CrossRef]
- Islam, M.A.; Magnusson, M.; Brown, R.J.; Ayoko, G.A.; Nabi, M.N.; Heimann, K. Microalgal Species Selection for Biodiesel Production Based on Fuel Properties Derived from Fatty Acid Profiles. Energies 2013, 6, 5676–5702. [Google Scholar] [CrossRef] [Green Version]
- Wu, H.; Miao, X. Biodiesel Quality and Biochemical Changes of Microalgae Chlorella Pyrenoidosa and Scenedesmus Obliquus in Response to Nitrate Levels. Bioresour. Technol. 2014, 170, 421–427. [Google Scholar] [CrossRef]
- Caporgno, M.P.; Mathys, A. Trends in Microalgae Incorporation into Innovative Food Products with Potential Health Benefits. Front. Nutr. 2018, 5, 58. [Google Scholar] [CrossRef]
- Hong, J.W.; Kim, O.H.; Jo, S.-W.; Kim, H.; Jeong, M.R.; Park, K.M.; Lee, K.I.; Yoon, H.-S. Biochemical Composition of a Korean Domestic Microalga Chlorella Vulgaris KNUA027. Microbiol. Biotechnol. Lett. 2016, 44, 400–407. [Google Scholar] [CrossRef]
- Ross, A.B.; Jones, J.M.; Kubacki, M.L.; Bridgeman, T. Classification of Macroalgae as Fuel and Its Thermochemical Behaviour. Bioresour. Technol. 2008, 99, 6494–6504. [Google Scholar] [CrossRef]
- Knothe, G. “Designer” Biodiesel: Optimizing Fatty Ester Composition to Improve Fuel Properties. Energy Fuels 2008, 22, 1358–1364. [Google Scholar] [CrossRef]
- Nascimento, I.A.; Marques, S.S.I.; Cabanelas, I.T.D.; Pereira, S.A.; Druzian, J.I.; de Souza, C.O.; Vich, D.V.; de Carvalho, G.C.; Nascimento, M.A. Screening Microalgae Strains for Biodiesel Production: Lipid Productivity and Estimation of Fuel Quality Based on Fatty Acids Profiles as Selective Criteria. Bioenergy Res. 2013, 6, 1–13. [Google Scholar] [CrossRef]
- Karmakar, G.; Ghosh, P.; Sharma, B. Chemically Modifying Vegetable Oils to Prepare Green Lubricants. Lubricants 2017, 5, 44. [Google Scholar] [CrossRef]
- Komolafe, O.; Velasquez Orta, S.B.; Monje-Ramirez, I.; Noguez, I.Y.; Harvey, A.P.; Orta Ledesma, M.T. Biodiesel Production from Indigenous Microalgae Grown in Wastewater. Bioresour. Technol. 2014, 154, 297–304. [Google Scholar] [CrossRef] [PubMed]
- Ji, F.; Liu, Y.; Hao, R.; Li, G.; Zhou, Y.; Dong, R. Biomass Production and Nutrients Removal by a New Microalgae Strain Desmodesmus sp. in Anaerobic Digestion Wastewater. Bioresour. Technol. 2014, 161, 200–207. [Google Scholar] [CrossRef] [PubMed]
- Santiago, A.F.; Calijuri, M.L.; Assemany, P.P.; Calijuri, M.D.C.; Dos Reis, A.J.D. Algal Biomass Production and Wastewater Treatment in High Rate Algal Ponds Receiving Disinfected Effluent. Environ. Technol. 2013, 34, 1877–1885. [Google Scholar] [CrossRef] [PubMed]
| Parameter | Value |
|---|---|
| Temperature | 25.8 °C |
| pH | 7.24 |
| Salinity | 0.5 |
| Conductivity | 913 μs cm−1 |
| Total solid (TS) | 0.013 g L−1 |
| Total dissolved solid (TDS) | 45.9 mg L−1 |
| Turbidity | 67.8 NTU |
| Chemical oxygen demand (COD) | 12.4 mg L−1 |
| Total nitrogen (TN) | 30.9 mg L−1 |
