Capillary Nanofiltration under Anoxic Conditions as Post-Treatment after Bank Filtration
Abstract
1. Introduction
2. Materials and Methods
2.1. Feed Water Source
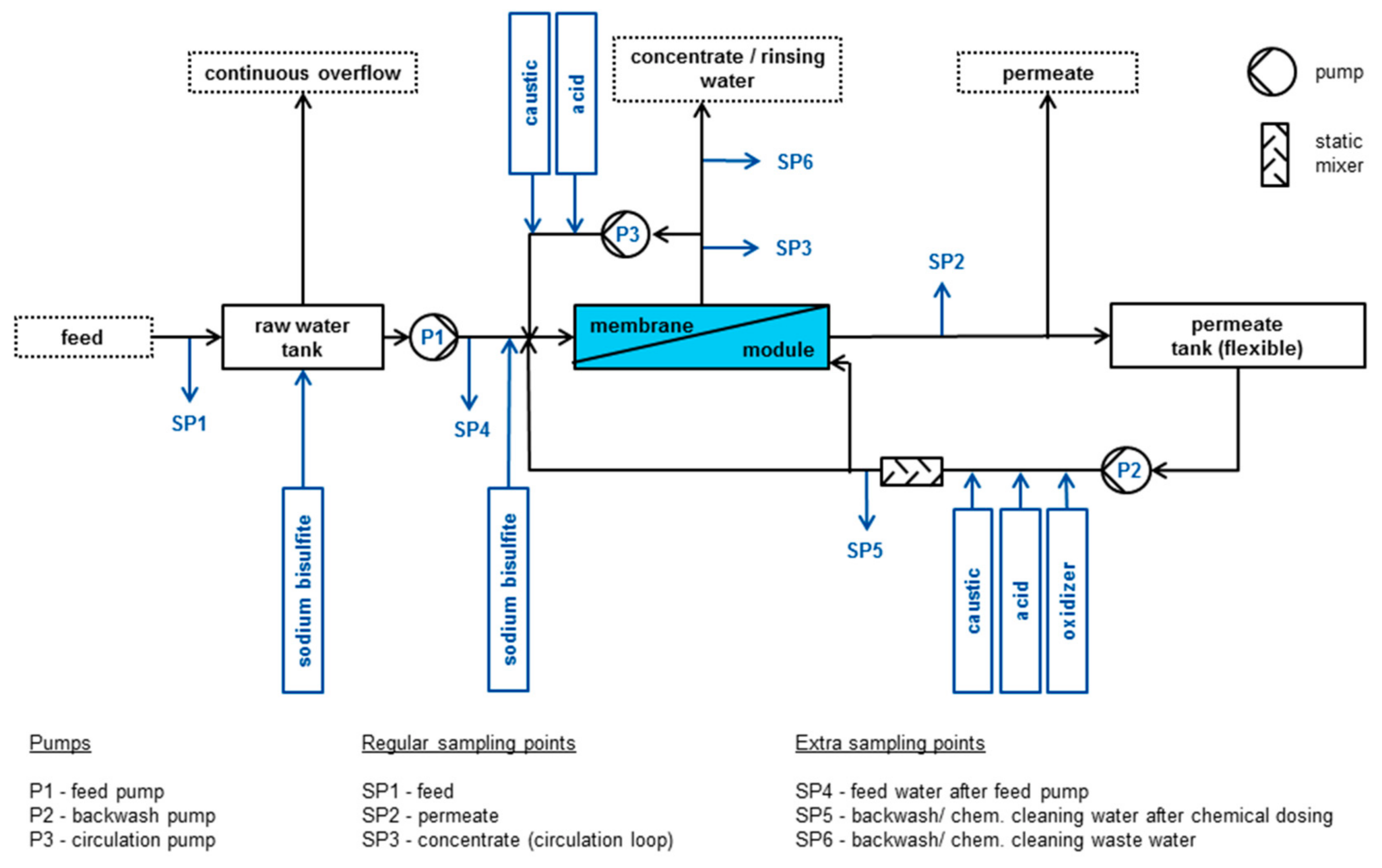
2.2. Capillary Nanofiltration
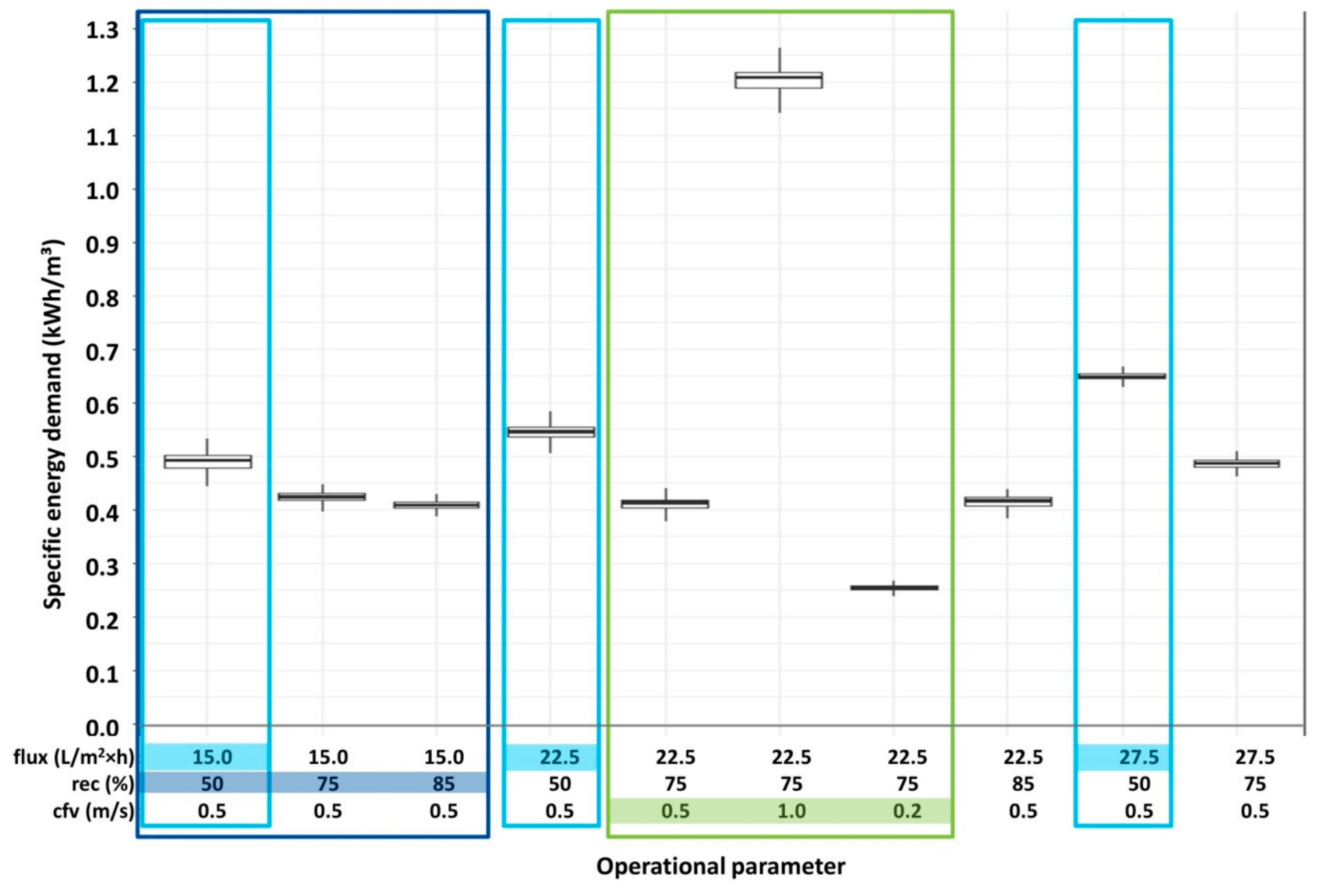
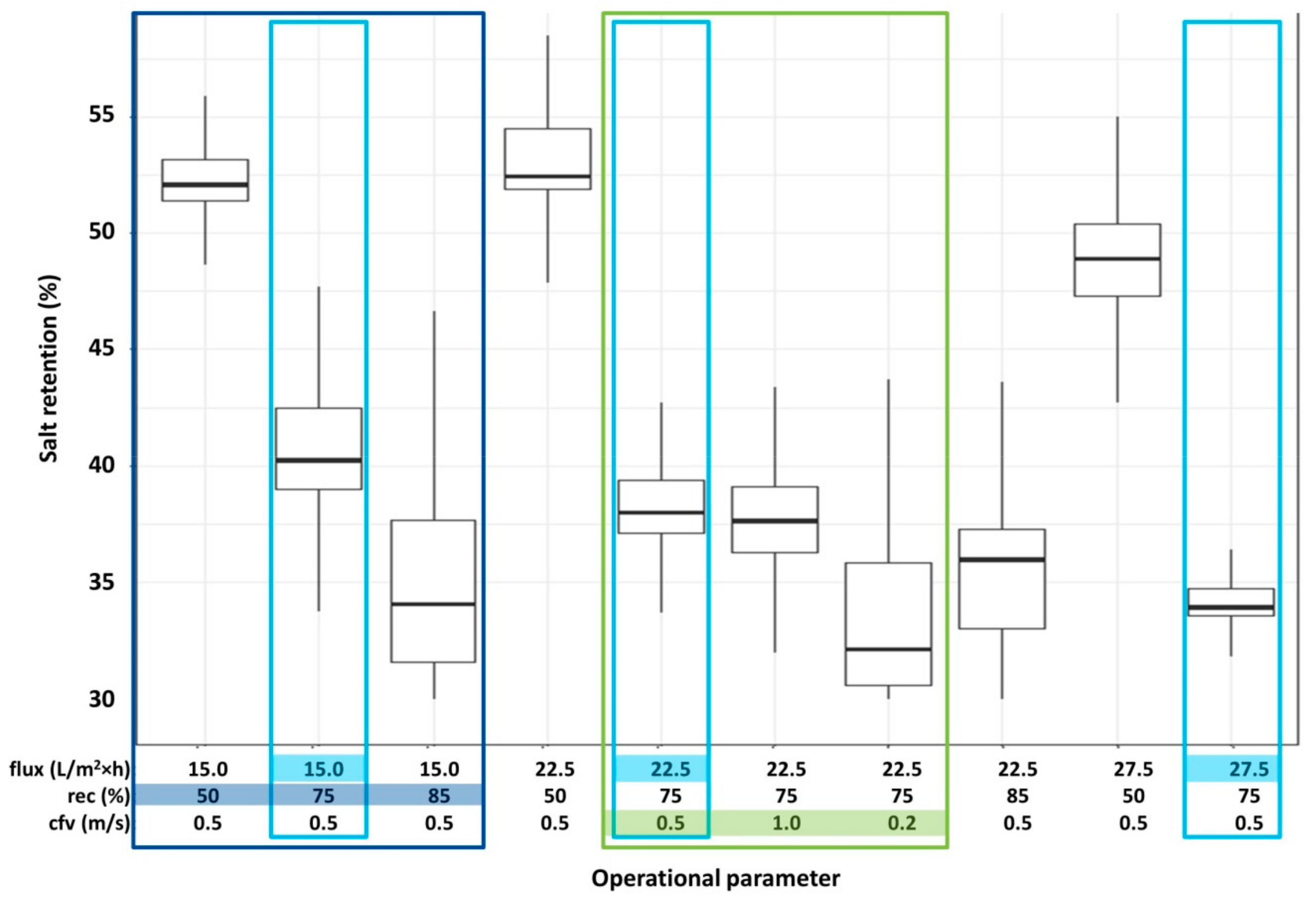
- flux (15/22.5/27.5/30 L/m2·h)
- recovery (rec) (50/75/85%)
- cross-flow velocity (cfv) (0.2/0.5/1.0 m/s)
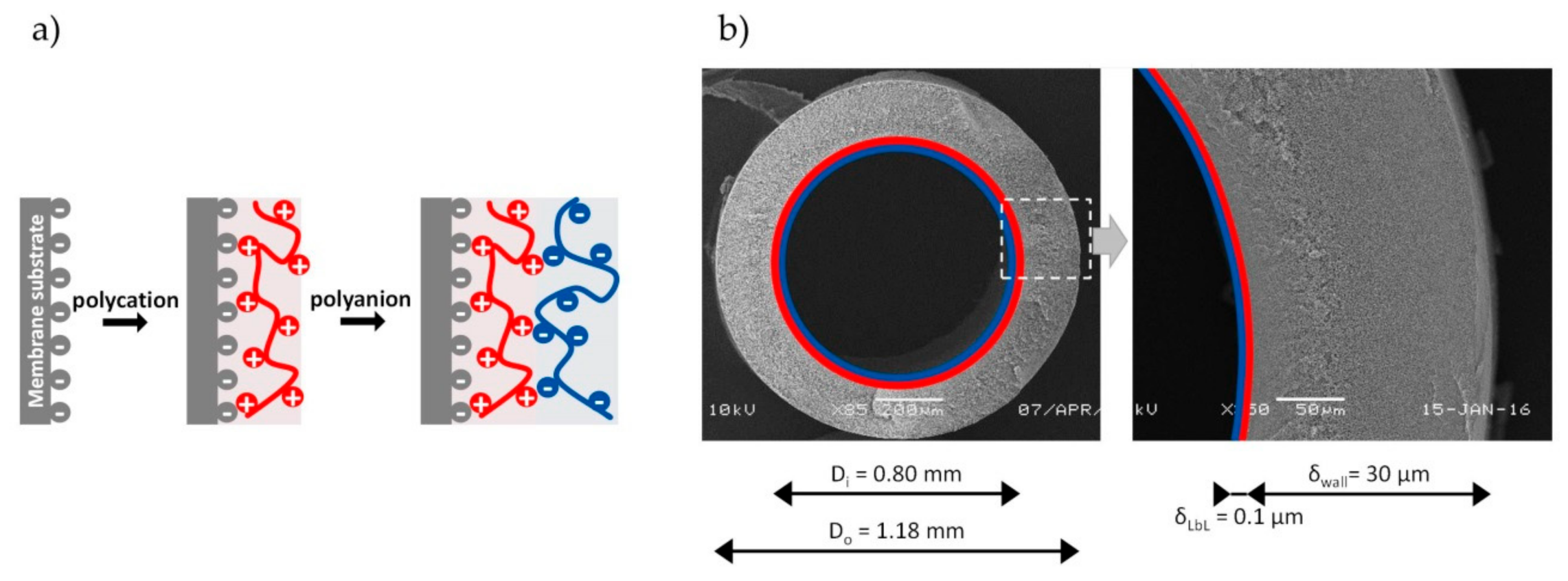
2.3. Membrane
2.4. Online Data and Water Analysis
2.5. Calculation of Energy Consumption
2.6. Chemical Cleaning
3. Results and Discussion
3.1. Operational Parameters and Energy Consumption
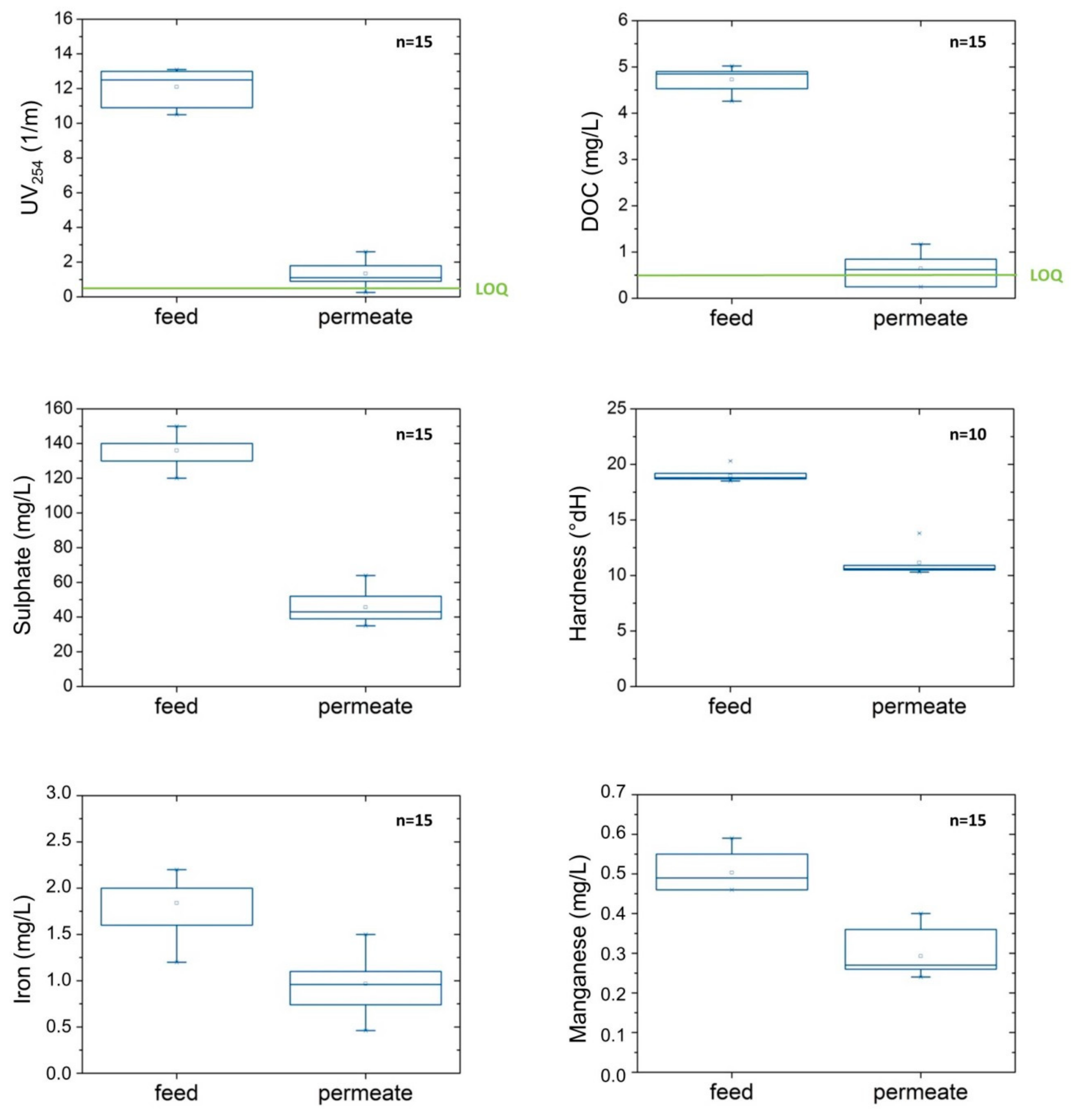
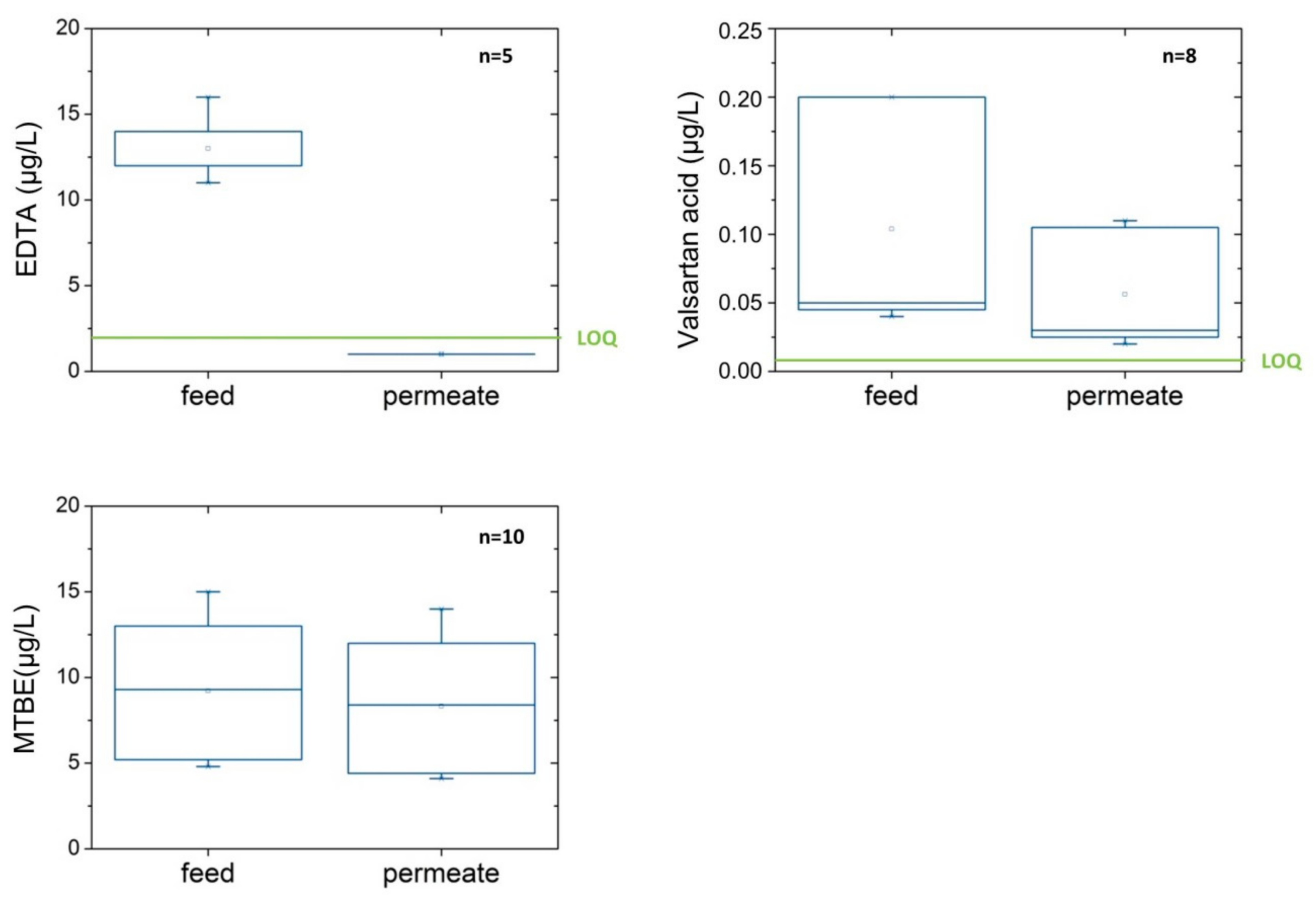
3.2. Retention of Compounds
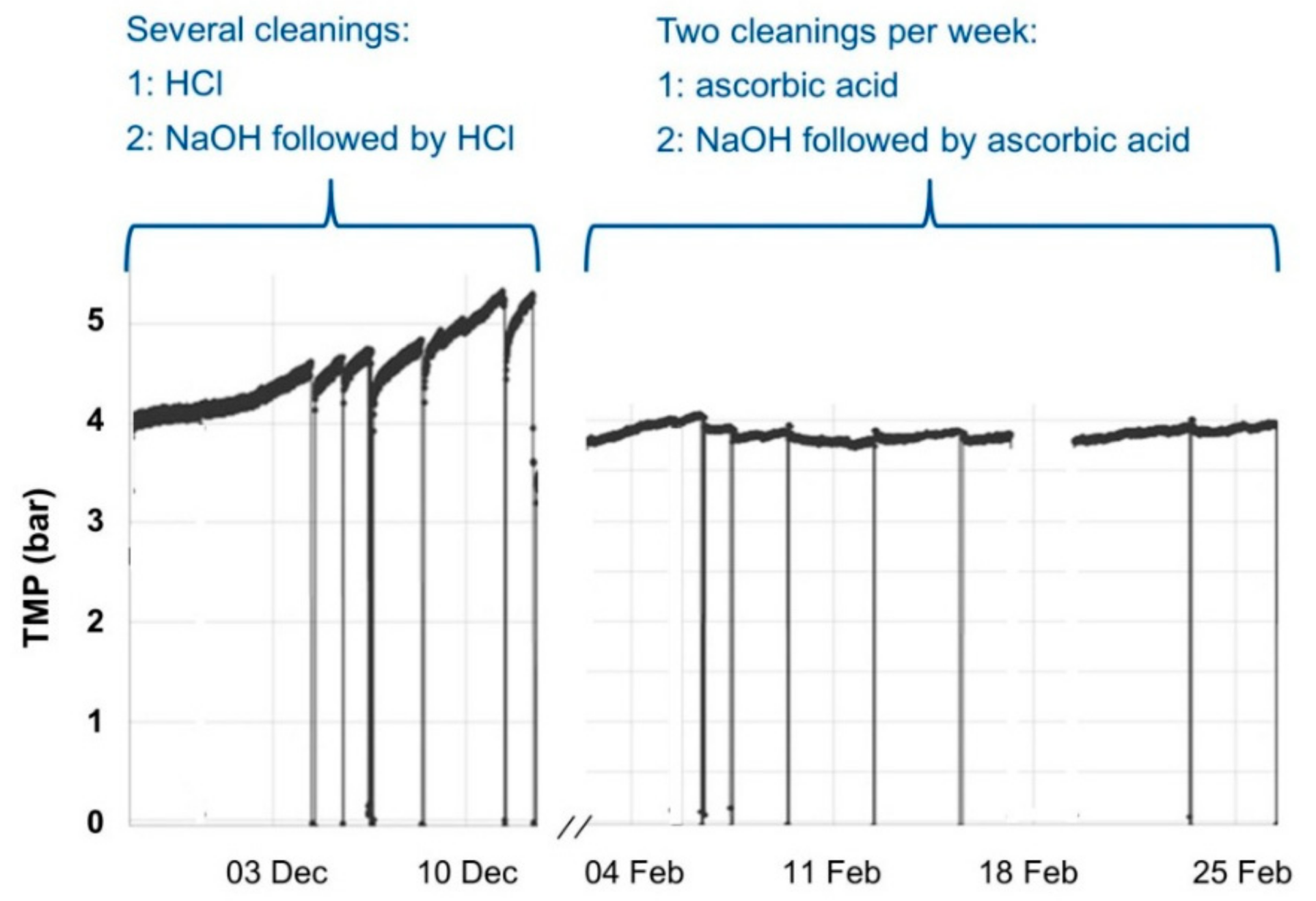
3.3. Hydraulical and Chemical Cleaning
3.4. Online Probes
3.5. Concentrate Discharge
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| capNF | capillary nanofiltration |
| CBZD | Carbamazepine-10,11-trans dihydrodiol |
| cfv | cross-flow velocity |
| DNEL | derived no-effect level |
| DOC | dissolved organic carbon |
| EDTA | Ethylendiamintetraacetic acid |
| FAA | Formylaminoantipyrine |
| LbL | Layer-by-Layer |
| LOQ | limit of quantification |
| MTBE | Methyl tertiary butyl ether |
| MW | molecular weight |
| MWCO | molecular weight cut-off |
| NF | nanofiltration |
| OMP | organic micropollutants |
| ORP | oxidation-reduction potential |
| PEMA | Phenylethyl-malonamide |
| PES | polyethersulfone |
| rec | recovery |
| TBA | Tertiary butyl alcohol |
| TMP | transmembrane pressure |
| UV254 | absorption of ultraviolet light (wave length: 254 nm) |
| ZLD | zero liquid discharge |
Appendix A
| Parameter | Unit | Feed | Permeate | Concentrate | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| (a) | (b) | (a) | (b) | ||||||||
| Temperature | °C | 12.8 4 | ±0.92 | 12.0 2 | ±0.12 | 13.0 5 | ±0.83 | 11.9 2 | ±0.15 | 13.1 6 | ±0.83 |
| pH | – | 7.0 4 | ±0.21 | 7.6 2 | ±0.31 | 7.0 5 | ±0.16 | 7.7 2 | ±0.34 | 7.1 6 | ±0.16 |
| ORP | mV | −100 8 | ±26 | – | – | −80 9 | ±25 | – | – | – | – |
| Oxygen | mg/L | <LOQ | – | – | 0.2 7 | ±0.14 | – | – | – | – | |
| Conductivity | µS/cm | 961 4 | ±80 | 932 2 | ± 19 | 744 5 | ±101 | 1162 2 | ±38 | 1549 5 | ±93 |
| Color436nm | 1/m | 0.3 1 | ±0.00 | <LOQ 2 | <LOQ 3 | 0.5 2 | ±0.00 | 1.0 3 | ±0.14 | ||
| UV254 | 1/m | 11.6 1 | ±0.13 | 4.3 2 | ±0.26 | 1.0 3 | ±0.14 | 19.0 2 | ±1.42 | 39.3 3 | ±3.18 |
| DOC | mg/L | 4.8 1 | ±0.15 | 1.9 2 | ±0.10 | 0.6 3 | ±0.02 | 7.4 2 | ±0.43 | 15.5 3 | ±1.06 |
