How Does Changing Ice-Out Affect Arctic versus Boreal Lakes? A Comparison Using Two Years with Ice-Out that Differed by More Than Three Weeks
Abstract
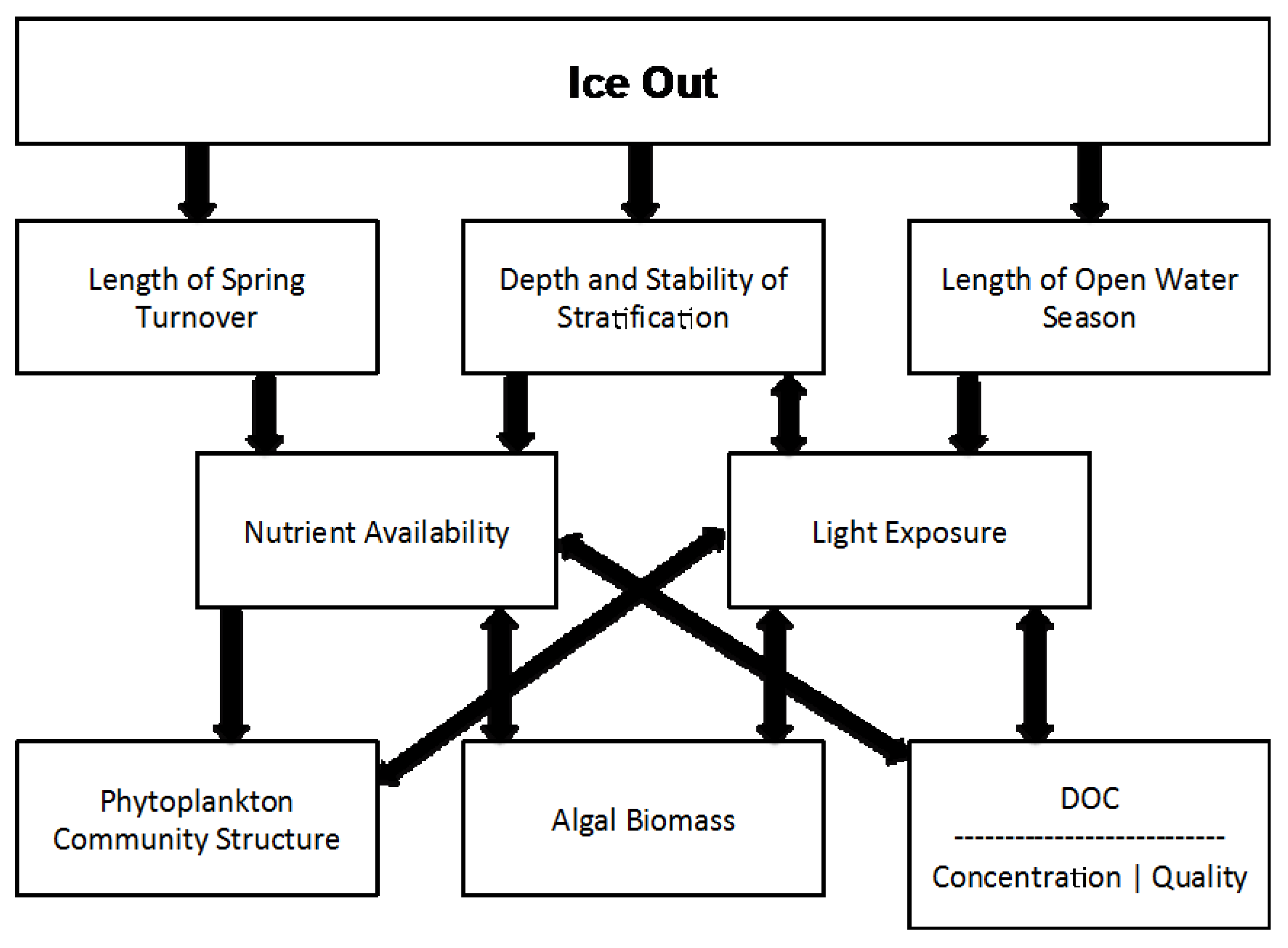
1. Introduction
2. Materials and Methods
2.1. Study Design
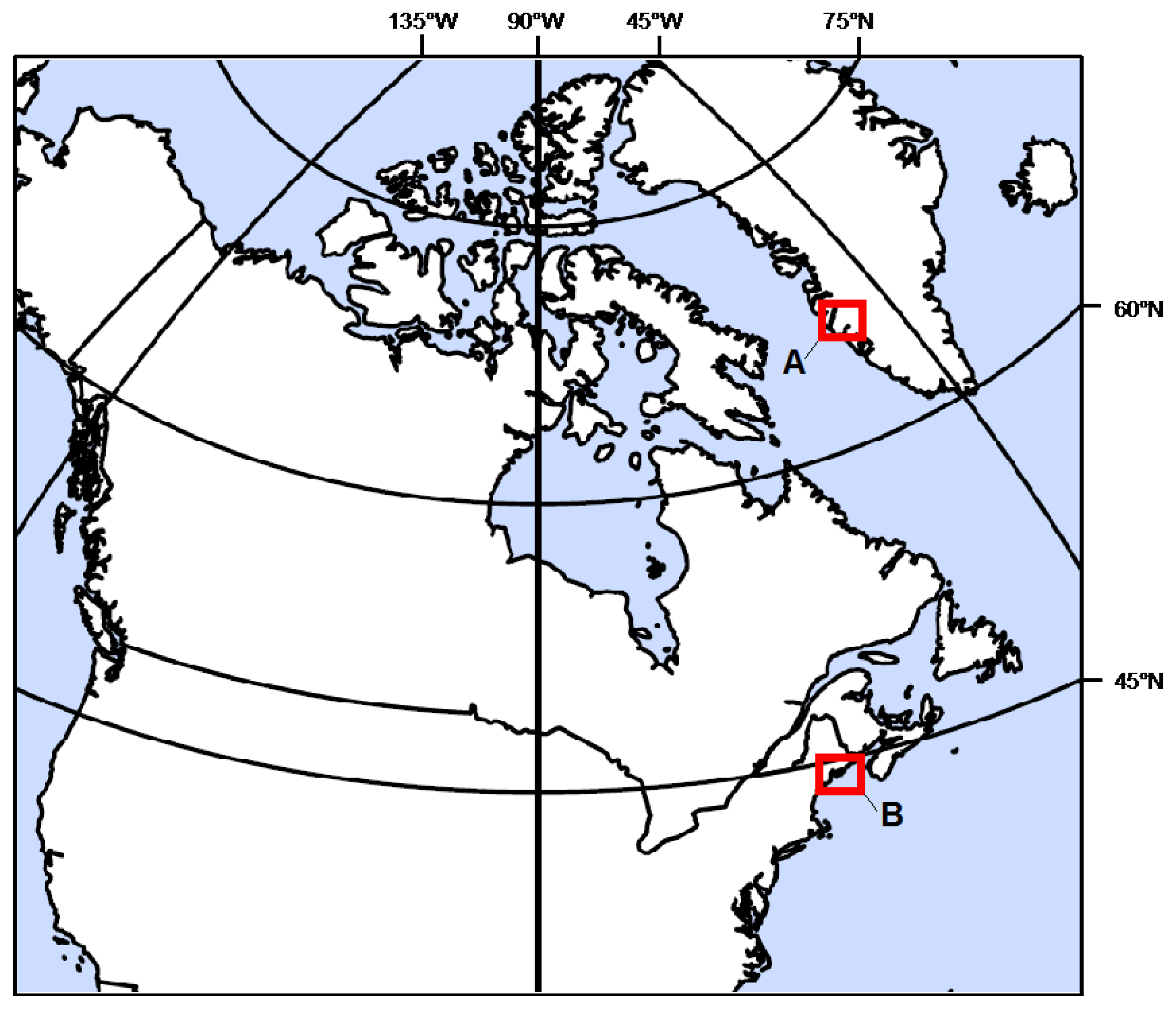
2.2. Site Description
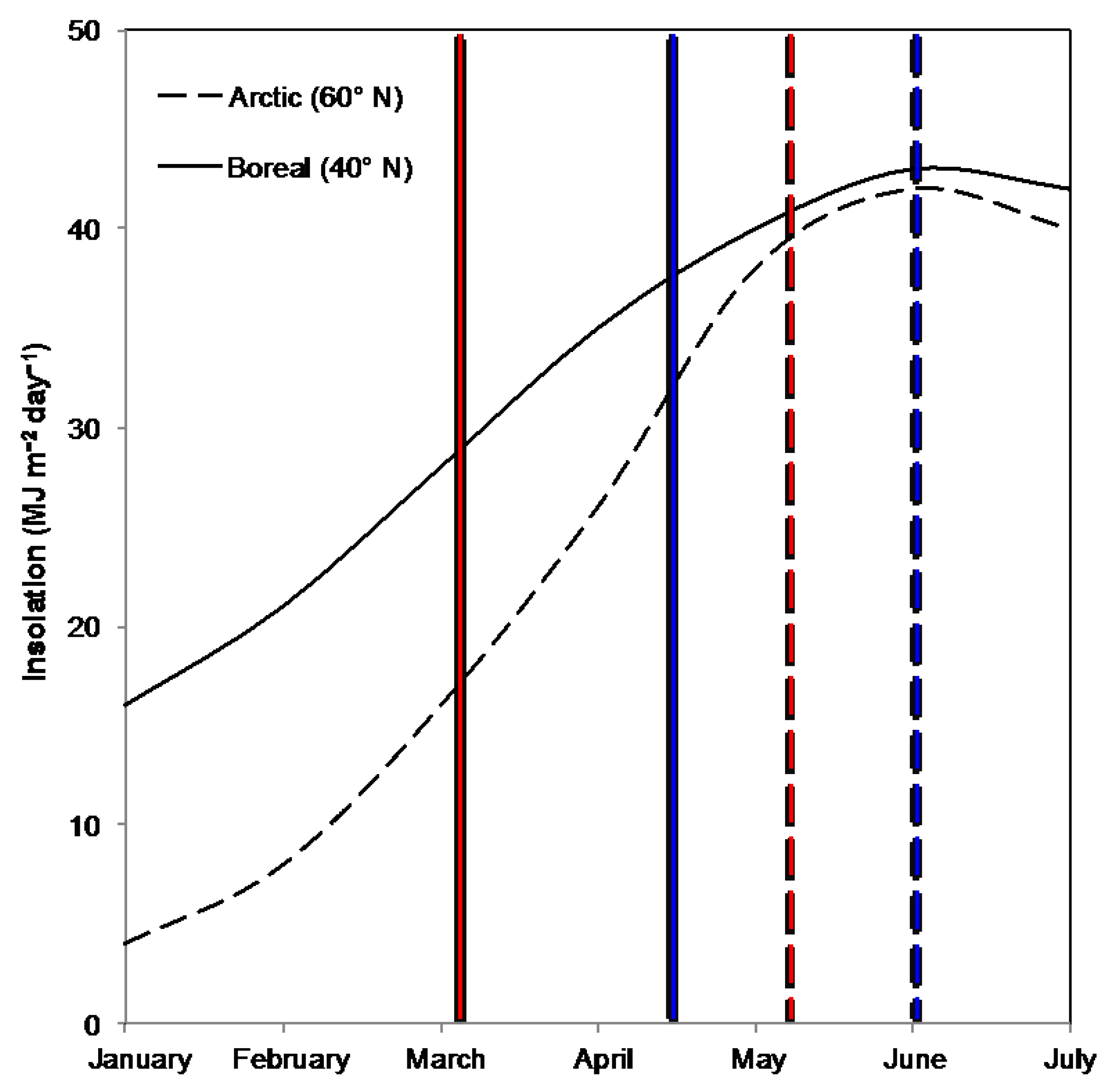
2.3. Climate Variables
2.4. Comparative Lake Sampling
2.4.1. Physical
2.4.2. Chemical
2.4.3. Biological
2.5. Data Analysis
3. Results
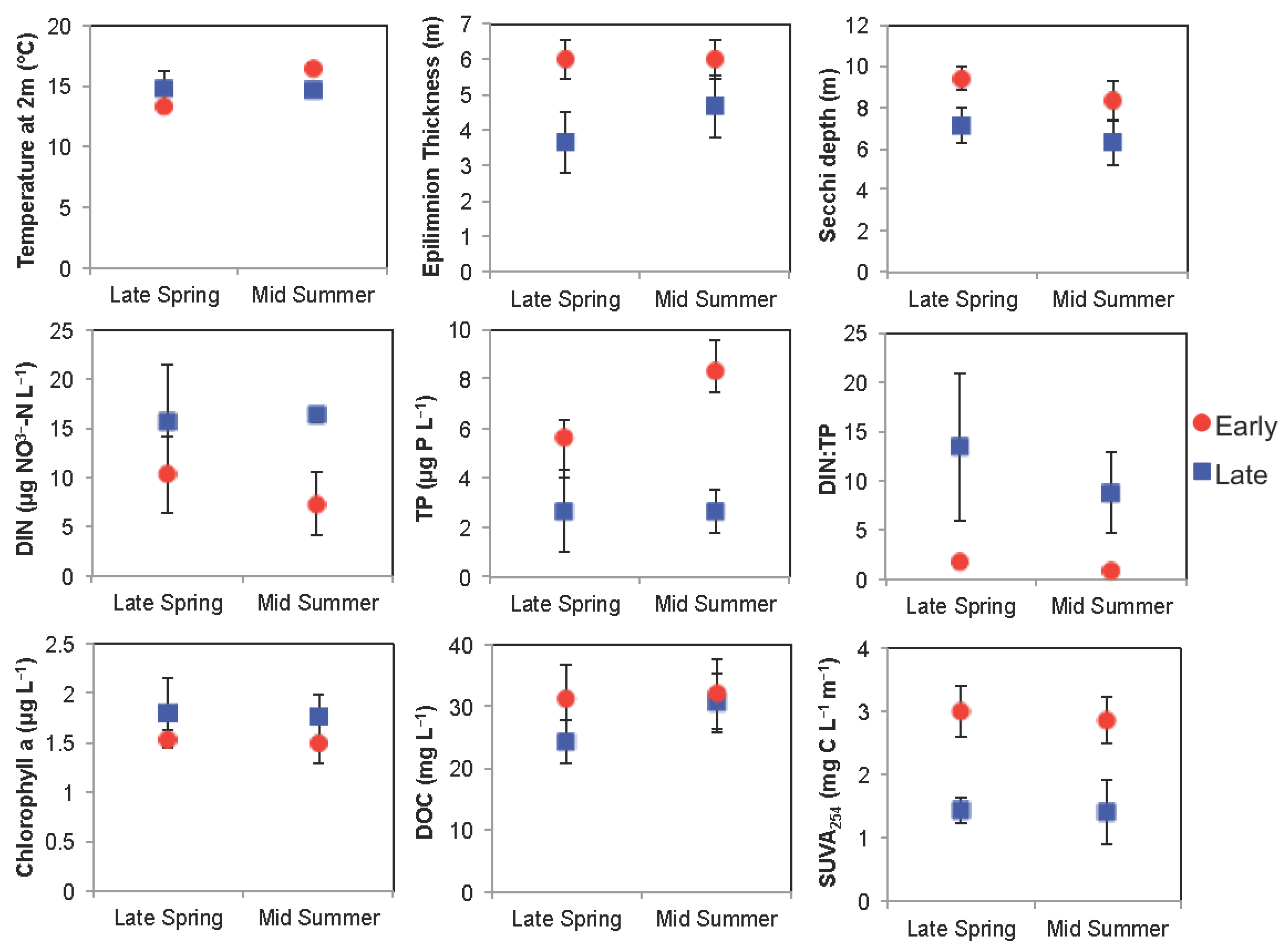
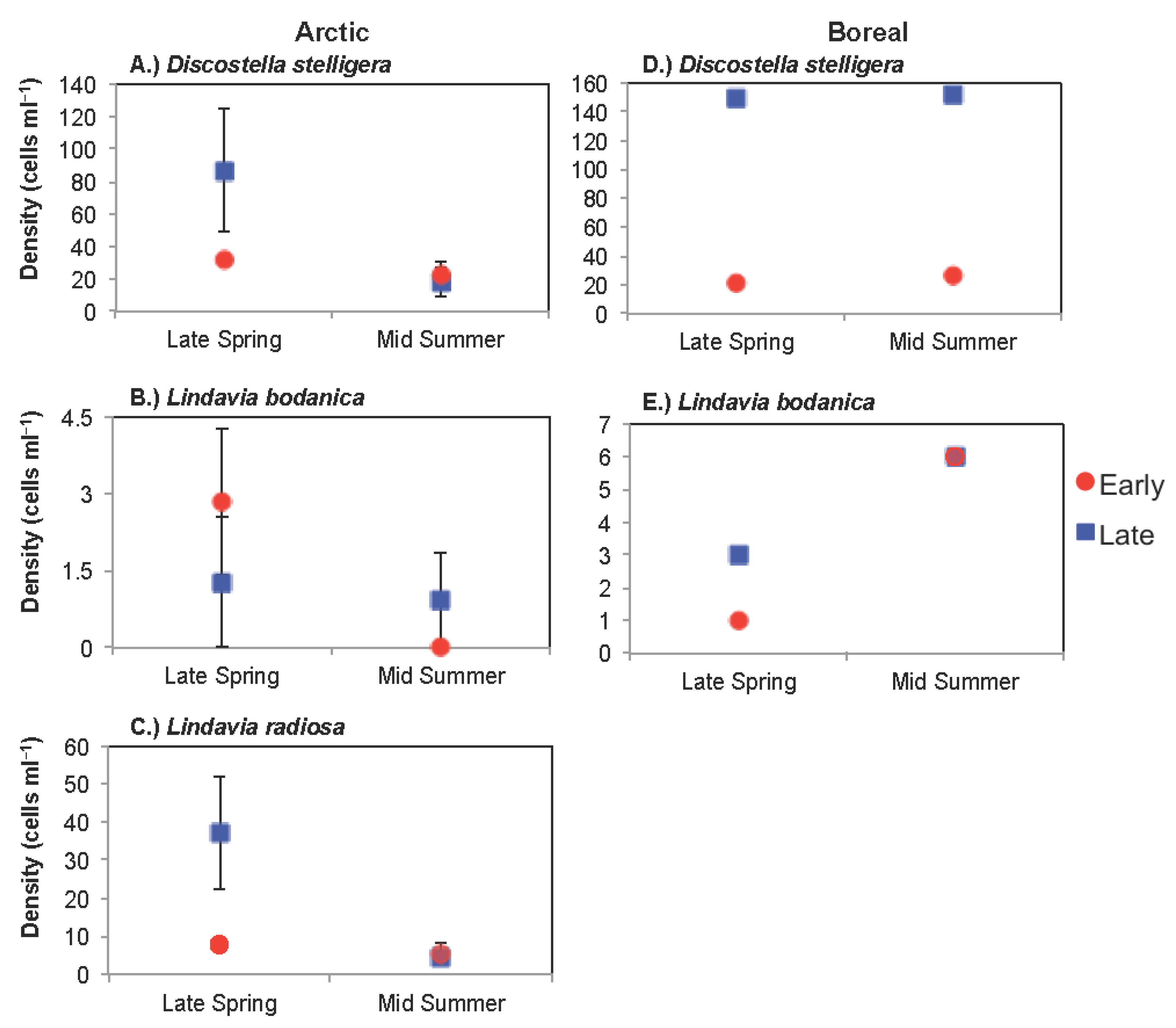
3.1. Arctic Region
3.1.1. Comparison of Spring Response across Early and Late Ice-out Years
3.1.2. Comparison of Summer Response across Early and Late Ice-Out Years
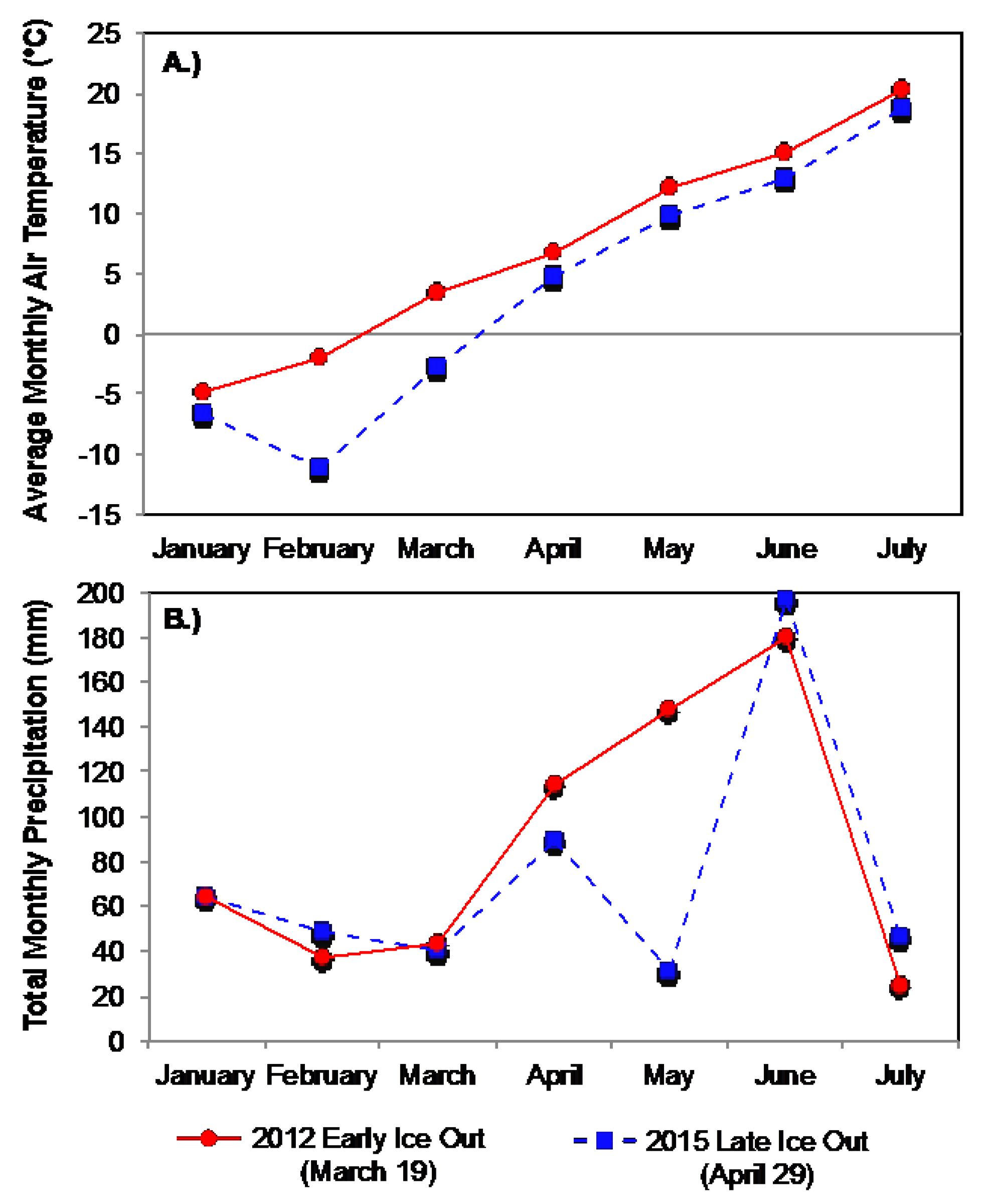
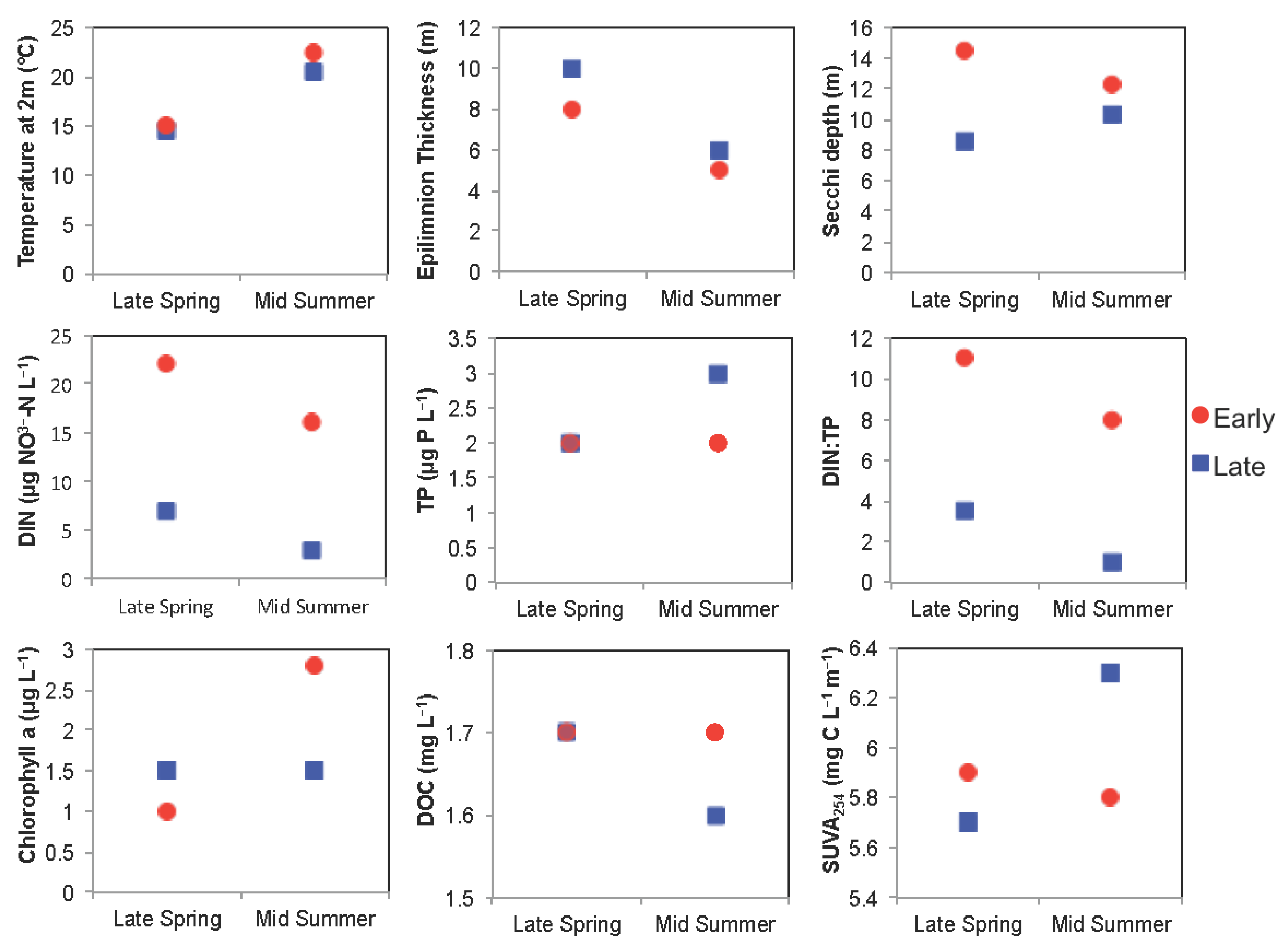
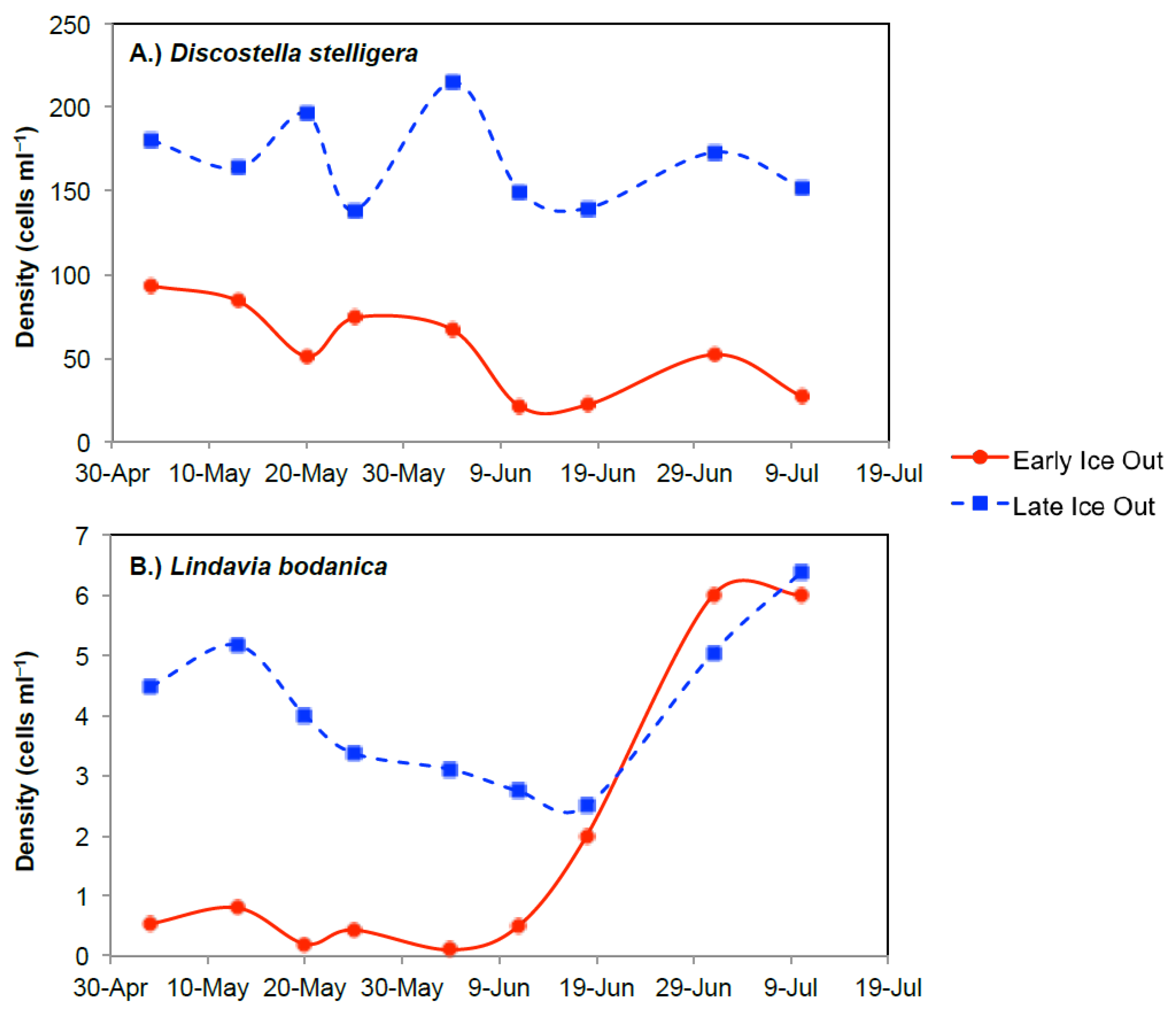
3.2. Boreal Region
3.2.1. Comparison of Spring Response across Early and Late Ice-Out Years
3.2.2. Comparison of Summer across Early and Late Ice-Out Years
4. Discussion
Acknowledgments
Author Contributions
Conflicts of Interest
Appendix A
| Year | Arctic (Greenland) | Boreal (Jordan Pond) |
|---|---|---|
| 2010 | 24 May | 22 March |
| 2011 | 14 June | 16 April |
| 2012 | 3 June | 19 March |
| 2013 | 9 June | 4 April |
| 2014 | 13 June | 14 April |
| 2015 | 17 June | 29 April |
| 2016 | 17 May | 17 March |
References
- Kuusisto, E. An analysis of the longest ice observation series made on Finish lakes. Aqua Fenn 1987, 17, 123–132. [Google Scholar]
