Study of Sludge Particles Formed during Coagulation of Synthetic and Municipal Wastewater for Increasing the Sludge Dewatering Efficiency
Abstract
1. Introduction
- (a)
- results of traditional jar tests,
- (b)
- CST measurements,
- (c)
- determination of the volumetric dimension (Dv) and respectively of the specific surface area (Sps).
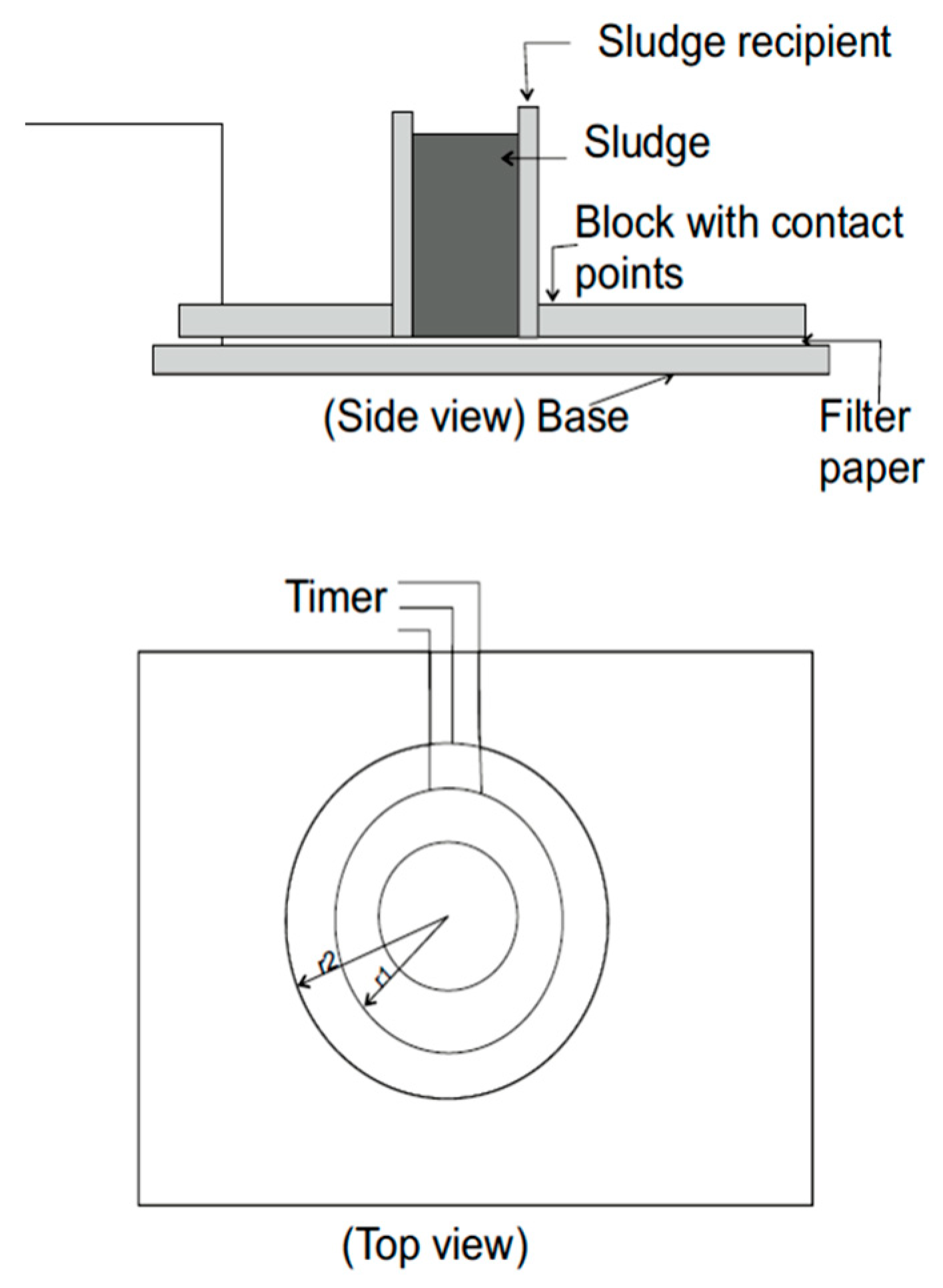
2. Materials and Methods
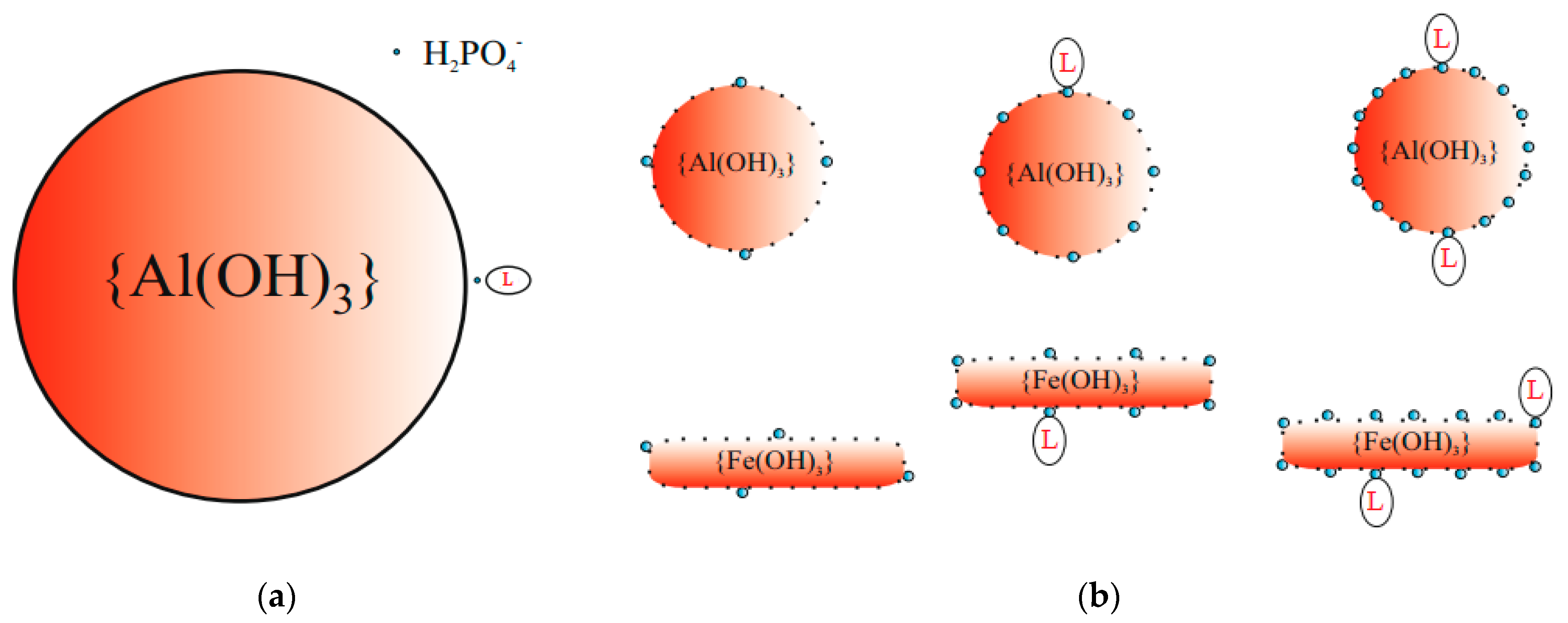
3. Results and Discussion
= 0.0012 + 0.165 + 0.0012 + 0.010 = 0.1685 (μm)
- (1)
- additional monitoring of the coagulation-flocculation process,
- (2)
- broader assessment of the “health”/quality of activated sludge,
- (3)
- better control and regulation of the process of municipal wastewater sludge dewatering.
