Near-Bed Monitoring of Suspended Sediment during a Major Flood Event Highlights Deficiencies in Existing Event-Loading Estimates
Abstract
:1. Introduction
2. Materials and Methods
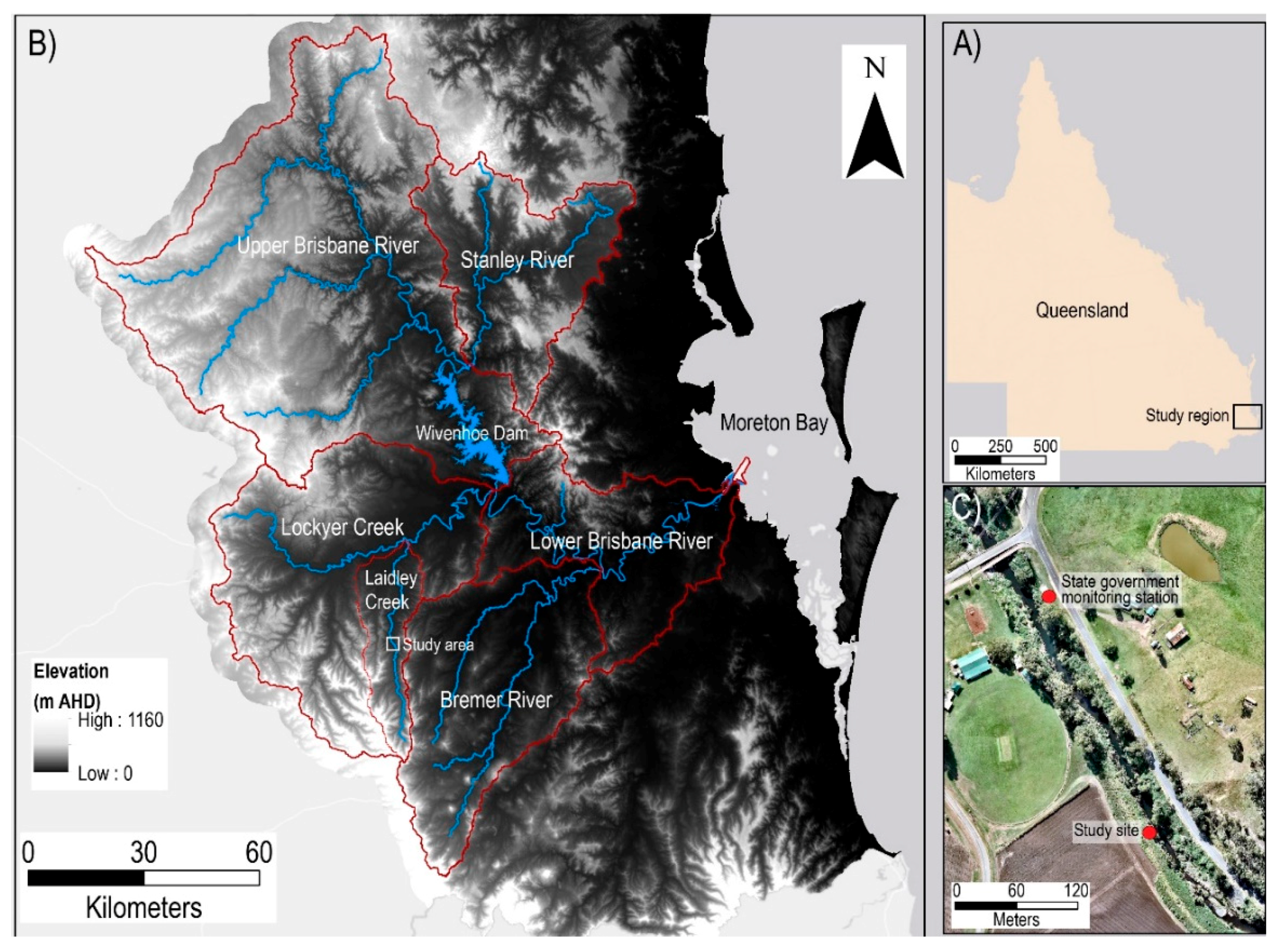
2.1. Study Site Description
2.2. Field Monitoring
2.3. Laboratory Analysis
2.4. Imagery Analysis
3. Results
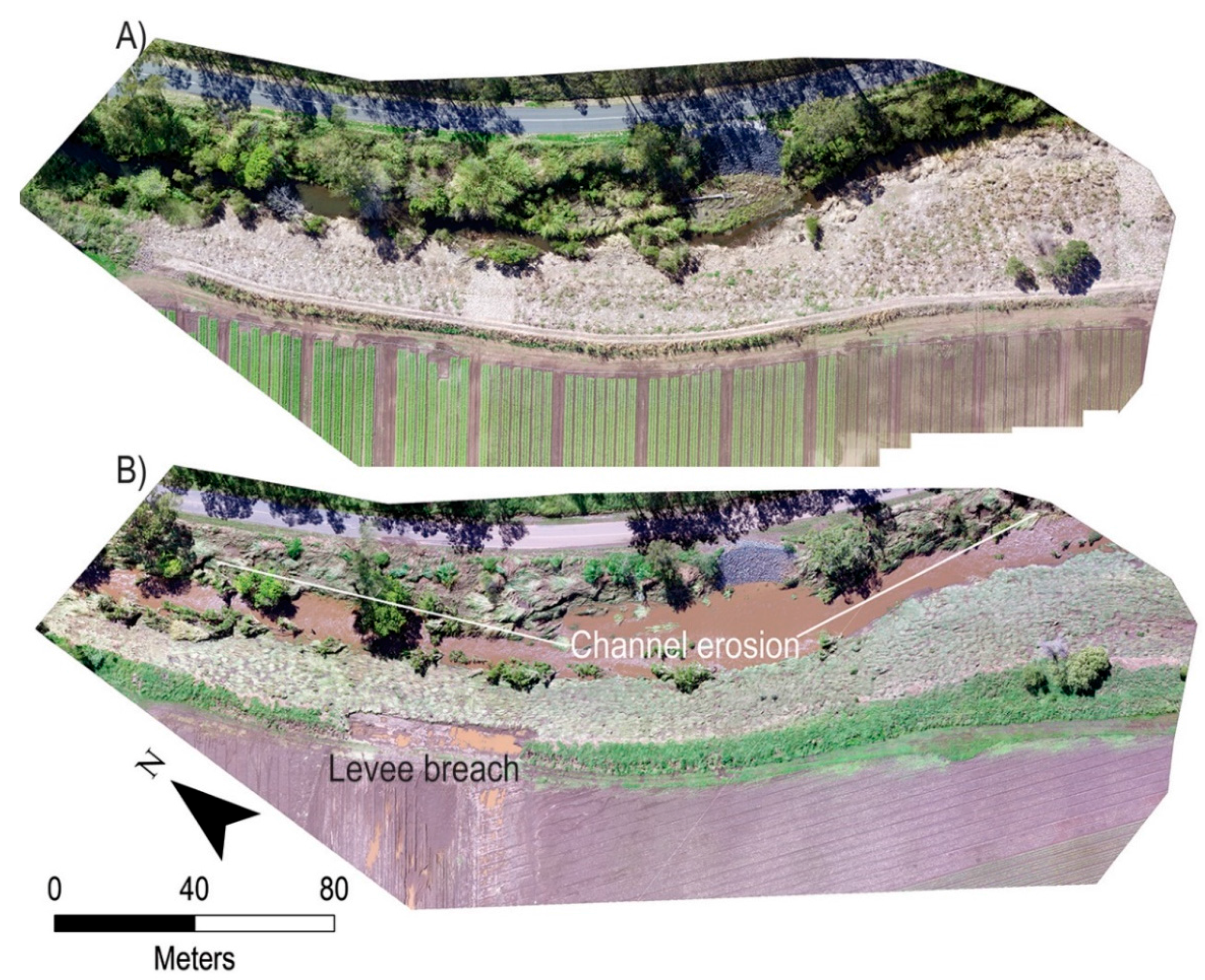
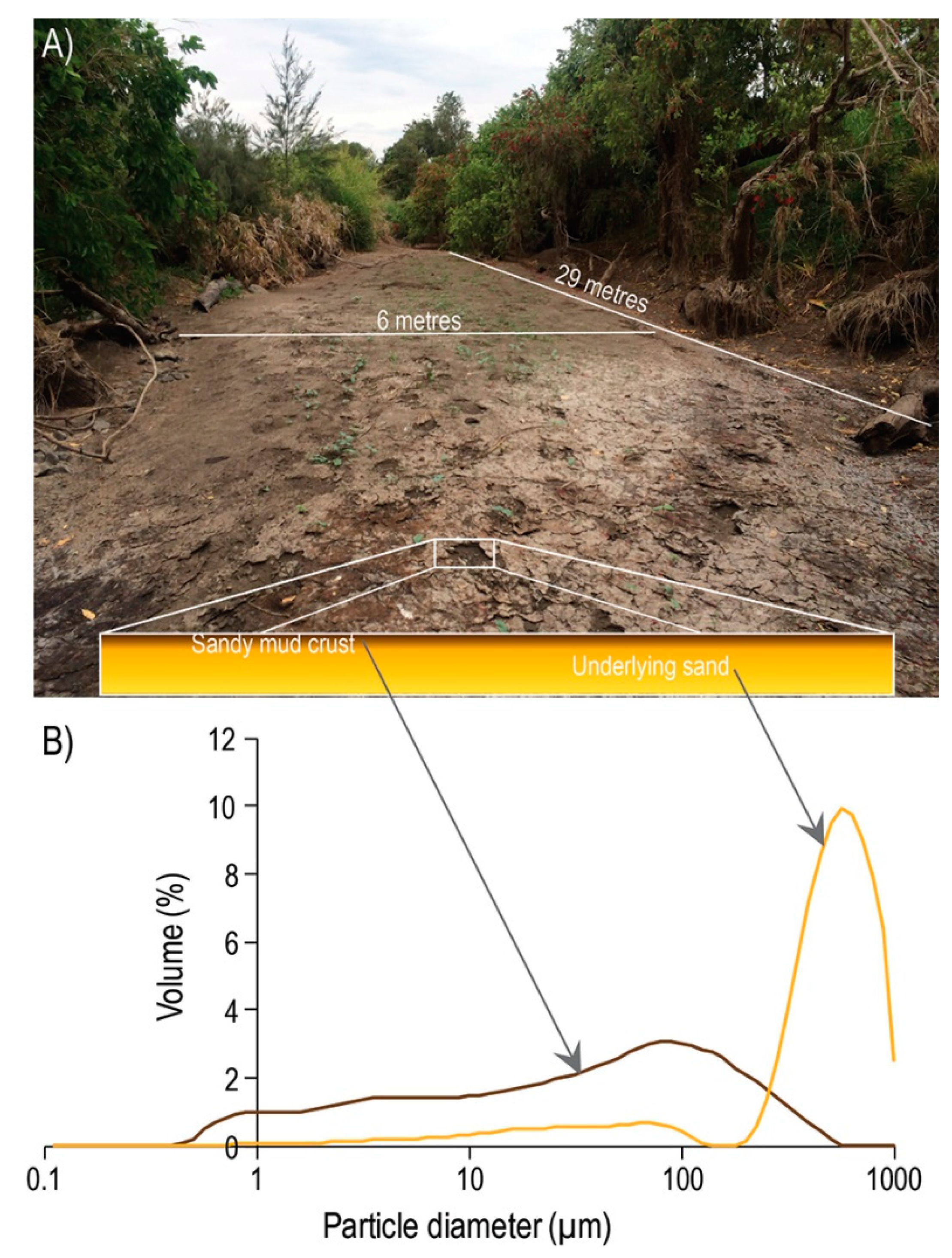
3.1. Time-Lapse and Drone Imagery
3.2. Suspended Sediment Dynamics
3.3. First Flush Phenomenon
4. Discussion
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
Appendix A

Appendix A.1. Sensor Assembly
- Drill a 21 mm hole in the top of two 50 mm PVC caps (Figure A2C) corresponding to the diameter of the lower section of the Amphenol Sensor (Figure A2A). Then, drill eight (10 mm) holes evenly spaced around the cap flange and four holes (3.5 mm) in the inner edges of one cap, cleaning all burrs. Ensure both caps are clean and glue the two caps back to back with PVC cement/glue.Figure A2. (A) Amphenol sensor. (B) Third PVC cap and cable gland. (C) Two caps produced in step 1 for forming the bottom of the sensor assembly.Figure A2. (A) Amphenol sensor. (B) Third PVC cap and cable gland. (C) Two caps produced in step 1 for forming the bottom of the sensor assembly.
