H3PO4-Activated Cattail Carbon Production and Application in Chromium Removal from Aqueous Solution: Process Optimization and Removal Mechanism
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Single Factor Affecting Adsorption Experiments
2.3. Box–Behnken Experimental Designs for Typha Angustifolia Activated Carbon Production and Cr(VI) Adsorption
2.4. Characterization of Typha Angustifolia Activated Carbon
2.5. Sorption-Desorption Experiments
3. Results and Discussion
3.1. Process Optimization of Typha Angustifolia Activated Carbon Production
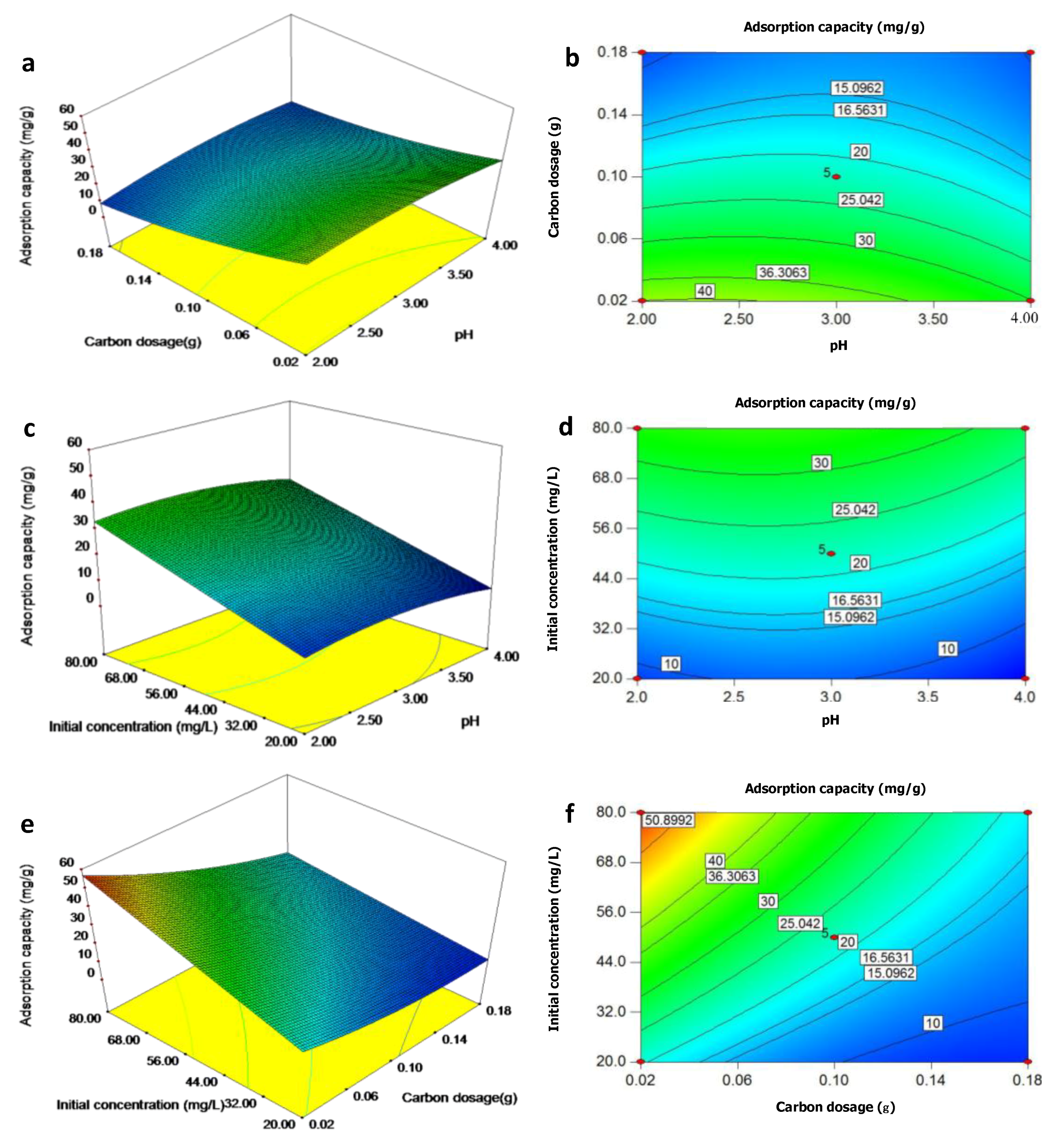
3.1.1. Statistical Analysis and Model Validation for Activated Carbon Preparation
3.1.2. Activated Carbon Yield and Cr Removal Responses
3.1.3. Optimum Parameters of Activated Carbon Preparation
3.2. Cr(VI) Adsorption on Typha Angustifolia Activated Carbon Affected by Single Factors
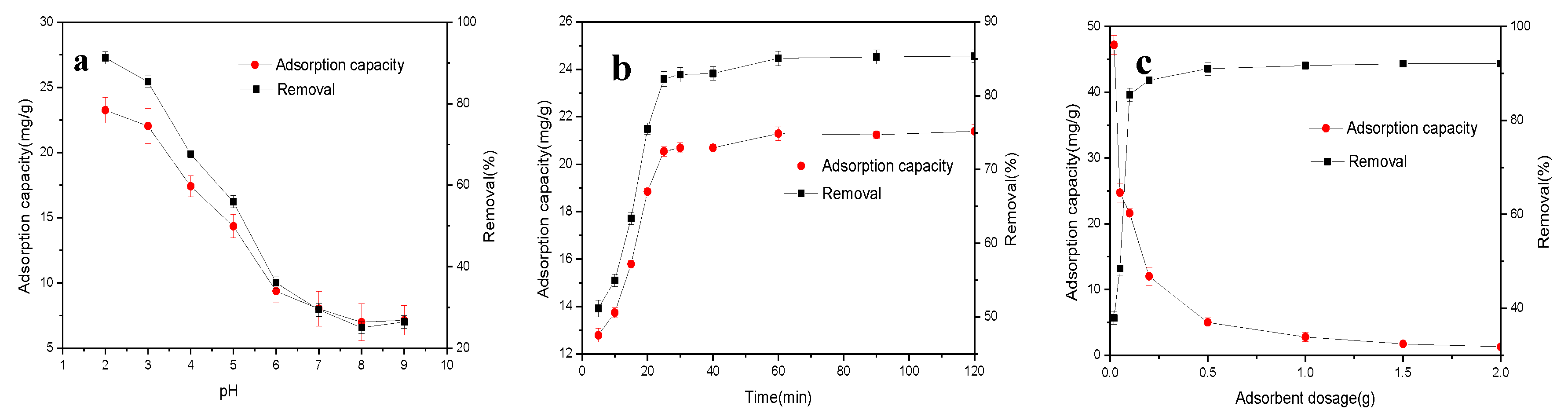
3.2.1. Effect of Initial Solution pH
3.2.2. Effect of Contact Time on Cr Removal and Adsorption Kinetic Analysis
3.2.3. Effect of Activated Carbon Dosage on Cr Removal and Adsorption Isotherms Analysis
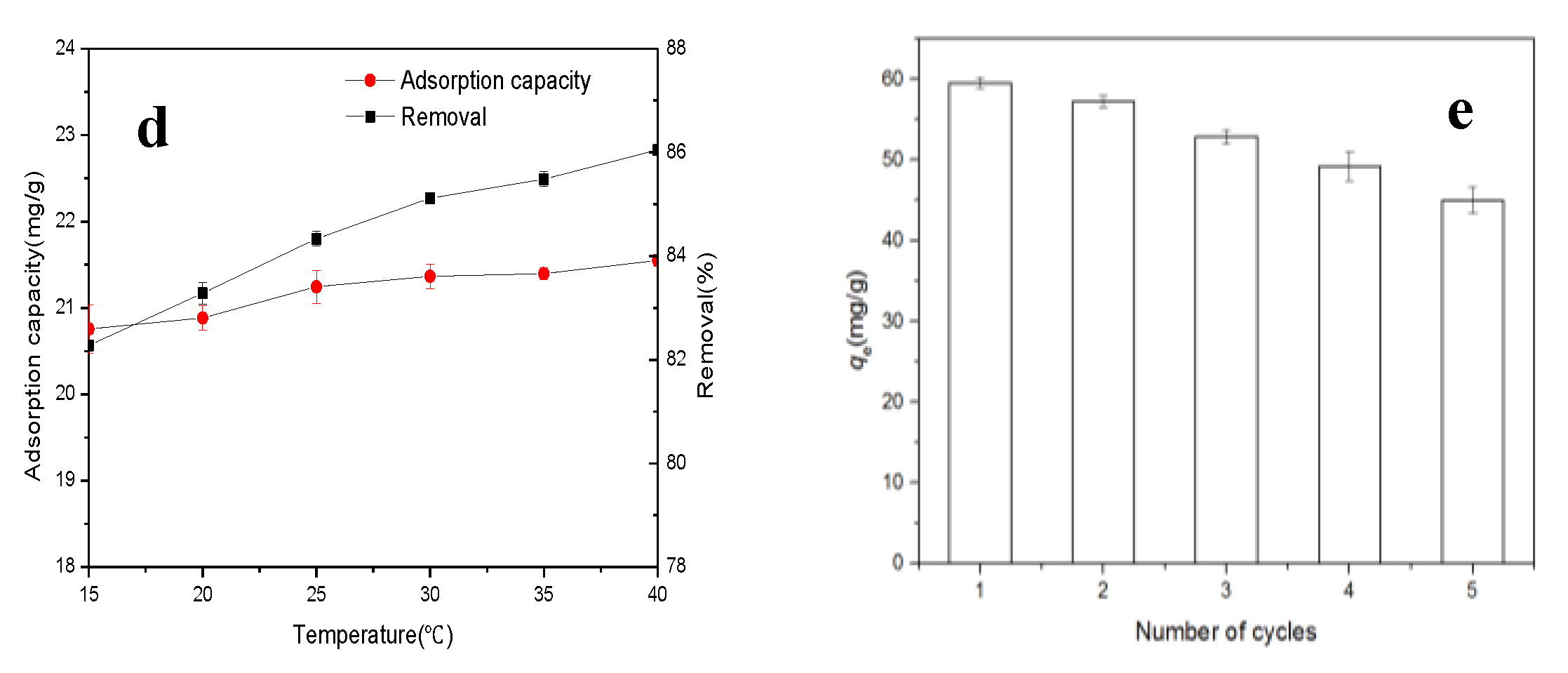
3.2.4. Effect of Temperature on Cr Removal and Adsorption Thermodynamics Analysis
3.3. Process Optimization for Cr Adsorption on Typha Angustifolia Activated Carbon
3.3.1. Statistical Analysis and Model Validation for Cr Sorption
3.3.2. Cr Sorption Capacity Response
3.3.3. Optimum Parameters of Cr Sorption Capacity
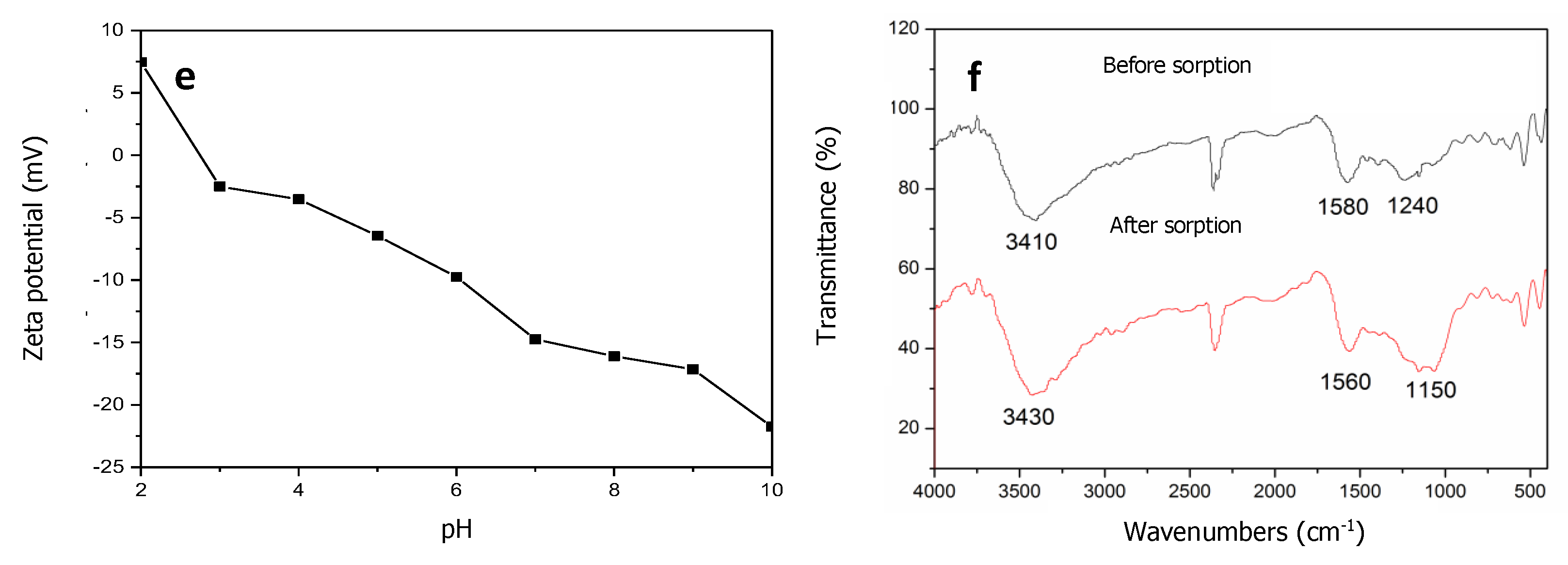
3.4. Characterization of Typha Angustifolia Activated Carbon and Cr Removal Mechanism
3.4.1. Physical Properties of Typha Angustifolia Activated Carbon
3.4.2. Chemical Properties of Typha Angustifolia Activated Carbon
3.5. Regeneration Evaluation of Typha Angustifolia Activated Carbon
4. Conclusions
Supplementary Materials
