Microbial Processing and Production of Aquatic Fluorescent Organic Matter in a Model Freshwater System
Abstract
1. Introduction
2. Materials and Methods
2.1. Laboratory Model Systems
2.2. Water Samples
2.2.1. Environmental Water Samples
2.2.2. Synthetic Water Samples and Supplemented Synthetic Water Samples
2.3. Fluorescence Measurements
2.4. Fluorescence Data Analysis
2.5. Bacterial Enumeration
3. Results
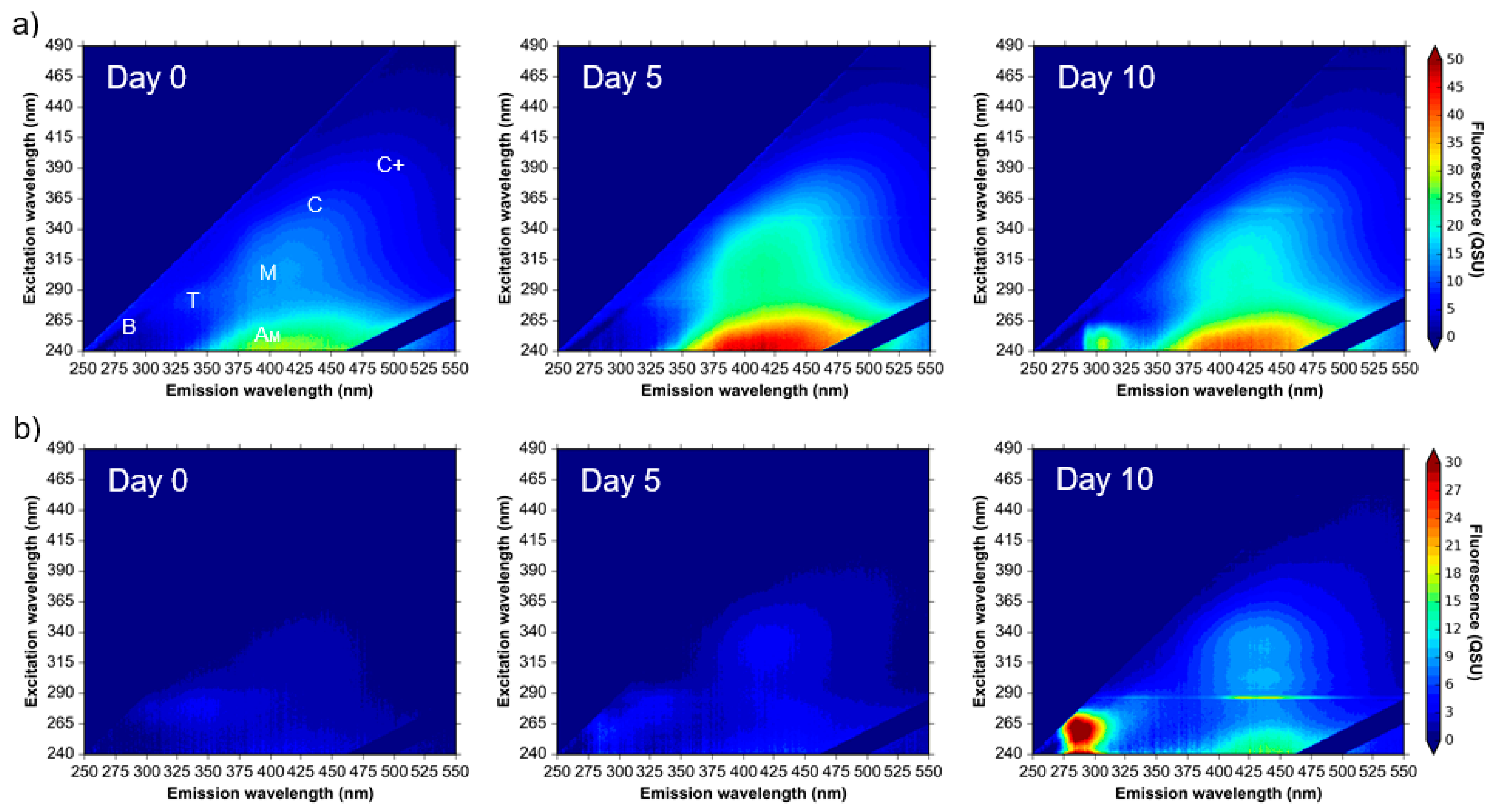
3.1. Microbial AFOM Processing over Time: Environmental Samples
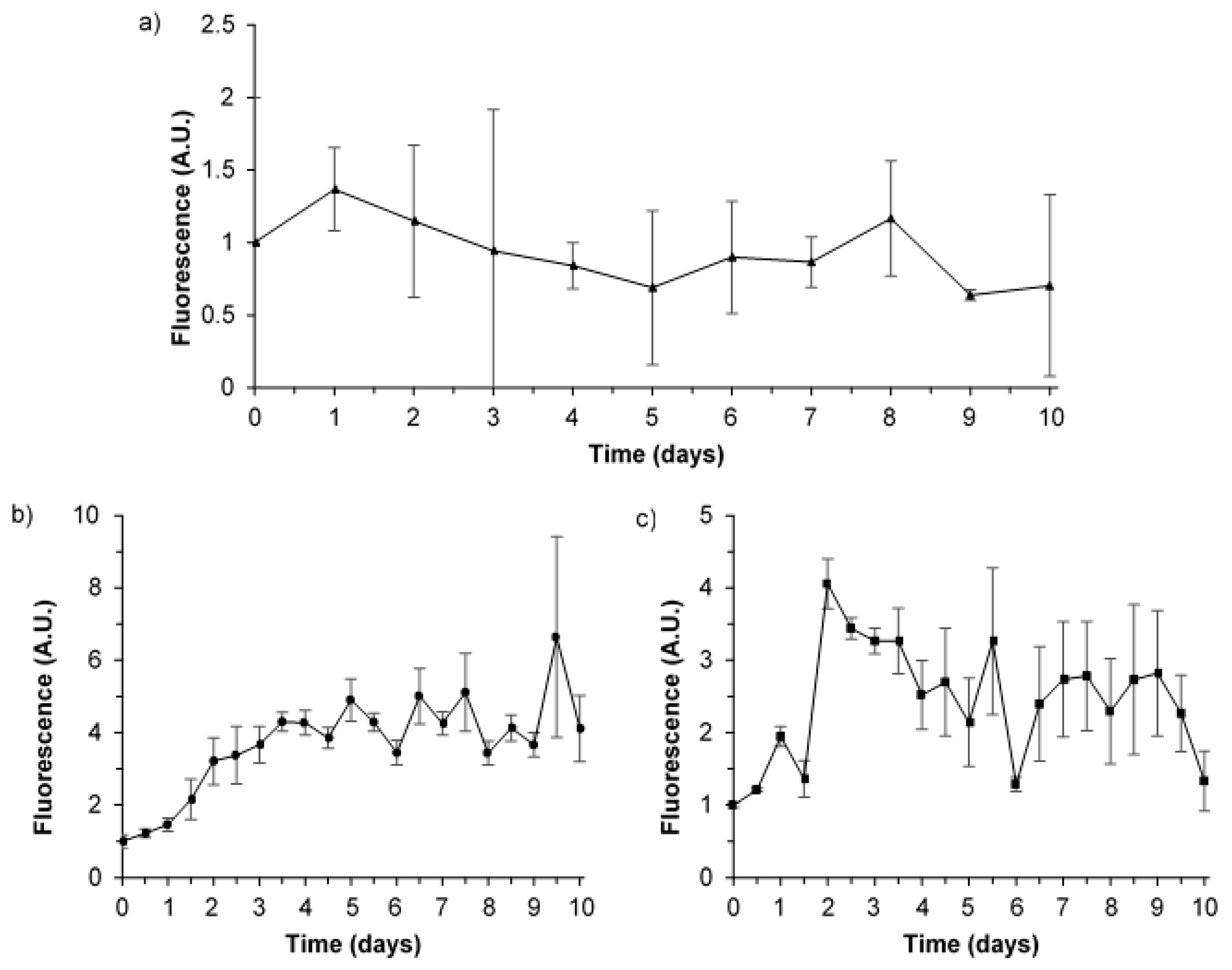
3.2. Bacterial AFOM Processing over Time: Supplemented Synthetic Samples
3.3. Bacterial AFOM Processing over Time: Synthetic Samples
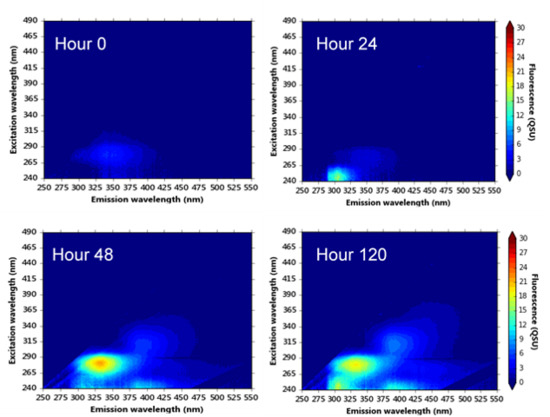
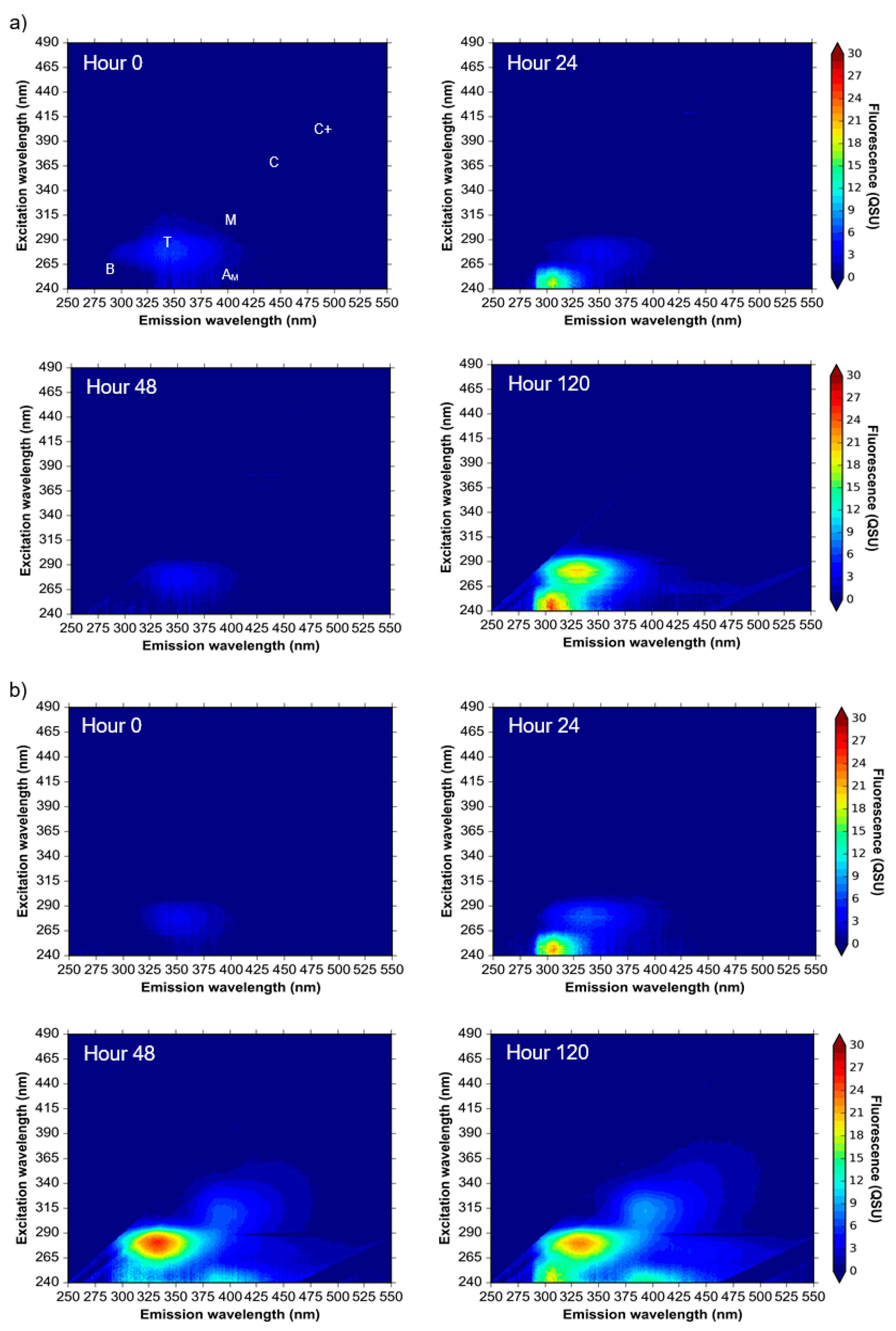
3.4. Hourly Monitoring of Synthetic Samples: Bacterial Growth and Fluorescence Development
4. Discussion
4.1. Microbially Engineered Protein-Like Fluorescence
Fluorescence, Microbes and Dissolved Oxygen
4.2. Microbially Engineered Humic-Like Fluorescence
4.3. Microbially Engineered AFOM: Hourly Fluorescence and Bacterial Enumeration Measurements
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Baker, A.; Spencer, R.G.M. Characterization of dissolved organic matter from source to sea using fluorescence and absorbance spectroscopy. Sci. Total Environ. 2004, 333, 217–232. [Google Scholar] [CrossRef] [PubMed]
- Larsen, L.G.; Aiken, G.R.; Harvey, J.W.; Noe, G.B.; Crimaldi, J.P. Using fluorescence spectroscopy to trace seasonal DOM dynamics, disturbance effects, and hydrologic transport in the Florida Everglades. J. Geophys. Res. Biogeosci. 2010, 115, 1–14. [Google Scholar] [CrossRef]
