Assessment of Water Quality and Thermal Stress for an Artificial Fish Shelter in an Urban Small Pond during Early Summer
Abstract
1. Introduction
2. Materials and Methods
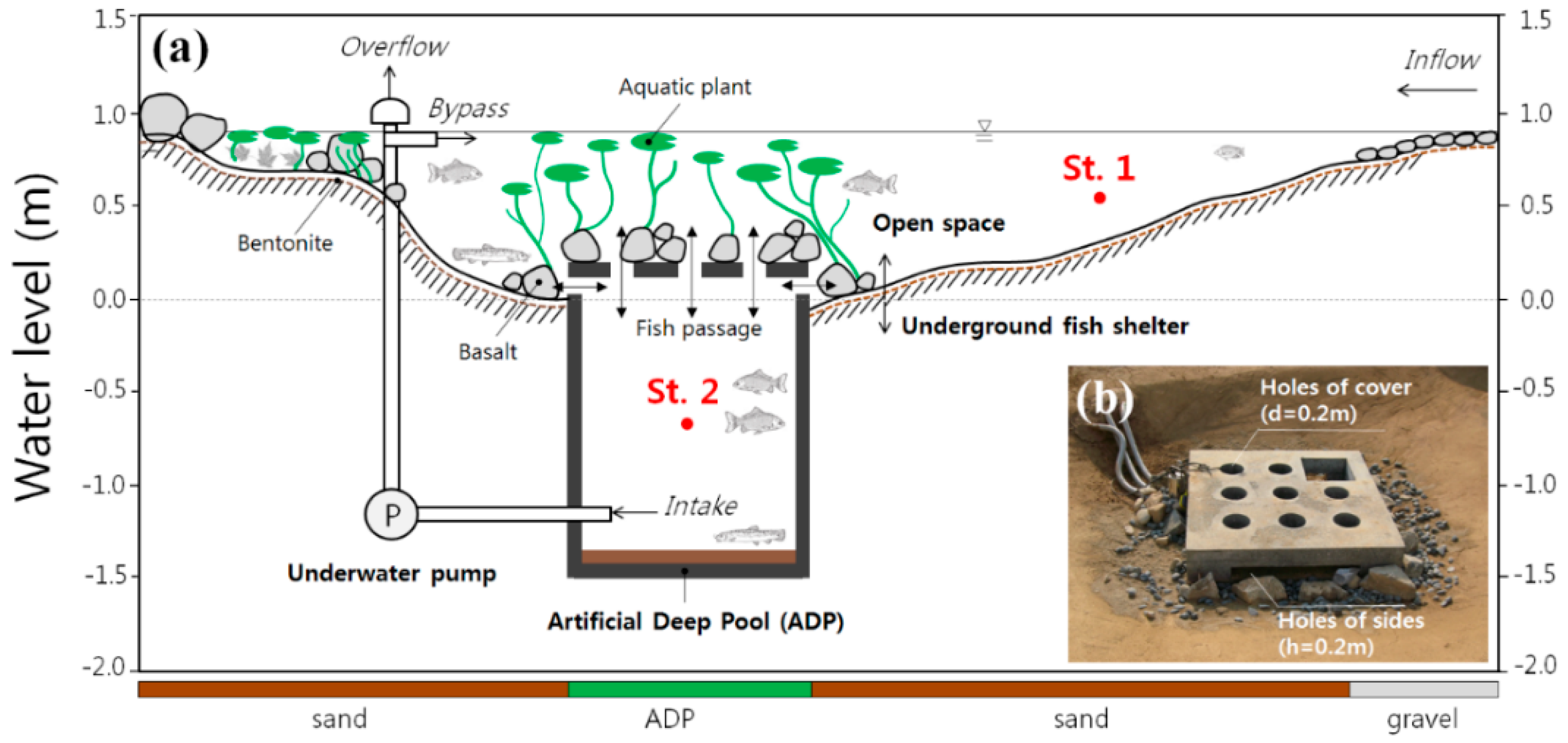
2.1. Study Site
2.2. Water Quality and Fish Monitoring Analysis
2.3. Statistical Analysis
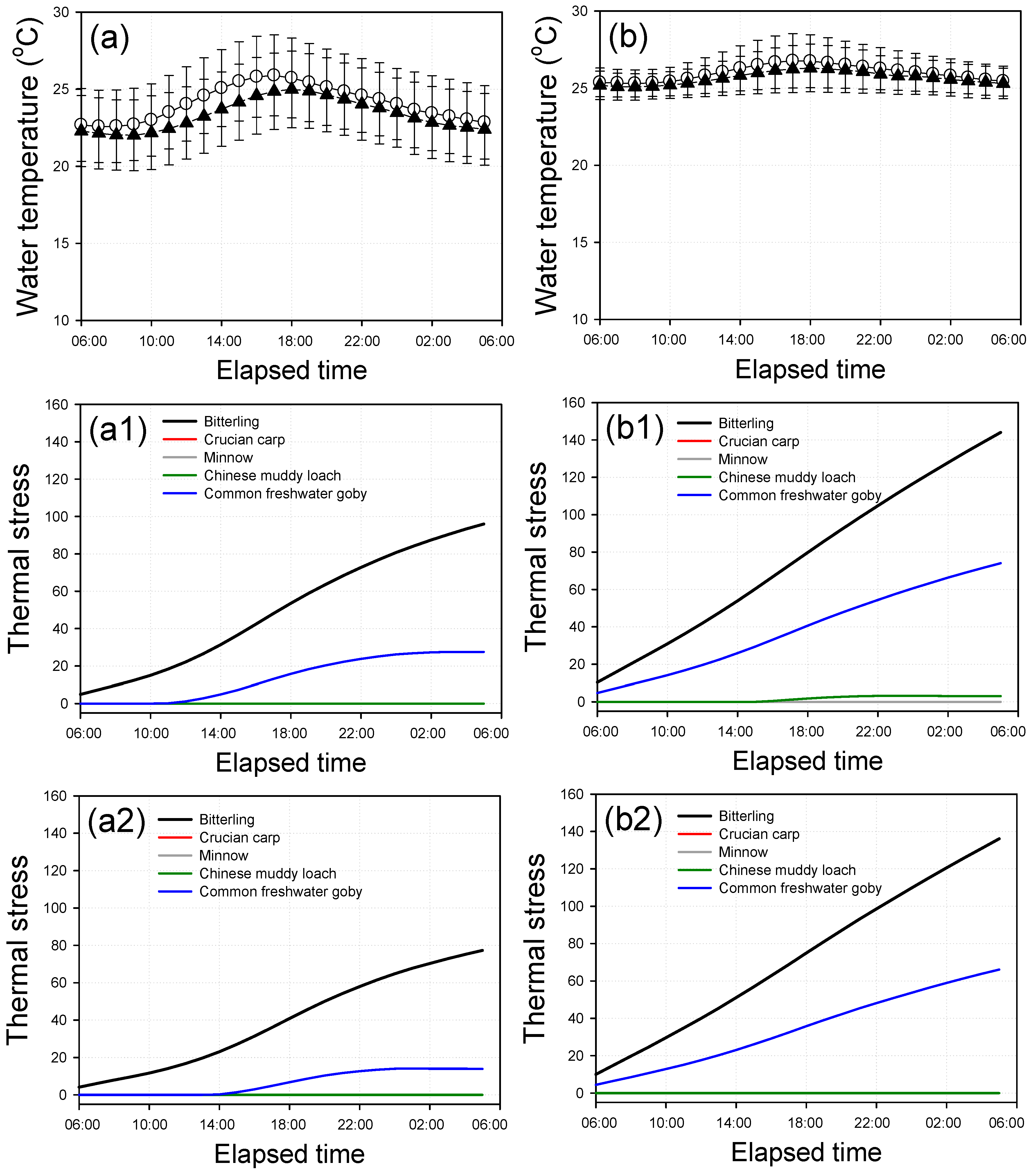
2.4. Thermal Stress Analysis
3. Results and Discussions
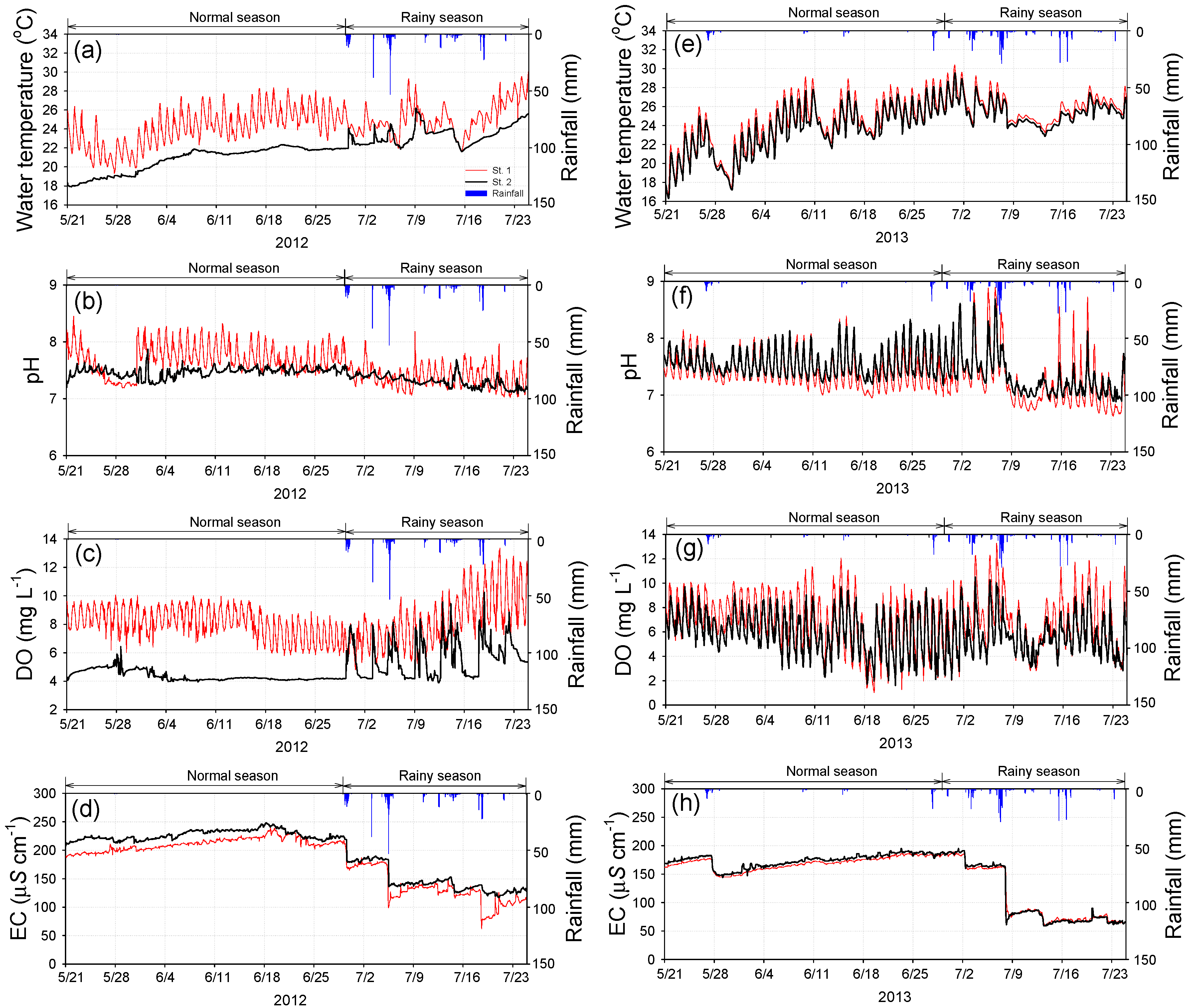
3.1. Water Temperature, pH, DO, and EC
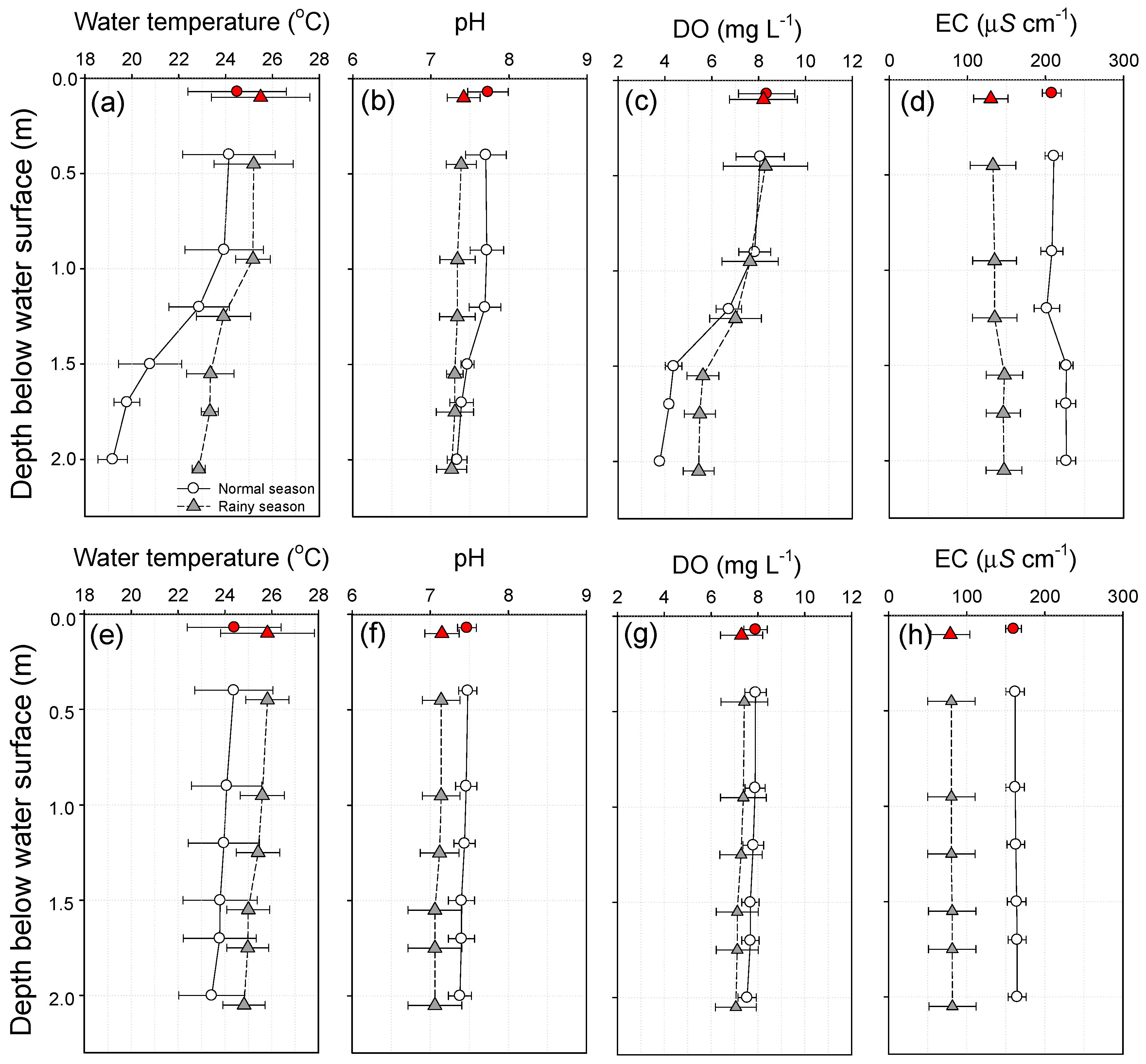
3.2. Physicochemical Water Quality
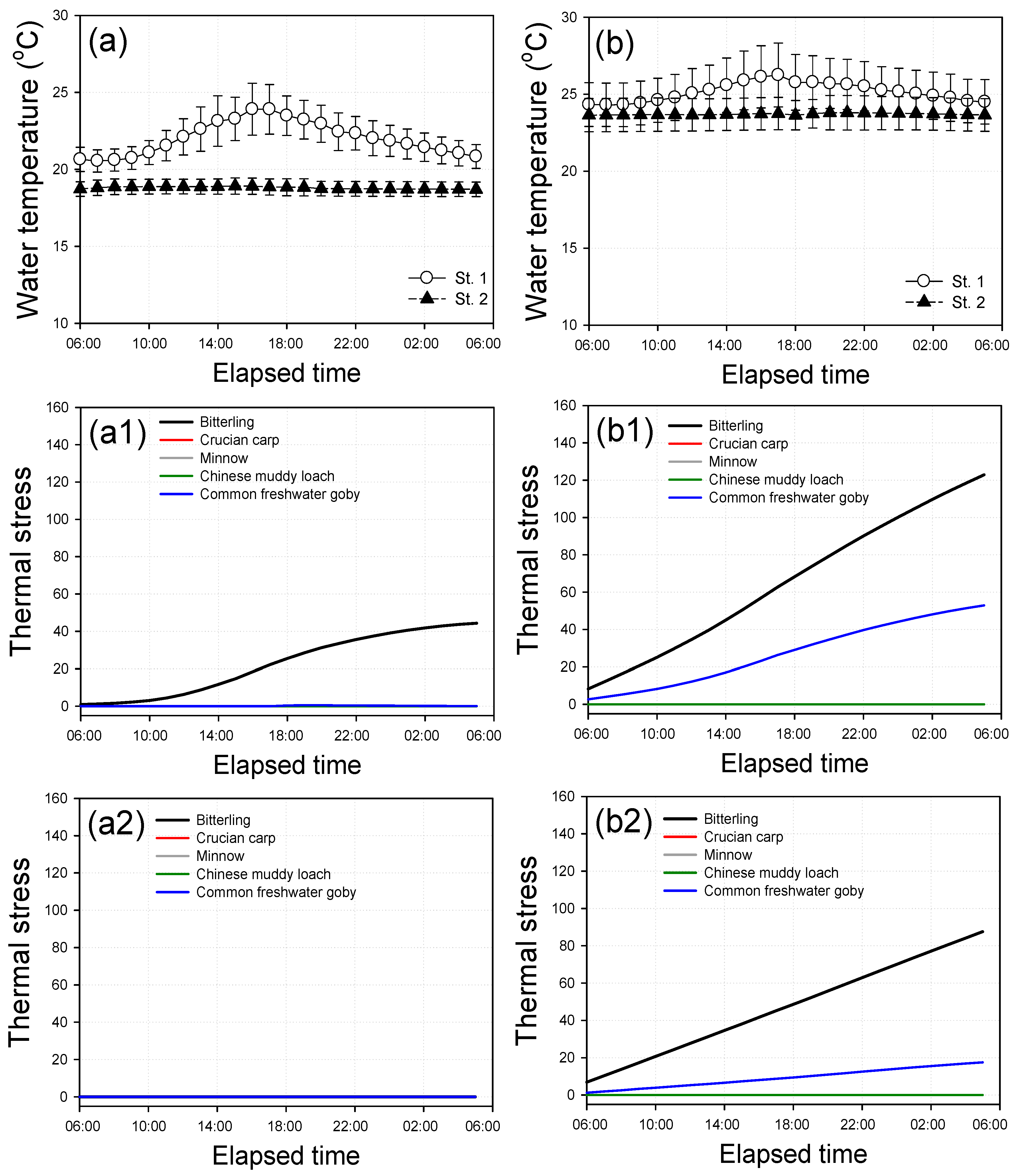
3.3. Thermal Stress of Fish
3.4. Diurnal Variations for Thermal Stress of Fish
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Davis, M.W. Fish stress and mortality can be predicted using reflex impairment. Fish Fish. 2010, 11, 1–11. [Google Scholar] [CrossRef]
- Amisah, S.; Cowx, I.G. Response of the fish populations of the River Don in South Yorkshire to water quality and habitat improvements. Environ. Pollut. 2000, 108, 191–199. [Google Scholar] [CrossRef]
- Mo, W.Y.; Cheng, Z.; Choi, W.M.; Man, Y.B.; Liu, Y.; Wong, M.H. Application of food waste based diets in polyculture of low trophic level fish Effect on fish growth, water quality and plankton density. Mar. Pollut. Bull. 2014, 85, 803–809. [Google Scholar] [CrossRef] [PubMed]
- Hokanson, K.E. Temperature requirement of some percids and adaptations to the seasonal temperature cycle. J. Fish. Res. Board Can. 1977, 34, 1524–1550. [Google Scholar] [CrossRef]
- Nicieza, A.G.; Metcalfe, N.B. Growth compensation in juvenile Atlantic salmon: Responses to depresses temperature and food availability. Ecology 1997, 78, 2385–2400. [Google Scholar] [CrossRef]
- Bridget, S.G.; Fisher, R. Temperature influences swimming speed, growth and larval duration in coral reef fish larval. J. Exp. Mar. Biol. Ecol. 2004, 299, 115–132. [Google Scholar]
- Byström, P.; Andersson, J.; Kiessling, A.; Eriksson, L.O. Size and temperature dependent foraging capacities and metabolism: Consequences for winter starvation mortality in fish. Oikos 2006, 115, 43–52. [Google Scholar] [CrossRef]