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Nomenclature
| sww | synthetic wastewater |
| nww | real municipal wastewater (natural origin) |
| CST | capillary suction time (s) |
| aaf | aggregation-agglomeration-flocculation |
| SD | standard deviation of the sample (% or mg/L) |
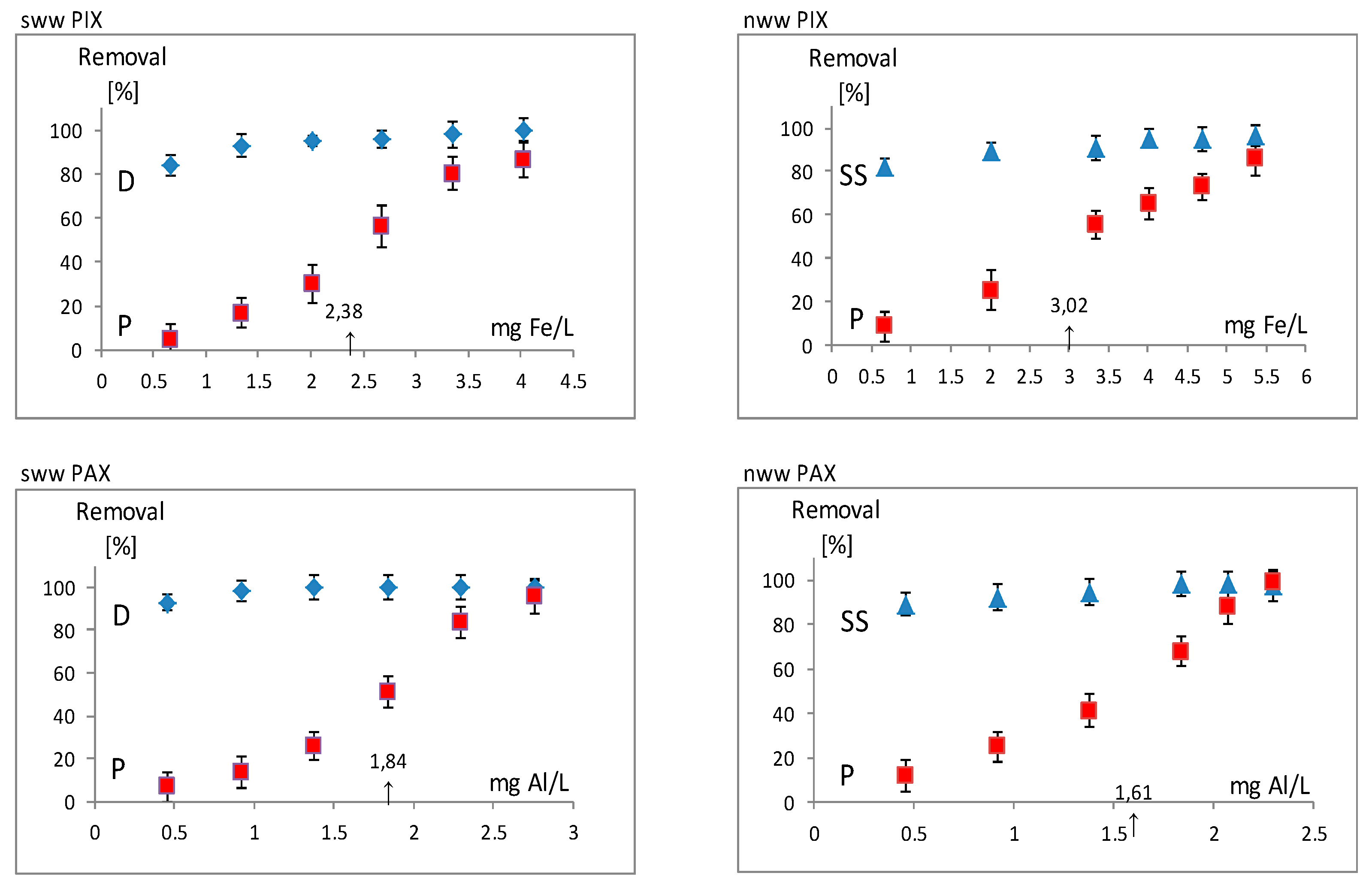
| fes | number of mg Fe3+ (PIX) which remove 50% P from 1 L of sww |
| als | umber of mg Al3+ (PAX) which remove 50% P from 1 L of sww |
| fen | number of mg Fe3+ (PIX) which remove 50% P from 1 L of nww |
| aln | number of mg Fe3+ (PIX) which remove 50% P from 1 L of nww |
| Dv | volumetric dimension (μm) |
| Sps | specific surface area (m2/g) |
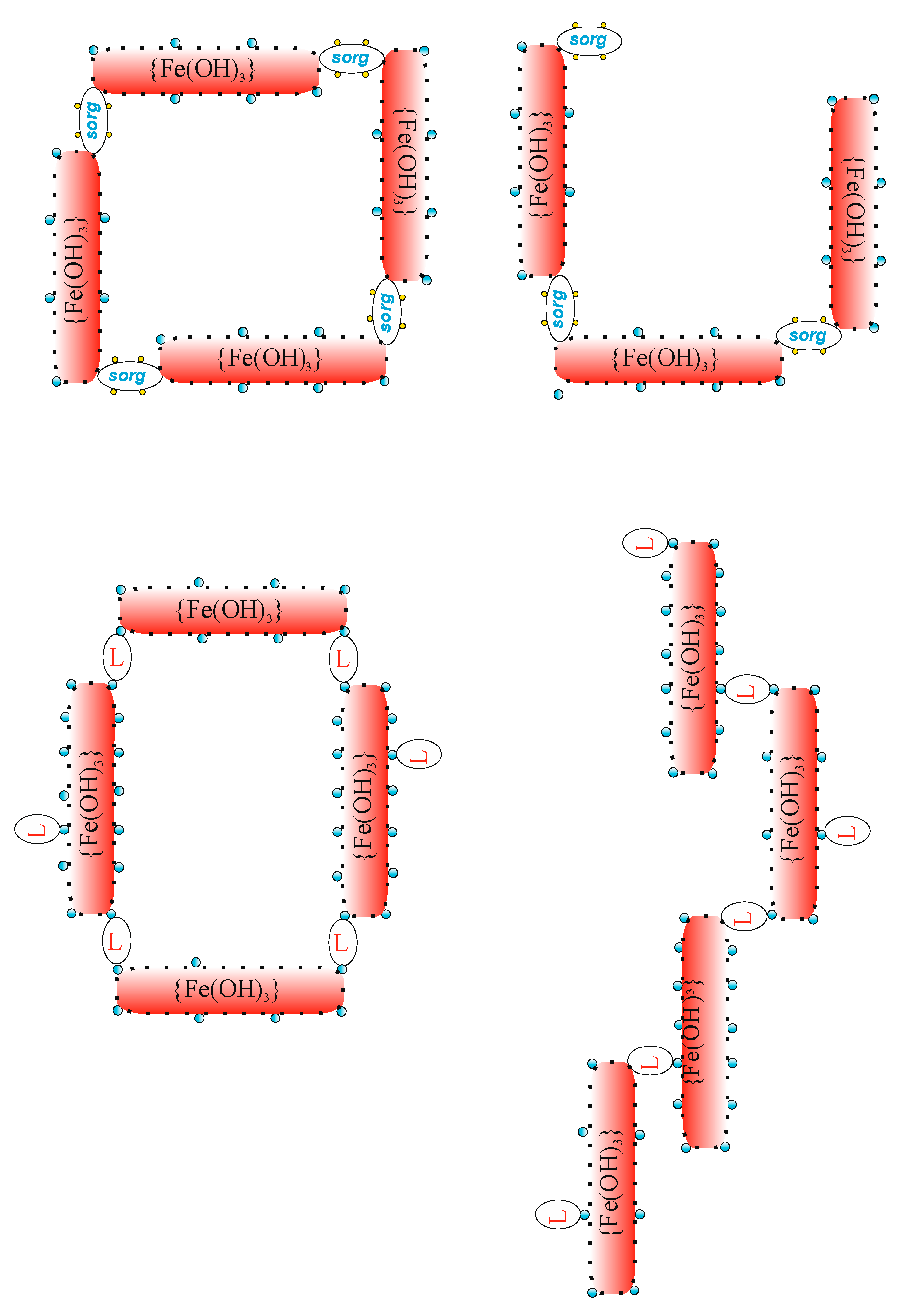
| L | disperse dye (Synthene Scarlet P3GL) |
| SS | Suspended Solids (mg/L) |
| prtc | colloidal particle {Fe(OH)3} or {Al(OH)3} |
| sorg | organic substances (responsible for COD in nww) |
References
- Duan, J.; Gregory, J. Coagulation by hydrolyzing metal salts. Adv Colloid Interface Sci. 2003, 100–102, 475–502. [Google Scholar] [CrossRef]
- The, C.Y.; Budiman, P.M.; Shak, K.P.; Wu, T.Y. Recent advancement of coagulation-flocculation and its application in wastewater treatment. Ind. Eng. Chem. Res. 2016, 55, 4363–4389. [Google Scholar]
- Zheng, H.; Shu, G.; Jiang, S.; Tshukudu, T.; Xiang, X.; Zhang, P.; He, Q. Investigations of coagulation-flocculation process by performance optimization, model prediction and fractal structure of flocs. Desalination 2011, 269, 148–156. [Google Scholar] [CrossRef]
- Chu, C.P.; Lee, D.J. Effect of pre-hydrolysis on floc structure. J. Environ. Manag. 2004, 71, 285–292. [Google Scholar] [CrossRef]