- Measure the diameter of the waterproof cable glands threaded section (≈19 mm) and drill the corresponding size hole in the centre of the third 50 mm PVC Cap. Clean the edges of the hole, ensuring there is no burr or raised markings. Secure the cable gland in the drilled hole (Figure A2B). Hot glue can be used on the nut of the gland to provide additional support when tightening.
- Cut a 75 mm length of 50 mm diameter PVC pipe, clean the edges of the cut.
- Cut the appropriate length of cable for the proposed deployment, noting that the length should be 500 mm greater than required.
- Strip 50 mm of outer black coating from both ends of the sensor cable. Then, strip a small amount (≈10 mm) from the red, yellow, and internal black wire covers. Cut the green cable off to the same level at the outer black coating (Figure A3A).
- Twist the ends of the exposed wires separately and apply lead-free solder.
- Remove the black cap from the Amphenol Sensor and then remove the sensor board from the clear housing. Cut the white plastic plug off the sensor board, and then solder the sensor cable wires to the uncovered pins (Figure A3A), covering connections in heat-shrink or electrical tape. Place the sensor board back into the housing and reattach the black cap, ensuring that the sensor cable exits through the hole. Seal the cable gap with hot glue.Figure A3. (A) Illustrating the cable soldered on the sensor board, ensuring the solder does not bridge between the sensors. The wires connected as; Red to A/C, Black to K, and Yellow to E. (B) Epoxy-filled sensor housing. The harder epoxy is first to provide a dense base, and hold the cable firmly in place to prevent movement of the soldered joint. (C) Silicon filling is used in the top to provide a more flexible housing fill so the cable can be positioned for the top cap. (D) Completed sensor assembly.Figure A3. (A) Illustrating the cable soldered on the sensor board, ensuring the solder does not bridge between the sensors. The wires connected as; Red to A/C, Black to K, and Yellow to E. (B) Epoxy-filled sensor housing. The harder epoxy is first to provide a dense base, and hold the cable firmly in place to prevent movement of the soldered joint. (C) Silicon filling is used in the top to provide a more flexible housing fill so the cable can be positioned for the top cap. (D) Completed sensor assembly.
- Place the sensor into the glued caps assembled in Step 1, ensuring that the lower section of the sensor is the on the side of shroud. Hot glue the sensor in this position, and slide the pipe down the cable. Glue the lower caps and the pipe together with PVC cement/glue (Figure A3D).
- Position so the opening in the pipe is facing up, and prepare the Araldite epoxy as per the manufacturer’s instructions. Pour epoxy into the pipe until 10–20 mm above the black cap, and agitate to remove the air voids while topping up with epoxy as required. Sit for 24–48 h until epoxy is fully set (Figure A3B).
- Fill the remaining void area with waterproof silicon (Figure A3C), and then pull the sensor cable through the cable gland. Use PVC cement/glue to affix the top cap to the pipe, and then tighten the gland around the cable to seal the assembly.
Appendix A.2. Case and Power Supply Assembly
- 11.
- Drill a hole corresponding to the diameter of the cable gland threaded section in the acrylonitrile butadiene styrene (ABS) case on the opposite side to the attachment strap. Clean burrs and remove the inner foam lining around the hole. Affix the second cable gland in the hole using hot glue to prevent excessive spinning of the nut.
- 12.
- Collect the battery holder and solder the red jumper wire to the red battery holder wire and the black jumper wire to the black battery holder wire. Cover the joints in heat-shrink or electrical tape.
- 13.
- Place batteries in the holder and test the voltage (7.2 V).
Appendix A.3. Electronics Board Assembly
- 14.
- Solder the stacking header to the outer holes in the prototyping board, and ensure that this can bridge to a DRFudino board (Figure A4B).
- 15.
- Once the stacking headers are in place, the inner headers are soldered in an upright orientation; this allows for easy wiring of the circuit.
- 16.
- Finally, the electronic components are added following the wiring pattern in Figure A4A.