| Fe total | mg/L | 1.8 1 | ±0.23 | 1.6 2 | ±0.17 | 0.6 3 | ±0.21 | 2.6 2 | ±0.35 | 3.3 3 | ±1.70 |
| Mn total | mg/L | 0.5 1 | ±0.02 | 0.4 2 | ±0.05 | 0.2 3 | ±0.00 | 0.6 2 | ±0.08 | 1.2 3 | ±0.14 |
| Sulphate | mg/L | 131 1 | ±0.92 | 73 2 | ±3.06 | 36 3 | ±0.71 | 200 2 | ±0.00 | 395 3 | ±7.07 |
| Parameter | Unit | Measuring Place | Measuring Probe | Measuring Method |
|---|---|---|---|---|
| Level | % | feed tank, | Endress + Hauser Liquiphant M | submerged gauge pressure sensor |
| permeate tank | Endress + Hauser Prosonic M | reflection ultrasonic pulses | ||
| T | °C | feed | Endress + Hauser Omnigrad M TR10 | resistance thermometer |
| Flow | m3/h | feed, concentrate, circulation, backwash | Endress + Hauser Promag 10P25 | magnetic induction |
| Pressure | bar | feed before/after restriction, feed, permeate, concentrate, backwash before/after restriction | Endress + Hauser Cerabar M | pressure-dependent change in capacitance |
| pH | - | feed, circulation | Endress + Hauser Memosens CPS16D | glass electrode with Ag/AgCl reference |
| ORP | mV | feed, permeate, concentrate (circulation) | Pt electrode with Ag/AgCl reference | |
| T | °C | circulation | NTC 30kΩ | |
| Conductivity | µS/cm | feed, permeate, concentrate (circulation) | Endress+Hauser Condumax CLS21D (old) Indumax CLS50D (new) | resistance between 2 electrodes (old) digital inductive (new) |
| Turbidity UV254 | NTU 1/m | feed, permeate | i::scan V1 Y04 | ISO7027/EPA 180.1 multi-wavelength photometer with narrow band light source |
| Parameter | Unit | LOQ | Measuring Method |
|---|---|---|---|
| Calcium | mg/L | 0.8 | DIN EN ISO 11885 (E22) |
| Color436nm | 1/m | 0.2 | DIN EN ISO 7887 (C01) |
| Conductivity | µS/cm | - | DIN EN 27888 (C08) |
| DOC | mg/L | 0.5 | DIN EN 1484 (H03) |
| Fe2+ | mg/L | 0.03 | DIN 38406-E01 |
| Fedissolved | mg/L | 0.03 | DIN EN ISO 11885 (E22) |
| Fetotal | mg/L | 0.03 | DIN EN ISO 11885 (E22) |
| Hardness | °dH | - | DIN 38409-H06 |
| Magnesium | mg/L | 0.1 | DIN EN ISO 11885 (E22) |
| Mndissolved | mg/L | 0.01 | DIN EN ISO 11885 (E22) |
| Mntotal | mg/L | 0.01 | DIN EN ISO 11885 (E22) |
| Sulphate | mg/L | 6.0 | DIN EN ISO 10304-1 (D20) |
| UV254 | 1/m | 0.5 | DIN 38404-C03 |
| Acid capacity | mmol/L | 0.02 | DIN 38409-H07-1/2 |
| Base capacity | mmol/L | 0.02 | DIN 38409-H07-2 |
| Calcite dissolving capacity | mmol/L | - | DIN 38404-C10-R3 |
| Chloride | mg/L | 5.0 | DIN EN ISO 10304-1 (D20) |
| Hydrogen carbonate | mg/L | - | calculated using DIN 38409-7 |
| Nitrate | mg/L | 0.2 | DIN EN ISO 10304-1 (D20) |
| Nitrate-N | mg/L | 0.05 | DIN EN ISO 10304-1 (D20) |
| Potassium | mg/L | 0.02 | DIN EN ISO 11885 (E22) |
