Potential Impact of In-Situ Oil Shale Exploitation on Aquifer System
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Area and Samples
2.2. Porosity Determination
2.2.1. Determination of Oil Shale Sample Weight
2.2.2. Determination of the Specific Gravity of Oil Shale Samples
2.2.3. Porosity Calculation
2.3. Water Absorption
2.4. Determination of Water Quality Indices in Oil Shale Soaking Liquid
2.5. Determination of Organic Matter in Oil Shale Immersion Samples
3. Results and Discussion
3.1. Impact of High-Temperature Pyrolysis on Physical and Hydraulic Properties of Oil Shale
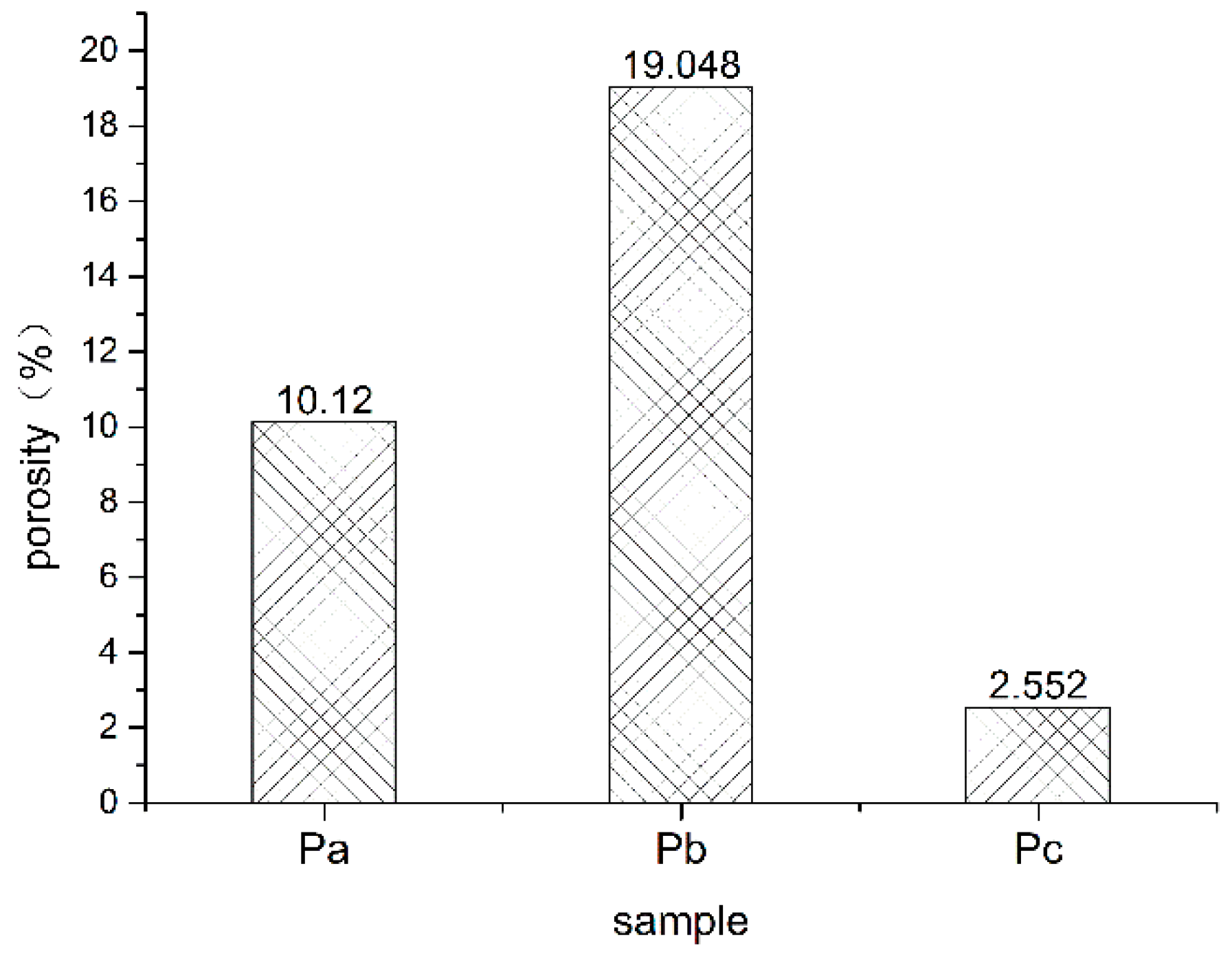
3.1.1. Change in Porosity
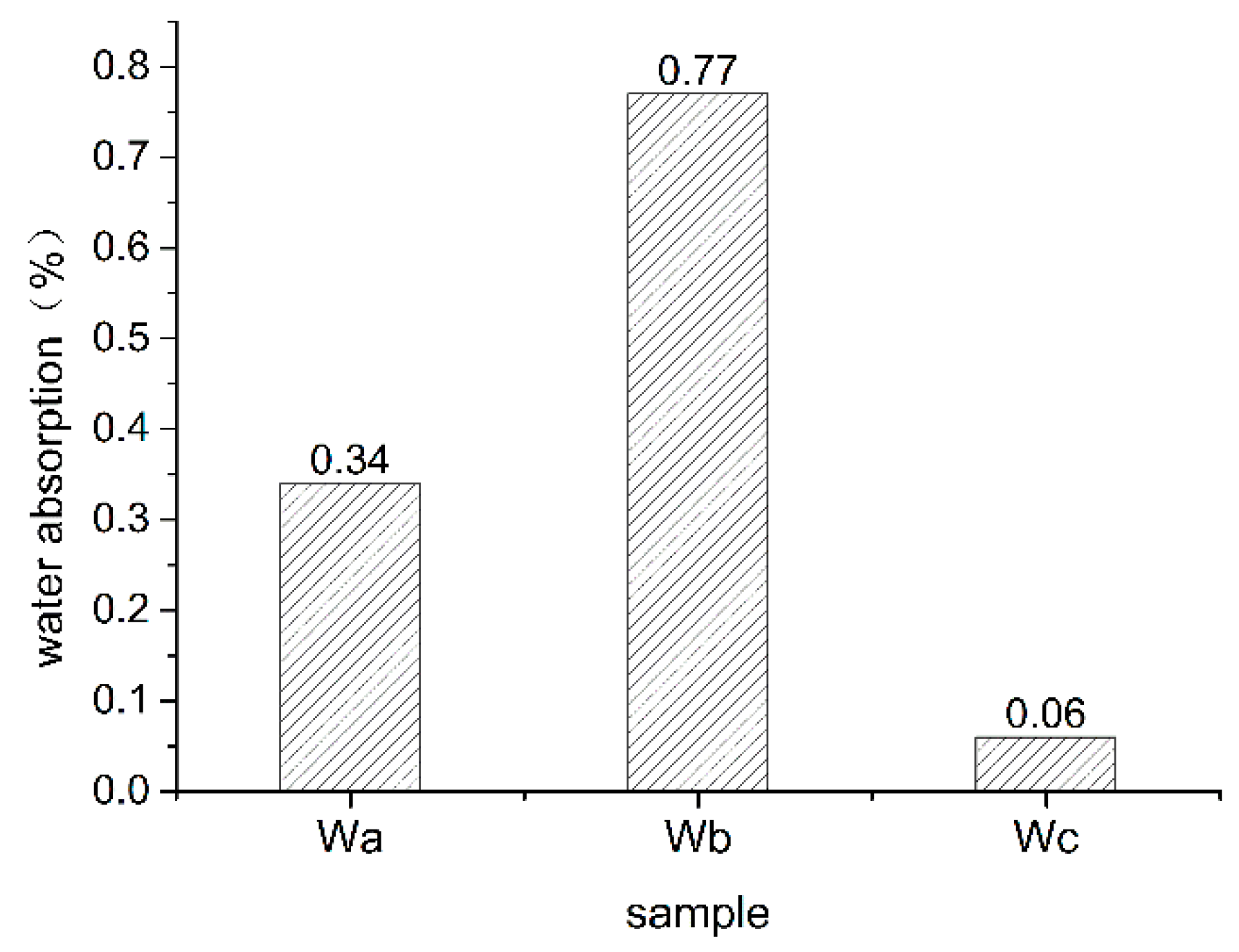
3.1.2. Change in Water Absorption
3.1.3. Analysis of Changes in Physical and Hydraulic Properties
3.2. Effect of Pyrolysis on Chemical Properties of Oil Shale
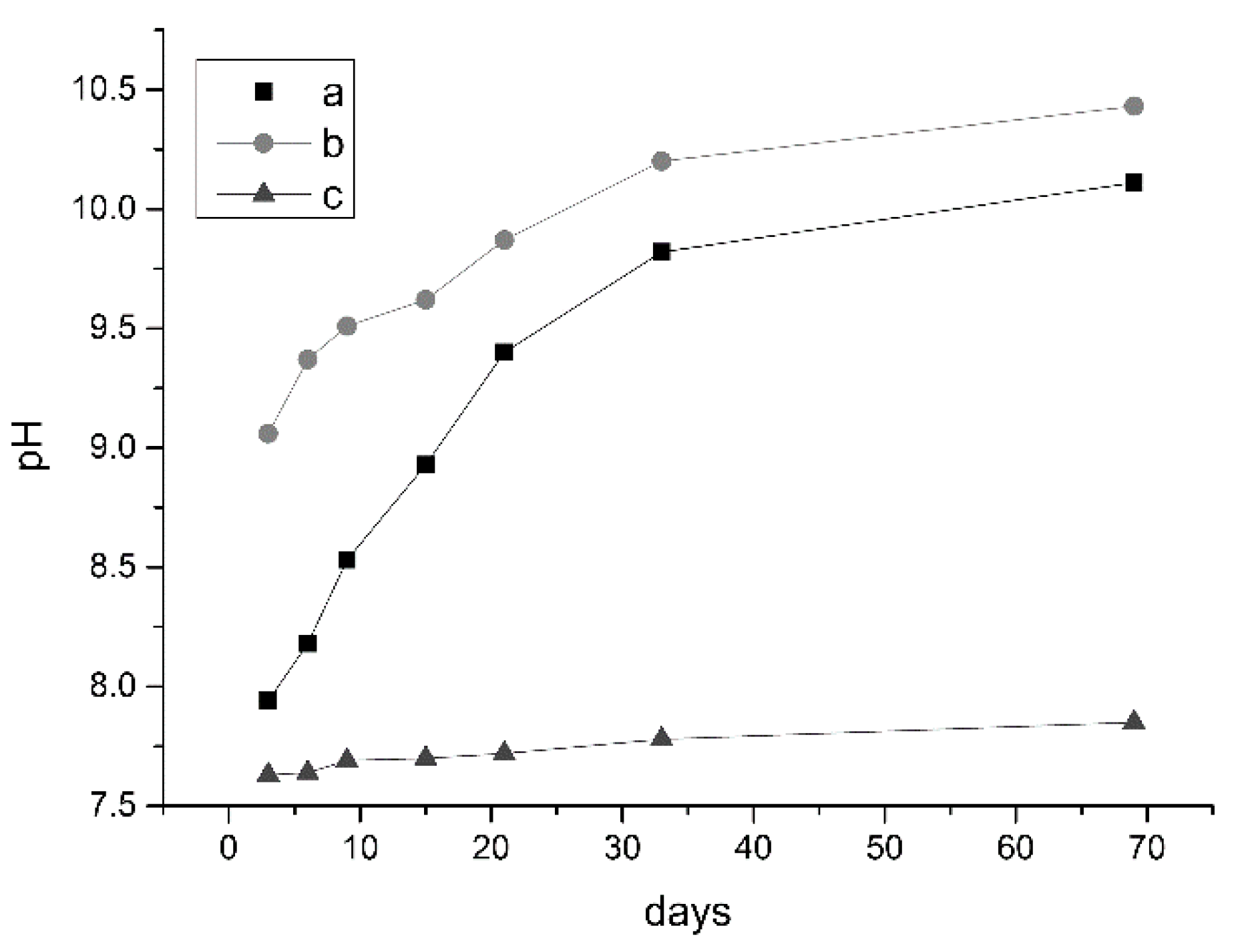
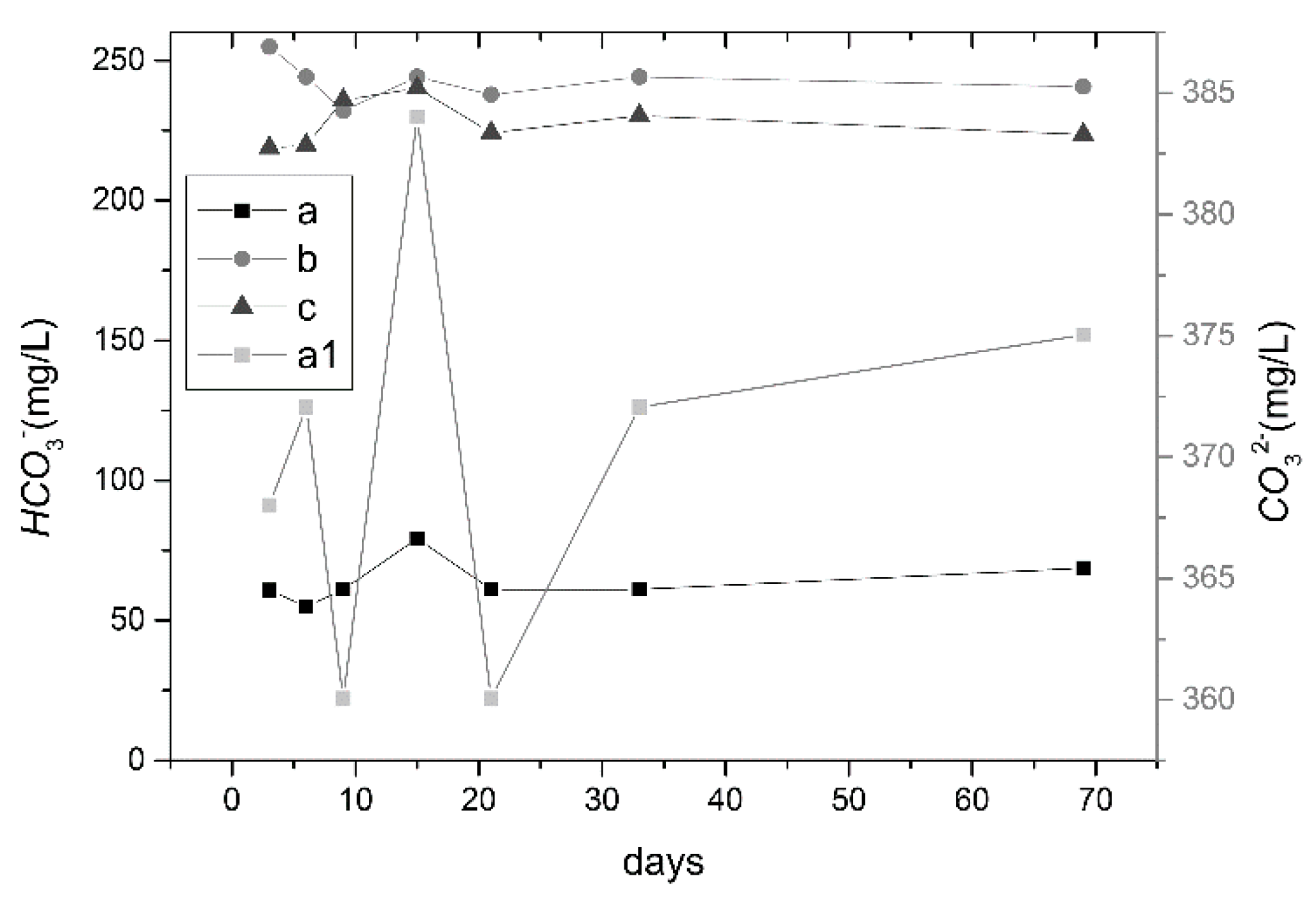
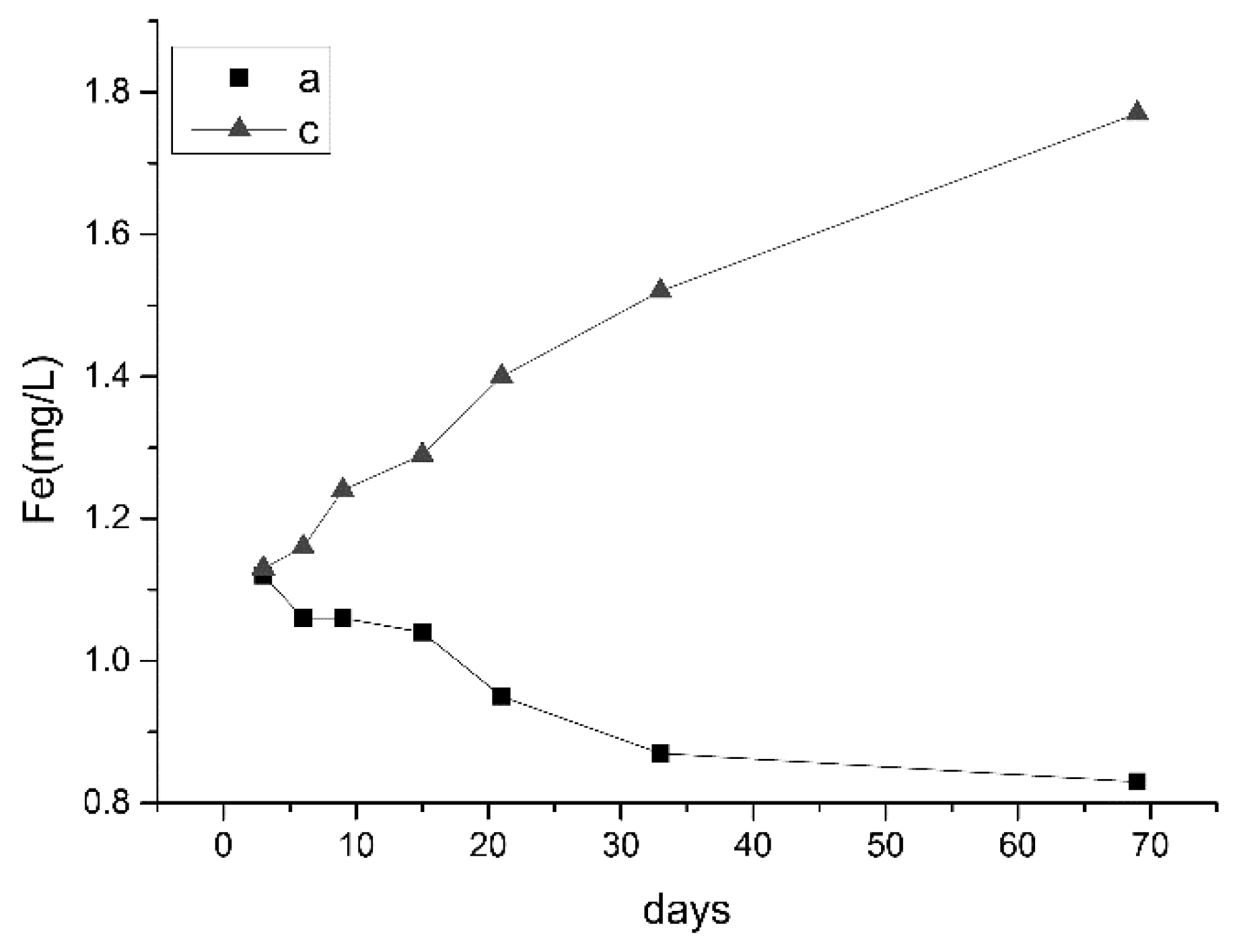
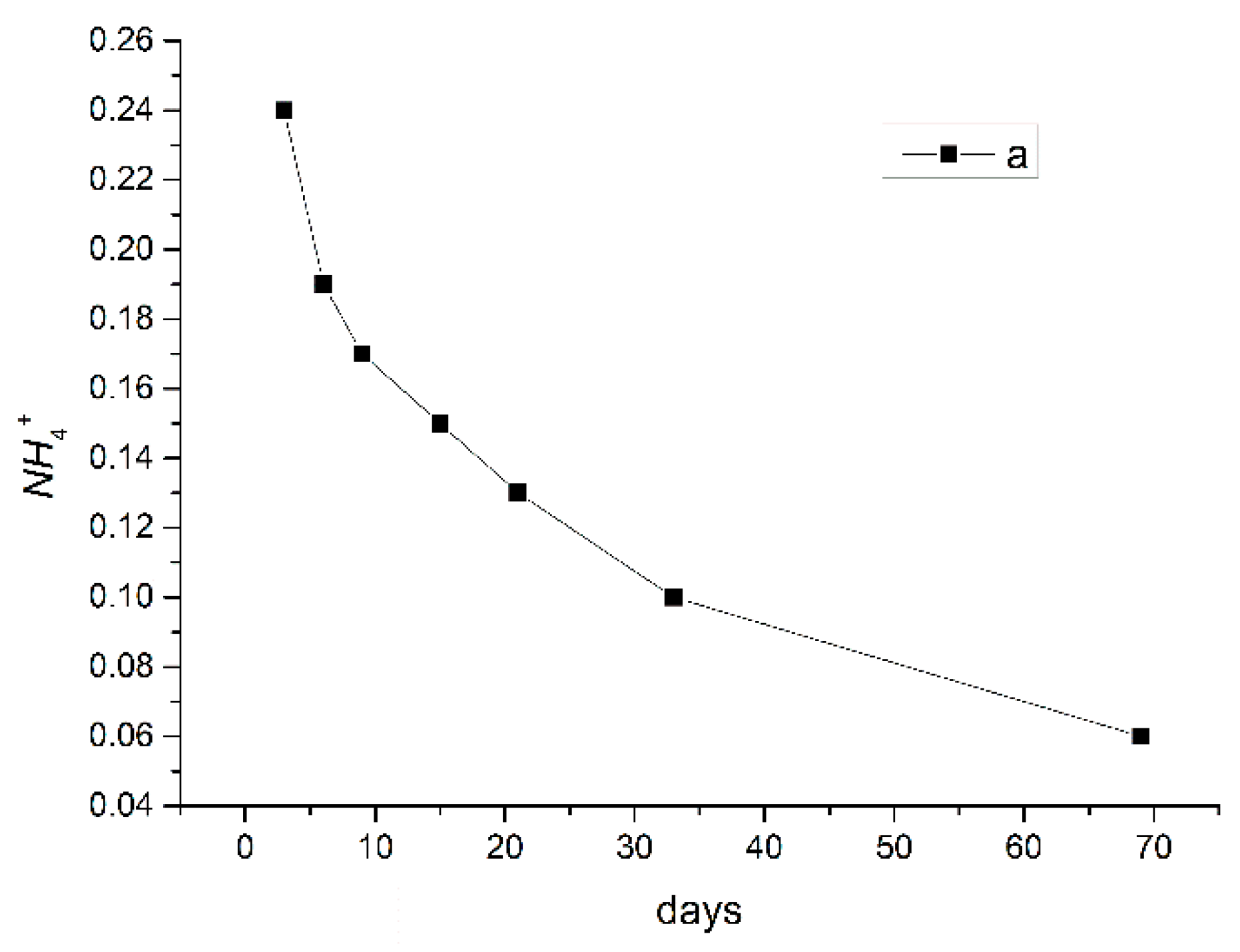
