Inhibition of Cationic Polymer-Induced Colloid Flocculation by Polyacrylic Acid
Abstract
1. Introduction
2. Materials and Methods
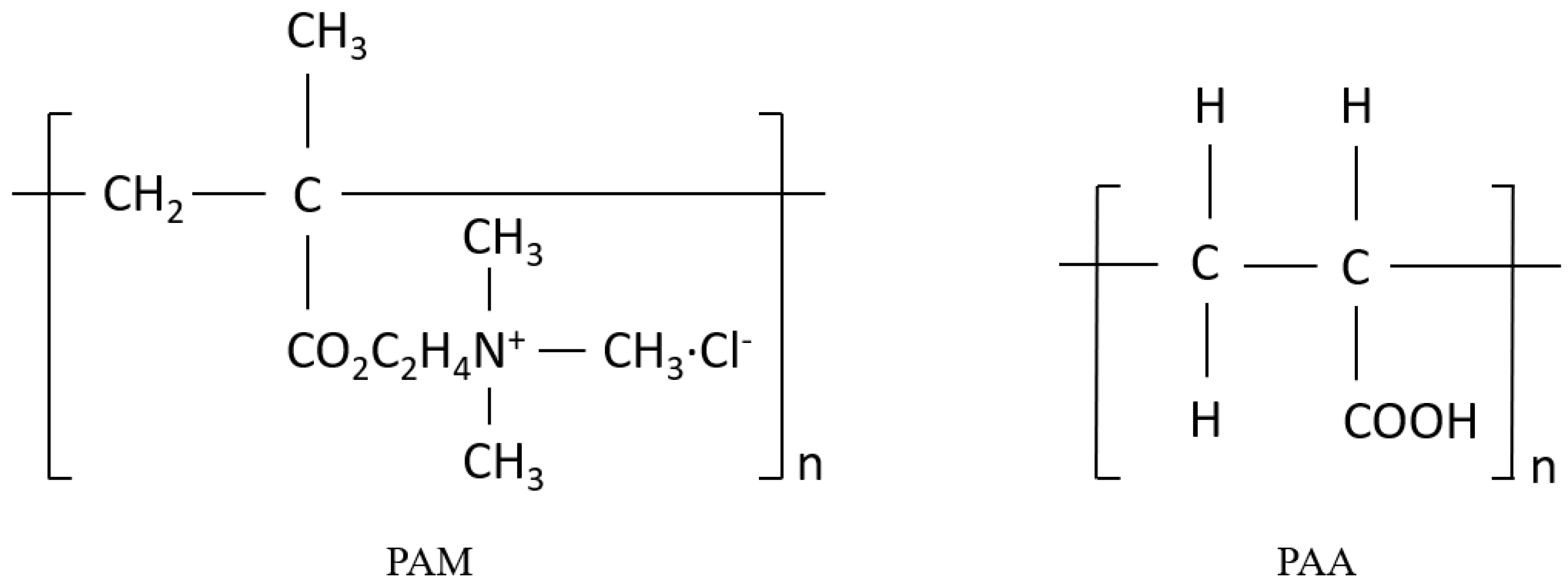
2.1. Materials
2.2. Analytical Principle
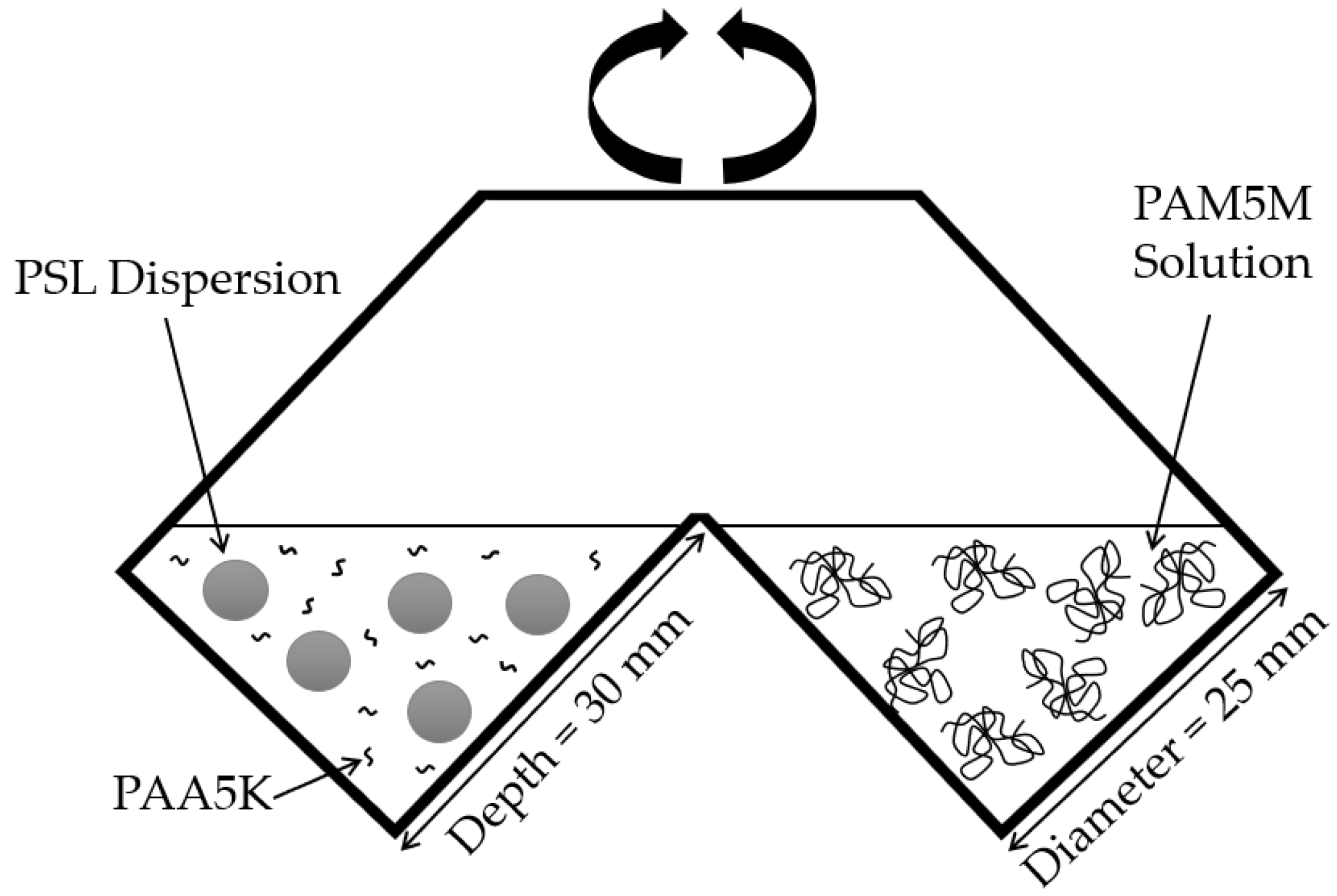
2.3. Particle Aggregation Analysis
2.4. Viscosity Measurements
3. Results
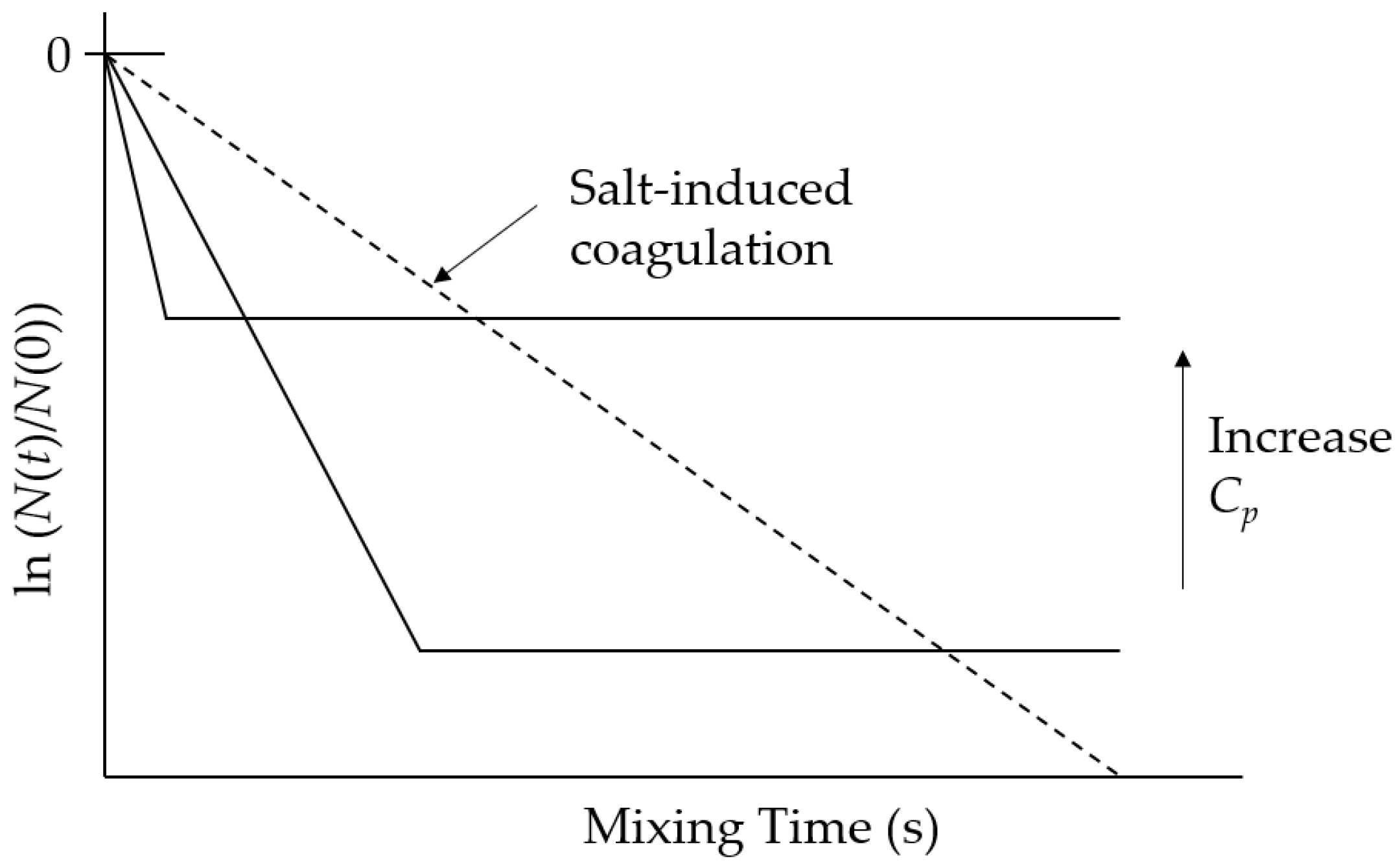
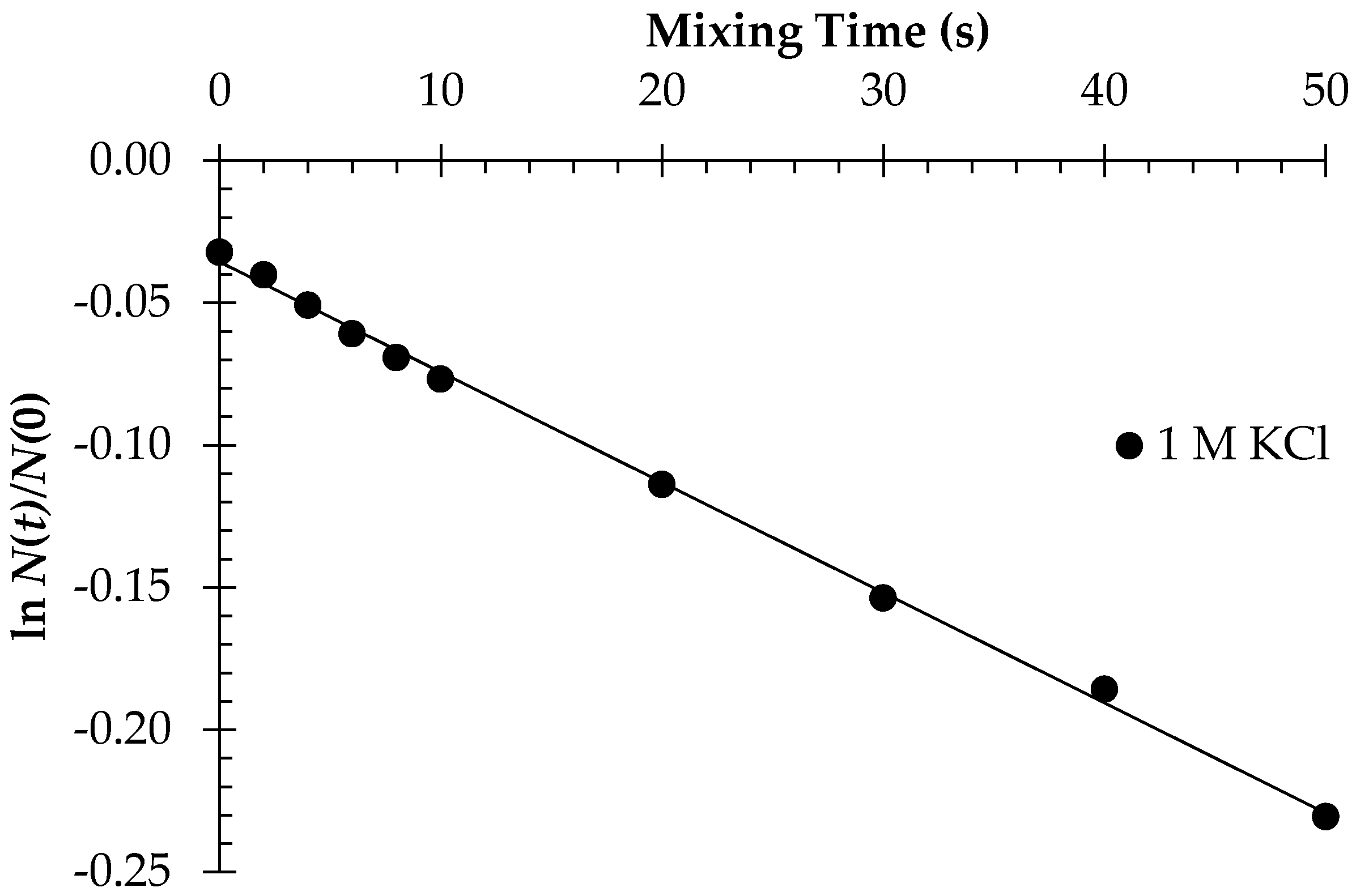
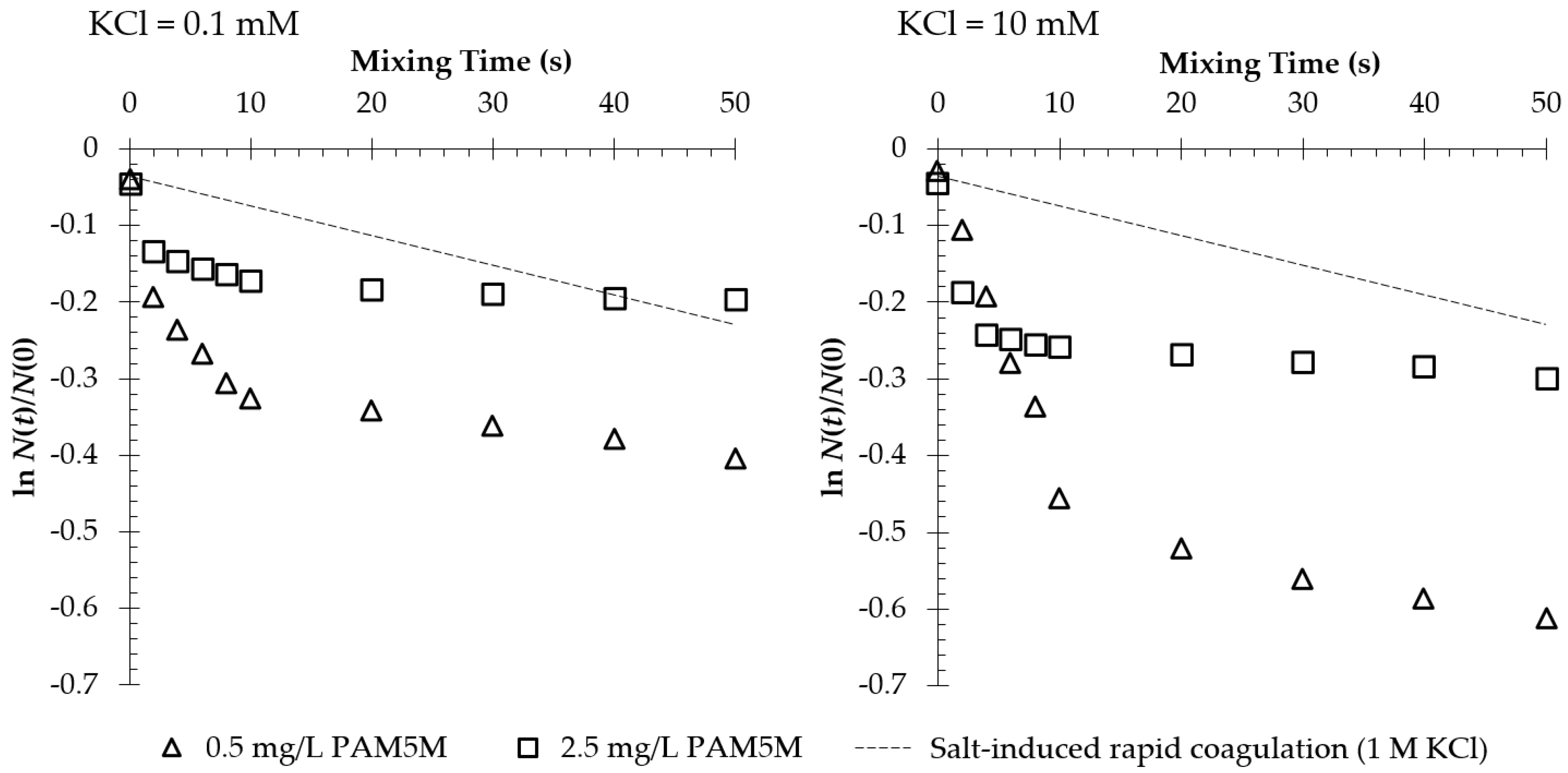
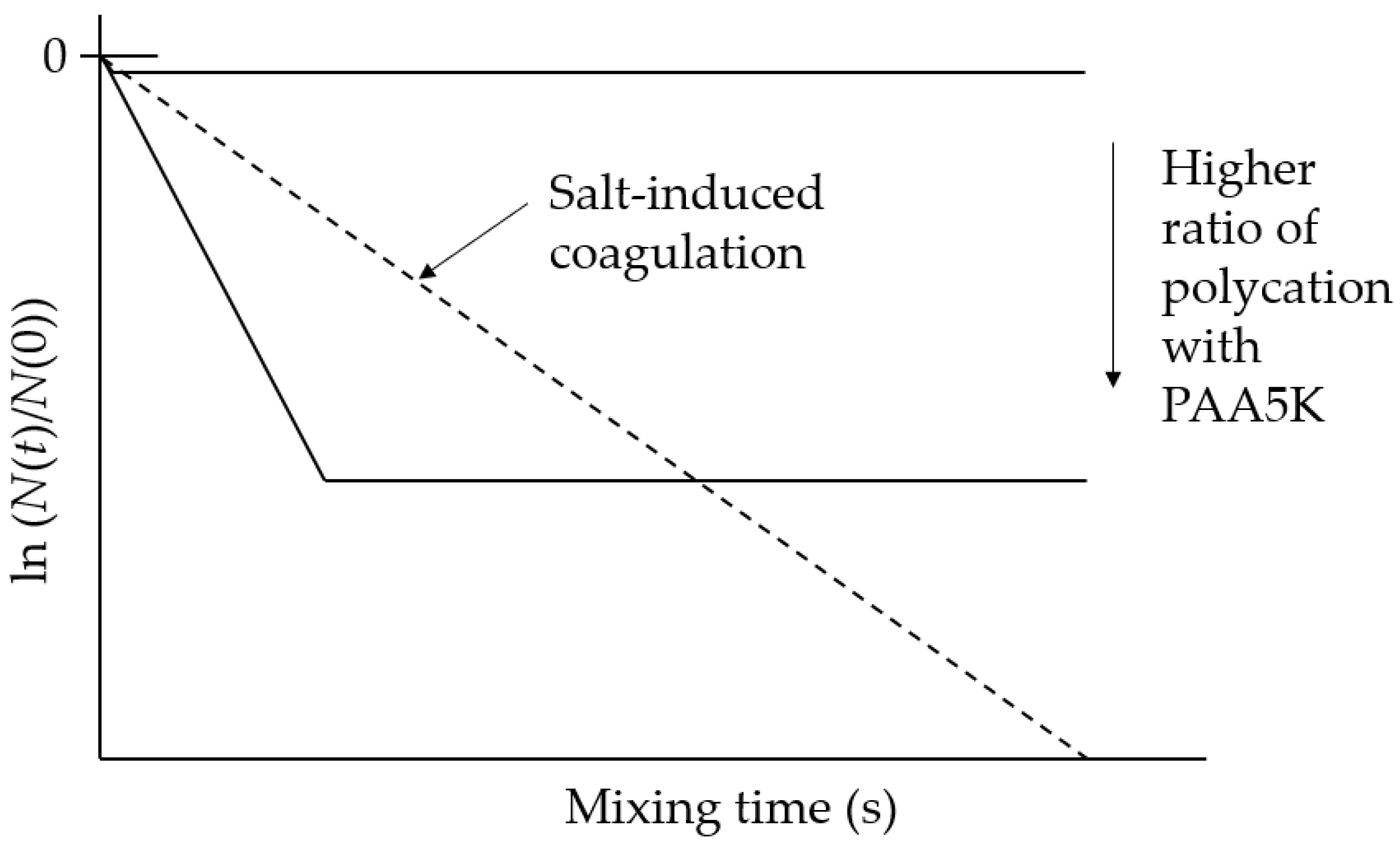
3.1. Polycation-Induced Flocculation
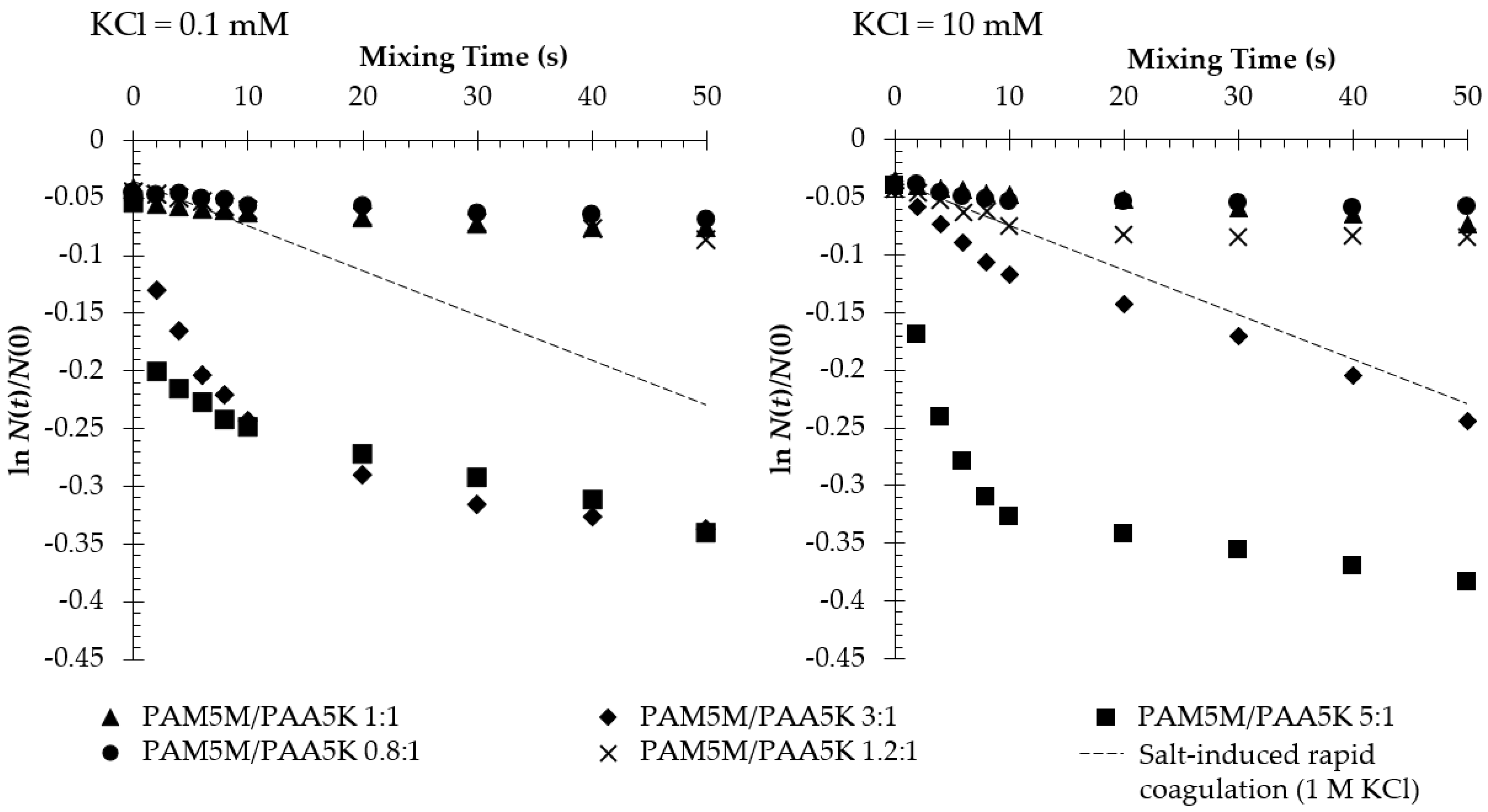
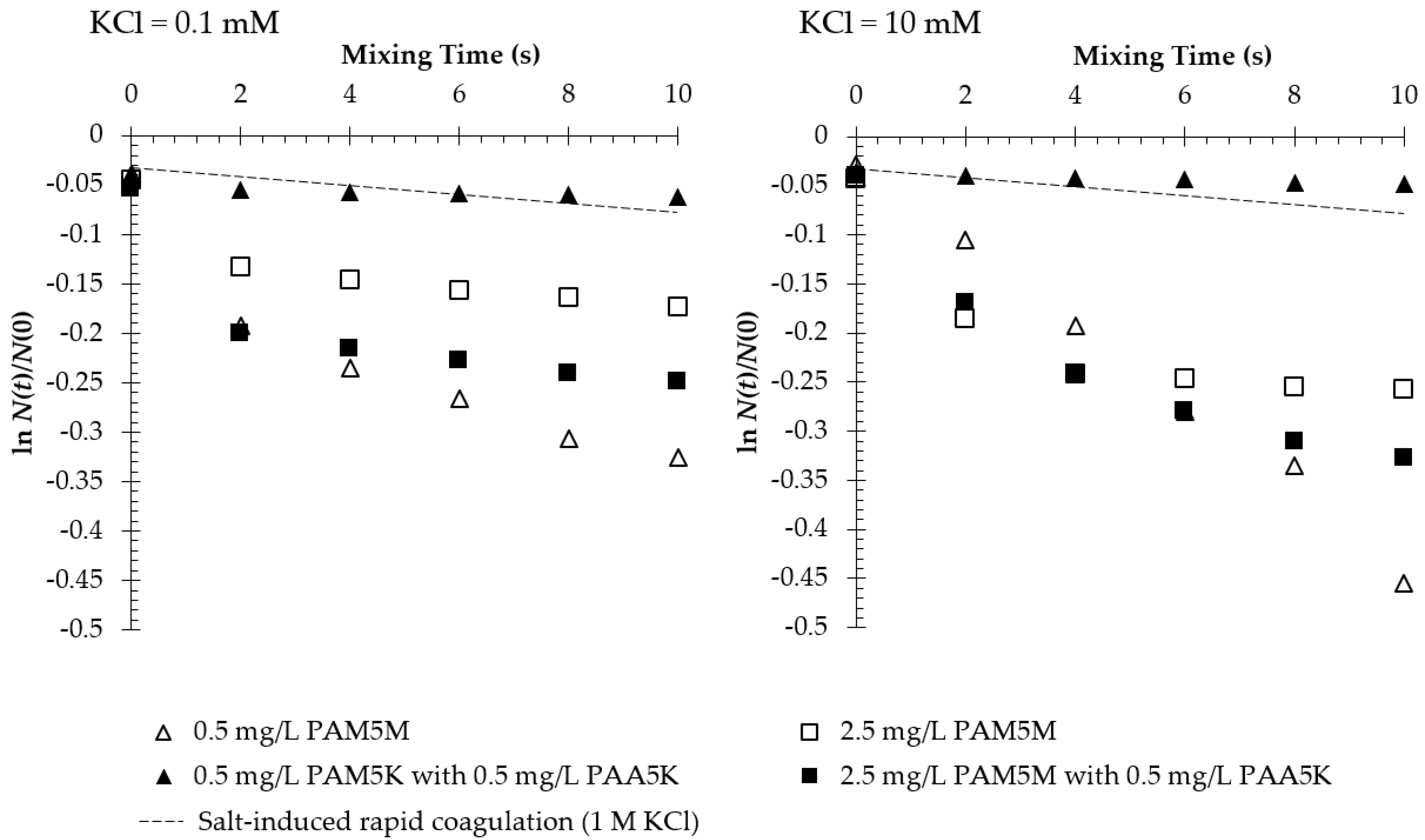
3.2. Flocculation in the Presence of Two Oppositely Charged Polymers
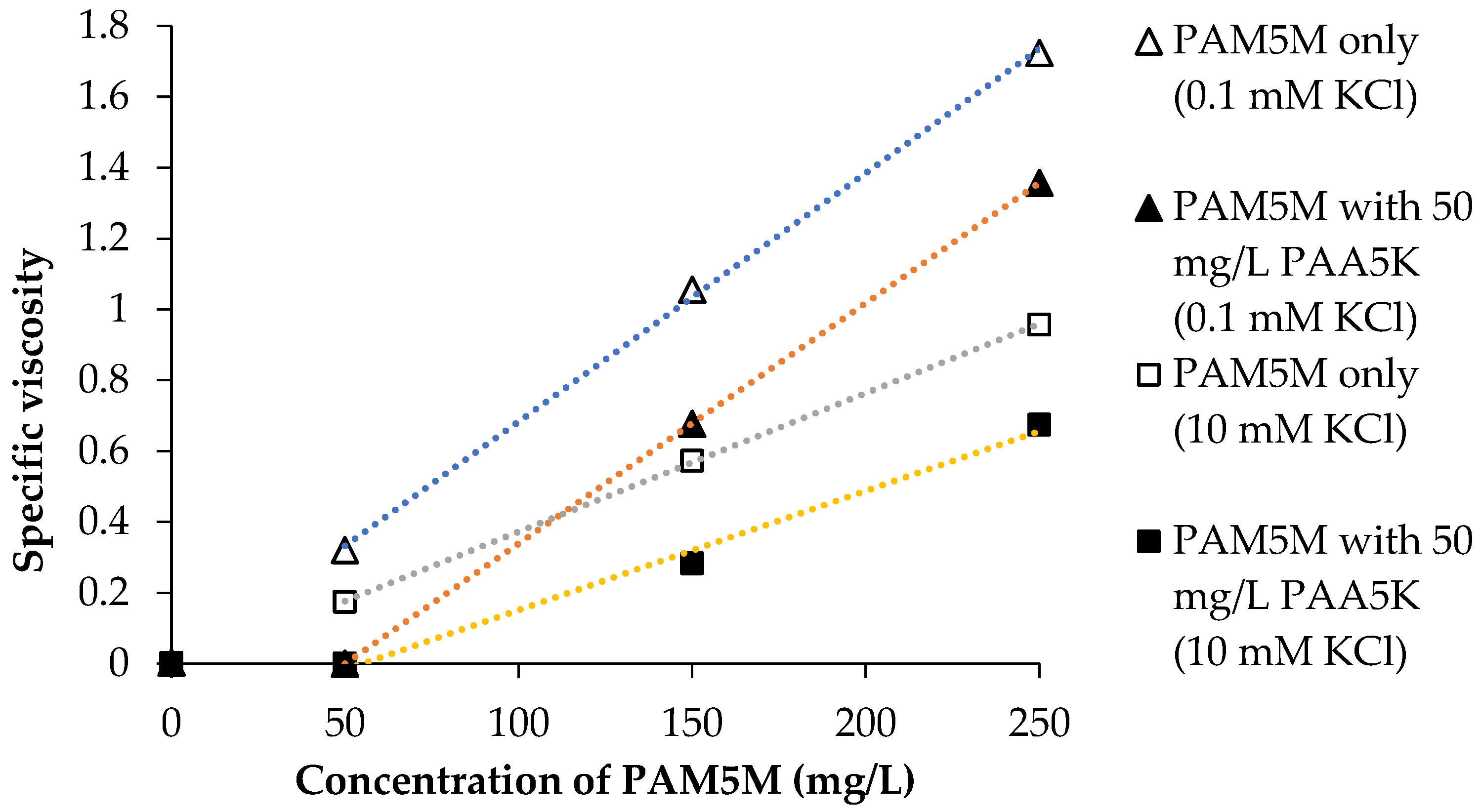
3.3. Reduction of PAM5M Viscosity
4. Discussion
4.1. Enhancement of Initial Flocculation Rate by PAM5M
4.2. Shrinkage of PAM5M in the Presence of PAA5K
4.3. Polyion Complex Formation
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Kadooka, H.; Kiso, Y.; Goto, S.; Tanaka, T.; Jami, M.S.; Iwata, M. Flocculation behavior of colloidal suspension by use of inorganic and polymer flocculants in powder form. J. Water Process. Eng. 2017, 18, 169–175. [Google Scholar] [CrossRef]