- Zhao, Y.X.; Shon, H.K.; Wang, Y.; Kim, J.H.; Yue, Q.Y. The effect of second coagulant dose on the regrowth of flocs formed by charge neutralization and sweep coagulation using titanium tetrachloride (TiCl4). J. Hazard Mater. 2011, 198, 70–77. [Google Scholar] [CrossRef]
- Fytili, D.; Zabaniotou, A. Utilization of sewage sludge in EU application of old and new methods—A review. Renew. Sustain. Energy Rev. 2008, 12, 116–140. [Google Scholar] [CrossRef]
- Verna, S.; Prased, B.; Mishra, I.M. Pretreatment of petrochemical wastewater by coagulation and the sludge characteristics. J. Hazard Mater. 2010, 178, 1055–1064. [Google Scholar] [CrossRef]
- Rohrsetzer, S.; Paszli, I.; Csempesz, F.; Ban, S. Colloidal stability of electrostatically stabilized sol particles. Part I: The role of hydration in coagulation and repeptization of ferric hydroxide sol. Colloid Polym. Sci. 1992, 270, 1243–1251. [Google Scholar] [CrossRef]
- Smoczyński, L.; Ratnaweera, H.; Kosobucka, M.; Kvaal, K.; Smoczyński, M. Image Analysis of Sludge Aggregates Obtained at Preliminary Treatment of Sewage. Water Sci. Technol. 2014, 70, 1048–1055. [Google Scholar] [CrossRef]
- Amirtharajah, A.; Mills, M.K. Rapid-mix design for mechanism of alum coagulation. JAWWA 1982, 74, 210–216. [Google Scholar] [CrossRef]
- Butler, E.; Hung, Y.T.; Yu, L.R.; Al Ahmad, M. Electrocoagulation in wastewater treatment. Water 2011, 3, 395–525. [Google Scholar] [CrossRef]
- Zaleschi, L.; Teodosiu, C.; Cretescu, I.; Rodrigo, M.A. A Comparative Study of Electrocoagulation and Chemical Coagulation Processes Applied for Wastewater Treatment. Environ. Eng. Manag. J. 2012, 11, 1517–1525. [Google Scholar]
- Smoczynski, L.; Munska, K.; Pierozynski, B. Electrocoagulation of synthetic dairy wastewater. Water Sci. Technol. 2013, 67, 404–409. [Google Scholar] [CrossRef]