- Lefébure, R. Effects of Temperature and Terrestrial Carbon on Fish Growth and Pelagic Food Web Efficiency; Department of Ecology and Environmental Science, Umeå University: Umeå, Sweden, 2012; pp. 1–28. [Google Scholar]
- Nowosad, J.; Targońska, K.; Chwaluczyk, R.; Kaszubowski, R.; Kucharczyk, D. Effect of temperature on the effectiveness of artificial reproduction of dace under laboratory and field conditions. J. Therm. Biol. 2014, 45, 62–68. [Google Scholar] [CrossRef]
- Bevelhimer, M.; Bennett, W. Assessing cumulative thermal stress in fish during chronic intermittent exposure to high temperatures. Environ. Sci. Policy 2000, 3, S211–S216. [Google Scholar] [CrossRef]
- Cheung, W.W.C.; Sarmiento, J.L.; Dunne, J.; Frölicher, T.L.; Lam, V.W.Y.; Palomares, M.L.D.; Watson, R.; Pauly, D. Shrinking of fishes exacerbates impacts of global ocean changes on marine ecosystems. Nat. Clim. Chang. 2013, 3, 254–258. [Google Scholar] [CrossRef]
- Basu, N.; Todgham, A.E.; Ackerman, P.A.; Bibeau, M.R.; Nakano, K.; Schulte, P.M.; Iwama, G.K. Heat shock protein genes and their functional significance in fish. Gene 2002, 295, 173–183. [Google Scholar] [CrossRef]
- Nakano, T.; Kameda, M.; Shoji, Y.; Hayashi, S.; Yamaguchi, T.; Sato, M. Effect of severe environmental thermal stress on redox state in salmon. Redox Biol. 2014, 2, 772–776. [Google Scholar] [CrossRef] [PubMed]
- Fuiman, L.A.; Magurran, A.E. Development of predator defenses in fishes. Rev. Fish Biol. Fish. 1994, 4, 145–183. [Google Scholar] [CrossRef]
- Almany, G.R. Differential effects of habitat complexity, predators and competitors on abundance of juvenile and adult coral reef fishes. Oecologia 2004, 141, 105–113. [Google Scholar] [CrossRef] [PubMed]
- Millidine, K.J.; Armstrong, J.D.; Metcalfe, N.B. Presence of shelter reduces maintenance metabolism of juvenile salmon. Funct. Ecol. 2006, 20, 839–845. [Google Scholar] [CrossRef]
- Finstad, A.G.; Einum, S.; Forseth, T.; Ugedal, O. Shelter availability affects behaviour, size-dependent and mean growth of juvenile Atlantic salmon. Freshw. Biol. 2007, 52, 1710–1718. [Google Scholar] [CrossRef]
- Yamamoto, K.C.; Freitas, C.E.C.; Zuanon, J.; Hurd, L.E. Fish diversity and species composition in small-scale artificial reefs inAmazonian floodplain lakes: Refugia for rare species? Ecol. Eng. 2014, 67, 165–170. [Google Scholar] [CrossRef]
