Relationship between Soil Salinization and Groundwater Hydration in Yaoba Oasis, Northwest China
Abstract
1. Introduction
2. Materials and Methods
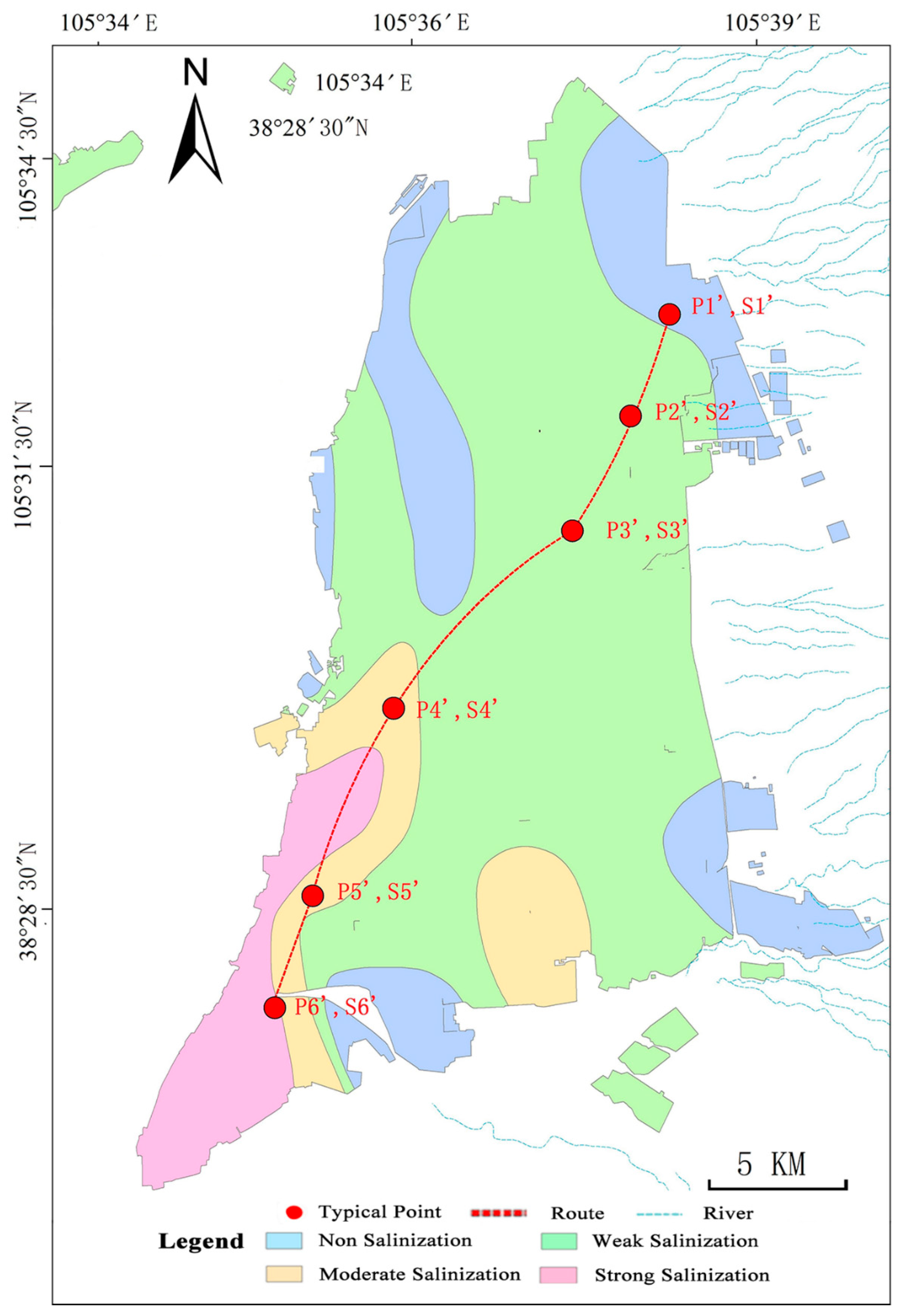
2.1. Study Area
2.2. Data Sources and Research Methods
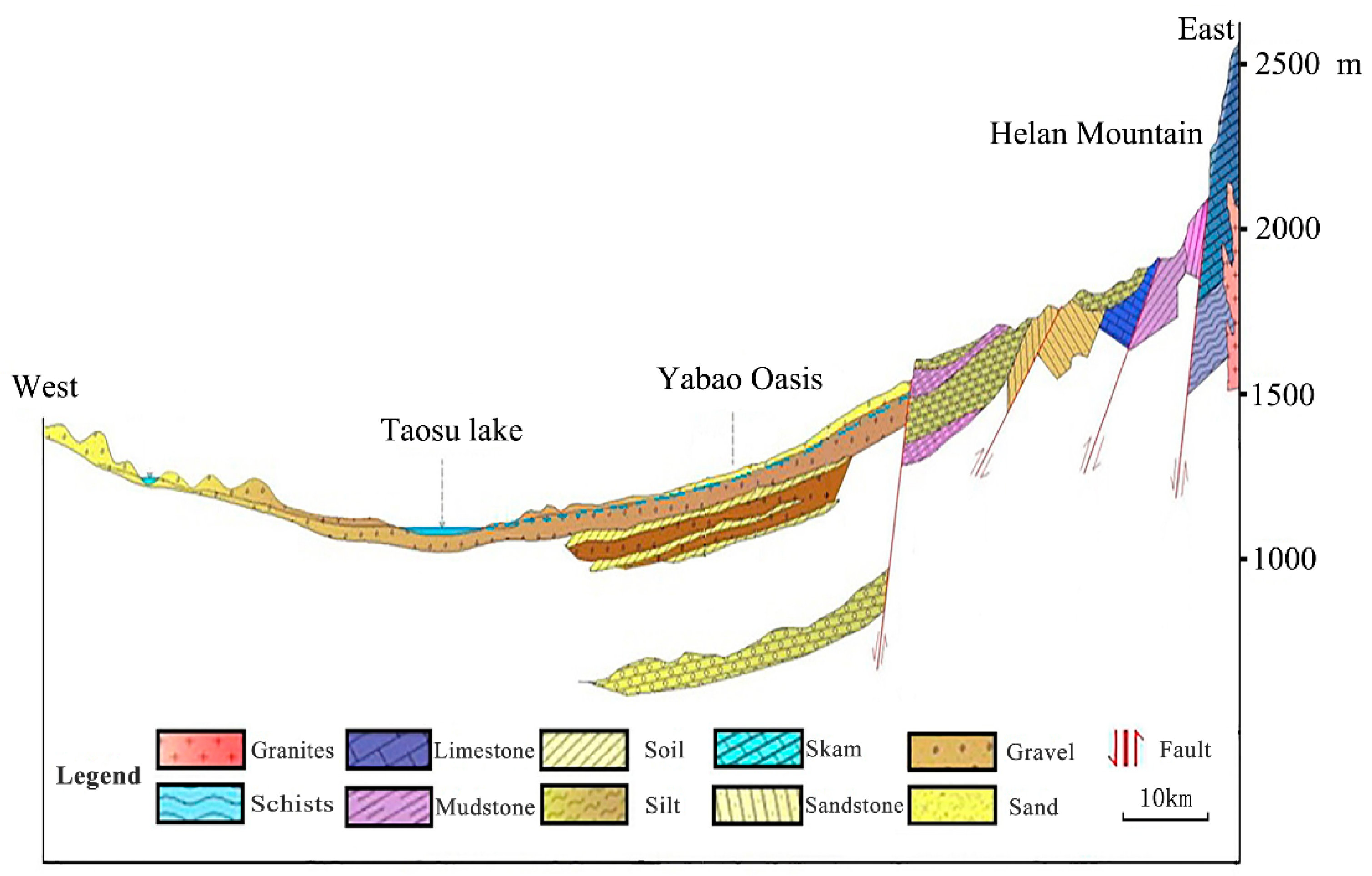
2.2.1. Geological and Hydrogeological Setting
2.2.2. Experiments
