The Effect of Bioaugmentation with Archaea on the Oxygen Uptake Rate in a Sequencing Batch Reactor
Abstract
1. Introduction
2. Materials and Methods
2.1. Experiment in the Sequencing Batch Reactor
2.2. Measurement of the Oxygen Uptake Rate (OUR)
3. Results and Discussion
4. Conclusions
- (1)
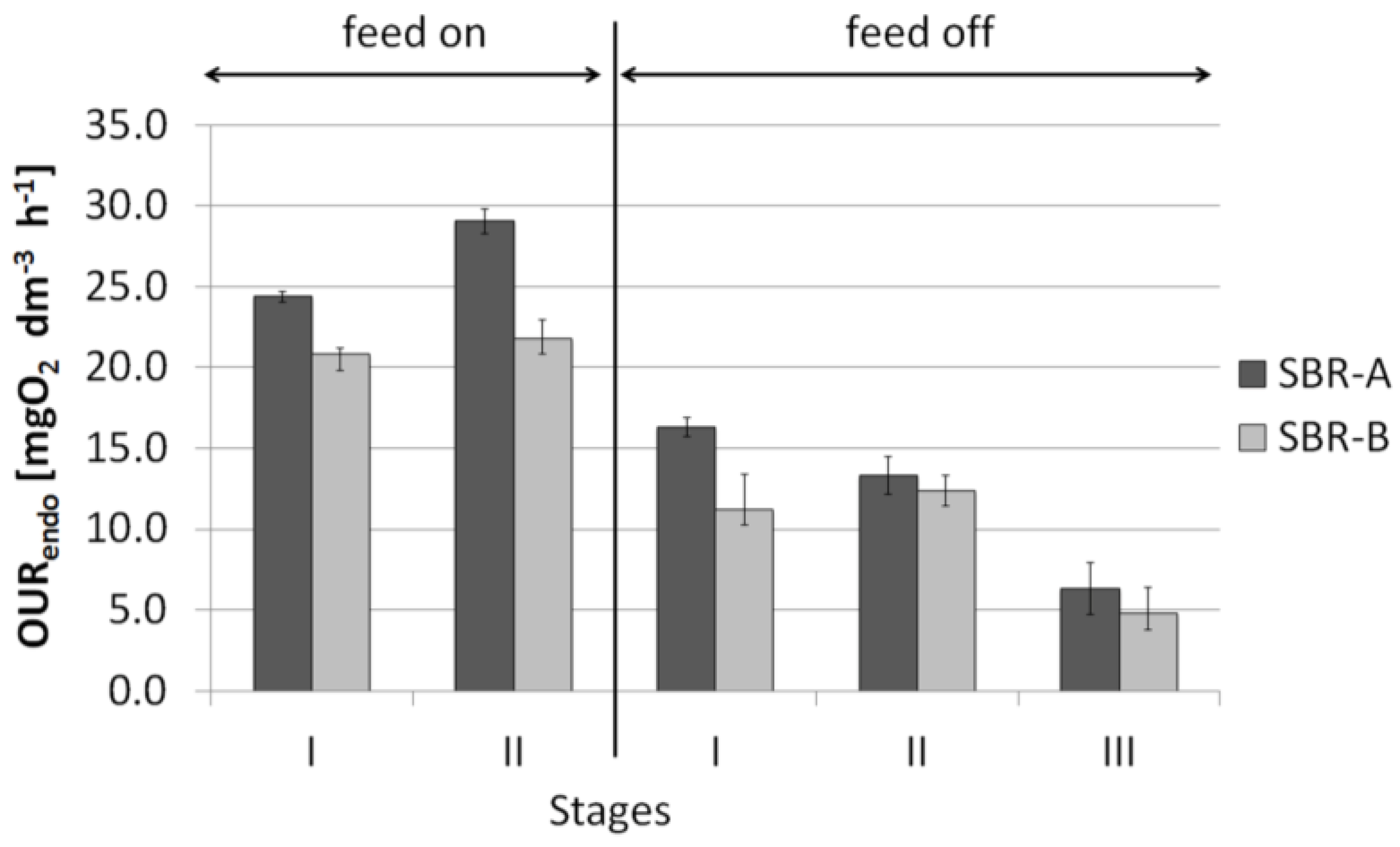
- It was observed that bioaugmentation with Archaea had a positive effect on both the endogenous and exogenous oxygen uptake rate of AS. The values of the OURendo and OURexo in the bioaugmented SBR was higher than in not bioaugmented SBR during the standard performance of the SBR bioreactor operating under sufficient substrate availability. The feeding inhibition of AS together with continuous aeration resulted in gradual stabilization and aerobic digestion of the bioaugmented and not bioaugmented AS, however in presence of Archaea the mentioned process is slower.