- Bieroza, M.Z.; Heathwaite, A.L. Unravelling organic matter and nutrient biogeochemistry in groundwater-fed rivers under baseflow conditions: Uncertainty in in situ high-frequency analysis. Sci. Total Environ. 2016, 572, 1520–1533. [Google Scholar] [CrossRef] [PubMed]
- Omori, Y.; Hama, T.; Ishii, M.; Saito, S. Vertical change in the composition of marine humic-like fluorescent dissolved organic matter in the subtropical western North Pacific and its relation to photoreactivity. Mar. Chem. 2011, 124, 38–47. [Google Scholar] [CrossRef]
- Reynolds, D.M. The Principles of Fluorescence. In Aquatic Organic Matter Fluorescence; Baker, A., Reynolds, D.M., Lead, J., Coble, P.G., Spencer, R.G.M., Eds.; Cambridge Environmental Chemistry Series; Cambridge University Press: Cambridge, UK, 2014; pp. 3–34. ISBN 9780521764612. [Google Scholar]
- Fox, B.G.; Thorn, R.M.S.; Anesio, A.M.; Reynolds, D.M. The in situ bacterial production of fluorescent organic matter; an investigation at a species level. Water Res. 2017, 125, 350–359. [Google Scholar] [CrossRef] [PubMed]
- Stedmon, C.A.; Markager, S. Resolving the variability of dissolved organic matter fluorescence in a temperate estuary and its catchment using PARAFAC analysis. Limnol. Oceanogr. 2005, 50, 686–697. [Google Scholar] [CrossRef]
- Pellerin, B.A.; Saraceno, J.F.; Shanley, J.B.; Sebestyen, S.D.; Aiken, G.R.; Wollheim, W.M.; Bergamaschi, B.A. Taking the pulse of snowmelt: In situ sensors reveal seasonal, event and diurnal patterns of nitrate and dissolved organic matter variability in an upland forest stream. Biogeochemistry 2012, 108, 183–198. [Google Scholar] [CrossRef]