2.2.3. Data Processing Method
2.2.4. Gray Correlation Analysis
3. Results and Discussion
3.1. Chemical Characteristics of Groundwater and Soil Salinization in Yaoba Oasis
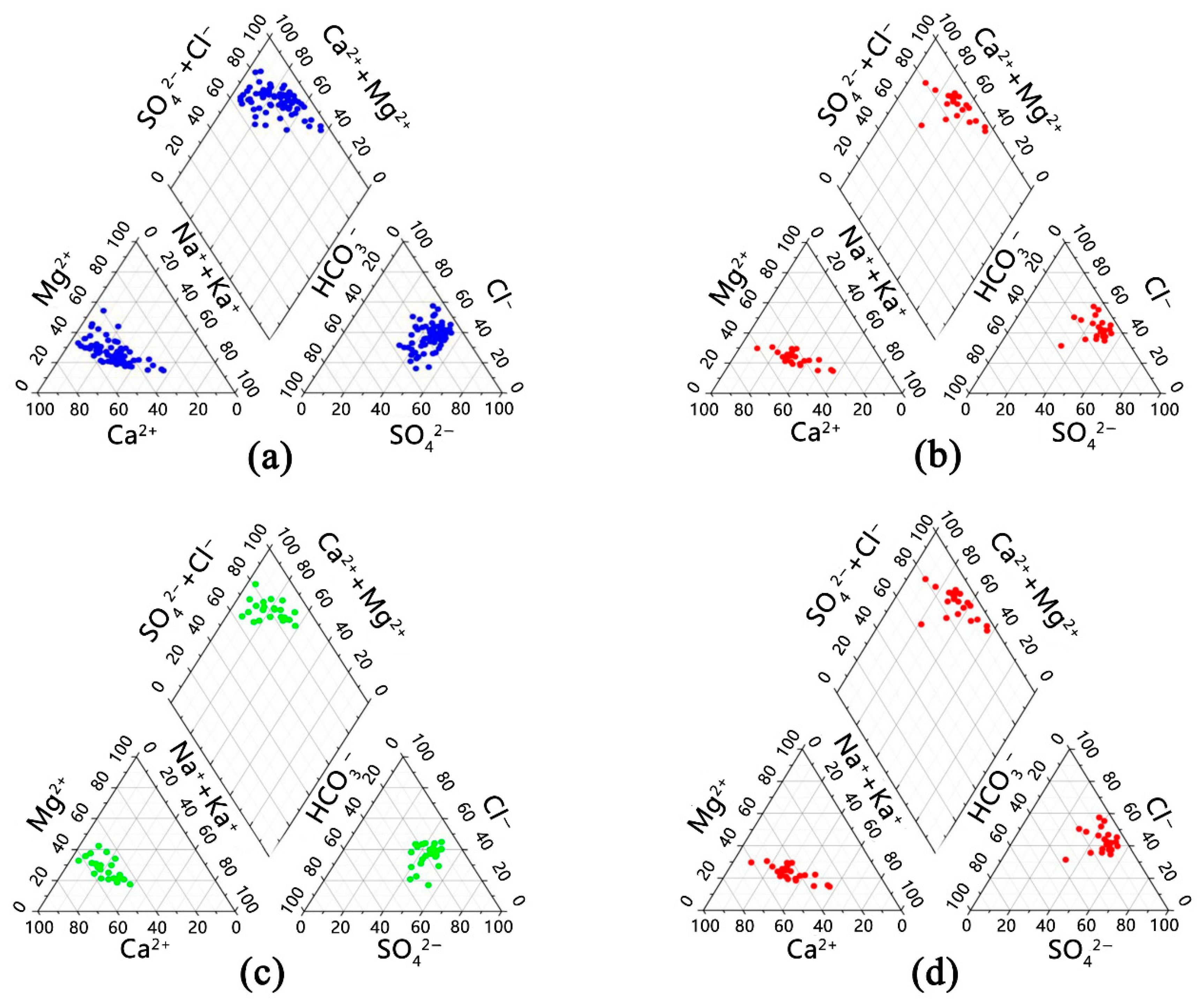
3.1.1. Chemical Characteristics of Groundwater
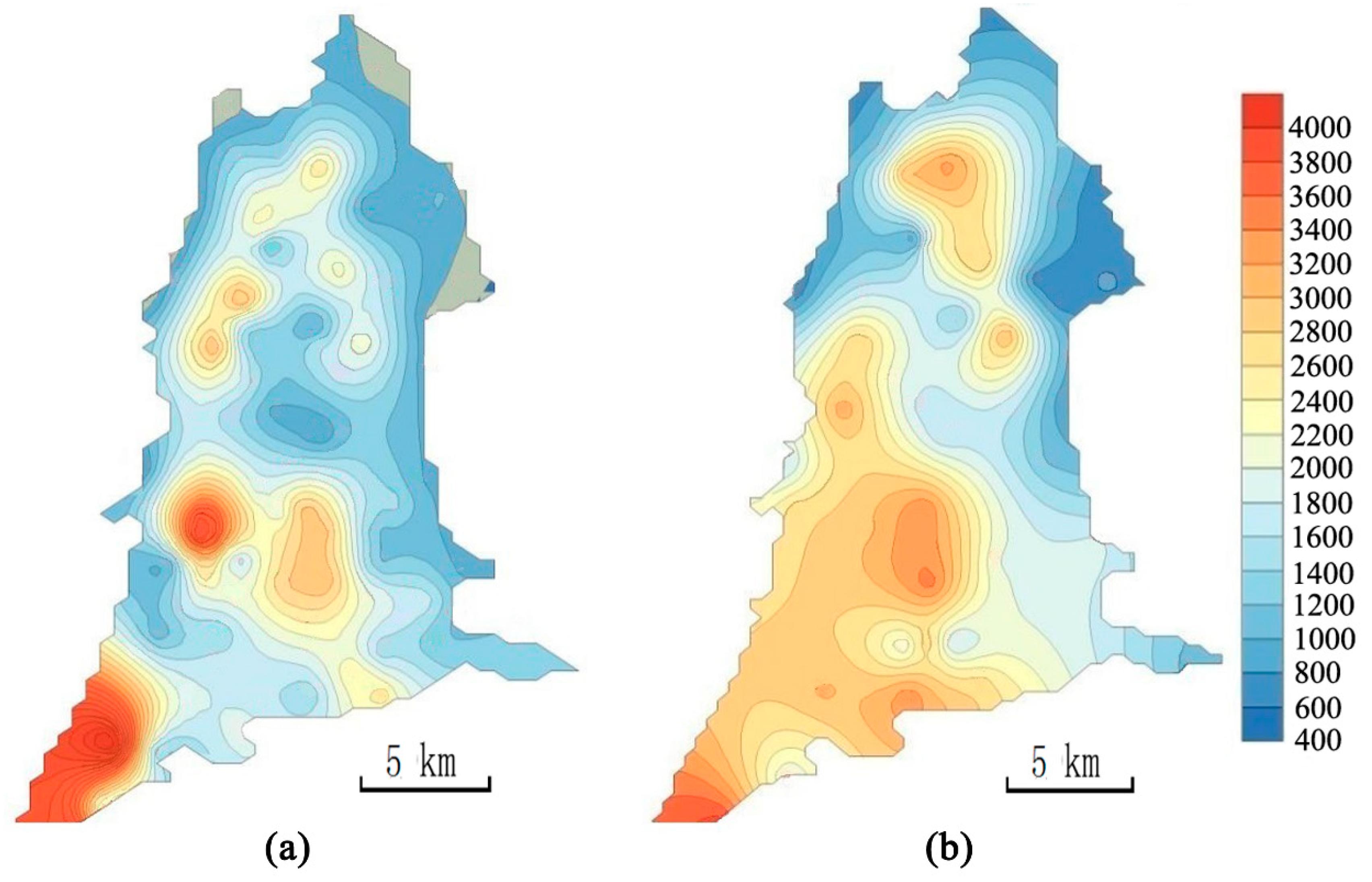
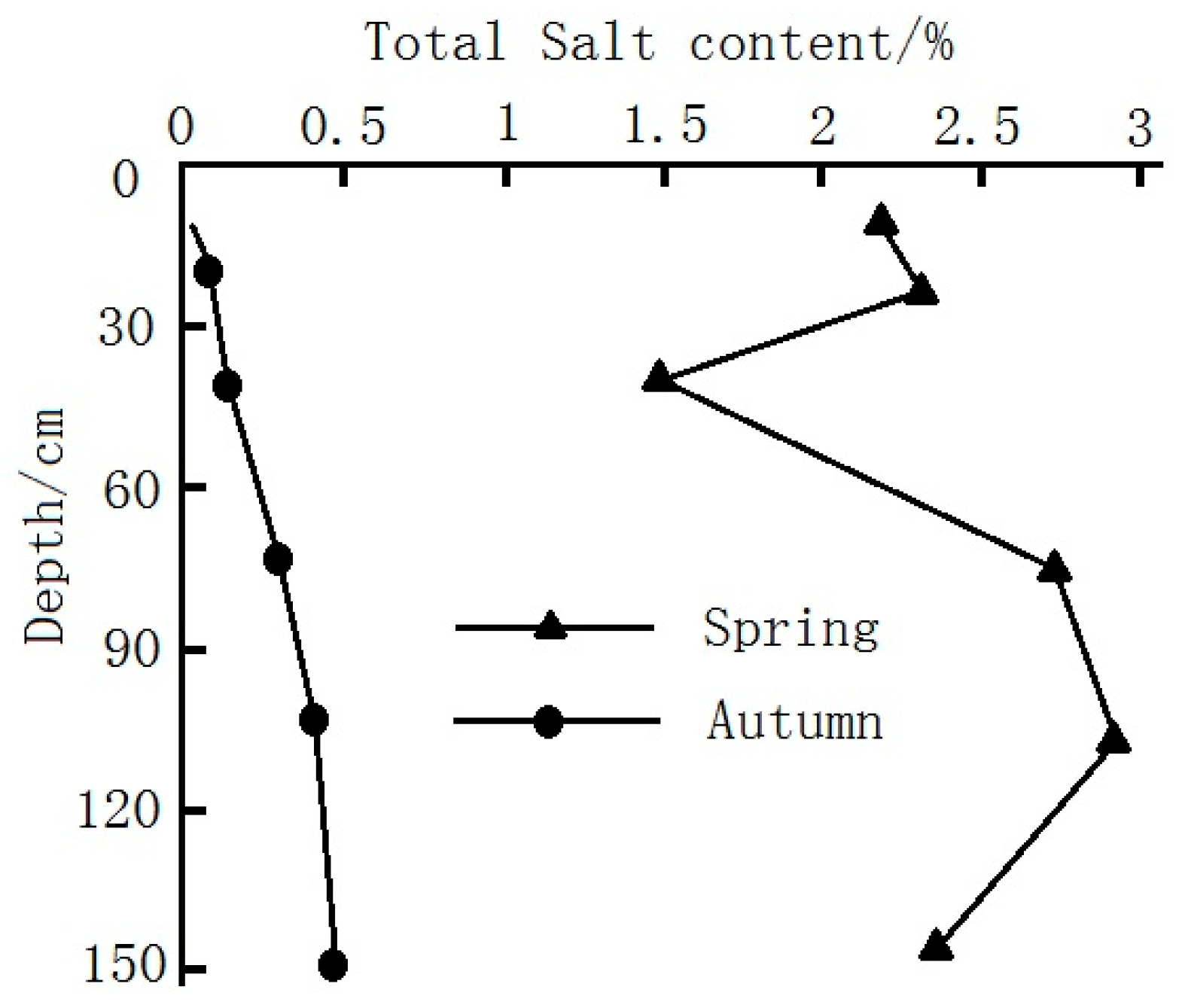