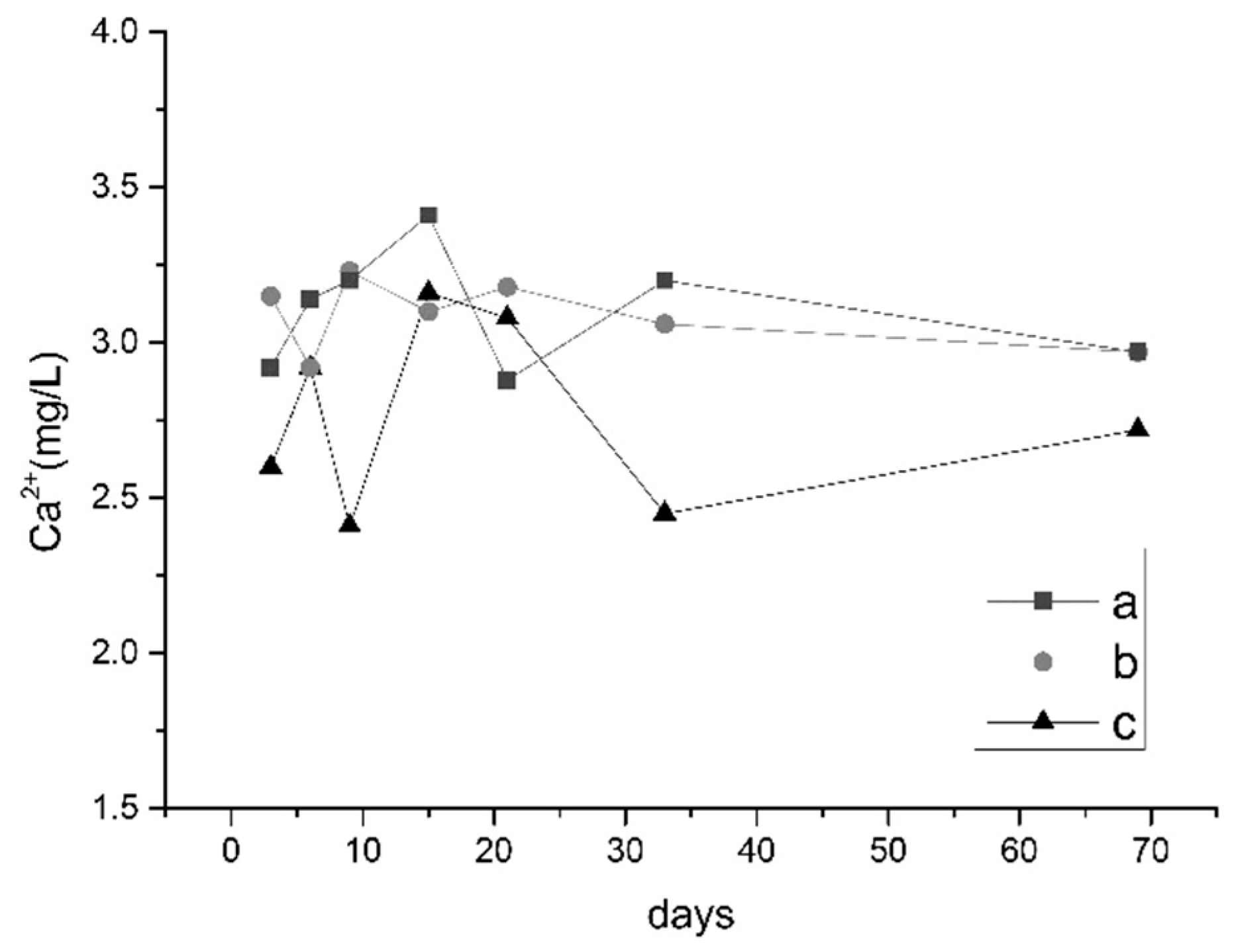
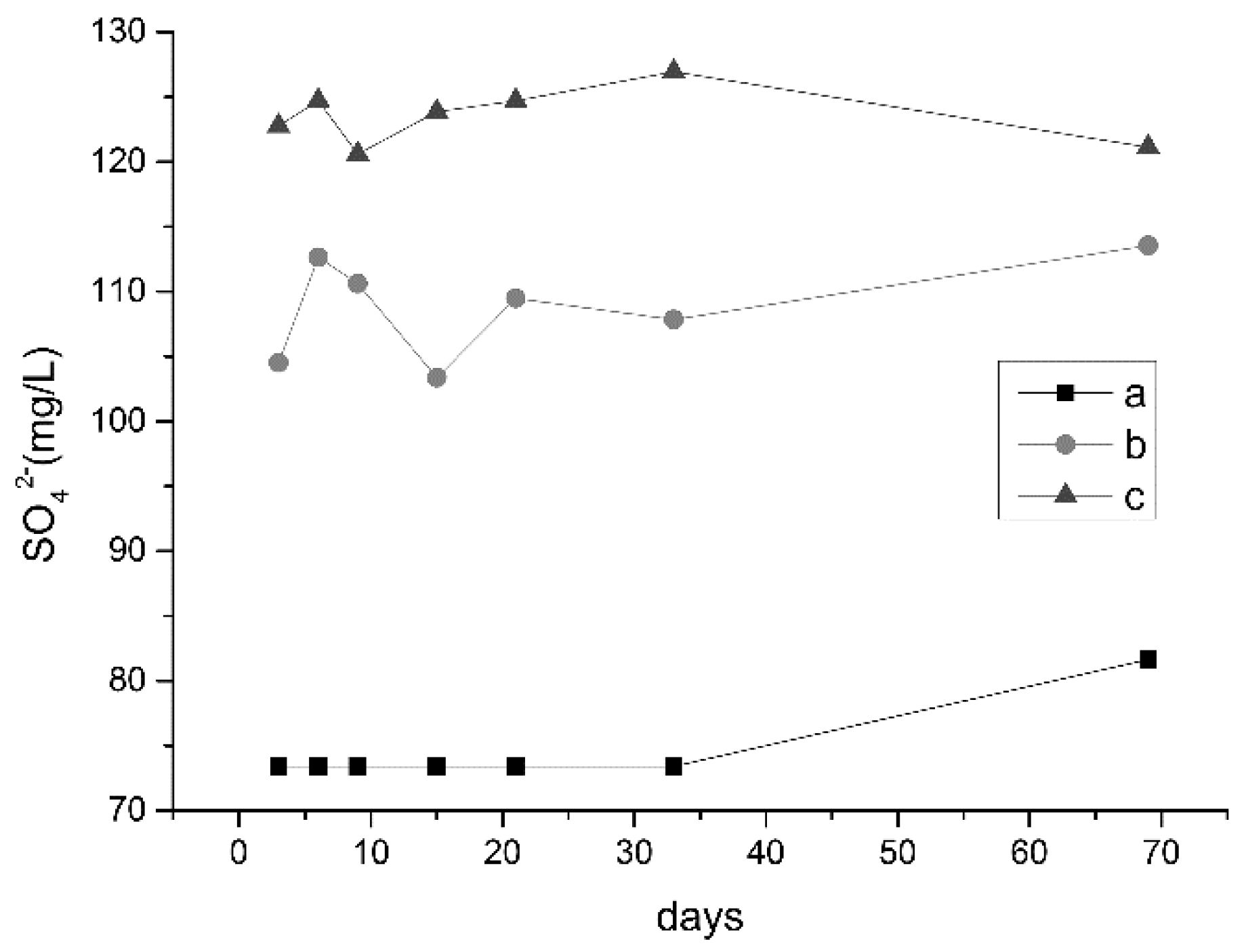
3.2.1. Changes in pH and Ion Concentrations
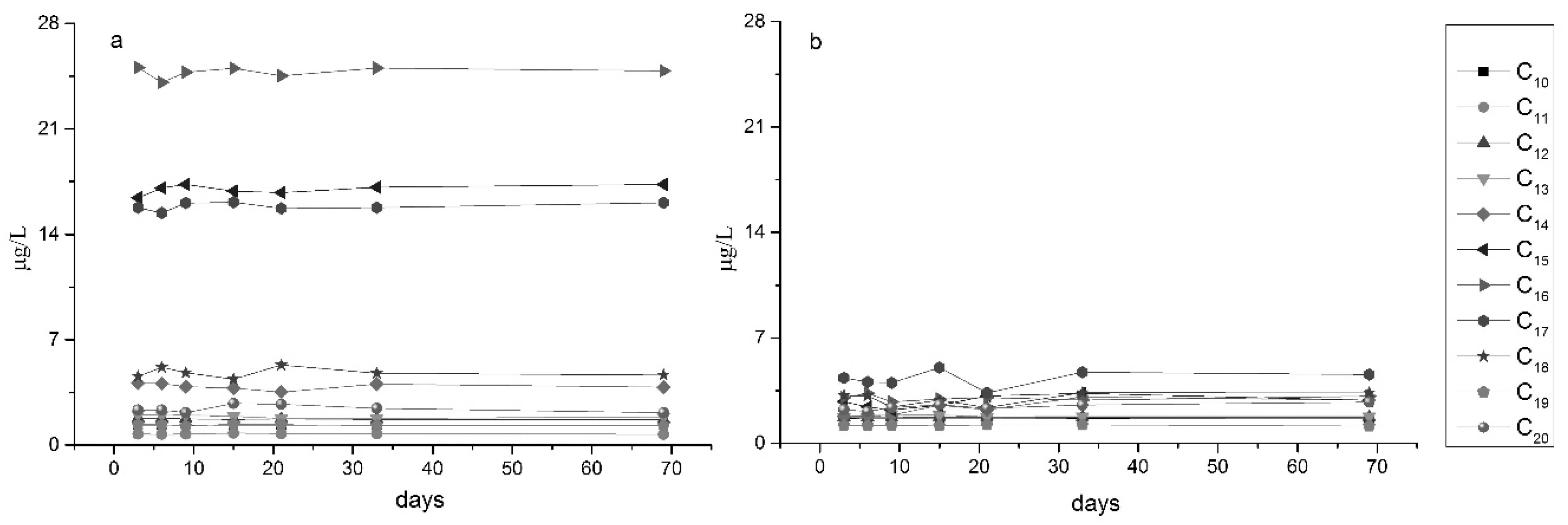
3.2.2. Changes in Organic Content
3.2.3. Analysis of Water Quality Changes
4. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Wang, H.; Zhao, Q.; Liu, H. Resource Distribution and Technical Progress of Oil Shale in China; Petroleum Industry Press: Beijing, China, 2013. [Google Scholar]
- Li, S.-Y.; Ma, Y.; Qian, J.-L. Global oil shale research, development and utilization today and an overview of three oil shale symposiums in 2011. Sino-Glob. Energy 2012, 17, 8–17, (In Chinese with English Abstract). [Google Scholar]
- Guan, X.; Dong, L.-I.; Han, D.-Y. Development and utilization progress of foreign oil shale resources. Contemp. Chem. Ind. 2015, 44, 80–82, (In Chinese with English Abstract). [Google Scholar]