- Zezin, A.; Mikheikin, S.; Rogacheva, V.; Zansokhova, M.; Sybachin, A.; Yaroslavov, A. Polymeric stabilizers for protection of soil and ground against wind and water erosion. Adv. Colloid Interface Sci. 2015, 226, 17–23. [Google Scholar] [CrossRef] [PubMed]
- Farrow, J.B.; Swift, J.D. A new procedure for assessing the performance of flocculants. Int. J. Miner. Process. 1996, 46, 263–275. [Google Scholar] [CrossRef]
- Rojas, J.; Hubbe, M.A. The dispersion science of papermaking. J. Dispers. Sci. Technol. 2005, 25, 713–732. [Google Scholar] [CrossRef]
- Bolto, B.; Gregory, J. Organic polyelectrolytes in water treatment. Water Res. 2007, 41, 2301–2324. [Google Scholar] [CrossRef] [PubMed]
- Gregory, J. Rates of flocculation of latex particles by cationic polymers. J. Colloid Interface Sci. 1973, 42, 448–456. [Google Scholar] [CrossRef]
- Adachi, Y.; Feng, L.; Kobayashi, M. Kinetics of flocculation of polystyrene latex particles in the mixing flow induced with high charge density polycation near the isoelectric point. Colloids Surf. A Physicochem. Eng. Asp. 2015, 471, 38–44. [Google Scholar] [CrossRef]
