Measured Solid State and Sub-Cooled Liquid Vapour Pressures of Benzaldehydes Using Knudsen Effusion Mass Spectrometry
Abstract
1. Introduction
2. Experimental
2.1. The Knudsen Effusion Mass Spectrometry System (KEMS)
2.2. Differential Scanning Calorimetry (DSC)
2.3. MOPAC2016
3. Theory
3.1. Sub-Cooled Correction
3.2. Vapour Pressure Predictive Techniques
4. Results and Discussion
4.1. Solid State Vapour Pressure
4.2. Sub-Cooled Liquid Vapour Pressure
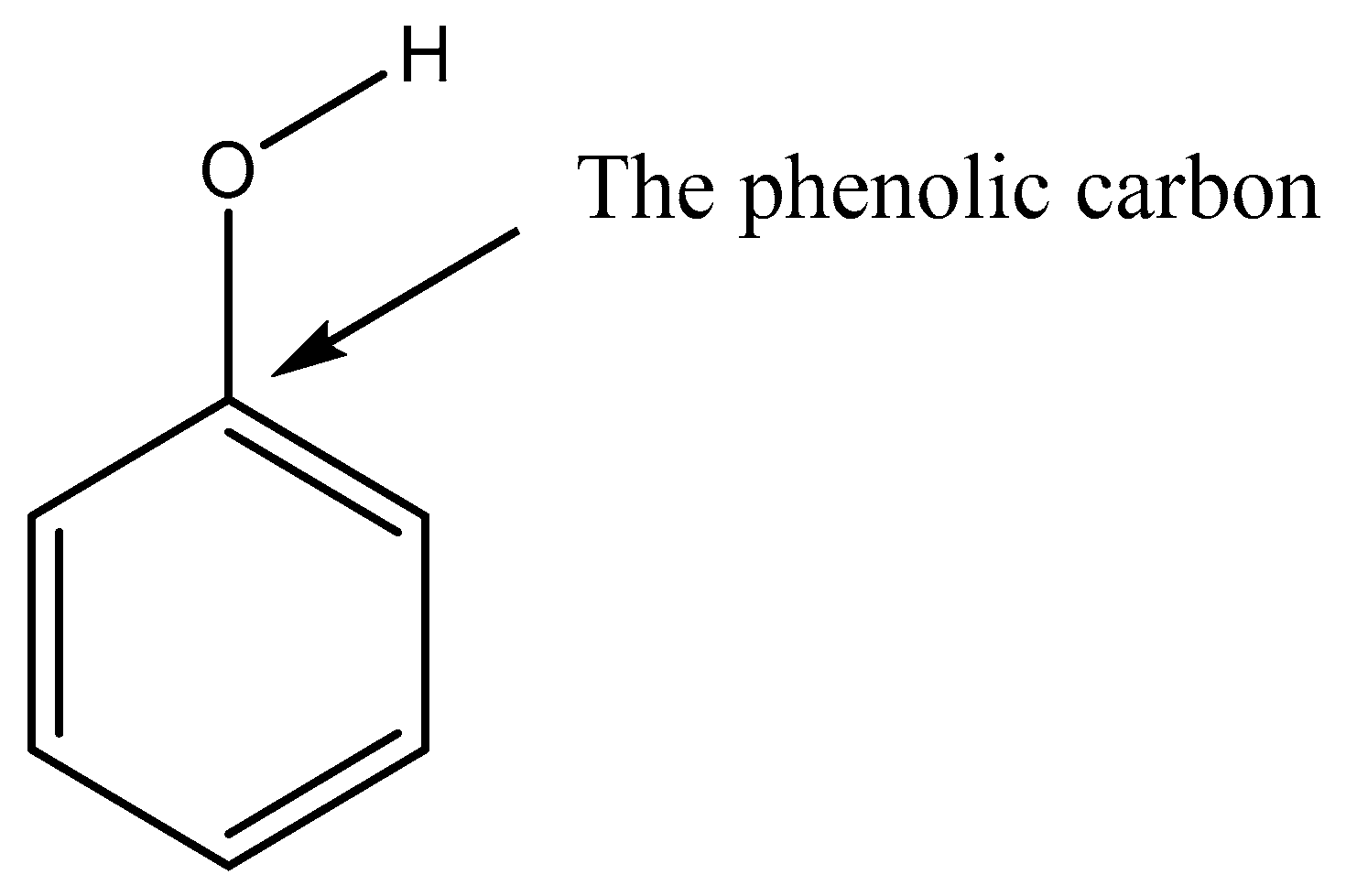
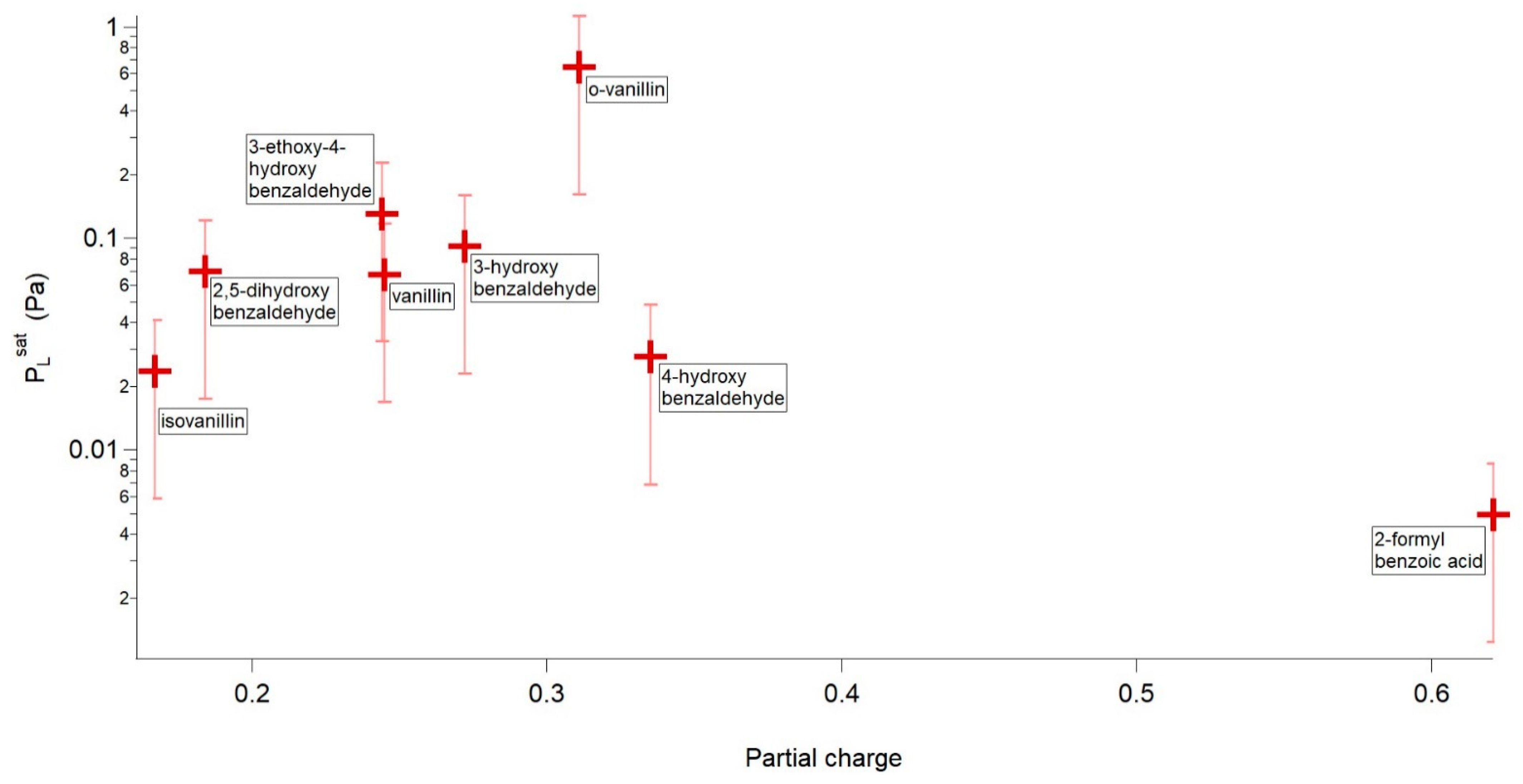
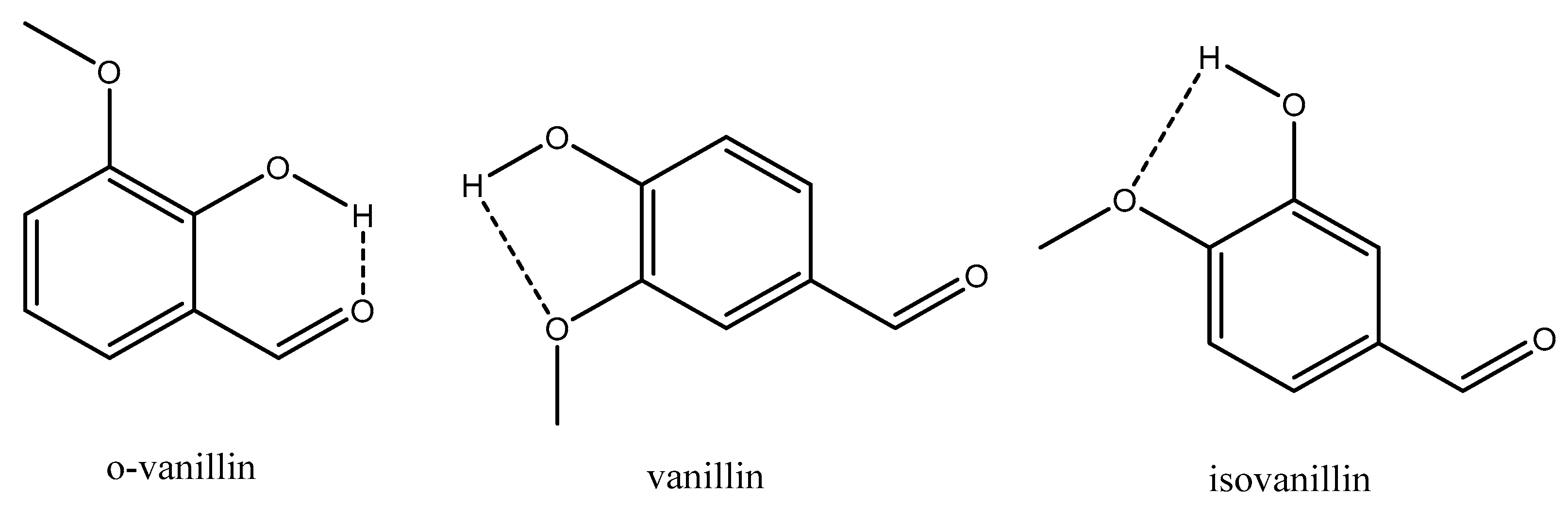
4.2.1. H-Bonding Compounds
4.2.2. Non H-Bonding Compounds
4.2.3. Comparisons between H-Bonding and non H-Bonding Compounds
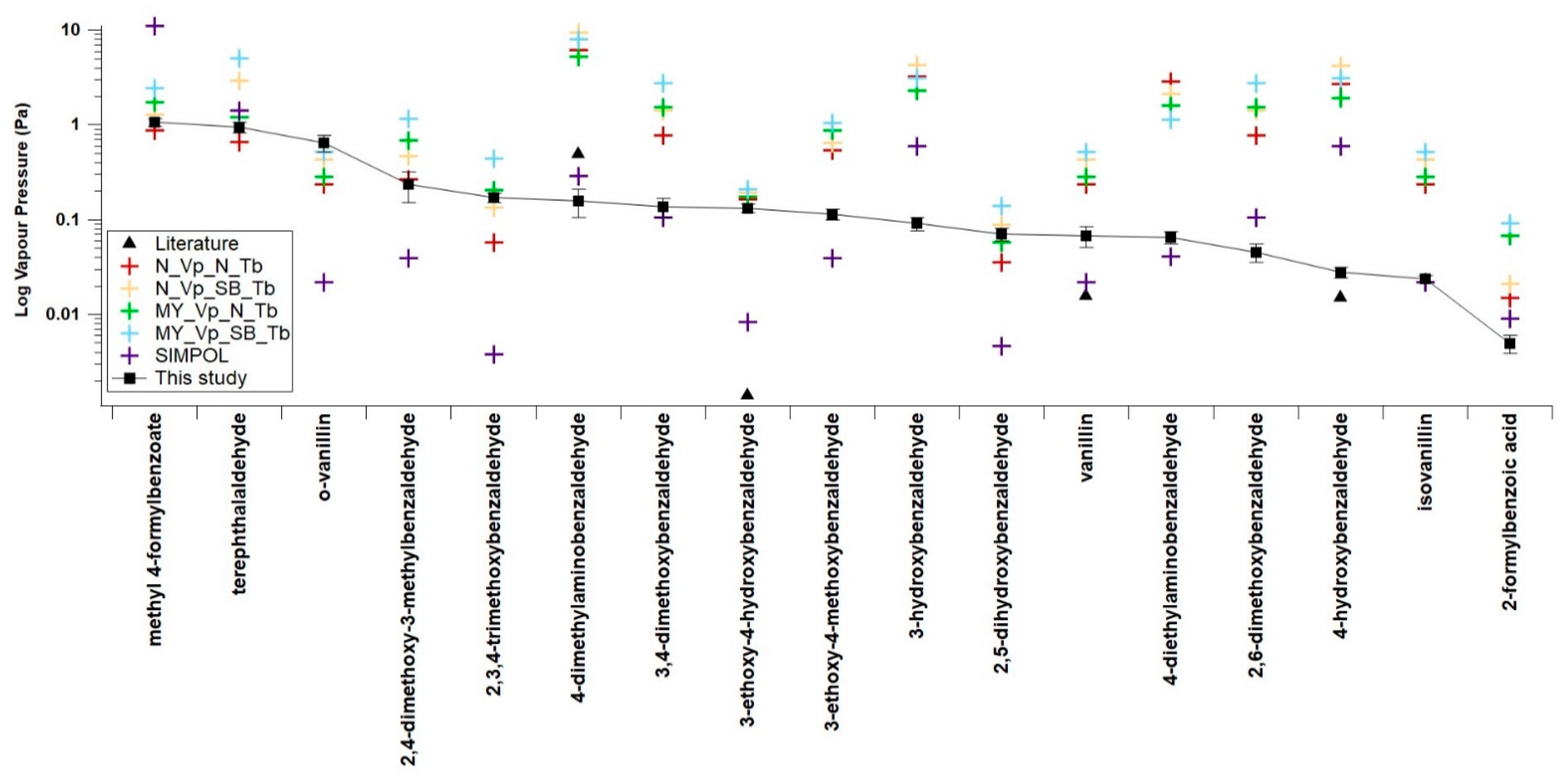
4.3. Comparisons with Estimations from GCMs
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Seinfeld, J.H.; Pandis, S.N. Atmospheric Chemistry and Physics: From Air Pollution to Climate Change, 3rd ed.; John Wiley & Sons, Incorporated: New York, NY, USA, 2016. [Google Scholar]
- Kanakidou, M.; Seinfeld, J.H.; Pandis, S.N.; Barnes, I.; Dentener, F.J.; Facchini, M.C.; van Dingenen, R.; Ervens, B.; Nenes, A.; Nielsen, C.J.; et al. Organic aerosol and global climate modelling: A review. Atmos. Chem. Phys. 2005, 5, 1053–1123. [Google Scholar] [CrossRef]
- Bilde, M.; Barsanti, K.; Booth, M.; Cappa, C.D.; Donahue, N.M.; Emanuelsson, E.U.; McFiggans, G.; Krieger, U.K.; Marcolli, C.; Topping, D.; et al. Saturation Vapor Pressures and Transition Enthalpies of Low-Volatility Organic Molecules of Atmospheric Relevance: From Dicarboxylic Acids to Complex Mixtures. Chem. Rev. 2015, 115, 4115–4156. [Google Scholar] [CrossRef] [PubMed]
