Predicting Taste and Odor Compounds in a Shallow Reservoir Using a Three–Dimensional Hydrodynamic Ecological Model
Abstract
:1. Introduction
2. Materials and Methods
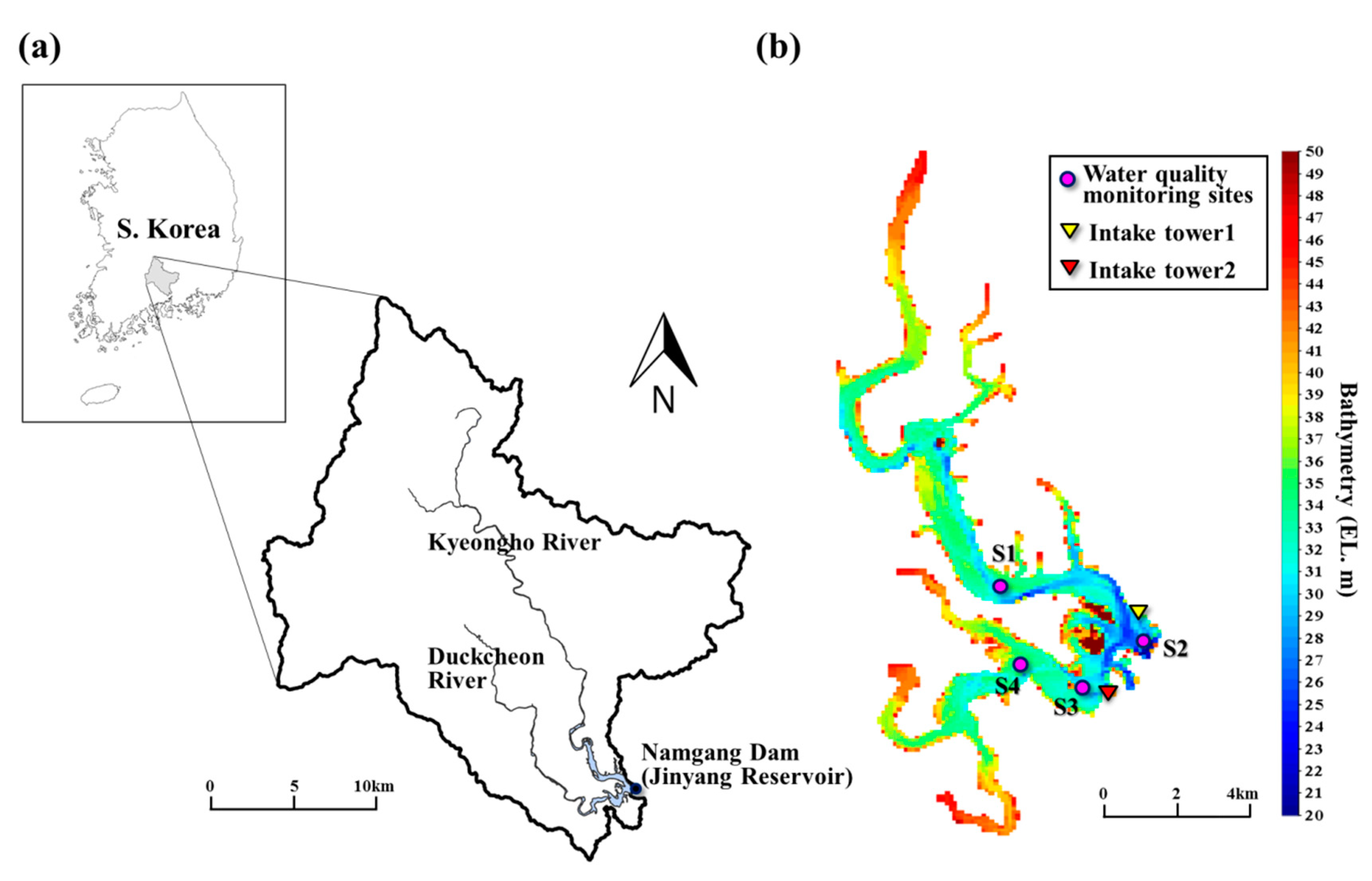
2.1. Description of the Study Site
2.2. Analysis of Water Quality, Phytoplankton and Taste and Odor Compounds
2.3. Model Description
2.4. Configuration and Application of the Model
2.5. Assessment of Model Performance
3. Results of the Field Survey for Algae and Taste and Odor Compounds
3.1. Meteorological and Hydrological Properties
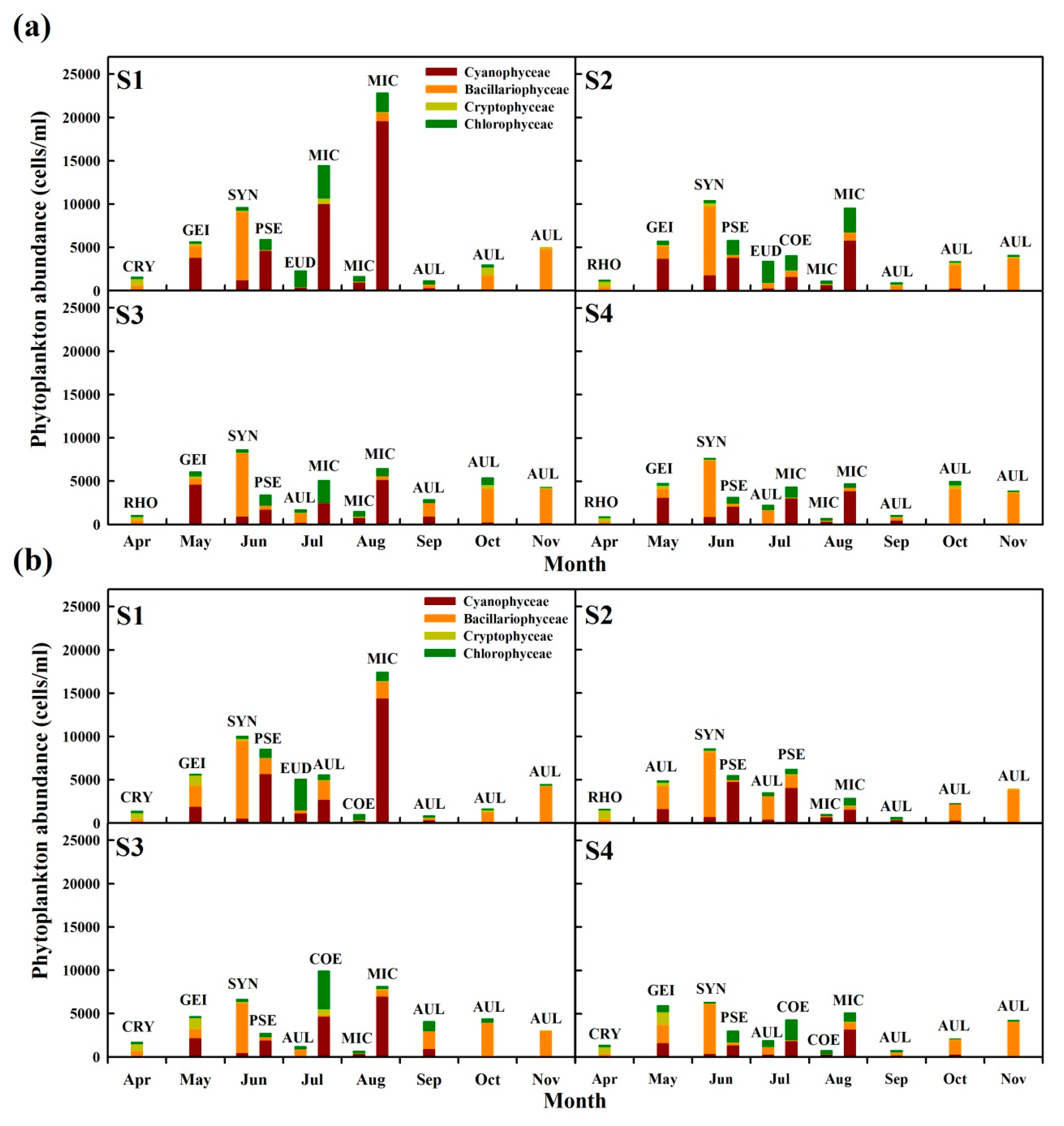
3.2. Characteristics of Algae and Taste and Odor Compound Occurrence
4. Model Prediction Results for Algae and Taste and Odor Compounds
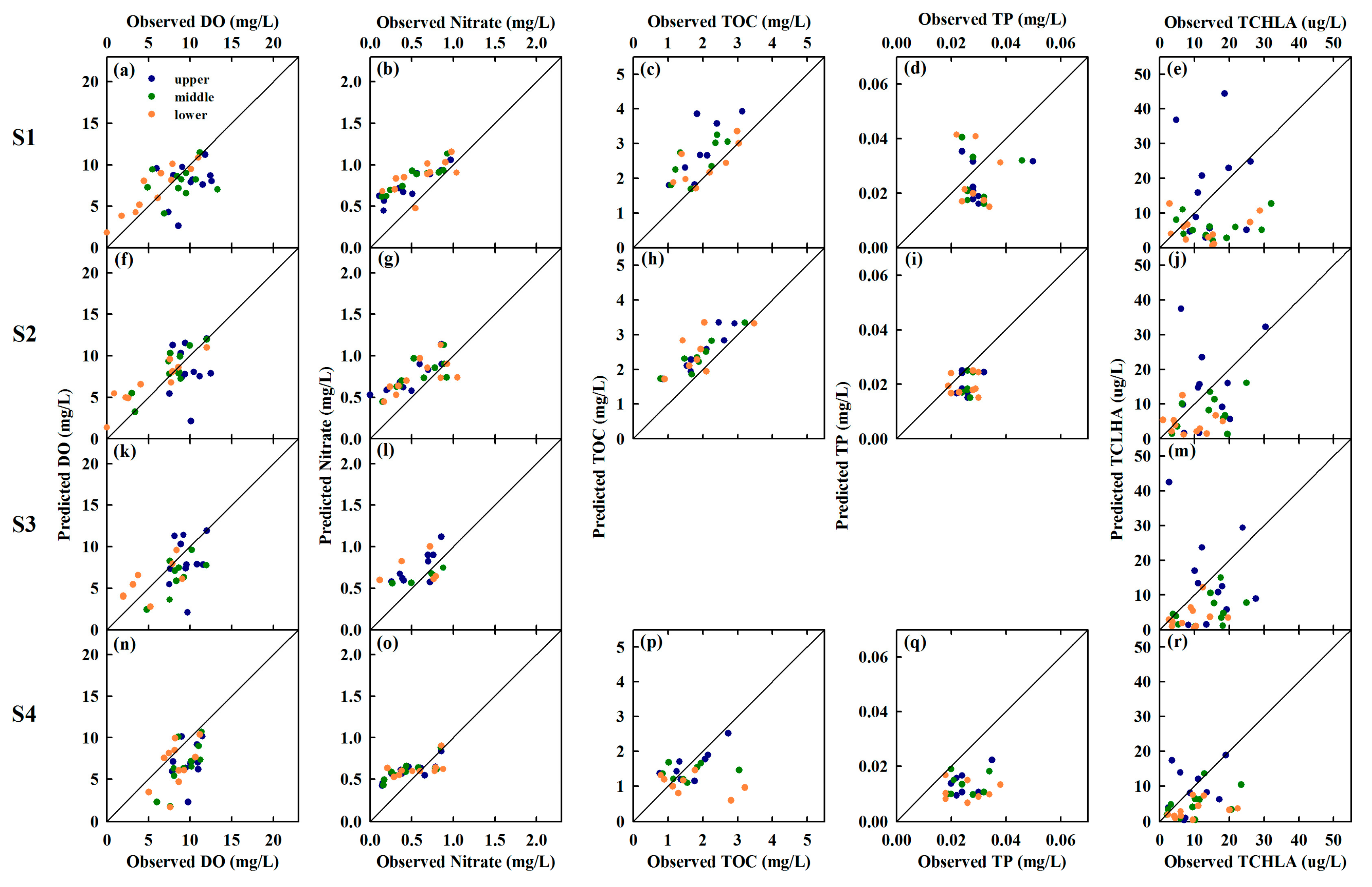
4.1. Model Validation
4.1.1. Water Quality
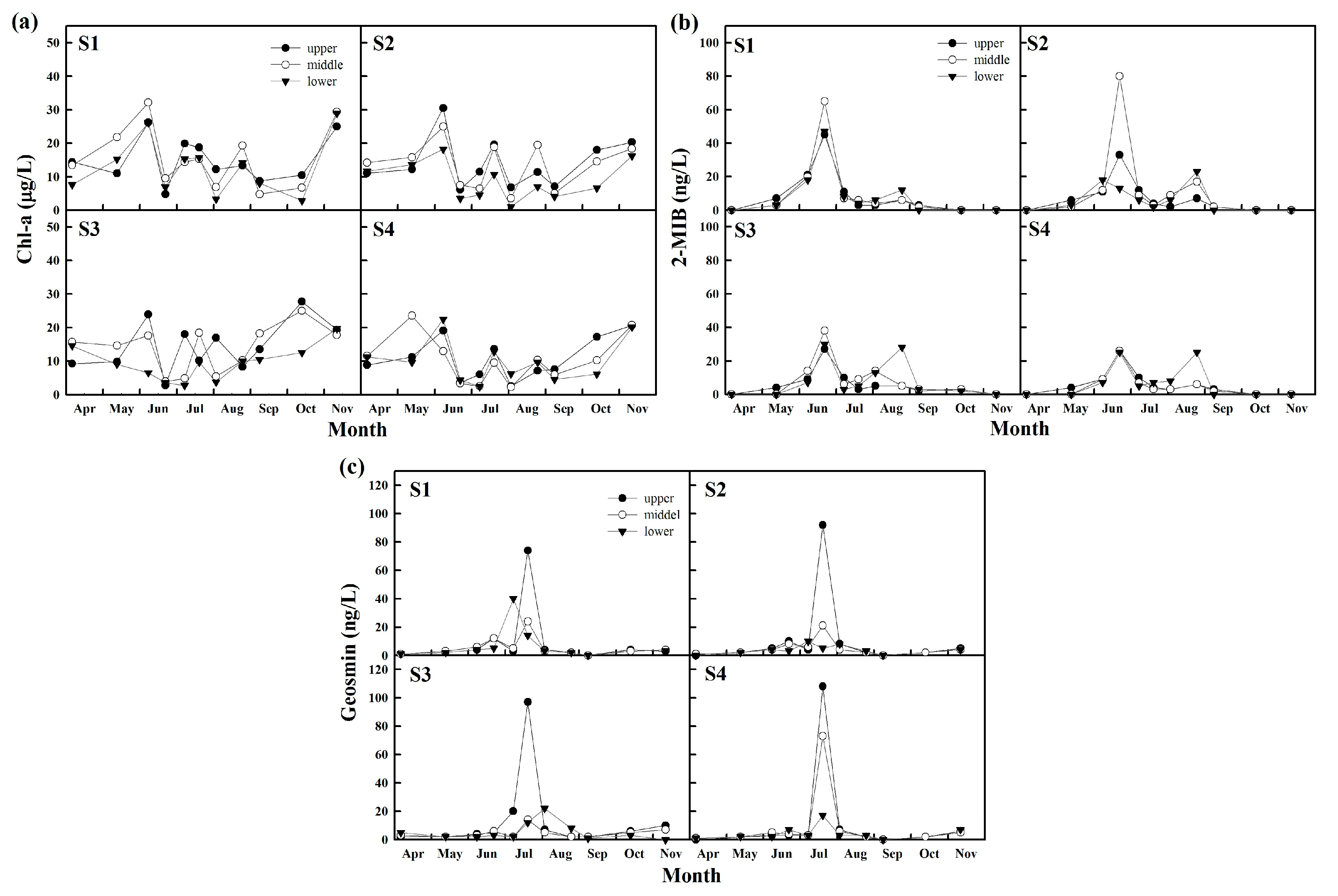
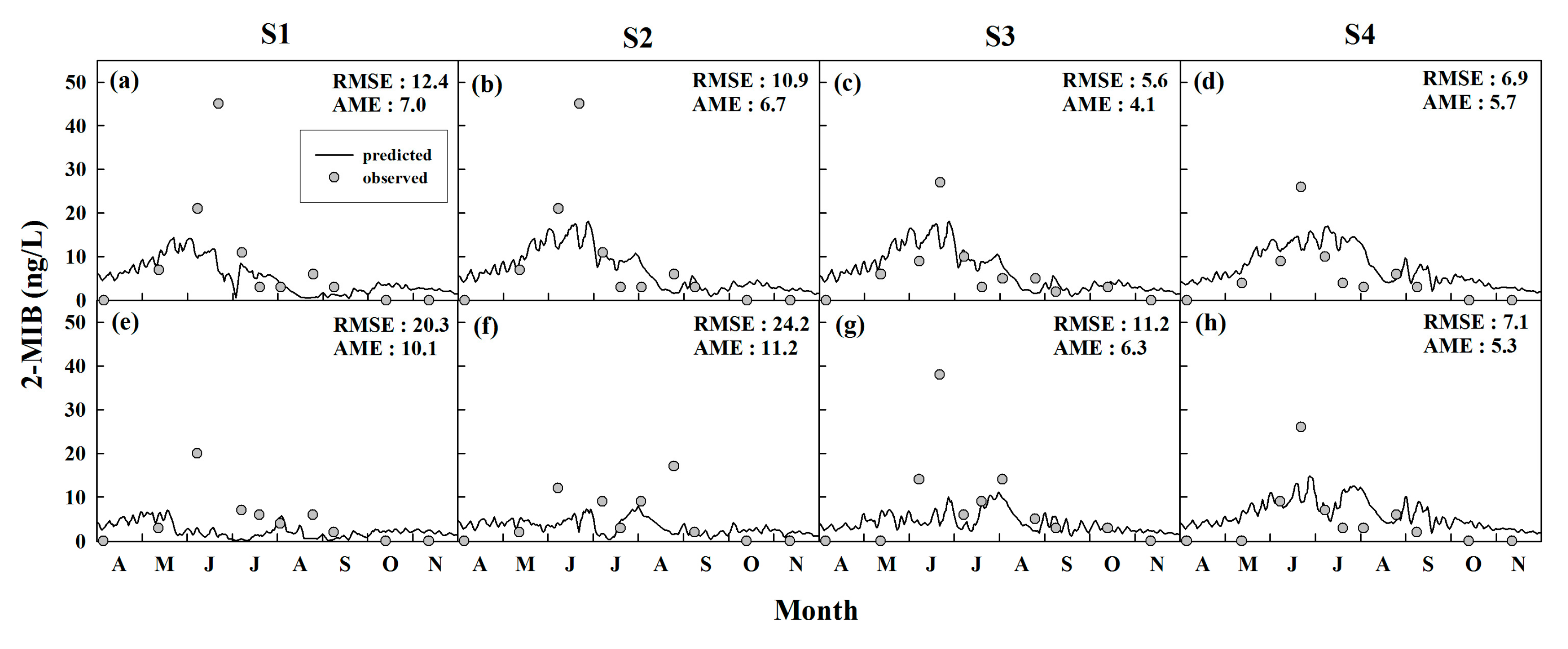
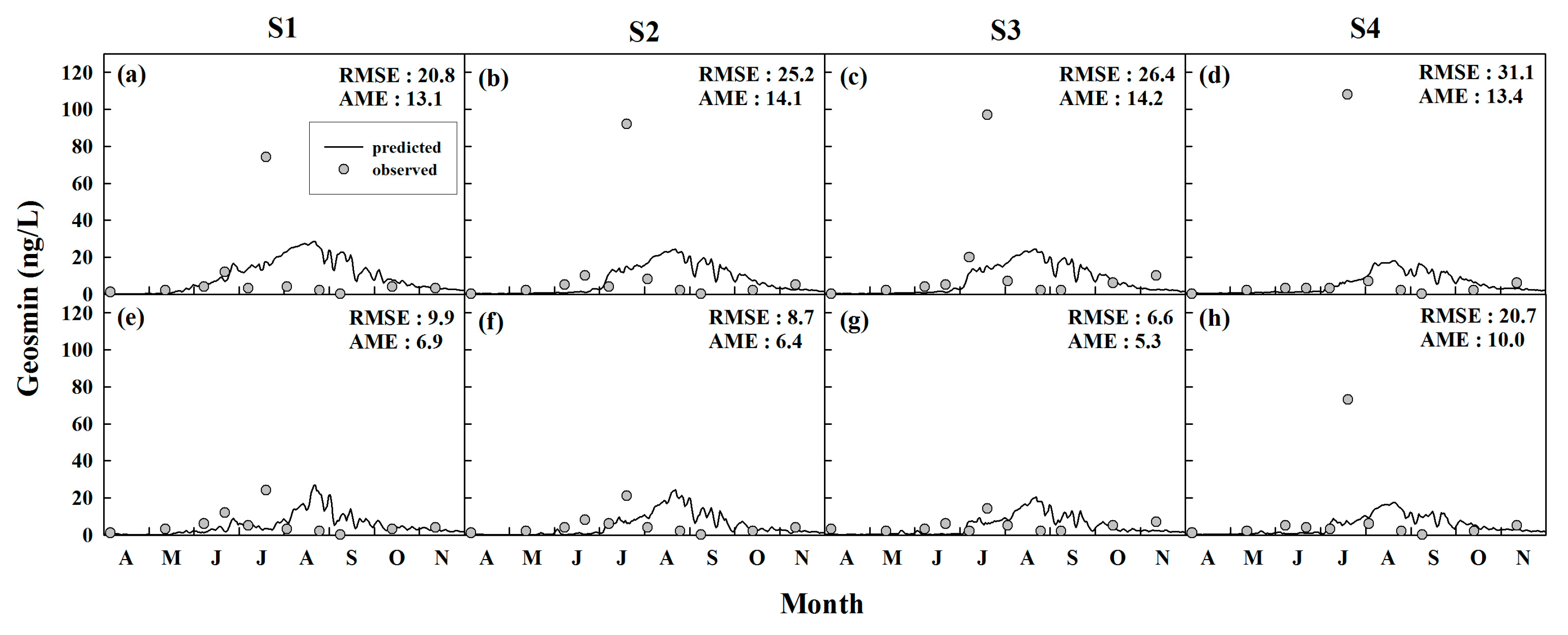
4.1.2. Algal-Derived Taste and Odor Compounds
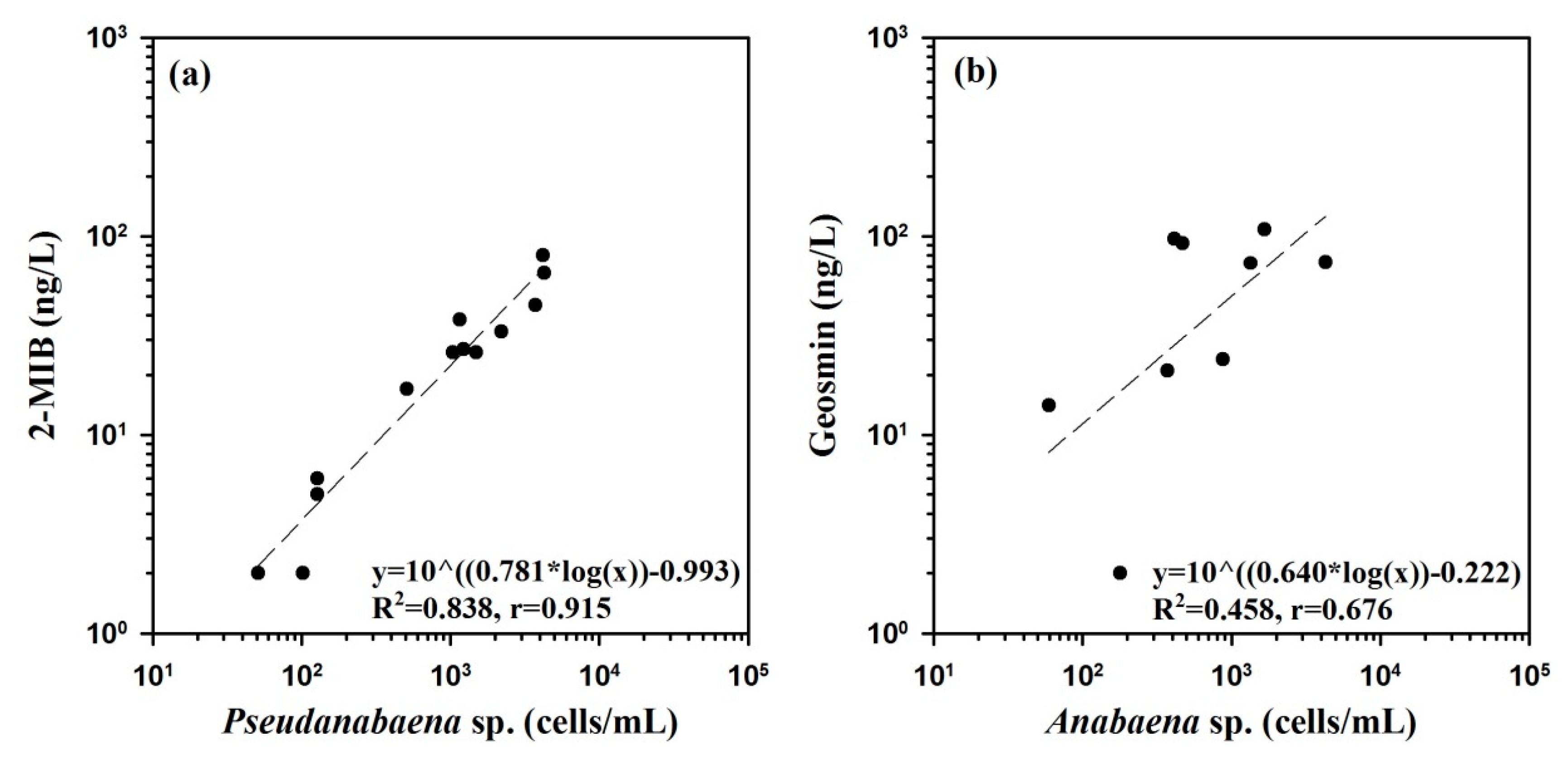
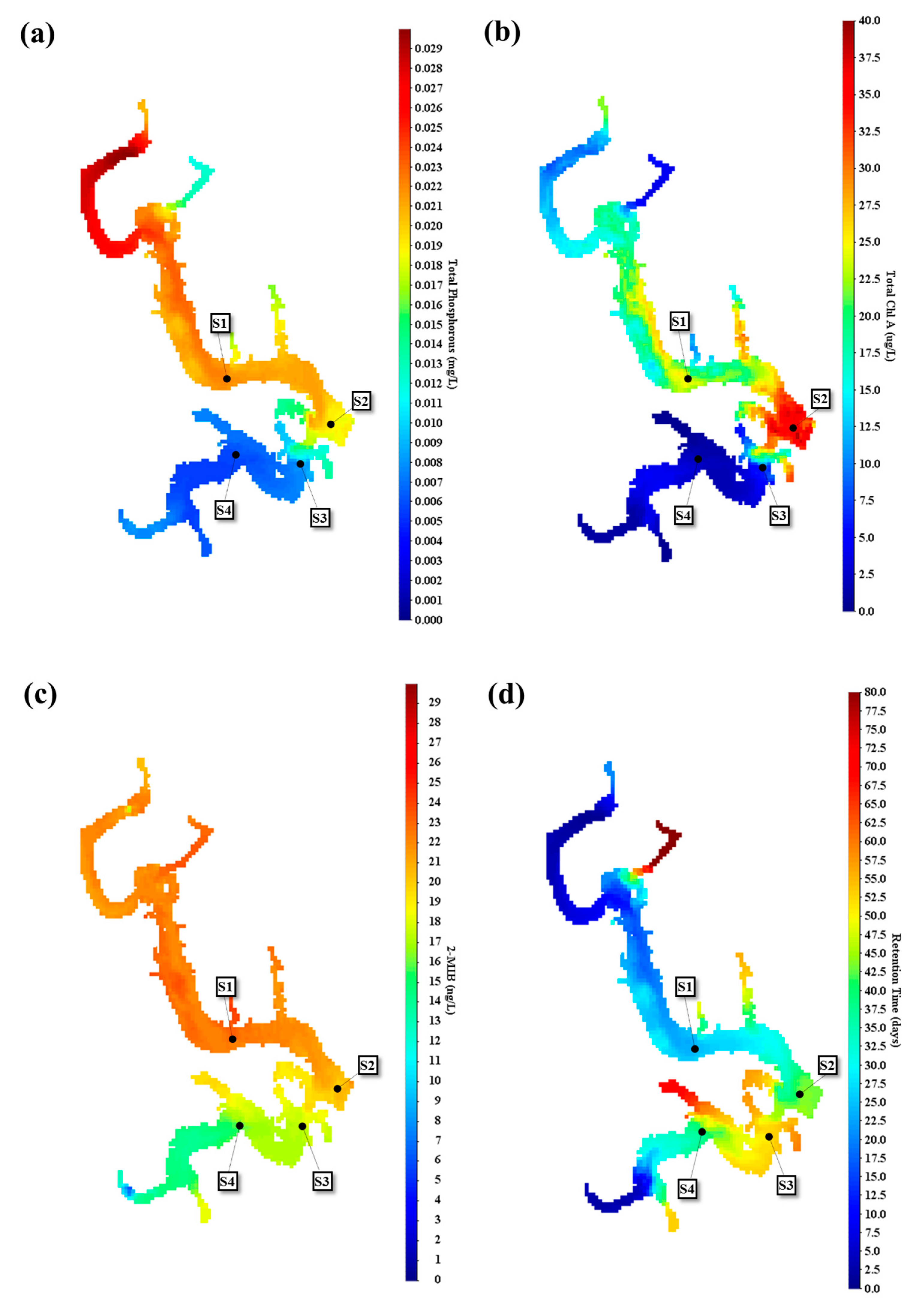
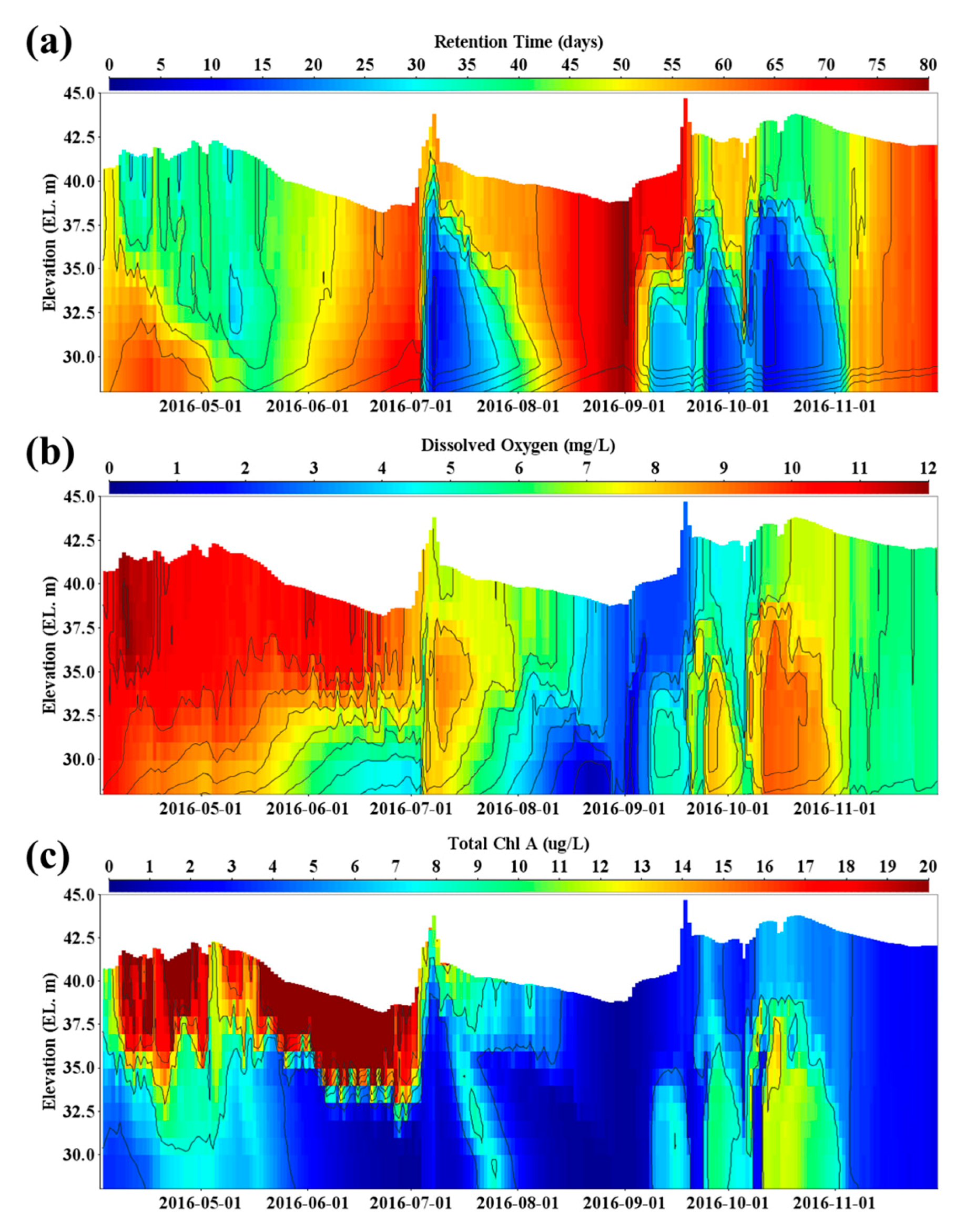
4.2. Effect of Spatiotemporal Physical–Water Quality Characteristics on the Occurrence of Taste and Odor Compounds
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Paerl, H.W.; Fulton, R.S.; Moisander, P.H.; Dyble, J. Harmful freshwater algal blooms, with an emphasis on cyanobacteria. Sci. World J. 2001, 1, 76–113. [Google Scholar] [CrossRef] [PubMed]
