One-Year Characterization and Reactivity of Isoprene and Its Impact on Surface Ozone Formation at A Suburban Site in Guangzhou, China
Abstract
- The characterization and reactivity of isoprene at a subtropical suburban site in Guangzhou were investigated.
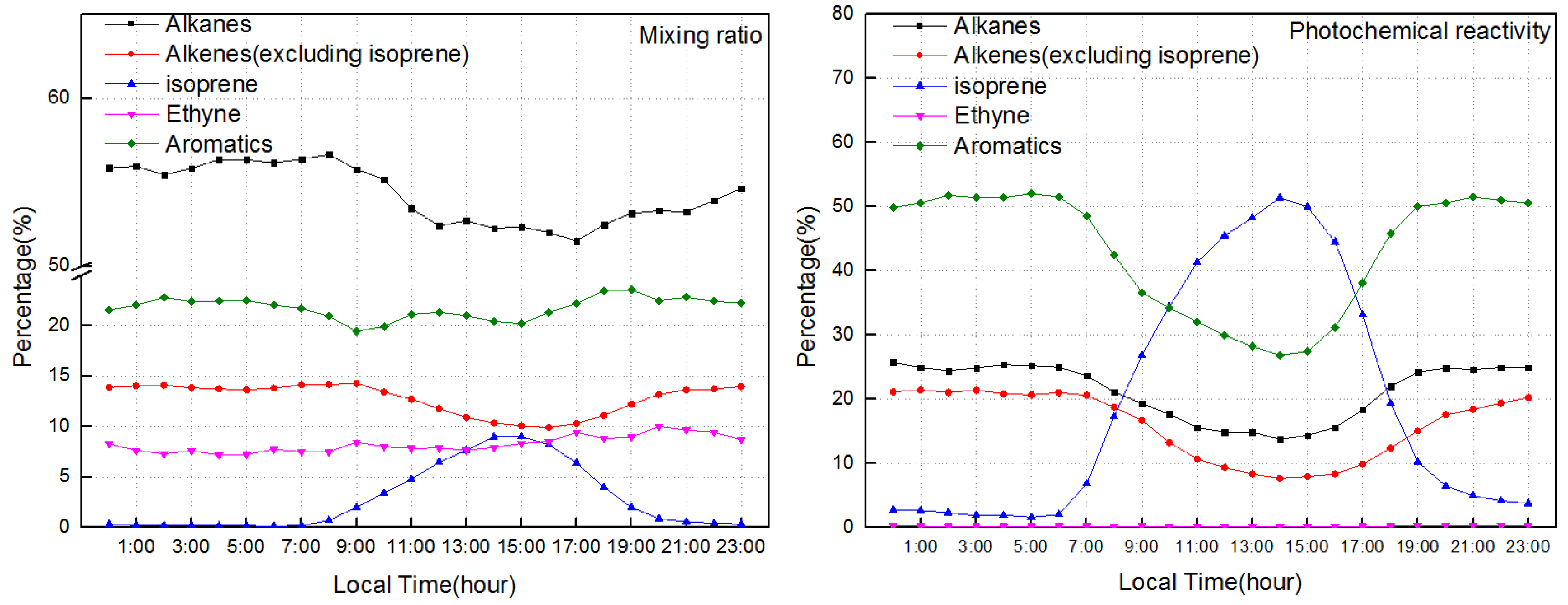
- Isoprene ranked first for OFP and propylene-equivalent mixing ratio during the O3 formation period.
