The Contribution of Coniferous Canopy to the Molecular Diversity of Dissolved Organic Matter in Rainfall
Abstract
1. Introduction
2. Materials and Methods
2.1. Site Description
2.2. Water Sampling
2.3. Chemical and Data Analyses
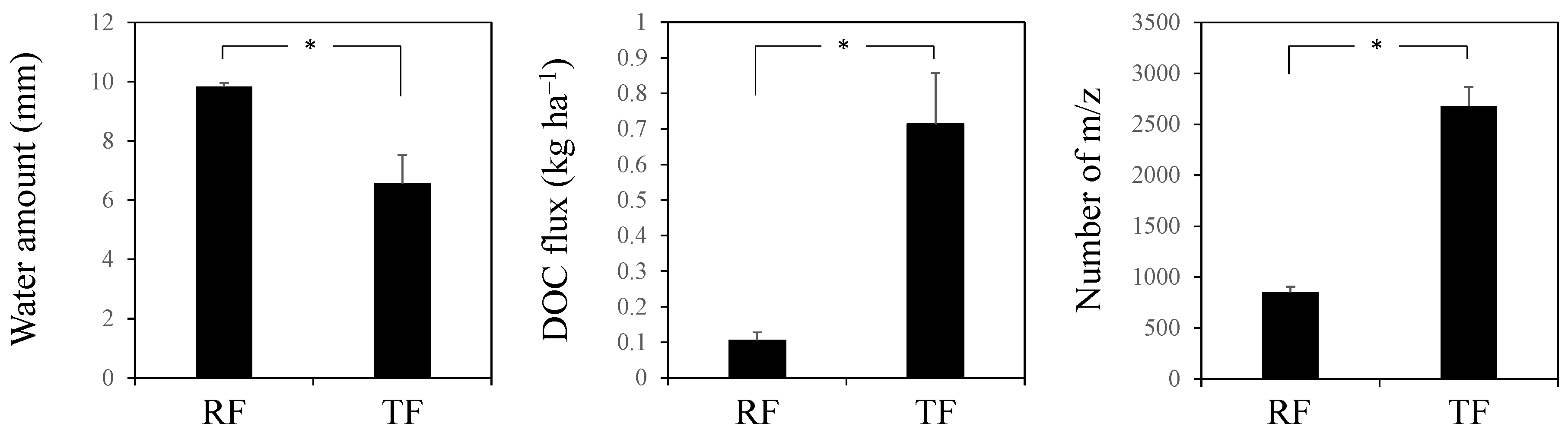
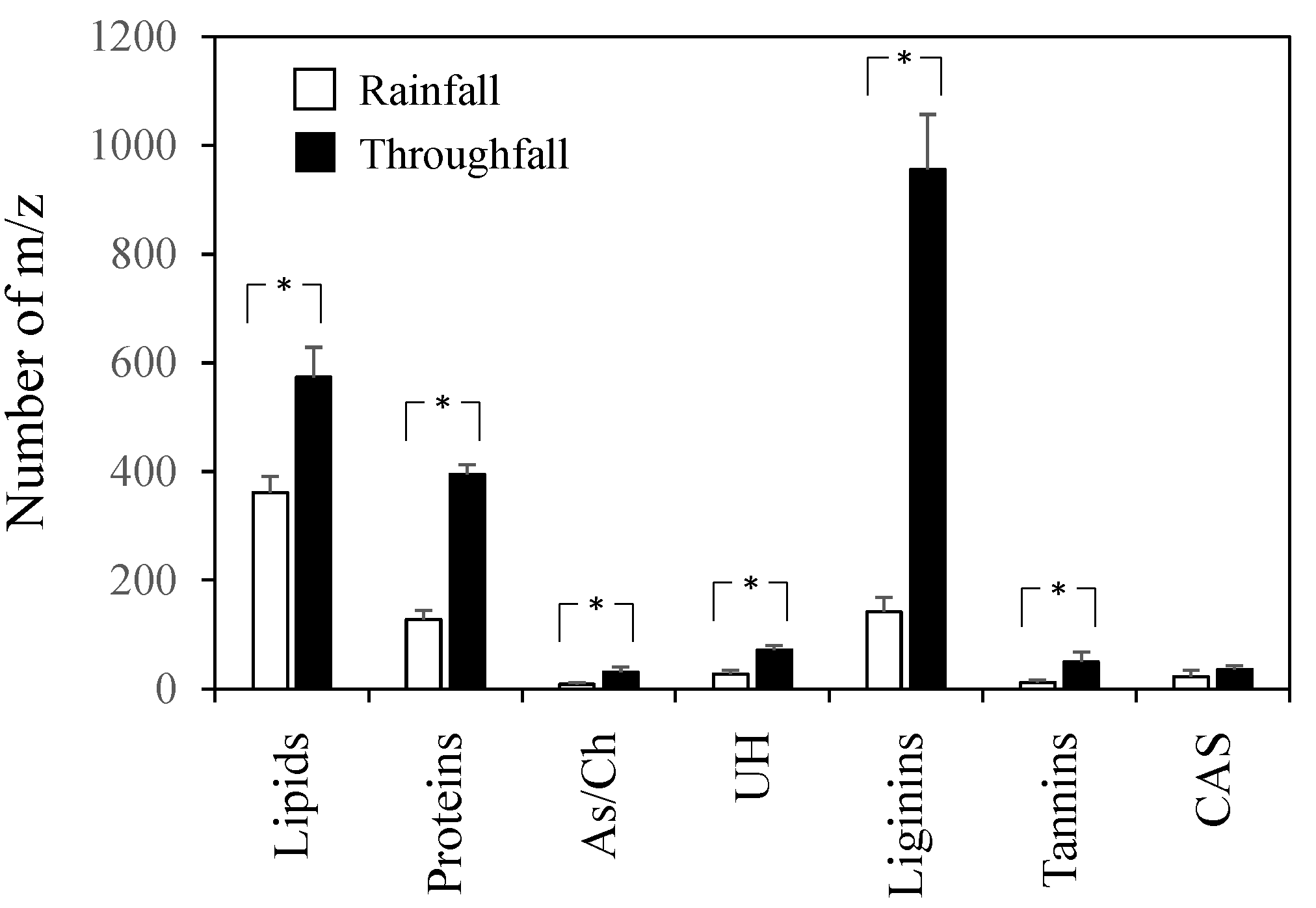
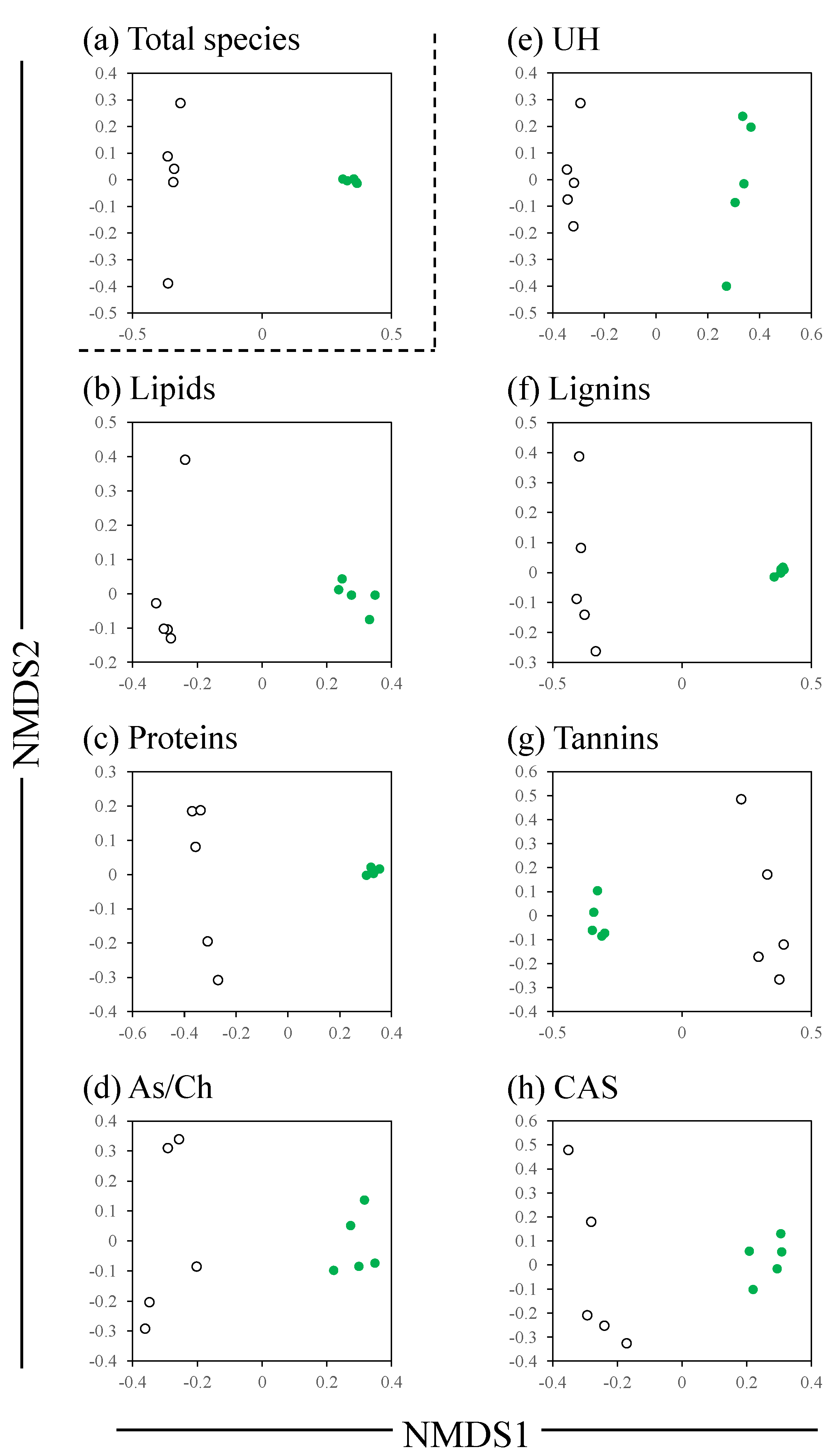
3. Results
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Forestry Agency of Japan. Annual Report on Trends in Forests and Forestry-Fiscal Year 2014; Zenrinkyo: Tokyo, Japan, 2014. [Google Scholar]
- Oohara, H. The necessity for the research on the relationship between transition of forest floor vegetation accompanying thinning and function of soil and water conservation in artificial sugi (Cryptomeria japonica) forest. Bull. For. For. Prod. Res. Inst. 2007, 6, 127–134. [Google Scholar]
- FAO (Food and Agriculture Organization of the United Nations). Global Forest Resources Assessment. Main Report; FAO Forest Paper 163; FAO: Rome, Italy, 2010; ISBN 978-92-5-106654-6. [Google Scholar]
- Oliver, B.G.; Thurman, E.M.; Malcolm, R.L. The contribution of humic substances to the acidity of colored natural waters. Geochim. Cosmochim. Acta 1983, 47, 2031–2035. [Google Scholar] [CrossRef]
- Green, S.A.; Blough, N.V. Optical absorption and fluorescence properties of chromophoric dissolved organic matter in natural waters. Limnol. Oceanogr. 1994, 39, 1903–1916. [Google Scholar] [CrossRef]
- Koenings, J.P.; Hooper, F.F. The influence of colloidal organic matter on iron and iron-phosphorus cycling in an acid bog lake1. Limnol. Oceanogr. 1976, 21, 684–696. [Google Scholar] [CrossRef]
- Jones, R.I.; Salonen, K.; Haan, H. Phosphorus transformations in the epilimnion of humic lakes: Abiotic interactions between dissolved humic materials and phosphate. Freshw. Biol. 1988, 19, 357–369. [Google Scholar] [CrossRef]
- Mierle, G.; Ingram, R. The role of humic substances in the mobilization of mercury from watersheds. Water Air Soil Pollut. 1991, 56, 349–357. [Google Scholar] [CrossRef]