- Sun, P.; Liu, Z.; Bai, Y.; Xu, Y.; Liu, R.; Meng, Q.; Hu, F. Accumulation stages and evolution characteristics of oil shale and coal in the Dunhua-Mishan fault zone, Northeast China. Oil Shale 2016, 33, 203–215. [Google Scholar] [CrossRef]
- Chen, X.-F.; Gao, W.-J.; Zhao, J. Status of exploiting and utilization of oil shale in China. Clean Coal Technol. 2010, 16, 29–31. [Google Scholar]
- Jalkanen, L.; Juhanoja, J. The effect of large anthropogenic particulate emissions on atmospheric aerosols, deposition and bioindicators in the eastern Gulf of Finland Region. Sci. Total Environ. 2000, 262, 123–136. [Google Scholar] [CrossRef]
- Song, Y.; Liu, Z.; Meng, Q.; Xu, J.; Sun, P.; Cheng, L.; Zheng, G. Multiple controlling factors of the enrichment of organic matter in the upper cretaceous oil shale sequences of the Songliao basin, ne China: Implications from geochemical analyses. Oil Shale 2016, 33, 142–166. [Google Scholar] [CrossRef]
- Zhong, S.; Tao, Y.; Li, X.; Li, T.; Zhang, F. Simulation and assessment of shale oil leakage during in situ oil shale mining. Oil Shale 2014, 31, 337–350. [Google Scholar]
