Groundwater Quality and Suitability for Different Uses in the Saloum Area of Senegal
Abstract
1. Introduction
2. Materials and Methods
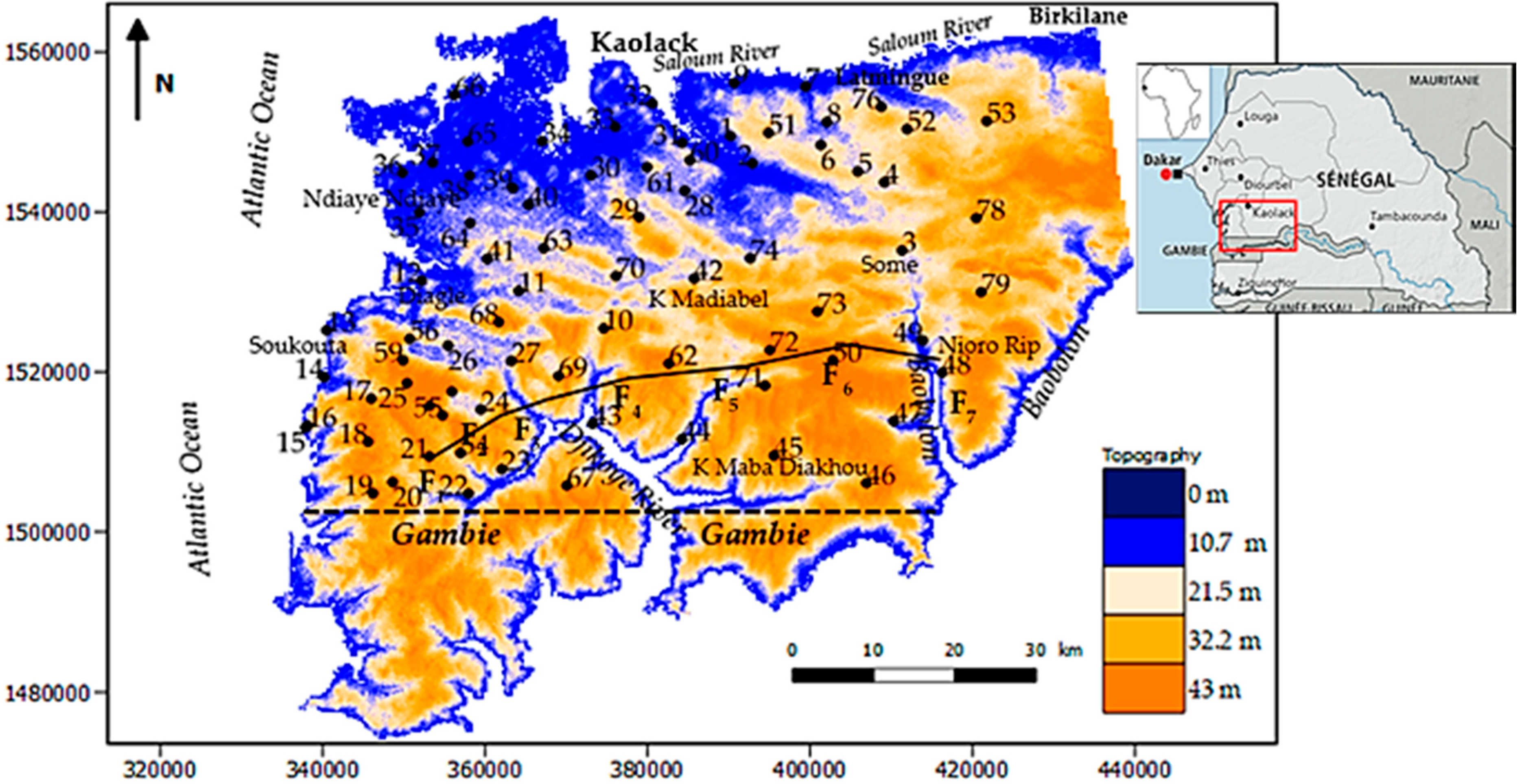
2.1. Location of the Study Area
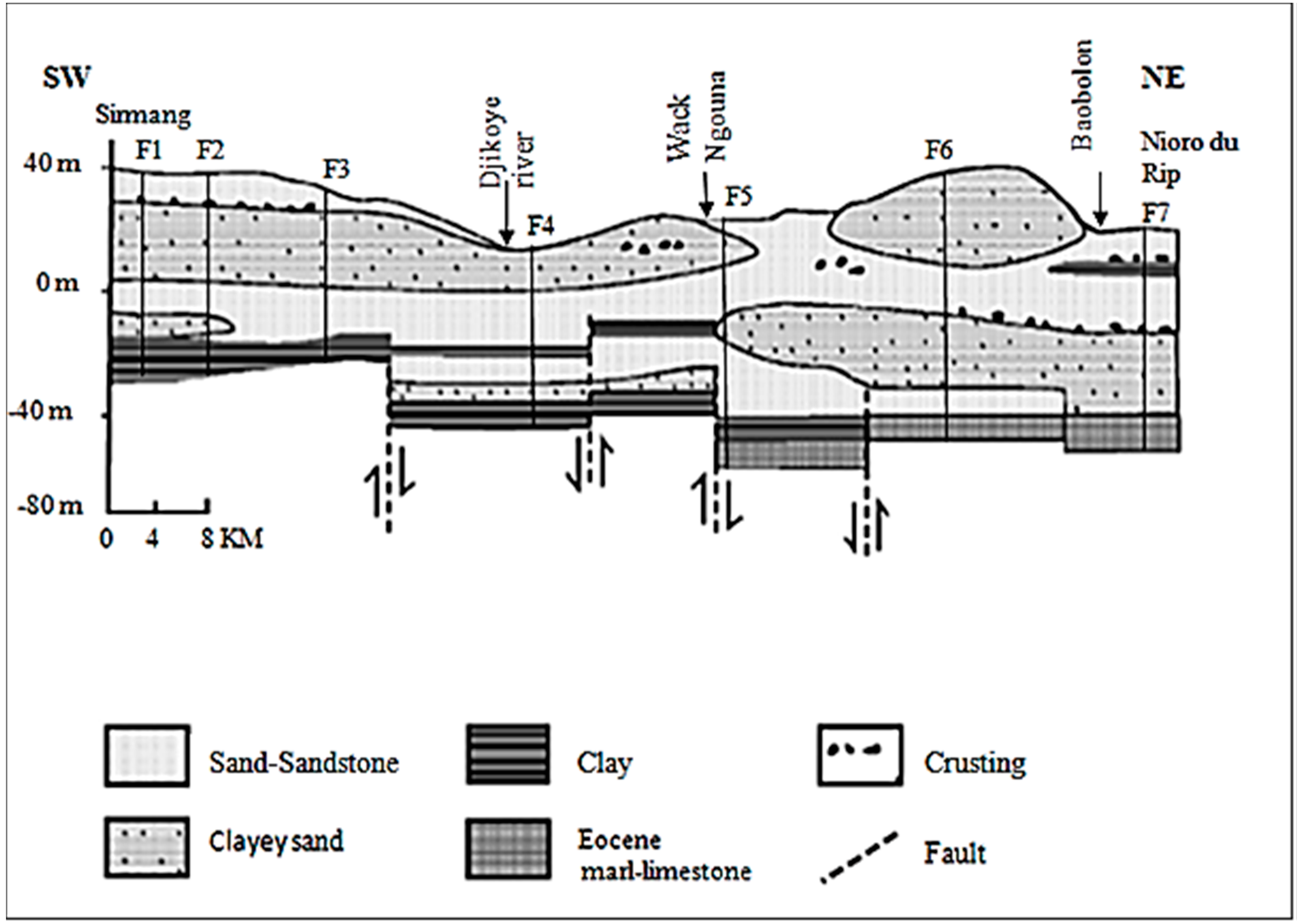
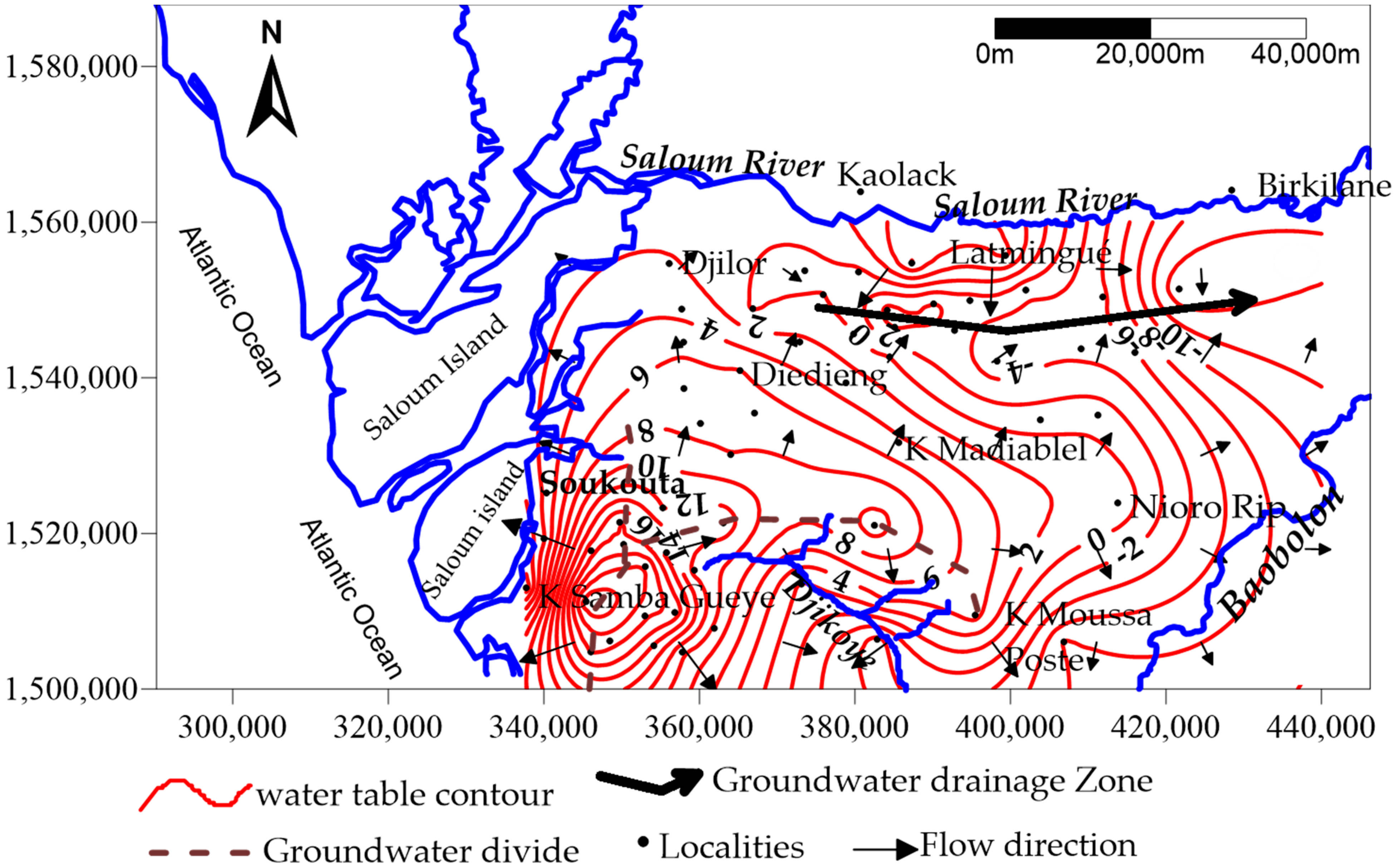
2.2. Geological and Hydrogeological Settings
2.3. Sample Collection and Analysis
3. Results and Discussion
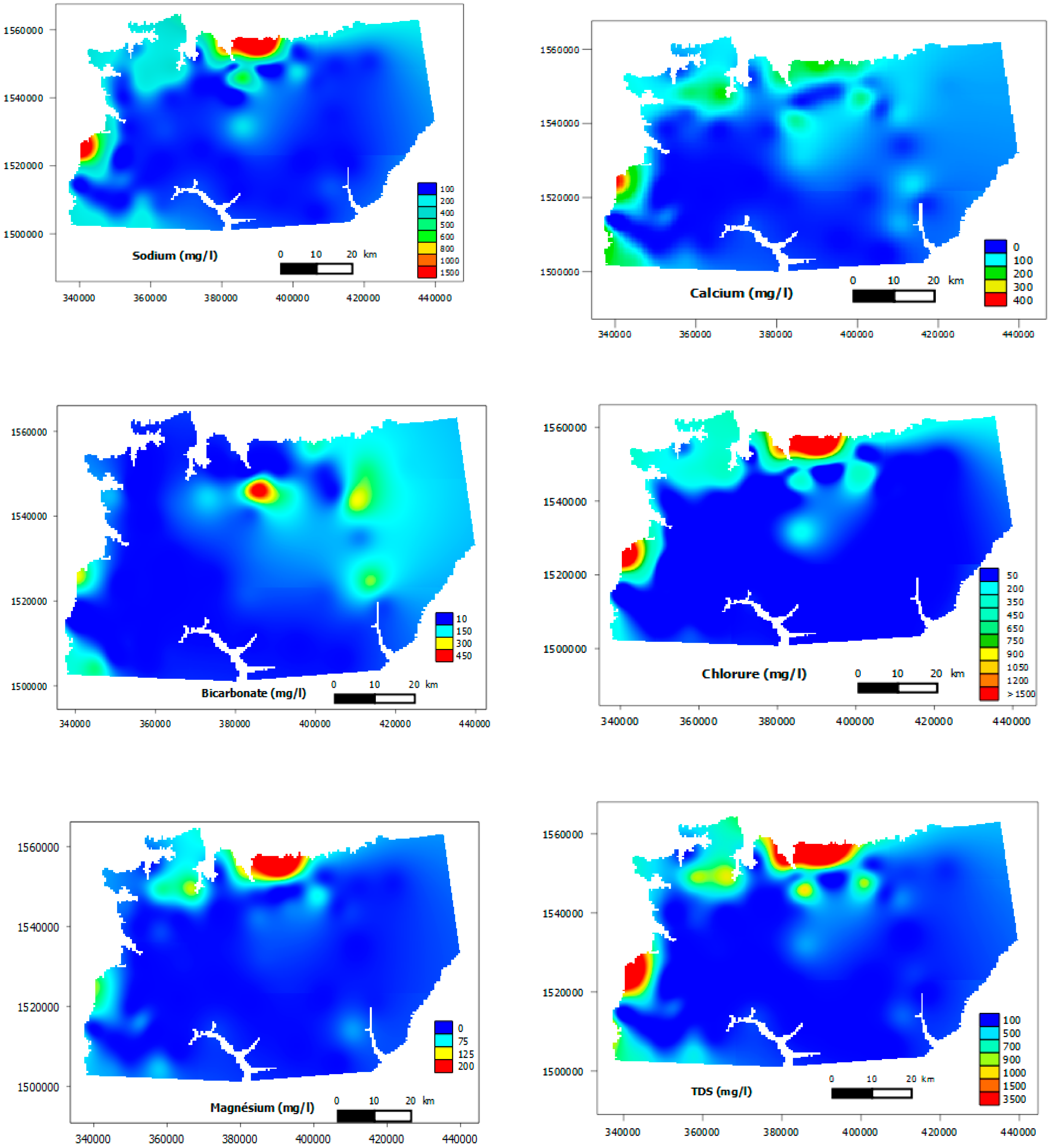
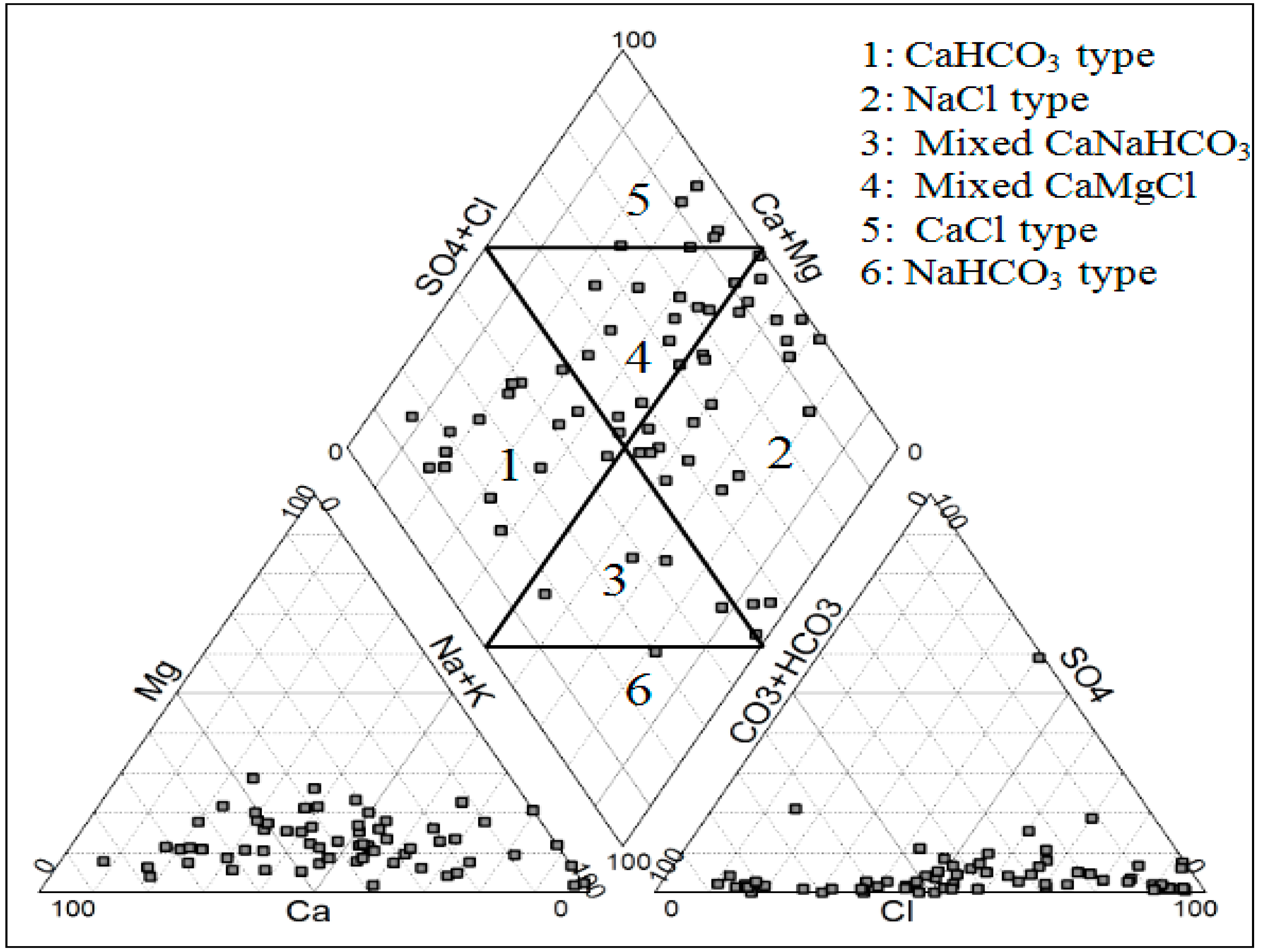
3.1. General Groundwater Chemistry
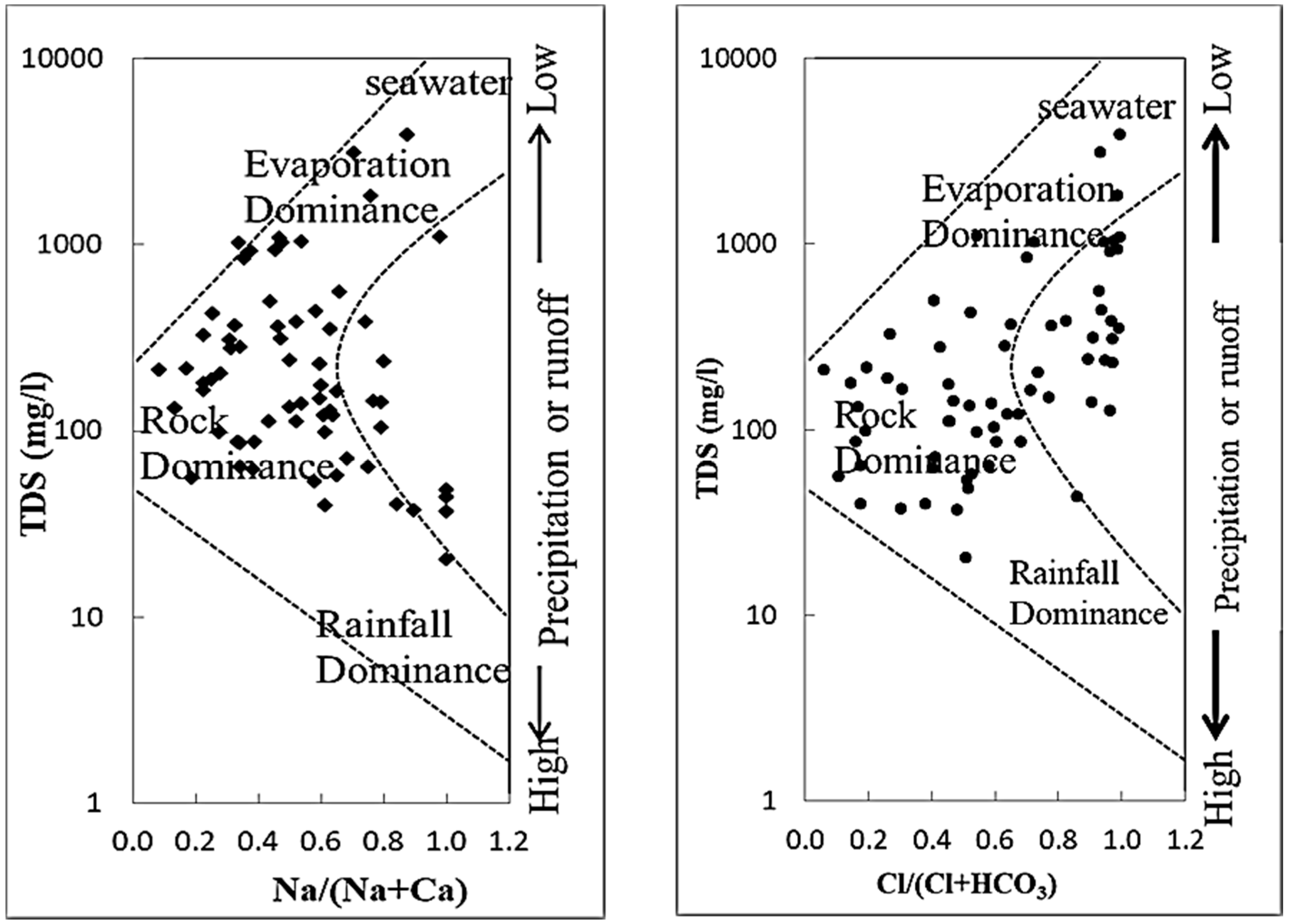
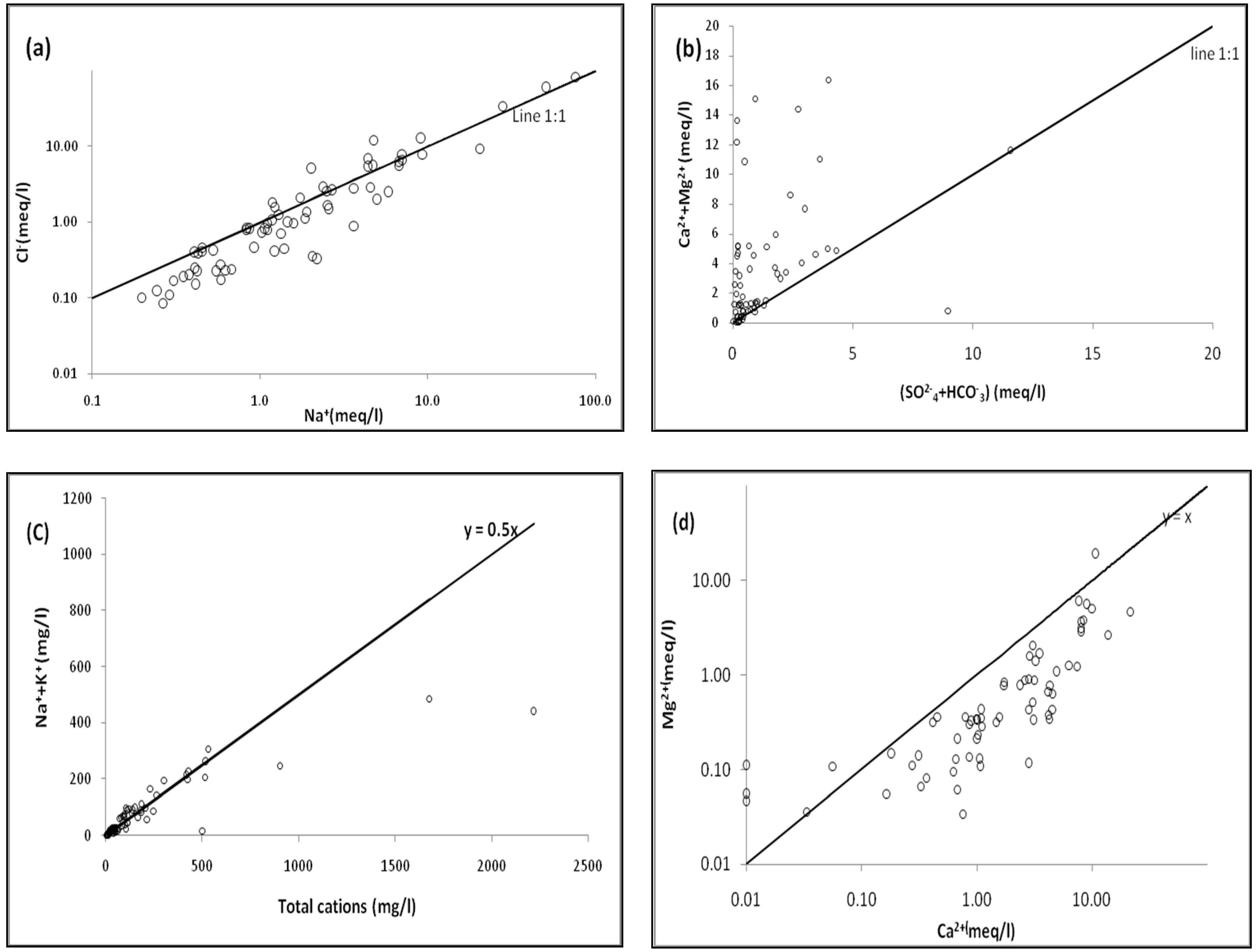
3.2. Binary Diagram and Geochemical Process
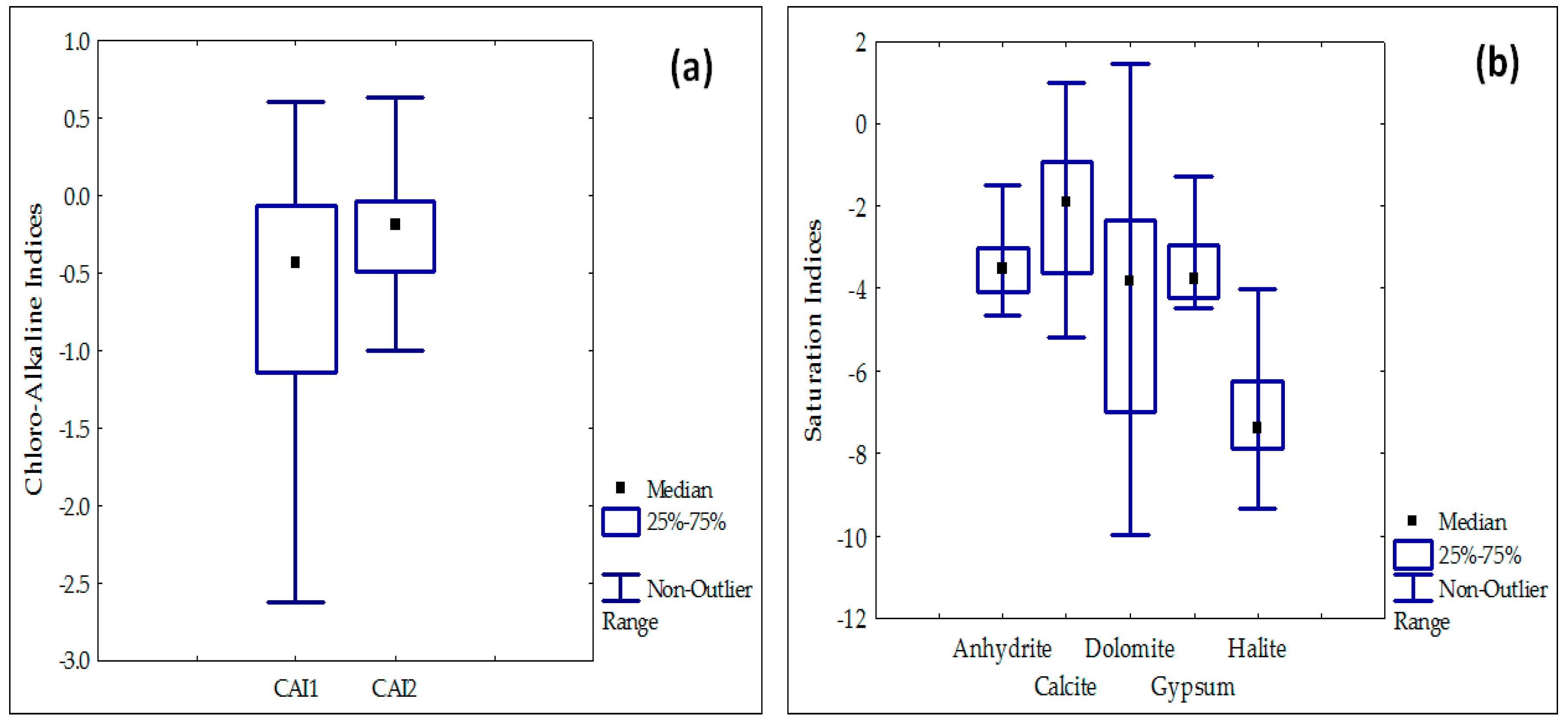
3.3. Mineral Saturation Indices
3.4. Drinking Water Quality
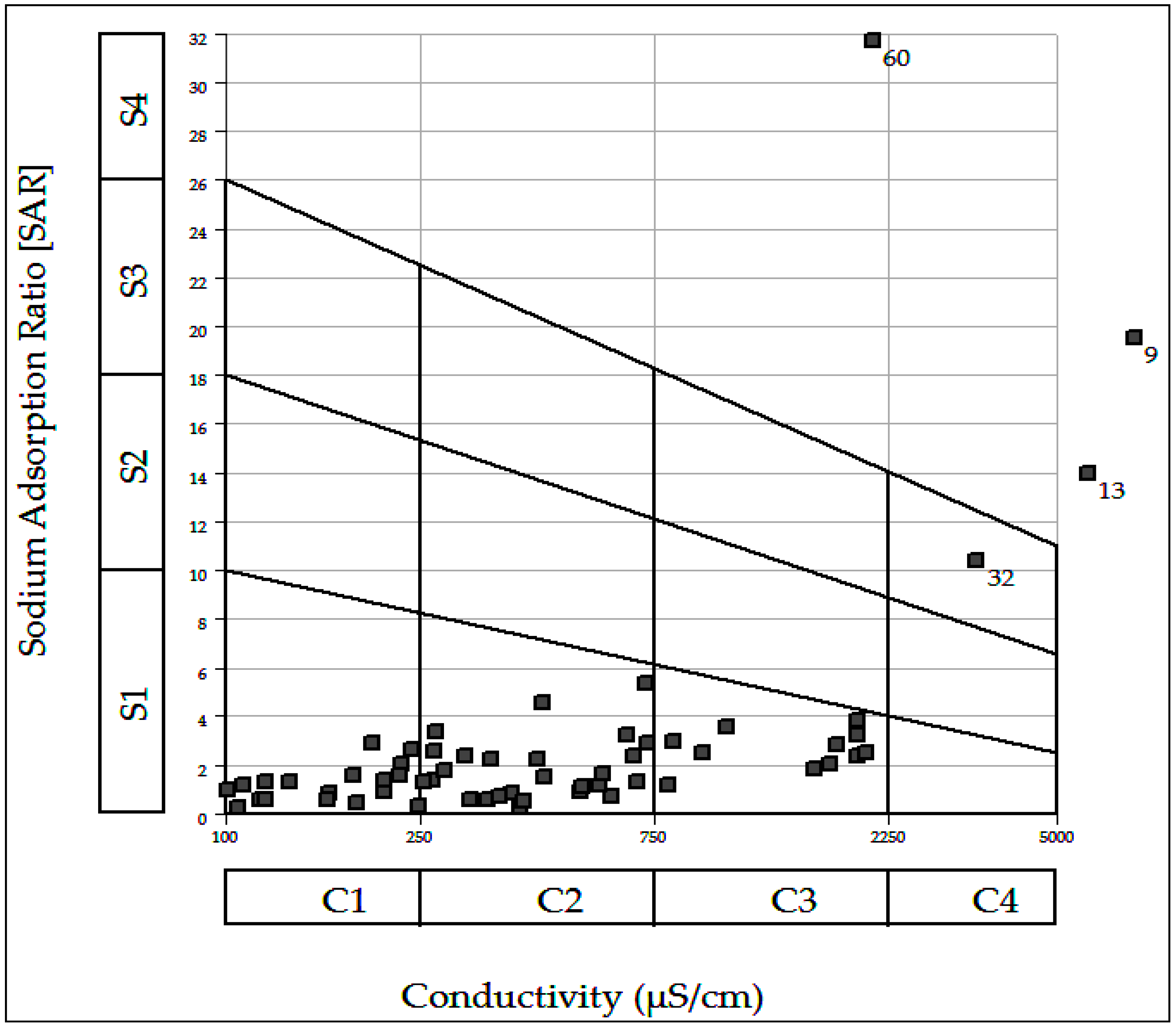
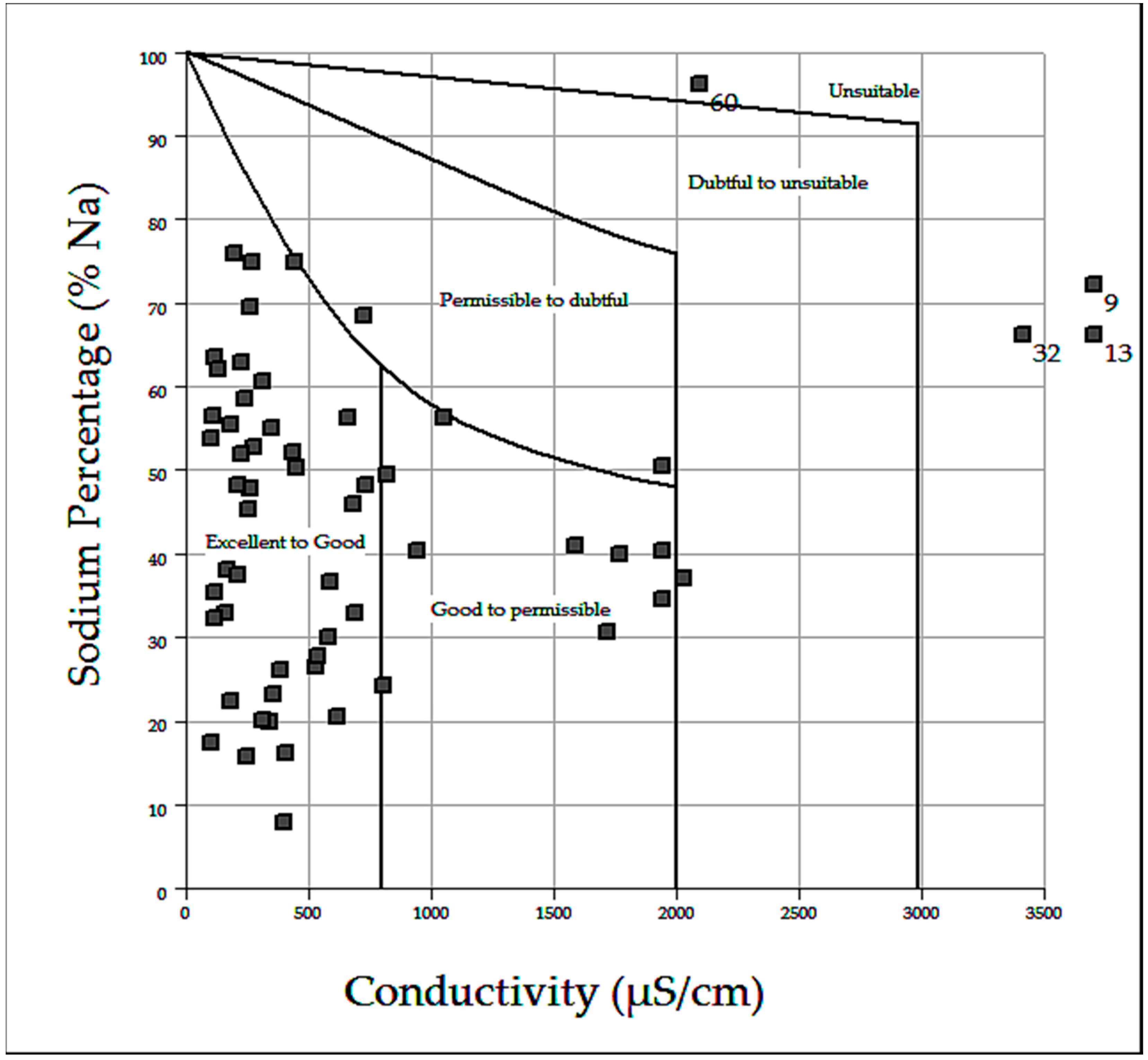
3.5. Suitability of Groundwater for Irrigation
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Rajmohan, N.; Elango, E.L. Identification and evolution of hydrogeochemical processes in the groundwater environment in an area of the Palar and Cheyyar River Basins, Southern India. Environ. Geol. 2004, 46, 47–61. [Google Scholar] [CrossRef]
- Jalali, M. Geochemistry characterization of groundwater in an agricultural area of Razan, Hamadan, Iran. Environ. Geol. 2009, 56, 1479–1488. [Google Scholar] [CrossRef]