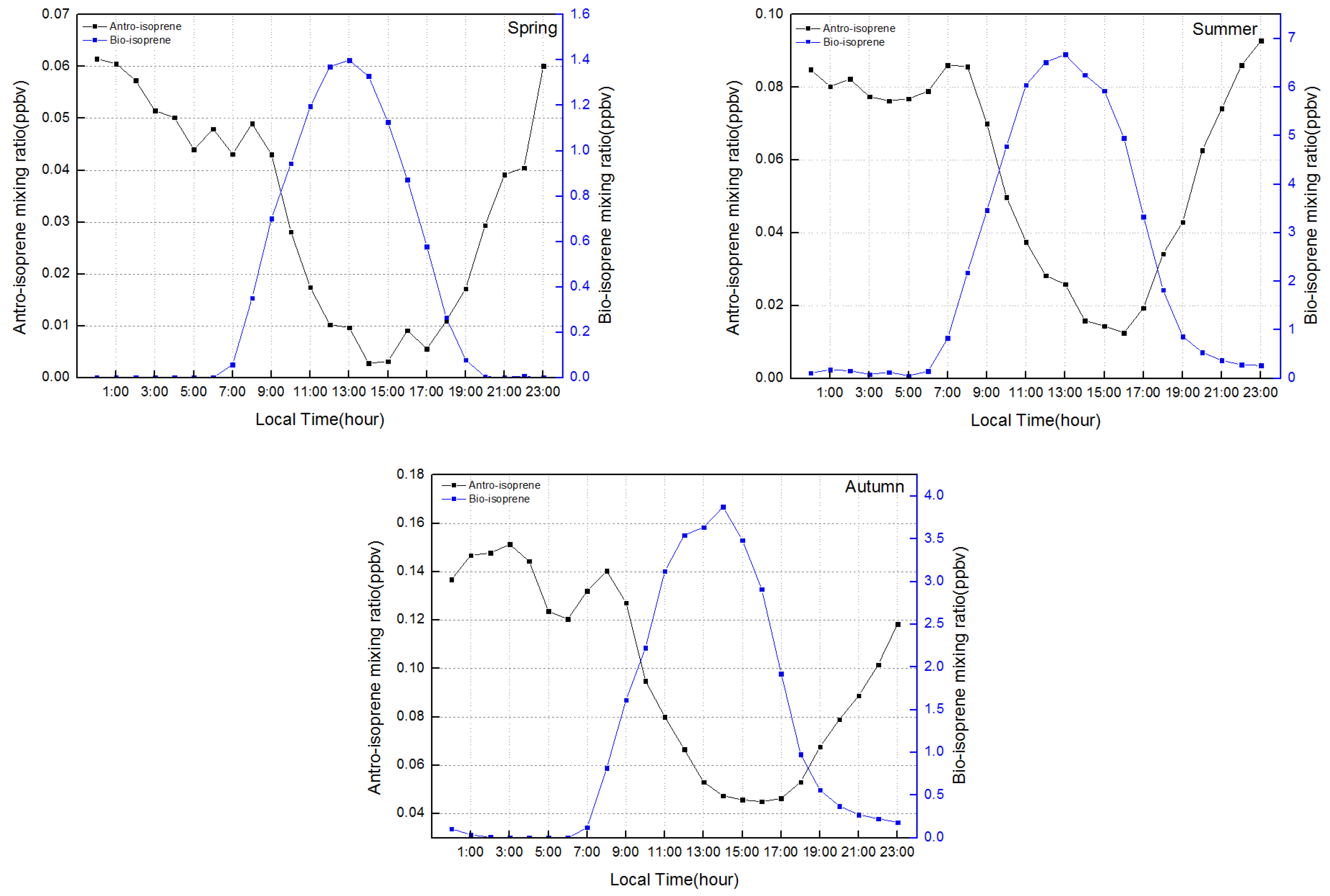
- Daytime biogenic isoprene emissions dominated over anthropogenic isoprene emissions in all four seasons.
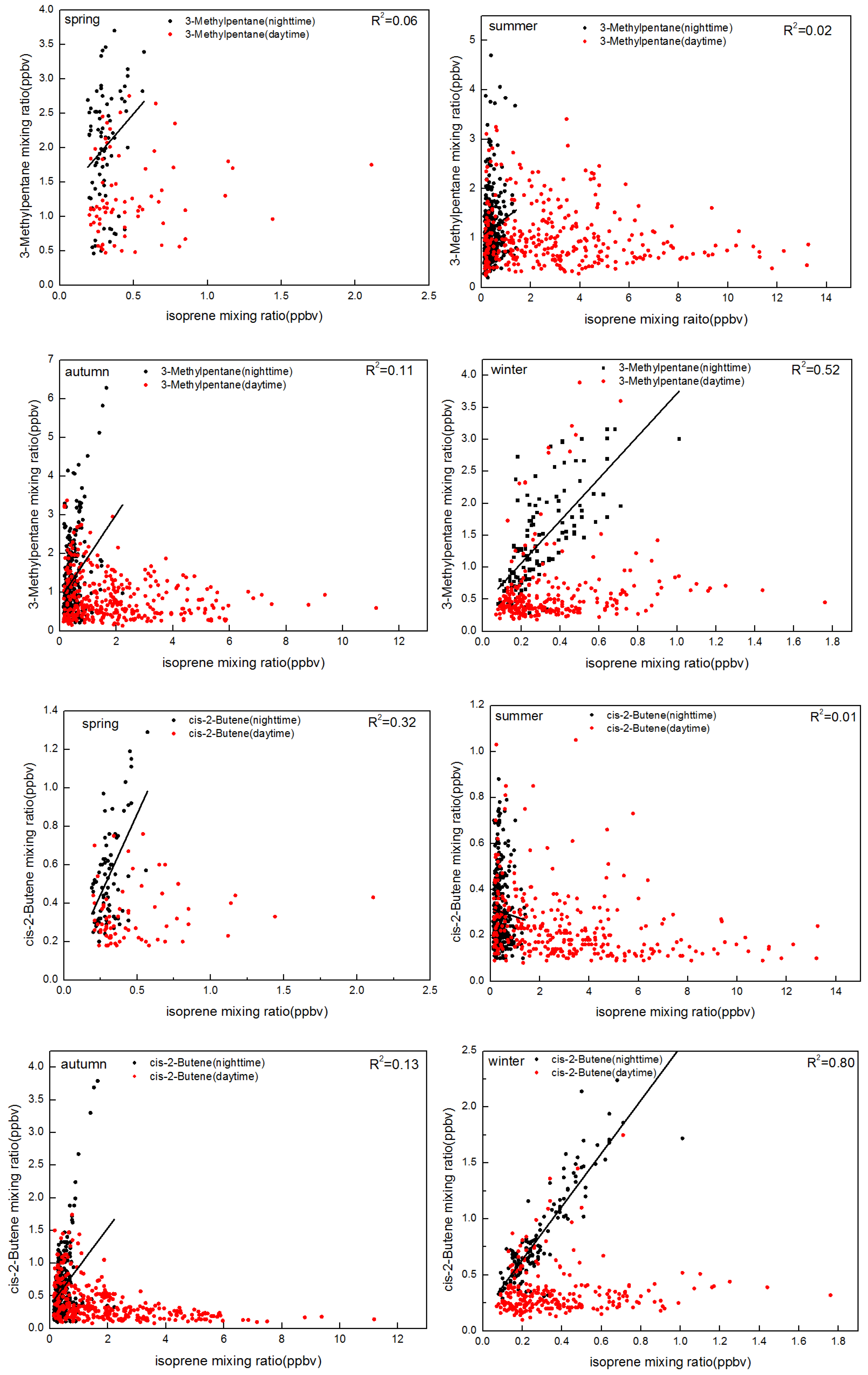
- Vehicular emissions contributed approximately 71.6%, 30.5%, and 33.1% of night-time isoprene emissions in spring, summer, and autumn, respectively.