- (2)
- The results indicate an increase in the OUR value of bioaugmented AS in comparison with non-bioaugmented one in exactly the same process conditions and greater invariability of the OUR level in the individual stages of the experiment. Therefore, it can be stated that Archaea exert a stabilizing effect on OUR of AS (increase the system’s resistance to external factors) and decrease its sensitivity both to changes in the quality of supplied wastewater and to disruption of substrate supply as well as prolonged aeration.
- (3)
- Because OUR is only one of the possible parameters describing AS, future work should be conducted, for instance related to influence of Archaea bioaugmentation on biogene congested bioreactors performance, bioreactors working in high range of temperatures, but also to describe reactions of eukaryotic organisms present in AS on supplementation with Archaea.
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| OUR | oxygen uptake rate |
| OURexo | exogenous oxygen uptake rate |
| OURendo | endogenous oxygen uptake rate |
| AS | activated sludge |
| SBR | sequencing batch reactor |
| SBR-A | bioreactor with bioaugmented activated sludge |
| SBR-B | non-bioreactor with bioaugmented activated sludge |
References
- Rittmann, B.; Whiteman, R. Bioaugmentation: A coming of age. Water Qual. Int. 1994, 1, 12–16. [Google Scholar]
- Herrero, M.; Stuckey, D.C. Bioaugmentation and its application in wastewater treatment: A review. Chemosphere 2015, 140, 119–128. [Google Scholar] [CrossRef] [PubMed]
- D’Annibale, A.; Ricci, M.; Leonardi, V.; Quaratino, D.; Mincione, E.; Petruccioli, M. Degradation of aromatic hydrocarbons by white-rot fungi in a historically contaminated soil. Biotechnol. Bioeng. 2005, 90, 723–731. [Google Scholar] [CrossRef] [PubMed]
- Mrozik, A.; Piotrowska-Seget, Z. Bioaugmentation as a strategy for cleaning up of soils contaminated with aromatic compounds. Microbiol. Res. 2010, 165, 363–375. [Google Scholar] [CrossRef] [PubMed]
- Zawierucha, I.; Malina, G.; Ciesielski, W.; Rychter, P. Effectiveness of Intrinsic Biodegradation Enhancement in Oil Hydrocarbons Contaminated Soil. Arch. Environ. Prot. 2014, 40, 101–113. [Google Scholar] [CrossRef]
- Nwaichi, E.O.; Frac, M.; Nwoha, P.A.; Eragbor, P. Enhanced Phytoremediation of Crude Oil-Polluted Soil by Four Plant Species: Effect of Inorganic and Organic Bioaugumentation. Int. J. Phytoremediat. 2015, 17, 1253–1261. [Google Scholar] [CrossRef] [PubMed]
- Stephenson, D.; Stephenson, T. Bioaugmentation for enhancing biological wastewater treatment. Biotechnol. Adv. 1992, 10, 549–559. [Google Scholar] [CrossRef]
- Schneider, I.D.; Topalova, Y.I. Effect of bioaugmentation on anaerobic wastewater treatment in the dairy industry. J. Dairy Sci. 2011, 94, 4389–4397. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Yang, G.; Chen, J.; Liu, Y. Enhanced Biodegradability of Aerobic Sludge by Bioaugmentation for Pulping Effluent Treatment. BioResources 2014, 9, 2143–2153. [Google Scholar] [CrossRef]