3.1.2. Characteristics of Soil Salinization
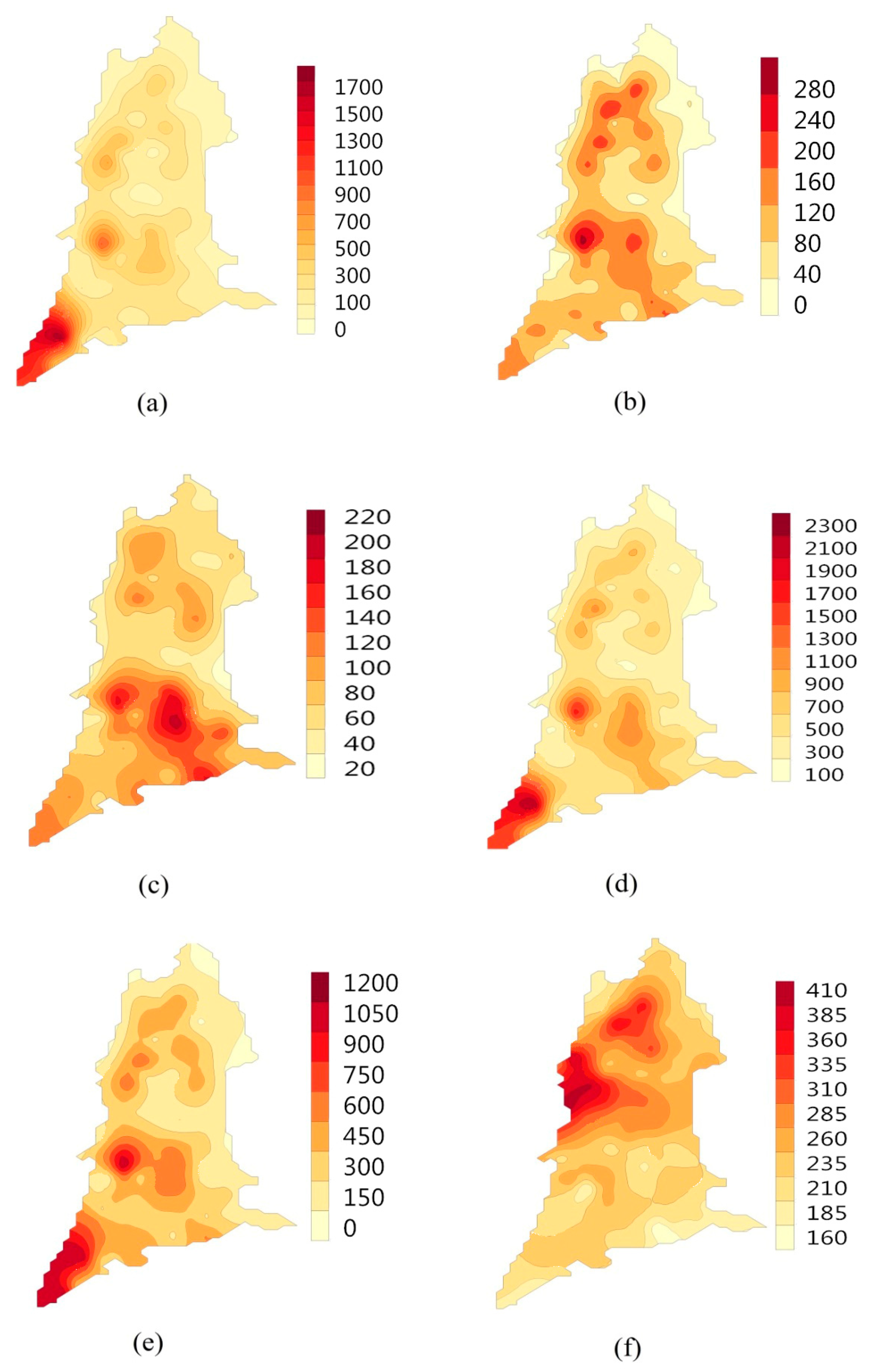
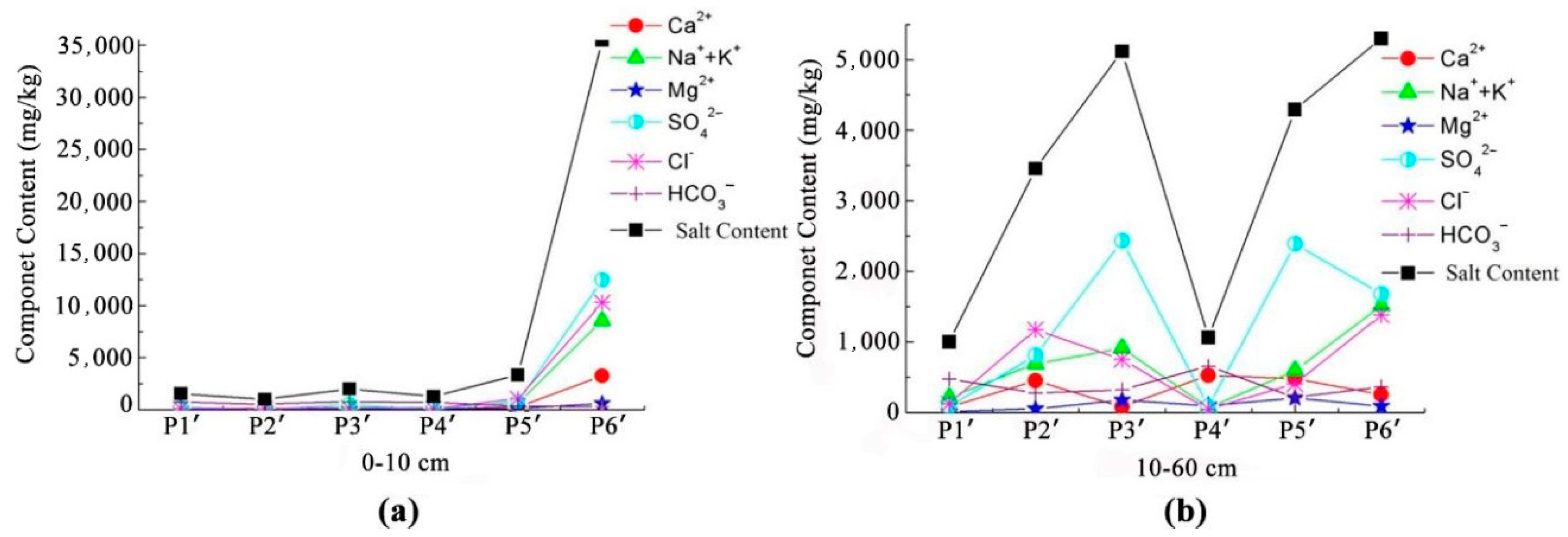
3.2. Relationship between Soil Salt Content and Chemical Components of Groundwater
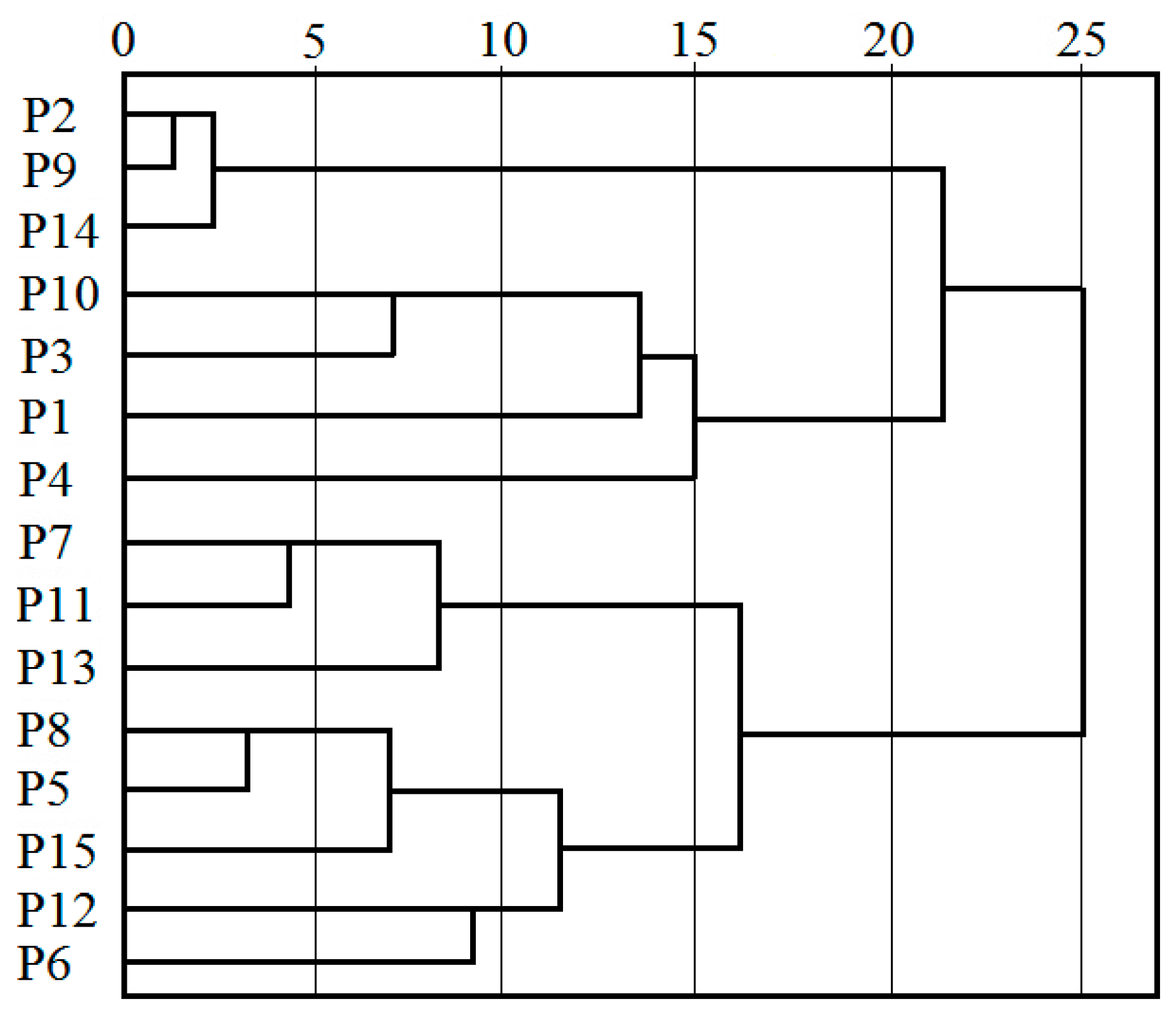
3.3. Correlation Analysis between Soil Salinity and Groundwater Quality
4. Conclusions
- Influence of irrigation: Farming and animal husbandry mainly use water from deep wells due to the climate characteristics of this area. When deep well irrigation is used frequently, a large amount of residual salt in the soil becomes active; with the strong evaporation of the soil, the accumulation of soil salinization reaches the surface.