- Coble, P.G.; Lead, J.; Baker, A.; Reynolds, D.M.; Spencer, R.G.M. Aquatic Organic Matter Fluorescence; Cambridge University Press: Cambridge, UK, 2014; ISBN 9780521764612. [Google Scholar]
- Khamis, K.; Bradley, C.; Hannah, D.M. Understanding dissolved organic matter dynamics in urban catchments: Insights from in situ fluorescence sensor technology. Wiley Interdiscip. Rev. Water 2017, 5, e1259. [Google Scholar] [CrossRef]
- Stolpe, B.; Zhou, Z.; Guo, L.; Shiller, A.M. Colloidal size distribution of humic- and protein-like fluorescent organic matter in the northern Gulf of Mexico. Mar. Chem. 2014, 164, 25–37. [Google Scholar] [CrossRef]
- Sorensen, J.P.R.; Vivanco, A.; Ascott, M.J.; Gooddy, D.C.; Lapworth, D.J.; Read, D.S.; Rushworth, C.M.; Bucknall, J.; Herbert, K.; Karapanos, I.; et al. Online fluorescence spectroscopy for the real-time evaluation of the microbial quality of drinking water. Water Res. 2018. [Google Scholar] [CrossRef]
- Baker, A.; Inverarity, R. Protein-like fluorescence intensity as a possible tool for determining river water quality. Hydrol. Process. 2004, 18, 2927–2945. [Google Scholar] [CrossRef]
- Hudson, N.; Baker, A.; Reynolds, D.M. Fluorescence Analysis of Dissolved Organic Matter in Natural, Waste and Polluted Water—A Review. River Res. Appl. 2007, 23, 631–649. [Google Scholar] [CrossRef]
- Hudson, N.; Baker, A.; Ward, D.; Reynolds, D.M.; Brunsdon, C.; Carliell-Marquet, C.; Browning, S. Can fluorescence spectrometry be used as a surrogate for the Biochemical Oxygen Demand (BOD) test in water quality assessment? An example from South West England. Sci. Total Environ. 2008, 391, 149–158. [Google Scholar] [CrossRef] [PubMed]