- Wang, T.K.; Audebert, R. Flocculation mechanisms of a silica suspension by some weakly cationic polyelectrolytes. J. Colloid Interface Sci. 1987, 119, 459–465. [Google Scholar] [CrossRef]
- Zhou, Y.; Franks, G.V. Flocculation mechanism induced by cationic polymers investigated by light scattering. Langmuir 2006, 22, 6775–6786. [Google Scholar] [CrossRef] [PubMed]
- Fleer, G.; Stuart, M.C.; Scheutjens, J.; Cosgrove, T.; Vincent, B. Polymers at Interfaces, 1st ed.; Chapman Hall: London, UK, 1993. [Google Scholar]
- De Gennes, P.G. Polymers at an interface; a simplified view. Adv. Colloid Interface Sci. 1987, 27, 189–209. [Google Scholar] [CrossRef]
- Netz, R.R.; Andelman, D. Neutral and charged polymers at interfaces. Phys. Rep. 2003, 380, 1–95. [Google Scholar] [CrossRef]
- Gregory, J. Polymer adsorption and flocculation in sheared suspensions. Colloids Surf. 1988, 31, 231–253. [Google Scholar] [CrossRef]
- Peng, P.; Garnier, G. Effect of cationic polyacrylamide adsorption kinetics and ionic strength on precipitated calcium carbonate flocculation. Langmuir 2010, 26, 16949–16957. [Google Scholar] [CrossRef] [PubMed]
- Adachi, Y. Dynamic aspects of coagulation and flocculation. Adv. Colloid Interface Sci. 1996, 56, 1–31. [Google Scholar] [CrossRef]