- McCarthy, H.E.; Cha, C.Y.; Bartel, W.J.; Burton, R.S. Development of the modified in situ oil-shale process. Aiche J. 1976, 72, 14–23. [Google Scholar]
- Liu, D.-X.; Wang, H.-Y.; Zheng, D.-W.; Fang, C.-H.; Ge, Z.-X. World progress of oil shale in-situ exploitation methods. Nat. Gas Ind. 2009, 25, 128–132, (In Chinese with English Abstract). [Google Scholar]
- Zheng, D.; Li, S.; Ma, G.; Wang, H. Autoclave pyrolysis experiments of Chinese Liushuhe oil shale to simulate in-situ underground thermal conversion. Oil Shale 2012, 29, 103–114. [Google Scholar] [CrossRef]
- Maaten, B.; Loo, L.; Konist, A.; Nesumajev, D.; Pihu, T.; Külaots, I. Decomposition kinetics of American, Chinese and Estonian oil shales kerogen. Oil Shale 2016, 33, 167–183. [Google Scholar] [CrossRef]
- Qian, J.; Yin, L. Oil Shale—Petroleum Alternative; Petroleum Industry Press: Beijing, China, 2010. [Google Scholar]
- Brandt, A.R. Converting oil shale to liquid fuels: Energy Inputs and greenhouse gas emissions of the shell in situ conversion process. Environ. Sci. Technol. 2008, 42, 7489–7495. [Google Scholar] [CrossRef] [PubMed]
- Tang, D. Rock and Soil Engineering; Geology Publishing House: Beijing, China, 1985. (In Chinese) [Google Scholar]
- Zhou, K.; Sun, Y.-H.; Li, Q.; Guo, W.; Lyu, S.-D.; Han, J. Experimental research about thermogravimetric analysis and thermal physical properties of Nong’an oil shale. Glob. Geology 2016, 35, 1178–1184. (In Chinese) [Google Scholar]
- Wang, W.; Ma, Y.; Li, S.; Shi, J.; Teng, J. Effect of temperature on the EPR properties of oil shale pyrolysates. Energy Fuels 2016, 30, 830–834. [Google Scholar] [CrossRef]
- Decora, A.W.; Kerr, R.D. Processing use, and characterization of shale oil products. Environ. Health Perspect. 1979, 30, 217–223. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Han, X.-X.; Jiang, X.-M.; Cui, Z.-G.; Yu, L.-J. Evolution of pore structure of oil shale particles during combustion. Proc. Chin. Soc. Electr. Eng. 2007, 27, 26–30. [Google Scholar]