| Sodium | mg/L | 0.5 | DIN EN ISO 11885 (E22) |
| Acesulfame | µg/L | 0.1 | DIN 38407-F47 |
| Candesartan | µg/L | 0.01 | DIN 38407-F47 |
| Carbamazepine | µg/L | 0.01 | DIN 38407-F47 |
| CBZD | µg/L | 0.02 | DIN 38407-F47 |
| EDTA | µg/L | 2.0 | DIN EN ISO 16588 (P10) |
| FAA | µg/L | 0.01 | DIN 38407-F47 |
| Gaba-lactam | µg/L | 0.01 | DIN 38407-F47 |
| Gabapentin | µg/L | 0.01 | DIN 38407-F47 |
| Iopamidol | µg/L | 0.02 | DIN 38407-F36 |
| MTBE | µg/L | 0.03 | DIN 38407-F43 |
| Olmesartan | µg/L | 0.01 | DIN 38407-F47 |
| PEMA | µg/L | 0.01 | DIN 38407-F47 |
| Primidone | µg/L | 0.01 | DIN 38407-F47 |
| TBA | µg/L | 1.0 | DIN 38407-F43 |
| Valsartan acid | µg/L | 0.01 | DIN 38407-F47 |
| Vinyl chloride | µg/L | 0.1 | DIN EN ISO 10301 (F04) |
| pH | - | 0.01 | Std. Methods 4500-H+ EPA 150.2 (SmarTROLL™) |
| T | °C | −5.0 | EPA 170.1 (SmarTROLL™) |
| ORP | mV | ±1400 | Std. Methods 2580 (SmarTROLL™) |
| Conductivity | µS/cm | 5.0 | Std. Methods 2510 EPA 120.1 (SmarTROLL™) |
| Oxygen | mg/L | 0.1 | EPA-approved In-Situ Methods 1002-8-2009 1003-8-2009 1004-8-2009 (SmarTROLL™) |
| Parameter | Molecular Weight (g/mol) 1 | Structure 1 | Parameter | Molecular Weight (g/mol) 1 | Structure 1 |
|---|---|---|---|---|---|
| Vinyl chloride | 62.50 |  | TBA | 74.12 |  |
| MTBE | 88.15 | 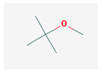 | Gabalactam | 153.225 |  |
| Gabapentin | 171.24 |  | Acesulfame | 201.24 | 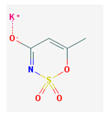 |
| PEMA | 206.24 | 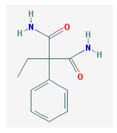 | Primidone | 218.252 | 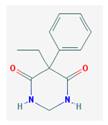 |
| FAA | 231.25 | 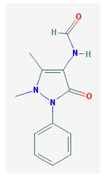 | Carba- mazepine | 236.27 |  |
| Valsartan acid | 266.08 | 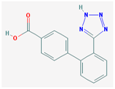 | CBZD | 270.29 |  |
| EDTA | 292.24 | 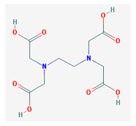 | Cande-sartan | 440.45 (candesartan) 610.66 (candesartan- cilexetil) | 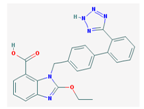 |
| Olmesartan | 446.51 | 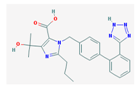 | Iopamidol | 777.08 | 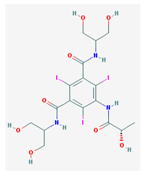 |
| Chemical | Concentration (wt. %) | Active stock Concentration (g/L) |
|---|---|---|
| HCl | 25 | 280 |
| NaOH | 35 | 483 |
| Ascorbic acid | 20 | 200 |
| NaHSO3 | 39 | 522.6 |
References
- Ascott, M.J.; Lapworth, D.J.; Gooddy, D.C.; Sage, R.C.; Karapanos, I. Impacts of extreme flooding on riverbank filtration water quality. Sci. Total Environ. 2016, 554–555, 89–101. [Google Scholar] [CrossRef] [PubMed]
- Przybyłek, J.; Dragon, K.; Kaczmarek, P.M.J. Hydrogeological investigations of river bed clogging at a river bank filtration site along the River Warta, Poland. Geologos 2017, 23, 201–214. [Google Scholar] [CrossRef]