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Demiral, H.; Demiral, İ.; Tümsek, F.; Karabacakoğlu, B. Adsorption of chromium(VI) from aqueous solution by activated carbon derived from olive bagasse and applicability of different adsorption models. Chem. Eng. J. 2008, 144, 188–196. [Google Scholar] [CrossRef]
- Wise, J.P., Jr.; Wise, J.T.F.; Wise, C.F.; Wise, S.S.; Gianios, C., Jr.; Xie, H.; Thompson, W.D.; Perkins, C.; Falank, C.; Wise, J.P., Sr. Concentrations of the genotoxic metals, chromium and nickel, in whales, tar balls, oil slicks, and released oil from the Gulf of Mexico in the immediate aftermath of the deepwater horizon oil crisis: Is genotoxic metal exposure part of the deepwater horizon legacy. Environ. Sci. Technol. 2014, 48, 2997–3006. [Google Scholar] [PubMed]
- Li, W.; Gong, X.; Xin, L.; Zhang, D.; Gong, H. Removal of Cr(VI) from low-temperature micro-polluted surface water by tannic acid immobilized powdered activated carbon. Bioresour. Technol. 2012, 113, 106–113. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Chen, S.; Zhang, H.; Wang, X. Removal behaviors and mechanisms of hexavalent chromium from aqueous solution by cephalosporin residue and derived chars. Bioresour. Technol. 2017, 238, 484–491. [Google Scholar] [CrossRef] [PubMed]
- Sahu, J.N.; Acharya, J.; Meikap, B.C. Response surface modeling and optimization of chromium(VI) removal from aqueous solution using Tamarind wood activated carbon in batch process. J. Hazard. Mater. 2009, 172, 818–825. [Google Scholar] [CrossRef] [PubMed]
- Sadyrbaeva, T.Z. Removal of chromium(VI) from aqueous solutions using a novel hybrid liquid membrane-electrodialysis process. Chem. Eng. Process. Process Intensif. 2015, 99, 183–191. [Google Scholar] [CrossRef]
- Dubey, S.P.; Sillanpaa, M.; Varma, R.S. Reduction of hexavalent chromium using Sorbaria sorbifolia aqueous leaf extract. Appl. Sci. 2018, 7, 715. [Google Scholar] [CrossRef]