- Winch, S.; Ridal, J.; Lean, D. Increased Metal Bioavailability Following Alteration of Freshwater Dissolved Organic Carbon by Ultraviolet B Radiation Exposure. Environ. Toxicol. 2002, 17, 267–274. [Google Scholar] [CrossRef]
- Qualls, R.G.; Haines, B.L. Biodegradability of Dissolved Organic Matter in Forest Throughfall, Soil Solution, and Stream Water. Soil Sci. Soc. Am. J. 1992, 56, 578–586. [Google Scholar] [CrossRef]
- Ward, N.D.; Keil, R.G.; Medeiros, P.M.; Brito, D.C.; Cunha, A.C.; Dittmar, T.; Yager, P.L.; Krusche, A.V.; Richey, J.E. Degradation of terrestrially derived macromolecules in the Amazon River. Nat. Geosci. 2013, 6, 530–533. [Google Scholar] [CrossRef]
- Likens, G.E.; Bormann, F.H. Biogeochemistry of a Forested Ecosystem; Springer: New York, NY, USA, 1995; ISBN 978-0-387-94351-0. [Google Scholar]
- Qualls, R.G.; Haines, B.L.; Swank, W.T. Fluxes of Dissolved Organic Nutrients and Humic Substances in a Deciduous Forest. Ecology 1991, 72, 254. [Google Scholar] [CrossRef]
- Schrumpf, M.; Zech, W.; Lehmann, J.; Lyaruu, H.V.C. TOC, TON, TOS and TOP in Rainfall, Throughfall, Litter Percolate and Soil Solution of a Montane Rainforest Succession at Mt. Kilimanjaro, Tanzania. Biogeochemistry 2006, 78, 361–387. [Google Scholar] [CrossRef]
- Currie, W.S.; Aber, J.D.; Mcdowell, W.H.; Boone, R.D.; Magill, A.H. Vertical transport of dissolved organic C and N under long-term N amendments in pine and hardwood forests. Biogeochemistry 1996, 35, 471–505. [Google Scholar] [CrossRef]
- David, M.B.; Driscoll, C.T. Aluminum speciation and equilibria in soil solutions of a Haplorthod in the Adirondack Mountains (New York, U.S.A.). Geoderma 1984, 33, 297–318. [Google Scholar] [CrossRef]
- Edmonds, R.L.; Thomas, T.B.; Blew, R.D. BIOGEOCIIEMISTRY OF AN OLD-GROWTH FORESTED WATERSHED, OLYMPIC NATIONAL PARK, WASHINGTON. J. Am. Water Resour. Assoc. 1995, 31, 409–419. [Google Scholar] [CrossRef]
- Matsumoto, Y.; Nishida, K.; Atsuta, Y.; Sakamoto, Y. Estimation of annual DOC flux of throughfall and stemflow in a temperate forest. Environ. Sci. 2004, 17, 379–388. [Google Scholar] [CrossRef]