- Baye, A.Y.; Razack, M.; Ayenew, T.; Zemedagegnehu, E. Hydrogeological and hydrochemical framework of Upper Awash River basin, Ethiopia: With special emphasis on inter-basins groundwater transfer between Blue Nile and Awash rivers. J. Afr. Earth Sci. 2012, 65, 46–60. [Google Scholar]
- Furi, W.; Razack, M.; Alemayu, T.A.; Ayenew, T.; Legesse, D. Fluoride enrichment mechanism and geospatial distribution in the volcanic aquifers of the Middle Awash basin, Northern Main Ethiopian Rift. J. Afr. Earth Sci. 2011, 60, 315–327. [Google Scholar] [CrossRef]
- Faye, S.; Cisse Faye, S.; Ndoye, S.; Faye, A. Hydrogeochemistry of the Saloum (Senegal) superficial coastal aquifer. Environ. Geol. 2003, 44, 127–136. [Google Scholar] [CrossRef]
- Faye, S.; Maloszewski, P.; Stichler, W.; Trimborn, P.; Cisse Faye, S.; Gaye, C.B. Groundwater salinization in the Saloum (Senegal) delta aquifer: Minor elements and isotopic indicators. Sci. Total Environ. 2005, 343, 243–259. [Google Scholar] [CrossRef] [PubMed]
- Abderamane, H.; Razack, M.; Vassolo, S. Hydrogeochemical and isotopic characterisation of the Groundwter in the Chari-Baguirmi depression. Republic of Chad. Environ. Earth Sci. 2013, 69, 2337–2350. [Google Scholar] [CrossRef]
- Ntanganedzeni, B.; Elumalai, V.; Rajmohan, N. Coastal Aquifer Contamination and Geochemical Processes Evaluation in Tugela Catchment, South Africa-Geochemical and Statistical Approaches. Water 2018, 10, 687. [Google Scholar] [CrossRef]
- Celestino, A.E.M.; Cruz, D.A.M.; Sánchez, E.M.O.; Reyes, F.G.; Soto, D.V. Groundwater Quality Assessment: An Improved Approach to K-Means Clustering, Principal Component Analysis and Spatial Analysis: A Case Study. Water 2018, 10, 437. [Google Scholar] [CrossRef]