- Paerl, H.W.; Otten, T.G. Harmful cyanobacterial blooms: Causes, consequences, and controls. Microb. Ecol. 2013, 65, 995–1010. [Google Scholar] [CrossRef] [PubMed]
- Chorus, I.; Bartram, J. Toxic Cyanobacteria in Water: A Guide to Their Public Health Consequences, Monitoring and Management; World Health Organization: London, UK; New York, NY, USA, 1999; ISBN 0-419-23930-8. [Google Scholar]
- Smith, V.H.; Denlinger, J.S.; de Noyelles, F.; Campbell, S.; Pan, S.; Randtke, S.J.; Blain, G.T.; Strasser, V.A. Managing taste and odor problems in a eutrophic drinking water reservoir. J. Lake Reserv. Manag. 2002, 18, 319–323. [Google Scholar] [CrossRef]
- Watson, S.B. Aquatic taste and odor: A primary signal of drinking-water integrity. J. Toxicol. Environ. Health 2004, 67, 1779–1795. [Google Scholar] [CrossRef] [PubMed]
- Srinivasan, R.; Sorial, G.A. Treatment of taste and odor causing compounds 2-methyl isoborneol and geosmin in drinking water: A critical review. J. Environ. Sci. 2011, 23, 1–13. [Google Scholar] [CrossRef]
- Hsieh, W.H.; Chang, D.W.; Lin, T.F. Occurrence and removal of earthy-musty odorants in two waterworks in Kinmen Island, Taiwan. J. Hazard. Toxic Radioact. Waste 2014, 18, 04014012. [Google Scholar] [CrossRef]