- Groterud, O.; Smoczyński, L. Removal of phosphorus and residual aluminium by a recirculating electrolysis of wastewater. Vatten 1986, 42, 293–296. [Google Scholar]
- Mikkelsen, L.H.; Keiding, K. Physico-chemical characteristics of full scale sewage sludge with implications to dewatering. Water Res. 2002, 36, 2451–2462. [Google Scholar] [CrossRef]
- Jin, B.; Wilen, B.M.; Lant, P. Impact of morphological, physical and chemical properties of sludge flocs on dewaterability of activated sludge. Chem. Eng. J. 2004, 98, 115–126. [Google Scholar] [CrossRef]
- Turchiuli, C.; Fargues, C. Influence of structural properties of alum and ferric flocs on sludge dewaterability. Chem. Eng. J. 2004, 103, 123–131. [Google Scholar] [CrossRef]
- Niu, M.; Zhang, W.; Wang, D.; Chen, Y.; Chen, R. Correlation of physiochemical properties and sludge dewaterability under chemical conditioning using inorganic coagulants. Bioresour. Technol. 2013, 144, 337–343. [Google Scholar] [CrossRef]
- Zhao, Y.Q. Correlations between floc physical properties and optimum polymer dosage in alum sludge conditioning and dewatering. Chem. Eng. J. 2003, 92, 227–235. [Google Scholar] [CrossRef]
- Zhao, P.; Ge, S.; Chen, Z.; Li, X. Study on pore characteristics of flocs and sludge dewaterability based on fractal methods (pore characteristics of flocs and sludge dewatering). Appl. Therm. Eng. 2013, 58, 217–223. [Google Scholar] [CrossRef]
- Sawalha, O.; Scholz, M. Assessment of capillary suction time (CST) test methodology. Environ. Technol. 2007, 28, 1377–1386. [Google Scholar] [CrossRef]
- Sawalha, O.; Scholz, M. Innovative enhancement of the design and precision of the capillary suction time testing device. Water Environ. Res. 2009, 81, 2344–2352. [Google Scholar] [CrossRef]