- Patureau, D.; Helloin, E.; Rustrian, E.; Bouchez, T.; Delgenes, J.P.; Moletta, R. Combined phosphate and nitrogen removal in a sequencing batch reactor using the aerobic denitrifier, Microvirgula aerodenitrificans. Water Res. 2001, 35, 189–197. [Google Scholar] [CrossRef]
- Jianlong, W.; Xiangchun, Q.; Libo, W.; Yi, Q.; Hegemann, W. Bioaugmentation as a tool to enhance the removal of refractory compound in coke plant wastewater. Process Biochem. 2002, 38, 777–781. [Google Scholar] [CrossRef]
- Fouratt, M.A.; Rhodes, J.S.; Smithers, C.M.; Love, N.G.; Stevens, A.M. Application of temperature gradient gel electrophoresis to the characterization of a nitrifying bioaugmentation product. FEMS Microbiol. Ecol. 2003, 43, 277–286. [Google Scholar] [CrossRef] [PubMed]
- Guo, J.; Wang, J.; Cui, D.; Wang, L.; Ma, F.; Chang, C.-C.; Yang, J. Application of bioaugmentation in the rapid start-up and stable operation of biological processes for municipal wastewater treatment at low temperatures. Bioresour. Technol. 2010, 101, 6622–6629. [Google Scholar] [CrossRef] [PubMed]
- Schauer-Gimenez, A.E.; Zitomer, D.H.; Maki, J.S.; Struble, C.A. Bioaugmentation for improved recovery of anaerobic digesters after toxicant exposure. Water Res. 2010, 44, 3555–3564. [Google Scholar] [CrossRef] [PubMed]
- Mannucci, A.; Munz, G.; Mori, G.; Makinia, J.; Lubello, C.; Oleszkiewicz, J.A. Modeling bioaugmentation with nitrifiers in membrane bioreactors. Water Sci. Technol. 2015, 71, 15–21. [Google Scholar] [CrossRef] [PubMed]
- Van Limbergen, V.; Top, E.M.; Verstraete, W. Bioaugmentation in activated sludge: Current features and future perspectives. Appl. Microbiol. Biotechnol. 1998, 50, 16–23. [Google Scholar] [CrossRef]
- Łagód, G.; Chomczyńska, M.; Montusiewicz, A.; Malicki, J.; Bieganowski, A. Proposal of measurement and visualization methods for dominance structures in the saprobe communities. Ecol. Chem. Eng. 2009, 16, 369–377. [Google Scholar]
- Bieganowski, A.; Łagód, G.; Ryżak, M.; Montusiewicz, A.; Chomczyńska, M.; Sochan, A. Measurement of activated sludge particle diameters using laser diffraction method. Ecol. Chem. Eng. 2012, 19, 597–608. [Google Scholar] [CrossRef][Green Version]
- Bartrolí, A.; Carrera, J.; Pérez, J. Bioaugmentation as a tool for improving the start-up and stability of a pilot-scale partial nitrification biofilm airlift reactor. Bioresour. Technol. 2011, 102, 4370–4375. [Google Scholar] [CrossRef] [PubMed]
- Babko, R.; Kuzmina, T.; Jaromin-Gleń, K.; Bieganowski, A. Bioindication assessment of activated sludge adaptation in lab-scale experiment. Ecol. Chem. Eng. 2014, 21. [Google Scholar] [CrossRef]
- Saravanane, R.; Murthy, D.V.S.; Krishnaiah, K. Bioaugmentation and treatment of cephalexin drug-based pharmaceutical effluent in an upflow anaerobic fluidized bed system. Bioresour. Technol. 2001, 76, 279–281. [Google Scholar] [CrossRef]
- Duran, M.; Tepe, N.; Yurtsever, D.; Punzi, V.L.; Bruno, C.; Mehta, R.J. Bioaugmenting anaerobic digestion of biosolids with selected strains of Bacillus, Pseudomonas, and Actinomycetes species for increased methanogenesis and odor control. Appl. Microbiol. Biotechnol. 2006, 73, 960–966. [Google Scholar] [CrossRef] [PubMed]