- Korth, W. Determination of Odour Compounds in Surface Water. Ph.D. Thesis, University of Wollongong, Wollongong, Australia, July 1992. [Google Scholar]
- Izaguirre, G.; Taylor, W.D. A Psedanabaena species from Castaic Lake, California, that produce 2-methylisoborneol. Water Res. 1998, 5, 1673–1677. [Google Scholar] [CrossRef]
- Watson, S.B. Cyanobacterial and eukaryotic algal odour compounds: Signal or by-product? A review of their biological activity. Phycologia 2003, 42, 332–350. [Google Scholar] [CrossRef]
- Smith, J.L.; Boyer, G.L.; Zimba, P.V. A review of cyanobacterial odorous and bioactive metabolites: Impacts and management alternatives in aquaculture. Aquaculture 2008, 280, 5–20. [Google Scholar] [CrossRef]
- Jüttner, F.; Watson, S.B. Biochemical and ecological control of geosmin and 2-Methylisoborneol in source waters. Appl. Environ. Microbiol. 2007, 73, 4395–4406. [Google Scholar] [CrossRef] [PubMed]
- Watson, S.B.; Monis, P.; Baker, P.; Giglio, S. Biochemistry and genetics of taste- and odor-producing cyanobacteria. Harmful Algae 2016, 54, 112–127. [Google Scholar] [CrossRef] [PubMed]
- Zhang, K.; Lin, T.F.; Zhang, T.; Li, C.; Gao, N. Characterization of typical taste and odor compounds formed by Microcystis aeruginosa. J. Environ. Sci. 2013, 25, 1539–1548. [Google Scholar] [CrossRef]