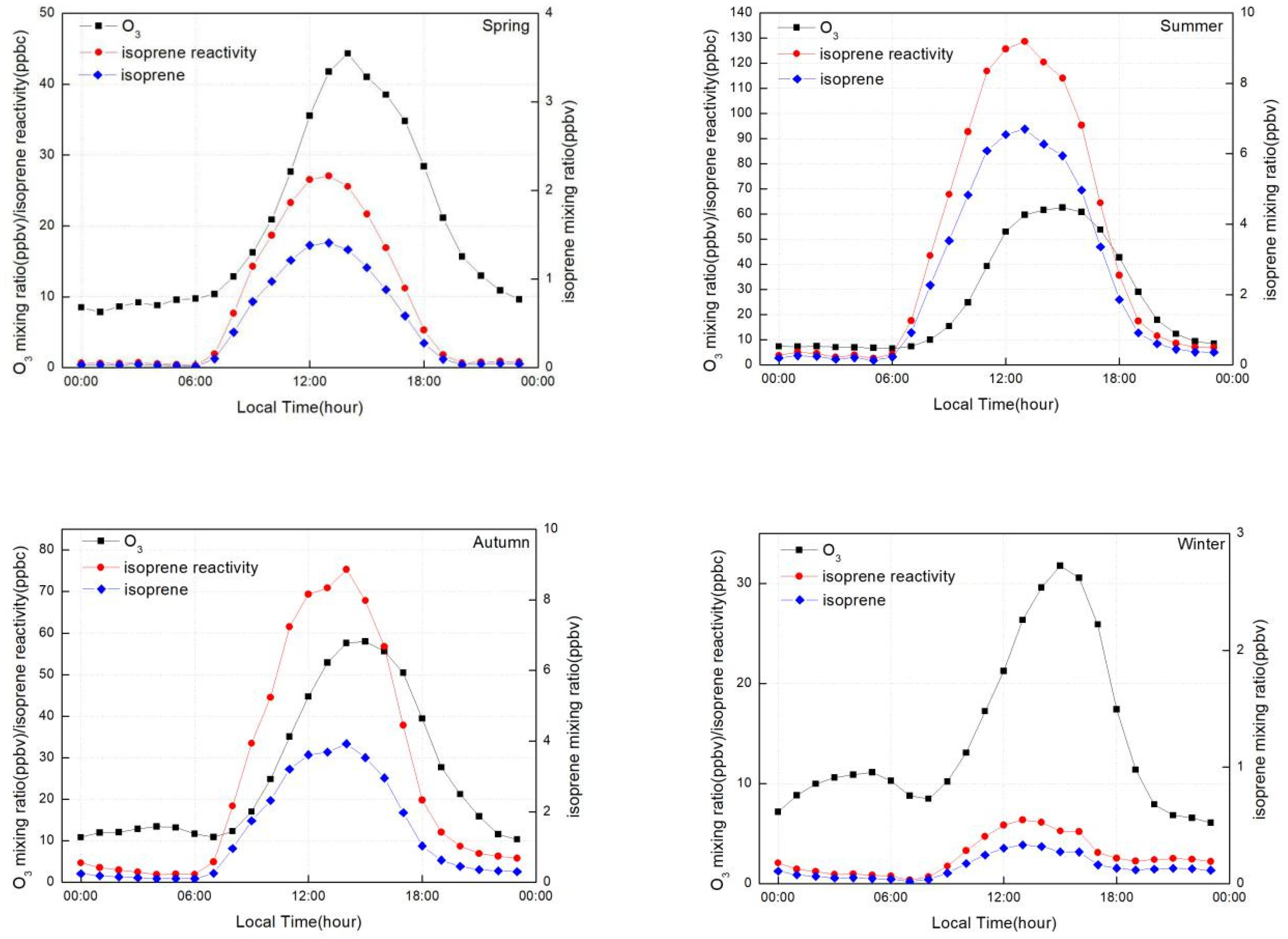
- The high (low) levels of isoprene could shape the strong (weak) and broad (sharp) peaks in the O3 profile.