- Scholtz, M.; Tapp, J. Development of the revised Capillary Suction Time (CST) test. Water Cond. Purif. 2006, 48, 46–52. [Google Scholar]
- Tuan, P.-A.; Sillanpaa, M. Migration of ions and organic matter during electro-dewatering of anaerobic sludge. J. Hazard Mater. 2010, 173, 54–61. [Google Scholar] [CrossRef]
- Lee, C.H.; Liu, J.C. Sludge dewaterability and floc structure in dual polymer conditioning. Adv. Environ. Res. 2001, 5, 129–136. [Google Scholar] [CrossRef]
- Meakin, P. The effects of reorganization processes on two dimensional cluster—Cluster aggregation. J. Colloid Interface Sci. 1986, 112, 187–194. [Google Scholar] [CrossRef]
- Yu, W.; Liu, T.; Gregory, J.; Li, G.; Liu, H.; Qua, J. Aggregation of nano-sized alum–humic primary particles. Sep. Purif. Technol. 2012, 99, 44–49. [Google Scholar] [CrossRef]
- Pastor-Satorras, R.; Rubí, J.M. Particle-cluster aggregation with dipolar interactions. Phys. Rev. E 1995, 51, 5994–6003. [Google Scholar] [CrossRef]
- Smoczyński, L.; Bukowski, Z.; Wardzyńska, R.; Załęska-Chróst, K. Dłużyńska, B. Simulation of coagulation, flocculation and sedimentation. Water Environ. Res. 2009, 81, 348–356. [Google Scholar] [CrossRef]
- Jullien, R.; Meakin, P. Simple models for the restructuring of 3-dimensional ballistic aggregates. J. Colloid Interface Sci. 1989, 127, 265–272. [Google Scholar] [CrossRef]
- Jarvis, P.; Jefferson, B.; Parsons, S.A. Measuring floc structural characteristics. Rev. Environ. Sci. Bio-Technol. 2005, 4, 1–18. [Google Scholar] [CrossRef]
- Chu, C.P.; Lee, D.J. Structural analysis of sludge flocs. Adv. Powder Technol. 2004, 15, 515–532. [Google Scholar] [CrossRef]