- Influence of irrigation water (groundwater) quality: The irrigation water of the study area is mainly supplied by groundwater from Helan Mountain and Tengger Desert. The parent material of the soil in the west of this area is lacustrine sediments; therefore, the soil contains considerable soluble alkali metals (mainly sodium). In this way, bivalent cations (mainly Ca2+ ions) in groundwater are exchanged with sodium in the soil in the recharge process, resulting in many soluble Na+ ions in groundwater in this area. The deep water quality worsens, and alkalinity increases. In this way, when the groundwater that contains high sodium ions is used for irrigation, alkalization occurs. The above discussion indicates that soil salinization in this region is closely related to groundwater quality.
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Nachshon, U. Cropland soil salinization and associated hydrology: Trends, processes and examples. Water 2018, 10, 1030. [Google Scholar]
- Shokrikuehni, S.M.S.; Norouzi Rad, M.; Webb, C.; Shokri, N. Impact of type of salt and ambient conditions on saline water evaporation from porous media. Adv. Water Resour. 2017, 105, 154–161. [Google Scholar] [CrossRef]
- Mau, Y.; Porporato, A. A dynamical system approach to soil salinity and sodicity. Adv. Water Resour. 2015, 83, 68–76. [Google Scholar] [CrossRef]
- Douaoui, A.E.K.; Nicolas, H.; Walter, C. Detecting salinity hazards within a semiarid context by means of combining soil and remote-sensing data. Geoderma 2017, 134, 217–230. [Google Scholar] [CrossRef]
- Geris, J.; Tetzlaff, D.; Mcdonnell, J.; Soulsby, C. The relative role of soil type and tree cover on water storage and transmission in northern headwater catchments. Hydrol. Process. 2015, 29, 1844–1860. [Google Scholar] [CrossRef]
- Song, Y.G.; Guo, Z.; Lu, Y.; Yan, D.; Liao, Z.; Liu, H.; Cui, Y. Pixel-level spatiotemporal analyses of vegetation fractional coverage variation and its influential factors in a desert steppe: A case study in Inner Mongolia, China. Water 2017, 9, 478. [Google Scholar] [CrossRef]
- Chen, Y.; Li, W.; Xu, C.; Ye, Z.; Chen, Y. Desert riparian vegetation and groundwater in the lower reaches of the tarim river basin. Environ. Earth Sci. 2015, 73, 547–558. [Google Scholar] [CrossRef]
- Gu, F.X.; Zhang, Y.D.; Chu, Y.; Shi, Q.D.; Pan, X.L. Primary analysis on groundwater, soil moisture and salinity in fukang oasis of southern junggar basin. Chin. Geogr. Sci. 2002, 12, 333–338. [Google Scholar] [CrossRef]
- Qian, J.; Wang, L.; Ma, L.; Lu, Y.H.; Zhao, W.; Zhang, Y. Multivariate statistical analysis of water chemistry in evaluating groundwater geochemical evolution and aquifer connectivity near a large coal mine, Anhui, China. Environ. Earth Sci. 2016, 75, 747. [Google Scholar] [CrossRef]
- Golshan, M.; Colombani, N.; Mastrocicco, M. Assessing aquifer salinization with multiple techniques along the southern caspian sea shore (Iran). Water 2018, 10, 348. [Google Scholar] [CrossRef]
- Rietz, D.N.; Haynes, R.J. Effects of irrigation-induced salinity and sodicity on soil microbial activity. Soil Biol. Biochem. 2003, 35, 845–854. [Google Scholar] [CrossRef]
- Xu, X.; Huang, G.; Sun, C.; Pereira, L.S.; Ramos, T.B.; Huang, Q.; Hao, Y. Assessing the effects of water table depth on water use, soil salinity and wheat yield: Searching for a target depth for irrigated areas in the upper yellow river basin. Agric. Water Manag. 2013, 125, 46–60. [Google Scholar] [CrossRef]
- Wang, Y.; Jiao, J.J. Origin of groundwater salinity and hydrogeochemical processes in the confined quaternary aquifer of the pearl river delta, China. J. Hydrol. 2012, 438–439, 112–124. [Google Scholar] [CrossRef]
- Bing, L.; Zhao, W.; Chang, X.; Li, S.; Zhang, Z.; Du, M. Water requirements and stability of oasis ecosystem in arid region, China. Environ. Earth Sci. 2010, 59, 1235. [Google Scholar]
