Evaluation of Key Antimicrobial Properties of Moringa oleifera in Relation to Its Use as a Hand-Washing Product
Abstract
1. Introduction
2. Materials and Methods
2.1. Antibacterial Properties of Minimally Processed M. oleifera Against Faecal Indicators Bacteria in Solution
2.1.1. Bacterial Strains
2.1.2. M. oleifera Preparations
2.1.3. Set-up and Estimation of Colony Forming Units (cfu/mL)
2.1.4. Quality Control
2.2. Hand-Washing Trial with Healthy Volunteers
2.2.1. Study Design
2.2.2. Subjects
2.2.3. M. oleifera Preparations
2.2.4. Contamination Procedure
2.2.5. Pre-Value Estimation
2.2.6. Hand-Washing Procedure
2.2.7. Post-Value Estimation
2.3. Bactericidal or Bacteriostatic Properties of M. oleifera in Potentially Reusable Aqueous Solution
2.4. Statistical Analysis
2.4.1. Die-Off Studies
2.4.2. Hand-Washing Trial
2.4.3. Rinse Water Bacterial Die-Off
3. Results
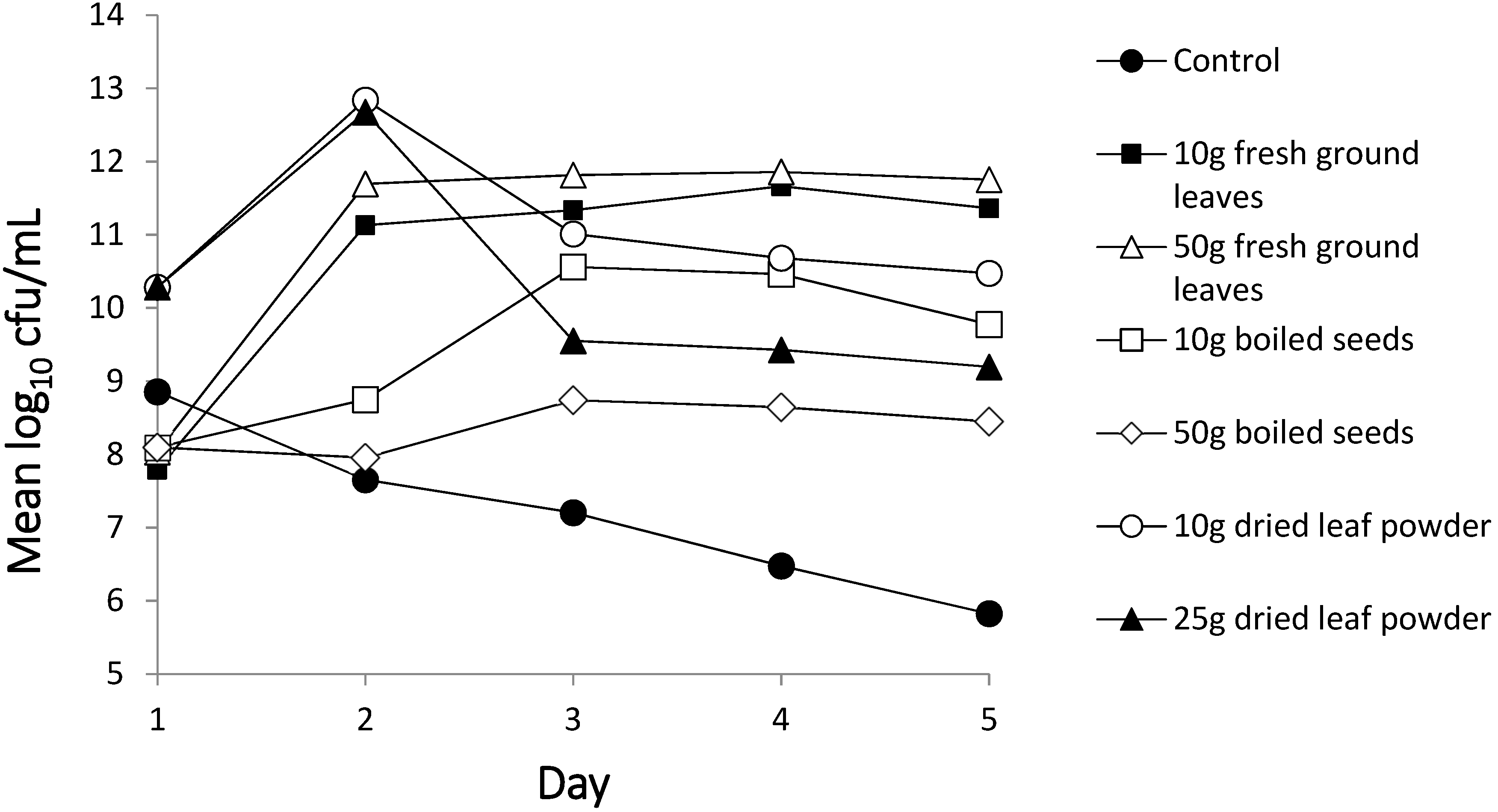
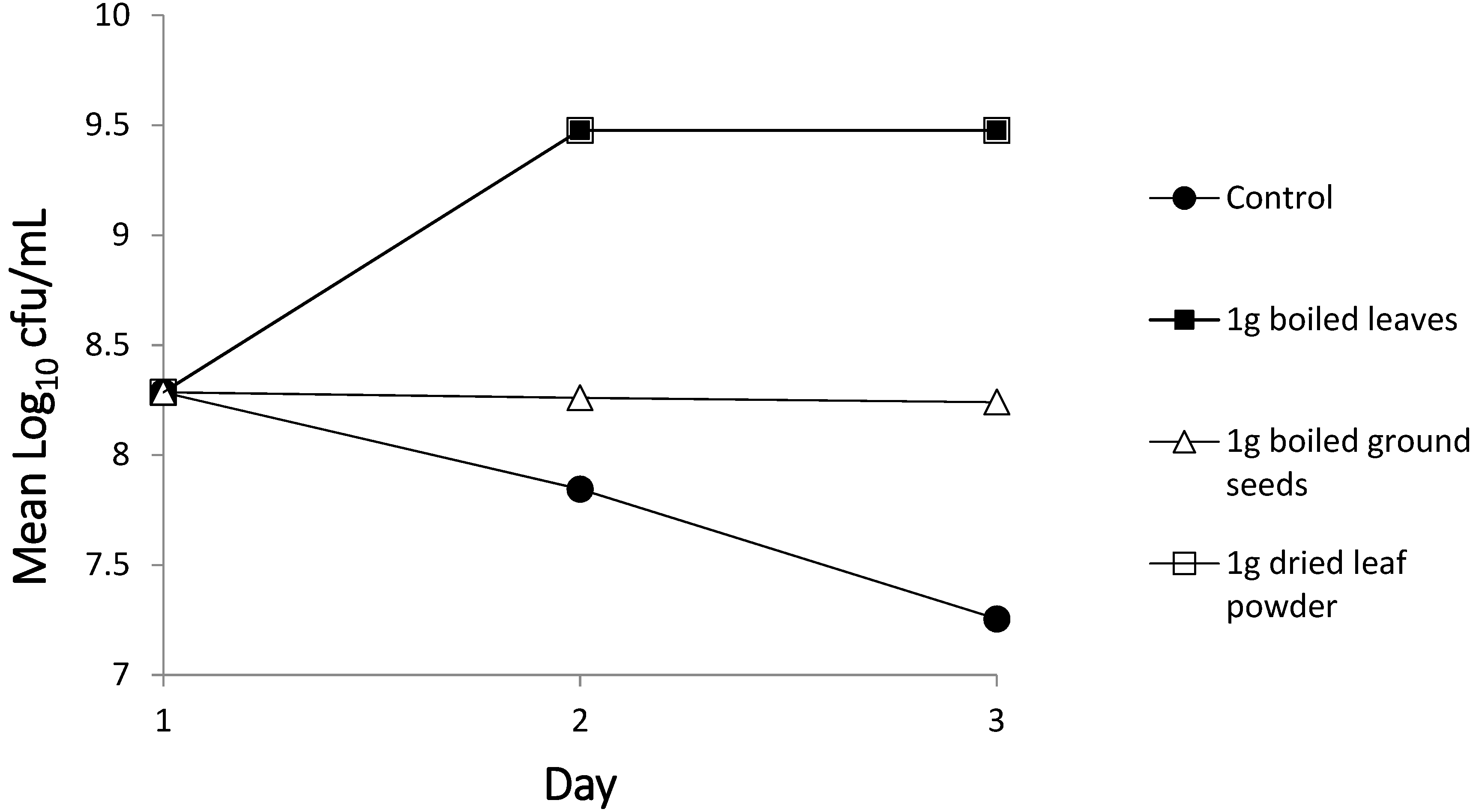
3.1. Die-Off Studies
3.1.1. Experiment to Determine Extent of M. oleifera Contamination
3.1.2. Antibacterial Activity of M. oleifera Against a Gram-Positive Bacterium
3.2. Hand-Washing Trial
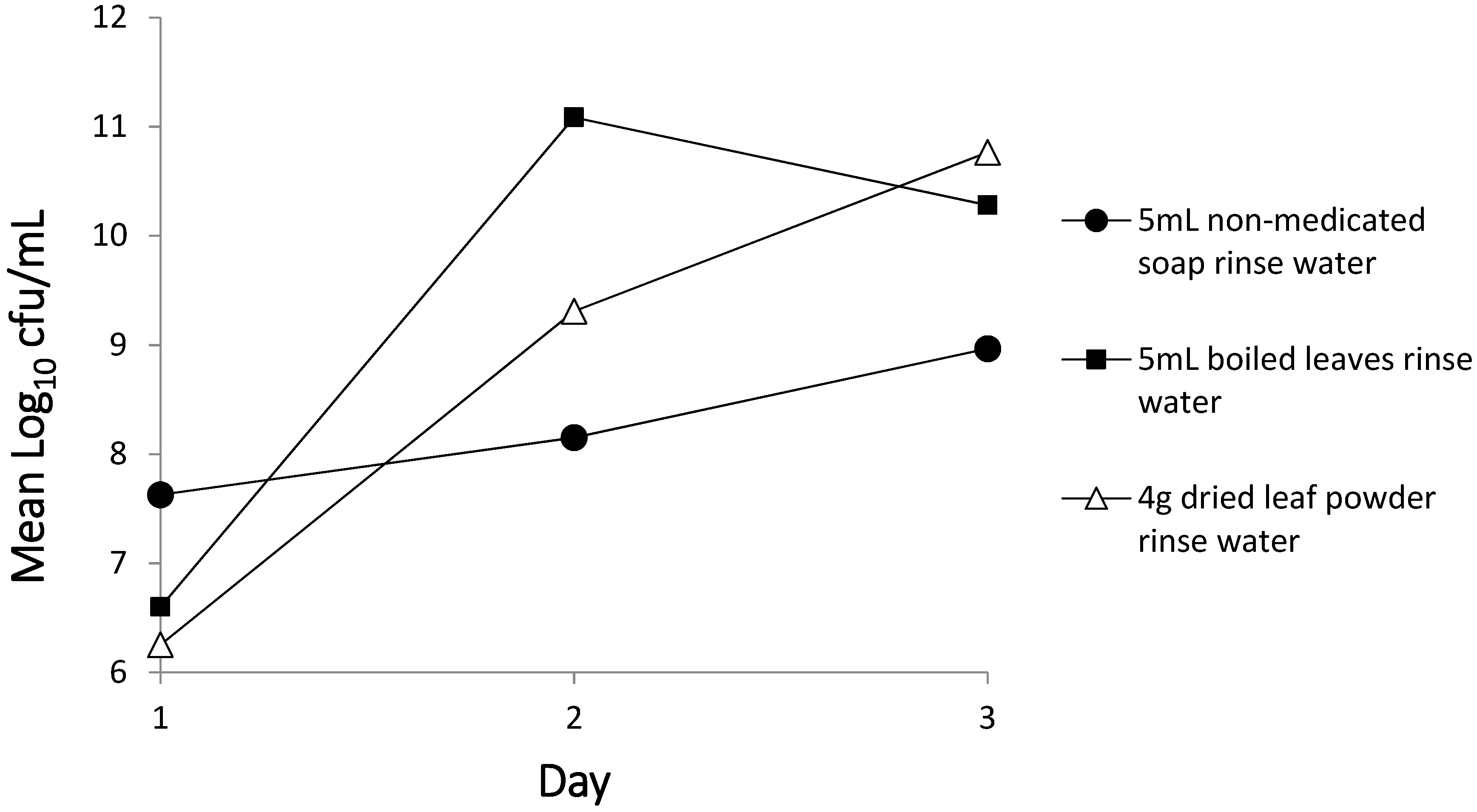
3.3. Rinse Water Collection
4. Discussion
4.1. Bacterial Die-Off Studies: Antibacterial Properties of M. oleifera Against Faecal Indicators Bacteria in Solution
4.2. Efficacy of M. oleifera as a Hand-Washing Product
4.3. Bacterial Die-Off in Rinse Water
4.4. Contamination of M. oleifera
4.5. Limitations
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Liu, L.; Johnson, H.L.; Cousens, S.; Perin, J.; Scott, S.; Lawn, J.E.; Rudan, I.; Campbell, H.; Cibulskis, R.; Li, M.; et al. Global, regional, and national causes of child mortality: An updated systematic analysis for 2010 with time trends since 2000. Lancet 2012, 379, 2151–2161. [Google Scholar] [CrossRef]
- Walker, C.L.F.; Perin, J.; Aryee, M.J.; Boschi-Pinto, C.; Black, R.E. Diarrhea incidence in low- and middle-income countries in 1990 and 2010: A systematic review. BMC Public Health 2012, 12, 220. [Google Scholar]