- Coble, P.G. Characterization of marine and terrestrial DOM in seawater using excitation-emission matrix spectroscopy. Mar. Chem. 1996, 51, 325–346. [Google Scholar] [CrossRef]
- Reynolds, D.M. Rapid and direct determination of tryptophan in water using synchronous fluorescence spectroscopy. Water Res. 2003, 37, 3055–3060. [Google Scholar] [CrossRef]
- Bridgeman, J.; Baker, A.; Carliell-Marquet, C.; Carstea, E. Determination of changes in wastewater quality through a treatment works using fluorescence spectroscopy. Environ. Technol. 2013, 34, 3069–3077. [Google Scholar] [CrossRef] [PubMed]
- Reynolds, D.M. The differentiation of biodegradable and non-biodegradable dissolved organic matter in wastewaters using fluorescence spectroscopy. J. Chem. Technol. Biotechnol. 2002, 77, 965–972. [Google Scholar] [CrossRef]
- Carstea, E.M.; Bridgeman, J.; Baker, A.; Reynolds, D.M. Fluorescence spectroscopy for wastewater monitoring: A review. Water Res. 2016, 95, 205–219. [Google Scholar] [CrossRef] [PubMed]
- Sorensen, J.P.R.; Baker, A.; Cumberland, S.A.; Lapworth, D.J.; MacDonald, A.M.; Pedley, S.; Taylor, R.G.; Ward, J.S.T. Real-time detection of faecally contaminated drinking water with tryptophan-like fluorescence: Defining threshold values. Sci. Total Environ. 2018, 622–623, 1250–1257. [Google Scholar] [CrossRef] [PubMed]
- Sorensen, J.P.R.; Sadhu, A.; Sampath, G.; Sugden, S.; Dutta Gupta, S.; Lapworth, D.J.; Marchant, B.P.; Pedley, S. Are sanitation interventions a threat to drinking water supplies in rural India? An application of tryptophan-like fluorescence. Water Res. 2016, 88, 923–932. [Google Scholar] [CrossRef]
- APHA AWWA WEF. Standard Methods for the Examination of Water and Wastewater, 20th ed.; American Public Health Association: Washington, DC, USA, 1999. [Google Scholar]