- Ahn, C.H.; Lee, S.; Song, H.M.; Bang, K.W.; Park, J.R.; Joo, J.C. Evaluation of the performance for an artificial deep pool (ADP) as an alternative fish shelter. Ecol. Eng. 2016, 93, 37–45. [Google Scholar] [CrossRef]
- Matthews, K.R.; Berg, N.H.; Azuma, D.L.; Lambert, T.R. Cool water formation and trout habitat use in a deep pool in the Sierra Nevada, California. Trans. Am. Fish. Soc. 1994, 123, 549–564. [Google Scholar] [CrossRef]
- Mercader, M.; Mercière, A.; Saragoni, G.; Cheminèe, A.; Crec’hriou, R.; Pastor, J.; Rider, M.; Dubas, R.; Lecaillon, G.; Boissery, P.; et al. Small artificial habitats to enhance the nursery function for juvenile fish in a large commercial port of the Mediterranean. Ecol. Eng. 2017, 105, 78–86. [Google Scholar] [CrossRef]
- Harvey, B.C.; Stewart, A.J. Fish size and habitat depth relationships in headwater streams. Oecologia 1991, 87, 336–342. [Google Scholar] [CrossRef] [PubMed]
- Goertzen, D.; Suhling, F. Promoting dragonfly diversity in cities: Major determinants and implications for urban pond design. J. Insect. Conserv. 2013, 17, 399–409. [Google Scholar] [CrossRef]
- Jrczak, T.; Wojtal-Frankiewicz, A.; Kaczkowski, Z.; Oleksińska, Z.; Bednarek, A.; Zalewski, M. Restoration of a shady urban pond-The pros and cons. J. Environ. Manag. 2018, 217, 919–928. [Google Scholar] [CrossRef] [PubMed]
- Byun, H.; Lee, D. Defining three rainy seasons and the hydrological summer monsoon in Korea using available water resources index. J. Meteor. Soc. Jpn. 2002, 80, 33–44. [Google Scholar] [CrossRef]
- Colt, J. Computation of Dissolved Gas Concentrations in Water as Functions of Temperature, Salinity, and Pressure; American Fisheries Society: Bethesda, MD, USA, 1984; pp. 1–154. [Google Scholar]
- Howmiller, R.P.; Sloey, W.E. A Horizontal water sampler for investigation of stratified waters. Limnol. Oceanogr. 1969, 14, 291–292. [Google Scholar] [CrossRef]
- American Public Health Association (APHA). Standard Methods for the Examination of Water and Wastewater, 21st ed.; American Public Health Association: Washington, DC, USA, 2005; pp. 9–72. [Google Scholar]