- Zhao, Y.Q. Settling behaviour of polymer flocculated water treatment sludge II: Effects of floc structure and floc packing. Sep. Purif. Technol. 2004, 35, 175–183. [Google Scholar] [CrossRef]
- Mandelbrot, B.B. The Fractal Geometry of Nature; Freeman: San Francisco, CA, USA, 1982. [Google Scholar]
- Leman, J.; Smoczynski, M.; Dolgan, T.; Dziuba, Z. Fractal analysis of structure of cow and goat β-lactoglobulin preparation. J. Food Technol. 2005, 42, 428–430. [Google Scholar]
- Yang, Z.; Yang, H.; Jiang, Z.; Huang, X.; Li, H.; Li, A.; Cheng, R. A new method for calculation of flocculation kinetics combining Smoluchowski model with fractal theory. Colloids Surf. A 2013, 423, 11–19. [Google Scholar] [CrossRef]
- Meng, F.G.; Zhang, H.M.; Li, Y.S.; Zhang, X.W.; Yang, F.L.; Xiao, J.N. Cake layer morphology in microfiltration of activated sludge wastewater based on fractal analysis. Sep. Purif. Technol. 2005, 44, 250–257. [Google Scholar] [CrossRef]
- Smoczynski, L.; Ratnaweera, H.; Kosobucka, M.; Smoczynski, M.; Pieczulis-Smoczynska, K.; Cretescu, I. The size of aggregates formed during coagulation and electrocoagulation of synthetic wastewater. J. Environ. Prot. Ecol. 2016, 17, 1160–1170. [Google Scholar]
- Wang, Y.; Gao, B.Y.; Xu, X.M.; Xu, G.Y. Characterization of floc size, strength and structure in various aluminum coagulants treatment. J. Colloid Interface Sci. 2009, 332, 354–359. [Google Scholar] [CrossRef]
- Smoczynski, L.; Kosobucka, M.; Smoczynski, M.; Ratnaweera, H.; Pieczulis-Smoczynska, K. Sizes of particles formed during municipal wastewater treatment. Water Sci. Technol. 2017, 75, 971–977. [Google Scholar]
- Smoczyński, L.; Ratnaweera, H.; Kosobucka, M.; Smoczyński, M.; Kalinowski, S.; Kvaal, K. Modelling the structure of sludge aggregates. Environ. Technol. 2016, 37, 1122–1132. [Google Scholar] [CrossRef]