- Booth, A.M.; Barley, M.H.; Topping, D.O.; Mcfiggans, G.; Garforth, A.; Percival, C.J. Solid state and sub-cooled liquid vapour pressures of substituted dicarboxylic acids using Knudsen Effusion Mass Spectrometry (KEMS) and Differential Scanning Calorimetry. Atmos. Chem. Phys. 2010, 10, 4879–4892. [Google Scholar] [CrossRef]
- Hallquist, M.; Wenger, J.C.; Baltensperger, U.; Rudich, Y.; Simpson, D.; Claeys, M.; Dommen, J.; Donahue, N.M.; George, C.; Goldstein, A.H.; et al. The formation, properties and impact of secondary organic aerosol: Current and emerging issues. Atmos. Chem. Phys. Atmos. Chem. Phys. 2009, 9, 5155–5236. [Google Scholar] [CrossRef]
- Nannoolal, Y.; Rarey, J.; Ramjugernath, D. Fluid Phase Equilibria Estimation of pure component properties Part 3. Estimation of the vapor pressure of non-electrolyte organic compounds via group contributions and group interactions. Fluid Phase Equilib. 2008, 269, 117–133. [Google Scholar] [CrossRef]
- Myrdal, P.B.; Yalkowsky, S.H. Estimating Pure Component Vapor Pressures of Complex Organic Molecules. Ind. Eng. Chem. Res. 1997, 36, 2494–2499. [Google Scholar] [CrossRef]
- Pankow, J.F.; Asher, W.E. SIMPOL.1: A simple group contribution method for predicting vapor pressures and enthalpies of vaporization of multifunctional organic compounds. Atmos. Chem. Phys. 2008, 8, 2773–2796. [Google Scholar] [CrossRef]
- Compernolle, S.; Ceulemans, K.; Müller, J.F. Evaporation: A new vapour pressure estimation methodfor organic molecules including non-additivity and intramolecular interactions. Atmos. Chem. Phys. 2011, 11, 9431–9450. [Google Scholar] [CrossRef]
- Barley, M.H.; McFiggans, G. The critical assessment of vapour pressure estimation methods for use in modelling the formation of atmospheric organic aerosol. Atmos. Chem. Phys 2010, 10, 749–767. [Google Scholar] [CrossRef]