- Zhang, X.; Qian, H.; Chen, J.; Qiao, L. Assessment of Groundwater Chemistry and Status in a Heavily Used Semi-Arid Region with Multivariate Statistical Analysis. Water 2014, 6, 2212–2232. [Google Scholar] [CrossRef]
- Li, X.; Wu, H.; Qian, H.; Gao, Y. Groundwater Chemistry Regulated by Hydrochemical Processes and Geological Structures: A Case Study in Tongchuan, China. Water 2018, 10, 338. [Google Scholar] [CrossRef]
- Yuan, J.; Xu, F.; Deng, G.; Tang, Y.; Li, P. Hydrogeochemistry of Shallow Groundwater in a Karst Aquifer System of Bijie City, Guizhou Province. Water 2017, 9, 625. [Google Scholar] [CrossRef]
- Krishna Kumar, S.; Logeshkumaran, A.; Magesh, N.S.; Godson, P.S.; Chandrasekar, N. Hydro-geochemistry and application of water quality index (WQI) for groundwater quality assessment, Anna Nagar, part of Chennai City, Tamil Nadu, India. Appl. Water Sci. 2015, 5, 335–343. [Google Scholar] [CrossRef]
- Diop, E.S. Tropical Holocene Estuaries. Comparative Study of the Physical Geography Features of the Rivers from the South of Saloum to the Mellcore (Guinea Republic). Ph.D. Thesis, University Louis Pasteur, Strasbourg, France, 1986. (In French). [Google Scholar]
- Conrad, G.; Lappartient, J.R. The Continental Terminal, its position within the Cenozoic geodynamic evolution of the Senegalo-mauritanian basin. J. Afr. Earth Sci. 1987, 6, 45–60. [Google Scholar]