- Kotloff, K.L.; Nataro, J.P.; Blackwelder, W.C.; Nasrin, D.; Farag, T.H.; Panchalingam, S.; Wu, Y.; Saw, S.O.; Sur, D.; Breiman, R.F.; et al. Burden and aetiology of diarrhoeal disease in infants and young children in developing countries (the Global Enteric Multicenter Study, GEMS): A prospective, case-control study. Lancet 2013, 382, 209–222. [Google Scholar] [CrossRef]
- Baker, K.K.; O’Reilly, C.E.; Levine, M.M.; Kotloff, K.L.; Nataro, J.P.; Ayers, T.L.; Farag, T.H.; Nasrin, D.; Blackwelder, W.C.; Wu, Y.; et al. Sanitation and Hygiene-Specific Risk Factors for Moderate-to-Severe Diarrhea in Young Children in the Global Enteric Multicenter Study, 2007–2011: Case-Control Study. PLoS Med. 2016, 13, e1002010. [Google Scholar] [CrossRef] [PubMed]
- Humanitarian Innovation Fund (HIF). WASH in Emergencies Problem Exploration Report: Handwashing; HIF: London, UK, 2016. [Google Scholar]
- WHO|Diarrhoeal Disease. Available online: http://www.who.int/mediacentre/factsheets/fs330/en/ (accessed on 29 August 2017).
- Cairncross, S.; Hunt, C.; Boisson, S.; Bosteon, K.; Curtis, V.; Fung, I.C.H.; Schmidt, W.P. Water, sanitation and hygiene for the prevention of diarrhoea. Int. J. Epidemiol. 2010, 39, i193–i205. [Google Scholar] [CrossRef] [PubMed]
- Bloomfield, S.F.; Aiello, A.E.; Cookson, B.; O’Boyle, C.; Larson, E.L. The effectiveness of hand hygiene procedures in reducing the risks of infections in home and community settings including handwashing and alcohol-based hand sanitizers. Am. J. Infect. Control 2007, 35, S27–S64. [Google Scholar] [CrossRef]
- Brilhante, R.S.N.; Sales, J.A.; Pereira, V.S.; Castelo-Branco, D.D.S.C.M.; De Aguiar Cordeiro, R.; De Souza Sampaio, C.M.; Paiva, M.D.A.N.; Dos Santos, J.B.F.; Sidrim, J.J.C.; Rocha, M.F.G. Research advances on the multiple uses of Moringa oleifera: A sustainable alternative for socially neglected population. Asian Pac. J. Trop. Med. 2017, 10, 621–630. [Google Scholar] [CrossRef] [PubMed]
- Sayeed, M.A.; Hossain, M.S.; Ehsanul, M.; Chowdhury, H.; Haque, M. In vitro antimicrobial activity of methanolic extract of Moringa oleifera Lam. Fruits. J. Pharmacogn. Phytochem. 2012, 1, 94–98. [Google Scholar]