- Adachi, Y.; Stuart, M.A.C.; Fokkink, R. Kinetics of turbulent coagulation studied by means of end-over-end rotation. J. Colloid Interface Sci. 1994, 165, 310–317. [Google Scholar] [CrossRef]
- Matsumoto, T.; Adachi, Y. Effect of ionic strength on the initial dynamics of flocculation of polystyrene latex with polyelectrolyte. J. Colloid Interface Sci. 1998, 204, 328–335. [Google Scholar] [CrossRef] [PubMed]
- Adachi, Y.; Wada, T. Initial stage dynamics of bridging flocculation of polystyrene latex spheres with polyethylene oxide. J. Colloid Interface Sci. 2000, 229, 148–154. [Google Scholar] [CrossRef] [PubMed]
- Aoki, K.; Adachi, Y. Kinetics of polyelectrolyte adsorption onto polystyrene latex particle studied using electrophoresis: Effects of molecular weight and ionic strength. J. Colloid Interface Sci. 2006, 300, 69–77. [Google Scholar] [CrossRef] [PubMed]
- Divakaran, R.; Sivasankara Pillai, V.N. Flocculation of river silt using chitosan. Water Res. 2002, 36, 2414–2418. [Google Scholar] [CrossRef]
- Ashmore, M.; Hearn, J.; Karpowicz, F. Flocculation of latex particles of varying surface charge densities by chitosans. Langmuir 2001, 17, 1069–1073. [Google Scholar] [CrossRef]