- Diluca, C. Hydrogeology of the Continental Terminal Aquifer between the Sine and the Gambia; Technical Report, BRGM DKR 76 DKR; BRGM Publ.: Orléans, France, 1976; p. 33. (In French) [Google Scholar]
- Lappartient, J.R. The Continental Terminal and the Early Pleistocene of the Senegalo-Mauritanian Basin. Stratigraphy, Sedimentology, Diagenesis, Alterations, Paleoshore Reconstitutions from the Ferralitic Formations. Ph.D. Thesis, University of Marseille, Provence, France, 1985. (In French). [Google Scholar]
- Edmunds, M.W. Groundwater Recharge in Senegal. In Technical Report Hydrogeology Series; Report WD/90/49R; British Geological Survey Publ.: Keyworth, UK, 1990; p. 162. [Google Scholar]
- Simler, R. Diagrammes; Laboratoire d’Hydrogéologie d’Avignon: Avignon, France, 2009. [Google Scholar]
- Gibbs, R.J. Mechanism controlling world water chemistry. Science 1970, 17, 1088–1090. [Google Scholar] [CrossRef]
- Schoeller, H. Qualitative evaluation of groundwater resources. In Methods and Techniques of Groundwater Investigation and Development; Water Resources Series, N° 33; UNESCO: Paris, France, 1967; pp. 44–52. [Google Scholar]
- Garrels, R.M.; Christ, C.L. Solutions, Minerals, and Equilibria; Freeman: Dallas, TX, USA, 1965; p. 450. [Google Scholar]
- Ammar, F.H.; Chkir, N.; Zouari, K.; Hamelin, B.; Deschamps, P.; Aigoun, A. Hydrogeochemical processes in the complex Terminal aquifer of southern Tunisia: An integrated investigation based on geochemical and multivariate statistical methods. J. Afr. Earth Sci. 2014, 100, 81–95. [Google Scholar] [CrossRef]