- Sutherland, D.L.; Turnbull, M.H.; Craggs, R.J. Increased pond depth improves algal productivity and nutrient removal in wastewater treatment high rate algal ponds. Water Res. 2014, 53, 271–281. [Google Scholar] [CrossRef] [PubMed]
- Englund, G.; Öhlund, G.; Hein, C.L.; Diehl, S. Temperature dependence of the functional response. Ecol. Lett. 2011, 14, 914–921. [Google Scholar] [CrossRef]
- Carveth, C.J.; Widmer, A.M.; Bonar, S.A.; Simms, J.R. An examination of the effects of chronic static and fluctuating temperature on the growth and survival of spikedace, Meda fulgida, with implications for management. J. Therm. Biol. 2007, 32, 102–108. [Google Scholar] [CrossRef]
- Johnstone, H.C.; Rahel, F.J. Assessing temperature tolerance of Bonneville cutthroat trout based on constant and cycling thermal regimes. Trans. Am. Fish. Soc. 2003, 132, 92–99. [Google Scholar] [CrossRef]
- Jobling, M. Temperature tolerance and the final preferendum-rapid mathods for the assessment of optimum growth temperature. J. Fish Biol. 1981, 19, 439–455. [Google Scholar] [CrossRef]
- Johnson, J.A.; Kelsch, S.W. Effects of evolutionary thermal environment on temperature-preference relationships in fishes. Environ. Biol. Fish. 1998, 53, 447–458. [Google Scholar] [CrossRef]
- Souchon, Y.; Tissot, L. Synthesis of thermal tolerances of the common freshwater fish species in large Western Europe rivers. Knowl. Manag. Aquat. Ecosyst. 2012, 405, 1–48. [Google Scholar] [CrossRef]
- Kramer, D.L. Dissolved oxygen and fish behavior. Environ. Biol. Fish. 1987, 18, 81–92. [Google Scholar] [CrossRef]
- Fang, X.; Stefan, H.G.; Eaton, J.G.; McCormick, J.H.; Alam, S.R. Simulation of thermal/dissolved oxygen habitat for fishes in lakes under different climate scenarios Part 2. Cold-water fish in the contiguous US. Ecol. Model. 2004, 172, 39–54. [Google Scholar] [CrossRef]
- Fang, X.; Stefan, H.G.; Eaton, J.G.; McCormick, J.H.; Alam, S.R. Simulation of thermal/dissolved oxygen habitat for fishes in lakes under different climate scenarios Part 3. Warm-water fish in the contiguous US. Ecol. Model. 2004, 172, 55–68. [Google Scholar] [CrossRef]
- Coutant, C.C. Complication of temperature preference data. J. Fish. Res. Board Can. 1977, 34, 739–745. [Google Scholar] [CrossRef]