- Schindler, D.W.; Beaty, K.G.; Fee, E.J.; Cruikshank, D.R.; DeBruyn, E.R.; Findlay, D.L.; Linsey, G.A.; Shearer, J.A.; Stainton, M.P.; Turner, M.A. Effects of climatic warming on lakes of the central boreal forest. Science 1990, 250, 967–970. [Google Scholar] [CrossRef] [PubMed]
- Livingstone, D.M. Large-scale climatic forcing detected in historical observations of lake ice break-up. Verh. Int. Ver. Theor. Angew. Limnol. 2000, 27, 2775–2783. [Google Scholar] [CrossRef]
- Magnuson, J.J.; Robertson, D.M.; Benson, B.J.; Wynne, R.H.; Livingstone, D.M.; Arai, T.; Assel, R.A.; Barry, R.G.; Card, V.; Kuusisto, E.; et al. Historical trends in lake and river ice cover in the Northern Hemisphere. Science 2000, 289, 1743–1746. [Google Scholar] [CrossRef] [PubMed]
- Futter, M.N. Pattern and trends in Southern Ontario lake ice phenology. Environ. Monit. Assess. 2003, 88, 431–444. [Google Scholar] [CrossRef] [PubMed]
- Sporka, F.; Livingstone, D.M.; Stuchlik, E.; Turek, J.; Galas, J. Water temperatures and ice cover in lakes of the Tatra Mountains. Biologia 2006, 61, S77–S90. [Google Scholar] [CrossRef]
- Adrian, R.; O’Reilly, C.M.; Zagarese, H.; Baines, S.B.; Hessen, D.O.; Keller, W.; Livingstone, D.M.; Sommaruga, R.; Straile, D.; Van Donk, E.; et al. Lakes as sentinels of change. Limnol. Oceanogr. 2009, 54, 2283–2297. [Google Scholar] [CrossRef] [PubMed]
- Weyhenmeyer, G.A.; Meili, M.; Livingstone, D.M. Systematic differences in the trend towards earlier ice-out on Swedish lakes along a latitudinal temperature gradient. Verh. Int. Ver. Theor. Angew. Limnol. 2005, 29, 257–260. [Google Scholar] [CrossRef]