- Mekatel, H.; Amokrane, S.; Benturki, A.; Nibou, D. Treatment of polluted aqueous solutions by Ni2+, Pb2+, Zn2+, Cr6+, Cd2+ and Co2+ ions by ion exchange process using faujasite zeolite. Procedia Eng. 2012, 33, 52–57. [Google Scholar] [CrossRef]
- Gottipati, R.; Mishra, S. Process optimization of adsorption of Cr(VI) on activated carbons prepared from plant precursors by a two-level full factorial design. Chem. Eng. J. 2010, 160, 99–107. [Google Scholar] [CrossRef]
- Han, Y.; Xi, C.; Xin, O.; Sohi, S.P.; Chen, J. Adsorption kinetics of magnetic biochar derived from peanut hull on removal of Cr(VI) from aqueous solution: Effects of production conditions and particle size. Chemosphere 2016, 145, 336–341. [Google Scholar] [CrossRef] [PubMed]
- Jiang, L.; Liu, S.; Liu, Y.; Zeng, G.; Guo, Y.; Yin, Y.; Huang, X. Enhanced adsorption of hexavalent chromium by a biochar derived from ramie biomass (Boehmeria nivea (L.) Gaud.) modified with β-cyclodextrin/poly (L-glutamic acid). Environ. Sci. Pollut. Res. 2017, 24, 23528–23537. [Google Scholar]
- Zhao, D.; Gao, X.; Wu, C.; Xie, R.; Feng, S.; Chen, C. Facile preparation of amino functionalized graphene oxide decorated with Fe3O4 nanoparticles for the adsorption of Cr(VI). Appl. Surf. Sci. 2016, 384, 1–9. [Google Scholar] [CrossRef]
- Chen, H.; Yan, T.; Jiang, F. Adsorption of Cr(VI) from aqueous solution on mesoporous carbon nitride. J. Taiwan Inst. Chem. Eng. 2014, 45, 1842–1849. [Google Scholar] [CrossRef]
- Hamadi, N.K.; Chen, X.D.; Farid, M.M.; Lu, M.G. Adsorption kinetics for the removal chromium(VI) aqueous solution by adsorbents derived from used tyres sawdust. Chem. Eng. J. 2001, 84, 95–105. [Google Scholar] [CrossRef]
- Karthikeyan, T.; Rajgopal, S.; Miranda, L.R. Chromium(VI) adsorption from aqueous solution by Hevea brasilinesis sawdust activated carbon. J. Hazard. Mater. 2005, 124, 192–199. [Google Scholar] [CrossRef] [PubMed]