- Tepe, N.; Yurtsever, D.; Mehta, R.J.; Bruno, C.; Punzi, V.L.; Duran, M. Odor control during post-digestion processing of biosolids through bioaugmentation of anaerobic digestion. Water Sci. Technol. J. Int. Assoc. Water Pollut. Res. 2008, 57, 589–594. [Google Scholar] [CrossRef] [PubMed]
- Jun, H.B.; Park, S.M.; Park, N.B.; Lee, S.H. Nitrogen removal and sludge reduction in a symbiotic activated sludge system between anaerobic archaea and bacteria. Water Sci. Technol. 2004, 50, 189–197. [Google Scholar] [PubMed]
- Anielak, A.M.; Polus, M. Kinetics of the sewage treatment involving an archaea-enriched system SBR, in Polish. Przem. Chem. 2015, 94, 1485–1487. [Google Scholar]
- Anielak, A.M.; Żaba, T.; Polus, M.; Beńko, P.; Bochnia, T.; Łuszczek, B. The effectiveness of muncipal sewage treatmentwith archaea in sequence biological reactorsand Bardenpho systems, in Polish. Przem. Chem. 2016, 95, 314–319. [Google Scholar] [CrossRef]
- Polus, M.; Anielak, A.M. The use of Archaea in the bioaugmentation of activated sludge as a method for the biological removal of nitrogen compounds. Tech. Trans. 2017, 5, 83–95. [Google Scholar]
- Szaja, A.; Łagód, G.; Drewnowski, J.; Sabba, F. Bioaugmentation of a sequencing batch reactor with Archaea for the treatment of reject water. J. Water Chem. Technol. 2016, 38, 238–243. [Google Scholar] [CrossRef]
- Park, H.-D.; Wells, G.F.; Bae, H.; Criddle, C.S.; Francis, C.A. Occurrence of Ammonia-Oxidizing Archaea in Wastewater Treatment Plant Bioreactors. Appl. Environ. Microbiol. 2006, 72, 5643–5647. [Google Scholar] [CrossRef] [PubMed]
- Limpiyakorn, T.; Sonthiphand, P.; Rongsayamanont, C.; Polprasert, C. Abundance of amoA genes of ammonia-oxidizing archaea and bacteria in activated sludge of full-scale wastewater treatment plants. Bioresour. Technol. 2011, 102, 3694–3701. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.L.; Xie, W.; Martin-Cuadrado, A.-B.; Rodriguez-Valera, F. Marine Group II Archaea, potentially important players in the global ocean carbon cycle. Front. Microbiol. 2015, 6, 1108. [Google Scholar] [CrossRef] [PubMed]
- Gray, N.D.; Miskin, I.P.; Kornilova, O.; Curtis, T.P.; Head, I.M. Occurrence and activity of Archaea in aerated activated sludge wastewater treatment plants. Environ. Microbiol. 2002, 4, 158–168. [Google Scholar] [CrossRef] [PubMed]
- Fredriksson, N.J.; Hermansson, M.; Wilén, B.-M. Diversity and dynamics of Archaea in an activated sludge wastewater treatment plant. BMC Microbiol. 2012, 12, 140. [Google Scholar] [CrossRef] [PubMed]
- Polus, M.; Anielak, A.M.; Zielina, M.; Ulman, E. Application of membrane processes for archaea isolation. In Monographs of the Environmental Engineering Committee; Konieczny, K., Korus, I., Eds.; Polish Academy of Sciences: Warsaw, Poland, 2014; pp. 381–391. [Google Scholar]
- Abell, G.C.J.; Ross, D.J.; Keane, J.; Holmes, B.H.; Robert, S.S.; Keough, M.J.; Eyre, B.D.; Volkman, J.K. Niche differentiation of ammonia-oxidising archaea (AOA) and bacteria (AOB) in response to paper and pulp mill effluent. Microb. Ecol. 2014, 67, 758–768. [Google Scholar] [CrossRef] [PubMed]
- ArchaeaSolutions, Inc. Available online: http://archaeasolutions.com/ (accessed on 19 March 2018).