- Jensen, O.P.; Benson, B.J.; Magnuson, J.J.; Card, V.M.; Futter, M.N.; Soranno, P.A.; Stewart, K.M. Spatial analysis of ice phenology trends across the Laurentian Great Lakes region during a recent warming period. Limnol. Oceanogr. 2007, 52, 2013–2026. [Google Scholar] [CrossRef]
- Beier, C.M.; Stella, J.C.; Dovciak, M.; McNulty, S.A. Local climatic drivers of changes in phenology at a boreal-temperate ecotone in eastern North America. Clim. Chang. 2012, 115, 399–417. [Google Scholar] [CrossRef]
- Benson, B.J.; Magnuson, J.J.; Jensen, O.P.; Card, V.M.; Hodgkins, G.; Korhonen, J.; Livingstone, D.M.; Stewart, K.M.; Weyhenmeyer, G.A.; Granin, N.G. Extreme events, trends and variability in Northern Hemisphere lake-ice phenology (1855–2005). Clim. Chang. 2012, 112, 299–323. [Google Scholar] [CrossRef]
- Smejkalova, T.; Edwards, M.; Jadunandan, D. Arctic lakes show strong decadal trend in earlier spring ice-out. Sci. Rep. 2016, 6, 38449. [Google Scholar] [CrossRef] [PubMed]
- Rühland, K.; Smol, J.P. Diatom shifts as evidence for recent Subarctic warming in a remote tundra lake, NWT, Canada. Paleogeogr. Paleoclimatol. Paleoecol. 2005, 226, 1–16. [Google Scholar] [CrossRef]
- Rühland, K.; Paterson, A.M.; Smol, J.P. Hemispheric-scale patterns of climate-related shifts in planktonic diatoms from North American and European lakes. Glob. Chang. Biol. 2008, 14, 2740–2754. [Google Scholar] [CrossRef]
- Rühland, K.; Paterson, A.M.; Smol, J.P. Lake diatom response to warming: Reviewing the evidence. J. Paleolimnol. 2015, 54, 1–35. [Google Scholar] [CrossRef]
- Smol, J.P.; Douglas, M.S. Crossing the final ecological threshold in high Arctic ponds. Proc. Natl. Acad. Sci. USA 2007, 104, 12395–12397. [Google Scholar] [CrossRef] [PubMed]