- Reynolds, W.W.; Casterlin, M.E. Effect of temperature on locomotor activity in the goldfish (Carassius auratus) and the bluegill (Lepomis macrochirus): Presence of an ‘activity well’ in the region of the final preferendum. Hydrobiologia 1979, 65, 3–5. [Google Scholar] [CrossRef]
- Coutant, C.C. Thermal preference: When does an asset become a liability? Environ. Biol. Fishes 1987, 18, 161–172. [Google Scholar] [CrossRef]
- Beitinger, T.L.; Bennett, W.A.; McCauley, R.W. Temperature tolerances of North American freshwater fishes exposed to dynamic changes in temperature. Environ. Biol. Fishes 2000, 58, 237–275. [Google Scholar] [CrossRef]
- Ford, T.; Bertinger, T.L. Temperature tolerance in the goldfish, Carassius auratus. J. Therm. Biol. 2004, 30, 147–152. [Google Scholar] [CrossRef]
- Korwin-Kossakowski, M. The influence of temperature during the embryonic period on larval growth and development in carp, Cyprinus carpio L., and gross carp, Ctenopharyngodon idelia (val.): Theoretical and practical aspects. Arch. Pol. Fish. 2008, 16, 231–314. [Google Scholar] [CrossRef]
- Takahara, T.; Honjo, M.N.; Uchii, K.; Minamoto, T.; Doi, H.; Ito, T.; Kawabata, Z. Effects of daily temperature variation on the survival of carp infected with Cyprinid herpesvirus 3. Aquaculture 2014, 433, 208–213. [Google Scholar] [CrossRef]
- Helfman, G.S.; Collette, B.B.; Facey, D.E.; Bowen, B.W. The Diversity of Fishes: Biology, Evolution, And Ecology, 2nd ed.; Wiley-Blackwell Publishing: Hoboken, NJ, USA, 2009; pp. 1–736. [Google Scholar]
- Chung, N.; Park, B.; Kim, K. Potential effect of increased water temperature on fish habitats in Han River watershed. J. Korean Soc. Water. Environ. 2011, 27, 314–321. [Google Scholar]
- Nam, M.; Choi, N.; Kim, S.; Seok, K.; Lee, J. Seedling production and rearing of pale chup, Zacco platypus (Temmink et Schlegel). J. Aquac. 1999, 12, 25–30. [Google Scholar]
- Van Vliet, M.T.H.; Ludwig, F.; Kabat, P. Global streamflow and thermal habitats of freshwater fishes under climate change. Clim. Chang. 2013, 121, 739–754. [Google Scholar] [CrossRef]
- Kang, H.; Hur, J.W.; Park, D. The effects of cold Water released from dams on Zacco platypus gonad maturation in the Nakdong River, South Korea. KSCE J. Civ. Eng. 2017, 21, 1473–1483. [Google Scholar] [CrossRef]