- Jiang, Q. Study on Physical and Chemical Properties of Oil Shale in Huadian City; Northeast Electric Power University: Jilin, China, 2006. (In Chinese) [Google Scholar]
- Meng, Q. Research on Petrologic and Geochemical Characteristics of Eocene Oil Shale and Its Enrichment Regularity, Huadian Basin; Jilin University: Jilin, China, 2010. (In Chinese) [Google Scholar]
- Bai, F.; Sun, Y.; Liu, Y.; Guo, M. Evaluation of the porous structure of Huadian oil shale during pyrolysis using multiple approaches. Fuel 2017, 187, 1–8. [Google Scholar] [CrossRef]
- Xi, F.-Y.; Zhao, X.-E.; Cai, X. Study on kinetics of oxidation of iron sulfur compounds. Appl. Chem. Ind. 2017, 5, 825–828. (In Chinese) [Google Scholar]
- Qiu, S. Experimental Study on the Impacts of Oil Shale in-Situ Pyrolysis on Groundwater Hydrochemical Characteristics; Jilin University: Jilin, China, 2016. (In Chinese) [Google Scholar]
- Song, W.N.; Dong, Y.L.; Zhou, G.J.; Ding, H.X.; Li, Z. Research summarization of structure-constitute and application of oil shale. J. Heilongjiang Hydraul. Eng. 2010, 3, 20. (In Chinese) [Google Scholar]
- Ying, H.-H.; Zhang, Z.-P.; Wu, Y.-B. Study on influence factors of acid solubility of fracturing proppant. Liaoning Chem. Ind. 2015, 44, 1052–1055, 1065. (In Chinese) [Google Scholar]
- Zhu, G.-Y.; Zhang, S.-C.; Liang, Y.-B.; Li, J. Alteration of thermochemical sulfate reduction to hydrocarbons. Acta Pet. Sin. 2005, 26, 48–52. [Google Scholar]
- Routson, R.C.; Wildung, R.E.; Bean, R.M. A Review of the environmental impact of ground disposal of oil shale wastes. J. Environ. Qual. 1979, 8, 14–19. [Google Scholar] [CrossRef]
| Time (min) | 14.16 | 15.29 | 18.67 | 21.35 | 24.04 | 25.01 | 27.84 |
|---|---|---|---|---|---|---|---|
| Component Chemical formula | C14H22O | C19H41NO3S | C18H24O | C7H5NS2 | C28H38O10 | C30H42Cl2N4O3 | C30H50 |
| Match probability | 50.90% | 52.90% | 52.70% | 65.50% | 57.00% | 50.20% | 57.90% |
| Time (min) | 11.99 | 12.62 | 13.82 | 14.82 | 14.96 | 15.97 | 23.18 |
|---|---|---|---|---|---|---|---|
| Component Chemical formula | C13H28 | C14H30 | C15H32 | C15H32 | C15H32 | C16H34 | C18H35NO |
| Match probability | 77.50% | 75.90% | 73.80% | 80.30% | 73.90% | 78.90% | 78.60% |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hu, S.; Xiao, C.; Jiang, X.; Liang, X. Potential Impact of In-Situ Oil Shale Exploitation on Aquifer System. Water 2018, 10, 649. https://doi.org/10.3390/w10050649
Hu S, Xiao C, Jiang X, Liang X. Potential Impact of In-Situ Oil Shale Exploitation on Aquifer System. Water. 2018; 10(5):649. https://doi.org/10.3390/w10050649
Chicago/Turabian StyleHu, Shuya, Changlai Xiao, Xue Jiang, and Xiujuan Liang. 2018. "Potential Impact of In-Situ Oil Shale Exploitation on Aquifer System" Water 10, no. 5: 649. https://doi.org/10.3390/w10050649
APA StyleHu, S., Xiao, C., Jiang, X., & Liang, X. (2018). Potential Impact of In-Situ Oil Shale Exploitation on Aquifer System. Water, 10(5), 649. https://doi.org/10.3390/w10050649