- Wiltse, B.; Paterson, A.M.; Findlay, D.L.; Cumming, B.F. Seasonal and decadal patterns in discostella (Bacillariophyceae) species from bi-weekly records of two boreal lakes (Experimental Lakes Area, Ontario Canada). J. Phycol. 2016, 52, 817–826. [Google Scholar] [CrossRef] [PubMed]
- Boeff, K.A.; Strock, K.E.; Saros, J.E. Evaluating planktonic diatom response to climate change across three lakes with differing morphometry. J. Paleolimnol. 2016, 56, 33–47. [Google Scholar] [CrossRef]
- Kienel, U.; Kirillin, G.; Brademann, B.; Plessen, B.; Lampe, R.; Brauer, A. Effects of spring warming and mixing duration on diatom deposition in deep Tiefer See, NE Germany. J. Paleolimnol. 2017, 57, 37–49. [Google Scholar] [CrossRef]
- Meis, S.; Thackeray, S.J.; Jones, I.D. Effects of recent climate change on phytoplanton phenology in a temperate lake. Freshwater Biol. 2009, 54, 1888–1898. [Google Scholar] [CrossRef]
- Peltomaa, E.; Ojala, A.; Holopainen, A.L.; Salonen, K. Changes in phytoplankton in a boreal lake during a 14-year period. Boreal Environ. Res. 2013, 18, 387–400. [Google Scholar]
- McBean, G. Arctic Climate Impact Assessment—Scientific Report; Cambridge University Press: Cambridge, UK, 2005. [Google Scholar]
- Screen, J.A.; Simmonds, I. The central role of diminishing sea ice in recent Arctic temperature amplification. Nature 2010, 464, 1334–1337. [Google Scholar] [CrossRef] [PubMed]
- Jeong, J.H.; Kug, J.S.; Linderholm, H.W.; Chen, D.; Kim, B.M.; Jun, S.Y. Intensified Arctic warming under greenhouse warming by vegetation-atmosphere-sea ice interaction. Environ. Res. Lett. 2014, 9, 94007. [Google Scholar] [CrossRef]