- O’Meara, S.; Booth, A.M.; Barley, M.H.; Topping, D.; Mcfiggans, G. An assessment of vapour pressure estimation methods. Phys. Chem. Chem. Phys. 2014, 16, 19453–19469. [Google Scholar] [CrossRef] [PubMed]
- Booth, A.M.; Bannan, T.; McGillen, M.R.; Barley, M.H.; Topping, D.O.; McFiggans, G.; Percival, C.J. The role of ortho, meta, para isomerism in measured solid state and derived sub-cooled liquid vapour pressures of substituted benzoic acids. RSC Adv. 2012, 2, 4430. [Google Scholar] [CrossRef]
- Dang, C.; Bannan, T.; Shelley, P.; Priestley, M.; Worrall, S.D.; Waters, J.; Coe, H.; Percival, C.J.; Topping, D. The effect of structure and isomerism on the vapour pressures of organic molecules and its potential atmospheric relevance. Aerosol. Sci. Technol. 2019, 53, 1–32. [Google Scholar] [CrossRef]
- Shelley, P.D.; Bannan, T.J.; Worrall, S.D.; Alfarra, M.R.; Krieger, U.K.; Percival, C.J.; Garforth, A.; Topping, D. Measured solid state and subcooled liquid vapour pressures of nitroaromatics using Knudsen effusion mass spectrometry. Atmos. Chem. Phys. 2020, 20, 8293–8314. [Google Scholar] [CrossRef]
- Caralp, F.; Foucher, V.; Lesclaux, R.; Wallington, T.J.; Michael Hurley, B.D. Atmospheric chemistry of benzaldehyde: UV absorption spectrum and reaction kinetics and mechanisms of the radical C6H5C(O)O2. Phys. Chem. Chem. Phys. 1999, 1, 3509–3517. [Google Scholar] [CrossRef]
- Baghi, R.; Helmig, D.; Guenther, A.; Duhl, T.; Daly, R. Contribution of flowering trees to urban atmospheric biogenic volatile organic compound emissions. Biogeosciences 2012, 9, 3777–3785. [Google Scholar] [CrossRef]
- Thiault, G.; Mellouki, A.; Le Bras, G.; Chakir, A.; Sokolowski-Gomez, N.; Daumont, D. UV-absorption cross sections of benzaldehyde, ortho-, meta-, and para-tolualdehyde. J. Photochem. Photobiol. A Chem. 2004, 162, 273–281. [Google Scholar] [CrossRef]