- Al-Othman, Z.A.; Ali, R.; Naushad, M. Hexavalent chromium removal from aqueous medium by activated carbon prepared from peanut shell: Adsorption kinetics, equilibrium and thermodynamic studies. Chem. Eng. J. 2012, 184, 238–247. [Google Scholar] [CrossRef]
- Gratuito, M.K.B.; Panyathanmaporn, T.; Chumnanklang, R.A.; Sirinuntawittaya, N.; Dutta, A. Production of activated carbon from coconut shell: Optimization using response surface methodology. Bioresour. Technol. 2008, 99, 4887–4895. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Liang, S.; Gao, J.; Ngo, H.H.; Guo, W.; Guo, Z. Enhancement of Cr(VI) removal by modifying activated carbon developed from Zizania caduciflora with tartaric acid during phosphoric acid activation. Chem. Eng. J. 2014, 246, 168–174. [Google Scholar] [CrossRef]
- Tang, C.; Song, J.; Hu, X.; Hu, X.; Zhao, Y.; Li, B.; Ou, D.; Peng, L. Exogenous spermidine enhanced Pb tolerance in Salix matsudana by promoting Pb accumulation in roots and spermidine, nitric oxide, and antioxidant system levels in leaves. Ecol. Eng. 2017, 107, 41–48. [Google Scholar] [CrossRef]
- Tang, C.; Shu, Y.; Zhang, R.; Li, X.; Song, J.; Li, B.; Zhang, Y.; Ou, D. Comparison removal and adsorption mechanism of cadmium and lead from aqueous solution by activated carbons prepared from Typha angustifolia and Salix matsudana. RSC Adv. 2017, 7, 16092–16103. [Google Scholar] [CrossRef]
- Toles, C.A.; Marshall, W.E.; Johns, M.M.; Wartelle, L.H.; Mcaloon, A. Acid-activated carbons from almond shells: physical, chemical and adsorptive properties and estimated cost of production. Bioresour. Technol. 2000, 71, 87–92. [Google Scholar] [CrossRef]
- Box, G.E.P.; Behnken, D.W. Simplex-sum designs: a class of second order rotatable designs derivable from those of first order. Ann. Math. Stat. 1960, 31, 838–864. [Google Scholar] [CrossRef]