- Peeters, F.; Straile, D.; Lorke, A.; Livingstone, D.M. Earlier onset of the spring phytoplankton bloom in lakes of the temperate zone in a warmer climate. Glob. Chang. Biol. 2007, 13, 1898–1909. [Google Scholar] [CrossRef]
- Livingstone, D.M. A change of climate provokes a change of paradigm: Taking leave of two tacit assumptions about physical lake forcing. Int. Rev. Hydrobiol. 2008, 93, 404–414. [Google Scholar] [CrossRef]
- Weyhenmeyer, G.A.; Meili, M.; Livingstone, D.M. Nonlinear temperature response of lake ice breakup. Geophys. Res. Lett. 2004, 31, L07203. [Google Scholar] [CrossRef]
- DeStasio, B.T.; Hill, D.K.; Kelinhans, J.M.; Nibbelink, N.P.; Magnuson, J.J. Potential effects of global climate change on small north-temperate lakes: Physics, fish and plankton. Limnol. Oceanogr. 1996, 41, 1136–1149. [Google Scholar] [CrossRef]
- Peeters, F.; Livingstone, D.M.; Goudsmit, G.H.; Kipfer, R.; Forster, R. Modeling 50 years of historical temperature profiles in a large central European lake. Limnol. Oceanogr. 2002, 47, 186–197. [Google Scholar] [CrossRef]
- Douglas, M.S.V.; Smol, J.P.; Pienitz, R.; Hamilton, P. Algal indicators of environmental change in Arctic and Antarctic lakes and ponds. In Long-Term Environmental Change in Arctic and Antarctic Lakes; Pienitz, R., Douglas, M.S.V., Smol, J.P., Eds.; Springer: Dordrecht, The Netherlands, 2004; pp. 117–157. [Google Scholar]
- Preston, D.L.; Caine, N.; McKnight, D.M.; Williams, M.W.; Hell, K.; Miller, M.P.; Hart, S.J.; Johnson, P.T.J. Climate regulates alpine lake ice cover phenology and aquatic ecosystem structure. Geophys. Res. Lett. 2016, 43, 5353–5360. [Google Scholar] [CrossRef]