- Dubtsov, S.N.; Dultseva, G.G.; Dultsev, E.N.; Skubnevskaya, G.I. Investigation of aerosol formation during benzaldehyde photolysis. J. Phys. Chem. B 2006, 110, 645–649. [Google Scholar] [CrossRef] [PubMed]
- Peng, C.-Y.; Yang, H.-H.; Lan, C.-H.; Chien, S.-M. Effects of the biodiesel blend fuel on aldehyde emissions from diesel engine exhaust. Atmos. Environ. 2008, 42, 906–915. [Google Scholar] [CrossRef]
- Magnusson, R.; Nilsson, C.; Andersson, B. Emissions of Aldehydes and Ketones from a Two-Stroke Engine Using Ethanol and Ethanol-Blended Gasoline as Fuel. Environ. Sci. Technol. 2002, 36, 1656–1664. [Google Scholar] [CrossRef]
- Hamilton, J.F.; Webb, P.J.; Lewis, A.C.; Reviejo, M.M. Quantifying small molecules in secondary organic aerosol formed during the photo-oxidation of toluene with hydroxyl radicals. Atmos. Environ. 2005, 39, 7263–7275. [Google Scholar] [CrossRef]
- Jenkin, M.E.; Saunders, S.M.; Wagner, V.; Pilling, M.J. Protocol for the development of the Master Chemical Mechanism, MCM v3 (Part B): Tropospheric degradation of aromatic volatile organic compounds. Atmos. Chem. Phys. 2003, 3, 181–193. [Google Scholar] [CrossRef]
- Bloss, C.; Wagner, V.; Jenkin, M.E.; Volkamer, R.; Bloss, W.J.; Lee, J.D.; Heard, D.E.; Wirtz, K.; Martin-Reviejo, M.; Rea, G.; et al. Development of a Detailed Chemical Mechanism (MCMv3.1) for the Atmospheric Oxidation of Aromatic Hydrocarbons. Atmos. Chem. Phys. 2005, 5, 641–644. [Google Scholar] [CrossRef]
- Bannan, T.J.; Booth, A.M.; Jones, B.T.; O’meara, S.; Barley, M.H.; Riipinen, I.; Percival, C.J.; Topping, D. Measured Saturation Vapor Pressures of Phenolic and Nitro-aromatic Compounds. Environ. Sci. Technol. 2017, 51, 3922–3928. [Google Scholar] [CrossRef] [PubMed]