- Otten, T.G.; Graham, J.L.; Harris, T.D.; Dreher, T.W. Elucidation of taste-and-odor producing bacteria and toxigenic cyanobacteria by shotgun metagenomics in a Midwestern drinking water supply reservoir. Appl. Environ. Microbiol. 2016, 82, 5410–5420. [Google Scholar] [CrossRef] [PubMed]
- Suurnäki, S.; Gomez-Saez, G.V.; Rantala-Ylinen, A.; Jokela, J.; Fewer, D.P.; Sivonen, K. Identification of geosmin and 2-methylisoborneol in cyanobacteria and molecular detection methods for the producers of these compounds. Water Res. 2015, 68, 56–66. [Google Scholar] [CrossRef] [PubMed]
- Su, M.; Yu, J.; Zhang, J.; Chen, H.; An, W.; Vogt, R.D.; Andersen, T.; Jia, D.; Wang, J.; Yang, M. MIB-producing cyanobacteria (Planktothrix sp.) in a drinking water reservoir: Distribution and odor producing potential. Water Res. 2015, 68, 444–453. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Shao, J.; Xu, Y.; Yan, B.; Li, R. Genetic basis for geosmin production by the water bloom-forming cyanobacterium, Anabaena ucrainica. Water 2015, 7, 175–187. [Google Scholar] [CrossRef]
- Westerhoff, P.M.; Hernandez, M.R.; Baker, L.; Sommerfeld, M. Seasonal occurrence and degradation of 2-methylisoborneol in water supply reservoirs. Water Res. 2005, 39, 4899–4912. [Google Scholar] [CrossRef] [PubMed]
- Dzialowski, A.R.; Smith, V.H.; Huggins, D.G.; de Noyelles, F.; Lim, N.C.; Baker, D.S.; Beury, J.H. Development of predictive models for geosmin-related taste and odor in Kansas, USA, drinking water reservoirs. Water Res. 2009, 43, 2829–2840. [Google Scholar] [CrossRef] [PubMed]