- Beyene, M.T.; Jain, S. Wintertime weather-climate variability and its links to early spring ice-out in Maine lakes. Limnol. Oceanogr. 2015, 60, 1890–1905. [Google Scholar] [CrossRef]
- Hampton, S.E.; Galloway, A.W.E.; Powers, S.M.; Ozersky, T.; Woo, K.H.; Batt, R.D.; Labou, S.G.; O’Reilly, C.M.; Sharma, S.; Lotting, N.R.; et al. Ecology under lake ice. Ecol. Lett. 2017, 20, 98–111. [Google Scholar] [CrossRef] [PubMed]
- De Senerpont Domis, L.N.; Elser, J.J.; Gsell, A.S.; Huszar, V.L.M.; Ibelings, B.W.; Jeppesen, E.; Kosten, S.; Mooij, W.M.; Roland, F.; Sommer, U.; et al. Plankton dynamics under different climatic conditions in space and time. Freshwater Biol. 2013, 58, 463–482. [Google Scholar] [CrossRef]
- Powers, S.M.; Labou, S.G.; Baulch, H.M.; Hunt, R.J.; Lottig, N.R.; Hampton, S.E.; Stanley, E.H. Ice duration drives winter nitrate accumulation in north temperate lakes. Limnol. Oceanogr. Lett. 2017, 2, 177–186. [Google Scholar] [CrossRef]
- Cory, R.M.; Ward, C.P.; Crump, B.C.; Kling, G.W. Sunlight controls water column processing of carbon in Arctic freshwaters. Science 2014, 345, 925–928. [Google Scholar] [CrossRef] [PubMed]
- Levine, M.A.; Whalen, S.C. Nutrient limitation of phytoplankton production in Alaskan Arctic foothill lakes. Hydrobiologia 2001, 455, 189–201. [Google Scholar] [CrossRef]
- Nielsen, A.B. Present Conditions in Greenland and the Kangerlussuaq Area, Working Report. Geological Survey of Denmark and Greenland. 2010. Available online: http://www.posiva.fi/files/1244/WR_2010-07web.pdf (assessed on 30 October 2017).