1. Introduction
2. Methodology
2.1. Site Description
2.2. Instrumentation
2.3. Data Analysis
2.3.1. The Effect of NMHCs on O3 Formation
2.3.2. Assessment of Biogenic and Anthropogenic Effects on Isoprene
3. Results and Discussion
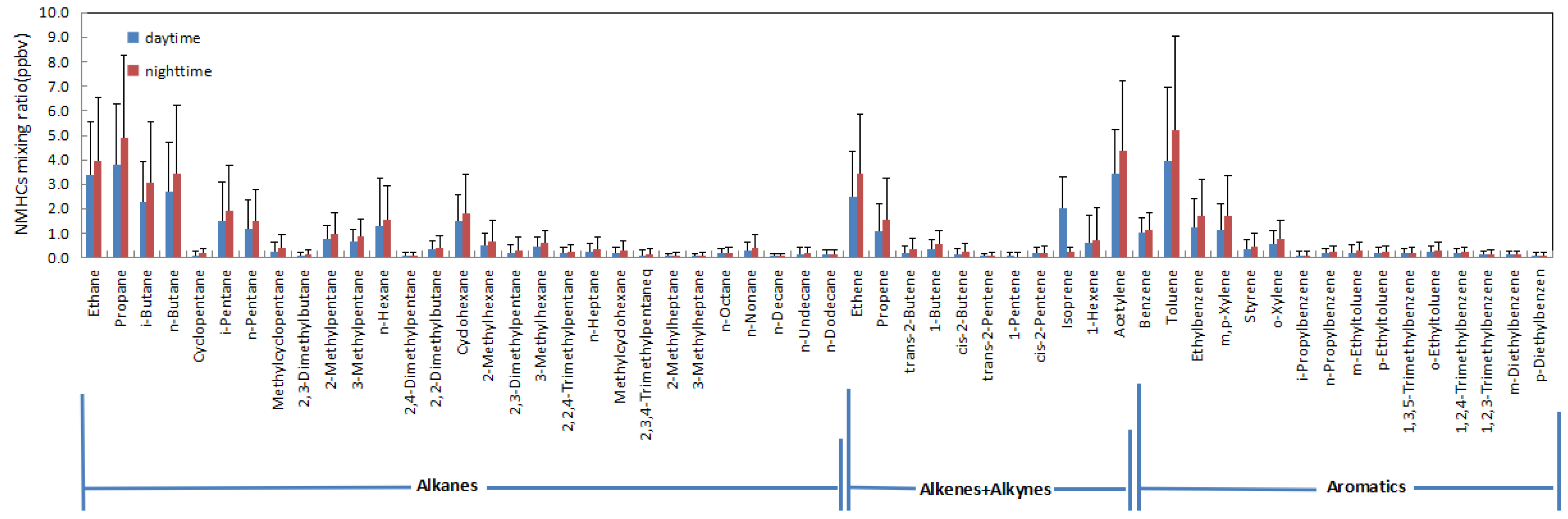
3.1. Overall Characteristics of NMHCs and Their Relative Potential to O3 Formation
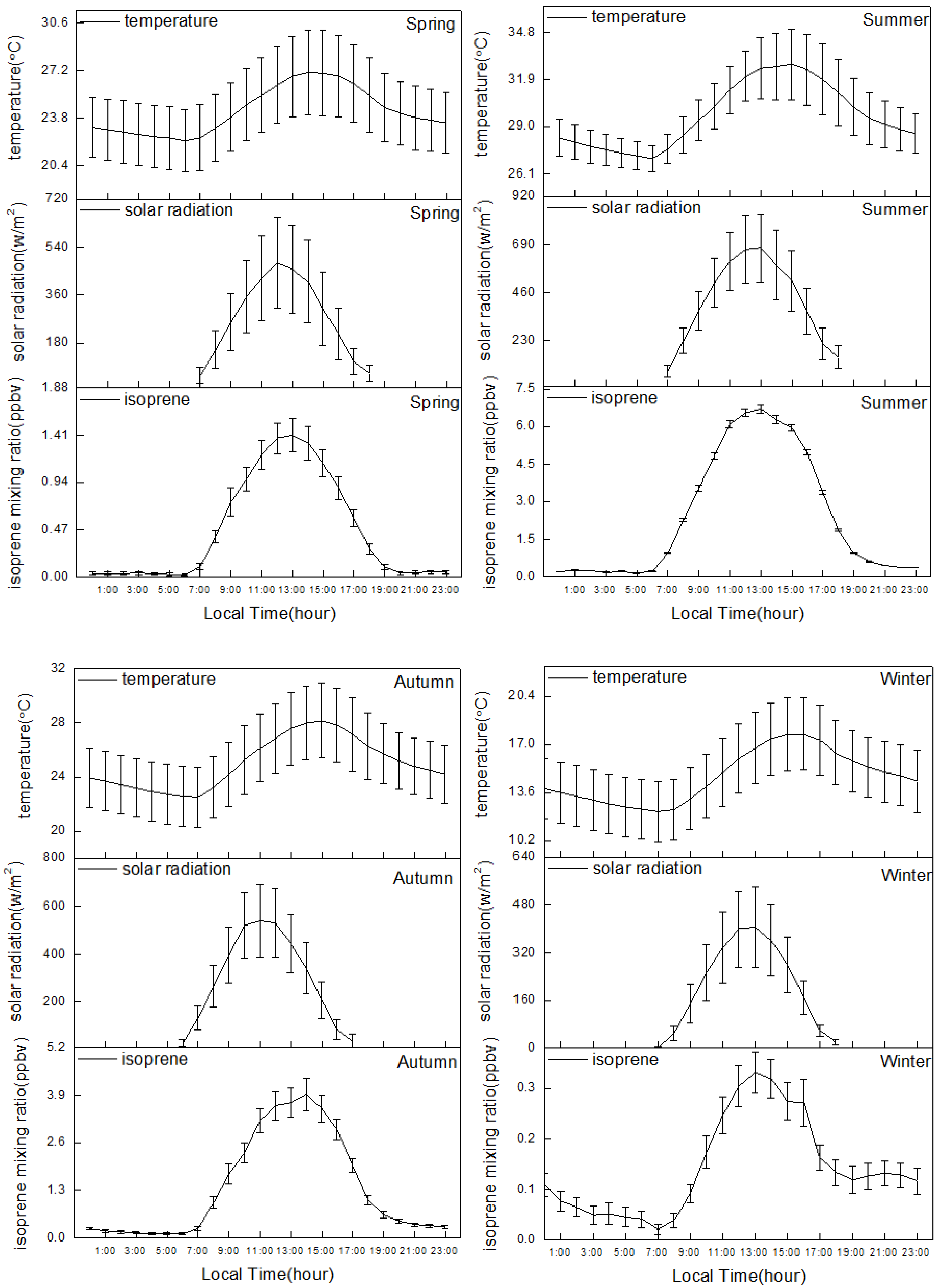
3.2. Biogenic and Anthropogenic Effects on Isoprene
3.3. Effect of Isoprene on Shaping the O3 Peak Profile
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Wang, T.; Wei, X.L.; Ding, A.J.; Poon, C.N.; Lam, K.S.; Li, Y.S.; Chan, L.Y.; Anson, M. Increasing surface ozone concentrations in the background atmosphere of Southern China, 1994–2007. Atmos. Chem. Phys. 2009, 9, 6217–6227. [Google Scholar] [CrossRef]
- Zheng, J.Y.; Zhong, L.J.; Wang, T.; Louie, P.K.K.; Li, Z.C. Ground-level ozone in the Pearl River Delta region: Analysis of data from a recently established regional air quality monitoring network. Atmos. Environ. 2010, 44, 814–823. [Google Scholar] [CrossRef]
- Zou, Y.; Charlesworth, E.; Yin, C.Q.; Yan, X.L.; Deng, X.J.; Li, F. The weekday/weekend ozone differences induced by the emissions change during summer and autumn in Guangzhou, China. Atmos. Environ. 2019, 199, 114–126. [Google Scholar] [CrossRef]
- Ran, L.; Zhao, C.S.; Xu, W.Y.; Lu, X.Q.; Han, M.; Lin, W.L.; Yan, P.; Xu, X.B.; Deng, Z.Z.; Ma, N.; et al. VOC reactivity and its effect on ozone production during the HaChi summer campaign. Atmos. Chem. Phys. 2011, 11, 4657–4667. [Google Scholar] [CrossRef]
- Zhong, L.J.; Louie, P.K.K.; Zheng, J.Y.; Yuan, Z.B.; Yue, D.L.; Ho, J.W.K.; Lau, A.K.H. Science-policy interplay: Air quality management in the Pearl River Delta region and Hong Kong. Atmos. Environ. 2013, 76, 3–10. [Google Scholar] [CrossRef]
- Wang, N.; Lyu, X.P.; Deng, X.J.; Guo, H.; Deng, T.; Li, Y.; Yin, C.Q.; Li, F.; Wang, S.Q. Assessment of regional air quality resulting from emission control in the Pearl River Delta region, southern China. Sci. Total Environ. 2016, 573, 1554–1565. [Google Scholar] [CrossRef]
- Li, J.F.; Lu, K.D.; Lv, W.; Li, J.; Zhong, L.J.; Ou, Y.B.; Cheng, D.H.; Huang, X.; Zhang, Y.H. Fast increasing of surface ozone concentrations in Pearl River Delta characterized by a regional air quality monitoring network during 2006–2011. J. Environ. Sci. 2014, 26, 23–26. [Google Scholar] [CrossRef]
- Wang, T.; Nie, W.; Gao, J.; Xue, L.K.; Gao, X.M.; Wang, X.F.; Qiu, J.; Poon, C.N.; Meinardi, S.; Blake, D.; et al. Air quality during the 2008 Beijing Olympics: Secondary pollutants and regional impact. Atmos. Chem. Phys. 2010, 10, 7603–7615. [Google Scholar] [CrossRef]
- Chameides, W.L.; Lindsay, R.W.; Richardson, J.; Kiang, C.S. The role of biogenic hydrocarbons in urban photochemical smog: Atlanta as a case study. Science. 1988, 241, 1473–1475. [Google Scholar] [CrossRef]