- Sen, R.; Swaminathan, T. Response surface modeling and optimization to elucidate and analyze the effects of inoculum age and size on surfactin production. Biochem. Eng. J. 2004, 21, 141–148. [Google Scholar] [CrossRef]
- Prahas, D.; Kartika, Y.; Indraswati, N.; Ismadji, S. Studies of cadmium(II), lead(II), nickel(II), cobalt(II) and chromium(VI) sorption on extracellular polymeric substances produced by Rhodococcus opacus and Rhodococcus rhodochrous. Chem. Eng. J. 2008, 140, 32–42. [Google Scholar] [CrossRef]
- Li, W.; Zhang, L.B.; Peng, J.H.; Li, N.; Zhu, X.Y. Preparation of high surface area activated carbons from tobacco stems with K2CO3 activation using microwave radiation. Ind. Crop Prod. 2008, 27, 341–347. [Google Scholar] [CrossRef]
- Cotton, A.F.; Wilkinson, G.; Bochmann, M.; Murillo, C.A. Advanced Inorganic Chemistry; Wiley: New Delhi, India, 1999. [Google Scholar]
- Palmer, C.D.; Puls, R.W. Natural Attenuation of Hexavalent Chromium in Groundwater and Soils; American Environmental Protection Agency: Washington, DC, USA, 1994. [Google Scholar]
- Stumm, W. The Inner-Sphere Surface Complex: A Key to Understanding Surface Reactivity; American Chemical Society: Washington, DC, USA, 1995. [Google Scholar]
- Yusof, A.M.; Malek, N.A.N.N. Removal of Cr(VI) and As(V) from aqueous solutions by HDTMA-modified zeolite Y. J. Hazard. Mater. 2009, 162, 1019–1024. [Google Scholar] [CrossRef] [PubMed]
- Kampalanonwat, P.; Supaphol, P. The study of competitive adsorption of heavy metal ions from aqueous solution by aminated polyacrylonitrile nanofiber mats. Energy Procedia 2014, 56, 142–151. [Google Scholar] [CrossRef]
- Nemeş, L.; Bulgariu, L. Optimization of process parameters for heavy metals biosorption onto mustard waste biomass. Open Chem. 2016, 14, 175–187. [Google Scholar] [CrossRef]