- Booth, A.M.; Markus, T.; Mcfiggans, G.; Percival, C.J.; Mcgillen, M.R.; Topping, D.O. Design and construction of a simple Knudsen Effusion Mass Spectrometer (KEMS) system for vapour pressure measurements of low volatility organics. Atmos. Meas. Tech. 2009, 2, 355–361. [Google Scholar] [CrossRef]
- Krieger, U.K.; Siegrist, F.; Marcolli, C.; Emanuelsson, E.U.; Gøbel, F.M.; Bilde, M.; Marsh, A.; Reid, J.P.; Huisman, A.J.; Riipinen, I.; et al. A reference data set for validating vapor pressure measurement techniques: Homologous series of polyethylene glycols. Atmos. Meas. Tech. 2018, 11, 49–63. [Google Scholar] [CrossRef]
- Booth, A.M.; Bannan, T.J.; Benyezzar, M.; Bacak, A.; Alfarra, M.R.; Topping, D.; Percival, C.J. Development of lithium attachment mass spectrometry—Knudsen effusion and chemical ionisation mass spectrometry (KEMS, CIMS). Analyst 2017, 142, 3666–3673. [Google Scholar] [CrossRef] [PubMed]
- Bannan, T.J.; Le Breton, M.; Priestley, M.; Worrall, S.D.; Bacak, A.; Marsden, N.A.; Mehra, A.; Hammes, J.; Hallquist, M.; Alfarra, M.R.; et al. A method for extracting calibrated volatility information from the FIGAERO-HR-ToF-CIMS and its experimental application. Atmos. Meas. Tech. 2019, 12, 1429–1439. [Google Scholar] [CrossRef]
- Hilpert, K. Potential of mass spectrometry for the analysis of inorganic high-temperature vapors. In Proceedings of the Fresenius’ Journal of Analytical Chemistry; Springer: Berlin/Heidelberg, Germany, 2001; Volume 370, pp. 471–478. [Google Scholar]
- Atomic Total Energies: Atomic Reference Data for Electronic Structure Calculations. Available online: https://physics.nist.gov/PhysRefData/Ionization/atom_index.html (accessed on 11 March 2021).