- Zhou, H.; Zhao, W.; Zhang, G. Varying water utilization of haloxylon ammodendron plantations in a desert-oasis ecotone. Hydrol. Process. 2016, 31, 825–835. [Google Scholar] [CrossRef]
- Sun, Z.; Ma, R.; Wang, Y.; Hu, Y.; Sun, L. Hydrogeological and hydrogeochemical control of groundwater salinity in an arid inland basin: Dunhuang basin, northwestern China. Hydrol. Process. 2016, 30, 1884–1902. [Google Scholar] [CrossRef]
- Eziz, M.; Yimit, H.; Mamat, Z.; Ning, X.U. The response of soil salinization to characters of groundwater in ili river basin. J. China Hydrol. 2012, 32, 14–20. [Google Scholar]
- Sai, J.; Lu, Y.; Wang, Z.; He, M. Characteristics of soil salinization in yaoba oasis of inner mongolia autonomous region. Bull. Soil Water Conserv. 2017, 5, 152–156. [Google Scholar]
- Bin, L.I.; Shi, H.B.; Yan, J.W.; Zhen, L.I.; Zhang, J.G.; Zhou, J. Relation between groundwater depth and soil water and salt after water saving reform in salinization irrigation district. J. Soil Water Conserv. 2014, 28, 167–172. [Google Scholar]
- Deng, B.S.; Halik, W.; Dang, J.H.; Zhang, Y.P.; Xuan, J.W. Coupled analysis of spatio-temporal variability of groundwater depth and soil salinity in keriya oasis. Arid. Land Geogr. 2015, 38, 599–607. [Google Scholar]
- Cao, J.R.; Xu, X.; Yu, H.; Liu, Y.J. Analysis for driving forces and ecological risk assessment of soil salinization in the yellow river delta. Adv. Mar. Sci. 2014, 32, 508–516. [Google Scholar]
- Wu, L.; Skaggs, T.H.; Shouse, P.J.; Ayars, J.E. State space analysis of soil water and salinity regimes in a loam soil underlain by shallow groundwater. Soil Sci. Soc. Am. J. 2001, 65, 1065–1074. [Google Scholar] [CrossRef]
- Liu, S.F.; Fang, Z.G.; Lin, Y. Study on a new definition of degree of grey incidence. J. Grey Syst. 2006, 9, 115–122. [Google Scholar]
- Fan, X.; Pedroli, B.; Liu, G.; Liu, Q.; Liu, H.; Shu, L. Soil salinity development in the yellow river delta in relation to groundwater dynamics. Land Degrad. Dev. 2012, 23, 175–189. [Google Scholar] [CrossRef]
- Qureshi, A.S.; Ahmad, W.; Ahmad, A.F.A. Optimum groundwater table depth and irrigation schedules for controlling soil salinity in central Iraq. Irrig. Drain. 2013, 62, 414–424. [Google Scholar] [CrossRef]
- Hui, B.; He, P.; Zhang, Y. Cyclic freeze–thaw as a mechanism for water and salt migration in soil. Environ. Earth Sci. 2015, 74, 675–681. [Google Scholar]
- Xia, J.; Zhang, S.; Zhao, X.; Liu, J.; Chen, Y. Effects of different groundwater depths on the distribution characteristics of soil- tamarix water contents and salinity under saline mineralization conditions. Catena 2016, 142, 166–176. [Google Scholar] [CrossRef]
- Abliz, A.; Tiyip, T.; Ghulam, A.; Halik, Ü.; Ding, J.L.; Sawut, M.; Zhang, F.; Nurmemet, I.; Abliz, A. Effects of shallow groundwater table and salinity on soil salt dynamics in the keriya oasis, northwestern China. Environ. Earth Sci. 2016, 75, 260. [Google Scholar] [CrossRef]
- Haj-Amor, Z.; Tóth, T.; Ibrahimi, M.K.; Bouri, S. Effects of excessive irrigation of date palm on soil salinization, shallow groundwater properties, and water use in a saharan oasis. Environ. Earth Sci. 2017, 76, 590. [Google Scholar] [CrossRef]
- Wichelns, D.; Qadir, M. Achieving sustainable irrigation requires effective management of salts, soil salinity, and shallow groundwater. Agric. Water Manag. 2015, 157, 31–38. [Google Scholar] [CrossRef]
- Xu, J.; Li, W.; Zhang, L.; Hong, Y.; Bi, X.; Yang, Y. Statistical analysis of groundwater chemistry of the tarim river lower reaches, northwest China. Environ. Earth Sci. 2012, 65, 1807–1820. [Google Scholar] [CrossRef]