- Hasholt, B.; Anderson, N.J. On the formation and stability of oligosaline lakes in the arid, low arctic area of Kangerlussuaq, south-west Greenland. In Proceedings of the Northern Research Basins-14th International Symposium and Workshop, Greenland, Denmark, 25–29 August 2003; C.A. Reitzel: Kobenhavn, Denmark, 2003; pp. 41–48. [Google Scholar]
- Saros, J.E.; Northington, R.M.; Anderson, D.S.; Anderson, N.J. A whole-lake experiment confirms a small centric diatom species as an indicator of changing lake thermal structure. Limnol. Oceanogr. Lett. 2016, 1, 27–35. [Google Scholar] [CrossRef]
- Brodersen, K.P.; Anderson, N.J. Subfossil insect remains (Chironomidae) and lake-water temperature inference in the Sisimiut-Kangerlussuaq region, southern West Greenland. Geol. Greenland Surv. Bull. 2000, 186, 78–82. [Google Scholar]
- Anderson, N.J.; Harriman, R.; Ryves, D.B.; Patrick, S.T. Dominant factors controlling variability in the ionic composition of West Greenland lakes. Arct. Antarct. Alp. Res. 2001, 33, 418–425. [Google Scholar] [CrossRef]
- Gilman, R.A.; Chapman, C.A.; Lowell, T.V.; Borns, H.W., Jr. The Geology of Mount Desert Island: A Visitors Guide to the Geology of Acadia National Park; Maine Geol. Surv., Department of Conservation: Augusta, GA, USA, 1988; pp. 1–50.
- Read, J.S.; Hamilton, D.P.; Jones, I.D.; Muraoka, K.; Winslow, L.A.; Kroiss, R.; Wu, C.H.; Gaiser, E. Derivation of lake mixing and stratification indices from high-resolution lake buoy data. Environ. Model Softw. 2011, 25, 1325–1336. [Google Scholar] [CrossRef]
- APHA (American Public Health Association). Standard Methods for the Examination of Water and Wastewater, 20th ed.; APHA: Washington, DC, USA, 2000. [Google Scholar]
- Bergström, A.K. The use of TN:TP and DIN:TP ratios as indicators for phytoplankton nutrient limitation in oligotrophic lakes affected by N deposition. Aquat. Sci. 2010, 72, 277–281. [Google Scholar] [CrossRef]
- Helms, J.R.; Stubbins, A.; Ritchie, J.D.; Minor, E.C.; Kieber, D.J.; Mopper, K. Absorption spectral slopes and slope ratios as indicators of molecular weight, source and photobleaching of chromophric dissolved organic matter. Limnol. Oceanogr. 2008, 53, 955–969. [Google Scholar] [CrossRef]
- Saros, J.E.; Stone, J.R.; Pederson, G.T.; Slemmons, K.E.H.; Spanbauer, T.; Schliep, A.; Cahl, D.; Williamson, C.E.; Engstrom, D.R. Climate-induced changes in lake ecosystem structure inferred from coupled neo- and paleoecological approaches. Ecology 2012, 93, 2155–2164. [Google Scholar] [CrossRef] [PubMed]
- Kirk, J.T.O. Light and Photosynthesis in Aquatic Ecosystems, 2nd ed.; Cambridge University Press: New York, NY, USA, 1994; pp. 40–45. ISBN 0521459664. [Google Scholar]
- Prowse, T.D.; Wrona, F.J.; Reist, J.D.; Gibson, J.J.; Hobbie, J.E.; Levesque, L.M.J.; Vincent, W.F. Climate change effects on hydroecology of Arctic freshwater ecosystems. AMBIO 2006, 35, 347–358. [Google Scholar] [CrossRef]
- King, J.R.; Shuter, B.J.; Zimmerman, A.P. Signals of climate trends and extreme events in thermal stratification pattern of multibasin Lake Opeongo, Ontario. Can. J. Fish. Aquat. Sci. 1999, 56, 847–852. [Google Scholar] [CrossRef]
- Weyhenmeyer, G.A.; Blenckner, T.; Pettersson, K. Changes of the plankton spring outburt related to the North Atlantic Oscillation. Limnol. Oceanogr. 1999, 44, 1788–1792. [Google Scholar] [CrossRef]
- Arvola, L.; George, G.; Livingstone, D.M.; Järvinen, M.; Blenckner, T.; Dokulil, M.T.; Jennings, E.; Aonghusa, C.N.; Nõges, P.; Nõges, T.; et al. The impact of the changing climate on the thermal characteristics of lakes. In The Impact of Climate Change on European Lakes; Aquatic Ecology Series; George, G., Ed.; Springer: Dordrecht, The Netherlands, 2009; Volume 4, pp. 85–102. ISBN 978-90-481-2944-7. [Google Scholar]
- Buffo, J.; Fritschen, L.J.; Murphy, J.L. Direct Solar Radiation on Various Slopes from 0 to 6 Degrees North Latitude; USDA Forest Service Research Paper, PNW-142; Pacific Northwest Forest and Range Experiment Station: Portland, OR, USA, 1972. [Google Scholar]
- Jeppesen, E.; Kronvang, B.; Olesen, J.E.; Larson, S.E.; Audet, J.; Søndergaard, M.; Hoffmann, C.C.; Andersen, H.E.; Lauridsen, T.L.; Liboriussen, L.; et al. Climate Change effects on nitrogen loading from cultivated catchments in Europe: Implications for nitrogen retention, ecological state of lakes and adaptation. Hydrobiologia 2011, 633, 1–21. [Google Scholar] [CrossRef]
- Fulton, R.S., III; Godwin, W.F.; Schaus, M.H. Water quality changes following nutrient loading reduction and biomanipulation in a large shallow subtropical lake, Lake Griffin, Florida, USA. Hydrobiologia 2015, 753, 243–263. [Google Scholar] [CrossRef]
- Bergström, A.-K.; Jannson, M. Atmospheric nitrogen deposition has cause nitrogen enrichment and eutrophication of lakes in the Northern Hemisphere. Glob. Chang. Biol. 2006, 12, 635–643. [Google Scholar] [CrossRef]
- Rip, W.J.; Ouboter, M.R.L.; Los, H.J. Impact of climatic fluctuations on Characeae biomass in a shallow, restored lake in the Netherlands. Hydrobiologia 2007, 584, 415–424. [Google Scholar] [CrossRef]