- Szilagyi, I.; Trefalt, G.; Tiraferri, A.; Maroni, P.; Borkovec, M. Polyelectrolyte adsorption, interparticle forces, and colloidal aggregation. Soft Matter 2014, 10, 2479–2502. [Google Scholar] [CrossRef] [PubMed]
- Kam, S.; Gregory, J. The interaction of humic substances with cationic polyelectrolytes. Water Res 2001, 35, 3557–3566. [Google Scholar] [CrossRef]
- Wang, H.; Adeleye, A.S.; Huang, Y.; Li, F.; Keller, A.A. Heteroaggregation of nanoparticles with biocolloids and geocolloids. Adv. Colloid Interface Sci. 2015, 226, 24–36. [Google Scholar] [CrossRef] [PubMed]
- Nikolaou, A.D.; Golfinopoulos, S.K.; Lekkas, T.D.; Kostopoulou, M.N. DBP Levels in Chlorinated Drinking Water: Effect of Humic Substances. Environ. Monit. Assess. 2004, 93, 301–319. [Google Scholar] [CrossRef] [PubMed]
- Matilainen, A.; Vepsäläinen, M.; Sillanpää, M. Natural organic matter removal by coagulation during drinking water treatment: A review. Adv. Colloid Interface Sci. 2010, 159, 189–197. [Google Scholar] [CrossRef] [PubMed]
- Pelton, R.; Lawrence, D. The influence of surfactants on latex flocculation with poly (diallyldimethyl ammonium chloride). Colloid Polym. Sci. 1989, 267, 907–914. [Google Scholar] [CrossRef]