- Libutti, A.; Cammerino, A.R.B.; Monteleone, M. Risk assessment of soil salinization due to tomato cultivation in mediterranean climate conditions. Water 2018, 10, 1503. [Google Scholar] [CrossRef]
- Zhenzhen, L.; Yang, J.; Liu, G.; Jinbiao, L.I.; Liu, H.; Bing, L.I. Relationship between soil salinization and groundwater characteristics in the yellow river delta. Acta Pedol. Sin. 2017, 54, 1377–1385. [Google Scholar]
- Seeboonruang, U. Relationship between groundwater properties and soil salinity at the;lower nam kam river basin in Thailand. Environ. Earth Sci. 2013, 69, 1803–1812. [Google Scholar] [CrossRef]
- Li, Y.; Lu, Y.D.; Li, H.J.; Wang, J.K.; Jiang, L. Groundwater dynamic characteristics and the influence on vegetation variation in the yaoba oasis. Ground Water 2012, 4, 030. [Google Scholar]
- Zheng, C.; Lu, Y.; Guo, X.; Li, H.; Sai, J.; Liu, X. Application of hydrus-1d model for research on irrigation infiltration characteristics in arid oasis of northwest China. Environ. Earth Sci. 2017, 76, 785. [Google Scholar] [CrossRef]
- Li, X.G.; Xia, W.; Lu, Y.D. Optimising the allocation of groundwater carrying capacity in a data-scarce region. Water SA 2010, 36, 451–460. [Google Scholar] [CrossRef]
- Yu, P.; Xu, H.; Liu, S.; Mu, Q.; Zhang, Q.; An, H.; Fu, J. Spatial distribution pattern changes of oasis soil types in manasi river basin, arid northwestern China. Catena 2011, 87, 253–259. [Google Scholar] [CrossRef]
- Wen, X.; Diao, M.; Wang, D.; Meng, G. Hydrochemical characteristics and salinization processes of groundwater in the shallow aquifer of eastern laizhou bay, China. Hydrological Processes 2012, 26, 2322–2332. [Google Scholar] [CrossRef]
- Visconti, F.; de Paz, J.M. Prediction of the soil saturated paste extract salinity from extractable ions, cation exchange capacity, and anion exclusion. Soil Res. 2012, 50, 536. [Google Scholar] [CrossRef]
- Noguchi, S.; Tsuboyama, Y.; Sidle, R.C.; Hosoda, I. Subsurface runoff characteristics from a forest hillslope soil profile including macropores, hitachi ohta, Japan. Hydrol. Process. 2010, 15, 2131–2149. [Google Scholar] [CrossRef]
- Deng, L.J.; Liu, L. Analysis on groundwater chemical composition based on gray relation. Ground Water 2009, 2, 17–19. [Google Scholar]
- Chao-Ming, L.I. Analysis relationship between every meteorological factor and evaporation with gray relation in shajingzi irrigation region. Ground Water 2009, 32, 98–100. [Google Scholar]
- Wei, G. Grey relational analysis model for dynamic hybrid multiple attribute decision making. Knowl.-Based Syst. 2011, 24, 672–679. [Google Scholar] [CrossRef]
- Foufoula-Georgiou, E.; Takbiri, Z.; Czuba, J.A.; Schwenk, J. The change of nature and the nature of change in agricultural landscapes: Hydrologic regime shifts modulate ecological transitions. Water Resour. Res. 2015, 51, 6649–6671. [Google Scholar] [CrossRef]


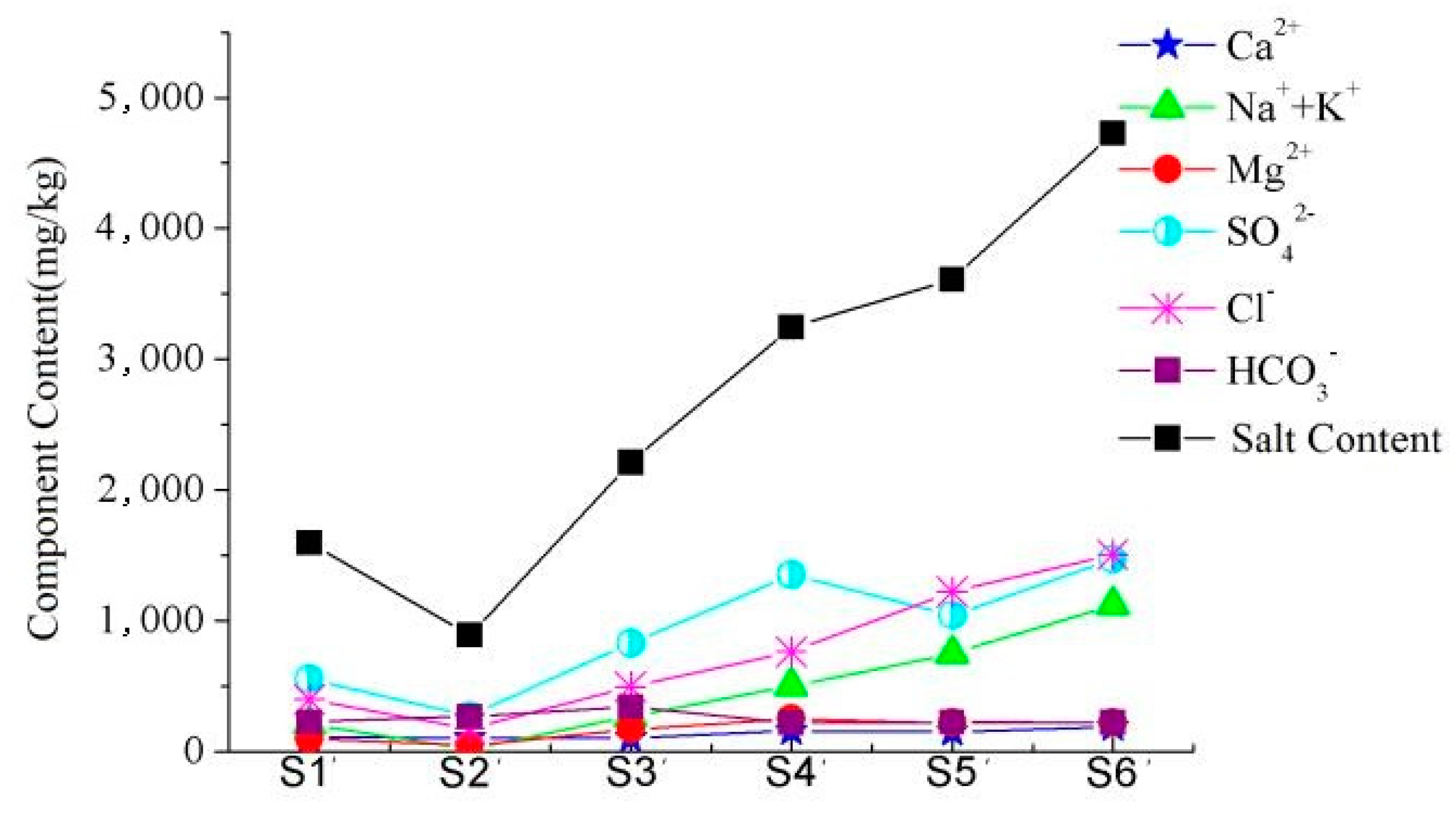
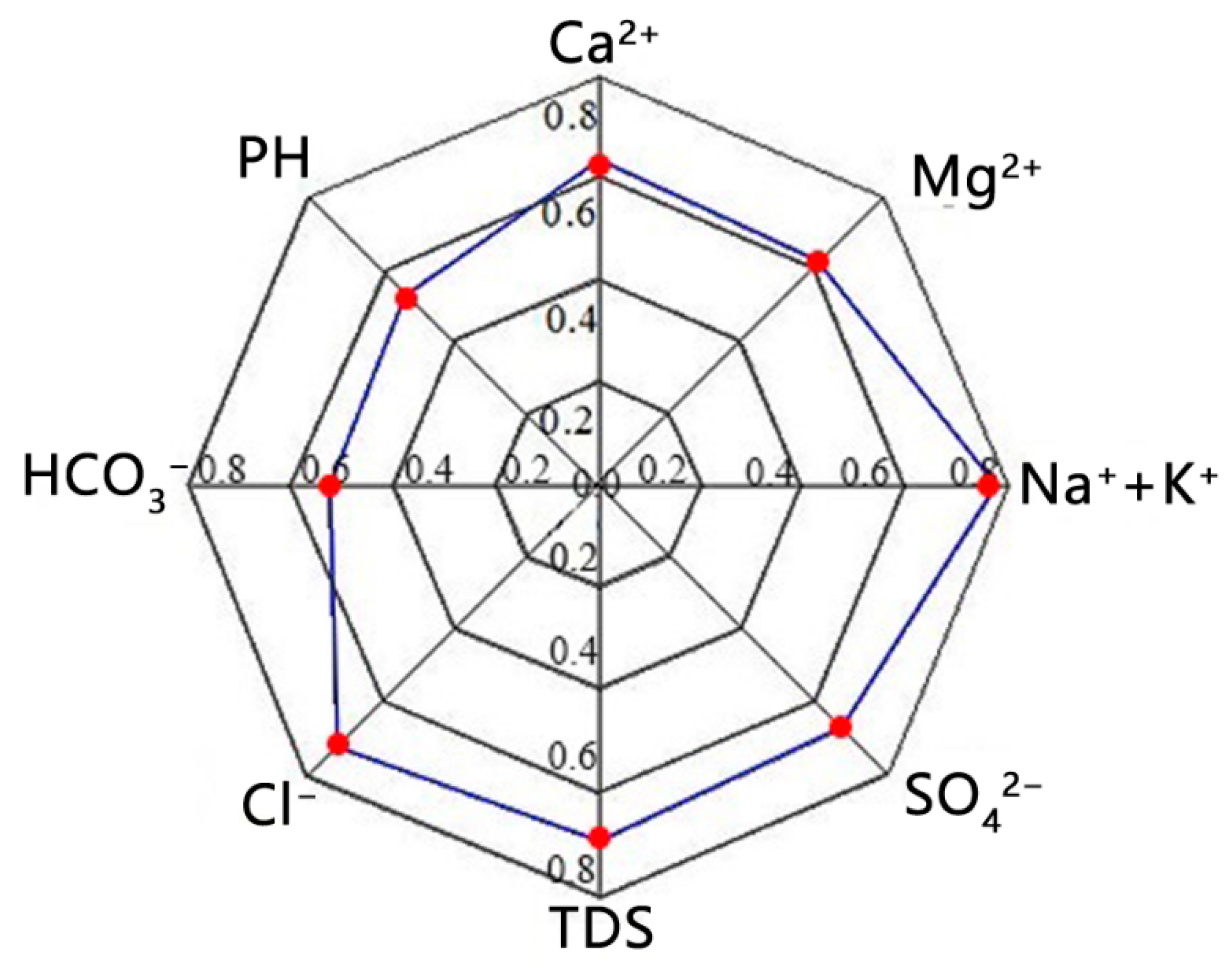

| Time | Statistics | EC (mS/cm) | pH | Content of Main Ions (mg/L) | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| K+ + Na+ | Ca2+ | Mg2+ | Cl− | SO42− | HCO3− | TDS | ||||
| Jun. | Min | 0.74 | 6.81 | 92.50 | 25.10 | 39.50 | 70.90 | 138.10 | 158.70 | 581.40 |
| Max | 5.79 | 7.99 | 1740.10 | 217.90 | 302.40 | 1241.00 | 2443.50 | 158.70 | 5845.60 | |
| Mean | 2.14 | 7.71 | 326.59 | 94.27 | 125.26 | 385.84 | 623.19 | 257.31 | 1683.81 | |
| SD | 1.03 | 0.17 | 239.39 | 42.22 | 59.75 | 251.28 | 389.20 | 55.23 | 939.03 | |
| CV (%) | 48.16 | 2.18 | 73.30 | 44.78 | 47.70 | 65.13 | 62.45 | 21.46 | 55.77 | |
| Nov. | Min | 0.44 | 7.09 | 90.10 | 35.10 | 42.50 | 53.20 | 192.10 | 128.10 | 580.80 |
| Max | 4.34 | 8.12 | 942.50 | 240.50 | 282.60 | 1134.60 | 1464.90 | 488.20 | 3860.90 | |
| Mean | 2.09 | 7.72 | 392.76 | 118.01 | 137.76 | 452.20 | 766.74 | 254.46 | 1994.70 | |
| SD | 0.22 | 1.27 | 206.01 | 60.27 | 71.14 | 309.06 | 368.08 | 68.66 | 955.71 | |