- Bohlen, J. Temperature and oxygen requirements of early life stages of the endangered spined loach, Cobitis taenia L. (Teleostei, Cobitidae) and implications for the management of natural populations. Arch. Hydrobiol. 2003, 157, 195–212. [Google Scholar] [CrossRef]
- Suk, H.Y.; Choe, J.C. Females prefer males with larger first dorsal fins in the common freshwater goby. J. Fish Biol. 2002, 61, 899–914. [Google Scholar] [CrossRef]


| Description | Normal Season | Rainy Season | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| St. 1 | St. 2 | St. 1 | St. 2 | |||||||||
| Mean ± S.D. | Max. | Min. | Mean ± S.D. | Max. | Min. | Mean ± S.D. | Max. | Min. | Mean ± S.D. | Max. | Min. | |
| Temperature (°C) a | 24.1 ± 2.0 | 28.4 | 19.3 | 20.4 ± 1.5 | 22.4 | 17.9 | 25.1 ± 1.7 | 30.1 | 21.7 | 23.4 ± 1.0 | 26.2 | 21.6 |
| pH a | 7.7 ± 0.3 | 8.5 | 7.2 | 7.5 ± 0.1 | 7.9 | 7.2 | 7.4 ± 0.2 | 8.2 | 7.0 | 7.3 ± 0.1 | 7.7 | 7.1 |
| DO (mg L−1) a | 8.1 ± 1.0 | 10.1 | 5.8 | 4.5 ± 0.4 | 6.4 | 4.0 | 8.3 ± 1.8 | 13.4 | 4.8 | 5.6 ± 1.3 | 10.3 | 3.9 |
| DO saturation (%) c | 96.2 ± 12.1 | 123.6 | 68.7 | 49.2 ± 3.3 | 69.8 | 44.5 | 101.1 ± 23.7 | 169.7 | 56.4 | 66.1 ± 15.5 | 120.1 | 46.0 |
| EC (μS cm−1) a | 211 ± 11 | 239 | 187 | 225 ± 8 | 248 | 208 | 133 ± 29 | 217 | 62 | 148 ± 24 | 223 | 117 |
| Turbidity (NTU) b | 8.8 ± 2.8 | 12.0 | 6.5 | 7.3 ± 1.2 | 8.3 | 6.0 | 6.0 ± 0.2 | 6.2 | 5.8 | 6.5 ± 0.6 | 7.2 | 6.1 |
| SS (mg L−1) b | 9.4 ± 1.9 | 11.5 | 7.8 | 5.1 ± 0.7 | 5.8 | 4.5 | 8.1 ± 0.3 | 8.4 | 7.8 | 8.3 ± 0.4 | 8.7 | 7.9 |
| TOC (mg L−1) b | 1.77 ± 0.14 | 1.79 | 1.73 | 1.75 ± 0.14 | 1.78 | 1.73 | 2.04 ± 0.22 | 2.42 | 1.75 | 1.99 ± 0.15 | 2.28 | 1.81 |
| DOC (mg L−1) b | 1.69 ± 0.15 | 1.74 | 1.61 | 1.71 ± 0.15 | 1.75 | 1.67 | 1.85 ± 0.11 | 2.05 | 1.69 | 1.91 ± 0.18 | 2.25 | 1.71 |
| BOD (mg L−1) b | 3.1 ± 0.5 | 3.5 | 2.6 | 3.1 ± 0.3 | 3.3 | 2.8 | 3.3 ± 0.2 | 3.5 | 3.2 | 3.1 ± 0.2 | 3.3 | 3.0 |
| CODMn (mg L−1) b | 2.8 ± 0.3 | 3.1 | 2.4 | 2.8 ± 0.3 | 3.1 | 2.3 | 2.6 ± 0.2 | 2.9 | 2.4 | 2.7 ± 0.1 | 2.9 | 2.5 |
| TN (mg L−1) b | 1.2 ± 0.1 | 1.3 | 1.1 | 1.2 ± 0.1 | 1.3 | 1.1 | 1.3 ± 0.1 | 1.4 | 1.2 | 1.3 ± 0.1 | 1.4 | 1.2 |
| NH3 (μg L−1) b | 159 ± 7 | 166 | 152 | 167 ± 10 | 178 | 158 | 160 ± 10 | 170 | 150 | 170 ± 13 | 182 | 156 |