| Ammonia (NH3-N) | 29.19 mg L−1 |
| Nitrate (NO3-N) | N.D. 1 |
| Total phosphorus (TP) | 2.29 mg L−1 |
| Element concentration | (mg L−1) |
| Na | 64.28 |
| Ca | 50.09 |
| K | 13.74 |
| Fe | 0.69 |
| Mg | 10.32 |
| P | 2.39 |
| S | 28.93 |
| Carbohydrate | Protein | Lipid | |
|---|---|---|---|
| Tetradesmus obliquus KNUA019 | 27.24 ± 2.39 | 34.29 ± 0.0 | 22.38 ± 3.42 |
| Asterarcys quadricellulare KNUA020 | 26.98 ± 1.91 | 35.78 ± 0.77 | 16.99 ± 2.56 |
| Desmodesmus sp. KNUA024 | 24.41 ± 0.99 | 39.66 ± 0.18 | 23.51 ± 3.62 |
| Pseudopediastrum sp. KNUA039 | 27.86 ± 3.49 | 32.15 ± 0.13 | 26.45 ± 3.28 |
| Tetradesmus obliquus KNUA019 | Asterarcys quadricellulare KNUA020 | Desmodesmus sp. KNUA024 | Pseudopediastrum sp. KNUA039 | |
|---|---|---|---|---|
| Proximate analysis (wt%) | ||||
| MC 1 | 5.9 ± 0.0 | 6.6 ± 0.2 | 5.9 ± 0.3 | 5.6 ± 0.1 |
| VM 2 | 90.6 ± 0.3 | 88.8 ± 0.1 | 88.9 ± 0.2 | 91.8 ± 0.5 |
| Ash | 3.7 ± 0.3 | 4.6 ± 0.3 | 5.2 ± 0.6 | 2.5 ± 0.6 |
| Ultimate analysis (wt%) | ||||
| C | 44.9 ± 0.4 | 41.6 ± 0.9 | 44.6 ± 0.8 | 46.1 ± 0.2 |
| H | 6.7 ± 0.1 | 6.6 ± 0.0 | 6.6 ± 0.2 | 7.1 ± 0.1 |
| O | 38.8 ± 0.8 | 40.9 ± 0.8 | 36.9 ± 0.4 | 38.8 ± 0.4 |
| N | 5.5 ± 0.0 | 5.7 ± 0.1 | 6.3 ± 0.0 | 5.1 ± 0.0 |
| S | 0.5 ± 0.0 | 0.6 ± 0.0 | 0.4 ± 0.0 | 0.4 ± 0.0 |
| CV 3 (MJ kg−1) | 19.5 ± 0.4 | 18.0 ± 0.4 | 19.5 ± 0.2 | 20.5 ± 0 |
| Tetradesmus obliquus KNUA019 | Asterarcys quadricellulare KNUA020 | Desmodesmus sp. KNUA024 | Pseudopediastrum sp. KNUA039 | |
|---|---|---|---|---|
| SV | 183 | 179 | 181 | 170 |
| IV | 150 | 131 | 170 | 95 |
| CN | 41.0 | 44.7 | 34.6 | 55.3 |
| DU | 114 | 102 | 122 | 91 |
| LCSF | 2.8 | 3.0 | 2.0 | 2.2 |
| CFPP | −7.7 | −7.0 | −10.1 | −9.7 |
| OS | 6.4 | 6.8 | 5.9 | 8 |
| υ | 3.64 | 3.76 | 3.39 | 3.92 |
| ρ | 0.89 | 0.88 | 0.89 | 0.83 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Do, J.-M.; Jo, S.-W.; Kim, I.-S.; Na, H.; Lee, J.H.; Kim, H.S.; Yoon, H.-S. A Feasibility Study of Wastewater Treatment Using Domestic Microalgae and Analysis of Biomass for Potential Applications. Water 2019, 11, 2294. https://doi.org/10.3390/w11112294
Do J-M, Jo S-W, Kim I-S, Na H, Lee JH, Kim HS, Yoon H-S. A Feasibility Study of Wastewater Treatment Using Domestic Microalgae and Analysis of Biomass for Potential Applications. Water. 2019; 11(11):2294. https://doi.org/10.3390/w11112294
Chicago/Turabian StyleDo, Jeong-Mi, Seung-Woo Jo, Il-Sup Kim, Ho Na, Jae Hak Lee, Han Soon Kim, and Ho-Sung Yoon. 2019. "A Feasibility Study of Wastewater Treatment Using Domestic Microalgae and Analysis of Biomass for Potential Applications" Water 11, no. 11: 2294. https://doi.org/10.3390/w11112294
APA StyleDo, J.-M., Jo, S.-W., Kim, I.-S., Na, H., Lee, J. H., Kim, H. S., & Yoon, H.-S. (2019). A Feasibility Study of Wastewater Treatment Using Domestic Microalgae and Analysis of Biomass for Potential Applications. Water, 11(11), 2294. https://doi.org/10.3390/w11112294