- Liu, Z.R.; Zhou, L.M.; Wei, P.; Zeng, K.; Wen, C.X.; Lan, H.H. Competitive adsorption of heavy metal ions on peat. J. China Univ. Min. Technol. 2008, 18, 255–260. [Google Scholar] [CrossRef]
- Bayo, J. Kinetic studies for Cd(II) biosorption from treated urban effluents by native grapefruit biomass (Citrus paradisi L.): The competitive effect of Pb(II), Cu(II) and Ni(II). Chem. Eng. J. 2012, 191, 278–287. [Google Scholar]
- De Lemos, M.J.S.; Silva, R.A. Turbulent flow over a layer of a highly permeable medium simulated with a diffusion-jump model for the interface. Int. J. Heat Mass Transf. 2006, 49, 546–556. [Google Scholar] [CrossRef]
- Örnek, A.; Özacar, M.; Şengil, İ.A. Adsorption of lead onto formaldehyde or sulphuric acid treated acorn waste: equilibrium and kinetic studies. Biochem. Eng. J. 2007, 37, 192–200. [Google Scholar] [CrossRef]
- Mohan, D.; Singh, K.P.; Singh, V.K. Trivalent chromium removal from wastewater using low cost activated carbon derived from agricultural waste material and activated carbon fabric cloth. J. Hazard. Mater. 2006, 135, 280–295. [Google Scholar] [CrossRef] [PubMed]
- Depci, T.; Kul, A.R.; Önal, Y. Competitive adsorption of lead and zinc from aqueous solution on activated carbon prepared from Van apple pulp: Study in single- and multi-solute systems. Chem. Eng. J. 2012, 200–202, 224–236. [Google Scholar]
- Chen, H.; Dai, G.; Zhao, J.; Zhong, A.; Wu, J.; Yan, H. Removal of copper(II) ions by a biosorbent-Cinnamomum camphora leaves powder. J. Hazard. Mater. 2010, 177, 228–236. [Google Scholar] [CrossRef] [PubMed]
- Futalan, C.M.; Kan, C.C.; Dalida, M.L.; Hsien, K.J.; Pascua, C.; Wan, M.W. Comparative and competitive adsorption of copper, lead, and nickel using chitosan immobilized on bentonite. Carbohydr. Polym. 2011, 83, 528–536. [Google Scholar] [CrossRef]