- Narkis, N.; Rebhun, M. Flocculation in presence of organic macromolecules of natural water and secondary effluents. Water Sci. Technol. 1997, 36, 85–91. [Google Scholar] [CrossRef]
- Lurie, M.; Rebhun, M. Effect of properties of polyelectrolytes on their interaction with particulates and soluble organics. Water Sci. Technol. 1997, 36, 93–101. [Google Scholar] [CrossRef]
- Miyajima, T. Metal complexation in polyelectrolyte solutions. In Physical Chemistry of Polyelectrolytes, 1st ed.; Radeva, T., Ed.; CRC Press: Boca Raton, FL, USA, 2001; Volume 99, pp. 829–874. [Google Scholar]
- Roger, G.M.; Durand-Vidal, S.; Bernard, O.; Mériguet, G.; Altmann, S.; Turq, P. Characterization of humic substances and polyacrylic acid: A high precision conductimetry study. Colloids Surf. A Physicochem. Eng. Asp. 2010, 356, 51–57. [Google Scholar] [CrossRef]
- Avena, M.J.; Vermeer, A.W.P.; Koopal, L.K. Volume and structure of humic acids studied by viscometry pH and electrolyte concentration effects. Colloids Surf. A Physicochem. Eng. Asp. 1999, 151, 213–224. [Google Scholar] [CrossRef]