- Smoczyński, L.; Ratnaweera, H.; Kosobucka, M.; Smoczyński, M. Image analysis of sludge aggregates. Sep. Purif. Technol. 2014, 122, 412–420. [Google Scholar] [CrossRef]
- Malvern Instruments Limited. A Basic Guide to Particle Characterization; Malvern Instruments Limited: Worcestershire, UK, 2012; pp. 1–26. [Google Scholar]
- Macedo, M.L.F.; Osawa, C.C.; Bertran, C.A. Sol-gel synthesis of transparent alumina gel and pure gamma alumina by urea hydrolysis of aluminum nitrate. J. Sol.-Gel. Sci. Technol. 2004, 30, 135–140. [Google Scholar] [CrossRef]
- Haas, W.; Zrinyi, M.; Kilian, H.G.; Heise, B. Structural analysis of anisometric colloidal iron(III)-hydroxide particles and particle-aggregates incorporated in poly(vinyl-acetate) networks. Colloid Polym. Sci. 1993, 271, 1024–1034. [Google Scholar] [CrossRef]
- Waite, T.D. Measurement and implications of flock structure in water and wastewater treatment. Colloids Surf. A 1999, 151, 27–41. [Google Scholar] [CrossRef]
- Smoczynski, L.; Wardzynska, R. Study on macroscopic aggregation of silica suspensions and sewage. J. Colloid Interface Sci. 1996, 183, 309–314. [Google Scholar] [CrossRef]
- Gregory, J. The role of floc density in solid-liquid separation. Filtr. Sep. 1998, 35, 367–371. [Google Scholar] [CrossRef]
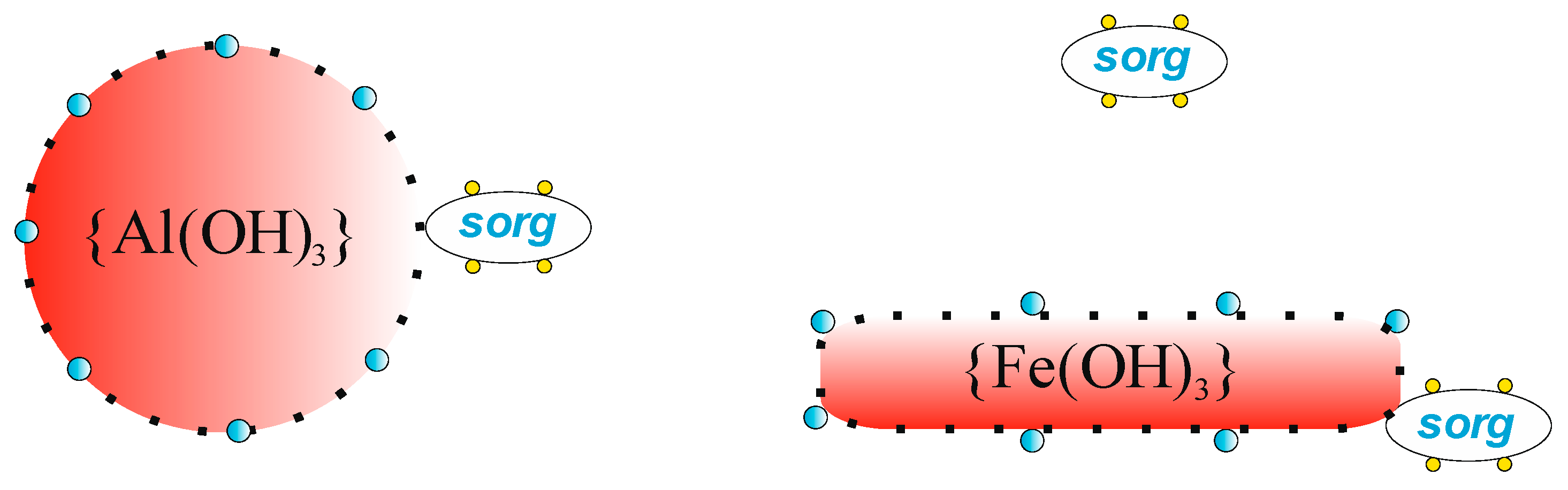
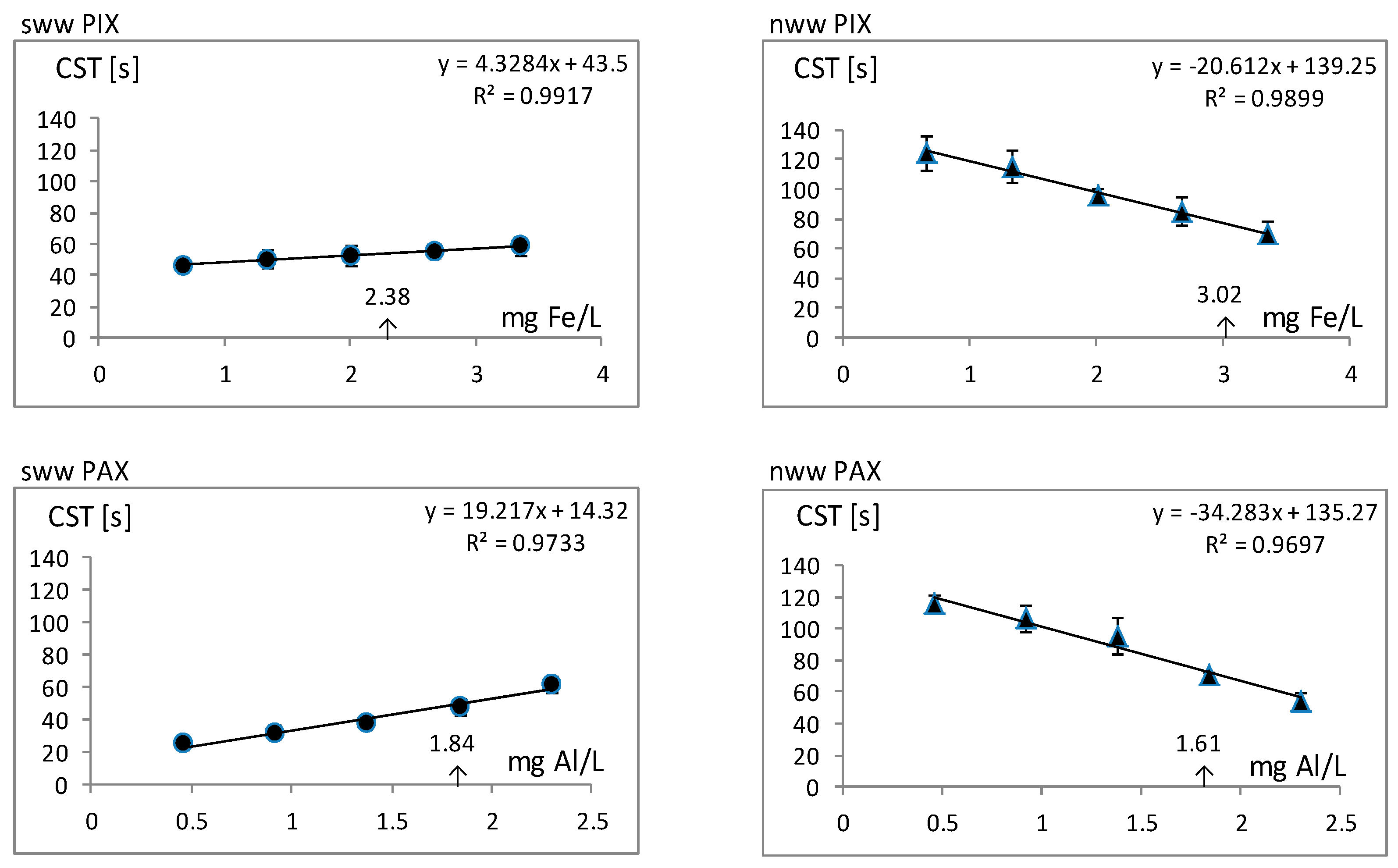
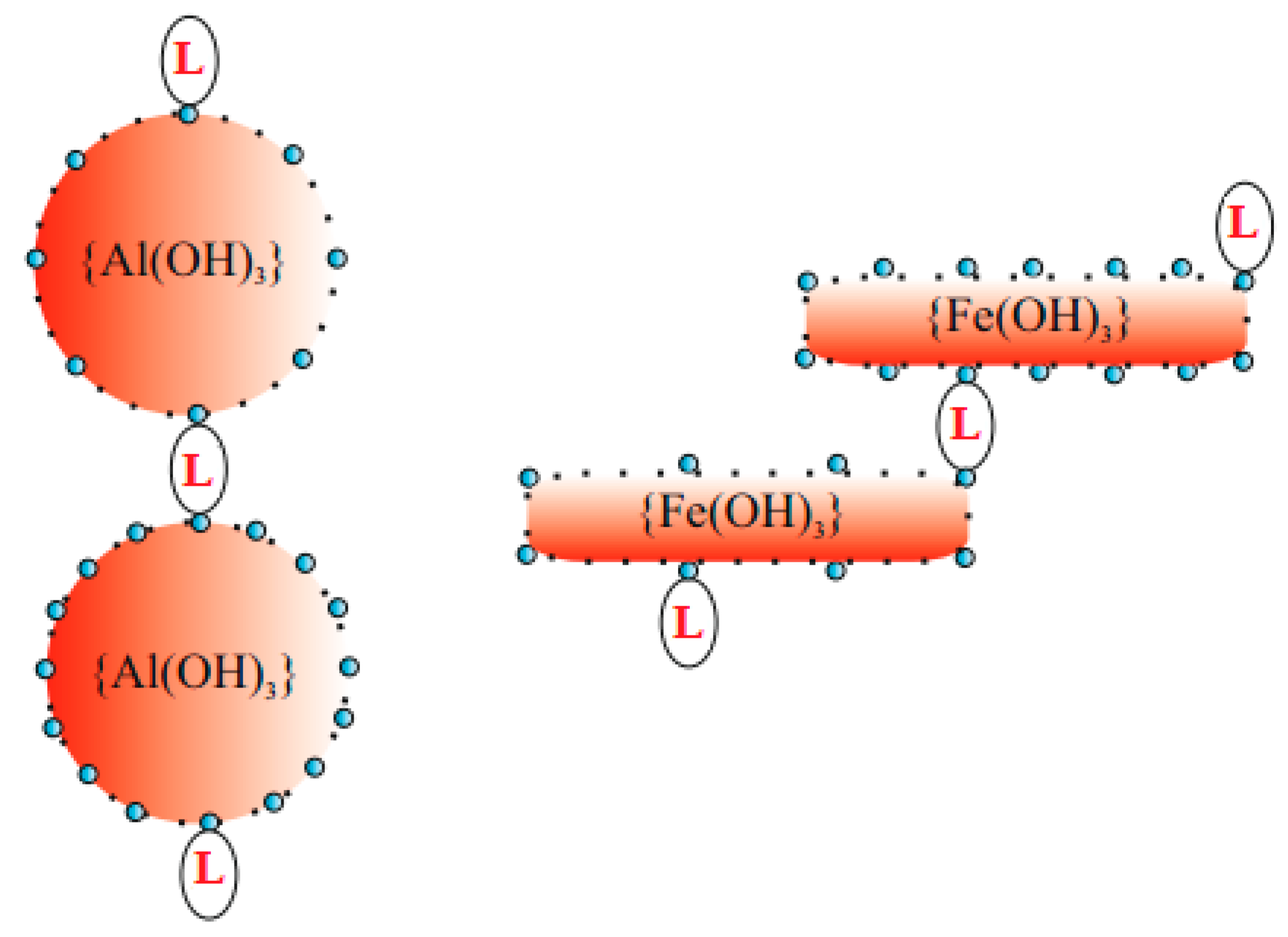
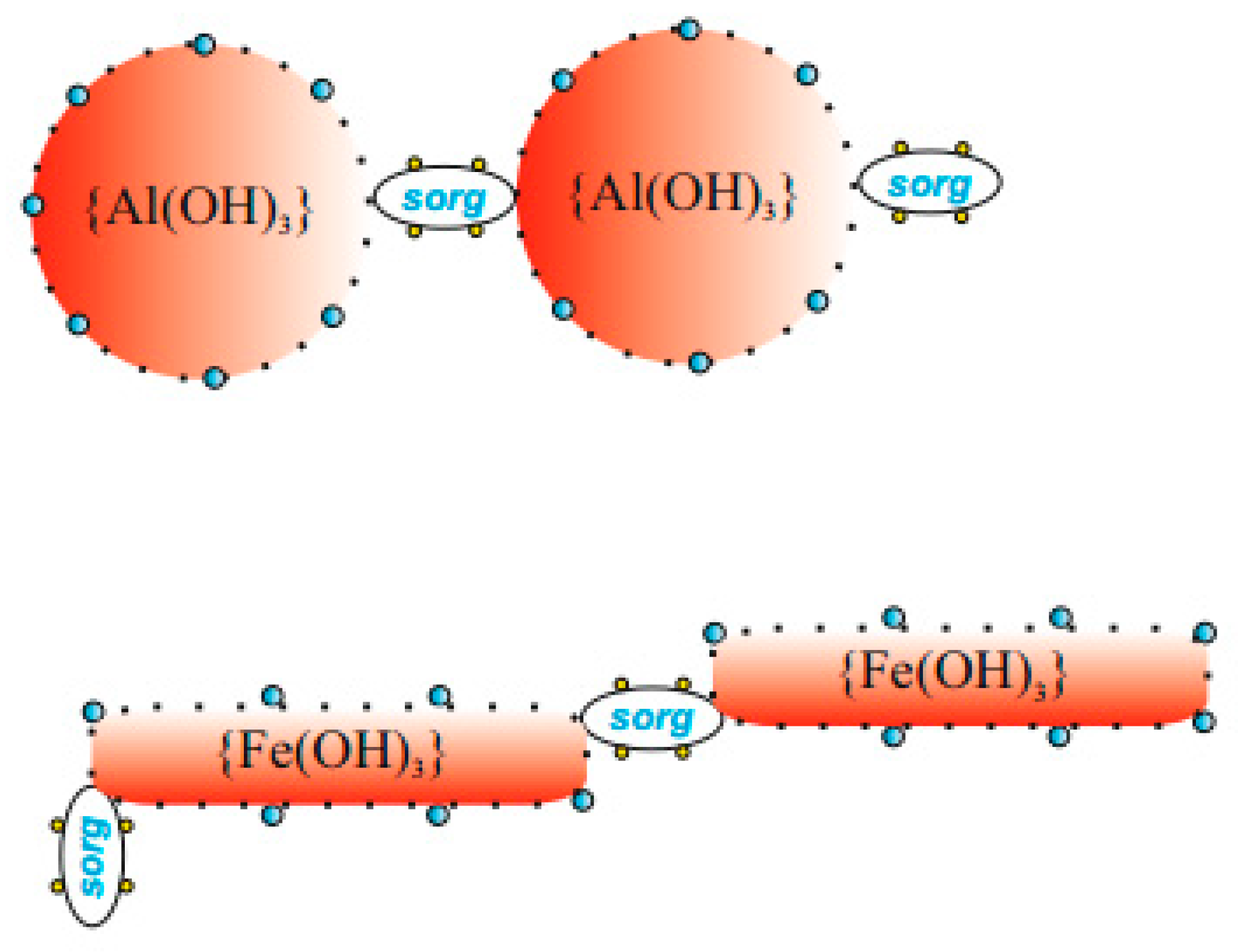
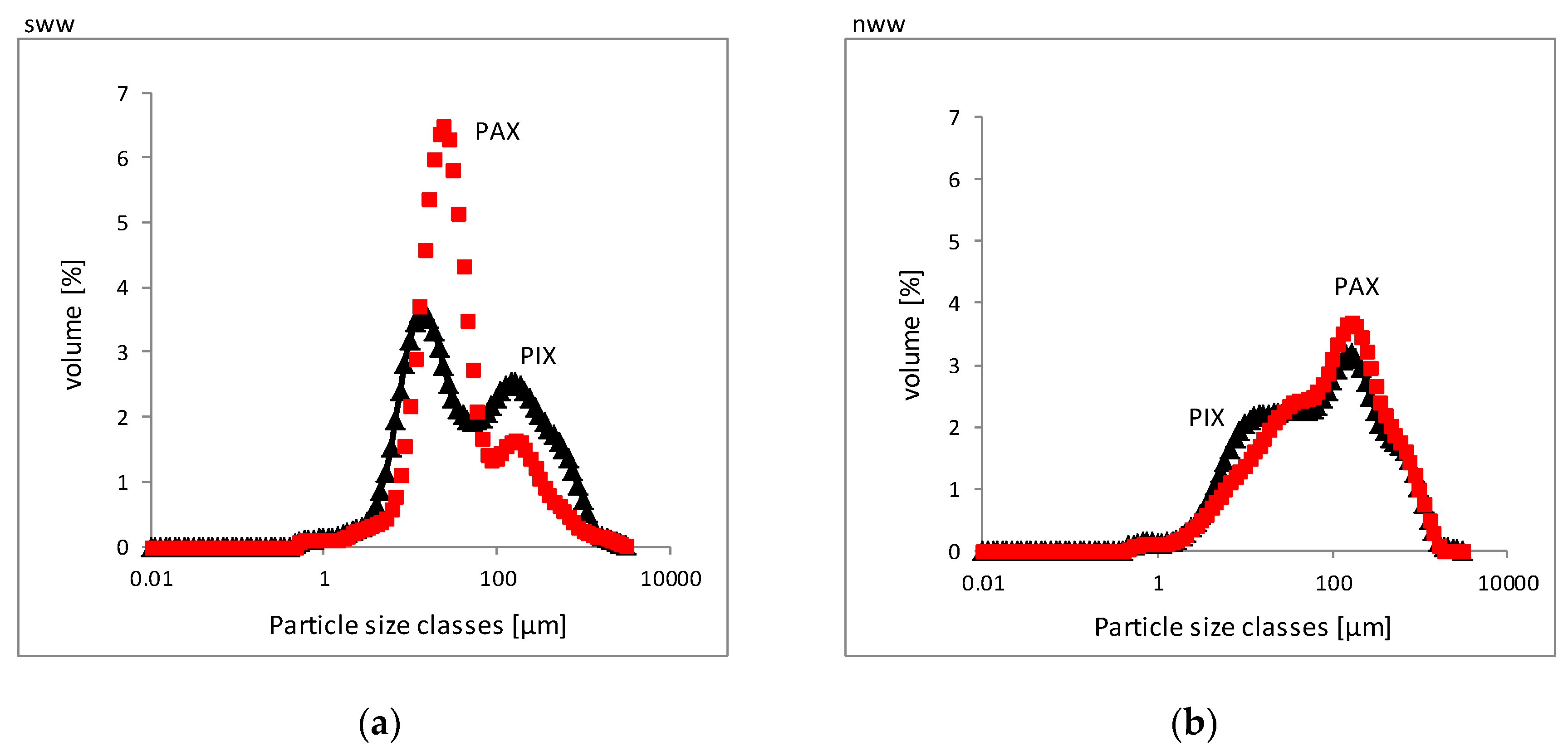
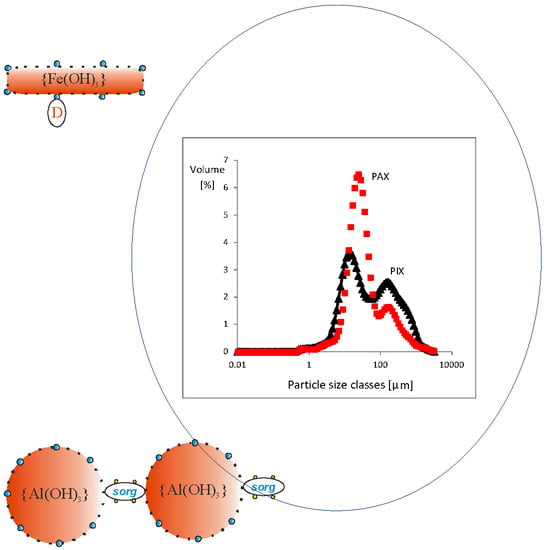
| Properties | PIX | PAX | PIX | PAX |
|---|---|---|---|---|
| sww/SD | sww/SD | nww/SD | nww/SD | |