- Mellec, A.; Meesenburg, H.; Michalzik, B. The importance of canopy-derived dissolved and particulate organic matter (DOM and POM)—Comparing throughfall solution from broadleaved and coniferous forests. Ann. For. Sci. 2010, 67, 411. [Google Scholar] [CrossRef]
- Likens, G.E.; Edgerton, E.S.; Galloway, J.N. The composition and deposition of organic carbon in precipitation. Tellus B 1983, 35B, 16–24. [Google Scholar] [CrossRef]
- Stubbins, A.; Silva, L.M.; Dittmar, T.; Van Stan, J.T. Molecular and Optical Properties of Tree-Derived Dissolved Organic Matter in Throughfall and Stemflow from Live Oaks and Eastern Red Cedar. Front. Earth Sci. 2017, 5, 1–13. [Google Scholar] [CrossRef]
- Guggenberger, G.; Zech, W. Composition and dynamics of dissolved carbohydrates and lignin-degradation products in two coniferous forests, N.E. Bavaria, Germany. Soil Biol. Biochem. 1994, 26, 19–27. [Google Scholar] [CrossRef]
- McDowell, W.H.; Likens, G.E. Origin, Composition, and Flux of Dissolved Organic Carbon in the Hubbard Brook Valley. Ecol. Monogr. 1988, 58, 177–195. [Google Scholar] [CrossRef]
- Sleighter, R.L.; Hatcher, P.G. The application of electrospray ionization coupled to ultrahigh resolution mass spectrometry for the molecular characterization of natural organic matter. J. Mass Spectrom. 2007, 42, 559–574. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Kramer, R.W.; Hatcher, P.G. Graphical Method for Analysis of Ultrahigh-Resolution Broadband Mass Spectra of Natural Organic Matter, the Van Krevelen Diagram. Anal. Chem. 2003, 75, 5336–5344. [Google Scholar] [CrossRef] [PubMed]
- Ide, J.; Ohashi, M.; Takahashi, K.; Sugiyama, Y.; Piirainen, S.; Kortelainen, P.; Fujitake, N.; Yamase, K.; Ohte, N.; Moritani, M.; et al. Spatial variations in the molecular diversity of dissolved organic matter in water moving through a boreal forest in eastern Finland. Sci. Rep. 2017, 7, 42102. [Google Scholar] [CrossRef] [PubMed]