- Marrufo, T.; Nazzaro, F.; Mancini, E.; Fratianni, F.; Coppola, R.; De Martino, L.; Agostinho, A.B.; De Feo, V. Chemical composition and biological activity of the essential oil from leaves of Moringa oleifera Lam. cultivated in Mozambique. Molecules 2013, 18, 10989–11000. [Google Scholar] [CrossRef] [PubMed]
- Mishra, S.P.; Singh, P.; Singh, S. Processing of Moringa oleifera leaves for human consumption. Bull. Environ. Pharmacol. Life Sci. 2012, 2, 28–31. [Google Scholar]
- Doerr, B. Moringa water treatment. In Environmental Chemistry Laboratory Manual Selected Analytical Method(s); International Institute for Infrastructural, Hydraulic and Environmental Engineering: Delft, The Nertherlands, 2005. [Google Scholar]
- Shebek, K.; Schantz, A.B.; Sines, I.; Lauser, K.; Velegol, S.; Kumar, M. The flocculating cationic polypeptide from Moringa oleifera seeds damages bacterial cell membranes by causing membrane fusion. Langmuir 2015, 31, 4496–4502. [Google Scholar] [CrossRef] [PubMed]
- Rani, N.Z.A.; Husain, K.; Kumolosasi, E. Moringa genus: A review of phytochemistry and pharmacology. Front. Pharmacol. 2018, 9, 108. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Chen, X.; Wu, A. Mini review on antimicrobial activity and bioactive compounds of Moringa oleifera. Med. Chem. 2016, 6, 578–582. [Google Scholar] [CrossRef]
- Rahman, M.M.; Sheikh, M.M.I.; Sharmin, S.A.; Islam, M.S.; Rahman, M.A.; Rahman, M.M.; Alam, M.F. Antibacterial activity of leaf juice and extracts of Moringa oleifera Lam. against some human pathogenic bacteria. CMU J. Nat. Sci. 2009, 8, 219–227. [Google Scholar]
- Kheir, S.M.; Kafi, S.K.; Elbir, H. The antimicrobial and phytochemical characteristic of Moringa oleifera seeds, leaves, and flowers. World J. Pharm. Res. 2015, 4, 258–271. [Google Scholar]
- Biran, A.; Schmidt, W.P.; Zeleke, L.; Emukule, H.; Khay, H.; Parker, J.; Peprah, D. Hygiene and sanitation practices amongst residents of three long-term refugee camps in Thailand, Ethiopia and Kenya. Trop. Med. Int. Heal. 2012, 17, 1133–1141. [Google Scholar] [CrossRef] [PubMed]