- Bradley, P.M. Environmental factors that influence cyanobacteria and geosmin occurrence in reservoirs. In Current Perspectives in Contaminant Hydrology and Water Resources Sustainability; Journey, C.A., Beaulieu, K.M., Bradley, P.M., Eds.; InTech: Rijeka, Croatia, 2013; pp. 27–54. [Google Scholar]
- Parinet, J.; Rodriguez, M.J.; Sérodes, J.B. Modelling geosmin concentrations in three sources of raw water in Quebec, Canada. Environ. Monit. Assess. 2013, 185, 95–111. [Google Scholar] [CrossRef] [PubMed]
- Bruder, S.R. Prediction of Spatial-Temporal Distribution of Algal Metabolites in Eagle Creek Reservoir, Indianapolis, IN. Master’s Thesis, Indiana University, Indianapolis, October 2012. [Google Scholar]
- Harris, T.D.; Graham, J.L. Predicting cyanobacterial abundance, microcystin, and geosmin in a eutrophic drinking-water reservoir using a 14-year dataset. Lake Reserv. Manag. 2017, 33, 32–48. [Google Scholar] [CrossRef]
- Chung, S.W.; Imberger, J.; Hipsey, M.R.; Lee, H.S. The influence of physical and physiological processes on the spatial heterogeneity of a Microcystis bloom in a stratified reservoir. Ecol. Model. 2014, 289, 133–149. [Google Scholar] [CrossRef]