- Wagner, P.; Kuttler, W. Biogenic and anthropogenic isoprene in the near-surface urban atmosphere-Acase study in Essen, Germany. Sci. Total Environ. 2014, 475, 104–115. [Google Scholar] [CrossRef]
- Carter, W.P.L. Development of ozone reactivity scales for volatile organic compounds. J. Air Waste Manag. Assoc. 1994, 44, 881–899. [Google Scholar] [CrossRef]
- Lee, B.S.; Wang, J.L. Concentration variation of isoprene and its implications for peak ozone concentration. Atmos. Environ. 2006, 40, 5486–5495. [Google Scholar] [CrossRef]
- Guenther, A.; Karl, T.; Harley, P.; Wiedinmyer, C.; Palmer, P.I.; Geron, C. Estimates of global terrestrial isoprene emissions using MEGAN (Model of Emissions of Gases and Aerosols from Nature). Atmos. Chem. Phys. 2006, 6, 3181–3210. [Google Scholar] [CrossRef]
- Borbon, A.; Fontaine, H.; Veillerot, M.; Locoge, N.; Galloo, J.C.; Guillermo, R. An investigation into the traffic-related fraction of isoprene at an urban location. Atmos. Environ. 2001, 35, 3749–3760. [Google Scholar] [CrossRef]
- Wang, J.L.; Chew, C.; Chang, C.Y.; Liao, W.C.; Lung, C.S.C.; Chen, W.N.; Lee, P.J.; Lin, P.H.; Chang, C.C. Biogenic isoprene in subtropical urban settings and implications for air quality. Atmos. Environ. 2013, 79, 369–379. [Google Scholar]
- Reimann, S.; Pierluigi, C.; Hofer, P. The anthropogenic fraction contribution to isoprene concentrations in a rural atmosphere. Atmos. Environ. 2000, 34, 109–115. [Google Scholar] [CrossRef]
- Ran, L.; Zhao, C.; Geng, F.; Tie, X.; Tang, X.; Peng, L.; Zhou, G.; Yu, Q.; Xu, J.; Guenther, A. Ozone photochemical production in urban Shanghai, China: Analysis based on ground level observations. J. Geophys. Res. 2009, 114. [Google Scholar] [CrossRef]
- Deng, T.; Wu, D.; Deng, X.J.; Tan, H.B.; Li, F.; Liao, B.T. A vertical sounding of severe haze process in Guangzhou area. Sci. China. Earth Sci. 2014, 57, 2650–2656. [Google Scholar] [CrossRef]
- Liu, L.; Tan, H.B.; Fan, S.J.; Cai, M.F.; Xu, H.B.; Lei, F.; Chan, P.W. Influence of aerosol hygroscopicity and mixing state on aerosol optical properties in the Pearl River Delta region, China. Sci. Total Environ. 2018, 627, 1560–1571. [Google Scholar] [CrossRef]
- Zou, Y.; Deng, X.J.; Zhu, D.; Gong, D.C.; Wang, H.; Li, F.; Tan, H.B.; Deng, T.; Mai, B.R.; Liu, X.T.; et al. Characteristics of 1 year of observational data of VOCs, NOx and O3 at a suburban site in Guangzhou, China. Atmos. Chem. Phys. 2015, 15, 6625–6636. [Google Scholar] [CrossRef]
- Tan, H.B.; Liu, L.; Fan, S.J.; Li, F.; Yin, Y.; Cai, M.F.; Chan, P.W. Aerosol optical properties and mixing state of black carbon in the Pearl River Delta, China. Atmos. Environ. 2016, 131, 196–208. [Google Scholar] [CrossRef]
- Main, H.H.; Roberts, P.T.; Hurwitt, S.B. Validation of PAMS VOC Data in the Mid-Atlantic Region. Report Prepared for MARAMA, Baltimore, MD. Sonoma Technology, Inc., Petaluma, 1999, CA STI-998481-1835-FR. Available online: http://www.marama.org/publications_folder/Validation_of_PAMS_Report_feb1999.pdf (accessed on 20 October 2012).