- Bloomfield, S.F.; Nath, K.J. Use of ash and mud for handwashing in low income communities. IFH 2009, 1014, 1–40. [Google Scholar]
- Phillips, R.M.; Vujcic, J.; Boscoe, A.; Handzel, T.; Aninyasi, M.; Cookson, S.T.; Blanton, C.; Blum, L.S.; Ram, P.K. Soap is not enough: Handwashing practices and knowledge in refugee camps, Maban County, South Sudan. Confl. Health 2015, 9, 39. [Google Scholar] [CrossRef] [PubMed]
- University at Buffalo, Oxfam Great Britain, and the US Centers for Disease Control and Prevention; Ram., P.K.; Blum, L.S.; Vujcic, J.; Phillips, R.M.; Boscoe, A.; Garang, A.J.; Handzel, T.; Thomas, A. Handwashing Behavior and Approaches to Handwashing Promotion in the Ongoing Humanitarian Emergency in South Sudan; University at Buffalo, Oxfam Great Britain, and the US Centers for Disease Control and Prevention: Buffalo, NY, USA, 2013. [Google Scholar]
- Islam, M.S.; Amin, N.; Pickering, A.J.; Ram, P.K.; Unicomb, L.; Najnin, N.; Homaira, N.; Ashraf, S.; Abedin, J.; Islam, M.S.; et al. Microbiological evaluation of the efficacy of soapy water to clean hands: A randomized, non-inferiority field trial. Am. J. Trop. Med. Hyg. 2014, 91, 415–423. [Google Scholar]
- Luby, S.P.; Halder, A.K.; Huda, T.; Unicomb, L.; Johnston, R.B. The effect of handwashing at recommended times with water alone and with soap on child diarrhea in rural Bangladesh: An observational study. PLoS Med. 2011, 8, e1001052. [Google Scholar] [CrossRef] [PubMed]
- Burton, M.; Cobb, E.; Donachie, P.; Judah, G.; Curtis, V.; Schmidt, W.P. The effect of handwashing with water or soap on bacterial contamination of hands. Int. J. Environ. Res. Public Health 2011, 8, 97–104. [Google Scholar] [CrossRef] [PubMed]
- Firth, J.; Balrah, V.; Muliyil, J.; Roy, S.; Michael, R.; Chandresekhar, R.; Kang, G. Point-of-use interventions to decrease contamination of drinking water: A randomized, controlled pilot study on efficacy, effectiveness, and acceptability of closed containers, Moringa oleifera, and in-home chlorination in rural South India. Am. J. Trop. Med. Hyg. 2010, 82, 759–765. [Google Scholar] [CrossRef] [PubMed]