- Kodama, H.; Miyajima, T.; Mori, M.; Takahashi, M.; Nishimura, H.; Ishiguro, S. A unified analytical treatment of the acid-dissociation equilibria of weakly acidic linear polyelectrolytes and the conjugate acids of weakly basic linear polyelectrolytes. Colloid Polym. Sci. 1997, 275, 938–945. [Google Scholar] [CrossRef]
- Van de Ven, T.G.M.; Mason, S.G. The microrheology of colloidal dispersions. Colloid Polym. Sci. 1977, 255, 794–804. [Google Scholar] [CrossRef]
- Adachi, Y.; Matsumoto, T. Dynamics of initial stage flocculation of polystyrene latex spheres with polyelectrolytes. Colloids Surf. A Physicochem. Eng. Asp. 1996, 113, 229–236. [Google Scholar] [CrossRef]
- Takahashi, T.; Takayama, K.; Machida, Y.; Nagai, T. Characteristics of polyion complexes of chitosan with sodium alginate and sodium polyacrylate. Int. J. Pharm. 1990, 61, 35–41. [Google Scholar] [CrossRef]
- Chen, S.; Zhang, Q.; Nakamoto, T.; Kawazoe, N.; Chen, G. Highly active porous scaffolds of collagen and hyaluronic acid prepared by suppression of polyion complex formation. J. Mater. Chem. B 2014, 2, 5612–5619. [Google Scholar] [CrossRef]
- Izumrudov, V.A.; Sybachin, A.V. Phase separation in solutions of polyelectrolyte complexes: The decisive effect of a host polyion. Polym. Sci. Ser. A 2006, 48, 1098–1104. [Google Scholar] [CrossRef]