- McKnight, D.M.; Boyer, E.W.; Westerhoff, P.K.; Doran, P.T.; Kulbe, T.; Anderson, D.T. Spectroflourometric characterization of dissolved organic matter for indication of precursor organic material and aromaticity. Limnol. Oceanogr. 2001, 46, 38–48. [Google Scholar] [CrossRef]
- Shimotori, K.; Omori, Y.; Hama, T. Bacterial production of marine humic-like fluorescent dissolved organic matter and its biogeochemical importance. Aquat. Microb. Ecol. 2009, 58, 55–66. [Google Scholar] [CrossRef]
- Mostofa, K.M.G.; Yoshioka, T.; Mottaleb, A.; Vione, D. Photobiogeochemistry of Organic Matter: Principles and Practices in Water Environments; Springer: Berlin/Heidelberg, Germany, 2013; ISBN 978-3642322228. [Google Scholar]
- Determann, S.; Lobbes, J.M.; Reuter, R.; Rullkötter, J. Ultraviolet fluorescence excitation and emission spectroscopy of marine algae and bacteria. Mar. Chem. 1998, 62, 137–156. [Google Scholar] [CrossRef]
- Baker, A. Spectrophotometric discrimination of river dissolved organic matter. Hydrol. Process. 2002, 16, 3203–3213. [Google Scholar] [CrossRef]
- Lapworth, D.J.; Kinniburgh, D.G. An R script for visualising and analysing fluorescence excitation-emission matrices (EEMs). Comput. Geosci. 2009, 35, 2160–2163. [Google Scholar] [CrossRef]
- Stedmon, C.A.; Bro, R. Characterizing dissolved organic matter fluorescence with parallel factor analysis: A tutorial. Limnol. Oceanogr. Methods 2008, 6, 572–579. [Google Scholar] [CrossRef]
- Molecular Probes Inc. LIVE/DEAD® BacLightTM Bacterial Viability Kits. Available online: https://tools.thermofisher.com/content/sfs/manuals/mp07007.pdf (accessed on 12 April 2016).