- Torondel, B.; Opare, D.; Brandberg, B.; Cobb, E.; Cairncross, S. Efficacy of Moringa oleifera leaf powder as a hand-washing product: A crossover controlled study among healthy volunteers. BMC Complement. Altern. Med. 2014, 14, 57. [Google Scholar] [CrossRef] [PubMed]
- Vujcic, J. Strategies & Challenges to Handwashing Promotion in Humanitarian Emergencies: Key Informant Interviews with Agency Experts; University at Buffalo: Buffalo, NY, USA, 2014. [Google Scholar]
- Tetteh-Quarcoo, P.B.; Anim-Baidoo, I.; Attah, S.K.; Baako, B.A.L.; Opintan, J.A.; Minamor, A.A.; Abdul-Rahman, M.; Ayeh-Kumi, P.F. Microbial content of ‘bowl water’ used for communal handwashing in preschools within Accra Metropolis, Ghana. Int. J. Microbiol. 2016, 2016, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Bulled, N.; Poppe, K.; Ramatsisti, K.; Sitsula, L.; Winegar, G.; Gumbo, J.; Dillingham, R.; Smith, J. Assessing the environmental context of hand washing among school children in Limpopo, South Africa. Water Int. 2017, 42, 568–584. [Google Scholar] [CrossRef] [PubMed]
- Ehiri, J.E.; Azubuike, M.C.; Ubbaonu, C.N.; Anyanwu, E.C.; Ibe, K.M.; Ogbonna, M.O. Critical control points of complementary food preparation and handling in eastern Nigeria. Bull. World Health Organ. 2001, 79, 423–433. [Google Scholar] [PubMed]
- Schmitt, R.; Bryan, F.L.; Jermini, M.; Chilufya, E.N.; Hakalima, A.T.; Zyuulu, M.; Mfume, E.; Mwandwe, C.; Mullungushi, E.; Lubasp, D. Hazards and critical control points of food preparation in homes in which persons had diarrhea in Zambia. J. Food Prot. 1997, 60, 161–171. [Google Scholar] [CrossRef]
- Dajaan, D.S.; Addo, H.O.; Ojo, L.; Amegah, K.E.; Loveland, F.; Bechala, B.D.; Benjamin, B.B. Hand washing knowledge and practices among public primary schools in the Kintampo Municipality of Ghana. Int. J. Community Med. Public Heal. 2018, 5, 2205. [Google Scholar] [CrossRef]
- Appiah-Brempong, E.; Harris, M.J.; Newton, S.; Gulis, G. Examining school-based hygiene facilities: A quantitative assessment in a Ghanaian municipality. BMC Public Health 2018, 18, 581. [Google Scholar] [CrossRef] [PubMed]