- Kaczmarek, P.M.J. Hydraulic conductivity changes in river valley sediments caused by river bank filtration—An analysis of specific well capacity. Geologos 2017, 23, 123–129. [Google Scholar] [CrossRef]
- Uhlmann, W.; Zimmermann, K. Fallanalyse der Sulfatbelastung in der Spree 2014/2015; Senatsverwaltung für Stadtentwicklung und Umwelt Berlin: Berlin, Germany, 2015. [Google Scholar]
- SenStadtUm Berlin and MWE Brandenburg. Sulfatgespräche der Länder Berlin und Brandenburg—Aktueller Sachstand und Maßnahmen zur Beherrschung der Bergbaulich Bedingten Stoffeinträge; Senatsverwaltung für Stadtentwicklung und Umwelt des Landes Berlin; Ministerium für Wirtschaft und Energie des Landes Brandenburg: Berlin, Germany, 2016. [Google Scholar]
- Galloway, J.N.; Aber, J.D.; Erisman, J.W.; Seitzinger, S.P.; Howarth, R.W.; Cowling, E.B.; Cosby, B.J. The Nitrogen Cascade. BioScience 2003, 53, 341–356. [Google Scholar] [CrossRef]
- Heberer, T. Occurrence, fate, and removal of pharmaceutical residues in the aquatic environment: A review of recent research data. Toxicol. Lett. 2002, 131, 5–17. [Google Scholar] [CrossRef]
- Destatis. Trinkwasser Wird Überwiegend aus Grundwasser Gewonnen; Statistisches Bundesamt Pressestelle: Wiesbaden, Germany, 2013. [Google Scholar]
- SenUVK. Grundwasser in BerlinVorkommen Nutzung Schutz Gefährdung; Senatsverwaltung für Umwelt, Verkehr und Klimaschutz: Berlin, Germany, 2007. [Google Scholar]
- Bundesgesundheitsamt. Verordnung über die Qualität von Wasser für den Menschlichen Gebrauch (Trinkwasserverordnung-TrinkwV). Available online: http://www.gesetze-im-internet.de/trinkwv_2001/BJNR095910001.html#BJNR095910001BJNG000201310 (accessed on 12 September 2018).
- SenStadt Berlin. Qualität des oberflächennahen Grundwassers, Senat für Stadtentwicklung und Umwelt, Umweltatlas. Available online: www.stadtentwicklung.berlin.de/umwelt/umweltatlas/ka204.htm (accessed on 8 April 2018).
- SenStadtUm. Das Grundwasser in Berlin—Bedeutung, Probleme, Sanierungskonzeptionen; Senatsverwaltung für Stadtentwicklung und Umwelt des Landes Berlin: Berlin, Germany, 1986. [Google Scholar]
- Wurl, J. Die geologischen, hydraulischen und hydrochemischen Verhältnisse in den südwestlichen Stadtbezirken von Berlin; Fachbereich Geowiss., FU: Berlin, Germany, 1995; 3-89582-003-2 978-3-89582-003-8. [Google Scholar]
- Schmidt, C.K.; Lange, F.T.; Brauch, H.-J.; Kühn, W. Experiences with Riverbank Filtration and Infiltration in Germany; DVGW-Water Technology Center (TZW): Karlsruhe, Germany, 2003. [Google Scholar]