| Sps (m2∙g−1) | 355/13.9 | 283/10.0 | 283/10.0 | 350/12.2 |
| Dv10 (μm) | 8.4/1.7 | 9.4/1.2 | 9.4/1.2 | 7.4/1.2 |
| Dv50 (μm) | 48.9/3.3 | 99.9/4.5 | 99.9/4.5 | 77.3/4.8 |
| Dv90 (μm) | 702/17.5 | 537/15.8 | 537/15.8 | 548/6.9 |
| Coagulant/Wastewater Type | Dv50 (μm) | Number of Units in a Floc |
|---|---|---|
| PIX/sww | 48.9 | 20,091 |
| PAX/sww | 32.4 | 9776 |
| PIX/nww | 99.9 | 70,138 |
| PAX/nww | 77.3 | 44,774 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Smoczynski, L.; Kalinowski, S.; Cretescu, I.; Smoczynski, M.; Ratnaweera, H.; Trifescu, M.; Kosobucka, M. Study of Sludge Particles Formed during Coagulation of Synthetic and Municipal Wastewater for Increasing the Sludge Dewatering Efficiency. Water 2019, 11, 101. https://doi.org/10.3390/w11010101
Smoczynski L, Kalinowski S, Cretescu I, Smoczynski M, Ratnaweera H, Trifescu M, Kosobucka M. Study of Sludge Particles Formed during Coagulation of Synthetic and Municipal Wastewater for Increasing the Sludge Dewatering Efficiency. Water. 2019; 11(1):101. https://doi.org/10.3390/w11010101
Chicago/Turabian StyleSmoczynski, Lech, Slawomir Kalinowski, Igor Cretescu, Michal Smoczynski, Harsha Ratnaweera, Mihaela Trifescu, and Marta Kosobucka. 2019. "Study of Sludge Particles Formed during Coagulation of Synthetic and Municipal Wastewater for Increasing the Sludge Dewatering Efficiency" Water 11, no. 1: 101. https://doi.org/10.3390/w11010101
APA StyleSmoczynski, L., Kalinowski, S., Cretescu, I., Smoczynski, M., Ratnaweera, H., Trifescu, M., & Kosobucka, M. (2019). Study of Sludge Particles Formed during Coagulation of Synthetic and Municipal Wastewater for Increasing the Sludge Dewatering Efficiency. Water, 11(1), 101. https://doi.org/10.3390/w11010101