- Moabi, N.A. Microbial Quality of Communal Hand Washing Water at African Funerals in the Mangaung Region. Magister Technologiae’s Thesis, Central University of Technology, Bloemfontein, South Africa, 2016. [Google Scholar]
- Steiner-Asiedu, M.; Van-Ess, S.; Papoe, M.; Setorglo, J.; Asiedu, D.K.; Anderson, A.K. Hand washing practices among school children in Ghana. Curr. Res. J. Soc. Sci. 2011, 3, 293–300. [Google Scholar]
- Environmental Health and Sanitation Directorate (EHSD) of The Ministry of Local Government and Rural Development (MLGRD); Water Directorate (WD) of The Ministry of Water Resources Works and Housing (MWRWH). Water Sanitation and Hygiene (WASH) Behaviour Change Communication (BCC) Strategy for the Urban Sub-Sector; MLGRD/MWRWH: Accra, Ghana, 2011.
- Byappanahalli, M.N.; Nevers, M.B.; Korajkic, A.; Staley, Z.R.; Harwood, V.J. Enterococci in the Environment. Microbiol. Mol. Biol. Rev. 2012, 76, 685–706. [Google Scholar] [CrossRef] [PubMed]
- Gorchev, H.G.; Ozolins, G. Guidelines for Drinking-Water Quality, 4th ed.; WHO Press: Geneva, Switzerland, 2011. [Google Scholar]
- Ferreira, R.S.; Napoleão, T.H.; Santos, A.F.S.; Sá, R.A.; Carneiro-da-Cunha, M.G.; Morais, M.M.C.; Silva-Lucca, R.A.; Oliva, M.L.V.; Coelho, L.C.B.B.; Paiva, P.M.G. Coagulant and antibacterial activities of the water-soluble seed lectin from Moringa oleifera. Lett. Appl. Microbiol. 2011, 53, 186–192. [Google Scholar] [CrossRef] [PubMed]
- Moura, M.C.; Napoleão, T.H.; Coriolano, M.C.; Paiva, P.M.G.; Figueiredo, R.C.B.Q.; Coelho, L.C.B.B. Water-soluble Moringa oleifera lectin interferes with growth, survival and cell permeability of corrosive and pathogenic bacteria. J. Appl. Microbiol. 2015, 119, 666–676. [Google Scholar] [CrossRef] [PubMed]
- Baylis, C.; Green, R.; Presland, F.; Baalham, T. Evaluation of Oxoid Selective E. coli/Coliform Chromogenic Medium Using Pure Cultures. 2003. Available online: http://jornades.uab.cat/workshopmrama/sites/jornades.uab.cat.workshopmrama/files/Brilliance_E.coli_coliform_agar_poster.pdf (accessed on 29 August 2017).