- Dobrowolski, R.; Szcześ, A.; Czemierska, M.; Jarosz-Wikołazka, A. Activated carbon from jackfruit peel waste by H3PO4 chemical activation: Pore structure and surface chemistry characterization. Bioresour. Technol. 2017, 225, 113–120. [Google Scholar] [CrossRef] [PubMed]
- Igberase, E.; Osifo, P.; Ofomaja, A. Chromium(VI) ion adsorption by grafted cross-linked chitosan beads in aqueous solution-a mathematical and statistical modeling study. Environ. Technol. 2017, 38, 3156–3166. [Google Scholar] [CrossRef] [PubMed]


| Models | Parameters | Cr | ||
|---|---|---|---|---|
| 50 mg/L | 100 mg/L | 150 mg/L | ||
| Pseudo-first-order model | qe,exp (mg/g) | 21.19 | 32.49 | 38.86 |
| qe,cal (mg/g) | 8.66 | 13.84 | 13.03 | |
| k1 (1/min) | 0.065 | 0.071 | 0.083 | |
| R2 | 0.911 | 0.907 | 0.948 | |
| χ2 | 0.347 | 0.439 | 0.312 | |
| RMSE | 0.589 | 0.662 | 0.559 | |
| Pseudo-second-order model | qe,cal (mg/g) | 22.08 | 33.78 | 39.68 |
| k2 (g/mg·min) | 0.012 | 0.008 | 0.012 | |
| R2 | 0.998 | 0.998 | 0.998 | |
| χ2 | 0.005 | 0.002 | 3.5 × 10−4 | |
| RMSE | 0.072 | 0.042 | 0.019 | |
| Elovich model | a | 22.09 | 13.18 | 8.44 |
| b | 4.092 | 4.617 | 3.039 | |
| R2 | 0.772 | 0.829 | 0.807 | |
| χ2 | 4.574 | 9.963 | 10.499 | |
| RMSE | 2.139 | 3.157 | 3.240 | |
| Intra-particle diffusion model | kid (mg/g·min−1/2) | 1.228 | 1.538 | 0.951 |
| R2 | 0.557 | 0.645 | 0.634 | |
| Models | Parameters | Cr | ||
|---|---|---|---|---|
| 15 °C | 25 °C | 35 °C | ||
| Langmuir | Qm (mg/g) | 43.92 | 43.26 | 43.01 |
| Ka (L/mg) | 0.090 | 0.109 | 0.132 | |
| R2 | 0.985 | 0.987 | 0.984 | |
| χ2 | 2.936 | 2.586 | 3.362 | |
| RMSE | 1.713 | 1.608 | 1.833 | |
| Freundlich | KF [(mg/g)/(mg/L)n] | 8.941 | 9.773 | 10.692 |
| 1/n | 2.956 | 3.112 | 3.269 | |
| R2 | 0.922 | 0.923 | 0.923 | |
| χ2 | 15.387 | 15.316 | 15.807 | |
| RMSE | 3.923 | 3.914 | 3.976 | |
| D–R | B (mol2/J2) | 1.1 × 10−5 | 7.2 × 10−6 | 5.5 × 10−6 |
| Qm (mg/g) | 37.06 | 36.92 | 37.53 | |
| R2 | 0.8874 | 0.8797 | 0.8750 | |
| χ2 | 19.400 | 20.952 | 22.393 | |