- Mauger, J.W.; Paruta, A.N.; Gerraughty, R.J. Solubilities of Sulfadiazine, Sulfisomidine, and Sulfadimethoxine in Several Normal Alcohols. J. Pharm. Sci. 1972, 61, 94–97. [Google Scholar] [CrossRef]
- Grant, D.J.W.; Mehdizadeh, M.; Chow, A.H.-L.; Fairbrother, J.E. Non-linear van’t Hoff solubility-temperature plots and their pharmaceutical interpretation. Int. J. Pharm. 1984, 18, 25–38. [Google Scholar] [CrossRef]
- Stewart, J.J. MOPAC2016; Stewart Computational Chemistry: Colorado Springs, CO, USA, 2016. [Google Scholar]
- Dewar, M.J.S.; Thiel, W. A semiempirical model for the two-center repulsion integrals in the NDDO approximation. Theor. Chim. Acta 1977, 46, 89–104. [Google Scholar] [CrossRef]
- Prausnitz, J.; Lichtenthaler, R.; de Azevedo, E. Molecular Thermodynamics of Fluid-Phase Equilibria; Pearson Education: Upper Saddle River, NJ, USA, 1998. [Google Scholar]
- Nannoolal, Y.; Rarey, J.; Ramjugernath, D.; Cordes, W. Estimation of pure component properties Part 1. Estimation of the normal boiling point of non-electrolyte organic compounds via group contributions and group interactions. Fluid Phase Equilib. 2004, 226, 45–63. [Google Scholar] [CrossRef]
- Joback, K.G.; Reid, R.C.; Reid, C. Estimation of pure-component properties from group-contributions. Chem. Eng. Commun. 1987, 157, 233–243. [Google Scholar] [CrossRef]
- Stein, S.E.; Brown, R.L. Estimation of Normal Boiling Points from Group Contributions. J. Chem. Inf. Comput. Sci. 1994, 34, 581–587. [Google Scholar] [CrossRef]
- Shelley, P.D.; Bannan, T.J.; Worrall, S.D.; Alfarra, M.R.; Percival, C.J.; Garforth, A.; Topping, D. PetrocShelley/Measured_Solid_State_and_Sub_Cooled_Liquid_Vapour_Pressures_of_Benzaldehydes_Using_KEMS_Data_Set: Pre Release; Zenodo: Genève, Switzerland, 2020. [Google Scholar] [CrossRef]
- Staikova, M.; Wania, F.; Donaldson, D.J. Molecular polarizability as a single-parameter predictor of vapour pressures and octanol–air partitioning coefficients of non-polar compounds: A priori approach and results. Atmos. Environ. 2004, 38, 213–225. [Google Scholar] [CrossRef]
- Staikova, M.; Messih, P.; Lei, Y.D.; Wania, F.; Donaldson, J.D. Prediction of Subcooled Vapor Pressures of Nonpolar Organic Compounds Using a One-Parameter QSPR. J. Chem. Eng. Data 2005, 50, 438–443. [Google Scholar] [CrossRef]
- Liang, C.; Gallagher, D.A. QSPR Prediction of Vapor Pressure from Solely Theoretically-Derived Descriptors. J. Chem. Inf. Comput. Sci. 1998, 38, 321–324. [Google Scholar] [CrossRef]
- Schwarzenbach, R.P.; Gschwend, P.M.; Imboden, D.M. Environmental Organic Chemistry; John Wiley & Sons, Incorporated: Hoboken, NJ, USA, 2016; ISBN 9781118767238. [Google Scholar]
- Remko, M.; Polcin, J. Theoretical study of the hydrogen bonding ability of phenol and its ortho, meta and para substituted derivatives. Adv. Mol. Relax. Interact. Process. 1977, 11, 249–254. [Google Scholar] [CrossRef]
- Stymne, B.; Stymne, H.; Wettermark, G. Substituent Effects in the Thermodynamics of Hydrogen Bonding as Obtained by Infrared Spectrometry. J. Am. Chem. Soc. 1973, 95, 3490–3494. [Google Scholar] [CrossRef]