- British Standards Institute. Chemical Disinfectants and Antiseptics. Hygienic Handwash. Test Method and Requirements (Phase 2/Step 2); British Standards Institute: London, UK, 1997. [Google Scholar]
- Whitman, W.B. (Ed.) Bergey’s Manual of Systematics of Archaea and Bacteria; John Wiley & Sons, Ltd.: Chichester, UK, 2015. [Google Scholar]
- Maslow, J.N.; Brecher, S.M.; Adams, K.S.; Durbin, A.; Loring, S.; Arbeit, R.D. Relationship between indole production and differentiation of Klebsiella species: Indole-positive and -negative isolates of Klebsiella determined to be clonal. J. Clin. Microbiol. 1993, 31, 2000–2003. [Google Scholar] [PubMed]
- Grimont, P.A.D.; Grimont, F.; Starr, M.P. Serratia species isolated from plants. Curr. Microbiol. 1981, 5, 317–322. [Google Scholar] [CrossRef]
- Schaible, U.E.; Kaufmann, S.H.E. Malnutrition and infection: Complex mechanisms and global impacts. PLoS Med. 2007, 4, e115. [Google Scholar] [CrossRef] [PubMed]
- Silhavy, T.J.; Kahne, D.; Walker, S. The bacterial cell envelope. Cold Spring Harb. Perspect. Biol. 2010, 2, a000414. [Google Scholar] [CrossRef] [PubMed]
- Ravikumar, K.; Sheeja, A.K. Water clarification using Moringa oleifera seed coagulant. In Proceedings of the 2012 International Conference on Green Technologies (ICGT), Trivandrum, India, 18–20 December 2012; pp. 64–70. [Google Scholar]
- Roberts, L.; Chartier, Y.; Chartier, O.; Malenga, G.; Toole, M.; Rodka, H. Keeping clean water clean in a Malawi refugee camp: A randomized intervention trial. Bull. World Health Organ. 2001, 79, 280–287. [Google Scholar] [PubMed]
- Witt, K.A. The Nutrient Content of Moringa oleifera Leaves; ECHO Research Note no.1; ECHO: North Fort Myers, FL, USA, 2013. [Google Scholar]
- Easton, J.H.; Lalor, M.; Gauthier, J.J.; Pitt, R. In-situ die-off of indicator bacteria and pathogens. In Proceedings of the AWRA’s 1999 Annual Water Resources Conference-Watershed Management to Protect Declining Species, Seattle, WA, USA, 5–9 December 1999; pp. 449–454. [Google Scholar]
- Van Elsas, J.D.; Semenov, A.V.; Costa, R.; Trevors, J.T. Survival of Escherichia coli in the environment: Fundamental and public health aspects. ISME J. 2011, 5, 173–183. [Google Scholar] [CrossRef] [PubMed]
- Cakmakci, M.L.; Evans, H.J.; Seidler, R.J. Characteristics of nitrogen-fixing Klebsiella oxytoca isolated from wheat roots. Plant Soil 1981, 61, 53–63. [Google Scholar] [CrossRef]
- Podschun, R.; Ullmann, U. Klebsiella spp. as nosocomial pathogens: Epidemiology, taxonomy, typing methods, and pathogenicity factors. Clin. Microbiol. Rev. 1998, 11, 589–603. [Google Scholar] [PubMed]
- Cabral, J.P.S. Water microbiology. Bacterial pathogens and water. Int. J. Environ. Res. Public Health 2010, 7, 3657–3703. [Google Scholar] [CrossRef] [PubMed]
- Ayeh-Kumi, P.F.; Tetteh-Quarcoo, P.B.; Duedu, K.O.; Obeng, A.S.; Addo-Osafo, K.; Mortu, S.; Asmah, R.H. A survey of pathogens associated with Cyperus esculentus L. (tiger nuts) tubers sold in a Ghanaian city. BMC Res. Notes 2014, 7, 343. [Google Scholar] [CrossRef] [PubMed]