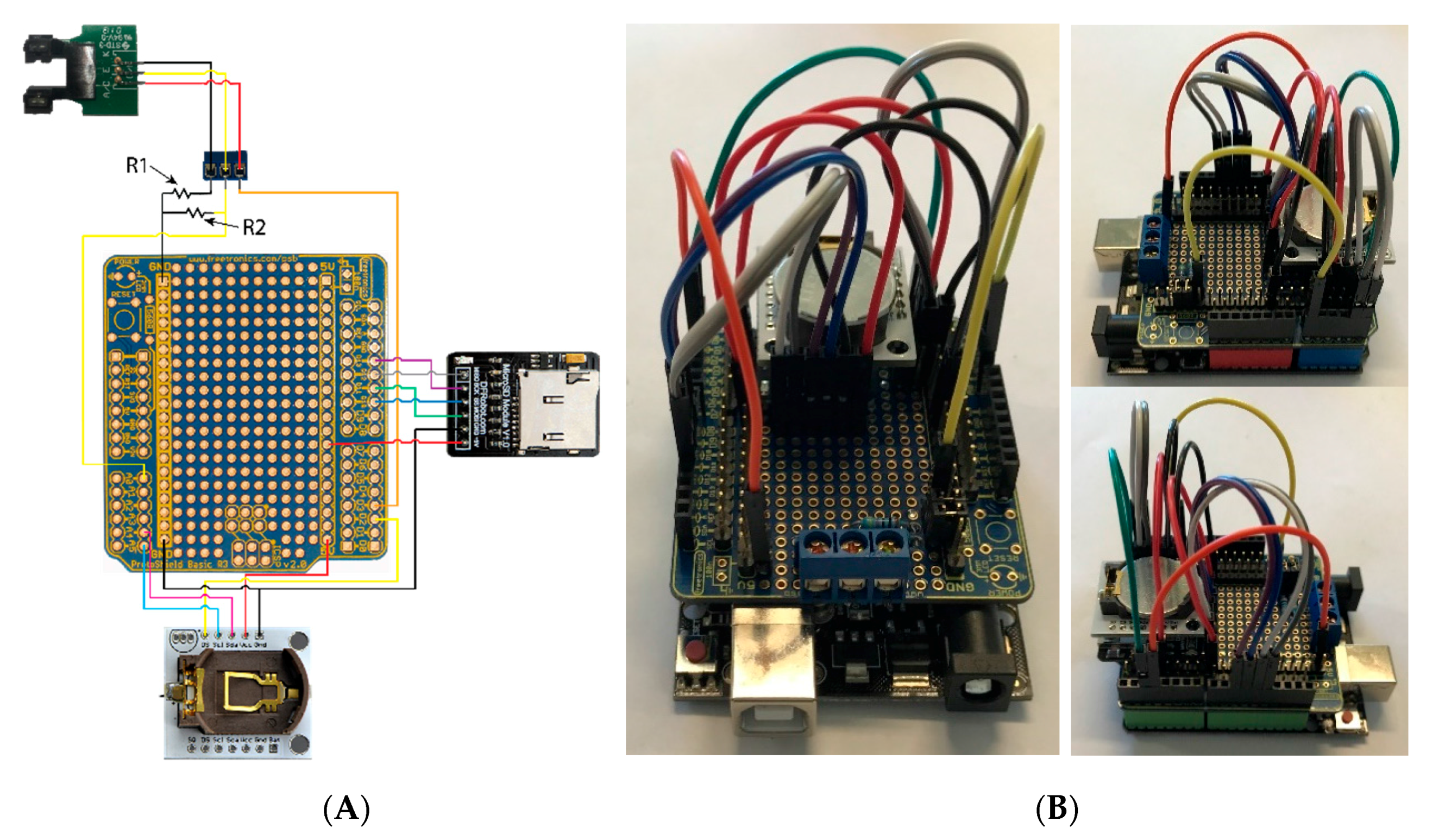
Appendix A.4. Final Assembly
- 17.
- Place components in the ABS housing case (Figure A1).
- 18.
- Connect the battery to the electronics board by plugging the red wire from the battery pack to Vin and the black wire to ground pin (GND). Ensure the red light on the electronics board lights up, then remove the battery wires until deployment.
- 19.
- Run the sensor cable through the cable gland in the housing and connect the wires as shown (Figure A4A). Before tightening the gland around the cable, ensure that there is extra cable length for board movement within the case.
Appendix A.5. Programing
- 20.
- Download the Arduino program from the Arduino website.
- 21.
- Install the relevant libraries, outlined in Code S1.
- 22.
- Connect the electronics board to the computer.
- 23.
- Set the real-time clocks using the corresponding code (Code S2).
- 24.
- Upload the sensor code to the board (Code S1). Note that the monitoring frequency can be adjusted within the code, the default is 10 s of monitoring.
- 25.
- Check that the code is running, the time is correct, and the sensor functions correctly (bits reduce when the sensor is blocked) by opening the serial monitoring portal.
Appendix A.6. Deployment
- 26.
- Connect the battery pack to the electronics board (Red: Vin, Black: Ground)
- 27.
- To check that the sensor is functioning correctly, log 3–5 monitoring intervals. Disconnect the battery, remove the storage device (SD) card, and check readings. If functioning correctly, reinsert the SD card and then reattach the battery.