- Tchobanoglous, G.; Burton, F.L.; Stensel, H.D. Wastewater Engineering: Treatment and Reuse; Metcalf & Eddy, McGraw-Hill Education: New York, NY, USA, 2004; ISBN 978-0-07-041878-3. [Google Scholar]
- Spanjers, H.; Vanrolleghem, P. Respirometry as a tool for rapid characterization of wastewater and activated sludge. Water Sci. Technol. 1995, 31, 105–114. [Google Scholar] [CrossRef]
- Young, J. Oxygen Uptake Rate as a Monitoring and Control Parameter for Activated Sludge Processes. In Proceedings of the 1999 WEF/Indiana WPCA/Purdue University Conference on Industrial Waste, Alexandria, VA, USA, 21–22 May 1999. [Google Scholar]
- Rybicki, S.M.; Cimochowicz-Rybicka, M. Dimensioning of Digestion Chamber for Upgrading of Gas Recovery at Wastewater Treatment Plant. Arch. Environ. Prot. 2014, 39, 105–112. [Google Scholar] [CrossRef][Green Version]
- Drewnowski, J. The impact of slowly biodegradable organic compounds on the oxygen uptake rate in activated sludge systems. Water Sci. Technol. 2014, 69, 1136–1144. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.Y.C.; Cheng, M.-D.; Mueller, J.T. Oxygen uptake rates for determining microbial activity and application. Water Res. 1985, 19, 373–381. [Google Scholar] [CrossRef]
- Zubrowska-Sudol, M.; Walczak, J. Effects of mechanical disintegration of activated sludge on the activity of nitrifying and denitrifying bacteria and phosphorus accumulating organisms. Water Res. 2014, 61, 200–209. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Trela, J.; Zubrowska-Sudol, M.; Plaza, E. Intermittent aeration in one-stage partial nitritation/anammox process. Ecol. Eng. 2015, 75, 413–420. [Google Scholar] [CrossRef]
- Gaudy, A.; Ekambaram, A.; Rozich, A. A respirometric method for biokinetic characterization of toxic waste. In Proceedings of the 43rd Purdue Industrial Waste Conference, West Lafayette, IN, USA, 10–12 May 1988. [Google Scholar]