- An, J.L.; Zhu, B.; Wang, H.L.; Li, Y.Y.; Lin, X.; Yang, H. Characteristics and source apportionment of VOCs measured in an industrial area of Nanjing, Yangtze River Delta, China. Atmos. Environ. 2014, 97, 206–214. [Google Scholar] [CrossRef]
- Dodge, M.C. Combined effects of organic reactivity and NMHC/NOx ratio on photochemical oxidant formation—A modeling study. Atmos. Environ. 1984, 18, 1657–1665. [Google Scholar] [CrossRef]
- Atkinson, R.; Arey, J. Atmospheric degradation of volatile organic compounds. Chem. Rev. 2003, 103, 4605–4638. [Google Scholar] [CrossRef]
- Chang, C.C.; Shao, M.; Chou, C.C.K.; Liu, S.C.; Wang, J.L.; Lee, K.Z.; Lai, C.H.; Zhu, T.; Lin, P.H. Biogenic isoprene and implications for oxidant levels in Beijing during the 2008 Olympic Games. Atmos. Chem. Phys. Discuss. 2013, 13, 25939–25967. [Google Scholar] [CrossRef]
- Kinoyama, M.; Nitta, H.; Watanabe, A.; Ueda, H. Acetone and isoprene concentrations in exhaled breath in healthy subjects. J. Health Sci. 2008, 54, 471–477. [Google Scholar] [CrossRef]
- Kushch, I.; Arendacká, B.; Štolc, S.; Mochalski, P.; Filipiak, W.; Schwarz, K.; Schwentner, L.; Schmid, A.; Dzien, A.; Lechleitner, M.; et al. Breath isoprene—aspects of normal physiology related to age, gender and cholesterol profile as determined in a proton transfer reaction mass spectrometry study. Clin. Chem. Lab. Med. 2008, 46, 1011–1018. [Google Scholar] [CrossRef]
- Zheng, J.Y.; Shao, M.; Che, W.W.; Zhang, L.J.; Zhong, L.J.; Zhang, Y.H.; Streets, D. Speciated VOC emission inventory and spatial patterns of ozone formation potential in the Pearl River Delta, China. Environ. Sci. Technol. 2009, 43, 8580–8586. [Google Scholar] [CrossRef] [PubMed]
- Guo, H.; Cheng, H.R.; Ling, Z.H.; Louie, P.K.K.; Ayoko, G.A. Which emission sources are responsible for the volatile organic compounds in the atmosphere of Pearl River Delta? J. Hazard. Mater. 2011, 188, 116–124. [Google Scholar] [CrossRef]
- Legreid, G.; Lööv, J.B.; Staehelin, J.; Hüglin, C.; Hill, M.; Buchmann, B.; Prevot, A.S.H.; Reimann, S. Oxygenated Volatile Organic Compounds (OVOCs) at an urban background site in Zürich (Europe): seasonal variation and source allocation. Atmos. Environ. 2007, 41, 8409–8423. [Google Scholar] [CrossRef]
- Hellén, H.; Tykkä, T.; Hakola, H. Importance of monoterpenes and isoprene inurban air in northern Europe. Atmos. Environ. 2012, 59, 59–66. [Google Scholar] [CrossRef]
- Lee, J.D.; Lewis, A.C.; Monks, P.S.; Jacob, M.; Hamilton, J.F.; Hopkins, J.R.; Watson, N.M.; Saxton, J.E.; Ennis, C.; Carpenter, L.J.; et al. Ozone photochemistry and elevated isoprene during the UK heatwave of August 2003. Atmos. Environ. 2003, 40, 7598–7613. [Google Scholar] [CrossRef]
- Khan, M.A.H.; Schlich, B.L.; Jenkin, M.E.; Shallcross, B.M.A.; Moseley, M.; Waler, C.; Morris, W.C.; Derwent, R.G.; Percival, C.J.; Shallcross, D.E. A two-decade anthropogenic and biogenic isoprene emissions study in a London urban background and a London urban traffic site. Atmosphere 2018, 9, 387. [Google Scholar] [CrossRef]
- Li, N.; He, Q.Y.; Greenberg, J.; Guenther, A.; Li, J.Y.; Cao, J.J.; Wang, J.; Liao, H.; Wang, Q.Y.; Zhang, Q. Impacts of biogenic and anthropogenic emissions on summertime ozone formation in the Guangzhou Basin, China. Atmos. Chem. Phys. 2018, 18, 7489–7507. [Google Scholar] [CrossRef]
- Tang, J.H.; Chan, L.Y.; Chang, C.C.; Liu, S.; Li, Y.S. Characteristics and sources of non-methane hydrocar-bons in background atmospheres of eastern, southwestern, and southern China. J. Geophys. Res.-Atmos. 2009, 114, D03304. [Google Scholar] [CrossRef]