- Parkhurst, D.L.; Apelo, C.A.J. User’s guide to PHREEQC (version 2)-A Computer program for speciation, batch-reaction, one-dimensional transport, and inverse geochemical calculations. United States Geological Survey. In Water Resources Investigations; Report 99-4259; USGS Publ.: Washington, DC, USA, 1999; p. 326. [Google Scholar]
- Touhari, F.; Meddi, M.; Mehaiguene, M.; Razack, M. Hydrogeochemical assessment of the Upper Cheliff groundwater (North West Algeria). Environ. Earth Sci. 2015, 73, 3043–3061. [Google Scholar] [CrossRef]
- WHO. Guidelines to Drinking Water Quality; World Health Organization: Geneva, Switzerland, 1983. [Google Scholar]
- WHO. Guideline for Drinking-Water Quality, 3rd ed.; World Health Organization: Geneva, Switzerland, 2004. [Google Scholar]
- Mukherjee, S.; Kumar, B.A.; Körtvélyessy, L. Assessment of groundwater quality in the south 24-Parganas, west Bengal coast, India. J. Environ. Hydrol. 2005, 13, 1–8. [Google Scholar]
- Wilcox, L.V. Classification and Use of Irrigation Water; Circular 969; US Department of Agriculture: Washington, DC, USA, 1955.
- USSL (United States Salinity Laboratory staff). Diagnosis and Improvement of Saline and Alkali Soils; Hand Book 60; US Department of Agricultural Soils; US Department of Agricultural (USDA): Washington, DC, USA, 1954; pp. 69–81.