| Sample | ap (nm) | |
|---|---|---|
| 10 mM KCl | 0.1 mM KCl | |
| PAA5K | 6.4 | 7.2 |
| PAM5M | 277 | 341 |
| Sample | ξ | |
|---|---|---|
| 10 mM KCl | 0.1 mM KCl | |
| 0.5 mg/L PAM5M | 8.76 | 10.54 |
| 2.5 mg/L PAM5M | 10.64 | N/A |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lim, V.H.; Yamashita, Y.; Doan, Y.T.H.; Adachi, Y. Inhibition of Cationic Polymer-Induced Colloid Flocculation by Polyacrylic Acid. Water 2018, 10, 1215. https://doi.org/10.3390/w10091215
Lim VH, Yamashita Y, Doan YTH, Adachi Y. Inhibition of Cationic Polymer-Induced Colloid Flocculation by Polyacrylic Acid. Water. 2018; 10(9):1215. https://doi.org/10.3390/w10091215
Chicago/Turabian StyleLim, Voon Huey, Yuji Yamashita, Yen Thi Hai Doan, and Yasuhisa Adachi. 2018. "Inhibition of Cationic Polymer-Induced Colloid Flocculation by Polyacrylic Acid" Water 10, no. 9: 1215. https://doi.org/10.3390/w10091215
APA StyleLim, V. H., Yamashita, Y., Doan, Y. T. H., & Adachi, Y. (2018). Inhibition of Cationic Polymer-Induced Colloid Flocculation by Polyacrylic Acid. Water, 10(9), 1215. https://doi.org/10.3390/w10091215