- Park, C.; Schade, G.; Boedeker, I. Characteristics of the flux of isoprene and its oxidation products in an urban area. J. Geophys. Res. 2011, 116, D21303. [Google Scholar] [CrossRef]
- Shao, M.; Czapiewski, K.V.; Heiden, A.C.; Kobel, K.; Komenda, M.; Koppmann, R.; Wildt, J. Volatile organic compound emissions from Scots pine: Mechanisms and description by algorithms. J. Geophys. Res. 2001, 106, 20483–20491. [Google Scholar] [CrossRef]
- Ling, Z.H.; Guo, H.; Cheng, H.R.; Yu, Y.F. Sources of ambient volatile organic compounds and their contributions to photochemical ozone formation at a site in the Pearl River Delta, southern China. Environ. Pollut. 2011, 159, 2310–2319. [Google Scholar] [CrossRef]
- Castellanos, P.; Stehr, J.W.; Dickerson, R.R.; Ehrman, S.H. The sensitivity of modeled ozone to the temporal distribution of point, area, and mobile source emissions in the eastern United States. Atmos. Environ. 2009, 43, 4603–4611. [Google Scholar] [CrossRef]

| Season | Meteorological Element | Minimum | Maximum | Mean | Median |
|---|---|---|---|---|---|
| Spring (March, April, May) | Temperature (°C) Solar Radiation (w/m2) | 9.4 0.1 | 35.5 886.5 | 22.6 271.2 | 22.9 199.9 |
| Summer (June, July, August) | Temperature (°C) Solar Radiation (w/m2) | 23.9 4.1 | 37.1 952.5 | 29.4 415.1 | 28.8 381.3 |
| Autumn (September, October, November) | Temperature (°C) Solar Radiation (w/m2) | 16.2 0.3 | 35.3 901.0 | 25.2 294.4 | 25.2 238.1 |
| Winter (December, January, February) | Temperature (°C) Solar Radiation (w/m2) | 4.7 0.1 | 26.9 693.5 | 14.2 206.8 | 14.1 135.3 |
| Compound | kaOH × 1012 | MIR b | Mixing Ratio (ppbv) (Parts Per Billion by Volume) |
|---|---|---|---|
| Alkanes | |||
| Ethane | 0.27 | 0.25 | 3.66 |
| Propane | 1.15 | 0.46 | 4.34 |
| i-Butane | 2.34 | 1.18 | 2.67 |
| n-Butane | 2.54 | 1.08 | 3.07 |
| Cyclopentane | 5.16 | 2.24 | 0.15 |
| i-Pentane | 3.90 | 1.36 | 1.72 |
| n-Pentane | 3.94 | 1.22 | 1.37 |
| Methylcyclopentane | 5.10 | 1.46 | 0.32 |
| 2,3-Dimethylbutane | 6.30 | 1.07 | 0.13 |
| 2,2-Dimethylbutane | 2.32 | 0.82 | 0.37 |
| 2-Methylpentane | 5.60 | 1.40 | 0.88 |
| 3-Methylpentane | 5.70 | 1.69 | 0.75 |
| n-Hexane | 5.60 | 1.14 | 1.43 |
| 2,4-Dimethylpentane | 5.70 | 1.11 | 0.37 |
| Cyclohexane | 7.49 | 1.14 | 1.65 |
| 2-Methylhexane | 6.90 | 1.09 | 0.58 |
| 2,3-Dimethylpentane | 5.10 | 1.25 | 0.26 |
| 3-Methylhexane | 5.10 | 1.50 | 0.52 |
| 2,2,4-Trimethylpentane | 3.68 | 1.20 | 0.22 |
| n-Heptane | 7.15 | 0.97 | 0.32 |
| Methylcyclohexane | 10.4 | 1.56 | 0.26 |
| 2,3,4-Trimethylpentane | 7.00 | 0.97 | 0.12 |
| 2-Methylheptane | 8.30 | 1.12 | 0.08 |
| 3-Methylheptane | 8.60 | 0.80 | 0.08 |
| n-Octane | 8.68 | 0.68 | 0.19 |
| n-Nonane | 10.20 | 0.59 | 0.35 |
| n-Decane | 11.60 | 0.52 | 0.03 |
| n-Undecane | 13.20 | 0.47 | 0.17 |
| n-Dodecane | 14.20 | 0.38 | 0.14 |
| Alkenes | |||
| Ethene | 8.50 | 7.40 | 2.99 |
| Propene | 26.30 | 11.57 | 1.32 |
| trans-2-Butene | 64.00 | 15.20 | 0.28 |
| 1-Butene | 31.40 | 9.57 | 0.44 |
| cis-2-Butene | 56.40 | 14.26 | 0.22 |
| trans-2-Pentene | 67.00 | 10.47 | 0.03 |
| 1-Pentene | 31.40 | 7.07 | 0.05 |
| cis-2-Pentene | 65.00 | 10.28 | 0.19 |
| Isoprene | 101.00 | 10.48 | 1.14 |
| 1-Hexene | – | – | 0.67 |
| Alkyne | |||
| Ethyne | 0.9 | 0.5 | 3.90 |
| Aromatics | |||
| Benzene | 1.23 | 0.42 | 1.09 |
| Toluene | 5.96 | 3.93 | 4.59 |
| Ethylbenzene | 6.96 | 2.96 | 1.48 |
| m,p-Xylene | 20.50 | 8.54 | 1.41 |
| Styrene | 58.00 | 1.66 | 0.41 |
| o-Xylene | 13.60 | 7.58 | 0.66 |
| i-Propylbenzene | 6.60 | 2.45 | 0.10 |
| n-Propylbenzene | 5.70 | 1.96 | 0.23 |
| m-Ethyltoluene | 18.60 | 7.39 | 0.25 |