- Kramer, G.D.; Herndl, G.J. Photo- and bioreactivity of chromophoric dissolved organic matter produced by marine bacterioplankton. Aquat. Microb. Ecol. 2004, 36, 239–246. [Google Scholar] [CrossRef]
- Shimotori, K.; Watanabe, K.; Hama, T. Fluorescence characteristics of humic-like fluorescent dissolved organic matter produced by various taxa of marine bacteria. Aquat. Microb. Ecol. 2012, 65, 249–260. [Google Scholar] [CrossRef]
- Parlanti, E.; Wörz, K.; Geoffroy, L.; Lamotte, M. Dissolved organic matter fluorescence spectroscopy as a tool to estimate biological activity in a coastal zone submitted to anthropogenic inputs. Org. Geochem. 2000, 31, 1765–1781. [Google Scholar] [CrossRef]
- Cammack, W.K.L.; Kalff, J.; Prairie, Y.T.; Smith, E.M. Fluorescent dissolved organic matter in lakes: Relationships with heterotrophic metabolism. Limnol. Oceanogr. 2004, 49, 2034–2045. [Google Scholar] [CrossRef]
- Ziervogel, K.; Osburn, C.; Brym, A.; Battles, J.; Joye, S.; D’souza, N.; Montoya, J.; Passow, U.; Arnosti, C. Linking Heterotrophic Microbial Activities with Particle Characteristics in Waters of the Mississippi River Delta in the Aftermath of Hurricane Isaac. Front. Mar. Sci. 2016, 3, 8. [Google Scholar] [CrossRef]
- Cooper, K.J.; Whitaker, F.F.; Anesio, A.M.; Naish, M.; Reynolds, D.M.; Evans, E.L. Dissolved organic carbon transformations and microbial community response to variations in recharge waters in a shallow carbonate aquifer. Biogeochemistry 2016, 129, 215–234. [Google Scholar] [CrossRef]
- Ogawa, H.; Amagai, Y.; Koike, I.; Kaiser, K.; Benner, R. Production of Refractory Dissolved Organic Matter by Bakteria. Science 2001, 292, 917–920. [Google Scholar] [CrossRef] [PubMed]
- Reynolds, D.M.; Ahmad, S.R. Rapid and direct determination of wastewater BOD values using a fluorescence technique. Water Res. 1997, 31, 2012–2018. [Google Scholar] [CrossRef]
- Cutrera, G.; Manfredi, L.; Valle, C.E.; González, J.F. On the determination of the kinetic parameters for the BOD test. Water SA 1999, 25, 377–380. [Google Scholar]
- Khamis, K.; Sorensen, J.P.R.; Bradley, C.; Hannah, D.M.; Lapworth, D.J.; Stevens, R. In situ tryptophan-like fluorometers: Assessing turbidity and temperature effects for freshwater applications. Environ. Sci. Process. Impacts 2015, 17, 740–752. [Google Scholar] [CrossRef] [PubMed]
- Reynolds, D.M.; Ahmad, S.R. The effect of metal ions on the fluorescence of sewage wastewater. Water Res. 1995, 29, 2214–2216. [Google Scholar] [CrossRef]
- Tanaka, K.; Kuma, K.; Hamasaki, K.; Yamashita, Y. Accumulation of humic-like fluorescent dissolved organic matter in the Japan Sea. Sci. Rep. 2014, 4, 5292. [Google Scholar] [CrossRef]
- Jiao, N.; Herndl, G.J.; Hansell, D.A.; Benner, R.; Kattner, G.; Wilhelm, S.W.; Kirchman, D.L.; Weinbauer, M.G.; Luo, T.; Chen, F.; Azam, F. Microbial production of recalcitrant dissolved organic matter: Long-term carbon storage in the global ocean. Nat. Rev. Microbiol. 2010, 8, 593. [Google Scholar] [CrossRef]