- Freeze, A.; Cherry, J. Groundwater; Prentice-Hall: Englewood Cliffs, NJ, USA, 1979; p. 604. [Google Scholar]
- Gilly, G.; Corrao, G.; Favilli, S. Concentrations of nitrates in drinking water and incidence of gastric carcinomas. First descriptive study of the Piemonate Region, Italy. Sci Total Environ. 1984, 34, 35–37. [Google Scholar] [CrossRef]
- Venkateswaran, S.; Karuppannan, S.; Vijay, S.P.; Kannan, R.; Malar, S.; Prabu, P. Hydro Chemical Characteristic and Groundwater Quality Assessment in Parts of Pambar River Basin, Tamil Nadu, India. Int. J. Innov. Technol. Explor. Eng. 2013, 3, 36–44. [Google Scholar]
- Piper, A.M. A graphic procedure in the chemical interpretation of water analysis. Am. Geophys. Union Trans. 1944, 25, 914–923. [Google Scholar] [CrossRef]
- Gibbs, R.J. Mechanism controlling world river water chemistry: Evaporation-crystallization process. Science 1971, 172, 871–872. [Google Scholar]
- Versluys, J. Subterranean water conditions in the coastal regions of the Netherlands. Econ. Geol. 1931, 26, 65–95. [Google Scholar] [CrossRef]
- Schoeller, H. Les échanges de base dans les eaux souterraines; trois exemples en Tunisie. Bull. Soc. Geol. Fr. 1934, 4, 389–420. [Google Scholar]
- Schoeller, H. Géochimie des eaux souterraines. Revue l’Instit. Français Pétrole 1956, 10, 230–234. [Google Scholar]
- Stuyfzand, P.J. A new hydrochemical classification of water types: Principles and application to the coastal dunes aquifer system of the Netherlands. In Proceedings of the 9th Salt Water Intrusion Meeting, Delft, The Netherlands, 12–16 May 1986; pp. 641–655. [Google Scholar]
- Stuyfzand, P.J. Base Exchange Indices as Indicators of salinization or Freshening of Coastal Aquifers. In Proceedings of the 20th Salt Water Intrusion Meeting, Naples, FL, USA, 23–27 June 2008; pp. 262–265. [Google Scholar]
- Batabyal, A.K.; Gupta, S. Fluoride-contaminated groundwater of Birbhum district, West Bengal, India: Interpretation of drinking and irrigation suitability and major geochemical processes using principal component analysis. Environ. Monit. Assess. 2017, 189–369. [Google Scholar] [CrossRef] [PubMed]
- Schoeller, H. Geochemistry of groundwater. In Groundwater Studies—An International Guide for Research and Practice; UNESCO: Paris, France, 1977; Volume 15, pp. 1–18. [Google Scholar]
- Kurumbein, W.C.; Graybill, F.A. An introduction to Statistical Models in Geology; McGraw Hill Book Company: New York, NY, USA, 1965. [Google Scholar]
- Mrazovac, S.; Vojinovic-Miloradov, M. Correlation of main physicochemical parameters of some groundwater in northern Serbia. J. Geochem. Explor. 2011, 108, 176–182. [Google Scholar] [CrossRef]
- Fisher, R.S.; Mulican, W.F. Hydrochemical evolution of sodium-sulfate and sodium-chloride groundwater beneath the Northern Chihuahuan desert, Trans-Pecos, Texas, USA. Hydrogeol. J. 1997, 10, 455–474. [Google Scholar] [CrossRef]
- Hiscock, K.M. Hydrogeology; Principles and Practice; Blackwell Publishing: Oxford, UK, 2005. [Google Scholar]
- Wang, P.; Yu, J.; Zhang, Y.; Liu, C. Groundwater recharge and hydrogeochemical evolution in the Ejina Basin, northwest China. J. Hydrol. 2013, 476, 72–86. [Google Scholar] [CrossRef]
- Stallard, R.F.; Edmond, J.M. Geochemistry of the Amazon: The influence of geology and weathering environment on the dissolved load. J. Geophys. Res. Oceans 1983, 88, 9671–9688. [Google Scholar] [CrossRef]
- Saravanan, K.; Srinivasamoorthy, K.; Prakash, R.; Gopinath, S.; Suma, C.S. An Evaluation of Hydrogeochemistry of Groundwater in Upper Vellar Sub-Basin using Mineral Stability and Solute Transport Modeling. International Conference on Water Resources, Coastal and Ocean Engineering (ICWRCOE). Aquat. Procedia 2015, 4, 1119–1125. [Google Scholar] [CrossRef]
- Cerling, T.E.; Pederson, B.L.; Damm, K.L.V. Sodium-calcium ion exchange in the weathering of shales: Implications for global weathering budgets. Geology 1989, 17, 552–554. [Google Scholar] [CrossRef]
- Datta, P.S.; Tyagui, S.K. Major ion chemistry of groundwater in Delhi area: Chemical weathering processes and groundwater regime. J. Geol. Soc. India 1996, 47, 179–188. [Google Scholar]
- Parkhurst, D.L.; Apelo, C.A.J. Description of Input and Examples for PHREEQC Version 3—A Computer Program for Speciation, Batch-Reaction, One-Dimensional Transport, and Inverse Geochemical Calculations. U.S. Department of the Interior. In U.S. Geological Survey Techniques and Methods; USGS Publ.: Washington, DC, USA, 2013; 6–A43. [Google Scholar]
- Garrels, R.M.; Mackenzie, F.T. Origin of the chemical compositions of some springs and lakes. In Equilibrium Concepts in Natural Water Systems; Stumm, W., Ed.; American Chemical Society: Washington, DC, USA, 1967; pp. 222–242. [Google Scholar]
- Srinivasamoorthy, K.; Chidambaram, S.; Prasanna, M.V.; Vasanthavigar, M.; John Peter, A.; Anandhan, P. Identification of major sources controlling groundwater chemistry from a hard rock terrain—A case study from Mettur Taluk, Salem district, Tamilnadu, India. J. Earth Syst. Sci. 2008, 117, 49. [Google Scholar] [CrossRef]
- Srinivasamoorthy, K.; Gopinath, M.; Chidambaram, S.; Vasanthavigar, M.; Sarma, V.S. Hydrochemical characterization and quality appraisal of groundwater from Pungar sub basin, Tamilnadu, India. J. King Saud Univ. Sci. 2014, 26, 37–52. [Google Scholar] [CrossRef]
- Hounslow, A.W. Water Quality Data: Analysis and Interpretation; CRC Press LLC: Boca Raton, FL, USA, 1995. [Google Scholar]
- Schroeder, H.A. Relations between hardness of water and death rates from certain chronic and degenerative diseases in the United States. J. Chron. Dis. 1960, 12, 586–591. [Google Scholar] [CrossRef]
- Hem, J.D. Study and Interpretation of Chemical Characteristics of Natural Water, 3rd ed.; Geological Survey Water-Supply 1985, Paper 2254; United States Government Printing Office: Washington, DC, USA.
- Sawyer, G.N.; McCarthy, D.L. Chemistry of Sanitary Engineers, 2nd ed.; McGraw Hill: New York, NY, USA, 1967; p. 518. [Google Scholar]
- Comly, H.H. Cyanosis in infants caused by nitrates in well water. J. Am. Med. Assoc. 1945, 129, 112–116. [Google Scholar] [CrossRef]
- Kumar, M.; Kumari, K.; Ramanathan, A.L.; Saxena, R.A. Comparative Evaluation of Groundwater Suitability for Irrigation and Drinking Purposes in Two Intensively Cultivated, Districts of Punjab, India. Environ. Geol. 2007, 53, 553–574. [Google Scholar] [CrossRef]
- Saleh, A.; Al-Ruwaih, F.; Shehata, M. Hydrogeochemical processes operating within the main aquifers of Kuwait. J. Arid. Environ. 1999, 42, 195–209. [Google Scholar] [CrossRef]