- Van Krevelen, D.W. Graphical-statistical method for the study of structure and reaction processes of coal. Fuel 1950, 29, 269–284. [Google Scholar]
- Ide, J.; Chiwa, M.; Higashi, N.; Maruno, R.; Mori, Y.; Otsuki, K. Determining storm sampling requirements for improving precision of annual load estimates of nutrients from a small forested watershed. Environ. Monit. Assess. 2012, 184, 4747–4762. [Google Scholar] [CrossRef]
- Ide, J.; Kume, T.; Wakiyama, Y.; Higashi, N.; Chiwa, M.; Otsuki, K. Estimation of annual suspended sediment yield from a Japanese cypress (Chamaecyparis obtusa) plantation considering antecedent rainfalls. For. Ecol. Manag. 2009, 257, 1955–1965. [Google Scholar] [CrossRef]
- Kume, T.; Tsuruta, K.; Komatsu, H.; Shinohara, Y.; Katayama, A.; Ide, J.; Otsuki, K. Differences in sap flux-based stand transpiration between upper and lower slope positions in a Japanese cypress plantation watershed. Ecohydrology 2016, 9, 1105–1116. [Google Scholar] [CrossRef]
- Parker, G.G. Throughfall and Stemflow in the Forest Nutrient Cycle. In Advances in Ecological Research; Academic Press: Cambridge, MA, USA, 1983; Volume 13, pp. 57–133. [Google Scholar]
- Dittmar, T.; Koch, B.; Hertkorn, N.; Kattner, G. A simple and efficient method for the solid-phase extraction of dissolved organic matter (SPE-DOM) from seawater. Limnol. Ocean. Methods 2008, 6, 230–235. [Google Scholar] [CrossRef]
- Reemtsma, T. Determination of molecular formulas of natural organic matter molecules by (ultra-) high-resolution mass spectrometry. J. Chromatogr. A 2009, 1216, 3687–3701. [Google Scholar] [CrossRef]
- Grannas, A.M.; Hockaday, W.C.; Hatcher, P.G.; Thompson, L.G.; Mosley-Thompson, E. New revelations on the nature of organic matter in ice cores. J. Geophys. Res. 2006, 111, D04304. [Google Scholar] [CrossRef]
- R Core Team R: A Language and Environment for Statistical Computing. Available online: https://www.r-project.org/ (accessed on 1 November 2018).
- Haider, K. Problems related to the humification processes in soils of temperate climates. In Soil Biochemistry 7; Stotzky, G., Bollag, J.-M., Eds.; CRC Press: Boca Raton, FL, USA, 1992; pp. 55–94. [Google Scholar]