- Robson, B.J.; Hamilton, D.P. Three-dimensional modelling of a Microcystis bloom event in the Swan River estuary, Western Australia. Ecol. Model. 2004, 174, 203–222. [Google Scholar] [CrossRef]
- Leon, L.F.; Smith, R.E.; Hipsey, M.R.; Bocaniov, S.A.; Higgins, S.N.; Hercky, R.E.; Antenucci, J.P.; Imberger, J.A.; Guildford, S.J. Application of a 3D hydrodynamic–biological model for seasonal and spatial dynamics of water quality and phytoplankton in Lake Erie. J. Great Lakes Res. 2011, 37, 41–53. [Google Scholar] [CrossRef]
- Yajima, H.; Choi, J. Changes in phytoplankton biomass due to diversion of an inflow into the Urayama Reservoir. Ecol. Eng. 2013, 58, 180–191. [Google Scholar] [CrossRef]
- Islam, M.N.; Kitazawa, D.; Park, H.D. Numerical modeling on toxin produced by predominant species of cyanobacteria within the ecosystem of Lake Kasumigaura, Japan. Procedia Environ. Sci. 2012, 13, 166–193. [Google Scholar] [CrossRef]
- Chung, S.W.; Chong, S.A.; Park, H.S. Development and applications of a predictive model for geosmin in North Han River, Korea. Procedia Eng. 2016, 154, 521–528. [Google Scholar] [CrossRef]
- APHA. Standard Methods for the Examination of Water and Wastewater, 20th ed.; American Public Health Association: Washington, DC, USA, 1998. [Google Scholar]
- Korea Meteorological Administration (KMA). Available online: http://www.weather.go.kr/weather/index.jsp (accessed on 1 May 2018).