- Hobbie, J.E.; Peterson, B.J.; Bettez, N.; Deegan, L.; O’Brien, W.J.; Kling, G.W.; Kipphut, G.W.; Bowden, W.B.; Hershey, A.E. Impact of global change on the biogeochemistry and ecology of an Arctic freshwater system. Polar Res. 1999, 18, 207–214. [Google Scholar] [CrossRef]
- Frey, K.E.; McClelland, J.W. Impacts of permafrost degradation on Arctic river biogeochemistry. Hydrol. Process. 2009, 23, 169–182. [Google Scholar] [CrossRef]
- Jeppesen, E.; Søndergaard, M.; Jensen, J.P.; Havens, K.E.; Anneville, O.; Carvalho, L.; Coveney, M.F.; Deneke, R.; Dokulil, M.T.; Foy, B.; et al. Lake responses to reduced nutrient loading-an analysis of contemporary long-term data from 35 case studies. Freshwater Biol. 2005, 50, 1747–1771. [Google Scholar] [CrossRef]
- Wilhelm, S.; Adrian, R. Impact of summer warming on the thermal characteristics of a polymictic lake and consequences for oxygen, nutrients and phytoplankton. Freshwater Biol. 2008, 53, 226–237. [Google Scholar] [CrossRef]
- Frey, K.E.; Siegel, D.I.; Smith, L.C. Geochemistry of west Siberian streams and their potential response to permafrost degradation. Water Resour. Res. 2007, 43, W03406. [Google Scholar] [CrossRef]
- Tank, S.E.; Frey, K.E.; Striegl, R.G.; Raymond, P.A.; Holmes, R.M.; McClelland, J.W.; Peterson, B.J. Landscape-level controls on dissolved organic carbon flux from diverse catchments of the circumboreal. Glob. Biogeochem. Cycles 2012, 26, GB0E02. [Google Scholar] [CrossRef]
- Saros, J.E.; Northington, R.M.; Osburn, C.L.; Burpee, B.T.; Anderson, N.J. Thermal stratification in small Arctic lakes of southwest Greenland affected by water transparency and epilimnetic temperatures. Limnol. Oceanogr. 2016, 61, 1530–1542. [Google Scholar] [CrossRef]
- Parker, B.R.; Vinebrooke, R.D.; Schindler, D.W. Recent climate extremes alter alpine lake ecosystems. Proc. Natl. Acad. Sci. USA 2008, 105, 12917–12931. [Google Scholar] [CrossRef] [PubMed]
- Persson, L. Trophic interactions in temperate lake ecosystems: A test of food chain theory. Am. Nat. 1992, 140, 59–84. [Google Scholar] [CrossRef]
- Jeppesen, E. The impact of nutrient state and lake depth on top down control in the pelagic zone of lakes: A study of 466 lakes from the temperate zone to the Arctic. Ecosystems 2003, 6, 313–325. [Google Scholar] [CrossRef]
- Hansson, L. Food-chain length alters community responses to global change in aquatic systems. Nat. Clim. Chang. 2012, 2, 1–6. [Google Scholar] [CrossRef]
- Kraemer, B.M.; Mehner, T.; Adrian, R. Reconciling the opposing effects of warming on phytoplankton biomass in 188 large lakes. Sci. Rep. 2017, 7, 10762. [Google Scholar] [CrossRef] [PubMed]
- Malik, H.I.; Saros, J.E. Effects of temperature, light and nutrients on five Cyclotella sensu lato taxa assessed with in situ experiments in Arctic lakes. J. Plankton Res. 2016, 38, 431–442. [Google Scholar] [CrossRef]
- Saros, J.E.; Anderson, N.J. The ecology of the planktonic diatom Cyclotella and its implications for global environmental change studies. Biol. Rev. 2015, 90, 522–541. [Google Scholar] [CrossRef] [PubMed]

| Region | Spring | Summer | |||
|---|---|---|---|---|---|
| Early Ice-Out | Late Ice-Out | Early Ice-Out | Late Ice-Out | ||
| Arctic | Year | 2016 | 2015 | 2016 | 2013 |
| Ice-out date | 18 May | 17 June | 18 May | 9 June | |
| Sampling dates | 28–30 June | 27 June–1 July | 15–17 July | 19–21 July | |
| Boreal | Year | 2012 | 2015 | 2012 | 2015 |
| Ice-out date | 19 March | 29 April | 19 March | 29 April | |
| Sampling dates | 11 June | 11 June | 12 July | 10 July | |
| Region | Lake | Lat | Long | Elevation (m) | Surface Area (km2) | Volume (×106 m3) | Max Depth (m) |
|---|---|---|---|---|---|---|---|
| Arctic | SS2 | 66.99 | −50.96 | 190 | 0.368 | 2.49 | 12 |
| SS85 | 66.98 | −51.06 | 195 | 0.246 | 0.94 | 11 | |
| SS1590 | 67.01 | −50.98 | 200 | 0.243 | 1.16 | 18 | |
| Boreal | Jordan | 44.33 | −68.26 | 83 | 0.800 | 17.4 | 45 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Warner, K.A.; Fowler, R.A.; Northington, R.M.; Malik, H.I.; McCue, J.; Saros, J.E. How Does Changing Ice-Out Affect Arctic versus Boreal Lakes? A Comparison Using Two Years with Ice-Out that Differed by More Than Three Weeks. Water 2018, 10, 78. https://doi.org/10.3390/w10010078
Warner KA, Fowler RA, Northington RM, Malik HI, McCue J, Saros JE. How Does Changing Ice-Out Affect Arctic versus Boreal Lakes? A Comparison Using Two Years with Ice-Out that Differed by More Than Three Weeks. Water. 2018; 10(1):78. https://doi.org/10.3390/w10010078
Chicago/Turabian StyleWarner, Kate A., Rachel A. Fowler, Robert M. Northington, Heera I. Malik, Joan McCue, and Jasmine E. Saros. 2018. "How Does Changing Ice-Out Affect Arctic versus Boreal Lakes? A Comparison Using Two Years with Ice-Out that Differed by More Than Three Weeks" Water 10, no. 1: 78. https://doi.org/10.3390/w10010078
APA StyleWarner, K. A., Fowler, R. A., Northington, R. M., Malik, H. I., McCue, J., & Saros, J. E. (2018). How Does Changing Ice-Out Affect Arctic versus Boreal Lakes? A Comparison Using Two Years with Ice-Out that Differed by More Than Three Weeks. Water, 10(1), 78. https://doi.org/10.3390/w10010078