Appendix A.7. Calibration
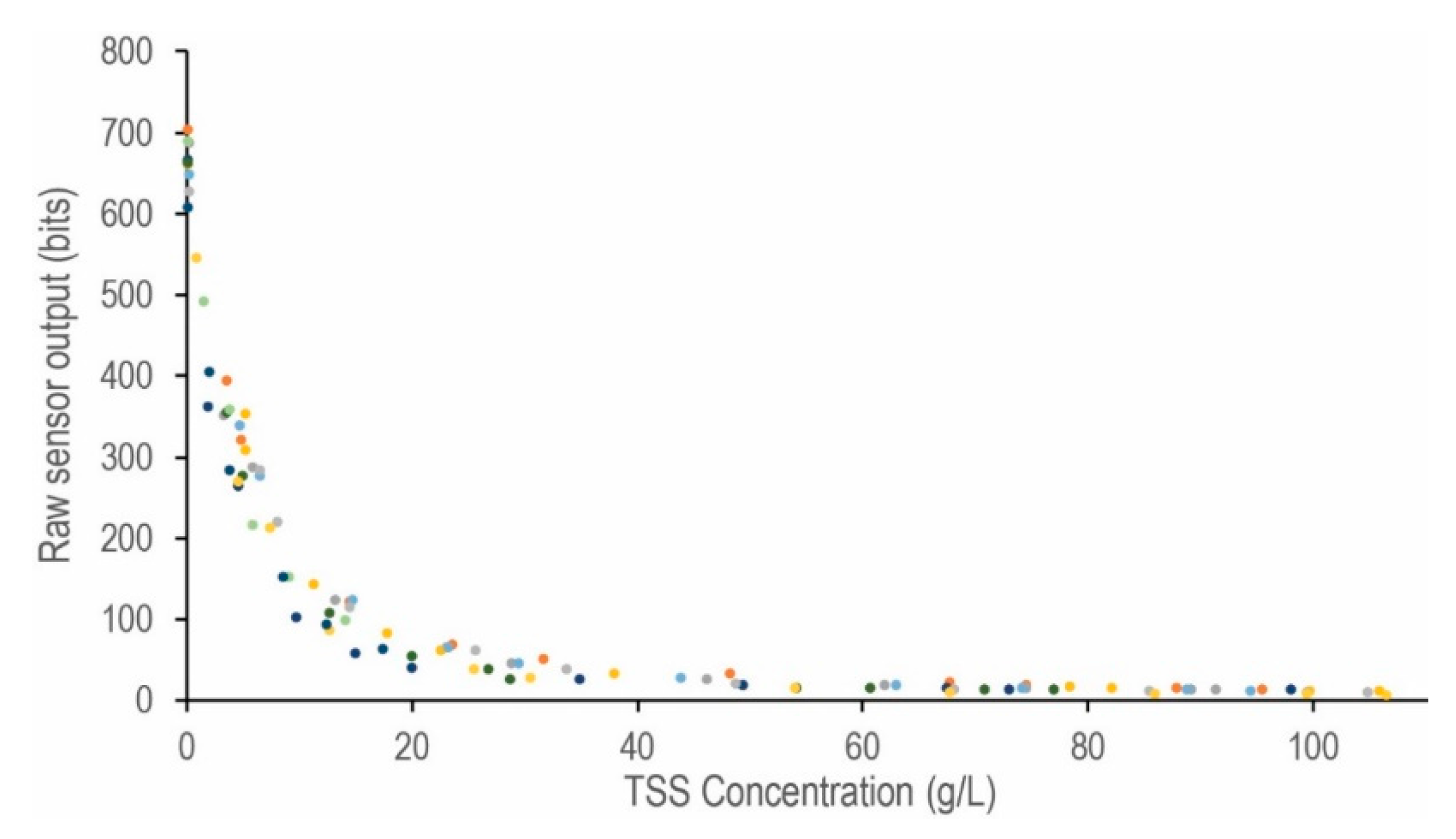
Appendix A.8. Required Parts
| Qty | Supplier | Part # | Cost (US$) | |
|---|---|---|---|---|
| Electronics Board | ||||
| DFRduino UNO R3—Arduino Compatible | 1 | DFRobot | DFR0216 | $15.19 |
| MicroSD card module for Arduino | 1 | DFRobot | DFR0229 | $3.97 |
| Real Time Clock Module (DS1307) V1.1 | 1 | DFRobot | DFR0151 | $3.36 |
| 10 Pcs 40 pin Headers Straight | 1 | DFRobot | FIT0084 | $2.37 |
| 4.7 k Ohm 0.5 Watt Metal Film Resistor | 1 | Jaycar | RR0588 | $0.42 |
| 470 Ohm 0.5 Watt Metal Film Resistor | 1 | Jaycar | RR0564 | $0.42 |
| 2 GB Class 4 microSD Card | 1 | Jaycar | XC4998 | $7.60 |
| 3 Way PCB Mount Screw Terminal—5 mm Pitch | 1 | Jaycar | HM3173 | $1.18 |
| ProtoShield Basic for Arduino | 1 | Freetronics | PSB | $2.29 |
| Shield stacking headers for arduino (R3 compatible) | 1 | Adafruit | 85 | $1.49 |
| Sub-total | $38.28 | |||
| Sensor Assembly | ||||
| Amphenol Advanced Sensors TSD-10 | 1 | Mouser Electronics | TSD-10 | $8.24 |
| 50 mm PVC DWV Pipe | 1 m | Bunnings | 4770087 | $5.40 |
| 50 mm PVC Caps | 3 | Bunnings | 4750164 | $6.30 |
| Multipurpose Water Proof Silicon | 1 | Bunnings | 1232705 | $4.08 |
| 5–10 mm DIA IP68 Waterproof Cable Glands—Pk.2 | 1 | Jaycar | HP0727 | $4.54 |
| Sealer Araldite Epoxy K106 | 1 | Blackwoods | 00405455 | $22.14 |
| 14/0.20 Black 4 Core Security Cable | 5 m | Altronics | W2355 | $8.40 |
| Sub-total | $59.11 | |||
| Housing and Power Supply | ||||
| Case Abs Blk IPX8 210 × 120 × 90 mm | 1 | Jaycar | HB6425 | $26.68 |
| 4 × D CELL 2 Rows of 2 battery holder | 1 | Jaycar | PH9222 | $2.25 |
| D cell Alkaline Batteries | 4 | $6.07 | ||
| Sub-total | $35.00 | |||
| Consumables | ||||
| Hot glue | ||||
| Lead free solder | ||||
| PVC cement for pressurised pipes | ||||
| Jumper shunts (links) | 2 | Jaycar | HM3240 | |
| Premium Female/Male ‘Extension’ Jumper Wires—40 × 3′′ (75 mm) | 1 | Adafruit | 825 | |
| Premium Female/Female Jumper Wires—40 × 3′′ (75 mm) | 1 | Adafruit | 794 | |
| Heat-shrink or electrical tape | ||||
| Sub-total | $15.27 | |||
| Total | $147.66 | |||
| Required Tools | ||||
| Soldering iron | ||||
| Hack saw | ||||
| Drill | ||||
| Drill bit (3.5 mm, 10 mm, 19 mm, and 21 mm) | ||||
| Wire strippers and cutters | ||||
| Phillips head screw driver #1, 2.5 mm shaft | ||||
| Snap-off or retracting knife (9 or 18 mm) | ||||
| Measurement tools (Ruler, Calipers) | ||||
| Multimeter |
References
- Pelletier, J.D. A spatially distributed model for the long-term suspended sediment discharge and delivery ratio of drainage basins. J. Geophys. Res. Earth Surf. 2012, 117. [Google Scholar] [CrossRef]