- Elango, L.; Kannan, R.; Senthil Kumar, M. Major ion chemistry and identification of hydrogeochemical processes of groundwater in part of Kancheepuram district, Tamil Nadu, Indian. J. Environ. Geosci. 2003, 10, 157–166. [Google Scholar]
- Collins, R.; Jenkins, A. The impact of agricultural land use on stream chemistry in the middle Hills of the Himalayas, Napal. J. Hydrol. 1996, 185, 71–86. [Google Scholar] [CrossRef]
- Subramani, T.; Elango, L.; Damodarasamy, S.R. Groundwater quality and its suitability for drinking and agricultural use in Chithar River Basin, Tamil Nadu, India. Environ. Geol. 2005, 47, 1099–1110. [Google Scholar] [CrossRef]
- Yidana, S.M. Groundwater classification using multivariate statistical methods: Southern Ghana. J. Afr. Earth Sci. 2010, 57, 455–469. [Google Scholar] [CrossRef]
| N° | Name | Ca | Mg | Na | K | HCO3 | Cl | SO4 | NO3 | TDS | pH | EC | RSC | TH |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Koumbal | 20.39 | 2.84 | 43.48 | 1.24 | 33.55 | 48.32 | 1.18 | 41.00 | 164.00 | 7.20 | 309.00 | −0.70 | 62.81 |
| 2 | Sikatoroum | 62.31 | 10.73 | 21.10 | 3.64 | 170.80 | 16.44 | 2.07 | 49.00 | 180.00 | 8.10 | 342.00 | −1.19 | 200.50 |
| 3 | Some | 21.37 | 1.30 | 15.49 | 1.97 | 76.25 | 8.38 | 1.46 | 2.92 | 87.00 | 7.60 | 163.00 | 0.08 | 58.85 |
| 4 | Touba Darou camp | 84.37 | 8.10 | 28.04 | 1.05 | 259.25 | 55.22 | 2.76 | 1.34 | 327.00 | 6.46 | 613.00 | −0.63 | 244.66 |
| 5 | Ndobene | 20.10 | 4.00 | 26.93 | 1.96 | 45.75 | 37.78 | 0.77 | 16.00 | 140.00 | 6.90 | 264.00 | −0.58 | 66.93 |
| 6 | Daga Youndou | 159.69 | 34.45 | 108.98 | 0.54 | 27.45 | 422.84 | 2.85 | 36.50 | 920.00 | 4.92 | 1720.00 | −10.35 | 542.76 |
| 7 | Latmingue | 155.90 | 37.10 | 100.30 | 128.70 | 179.95 | 376.29 | 34.91 | 50.00 | 850.00 | 6.45 | 1590.00 | −7.88 | 544.33 |
| 8 | Keur Mamour Lo | 61.11 | 6.22 | 25.21 | 1.83 | 46.65 | 72.80 | 11.83 | 48.12 | 205.00 | 7.40 | 385.00 | −2.80 | 178.69 |
| 9 | Koilal | 214.00 | 228.40 | 1726.10 | 49.70 | 33.55 | 2886.18 | 316.3 | 27.94 | 3900.00 | 5.30 | 7160.00 | −28.92 | 1486.67 |
| 10 | Ndrame Scale | 0.00 | 0.67 | 8.64 | 1.26 | 13.42 | 7.21 | 0.00 | 4.62 | 37.10 | 7.20 | 69.40 | 0.17 | 2.78 |
| 11 | Ndrame Ibra | 8.29 | 3.82 | 35.38 | 2.67 | 6.10 | 37.92 | 1.20 | 46.09 | 142.00 | 7.30 | 267.00 | −0.63 | 36.66 |
| 12 | Diagle | 15.12 | 0.42 | 26.51 | 0.76 | 24.40 | 25.98 | 2.40 | 29.05 | 121.00 | 7.50 | 229.00 | −0.39 | 39.53 |
| 13 | Soukouta | 427.40 | 57.20 | 1158.90 | 35.80 | 268.40 | 2154.45 | 30.89 | 23.29 | 3110.00 | 7.70 | 5740.00 | −21.63 | 1306.83 |
| 14 | Nema Ba | 160.40 | 43.30 | 211.90 | 102.00 | 12.20 | 275.86 | 544.9 | 12.51 | 1040.00 | 4.50 | 1940.00 | −11.37 | 581.42 |
| 15 | Missira 1 | 170.70 | 28.32 | 150.50 | 64.50 | 162.50 | 232.86 | 80.81 | 293.45 | 1030.00 | 6.79 | 1940.00 | −8.18 | 544.75 |
| 16 | Missira2 | 6.52 | 0.80 | 10.30 | 0.30 | 23.79 | 14.35 | 1.41 | 1.97 | 54.00 | 5.63 | 101.00 | 0.00 | 19.63 |
| 17 | Nema Nding | 55.37 | 16.79 | 92.91 | 0.66 | 22.20 | 122.08 | 13.20 | 163.97 | 443.00 | 3.85 | 819.00 | −3.78 | 208.39 |
| 18 | Dassilame Soce | 0.67 | 0.43 | 6.60 | 0.89 | 15.25 | 3.86 | 0.00 | 5.09 | 37.70 | 6.10 | 71.00 | 0.18 | 3.49 |
| 19 | Karang | 97.39 | 9.49 | 114.03 | 4.87 | 179.95 | 101.37 | 11.65 | 168.41 | 500.00 | 6.23 | 940.00 | −2.69 | 283.03 |
| 20 | Sirmang | 19.33 | 3.83 | 32.03 | 0.00 | 44.40 | 31.65 | 3.65 | 33.40 | 175.00 | 6.90 | 350.00 | −0.55 | 64.26 |
| 21 | keur Samba Gueye | 3.34 | 0.67 | 6.05 | 0.00 | 24.40 | 3.01 | 0.00 | 3.18 | 40.20 | 7.50 | 76.50 | 0.18 | 11.16 |
| 22 | keur Seyni Gueye | 45.72 | 20.97 | 68.81 | 16.35 | 50.20 | 119.69 | 10.00 | 102.50 | 560.00 | 4.10 | 1050.00 | −3.18 | 201.67 |
| 23 | Keur Momar Sokh | 10.48 | 1.15 | 34.82 | 2.87 | 34.40 | 14.49 | 1.30 | 49.83 | 144.00 | 5.43 | 269.00 | −0.05 | 30.98 |
| 24 | Bambadalo Thiakh | 3.68 | 1.77 | 12.53 | 1.14 | 10.07 | 8.04 | 1.20 | 21.47 | 64.00 | 6.30 | 121.00 | −0.16 | 16.57 |
| 25 | keur Lahine Fatim | 7.31 | 0.98 | 27.98 | 0.56 | 18.30 | 15.76 | 2.80 | 35.14 | 104.00 | 7.60 | 199.00 | −0.15 | 22.38 |
| 26 | Nioro Alassane Tall | 0.00 | 0.55 | 14.25 | 0.45 | 13.12 | 8.10 | 1.18 | 9.04 | 48.60 | 7.90 | 80.50 | 0.17 | 2.31 |
| 27 | Keur Saloum Diane | 0.00 | 0.07 | 5.56 | 0.25 | 7.38 | 4.42 | 0.00 | 2.44 | 20.60 | 7.30 | 38.60 | 0.12 | 0.30 |
| 28 | Lamaram Badiane | 88.00 | 9.90 | 50.20 | 10.90 | 153.35 | 105.75 | 5.81 | 47.99 | 426.00 | 6.09 | 805.00 | −2.69 | 261.25 |
| 29 | Ndiagne | 21.83 | 4.30 | 19.08 | 1.22 | 61.00 | 29.65 | 1.44 | 8.76 | 112.00 | 7.50 | 210.00 | −0.44 | 72.52 |
| 30 | Ndiakhate | 46.60 | 1.40 | 9.80 | 4.80 | 118.95 | 13.62 | 1.36 | 5.87 | 132.00 | 6.53 | 249.00 | −0.49 | 122.33 |
| 31 | Sama Toucouleur | 46.30 | 11.10 | 29.50 | 2.10 | 103.70 | 54.56 | 4.25 | 34.42 | 280.00 | 6.47 | 530.00 | −1.52 | 162.00 |
| 32 | Dabane Galadio | 178.20 | 67.00 | 639.50 | 21.80 | 30.50 | 1195.78 | 107.4 | 25.81 | 1830.00 | 5.48 | 3410.00 | −13.91 | 724.67 |
| 33 | Tiofior | 52.00 | 10.70 | 90.30 | 4.50 | 13.05 | 194.25 | 2.93 | 28.37 | 352.00 | 4.84 | 661.00 | −3.26 | 174.58 |
| 34 | Bandoulou Toucou | 181.60 | 61.30 | 187.30 | 48.50 | 55.75 | 556.18 | 7.96 | 170.84 | 1030.00 | 5.91 | 1940.00 | −13.19 | 709.42 |
| 35 | Tallene | 13.14 | 1.56 | 9.31 | 0.95 | 36.30 | 14.26 | 0.72 | 2.80 | 62.00 | 6.60 | 118.00 | −0.19 | 39.35 |
| 36 | Lérane Kolli | 70.20 | 13.00 | 44.30 | 12.70 | 114.55 | 102.66 | 12.15 | 36.85 | 369.00 | 6.90 | 692.00 | −2.69 | 229.67 |
| 37 | Ndiaye ndiaye S. | 90.00 | 7.70 | 45.10 | 8.50 | 29.15 | 182.79 | 2.63 | 18.04 | 308.00 | 4.50 | 579.00 | −4.65 | 257.08 |
| 38 | Sorom1 | 46.30 | 9.30 | 112.20 | 3.50 | 22.20 | 196.94 | 3.16 | 39.05 | 385.00 | 6.80 | 723.00 | −2.71 | 154.50 |
| 39 | Thianda Thiamene | 71.80 | 4.60 | 70.50 | 6.60 | 88.80 | 128.40 | 5.27 | 47.95 | 364.00 | 7.40 | 682.00 | −2.51 | 198.67 |
| 40 | Keur Mat Thiam | 20.34 | 2.51 | 25.39 | 1.09 | 57.95 | 27.99 | 0.95 | 12.98 | 112.00 | 7.30 | 210.00 | −0.27 | 61.32 |
| 41 | Passi Diemoul | 19.79 | 4.15 | 33.33 | 1.55 | 28.30 | 45.68 | 10.00 | 21.76 | 150.00 | 6.70 | 280.00 | −0.87 | 66.78 |
| 42 | Keur Madiabel | 77.10 | 9.40 | 97.40 | 3.10 | 73.20 | 198.75 | 10.81 | 22.39 | 386.00 | 7.40 | 728.00 | −3.42 | 231.92 |
| 43 | Koular | 24.80 | 9.50 | 35.60 | 32.20 | 20.25 | 84.66 | 4.76 | 48.36 | 239.00 | 5.30 | 448.00 | −1.69 | 101.58 |
| 44 | Koutango | 17.29 | 3.66 | 10.33 | 4.66 | 18.30 | 26.17 | 3.43 | 23.08 | 86.00 | 7.30 | 162.00 | −0.86 | 58.46 |
| 45 | Keur Maba Diakhou | 6.23 | 1.67 | 13.32 | 0.22 | 14.95 | 9.67 | 2.30 | 18.21 | 58.00 | 5.07 | 109.00 | −0.20 | 22.55 |
| 46 | Keur Moussa Poste | 1.14 | 1.31 | 7.01 | 0.32 | 16.78 | 5.95 | 0.00 | 5.48 | 40.30 | 4.80 | 75.90 | 0.11 | 8.29 |