- Hodges, B.R.; Dallimore, C. Estuary, Lake and Coastal Ocean Model: ELCOM. v2.2 Science Manual; University of Western Australia Technical Publication: Perth, Australia, 2006; pp. 1–32. [Google Scholar]
- Hipsey, M.R.; Romero, J.R.; Antenucci, J.P.; Hamilton, D. Computational Aquatic Ecosystem Dynamics Model: CAEDYM v2.3 Science Manual; University of Western Australia Technical Publication: Perth, Australia, 2006; pp. 36–54. [Google Scholar]
- Nalewajko, C.; Murphy, T.P. Effects of temperature, and availability of nitrogen and phosphorus on the abundance of Anabaena and Microcystis in Lake Biwa, Japan: An experimental approach. Limnology 2001, 2, 45–48. [Google Scholar] [CrossRef]
- Li, Z.; Hobson, P.; An, W.; Burch, M.; House, J.; Yang, M. Earthy odor compounds production and loss in three cyanobacterial cultures. Water Res. 2012, 46, 5165–5173. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cai, Y.; Kong, F. Diversity and dynamics of picocyanobacteria and the bloom-forming cyanobacteria in a large shallow eutrophic lake (lake Chaohu, China). J. Limnol. 2013, 72, e38. [Google Scholar] [CrossRef]
- Mowe, M.; Mitrovic, S.; Lim, R.; Furey, A.; Yeo, D. Tropical cyanobacterial blooms: A review of prevalence, problem taxa, toxins and influencing environmental factors. J. Limnol. 2014, 74. [Google Scholar] [CrossRef]
- Li, X.; Dreher, T.W.; Li, R. An overview of diversity, occurrence, genetics and toxin production of bloom-forming Dolichospermum (Anabaena) species. Harmful Algae 2016, 54, 54–68. [Google Scholar] [CrossRef] [PubMed]
- Lu, X.; Tian, C.; Pei, H.; Hu, W.; Xie, J. Environmental factors influencing cyanobacteria community structure in Dongping Lake, China. J. Environ. Sci. 2013, 25, 2196–2206. [Google Scholar] [CrossRef]
- Kakimoto, M.; Ishikawa, T.; Miyagi, A.; Saito, K.; Miyazaki, M.; Asaedaa, T.; Yamaguchi, M.; Uchimiya, H.; Yamada, M. Culture temperature affects gene expression and metabolic pathways in the 2-methylisoborneol-producing cyanobacterium Pseudanabaena galeata. J. Plant Physiol. 2014, 171, 292–300. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Li, R. Effects of light and temperature on the odor production of 2-Methylisoborneol-producing Pseudanabaena sp. and geosmin-producing Anabaena ucrainica (cyanobacteria). Biochem. Syst. Ecol. 2015, 58, 219–226. [Google Scholar] [CrossRef]
- Zhaoa, H.; Wang, Y.; Yang, L.; Yuan, L.; Peng, D. Relationship between phytoplankton and environmental factors inlandscape water supplemented with reclaimed water. Ecol. Indic. 2015, 58, 113–121. [Google Scholar] [CrossRef]
- Rosińska, J.; Kozak, A.; Dondajewska, R.; Gołdyn, R. Cyanobacteria blooms before and during the restoration process of a shallow urban lake. J. Environ. Manag. 2017, 198, 340–347. [Google Scholar] [CrossRef] [PubMed]
- Chung, S.W.; Hipsey, M.R.; Imberger, J. Modelling the propagation of turbid density inflows into a stratified lake: Daecheong Reservoir, Korea. Environ. Model. Softw. 2009, 24, 1462–1482. [Google Scholar] [CrossRef]
- Vilhena, L.C.; Hillmer, I.; Imberger, J. The role of climate change in the occurrence of algal blooms: Lake Burragorang, Australia. Limnol. Oceanogr. 2010, 55, 1188–1200. [Google Scholar] [CrossRef] [Green Version]
- Linden, L.; Daly, R.I.; Burch, M.D. Suitability of a coupled hydrodynamic water quality model to predict changes in water quality from altered meteorological boundary conditions. Water 2015, 7, 348–361. [Google Scholar] [CrossRef]
- Westernhagen, N.V. Measurements and Modelling of Eutrophication Processes in Lake Rotoiti, New Zealand. Ph.D. Thesis, The University of Waikato, Hamilton, New Zealand, 2010. [Google Scholar]
- Missaghi, S.; Hondzo, M. Evaluation and application of a three-dimensional water quality model in a shallow lake with complex morphometry. Ecol. Model. 2010, 221, 1512–1525. [Google Scholar] [CrossRef]
- K-Water. Development of the Predictive Model and Analysis of Occurrence Characteristics for T&Os in Nakdong River; Korea Water Resources Corporation (K-Water) Technical Report; K-Water: Daejeon, Korea, 2015; pp. 46–54. [Google Scholar]
- Li, Z.; Yu, J.; Yang, M.; Zhang, D.; Burch, M.D.; Han, W. Cyanobacterial population and harmful metabolites dynamics during a bloom in Yanghe Reservoir, North China. Harmful Algae 2010, 9, 481–488. [Google Scholar] [CrossRef] [Green Version]
- Jones, G.J.; Korth, W. In situ production of volatile odor compounds by river and reservoir phytoplankton populations in Australia. Water Sci. Technol. 1995, 31, 145–153. [Google Scholar] [CrossRef]
- Oh, H.S.; Lee, C.S.; Srivastava, A.; Oh, H.M.; Ahn, C.Y. Effects of environmental factors on cyanobacterial production of odorous compounds: Geosmin and 2-methylisoborneol. J. Microbiol. Biotechnol. 2017, 27, 1316–1323. [Google Scholar] [CrossRef] [PubMed]
- Bertone, E.; Burford, M.A.; Hamilton, D.P. Fluorescence probes for real-time remote cyanobacteria monitoring: A review of challenges and opportunities. Water Res. 2018, 141, 152–162. [Google Scholar] [CrossRef] [PubMed]
| Month | Meteorological Data | Hydrological Data | ||||||
|---|---|---|---|---|---|---|---|---|
| Monthly Average Air Temperature (°C) | Rainfall (mm) | Monthly Average Inflow (m3/s) | Monthly Average Outflow (m3/s) | |||||
| 2016 | 1981–2010 (1) | 2016 | 1981–2010 | 2016 | 1976–2015 (2) | 2016 | 1976–2015 | |
| January | −0.2 | −0.1 | 35.4 | 32.9 | 16.7 | 11.9 | 25.9 | 13.6 |