- Galy, V.; Peucker-Ehrenbrink, B.; Eglinton, T. Global carbon export from the terrestrial biosphere controlled by erosion. Nature 2015, 521, 204–207. [Google Scholar] [CrossRef] [PubMed]
- Shellberg, J.G.; Brooks, A.P.; Rose, C.W. Sediment production and yield from an alluvial gully in northern Queensland, Australia. Earth Surf. Process. Landf. 2013, 38, 1765–1778. [Google Scholar] [CrossRef]
- Wallbrink, P.J. Quantifying the erosion processes and land-uses which dominate fine sediment supply to Moreton Bay, Southeast Queensland, Australia. J. Environ. Radioact. 2004, 76, 67–80. [Google Scholar] [CrossRef] [PubMed]
- Olley, J.; Burton, J.; Smolders, K.; Pantus, F.; Pietsch, T. The application of fallout radionuclides to determine the dominant erosion process in water supply catchments of subtropical South-east Queensland, Australia. Hydrol. Process. 2013, 27, 885–895. [Google Scholar] [CrossRef]
- Coates-Marnane, J.; Olley, J.; Burton, J.; Sharma, A. Catchment clearing accelerates the infilling of a shallow subtropical bay in east coast Australia. Estuar. Coast. Shelf Sci. 2016, 174, 27–40. [Google Scholar] [CrossRef]
- Lockington, J.R.; Albert, S.; Fisher, P.L.; Gibbes, B.R.; Maxwell, P.S.; Grinham, A.R. Dramatic increase in mud distribution across a large sub-tropical embayment, Moreton Bay, Australia. Mar. Pollut. Bull. 2016, 116, 491–497. [Google Scholar] [CrossRef] [PubMed]
- Grinham, A.; Gibbes, B.; Gale, D.; Watkinson, A.; Bartkow, M. Extreme rainfall and drinking water quality: A regional perspective. In Water Pollution XI; Brebbia, C.A., Ed.; WIT: Southampton, UK, 2012; Volume 164, pp. 183–194. [Google Scholar]
- Hubble, T.; Docker, B.; Rutherfurd, I. The role of riparian trees in maintaining riverbank stability: A review of Australian experience and practice. Ecol. Eng. 2010, 36, 292–304. [Google Scholar] [CrossRef]
- Olley, J.; Wilkinson, S.; Caitcheon, G.; Read, A. Protecting Moreton Bay: How can we reduce sediment and nutrient loads by 50%. In Proceedings of the 9th International River Symposium, Brisbane, Australia, 4–7 September 2006; Volume 47, p. 19. [Google Scholar]
- Kemp, J.; Olley, J.M.; Ellison, T.; McMahon, J. River response to european settlement in the subtropical Brisbane River, Australia. Anthropocene 2015, 11, 48–60. [Google Scholar] [CrossRef]
- Department of Natural Resource Management. Water Monitoring Information Portal. Available online: https://watermonitoring.information.qld.gov.au (accessed on 25 April 2017).
- Australian Government Bureau of Meteorology (BOM). Queensland Rainfall and River Height Data, Stony Creek rd Gauge. Available online: http://www.bom.gov.au/qld/flood/rain_river.shtml (accessed on 2 April 2017).