- Çokgör, E.U.; Sözen, S.; Orhon, D.; Henze, M. Respirometric analysis of activated sludge behaviour—I. Assessment of the readily biodegradable substrate. Water Res. 1998, 32, 461–475. [Google Scholar] [CrossRef]
- Pollice, A.; Laera, G.; Blonda, M. Biomass growth and activity in a membrane bioreactor with complete sludge retention. Water Res. 2004, 38, 1799–1808. [Google Scholar] [CrossRef] [PubMed]
- Welch, B.L. The generalisation of student’s problems when several different population variances are involved. Biometrika 1947, 34, 28–35. [Google Scholar] [PubMed]
- Henze, M.; Harremoes, P.; Cour Jansen, J.; Arvin, E. Wastewater Treatment—Biological and Chemical Processes; Springer: Berlin, Germany, 2002. [Google Scholar]
- Van Haandel, C.; Van Der Lubbe, J. Handbook of Biological Wastewater Treatment: Design and Optimization of Activated Sludge Systems, 2nd ed.; IWA Publishing: London, UK, 2012. [Google Scholar]
- Puig, S.; Corominas, L.; Colomer, J.; Balaguer, M.D.; Colprim, J. On-line oxygen uptake rate as a new tool for monitoring and controlling the SBR process. In Computer Aided Chemical Engineering; Puigjaner, L., Espuña, A., Eds.; Elsevier: New York, NY, USA, 2005; pp. 1291–1296. [Google Scholar]
- Avcioglu, E.; Orhon, D.; Sözen, S. A new method for the assessment of heterotrophic endogenous respiration rate under aerobic and anoxic conditions. Water Sci. Technol. 1998, 38, 95–103. [Google Scholar] [CrossRef]
- Cokgor, E.U.; Okutman Tas, D.; Zengin, G.E.; Insel, G. Effect of stabilization on biomass activity. J. Biotechnol. 2012, 157, 547–553. [Google Scholar] [CrossRef] [PubMed]
- Bernard, S.; Gray, N.F. Aerobic digestion of pharmaceutical and domestic wastewater sludges at ambient temperature. Water Res. 2000, 34, 725–734. [Google Scholar] [CrossRef]
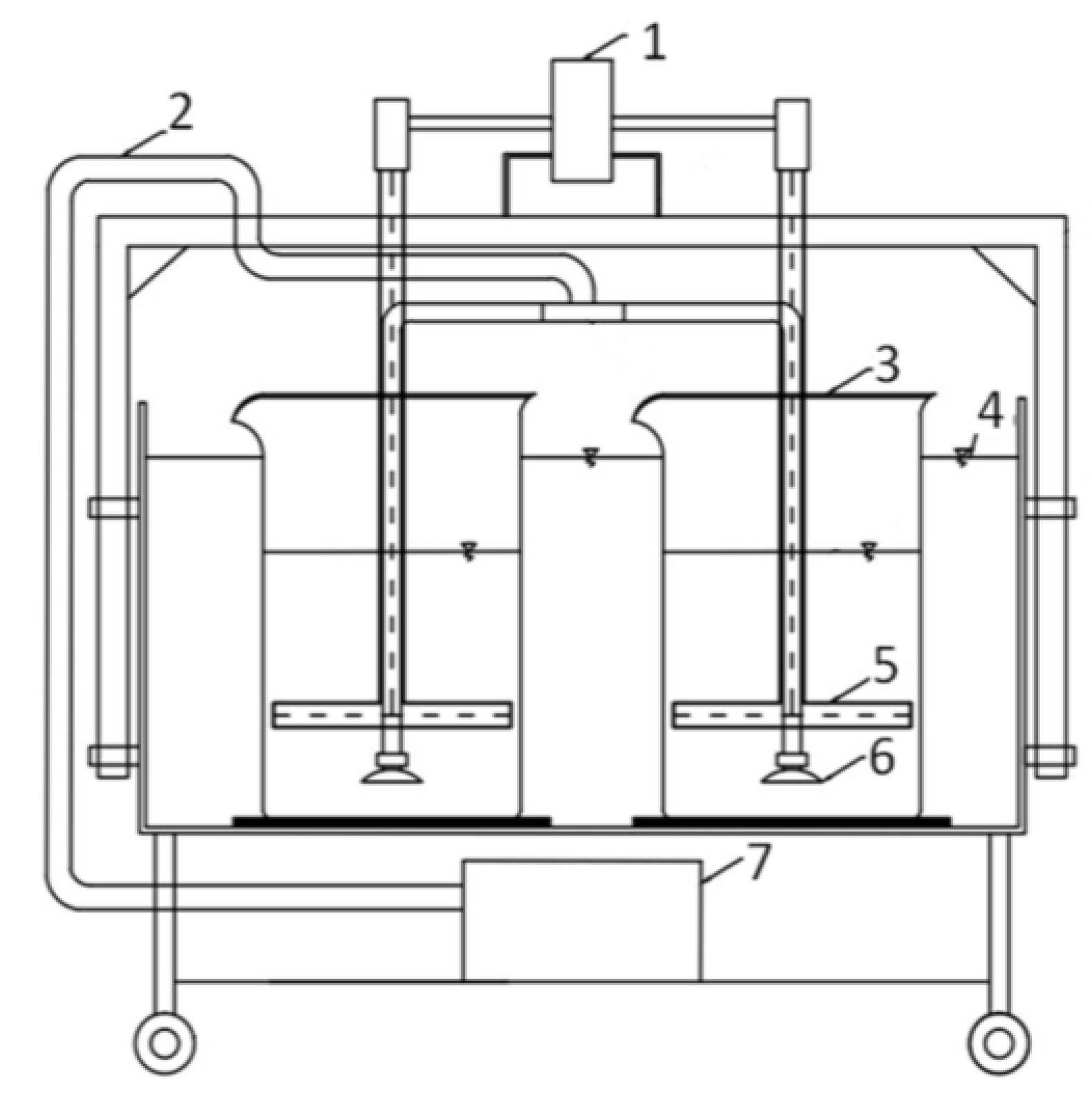