| RMSE | 4.404 | 4.577 | 4.732 | |
| Temkin | KTE | 1.212 | 1.540 | 1.959 |
| b | 282.04 | 302.97 | 321.73 | |
| R2 | 0.9775 | 0.9797 | 0.9816 | |
| χ2 | 3.936 | 3.545 | 3.290 | |
| RMSE | 1.984 | 1.883 | 1.814 | |
| Metal | T (°C) | n | Ka | RLmax | RLmin | B (mol2/J2) | E (kJ/mol) |
| 288 | 2.956 | 0.090 | 0.357 | 0.053 | 1.1 × 10−5 | 0.302 | |
| Cr | 298 | 3.112 | 0.109 | 0.314 | 0.046 | 7.2 × 10−6 | 0.373 |
| 308 | 3.269 | 0.132 | 0.275 | 0.036 | 5.5 × 10−6 | 0.426 | |
| lnKc | ΔG0 (kJ/mol) | ΔH0 (kJ/mol) | ΔS0 (J/mol·K) | ||||
| 288 | 1.534 | −11.08 | 8.49 | 42.33 | |||
| 293 | 1.595 | −12.01 | |||||
| Cr | 298 | 1.691 | −13.44 | ||||
| 303 | 1.740 | −14.35 | |||||
| 308 | 1.765 | −14.96 | |||||
| 313 | 1.815 | −15.98 | |||||
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shu, Y.; Tang, C.; Hu, X.; Jiang, L.; Hu, X.; Zhao, Y. H3PO4-Activated Cattail Carbon Production and Application in Chromium Removal from Aqueous Solution: Process Optimization and Removal Mechanism. Water 2018, 10, 754. https://doi.org/10.3390/w10060754
Shu Y, Tang C, Hu X, Jiang L, Hu X, Zhao Y. H3PO4-Activated Cattail Carbon Production and Application in Chromium Removal from Aqueous Solution: Process Optimization and Removal Mechanism. Water. 2018; 10(6):754. https://doi.org/10.3390/w10060754
Chicago/Turabian StyleShu, Yan, Chunfang Tang, Xinjiang Hu, Luhua Jiang, Xi Hu, and Yunlin Zhao. 2018. "H3PO4-Activated Cattail Carbon Production and Application in Chromium Removal from Aqueous Solution: Process Optimization and Removal Mechanism" Water 10, no. 6: 754. https://doi.org/10.3390/w10060754
APA StyleShu, Y., Tang, C., Hu, X., Jiang, L., Hu, X., & Zhao, Y. (2018). H3PO4-Activated Cattail Carbon Production and Application in Chromium Removal from Aqueous Solution: Process Optimization and Removal Mechanism. Water, 10(6), 754. https://doi.org/10.3390/w10060754