- Gian, J. Improving the Testing of Sedimentation Processes—Development of a Large Column Test and Observations of Solid Concentration Using Turbidity Measurements. Bachelor’s Thesis, The Unversity of Queensland, Brisbane, Australia, 2016. [Google Scholar]
- American Public Health Association; American Water Works Association; Water Pollution Federation. Standard Methods for the Examination of Water and Wastewater, 22nd ed.; American Public Health Association: Washinton, DC, USA, 2012. [Google Scholar]
- Sperazza, M.; Moore, J.; Hendrix, M. High-resolution particle size analysis of naturally occurring very fine-grained sediment through laser diffractometry. J. Sediment. Res. 2004, 74, 736–743. [Google Scholar] [CrossRef]
- Folk, R.L. Petrology of Sedimentary Rocks; Hemphill Pub. Co.: Austin, TX, USA, 1974. [Google Scholar]
- Horowitz, A.J. A review of selected inorganic surface water quality-monitoring practices: Are we really measuring what we think, and if so, are we doing it right? Environ. Sci. Technol. 2013, 47, 2471–2486. [Google Scholar] [CrossRef] [PubMed]
- Gray, J.R.; Glysson, G.D.; Turcios, L.M.; Schwarz, G.E. Comparability of suspended-sediment concentration and total suspended solids data. In US Geological Survey Water-Resources Investigations Report 00-4191; U.S. Geological Survey: Reston, VA, USA, 2000. [Google Scholar]
- Orr, D.N.; Turner, R.D.R.; Thomson, B.; Ferguson, B.; Newham, M.; Wallace, R.; Huggins, R.; Severino, Z. South East Queensland Water Quality Statistics for Event Flow and Base Flow Conditions: 2002–2015; Department of Science, Information Technology and Innovation.: Brisbane, Australia, 2017. [Google Scholar]
- Obermann, M.; Rosenwinkel, K.-H.; Tournoud, M.-G. Investigation of first flushes in a medium-sized mediterranean catchment. J. Hydrol. 2009, 373, 405–415. [Google Scholar] [CrossRef]
- Kerr, J.G.; Burford, M.; Olley, J.; Udy, J. Phosphorus sorption in soils and sediments: Implications for phosphate supply to a subtropical river in Southeast Queensland, Australia. Biogeochemistry 2011, 102, 73–85. [Google Scholar] [CrossRef]
- Garzon-Garcia, A.; Olley, J.M.; Bunn, S.E. Controls on carbon and nitrogen export in an eroding catchment of South-eastern Queensland, Australia. Hydrol. Process. 2015, 29, 739–751. [Google Scholar] [CrossRef]
- Dankers, P.J.T. On the Hindered Settling of Suspensions of Mud and Mud-Sand Mixtures. Ph.D. Thesis, Delft University of Technology, Delft, The Netherlands, 2006. [Google Scholar]
- Rijn, L.C.V. Principles of Sedimentation and Erosion Engineering in Rivers, Estuaries and Coastal Seas Leo C. van Rijn; Aqua Publications: Amsterdam, The Netherlands, 2005. [Google Scholar]
- Amy, L.A.; Talling, P.J.; Edmonds, V.O.; Sumner, E.J.; Lesueur, A. An experimental investigation of sand–mud suspension settling behaviour: Implications for bimodal mud contents of submarine flow deposits. Sedimentology 2006, 53, 1411–1434. [Google Scholar] [CrossRef]
- Gettel, M.; Gulliver, J.S.; Kayhanian, M.; DeGroot, G.; Brand, J.; Mohseni, O.; Erickson, A.J. Improving suspended sediment measurements by automatic samplers. J. Environ. Monit. 2011, 13, 2703–2709. [Google Scholar] [CrossRef] [PubMed]
- Devlin, M.; Waterhouse, J.; Brodie, J. Community and connectivity: Summary of a community based monitoring program set up to assess the movement of nutrients and sediments into the Great Barrier Reef during high flow events. Water Sci. Technol. 2001, 43, 121–131. [Google Scholar] [PubMed]
- Selbig, W.R. Characterizing the distribution of particles in urban stormwater: Advancements through improved sampling technology. Urban Water J. 2015, 12, 111–119. [Google Scholar] [CrossRef]