| NO3− (μg L−1) b | 1043 ± 25 | 1070 | 1020 | 1039 ± 22 | 1063 | 1021 | 1033 ± 21 | 1050 | 1010 | 1034 ± 14 | 1048 | 1.021 |
| TP (μg L−1) b | 36 ± 3 | 40 | 34 | 40 ± 5 | 45 | 36 | 42 ± 4 | 47 | 39 | 43 ± 2 | 45 | 42 |
| PO43− (μg L−1) b | 22 ± 3 | 25 | 19 | 24 ± 4 | 29 | 21 | 16 ± 2 | 18 | 14 | 18 ± 4 | 21 | 14 |
| Chl-a (μg L−1) b | 8.5 ± 1.1 | 9.6 | 7.5 | 1.1 ± 0.4 | 1.5 | 0.8 | 13.5 ± 1.2 | 14.7 | 12.4 | 0.7 ± 0.6 | 1.2 | ND d |
| Description | Normal Season | Rainy Season | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| St. 1 | St. 2 | St. 1 | St. 2 | |||||||||
| Mean ± S.D. | Max. | Min. | Mean ± S.D. | Max. | Min. | Mean ± S.D. | Max. | Min. | Mean ± S.D. | Max. | Min. | |
| Temperature (°C) a | 24.0 ± 2.7 | 29.1 | 16.6 | 23.3 ± 2.5 | 28.1 | 16.3 | 26.0 ± 1.4 | 30.4 | 23.3 | 25.4 ± 1.3 | 29.5 | 22.8 |
| pH a | 7.4 ± 0.3 | 8.4 | 7.0 | 7.6 ± 0.2 | 8.3 | 7.2 | 7.2 ± 0.5 | 8.9 | 6.6 | 7.3 ± 0.4 | 8.7 | 6.9 |
| DO (mg L−1) a | 6.9 ± 2.2 | 12.1 | 1.0 | 6.0 ± 1.7 | 9.7 | 1.6 | 6.7 ± 2.3 | 13.3 | 2.0 | 5.6 ± 1.6 | 10.5 | 2.1 |
| DO saturation (%) c | 81.7 ± 27.1 | 154.0 | 12.0 | 70.3 ± 20.8 | 122.7 | 19.3 | 85.1 ± 30.9 | 176.6 | 29.0 | 68.3 ± 20.8 | 133.3 | 16.6 |
| EC (μS cm−1) a | 169 ± 11 | 187 | 145 | 174 ± 11 | 195 | 144 | 107 ± 47 | 189 | 60 | 107 ± 49 | 195 | 59 |
| Turbidity (NTU) b | 8.7 ± 1.7 | 12.0 | 6.5 | 8.0 ± 1.0 | 8.9 | 6.0 | 7.8 ± 1.8 | 10.1 | 5.8 | 8.8 ± 2.5 | 12.5 | 6.1 |
| SS (mg L−1) b | 7.7 ± 2.1 | 11.5 | 5.1 | 5.6 ± 0.6 | 6.3 | 4.5 | 9.5 ± 1.7 | 12.1 | 7.8 | 10.0 ± 1.9 | 13.1 | 7.9 |
| TOC (mg L−1) b | 2.20 ± 0.11 | 2.40 | 2.04 | 2.50 ± 0.18 | 2.76 | 2.22 | 2.25 ± 0.14 | 2.51 | 2.05 | 2.38 ± 0.14 | 2.54 | 2.10 |
| DOC (mg L−1) b | 2.16 ± 0.11 | 2.35 | 1.97 | 2.45 ± 0.18 | 2.75 | 2.19 | 2.16 ± 0.12 | 2.35 | 1.97 | 2.29 ± 0.12 | 2.50 | 2.07 |
| BOD (mg L−1) b | 3.2 ± 0.3 | 3.5 | 2.6 | 3.2 ± 0.2 | 3.3 | 2.8 | 3.2 ± 0.2 | 3.5 | 3.0 | 3.2 ± 0.1 | 3.3 | 3.0 |
| CODMn (mg L−1) b | 3.6 ± 0.1 | 3.8 | 3.4 | 3.5 ± 0.1 | 3.7 | 3.3 | 3.4 ± 0.2 | 3.8 | 3.2 | 3.4 ± 0.1 | 3.6 | 3.3 |
| TN (mg L−1) b | 1.2 ± 0.1 | 1.3 | 1.1 | 1.2 ± 0.1 | 1.3 | 1.1 | 1.2 ± 0.1 | 1.4 | 1.0 | 1.2 ± 0.1 | 1.4 | 1.1 |
| NH3 (μg L−1) b | 146 ± 15 | 166 | 124 | 150 ± 19 | 178 | 127 | 138 ± 23 | 170 | 109 | 146 ± 26 | 182 | 118 |