- Daubert, T.E.; Danner, R.P. Physical and Thermodynamic Properties of Pure Chemicals: Data Compilation; Hemisphere Pub. Corp.: New York, NY, USA, 1989. [Google Scholar]
- Yaws, C.L. Handbook of Vapor Pressure; Gulf Pub. Co.: Houston, TX, USA, 1994. [Google Scholar]
| Compound | Structure | CAS | Supplier |
|---|---|---|---|
| Vanillin (4-hydroxy-3-methoxybenzaldehyde) |  | 121-33-5 | Sigma Aldrich |
| Isovanillin (3-hydroxy-4-methoxybenzaldehyde) |  | 621-59-0 | Sigma Aldrich |
| o-vanillin (2-hydroxy-3-methoxybenzaldehyde) |  | 148-53-8 | Fisher Scientific |
| 3-hydroxybenzaldehyde |  | 100-83-4 | Sigma Aldrich |
| 4-hydroxybenzaldehyde |  | 123-08-0 | Fisher Scientific |
| 2,5-dihydroxybenzaldehyde |  | 1194-98-5 | Fisher Scientific |
| 3-ethoxy-4-hydroxybenzaldehyde |  | 121-32-4 | Sigma Aldrich |
| 2-formylbenzoic acid |  | 119-67-5 | Fisher Scientific |
| 4-dimethylaminobenzaldehyde |  | 100-10-7 | Fisher Scientific |
| 4-diethylaminobenzaldehyde |  | 120-21-8 | Sigma Aldrich |
| methyl-4-formylbenzoate |  | 1571-08-0 | Sigma Aldrich |
| terephthalaldehyde |  | 623-27-8 | Sigma Aldrich |
| 3,4-dimethoxybenzaldehyde |  | 120-14-9 | Sigma Aldrich |
| 2,6-dimethoxybenzaldehyde | 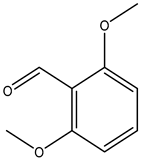 | 3392-97-0 | Alfa Aesar |
| 3-ethoxy-4-methoxybenzaldehyde | 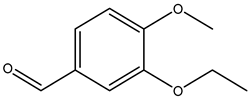 | 1131-52-8 | Sigma Aldrich |
| 2,4-dimethoxy-3-methylbenzaldehyde | 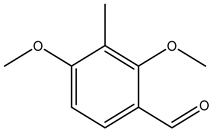 | 7149-92-0 | Sigma Aldrich |
| 2,3,4-trimethoxybenzaldehyde | 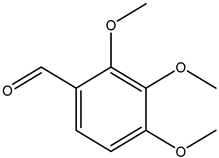 | 2103-57-3 | Sigma Aldrich |
| Compound | P298 (Pa) | ΔHsub (kJ mol−1) | ΔSsub (J mol−1 K−1) |
|---|---|---|---|
| Methyl 4-formylbenzoate | 3.97 × 10−1 | 75.98 | 247.24 |
| terephthalaldehyde | 2.34 × 10−1 | 76.66 | 245.10 |
| 2,3,4-trimethoxybenzaldehyde | 1.11 × 10−1 | 87.51 | 275.35 |
| 2,4-dimethoxy-3-methylbenzaldehyde | 1.09 × 10−1 | 79.02 | 246.18 |
| 3,4-dimethoxybenzaldehyde | 6.64 × 10−2 | 91.58 | 284.67 |
| 3-ethoxy-4-methoxybenzaldehyde | 5.72 × 10−2 | 94.49 | 293.23 |
| 4-dimethylaminobenzaldehyde | 5.19 × 10−2 | 95.42 | 295.11 |
| 4-diethylaminobenzaldehyde | 4.44 × 10−2 | 92.28 | 283.70 |
| 2,6-dimethoxybenzaldehyde | 7.29 × 10−3 | 118.09 | 355.16 |
| o-vanillin | 3.88 × 10−1 | 67.75 | 219.30 |
| 3-ethoxy-4-hydroxybenzaldehyde | 3.14 × 10−2 | 100.35 | 307.95 |
| Vanillin | 2.14 × 10−2 | 108.16 | 330.77 |
| 2,5-dihydroxybenzaldehyde | 1.63 × 10−2 | 102.50 | 309.66 |
| 3-hydroxybenzaldehyde | 1.58 × 10−2 | 109.90 | 334.17 |
| 4-hydroxybenzaldehyde | 5.86 × 10−3 | 107.76 | 318.80 |
| Isovanillin | 3.43 × 10−3 | 119.00 | 352.12 |
| 2-formylbenzoic acid | 1.11 × 10−3 | 114.82 | 328.51 |
| Compound | P298 (Pa) | Tm (K) | ΔHfus (kJ mol−1) | ΔSfus (J mol−1 K−1) | Partial Charge of the Phenolic/Carboxylic Carbon |
|---|---|---|---|---|---|
| o-vanillin | 6.44 × 10−1 | 320.09 | 19.06 | 59.55 | 0.311 |
| 3-ethoxy-4-hydroxybenzaldehyde | 1.31 × 10−1 | 351.70 | 25.27 | 71.85 | 0.244 |
| 3-hydroxybenzaldehyde | 9.21 × 10−2 | 378.98 | 23.19 | 61.20 | 0.272 |