| 47 | Porokhane | 40.80 | 15.10 | 22.40 | 3.70 | 59.65 | 55.59 | 13.26 | 45.30 | 284.00 | 7.10 | 535.00 | −2.30 | 164.92 |
| 48 | Nioro du Rip | 21.78 | 5.18 | 9.35 | 1.18 | 64.05 | 8.77 | 16.56 | 7.45 | 99.00 | 6.15 | 186.00 | −0.46 | 76.04 |
| 49 | Paoskoto | 91.50 | 5.20 | 9.70 | 0.00 | 228.75 | 18.01 | 8.93 | 6.09 | 212.00 | 6.41 | 400.00 | −1.24 | 250.42 |
| 50 | Taiba Niassene | 17.15 | 1.66 | 4.53 | 0.51 | 52.16 | 3.56 | 1.01 | 0.97 | 56.00 | 5.68 | 106.00 | −0.14 | 49.80 |
| 51 | Thiamene | 22.17 | 3.49 | 25.36 | 1.93 | 54.90 | 35.31 | 1.08 | 11.59 | 134.00 | 6.90 | 254.00 | −0.49 | 69.99 |
| 52 | Keur Ali Bassine | 83.27 | 4.13 | 19.85 | 1.33 | 207.40 | 29.17 | 3.09 | 10.93 | 216.00 | 6.91 | 406.00 | −1.10 | 225.38 |
| 53 | Keur Goumba Gueye | 62.14 | 4.00 | 23.50 | 0.76 | 128.10 | 46.02 | 4.37 | 12.21 | 190.00 | 6.69 | 361.00 | −1.33 | 172.01 |
| 54 | Keur Ayip KA | 15.20 | 3.90 | 70.50 | 2.70 | 53.05 | 61.39 | 3.49 | 48.75 | 236.00 | 4.95 | 443.00 | −0.21 | 54.25 |
| 55 | Thila babou Diarra | 10.81 | 4.40 | 19.93 | 1.23 | 14.20 | 18.59 | 1.71 | 43.06 | 121.00 | 6.90 | 227.00 | −0.67 | 45.39 |
| 56 | Keur Mama Lamine | 0.00 | 1.34 | 9.49 | 0.36 | 1.53 | 5.36 | 1.94 | 14.51 | 44.00 | 7.00 | 83.20 | −0.08 | 5.57 |
| 57 | keur Aliou DIOP | 56.30 | 17.50 | 57.60 | 1.90 | 199.15 | 54.27 | 2.23 | 50.00 | 313.00 | 6.90 | 588.00 | −0.98 | 213.67 |
| 58 | Ngayene Thiébou | 123.40 | 52.50 | 124.10 | 30.20 | 63.05 | 244.49 | 10.44 | 341.50 | 1090.00 | 4.20 | 2030.00 | −9.44 | 527.25 |
| 59 | Simon Diene | 5.49 | 1.35 | 13.42 | 1.71 | 15.25 | 8.13 | 2.84 | 18.75 | 71.00 | 7.30 | 135.00 | −0.14 | 19.37 |
| 60 | Keur Soce Sogo | 9.20 | 4.40 | 467.50 | 20.30 | 472.75 | 326.31 | 59.12 | 0.00 | 1110.00 | 8.30 | 2090.00 | 6.93 | 41.33 |
| 61 | Keur Niegne Wolof | 50.60 | 5.20 | 18.90 | 0.00 | 109.80 | 37.95 | 1.27 | 13.11 | 166.00 | 8.10 | 316.00 | −1.15 | 148.17 |
| 62 | Keur Mamour Koumba | 13.55 | 0.75 | 7.99 | 0.17 | 54.90 | 6.80 | 1.43 | 2.97 | 64.00 | 6.06 | 121.00 | 0.16 | 36.99 |
| 63 | Keur Mor Khouredia | 13.57 | 2.54 | 24.37 | 1.59 | 42.70 | 29.05 | 0.73 | 7.31 | 98.00 | 7.10 | 183.00 | −0.19 | 44.48 |
| 64 | Keur Abdou Yassine | 22.50 | 7.20 | 40.80 | 8.50 | 33.05 | 59.30 | 2.25 | 52.00 | 231.00 | 4.01 | 434.00 | −1.17 | 86.25 |
| 65 | Djalaba Sintye | 147.50 | 30.40 | 139.90 | 20.80 | 36.10 | 277.60 | 8.64 | 311.01 | 940.00 | 4.70 | 1770.00 | −9.27 | 495.42 |
| 66 | Djilor | 31.50 | 4.40 | 51.30 | 2.80 | 16.10 | 94.43 | 1.15 | 35.30 | 128.00 | 5.80 | 240.00 | −1.67 | 97.08 |
| Parameters | Min | Max | Average | Median | Standard Deviation |
|---|---|---|---|---|---|
| HCO3− (mg L−1) | 1.5 | 473.0 | 7223.0 | 43.5 | 82.5 |
| Cl− (mg L−1) | 3.0 | 2886.0 | 177.3 | 41.8 | 457.7 |
| NO3− (mg L−1) | 0.0 | 342.0 | 44.4 | 23.1 | 69.4 |
| SO42− (mg L−1) | 0.0 | 545.0 | 21.4 | 2.8 | 77.7 |
| K+ (mg L−1) | 0.0 | 128.7 | 10.5 | 1.9 | 22.9 |
| Na+ (mg L−1) | 4.5 | 1726.0 | 103.1 | 28.0 | 263.3 |
| Ca2+ (mg L−1) | 0.0 | 428.0 | 56.7 | 23.6 | 71.0 |
| Mg2+ (mg L−1) | 0.1 | 228.0 | 14.1 | 4.4 | 30.9 |
| pH | 3.8 | 8.3 | 6.9 | 6.7 | 1.1 |
| EC (µS cm−1) | 38.6 | 7160.0 | 763.1 | 346.0 | 1213.6 |
| TDS (mg L−1) | 20.6 | 3900.0 | 408.6 | 177.5 | 657.5 |
| HCO3− | Cl− | NO3− | SO42− | K+ | Na+ | Ca2+ | Mg2+ | pH | EC | TDS | |
| HCO3− (mg/L) | 1.00 | ||||||||||
| Cl− (mg/L) | 0.17 | 1.00 | |||||||||
| NO3− (mg/L) | −0.08 | 0.07 | 1.00 | ||||||||
| SO42− (mg/L) | −0.02 | 0.47 | −0.06 | 1.00 | |||||||
| K+ (mg/L) | 0.08 | 0.51 | 0.22 | 0.86 | 1.00 | ||||||
| Na+ (mg/L) | 0.25 | 0.99 | 0.07 | 0.50 | 0.53 | 1.00 | |||||
| Ca2+ (mg/L) | 0.32 | 0.78 | 0.31 | 0.39 | 0.60 | 0.72 | 1.00 | ||||
| Mg2+ (mg/L) | 0.01 | 0.90 | 0.19 | 0.60 | 0.58 | 0.90 | 0.63 | 1.00 | |||
| pH | 0.43 | −0.02 | −0.17 | −0.48 | −0.46 | −0.01 | −0.10 | −0.12 | 1.00 | ||
| E C (µs/cm) | 0.23 | 0.98 | 0.15 | 0.54 | 0.61 | 0.98 | 0.84 | 0.90 | −0.07 | 1.00 | |
| TDS (mg/L) | 0.27 | 0.98 | 0.15 | 0.54 | 0.61 | 0.98 | 0.83 | 0.90 | −0.05 | 1.00 | 1.00 |
| Parameters | WHO International Standard (1983, 2004) | Wells Exceeding Permissible Limits (Figure 1) | Undesirable Effect | |
|---|---|---|---|---|
| Desired Limit | Maximum Allowable Limits | |||
| pH | 6.5–8.5 | 9,2 | 6, 9, 14, 17, 22, 33, 37, 46, 54, 58, 64, 65 | Taste |
| TH (mg/L) | 100 | 500 | 6, 7, 9, 13, 14, 15, 32, 34, 58 | Scale formation |
| Na (mg/L) | - | 200 | 9, 13, 14, 32, 34, 60 | Taste |
| Ca (mg/L) | 75 | 200 | 9, 13, 15, 34 | Scale formation |
| Mg (mg/L) | 50 | 150 | 9 | |
| Cl (mg/L) | 250 | 600 | 9, 13, 32 | Salty Taste |
| SO4 (mg/L) | 200 | 500 | 14 | Taste |
| NO3 (mg/L) | 50 | - | 15, 17, 19, 22, 34, 58, 65 | Methaemoglobinaemia in infant an [60] |
| Based on TDS (mg/L) | Water Class | Number of Samples |
|---|---|---|
| <300 | Excellent | 43 |
| 300–600 | Good | 12 |
| 600–900 | Fair | 1 |
| 900–1200 | Poor | 7 |
| >1200 | Unacceptable | 3 |
| Classes of Water | Parameters | Suitable for Irrigation | |||
|---|---|---|---|---|---|
| TDS (mg/L) | SO42− (mg/L) | Cl− (mg/L) | EC (mS/cm) | ||
| I | 0–700 (55) | 0–192 (64) | 0–142 (51) | 0–0.75 (51) | Excellent to good for irrigation |
| II | 700–2000 (9) | 192–480 (1) | 142–355 (10) | 0.75–2.25 (12) | Good to injurious suitable soil |
| III | >2000 (2) | >480 (1) | >355 (5) | >2.25 (3) | Unfit for irrigation |
| SAR | Suitability for Irrigation |
|---|---|
| 1–10 (90.0%) | Suitable for all types of crops and soil except for those crops sensitive to sodium |
| 10–18 (4.0%) | Suitable for coarsed textured or organic soil with permeability |
| 18–26 (1.5%) | Harmful for almost all soil |
| >26 (4.5%) | Unsuitable for irrigation |
| RSC (meq/L) | Remark on Quality | Representing Wells |
|---|---|---|
| <1.25 | Good | All wells except No.60 |
| 1.25–2.5 | Doubtful | Nil |
| >2.5 | Unsuitable | 60 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ndoye, S.; Fontaine, C.; Gaye, C.B.; Razack, M. Groundwater Quality and Suitability for Different Uses in the Saloum Area of Senegal. Water 2018, 10, 1837. https://doi.org/10.3390/w10121837
Ndoye S, Fontaine C, Gaye CB, Razack M. Groundwater Quality and Suitability for Different Uses in the Saloum Area of Senegal. Water. 2018; 10(12):1837. https://doi.org/10.3390/w10121837
Chicago/Turabian StyleNdoye, Seyni, Claude Fontaine, Cheikh Becaye Gaye, and Moumtaz Razack. 2018. "Groundwater Quality and Suitability for Different Uses in the Saloum Area of Senegal" Water 10, no. 12: 1837. https://doi.org/10.3390/w10121837
APA StyleNdoye, S., Fontaine, C., Gaye, C. B., & Razack, M. (2018). Groundwater Quality and Suitability for Different Uses in the Saloum Area of Senegal. Water, 10(12), 1837. https://doi.org/10.3390/w10121837