- Ziegler, D. Untersuchung zur Nachhaltigen Wirkung der Uferfiltration im Wasserkreislauf Berlins; Technische Universität Berlin: Berlin, Germany, 2001. [Google Scholar]
- Darbi, A.; Viraraghavan, T.; Jin, Y.-C.; Braul, L.; Corkal, D. Sulfate Removal from Water. Water Qual. Res. J. Can. 2003, 88, 169–182. [Google Scholar] [CrossRef]
- Lipp, P.; Gronki, T.; Lueke, J.; Lanfervoss, A.; Baldauf, G. Sulphate Removal from Ground Water—A Case Study. gwf-Wasser/Abwasser 2011, 152, 46–51. [Google Scholar]
- Cao, W.; Dang, Z.; Zhou, X.-Q.; Yi, X.-Y.; Wu, P.-X.; Zhu, N.-W.; Lu, G.-N. Removal of sulphate from aqueous solution using modified rice straw: Preparation, characterization and adsorption performance. Carbohydr. Polym. 2011, 85, 571–577. [Google Scholar] [CrossRef]
- Liu, H.; Li, F.; Zhang, G. Experimental Study on Adsorption Removal of Sulfate with Synthesized Zeolite Made from Fly Ash. In Proceedings of the 4th International Conference on Bioinformatics and Biomedical Engineering, Chengdu, China, 18–20 June 2010. [Google Scholar]
- Rao, S.M.; Sridharan, A. Mechanism of Sulfate Adsorption by Kaolinite. Clays Clay Miner. 1984, 32, 414–418. [Google Scholar] [CrossRef]
- Lindner, K.; Knepper, T.P.; Müller, J.; Karrenbrock, F.; Rörden, O.; Brauch, H.-J.; Sacher, F. Entwicklung von Verfahren zur Bestimmung und Beurteilung der Trinkwassergängigkeit von Organischen Einzelstoffen; Internationale Arbeitsgemeinschaft der Wasserwerke im Rheineinzugsgebiet: Köln, Germany, 2000. [Google Scholar]
- Lindner, K.; Knepper, T.P.; Müller, J.; Karrenbrock, F.; Rörden, O.; Juchem, H.; Brauch, H.-J.; Sacher, F. Bestimmung und Beurteilung der Mikrobiellen Abbaubarkeit von Organischen Einzelstoffen bei Umweltrelevanten Konzentrationen in Gewässern; Internationale Arbeitsgemeinschaft der Wasserwerke im Rheineinzugsgebiet: Köln, Germany, 2003. [Google Scholar]
- Wisotzky, F.; Jakschik, S.; Denzig, D. Herkunft und Dauer einer EDTA-Belastung im Grundwasser zweier Trinkwassereinzugsgebiete. gwf-Wasser/Abwasser 2010, 3, 278–284. [Google Scholar]
- Krueger-Marondel, J. Senatsverwaltung für Umwelt, Verkehr und Klimaschutz, Berlin, Germany. Personal communication, 2018. [Google Scholar]
- Samhaber, W.M. Erfahrungen und Anwendungspotential der Nanofiltration; Institut für Verfahrenstechnik, Johannes Kepler Universität Linz: Linz, Austria, 2006. [Google Scholar]
- Cadotte, J.; Forester, R.; Kim, M.; Petersen, R.; Stocker, T. Nanofiltration membranes broaden the use of membrane separation technology. Desalination 1988, 70, 77–88. [Google Scholar] [CrossRef]