- Amoah, P.; Drechsel, P.; Abaidoo, R.C.; Klutse, A. Effectiveness of common and improved sanitary washing methods in selected cities of West Africa for the reduction of coliform bacteria and helminth eggs on vegetables. Trop. Med. Int. Heal. 2007, 12, 40–50. [Google Scholar] [CrossRef] [PubMed]
- Wu, Z.; Peng, Y.; Guo, L.; Li, C. Root colonization of encapsulated Klebsiella oxytoca Rs-5 on cotton plants and its promoting growth performance under salinity stress. Eur. J. Soil Biol. 2014, 60, 81–87. [Google Scholar] [CrossRef]
- Pegues, D.A.; Shireley, L.A.; Riddle, C.F.; Anderson, R.L.; Vess, R.W.; Hill, B.C.; Jarvis, W.R. Serratia marcescens surgical wound infection following breast reconstruction. Am. J. Med. 1991, 91, 173S–178S. [Google Scholar] [CrossRef]
- Zhu, J.-K. Plant salt tolerance. Trends Plant Sci. 2001, 6, 66–71. [Google Scholar] [CrossRef]
- Yousef, A.E.; Juneja, V.K. (Eds.) Microbial Stress Adaptation and Food Safety; CRC Press: Boca Raton, FL, USA, 2003; Volume 3. [Google Scholar]
| Colour of Colonies | Gram Stain | TSI—Slope | TSI—Butt | TSI—H2S | TSI—Gas | Citrate | Urea | Indole | Motility | Species Identified |
|---|---|---|---|---|---|---|---|---|---|---|
| Dark pink | Gram-negative rods | Yellow | Yellow | − | + | + | + | + | − | Klebsiella oxytoca |
| Light pink | Gram-negative rods | Yellow | Yellow | − | + | + | + | + | − | Klebsiella oxytoca |
| Orange | Gram-negative rods | Red | Yellow | − | − | + | − | − | + | Serratia spp. |
| Treatment | Mean Pre-Value (log10 cfu/mL) | Mean Post-Value (log10 cfu/mL) | Mean log10 cfu/mL Reduction | Standard Deviation | p-Value of Difference in Mean Compared to Control |
|---|---|---|---|---|---|
| Control (5 mL regular, non-medicated soap) | 8.86 | 5.49 | 3.37 | 0.76 | - |
| 5 mL boiled M. oleifera leaves | 8.90 | 6.32 | 2.57 | 0.26 | 0.005 |
| 4 g dry M. oleifera leaf powder | 9.03 | 7.01 | 2.02 | 0.44 | <0.001 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Clark, Y.N.-B.; Jimenez, M.; Raso, E.; Antwi, L.; Ofosu-Appiah, L.H.; Opare, D.; Torondel, B. Evaluation of Key Antimicrobial Properties of Moringa oleifera in Relation to Its Use as a Hand-Washing Product. Water 2018, 10, 1154. https://doi.org/10.3390/w10091154
Clark YN-B, Jimenez M, Raso E, Antwi L, Ofosu-Appiah LH, Opare D, Torondel B. Evaluation of Key Antimicrobial Properties of Moringa oleifera in Relation to Its Use as a Hand-Washing Product. Water. 2018; 10(9):1154. https://doi.org/10.3390/w10091154
Chicago/Turabian StyleClark, Yasaman Naemi-Baghshomali, Monica Jimenez, Enrique Raso, Loretta Antwi, Lawrence H. Ofosu-Appiah, David Opare, and Belen Torondel. 2018. "Evaluation of Key Antimicrobial Properties of Moringa oleifera in Relation to Its Use as a Hand-Washing Product" Water 10, no. 9: 1154. https://doi.org/10.3390/w10091154
APA StyleClark, Y. N.-B., Jimenez, M., Raso, E., Antwi, L., Ofosu-Appiah, L. H., Opare, D., & Torondel, B. (2018). Evaluation of Key Antimicrobial Properties of Moringa oleifera in Relation to Its Use as a Hand-Washing Product. Water, 10(9), 1154. https://doi.org/10.3390/w10091154