- Fukuzaki, K.; Imai, I.; Fukushima, K.; Ishii, K.I.; Sawayama, S.; Yoshioka, T. Fluorescent characteristics of dissolved organic matter produced by bloom-forming coastal phytoplankton. J. Plankton Res. 2014, 36, 685–694. [Google Scholar] [CrossRef]
- Guillemette, F.; del Giorgio, P.A. Simultaneous consumption and production of fluorescent dissolved organic matter by lake bacterioplankton. Environ. Microbiol. 2012, 14, 1432–1443. [Google Scholar] [CrossRef]
- Timko, S.A.; Maydanov, A.; Pittelli, S.L.; Conte, M.H.; Cooper, W.J.; Koch, B.P.; Schmitt-Kopplin, P.; Gonsior, M. Depth-dependent photodegradation of marine dissolved organic matter. Front. Mar. Sci. 2015, 2, 66. [Google Scholar] [CrossRef]
- Kallenbach, C.M.; Frey, S.D.; Grandy, A.S. Direct evidence for microbial-derived soil organic matter formation and its ecophysiological controls. Nat. Commun. 2016, 7, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.; Sultana, C.M.; Collins, D.B.; Santander, M.V.; Axson, J.L.; Malfatti, F.; Cornwell, G.C.; Grandquist, J.R.; Deane, G.B.; Stokes, M.D.; et al. Advancing Model Systems for Fundamental Laboratory Studies of Sea Spray Aerosol Using the Microbial Loop. J. Phys. Chem. A 2015, 119, 8860–8870. [Google Scholar] [CrossRef] [PubMed]
- Asmala, E.; Autio, R.; Kaartokallio, H.; Stedmon, C.A.; Thomas, D.N. Processing of humic-rich riverine dissolved organic matter by estuarine bacteria: Effects of predegradation and inorganic nutrients. Aquat. Sci. 2014, 76, 451–463. [Google Scholar] [CrossRef]
- Jørgensen, L.; Stedmon, C.A.; Granskog, M.A.; Middelboe, M. Tracing the long-term microbial production of recalcitrant fluorescent dissolved organic matter in seawater. Geophys. Res. Lett. 2014, 41, 2481–2488. [Google Scholar] [CrossRef]
- Baker, A.; Cumberland, S.A.; Bradley, C.; Buckley, C.; Bridgeman, J. To what extent can portable fluorescence spectroscopy be used in the real-time assessment of microbial water quality? Sci. Total Environ. 2015, 532, 14–19. [Google Scholar] [CrossRef] [PubMed]
- Sommerwerk, N.; Hein, T.; Schneider-Jacoby, M.; Baumgartner, C.; Ostojić, A.; Siber, R.; Bloesch, J.; Paunović, M.; Tockner, K. Chapter 3—The Danube River Basin. In Rivers of Europe; Tockner, K., Uehlinger, U., Robinson, C.T., Eds.; Academic Press: London, UK, 2009; pp. 59–112. ISBN 978-0-12-369449-2. [Google Scholar]
- Sorensen, J.P.R.; Lapworth, D.J.; Marchant, B.P.; Nkhuwa, D.C.W.; Pedley, S.; Stuart, M.E.; Bell, R.A.; Chirwa, M.; Kabika, J.; Liemisa, M.; et al. In situ tryptophan-like fluorescence: A real-time indicator of faecal contamination in drinking water supplies. Water Res. 2015, 81, 38–46. [Google Scholar] [CrossRef]
- Sorensen, J.P.R.; Lapworth, D.J.; Read, D.S.; Nkhuwa, D.C.W.; Bell, R.A.; Chibesa, M.; Chirwa, M.; Kabika, J.; Liemisa, M.; Pedley, S. Tracing enteric pathogen contamination in sub-Saharan African groundwater. Sci. Total Environ. 2015, 538, 888–895. [Google Scholar] [CrossRef]
- Cumberland, S.; Bridgeman, J.; Baker, A.; Sterling, M.; Ward, D. Fluorescence spectroscopy as a tool for determining microbial quality in potable water applications. Environ. Technol. 2012, 33, 687–693. [Google Scholar] [CrossRef]