- Raman, L.P.; Cheryna, M.; Rajagopalan, N. Consider nanofiltration for membrane separations. Chem. Eng. Prog. 1994, 90, 68–74. [Google Scholar]
- Hilal, N.; Al-Zoubi, H.; Darwish, N.A.; Mohamma, A.W.; Abu Arabi, M. A comprehensive review of nanofiltration membranes: Treatment, pretreatment, modelling, and atomic force microscopy. Desalination 2004, 170, 281–308. [Google Scholar] [CrossRef]
- Radier, R.G.J.; van Oers, C.W.; Steenbergen, A.; Wessling, M. Desalting a process cooling water using nanofiltration. Sep. Purif. Technol. 2001, 22–23, 159–168. [Google Scholar] [CrossRef]
- Hiemstra, P.; van Paassen, J.; Rietman, B.; Verdouw, J. Aerobic versus Anaerobic Nanofiltration: Fouling of Membranes. In Proceedings of the AWWA Membrane Technology Conference, Long Beach, CA, USA, 28 February–3 March 1999; pp. 55–82. [Google Scholar]
- Nederlof, M.M.; Kruithof, J.C.; Taylor, J.S.; van der Kooij, D.; Schippers, J.C. Comparison of NF/RO membrane performance in integrated membrane systems. Desalination 2000, 131, 257–269. [Google Scholar] [CrossRef]
- Vrouwenvelder, J.S.; Kruithof, J.C.; Van Loosdrecht, M.C.M. Integrated approach for biofouling control. Water Sci. Technol. 2010, 62, 2477–2490. [Google Scholar] [CrossRef] [PubMed]
- Beyer, F.; Rietman, B.M.; Zwijnenburg, A.; van den Brink, P.; Vrouwenvelder, J.S.; Jarzembowska, M.; Laurinonyte, J.; Stams, A.J.M.; Plugge, C.M. Long-term performance and fouling analysis of full-scale direct nanofiltration (NF) installations treating anoxic groundwater. J. Membr. Sci. 2014, 468, 339–348. [Google Scholar] [CrossRef]
- Potreck, J. Enhanced membrane retention and mass transfer through smart surface modification. In Proceedings of the EuroMebrane 2015, Aachen, Germany, 6–10 September 2015. [Google Scholar]
- Vriezekolk, E.; Pacak, A.; Potreck, J. Poster presentation: Layer-by-layer nanofiltration membranes; From fundamental research to large-scale membrane modules. In Proceedings of the Imagine Membrane, Azores, Portugal, 24–29 September 2017. [Google Scholar]
- Rustler, M. Aquanes Report; Version v.0.5.0; Zenodo: Genève, Switzerland, 2018. [Google Scholar] [CrossRef]
- R Development Core Team. R: A Language and Environment for Statistical Computing. 2017. Available online: www.R-project.org/ (accessed on 12 September 2018).
- Sethi, S.; Wiesner, M.R. Cost Modeling and Estimation of Crossflow Membrane Filtration Processes. Environ. Eng. Sci. 2000, 17, 61–79. [Google Scholar] [CrossRef]