| CV (%) | 60.61 | 2.90 | 52.45 | 51.07 | 51.64 | 68.35 | 48.01 | 26.98 | 47.91 | |
| Class | Non-Salinization | Weak Salinization | Medium Salinization | Strong Salinization |
|---|---|---|---|---|
| Salt content (g/kg) | <1 | 1–2 | 2–4 | >4 |
| Number of samples | 12 | 24 | 5 | 3 |
| Percent (%) | 27.3 | 54.5 | 11.4 | 6.8 |
| Depth/cm | pH | Total Salt Content/% | CO32− | HCO3− | Cl− | SO42− | Ca2+ | Mg2+ | K+ | Na+ | |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 0–10 | Mean | 8.39 | 0.9 | 0.02 | 0.34 | 7.69 | 3.56 | 3.79 | 1.32 | 0.10 | 10.86 |
| Max | 8.6 | 2.23 | 0.06 | 0.4 | 18.75 | 16.42 | 12.95 | 4.52 | 0.25 | 34.41 | |
| Min | 7.9 | 0.03 | 0 | 0.26 | 0.12 | 0.05 | 0.1 | 0.14 | 0.01 | 0.13 | |
| Std. | 0.29 | 1.13 | 0.03 | 0.06 | 10.03 | 7.19 | 6.14 | 2.14 | 0.1 | 15.33 | |
| CV | 0.03 | 1.27 | 1.05 | 0.18 | 1.3 | 2.02 | 1.62 | 1.62 | 1.04 | 1.41 | |
| 10–30 | Mean | 8.52 | 0.71 | 0.08 | 0.38 | 4.94 | 5.26 | 2.6 | 0.46 | 0.08 | 8.18 |
| Max | 9.25 | 2.36 | 0.29 | 0.8 | 12.3 | 22.19 | 11.9 | 1.1 | 0.31 | 24.35 | |
| Min | 8.1 | 0.02 | 0 | 0.24 | 0.18 | 0 | 0.1 | 0.07 | 0 | 0.16 | |
| Std. | 0.43 | 1 | 0.12 | 0.24 | 6.12 | 9.56 | 5.2 | 0.48 | 0.15 | 11 | |
| CV | 0.05 | 1.42 | 1.39 | 0.61 | 1.24 | 82 | 2 | 1.06 | 1.92 | 1.34 | |
| 30–60 | Mean | 8.6 | 0.63 | 0.18 | 0.41 | 5.84 | 3.35 | 2.97 | 0.6 | 0.05 | 7.26 |
| Max | 9.15 | 1.48 | 0.84 | 0.77 | 14.17 | 10.3 | 11.11 | 1.78 | 0.12 | 21 | |
| Min | 8.4 | 0.04 | 0 | 0.22 | 0.3 | 0 | 0.14 | 0.1 | 0 | 0.33 | |
| Std. | 0.31 | 0.73 | 0.37 | 0.22 | 6.84 | 4.6 | 5.43 | 0.8 | 0.O5 | 9.11 | |
| CV | 0.04 | 1.17 | 2 | 0.53 | 1.17 | 1.37 | 1.83 | 33 | 1.21 | 1.25 | |
| 60–90 | Mean | 8.62 | 0.89 | 0.l9 | 0.5 | 6.88 | 4.48 | 1.08 | 0.44 | 0.09 | l2.28 |
| Max | 9.4 | 2.66 | 0.86 | 0.79 | 19.66 | 20.24 | 3.94 | 0.5 | 0.31 | 37.4 | |
| Min | 8.35 | 0.06 | 0 | 0.3 | 0.31 | 0 | 0.09 | 0.35 | 0 | 0.43 | |
| Std. | 0.44 | l.10 | 0.38 | 0.19 | 8.57 | 8.82 | 1.91 | 0.07 | 0.15 | 16.09 | |
| CV | 0.05 | 1.25 | 1.93 | 0.37 | 1.25 | 1.97 | 1.76 | 0.l5 | 1.77 | 1.31 | |
| 90–120 | Mean | 8.86 | 0.84 | 0.25 | 0.41 | 7.03 | 5.6 | 2.89 | 0.28 | 0.02 | 9.93 |
| Max | 9.65 | 2.95 | 1.06 | 0.6 | 24.49 | 21.46 | 12.4 | 0.5 | 0.03 | 33.33 | |
| Min | 8.35 | 0.05 | 0 | 0.28 | 0.l5 | 0 | 0.05 | 0.09 | 0.01 | 0.27 | |
| Std. | 0.5 | 1.21 | 0.46 | 0.13 | 10.04 | 8.96 | 5.36 | 0.2 | 0.01 | 13.64 | |
| CV | 0.06 | l.44 | 1.81 | 0.32 | 1.43 | I.60 | 1.86 | 0.72 | 0.55 | 1.37 | |
| 120–140 | Mean | 8.67 | 0.83 | 0.06 | 0.5 | 6.57 | 5.1 | 1.75 | 0.58 | 0.12 | 12.02 |
| Max | 8.95 | 2.61 | 0.29 | 0.8 | 19.78 | 17.91 | 4.9 | 1.17 | 0.41 | 39.14 | |
| Min | 8.35 | 0.08 | 0 | 0.28 | 0.28 | 0.36 | 0.11 | 0.25 | 0.01 | 0.6 | |
| Std. | 0.25 | 1.05 | 0.13 | 0.27 | 8.02 | 7.31 | 2.21 | 0.41 | 0.19 | 16.14 | |
| CV | 0.03 | 1.26 | 1.98 | 0.55 | 1.22 | 1.43 | 1.26 | 0.71 | 1.57 | 1.34 | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cui, G.; Lu, Y.; Zheng, C.; Liu, Z.; Sai, J. Relationship between Soil Salinization and Groundwater Hydration in Yaoba Oasis, Northwest China. Water 2019, 11, 175. https://doi.org/10.3390/w11010175
Cui G, Lu Y, Zheng C, Liu Z, Sai J. Relationship between Soil Salinization and Groundwater Hydration in Yaoba Oasis, Northwest China. Water. 2019; 11(1):175. https://doi.org/10.3390/w11010175
Chicago/Turabian StyleCui, Guoqing, Yudong Lu, Ce Zheng, Zhiheng Liu, and Jiamei Sai. 2019. "Relationship between Soil Salinization and Groundwater Hydration in Yaoba Oasis, Northwest China" Water 11, no. 1: 175. https://doi.org/10.3390/w11010175
APA StyleCui, G., Lu, Y., Zheng, C., Liu, Z., & Sai, J. (2019). Relationship between Soil Salinization and Groundwater Hydration in Yaoba Oasis, Northwest China. Water, 11(1), 175. https://doi.org/10.3390/w11010175