- Wu, X.; Wu, L.; Liu, Y.; Zhang, P.; Li, Q.; Zhou, J.; Hess, N.J.; Hazen, T.C.; Yang, W.; Chakraborty, R. Microbial Interactions with Dissolved Organic Matter Drive Carbon Dynamics and Community Succession. Front. Microbiol. 2018, 9, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Michalzik, B.; Kalbitz, K.; Park, J.; Solinger, S. Fluxes and concentrations of dissolved organic carbon and nitrogen—A synthesis for temperate forests. Biogeochemistry 2011, 52, 173–205. [Google Scholar] [CrossRef]
- Kalbitz, K.; Solinger, S.; Park, J.-H.; Michalzik, B.; Matzner, E. Controls on the dynamics of dissolved organic matter in soils: A review. Soil Sci. 2000, 165, 277–304. [Google Scholar] [CrossRef]
- O’Donnell, J.A.; Aiken, G.R.; Butler, K.D.; Guillemette, F.; Podgorski, D.C.; Spencer, R.G.M. DOM composition and transformation in boreal forest soils: The effects of temperature and organic-horizon decomposition state. J. Geophys. Res. Biogeosci. 2016, 121, 2727–2744. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ide, J.; Makita, N.; Jeong, S.; Yamase, K.; Ohashi, M. The Contribution of Coniferous Canopy to the Molecular Diversity of Dissolved Organic Matter in Rainfall. Water 2019, 11, 167. https://doi.org/10.3390/w11010167
Ide J, Makita N, Jeong S, Yamase K, Ohashi M. The Contribution of Coniferous Canopy to the Molecular Diversity of Dissolved Organic Matter in Rainfall. Water. 2019; 11(1):167. https://doi.org/10.3390/w11010167
Chicago/Turabian StyleIde, Jun’ichiro, Naoki Makita, Seonghun Jeong, Keitaro Yamase, and Mizue Ohashi. 2019. "The Contribution of Coniferous Canopy to the Molecular Diversity of Dissolved Organic Matter in Rainfall" Water 11, no. 1: 167. https://doi.org/10.3390/w11010167
APA StyleIde, J., Makita, N., Jeong, S., Yamase, K., & Ohashi, M. (2019). The Contribution of Coniferous Canopy to the Molecular Diversity of Dissolved Organic Matter in Rainfall. Water, 11(1), 167. https://doi.org/10.3390/w11010167