| NO3− (μg L−1) b | 1048 ± 18 | 1070 | 1020 | 720 ± 437 | 1063 | 102 | 1.011 ± 55 | 1051 | 892 | 986 ± 69 | 1048 | 850 |
| TP (μg L−1) b | 33 ± 4 | 40 | 27 | 35 ± 5 | 45 | 29 | 35 ± 8 | 47 | 26 | 37 ± 7 | 45 | 29 |
| PO43− (μg L−1) b | 24 ± 3 | 28 | 19 | 25 ± 3 | 29 | 21 | 19 ± 3 | 22 | 14 | 21 ± 4 | 25 | 14 |
| Chl-a (μg L−1) b | 10.4 ± 2.6 | 15.1 | 7.5 | 3.3 ± 2.2 | 6.1 | 0.8 | 14.5 ± 1.5 | 16.6 | 12.4 | 3.5 ± 2.9 | 7.1 | ND d |
| Family | Species | RA a (%) | OT b (°C) | FPT c (°C) | ST d (°C) | LT e (°C) | FPT of This Study (°C) | References |
|---|---|---|---|---|---|---|---|---|
| Cyprinidae (Acheilognathinae) | Bitterling (Rhodeus uyekii) | 57.9 | 12.0–24.3 | 20.2 | 15.0–21.0 | 36.5 | 20.2 | [34,35] |
| Cyprinidae | Crucian carp (Carassius auratus) | 17.8 | 25.0–30.0 | 28.0–35.8 | 26.0–28.0 | 38.6–43.6 | 30.0 | [33,40,42,43,44] |
| Danioninae | Minnow (Zacco platypus) | 12.1 | 28.0–30.5 | 29.0 | 26.0 | 32.0 | 29.0 | [48,49,50] |
| Cobitidae | Chinese muddy loach (Misgurmus mizolepis) | 6.5 | 17.8–26.1 | 26.1 | 18.0–26.0 | 30.8 | 26.1 | [51] |
| Gobiidae | Common freshwater goby (Rhinogobius brunneus) | 5.6 | 17.0–21.0 | ND f | 9.0–15.0 | ND f | 23.0 | [52] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ahn, C.H.; Lee, S.; Song, H.M.; Park, J.R.; Joo, J.C. Assessment of Water Quality and Thermal Stress for an Artificial Fish Shelter in an Urban Small Pond during Early Summer. Water 2019, 11, 139. https://doi.org/10.3390/w11010139
Ahn CH, Lee S, Song HM, Park JR, Joo JC. Assessment of Water Quality and Thermal Stress for an Artificial Fish Shelter in an Urban Small Pond during Early Summer. Water. 2019; 11(1):139. https://doi.org/10.3390/w11010139
Chicago/Turabian StyleAhn, Chang Hyuk, Saeromi Lee, Ho Myeon Song, Jae Roh Park, and Jin Chul Joo. 2019. "Assessment of Water Quality and Thermal Stress for an Artificial Fish Shelter in an Urban Small Pond during Early Summer" Water 11, no. 1: 139. https://doi.org/10.3390/w11010139
APA StyleAhn, C. H., Lee, S., Song, H. M., Park, J. R., & Joo, J. C. (2019). Assessment of Water Quality and Thermal Stress for an Artificial Fish Shelter in an Urban Small Pond during Early Summer. Water, 11(1), 139. https://doi.org/10.3390/w11010139