- Blaen, P.J.; Khamis, K.; Lloyd, C.E.M.; Bradley, C.; Hannah, D.; Krause, S. Real-time monitoring of nutrients and dissolved organic matter in rivers: Capturing event dynamics, technological opportunities and future directions. Sci. Total Environ. 2016, 569–570, 647–660. [Google Scholar] [CrossRef] [PubMed]
- Asmala, E.; Kaartokallio, H.; Carstensen, J.; Thomas, D.N. Variation in Riverine Inputs Affect Dissolved Organic Matter Characteristics throughout the Estuarine Gradient. Front. Mar. Sci. 2016, 2, 125. [Google Scholar] [CrossRef]
- Makarewicz, A.; Kowalczuk, P.; Sagan, S.; Granskog, M.A.; Pavlov, A.K.; Zdun, A.; Borzycka, K.; Zabłocka, M. Characteristics of Chromophoric and Fluorescent Dissolved Organic Matter in the Nordic Seas. Ocean Sci. Discuss. 2018, 14, 543–562. [Google Scholar] [CrossRef]


| PARAFAC Component | Named Fluorescence Peak * | λex/λem (nm) | Description | Present in Sample Type | ||
|---|---|---|---|---|---|---|
| EW | SW | SSW | ||||
| 1 | T Tryptophan-like | 275/340 | Autochthonous peak, described unanimously in the literature as microbially-derived and associated with protein presence. | ✔ | ✔ | ✔ |
| 2 | M AM Humic-like (Marine-like) | 300/410 240/410 | Biologically or photochemically degraded terrestrial humic-like, sometimes referred to as ‘marine-like’ OM [16]. | ✔ | ✔ | ✔ |
| 3 | B Tyrosine-like | 265/290 | Autochthonous peak, described as microbially-derived and associated with amino-acid and protein presence. | ✔ | ✔ | |
| 4 | C+ C Humic-like | 260/490 390/490 260/430 360/430 | Double maxima components of terrestrial allochthonous higher molecular weight aromatic compounds, noted as humic-like fluorescence. | ✔ | ✔ | ✔ |
| 5 | B | 250/300 | Region similar to Peak B, associated with autochthonous production or possible photodegraded OM. Also similar to BTEX fluorescence signature. | ✔ | ✔ | ✔ |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fox, B.G.; Thorn, R.M.S.; Anesio, A.M.; Cox, T.; Attridge, J.W.; Reynolds, D.M. Microbial Processing and Production of Aquatic Fluorescent Organic Matter in a Model Freshwater System. Water 2019, 11, 10. https://doi.org/10.3390/w11010010
Fox BG, Thorn RMS, Anesio AM, Cox T, Attridge JW, Reynolds DM. Microbial Processing and Production of Aquatic Fluorescent Organic Matter in a Model Freshwater System. Water. 2019; 11(1):10. https://doi.org/10.3390/w11010010
Chicago/Turabian StyleFox, Bethany G., Robin M.S. Thorn, Alexandre M. Anesio, Timothy Cox, John W. Attridge, and Darren M. Reynolds. 2019. "Microbial Processing and Production of Aquatic Fluorescent Organic Matter in a Model Freshwater System" Water 11, no. 1: 10. https://doi.org/10.3390/w11010010
APA StyleFox, B. G., Thorn, R. M. S., Anesio, A. M., Cox, T., Attridge, J. W., & Reynolds, D. M. (2019). Microbial Processing and Production of Aquatic Fluorescent Organic Matter in a Model Freshwater System. Water, 11(1), 10. https://doi.org/10.3390/w11010010