| February | 2.7 | 2.1 | 76.4 | 43.0 | 36.3 | 18.7 | 25.4 | 18.0 |
| March | 7.9 | 6.8 | 121.0 | 72.1 | 62.4 | 28.9 | 63.5 | 29.1 |
| April | 13.9 | 12.8 | 299.7 | 118.2 | 110.5 | 46.2 | 102.6 | 41.7 |
| May | 18.2 | 17.6 | 153.9 | 122.8 | 65.1 | 42.4 | 87.1 | 48.0 |
| June | 21.9 | 21.5 | 113.8 | 213.0 | 20.7 | 75.1 | 33.8 | 78.4 |
| July | 25.6 | 25.1 | 161.0 | 300.0 | 118.6 | 192.0 | 109.7 | 186.3 |
| August | 26.7 | 25.7 | 74.8 | 316.9 | 17.2 | 201.1 | 28.3 | 199.2 |
| September | 21.5 | 21.0 | 407.5 | 184.5 | 106.2 | 131.0 | 75.6 | 133.8 |
| October | 16.0 | 14.5 | 170.1 | 45.0 | 128.2 | 29.0 | 121.7 | 33.2 |
| November | 8.3 | 7.7 | 37.0 | 45.0 | 20.1 | 19.6 | 32.4 | 18.5 |
| December | 3.2 | 2.0 | 84.0 | 19.2 | 34.2 | 15.8 | 28.0 | 17.5 |
| Parameters | Variable | Unit | Assigned Value | Reference |
|---|---|---|---|---|
| Horizontal eddy diffusivity | DX | m2/s | 1 | |
| Bottom drag coefficient | CD | 0.005 | 0.005 1) | |
| Extinction coefficient for NIR (700–2000 nm) | λNIR | /m | 1 | 1 1) |
| Extinction coefficient for PAR (400–700 nm) | λPAR | /m | 0.25 | 0.25 1) |
| Extinction coefficient for UVA (320–400 nm) | λUVA | /m | 1 | 1 1) |
| Extinction coefficient for UVB (300–320 nm) | λUVB | /m | 1 | 1 1) |
| Mean albedo for short-wave radiation | αSW | 0.08 | 0.08 1) |
| Parameter | Description | Unit | Assigned Value | Reference | |||
|---|---|---|---|---|---|---|---|
| CYANO1 | CYANO2 | FDIAT | |||||
| Phytoplankton parameters | μmax | Maximum specific growth rate | /day | 0.5 | 0.5 | 1.2 | 1.1 1)–1.22) |
| Ycc | Average ratio of C to Chl-a | mg-C/mg-Chl.a | 40 | 40 | 20 | 40 1) | |
| ISt | Light saturation for max. production | uE/m2/s | 130 | 130 | 70 | 100 2)–130 1) | |
| Kpa | Half saturation constant for P | mg/L | 0.005 | 0.005 | 0.005 | 0.008 1),2) | |
| Kna | Half saturation constant for N | mg/L | 0.045 | 0.045 | 0.050 | 0.030 2) | |
| KCa | Half saturation constant for C | mg/L | 2 | 2 | 2 | - | |
| Ksa | Half saturation constant for Si | mg/L | 0 | 0 | 0.15 | - | |
| INmin/max | Min/max internal N concentration | mg-N/mg-Chl.a | 2.0/4.0 | 2.0/4.0 | 2.0/4.0 | - | |
| IPmin/max | Min/max internal P concentration | mg-P/mg-Chl.a | 0.1/0.6 | 0.1/0.6 | 0.1/0.6 | - | |
| ICmin/max | Min/max internal C concentration | mg-C/mg-Chl.a | 15/80 | 15/80 | 15/80 | - | |
| vT | Temperature multiplier for growth | - | 1.06 | 1.06 | 1.06 | 1.07 2) | |
| Tsta | Standard temperature | oC | 20.0 | 20.0 | 20.0 | 20.0 1),2) | |
| Topt | Optimum temperature | oC | 24.5 | 26.0 | 18.0 | 22.0 2)–30.0 1) | |
| Tmax | Maximum temperature | oC | 32.0 | 36.0 | 32.0 | 35.0 2)–39.0 1) | |
| kr | Respiration rate coefficient | /day | 0.12 | 0.12 | 0.08 | 0.070 1)–0.1 2) | |
| vR | Temperature multiplier for respiration | - | 1.08 | 1.08 | 1.08 | - | |
| Nutrient parameters | koN2 | Denitrification rate coefficient (20 °C) | /day | 0.01 | 0.03 3)–0.15 4) | ||
| KN2 | Inverted half-saturation constant for effect of oxygen on denitrification | mg/L | 0.5 | 0.44 4) | |||
| vON | Temperature multiplier for denitrification | - | 1.08 | 1.08 3) | |||
| koNH | Nitrification rate coefficient (20 °C) | /day | 0.050 | 0.065 3) –0.25 4) | |||
| KOn | Half-saturation constant for effect of oxygen on nitrification | mg/L | 2 | 0.5 3) –0.75 4) | |||
| vN2 | Temperature multiplier for nitrification | - | 1.08 | 1.08 1) | |||
| PON1max | Rate coefficient of PONL to DONL (20 °C) | /day | 0.010 | 0.003 3) –0.005 4) | |||
| DON1max | Rate coefficient of DONL to NH4 (20 °C) | /day | 0.003 | 0.008 3) –0.08 4) | |||
| POP1max | Rate coefficient of POPL to DOPL (20 °C) | /day | 0.01 | 0.02 3) –0.03 4) | |||
| DOP1max | Rate coefficient of DOPL to PO4 (20 °C) | /day | 0.005 | 0.025 3) –0.25 4) | |||
| Variables | Root-Mean-Square Error (RMSE) | Absolute Mean Error (AME) | ||||||
|---|---|---|---|---|---|---|---|---|
| S1 | S2 | S3 | S4 | S1 | S2 | S3 | S4 | |
| DO (mg/L) | 2.7 | 2.5 | 2.7 | 3.4 | 2.1 | 1.9 | 2.2 | 2.8 |
| NO3-N (mg/L) | 0.321 | 0.274 | 0.243 | 0.213 | 0.279 | 0.237 | 0.209 | 0.182 |
| TOC (mg/L) | 0.8 | 0.8 | - | 0.7 | 0.6 | 0.5 | - | 0.4 |
| TP (mg/L) | 0.016 | 0.009 | - | 0.014 | 0.013 | 0.006 | - | 0.010 |
| Chl-a (μg/L) | 13.4 | 9.8 | 11.7 | 8.5 | 10.6 | 7.4 | 8.6 | 6.3 |
| Parameters | Variable | Unit | Assigned Value |
|---|---|---|---|
| Degradation rate constant | /d | 0.14 | |
| Minimum internal metabolite concentration | 0 | ||
| Internal metabolite concentration during maximum production | 0.00324 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chong, S.; Lee, H.; An, K.-G. Predicting Taste and Odor Compounds in a Shallow Reservoir Using a Three–Dimensional Hydrodynamic Ecological Model. Water 2018, 10, 1396. https://doi.org/10.3390/w10101396
Chong S, Lee H, An K-G. Predicting Taste and Odor Compounds in a Shallow Reservoir Using a Three–Dimensional Hydrodynamic Ecological Model. Water. 2018; 10(10):1396. https://doi.org/10.3390/w10101396
Chicago/Turabian StyleChong, Suna, Heesuk Lee, and Kwang-Guk An. 2018. "Predicting Taste and Odor Compounds in a Shallow Reservoir Using a Three–Dimensional Hydrodynamic Ecological Model" Water 10, no. 10: 1396. https://doi.org/10.3390/w10101396
APA StyleChong, S., Lee, H., & An, K.-G. (2018). Predicting Taste and Odor Compounds in a Shallow Reservoir Using a Three–Dimensional Hydrodynamic Ecological Model. Water, 10(10), 1396. https://doi.org/10.3390/w10101396