- Arbeitsgemeinschaft Trinkwassertalsperren e. V.; Bundesverband der Energie und Wasserwirtschaft e. V.; Deutscher Bund der verbandlichen Wasserwirtschaft e. V.; Deutscher Verein des Gas und Wasserfaches e. V.—Technisch-wissenschaftlicher Verein; Deutsche Vereinigung für Wasserwirtschaft, Abwasser und Abfall e. V.; Verband kommunaler Unternehmen e. V. Branchenbild der deutschen Wasserwirtschaft 2015; wvgw Wirtschafts- und Verlagsgesellschaft Gas und Wasser: Bonn, Germany, 2015. [Google Scholar]
- Keucken, A.; Wang, Y.; Tng, K.H.; Leslie, G.; Spanjer, T.; Köhler, S.J. Optimizing hollow fibre nanofiltration for organic matter rich lake water. Water 2016, 8, 430. [Google Scholar] [CrossRef]
- Heidfors, I.; Vredenbregt, L.H.J.; Homes, A.; van Es, M.B. Pilot testing with hollow fiber nano filtration membranes for removal of NOM from surface water in Sweden. In Proceedings of the NOM 6 2015 (6th Specialist Conference on Natural Organic Matter in Drinking Water), Malmö, Sweden, 7–10 September 2015. [Google Scholar]
- Dieter, H.H.; Frank, D.; Gihr, R.; Konietzka, R.; Moll, B.; Stockerl, R.; von der Trenck, T.; Schudoma, D.; Zedler, B.; Brodsky, J. Ableitung von Geringfügigkeitsschwellenwerten für das Grundwasser—Aktualisierte und Überarbeitete Fassung 2016. Available online: www.lawa.de/documents/Geringfuegigkeits_Bericht_Seite_001-028_6df.pdf (accessed on 12 September 2018).
- Möller, K.; Kade, N.; Havermeier, L.; Paproth, F.; Burgschweiger, J.; Wittstock, E.; Günther, M.; Naumann, K.; Broll, J. Wasserversorgungskonzept für Berlin und für das von den BWB Versorgte Umland (Entwicklung bis 2040); Berliner Wasserbetriebe: Berlin, Germany, 2008. [Google Scholar]
- Zhang, Z.; Bligh, M.W.; Waite, T.D. Ascorbic acid-mediated reductive cleaning of iron-fouled membranes from submerged membrane bioreactors. J. Membr. Sci. 2015, 477, 194–202. [Google Scholar] [CrossRef]
- Zhang, Z.; Bligh, M.W.; Wang, Y.; Leslie, G.L.; Bustamante, H.; Waite, T.D. Cleaning strategies for iron-fouled membranes from submerged membrane bioreactor treatment of wastewaters. J. Membr. Sci. 2015, 475, 9–21. [Google Scholar] [CrossRef]
- Müller, U.; Baldauf, G.; Osmera, S.; Göttsche, R. Erfassung und Bewertung von Nanofiltrations und Niederdruckumkehrosmoseanlagen in der Öffentlichen Wasserversorgung in Deutschland; DVGW-Technologiezentrum Wasser Karlsruhe (TZW): Karlsruhe, Germany, 2009. [Google Scholar]
- Semblante, G.U.; Lee, J.Z.; Lee, L.Y.; Ong, S.L.; Ng, H.Y. Brine pre-treatment technologies for zero liquid discharge systems. Brine Pre-Treat. Technol. Zero Liq. Discharge Syst. 2018, 441, 96–111. [Google Scholar] [CrossRef]
- PubChem Substance and Compound databases. PubChem—Open Chemistry Database. Available online: https://pubchem.ncbi.nlm.nih.gov (accessed on 22 August 2018).