| 2,5-dihydroxybenzaldehyde | 7.02 × 10-2 | 373.15 | 20.16 | 54.02 | 0.329 (intra) 0.184 (inter) |
| Vanillin | 6.73 × 10−2 | 356.82 | 18.88 | 52.90 | 0.245 |
| 4-hydroxybenzaldehyde | 2.78 × 10−2 | 391.40 | 18.60 | 47.53 | 0.335 |
| Isovanillin | 2.36 × 10−2 | 390.34 | 23.20 | 59.44 | 0.167 |
| 2-formylbenzoic acid | 4.96 × 10−3 | 375.12 | 20.36 | 54.28 | 0.621 |
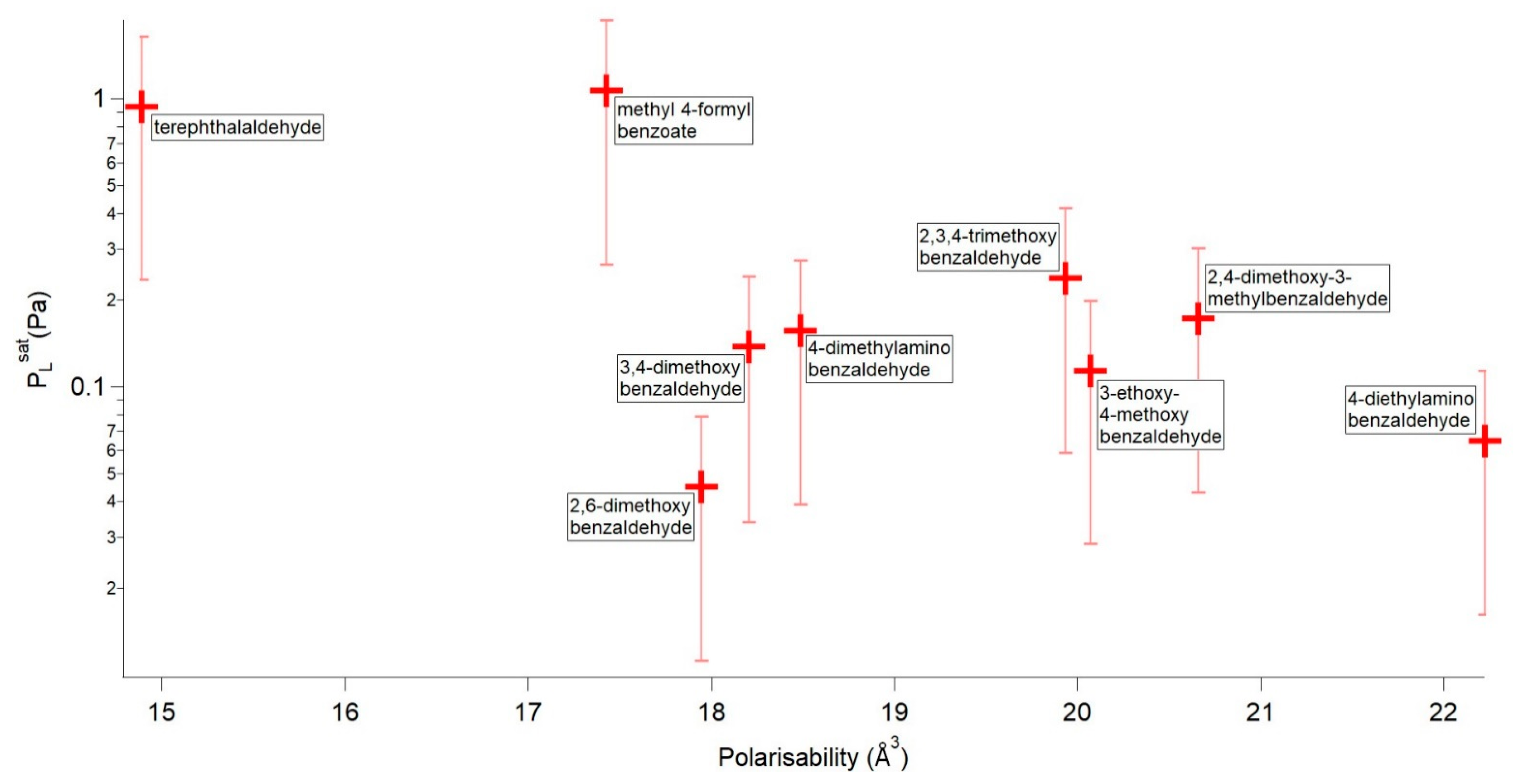
| Compound | P298 (Pa) | Tm (K) | ΔHfus (kJ mol−1) | ΔSfus (J mol−1 K−1) | αm (Å3) |
|---|---|---|---|---|---|
| methyl 4-formylbenzoate | 1.07 × 100 | 337.21 | 22.48 | 66.66 | 17.424 |
| terephthalaldehyde | 9.43 × 10−1 | 390.06 | 16.82 | 43.11 | 14.888 |
| 2,4-dimethoxy-3-methylbenzaldehyde | 2.38 × 10−1 | 327.43 | 22.81 | 69.66 | 19.931 |
| 2,3,4-trimethoxybenzaldehyde | 1.73 × 10−1 | 313.63 | 22.64 | 72.17 | 20.658 |
| 4-dimethylaminobenzaldehyde | 1.57 × 10−1 | 349.37 | 20.24 | 57.93 | 18.488 |
| 3,4-dimethoxybenzaldehyde | 1.38 × 10−1 | 321.53 | 20.77 | 64.61 | 18.206 |
| 3-ethoxy-4-methoxybenzaldehyde | 1.14 × 10−1 | 324.96 | 21.67 | 66.67 | 20.071 |
| 4-diethylaminobenzaldehyde | 6.49 × 10−2 | 314.53 | 18.59 | 59.11 | 22.224 |
| 2,6-dimethoxybenzaldehyde | 4.50 × 10−2 | 373.19 | 25.16 | 67.43 | 17.944 |
| N_VP_N_Tb | N_VP_SB_Tb | MY_VP_N_Tb | MY_VP_SB_Tb | SIMPOL | |
|---|---|---|---|---|---|
| Average difference (orders of magnitude) | 0.60 | 0.82 | 0.77 | 0.98 | −0.20 |
| Compounds | N_VP_N_Tb | N_VP_SB_Tb | MY_VP_N_Tb | MY_VP_SB_Tb | SIMPOL |
|---|---|---|---|---|---|
| This study | 0.60 | 0.82 | 0.77 | 0.98 | −0.20 |
| Non H-bonding—this study | 0.58 | 0.81 | 0.80 | 1.02 | −0.15 |
| H-Bonding—this study | 0.61 | 0.83 | 0.72 | 0.93 | −0.24 |
| Nitrobenzaldehyde from Shelley et al. (2020) [14] | 3.18 | 2.50 | 3.17 | 2.46 | 0.29 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shelley, P.; Bannan, T.J.; Worrall, S.D.; Alfarra, M.R.; Percival, C.J.; Garforth, A.; Topping, D. Measured Solid State and Sub-Cooled Liquid Vapour Pressures of Benzaldehydes Using Knudsen Effusion Mass Spectrometry. Atmosphere 2021, 12, 397. https://doi.org/10.3390/atmos12030397
Shelley P, Bannan TJ, Worrall SD, Alfarra MR, Percival CJ, Garforth A, Topping D. Measured Solid State and Sub-Cooled Liquid Vapour Pressures of Benzaldehydes Using Knudsen Effusion Mass Spectrometry. Atmosphere. 2021; 12(3):397. https://doi.org/10.3390/atmos12030397
Chicago/Turabian StyleShelley, Petroc, Thomas J. Bannan, Stephen D. Worrall, M. Rami Alfarra, Carl J. Percival, Arthur Garforth, and David Topping. 2021. "Measured Solid State and Sub-Cooled Liquid Vapour Pressures of Benzaldehydes Using Knudsen Effusion Mass Spectrometry" Atmosphere 12, no. 3: 397. https://doi.org/10.3390/atmos12030397
APA StyleShelley, P., Bannan, T. J., Worrall, S. D., Alfarra, M. R., Percival, C. J., Garforth, A., & Topping, D. (2021). Measured Solid State and Sub-Cooled Liquid Vapour Pressures of Benzaldehydes Using Knudsen Effusion Mass Spectrometry. Atmosphere, 12(3), 397. https://doi.org/10.3390/atmos12030397