| Parameter | Unit | Mean Value ± Standard Deviation |
|---|---|---|
| Chemical oxygen demand (COD) | mg∙dm−3 | 899 ± 81.0 |
| Total suspended solids (TSS) | mg∙dm−3 | 297 ± 24.3 |
| Total nitrogen (TN) | mg∙dm−3 | 105 ± 7.8 |
| Ammonia nitrogen (N-NH4+) | mg∙dm−3 | 88.2 ± 2.2 |
| Total phosphorus (TP) | mg∙dm−3 | 11.8 ± 0.7 |
| pH | pH | 7.8 ± 0.3 |
| Parameter | Unit | Mean Value ± Standard Deviation |
|---|---|---|
| Chemical oxygen demand (COD) | mg∙dm−3 | 22 ± 1.0 |
| Volatile fatty acids (VFA) | mg∙dm−3 | 21 ± 1.0 |
| Total solids (TS) | mg∙g−1 | 0.47 ± 1.0 |
| Volatile solids (VS) | mg∙g−1 | 0.042 ± 1.0 |
| Total suspended solids (TSS) | mg∙dm−3 | 6 ± 1.0 |
| Total nitrogen (TN) | mg∙dm−3 | 75 ± 1.0 |
| Ammonia nitrogen (N-NH4+) | mg∙dm−3 | 0.4 ± 0.02 |
| Total phosphorus (TP) | mg∙dm−3 | 0.17 ± 0.03 |
| Alkalinity | mg∙dm−3 | 330 ± 0.03 |
| pH | pH | 7.16 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Szaja, A.; Łagód, G.; Jaromin-Gleń, K.; Montusiewicz, A. The Effect of Bioaugmentation with Archaea on the Oxygen Uptake Rate in a Sequencing Batch Reactor. Water 2018, 10, 575. https://doi.org/10.3390/w10050575
Szaja A, Łagód G, Jaromin-Gleń K, Montusiewicz A. The Effect of Bioaugmentation with Archaea on the Oxygen Uptake Rate in a Sequencing Batch Reactor. Water. 2018; 10(5):575. https://doi.org/10.3390/w10050575
Chicago/Turabian StyleSzaja, Aleksandra, Grzegorz Łagód, Katarzyna Jaromin-Gleń, and Agnieszka Montusiewicz. 2018. "The Effect of Bioaugmentation with Archaea on the Oxygen Uptake Rate in a Sequencing Batch Reactor" Water 10, no. 5: 575. https://doi.org/10.3390/w10050575
APA StyleSzaja, A., Łagód, G., Jaromin-Gleń, K., & Montusiewicz, A. (2018). The Effect of Bioaugmentation with Archaea on the Oxygen Uptake Rate in a Sequencing Batch Reactor. Water, 10(5), 575. https://doi.org/10.3390/w10050575