| Parameter | Unit | Feed | Permeate * | Concentrate * | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Temperature | °C | 12.8 | ±0.92 | (n = 60) | 13.0 | ±0.83 | (n = 51) | 13.1 | ±0.83 | (n = 33) |
| pH | – | 7.0 | ±0.21 | (n = 60) | 7.0 | ±0.16 | (n = 51) | 7.1 | ±0.16 | (n = 33) |
| ORP | mV | −100 | ±26 | (n = 17) | -80 | ±25 | (n = 16) | n.d. ** | ||
| Oxygen | mg/l | <LOQ | (n = 16) | 0.2 | ±0.14 | (n = 12) | n.d. ** | |||
| Conductivity | µS/cm | 961 | ±80 | (n = 60) | 744 | ±101 | (n = 51) | 1549 | ±93 | (n = 51) |
| Color436nm | 1/m | 0.3 | ±0.00 | (n = 5) | <LOQ | (n = 2) | 1.0 | ±0.14 | (n = 2) | |
| UV254 | 1/m | 11.6 | ±0.13 | (n = 5) | 1.0 | ±0.14 | (n = 2) | 39.3 | ±3.18 | (n = 2) |
| DOC | mg/l | 4.8 | ±0.15 | (n = 5) | 0.6 | ±0.02 | (n = 2) | 15.5 | ±1.06 | (n = 2) |
| Parameter | Unit | Feed: Drinking Water 1 | Feed: Well Water 2 | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Feed | Retention (%) | Feed | Retention (%) | ||||||||
| UV254 | 1/m | 8.7 | ±0.23 | 85 | ±6.5 | (n = 9) | 12.1 | ±1.01 | 89 | ±5.5 | (n = 15) |
| DOC | mg/L | 3.8 | ±0.21 | 82 | ±7.8 | (n = 11) | 4.7 | ±0.24 | 87 | ±6.4 | (n = 15) |
| Sulphate | mg/L | 149.1 | ±9.44 | 71 | ±8.5 | (n = 11) | 136.0 | ±7.37 | 67 | ±5.5 | (n = 16) |
| Mg | mg/L | 10.7 | ±0.43 | 64 | ±13.8 | (n = 5) | 11.7 | ±0.49 | 50 | ±6.0 | (n = 10) |
| Hardness | °dH | 20.5 | ±0.66 | 55 | ±14.7 | (n = 5) | 18.8 | ±0.51 | 41 | ±5.2 | (n = 10) |
| Ca | mg/L | 128.6 | ±4.93 | 54 | ±14.8 | (n = 5) | 115.1 | ±3.16 | 40 | ±5.0 | (n =10) |
| Fetotal | mg/L | <LOQ | – | (n = 11) | 1.8 | ±0.29 | 48 | ±8.7 | (n = 15) | ||
| Fe2+ | mg/L | <LOQ | – | (n = 11) | 1.6 | ±0.28 | 48 | ±7.7 | (n = 15) | ||
| Fedissolved | mg/L | <LOQ | – | (n = 11) | 1.8 | ±0.31 | 49 | ±8.0 | (n = 14) | ||
| Mntotal | mg/L | <LOQ | – | (n = 11) | 0.5 | ±0.05 | 42 | ±5.8 | (n = 15) | ||
| Mndissolved | mg/L | <LOQ | – | (n = 11) | 0.5 | ±0.05 | 42 | ±6.0 | (n = 15) | ||
| Conductivity | µS/cm | 948.0 | ±71.0 | 32 | ±5.8 | (n = 11) | 970.0 | ±78.3 | 22 | ±4.3 | (n = 40) |
| Color | 1/m | 0.2 | ±0.04 | – | (n = 5) 4 | 0.3 | ±0.05 | – | (n = 15) 4 | ||
| EDTA | µg/L | 11.0 | ±0.00 | 84 | ±10.3 | (n = 2) | 12.3 | ±3.73 | 92 | ±1.1 | (n = 5) |
| Gabapentin | µg/L | 0.1 | ±0.04 | 55 | ±12.1 | (n = 4) | 0.05 | ±0.03 | 46 | ±10.6 | (n = 8) 3 |
| Valsartan acid | µg/L | 0.2 | ±0.06 | 58 | ±11.5 | (n = 4) | 0.1 | ±0.08 | 44 | ±10.2 | (n = 8) |
| FAA | µg/L | 0.05 | ±0.01 | 57 | ±13.9 | (n = 4) | 0.1 | ±0.01 | 41 | ±7.3 | (n = 8) 3 |
| MTBE | µg/L | 0.6 | ±0.32 | 14 | ±13.6 | (n = 3) | 9.2 | ±4.04 | 10 | ±9.2 | (n = 10) |
| TBA | µg/L | <LOQ | – | (n = 3) | 5.4 | ±2.87 | 8 | ±6.7 | (n = 10) | ||
| Acesulfame | µg/L | 0.7 | ±0.16 | 24 | ±9.5 | (n = 4) | 0.7 | ±0.08 | −5 | ±2.1 | (n = 8) |
| Vinyl chloride | µg/L | <LOQ | – | (n = 4) | 1.6 | ±0.47 | −13 | ±14.1 | (n = 10) | ||
| Parameter | Unit | HFW 1000 | HF-TNF |
|---|---|---|---|
| MWCO | Da | ~1000 | ~200–300 |
| Flux | L/(m2·h) | 10–20 | 22.5 |
| Permeability | L/(m2·h·bar) | 11.0–13.6 | 6.0–7.7 |
| Resistance × 1013 | 1/m | 2.65–3.23 | 4.6–6.3 |
| TMP | bar | 1.1–2.8 | 3.6–4.6 |
| Pressure drop | bar | 0.5–0.7 | 0.35–0.50 |
| Sulphate removal | % | n.d. * | 67 ± 5.5 |
| DOC removal | % | 70–80 | 87 ± 6.4 |
| UV254 removal | % | 80–90 | 89 ± 5.5 |
| Hardness removal | % | <20 | 41 ± 5.2 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jährig, J.; Vredenbregt, L.; Wicke, D.; Miehe, U.; Sperlich, A. Capillary Nanofiltration under Anoxic Conditions as Post-Treatment after Bank Filtration. Water 2018, 10, 1599. https://doi.org/10.3390/w10111599
Jährig J, Vredenbregt L, Wicke D, Miehe U, Sperlich A. Capillary Nanofiltration under Anoxic Conditions as Post-Treatment after Bank Filtration. Water. 2018; 10(11):1599. https://doi.org/10.3390/w10111599
Chicago/Turabian StyleJährig, Jeannette, Leo Vredenbregt, Daniel Wicke, Ulf Miehe, and Alexander Sperlich. 2018. "Capillary Nanofiltration under Anoxic Conditions as Post-Treatment after Bank Filtration" Water 10, no. 11: 1599. https://doi.org/10.3390/w10111599
APA StyleJährig, J., Vredenbregt, L., Wicke, D., Miehe, U., & Sperlich, A. (2018). Capillary Nanofiltration under Anoxic Conditions as Post-Treatment after Bank Filtration. Water, 10(11), 1599. https://doi.org/10.3390/w10111599