| p-Ethyltoluene | 11.80 | 4.39 | 0.21 |
| 1,3,5-Trimethylbenzene | 56.70 | 11.75 | 0.21 |
| o-Ethyltoluene | 11.90 | 5.54 | 0.27 |
| 1,2,4-Trimethylbenzene | 32.50 | 8.83 | 0.21 |
| 1,2,3-Trimethylbenzene | 32.70 | 11.94 | 0.15 |
| m-Diethylbenzene | 15.00 | 7.08 | 0.12 |
| p-Diethylbenzene | 10.00 | 4.39 | 0.11 |
| Site | Location | Year | Season | Mean Mixing Ratio or Range (ppbv) | Reference |
|---|---|---|---|---|---|
| National Taiwan University (Urban Site) | Taipei (25° 00′ N/121° 34′ E) | 2011 | Summer | 0.72 (daily) | [15] |
| Hualin Weather Station (Rural Site) | Northern Taiwan (24° 53′ N/121° 34′ E) | 2011 | Summer | 0.70 (daily) | [15] |
| Kasernenhof (Urban Background Site) | Zurich (47° 22′ N/8° 31′ E) | 2005 | Summer | 0.16 (daily) | [31] |
| Rooftop Site (Urban Site) | Essen (51° 27′ N/7° 00′ E) | 2011 | Summer | 0.09 (daily) | [10] |
| SMEARIII (Urban Background Site) | Helsinki (60° 12′ N/24° 58′ E) | 2011 | Summer | 0.11 (daily) | [32] |
| Writtle College (Rural Site) | Essex (51° 44′ N/0° 25′ E) | 2003 | Summer | 0.13 (daytime) | [33] |
| Eltham (Suburban Background Site) | London (51° 27′ N/0° 03′ E) | 1997–2017 | Summer | 0.13 (daily) | [34] |
| Marylebone Road (Urban Site) | London (51°27′ N/0°03′ E) | 1997–2017 | Summer | 0.15 (daily) | [34] |
| Six Sites (Rural Sites) | Xi an (32° 42′–33° 47′ N/107°44′–109°24′ E) | 2011 | Summer | 0.1–3.8 (daytime) | [35] |
| Jianfeng Mountain (Remote Site) | HainanProvince (18° 40′ N, 108° 49′ E) | 2004 | Spring | 0.55 (daily) | [36] |
| Tengchong Mountain (Remote Site) | Yunnan Province (24° 570 N, 98° 29′ E) | 2004 | Spring | 0.08 (daily) | [36] |
| Lin’an Station (Rural Site) | Zhejiang Province (30° 30′ N, 119° 75′ E) | 2004 | Spring | 0.40 (daily) | [36] |
| Tengyue Town (Urban Site) | Yunnan Province (25° 01′ N, 98° 30′ E) | 2004 | Spring | 0.13 (daily) | [36] |
| Peking University (Urban Site) | Beijing (39° 59′ N/116° 18′ E) | 2008 | Summer | 0.45 (daily) | [26] |
| 3–4 km North of Downtown Houston | Houston (29° 47′ N/95° 21′ W) | 2008 | Summer | 0.63 (daily) | [37] |
| GPACS (Suburban Site) | Guangzhou (23° 00′ N/113° 21′ E) | 2011 | Spring | 0.40 (daily) | This study |
| GPACS (Suburban Site) | Guangzhou (23° 00′ N/113° 21′ E) | 2011 | Summer | 2.20 (daily) | This study |
| GPACS (Suburban Site) | Guangzhou (23° 00′ N/113° 21′ E) | 2011 | Autumn | 1.40 (daily) | This study |
| GPACS (Suburban Site) | Guangzhou (23° 00′ N/113° 21′ E) | 2011 | Winter | 0.13 (daily) | This study |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zou, Y.; Deng, X.J.; Deng, T.; Yin, C.Q.; Li, F. One-Year Characterization and Reactivity of Isoprene and Its Impact on Surface Ozone Formation at A Suburban Site in Guangzhou, China. Atmosphere 2019, 10, 201. https://doi.org/10.3390/atmos10040201
Zou Y, Deng XJ, Deng T, Yin CQ, Li F. One-Year Characterization and Reactivity of Isoprene and Its Impact on Surface Ozone Formation at A Suburban Site in Guangzhou, China. Atmosphere. 2019; 10(4):201. https://doi.org/10.3390/atmos10040201
Chicago/Turabian StyleZou, Yu, Xue Jiao Deng, Tao Deng, Chang Qin Yin, and Fei Li. 2019. "One-Year Characterization and Reactivity of Isoprene and Its Impact on Surface Ozone Formation at A Suburban Site in Guangzhou, China" Atmosphere 10, no. 4: 201. https://doi.org/10.3390/atmos10040201
APA StyleZou, Y., Deng, X. J., Deng, T., Yin, C. Q., & Li, F. (2019). One-Year Characterization and Reactivity of Isoprene and Its Impact on Surface Ozone Formation at A Suburban Site in Guangzhou, China. Atmosphere, 10(4), 201. https://doi.org/10.3390/atmos10040201