- Trimble, S.W. A sediment budget for Coon Creek basin in the driftless area, Wisconsin, 1853–1977. Am. J. Sci. 1983, 283, 454–474. [Google Scholar] [CrossRef]
- Wallbrink, P.J.; Murray, A.S.; Olley, J.M.; Olive, L.J. Determining sources and transit times of suspended sediment in the Murrumbidgee River, New South Wales, Australia, using fallout 137Cs and 210Pb. Water Resour. Res. 1998, 34, 879–887. [Google Scholar] [CrossRef]
- Dietrich, W.E. Settling velocity of natural particles. Water Resour. Res. 1982, 18, 1615–1626. [Google Scholar] [CrossRef]
- Beylich, A.A. Quantitative studies on sediment fluxes and sediment budgets in changing cold environments—Potential and expected benefit of coordinated data exchange and the unification of methods. Landf. Anal. 2007, 5, 9–10. [Google Scholar]
- Walling, D. Suspended sediment and solute response characteristics of the River Exe, Devon, England. Res. Fluv. Syst. 1978, 169–197. [Google Scholar]
- Walling, D. Suspended sediment and solute yields from a small catchment prior to urbanization. Fluv. Process. Instrum. Watersheds 1974, 6, 169–192. [Google Scholar]
- Burt, T.; Gardiner, A. The permanence of stream networks in Britain: Some further comments. Earth Surf. Process. Landf. 1982, 7, 327–332. [Google Scholar] [CrossRef]
- Foster, D.R. Land-use history (1730–1990) and vegetation dynamics in Central New England, USA. J. Ecol. 1992, 80, 753–771. [Google Scholar] [CrossRef]
- Slattery, M.C.; Burt, T.P. Particle size characteristics of suspended sediment in hillslope runoff and stream flow. Earth Surf. Process. Landf. 1997, 22, 705–719. [Google Scholar] [CrossRef]
- Lenzi, M.A.; Marchi, L. Suspended sediment load during floods in a small stream of the Dolomites (Northeastern Italy). Catena 2000, 39, 267–282. [Google Scholar] [CrossRef]
- Cai, W.; Rensch, P. The 2011 Southeast Queensland extreme summer rainfall: A confirmation of a negative pacific decadal oscillation phase? Geophys. Res. Lett. 2012, 39. [Google Scholar] [CrossRef]



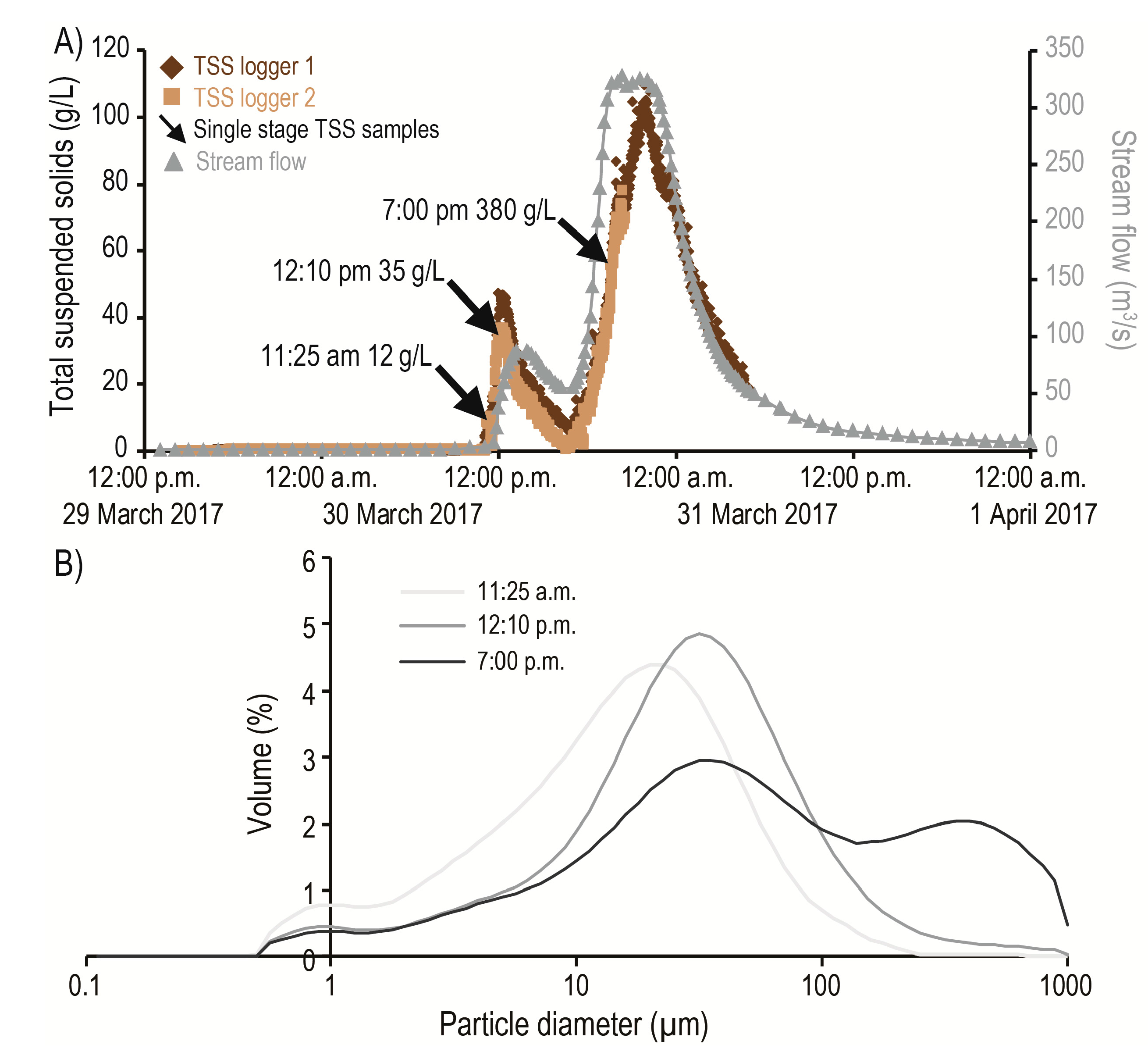

| Study | Sample | TP | TN |
|---|---|---|---|
| Present study | 12:10 p.m. | 8.83 | 18.71 |
| 12:10 p.m. washload | 8.32 | 19.24 | |
| 7:00 p.m. | 8.04 | 19.34 | |
| 7:00 p.m. washload | 8.03 | 16.47 | |
| Long-term monitoring 1 | 2002–2015 | 0.82 (0.48–1.95) | 1.72 (1.04–3.29) |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Grinham, A.; Deering, N.; Fisher, P.; Gibbes, B.; Cossu, R.; Linde, M.; Albert, S. Near-Bed Monitoring of Suspended Sediment during a Major Flood Event Highlights Deficiencies in Existing Event-Loading Estimates. Water 2018, 10, 34. https://doi.org/10.3390/w10020034
Grinham A, Deering N, Fisher P, Gibbes B, Cossu R, Linde M, Albert S. Near-Bed Monitoring of Suspended Sediment during a Major Flood Event Highlights Deficiencies in Existing Event-Loading Estimates. Water. 2018; 10(2):34. https://doi.org/10.3390/w10020034
Chicago/Turabian StyleGrinham, Alistair, Nathaniel Deering, Paul Fisher, Badin Gibbes, Remo Cossu, Michael Linde, and Simon Albert. 2018. "Near-Bed Monitoring of Suspended Sediment during a Major Flood Event Highlights Deficiencies in Existing Event-Loading Estimates" Water 10, no. 2: 34. https://doi.org/10.3390/w10020034
APA StyleGrinham, A., Deering, N., Fisher, P., Gibbes, B., Cossu, R., Linde, M., & Albert, S. (2018). Near-Bed Monitoring of Suspended Sediment during a Major Flood Event Highlights Deficiencies in Existing Event-Loading Estimates. Water, 10(2), 34. https://doi.org/10.3390/w10020034







