Description of Genetic Variants in BRCA Genes in Mexican Patients with Ovarian Cancer: A First Step towards Implementing Personalized Medicine
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Population and DNA Extraction
2.2. DNA Sequencing
2.3. Variant Calling, Filtering, and Annotation
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ferlay, J.; Soerjomataram, I.; Dikshit, R.; Eser, S.; Mathers, C.; Rebelo, M.; Parkin, D.M.; Forman, D.; Bray, F. Cancer incidence and mortality worldwide: Sources, methods and major patterns in GLOBOCAN 2012. Int. J. Cancer 2014, 136. [Google Scholar] [CrossRef] [PubMed]
- Malvezzi, M.; Carioli, G.; Rodriguez, T.; Negri, E.; La Vecchia, C. Global trends and predictions in ovarian cancer mortality. Ann. Oncol. 2016, 27, 2017–2025. [Google Scholar] [CrossRef] [PubMed]
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics, 2018. CA Cancer J. Clin. 2018, 68, 7–30. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Xu, Y.; Yu, C.-X. Proteomic analysis of serum of women with elevated CA-125 to differentiate malignant from benign ovarian tumors. Asian Pac. J. Cancer Prev. 2012, 13, 3265–3270. [Google Scholar] [CrossRef] [PubMed]
- Nossov, V.; Amneus, M.; Su, F.; Lang, J.; Janco, J.M.; Reddy, S.T.; Farias-Eisner, R. The early detection of ovarian cancer: From traditional methods to proteomics. Can we really do better than serum CA-125? Am. J. Obstet. Gynecol. 2008, 199, 215–223. [Google Scholar] [CrossRef] [PubMed]
- Barroilhet, L.; Vitonis, A.; Shipp, T.; Muto, M.; Benacerraf, B. Sonographic predictors of ovarian malignancy. J. Clin.Ultrasound 2013, 41, 269–274. [Google Scholar] [CrossRef] [PubMed]
- Jacobs, I.J.; Menon, U.; Ryan, A.; Gentry-Maharaj, A.; Burnell, M.; Kalsi, J.K.; Amso, N.N.; Apostolidou, S.; Benjamin, E.; Cruickshank, D.; et al. Ovarian cancer screening and mortality in the UK Collaborative Trial of Ovarian Cancer Screening (UKCTOCS): A randomised controlled trial. Lancet 2016, 387, 945–956. [Google Scholar] [CrossRef]
- Committee on Gynecologic Practice. Committee opinion No. 716: The role of the obstetrician-gynecologist in the early detection of epithelial ovarian cancer in women at average risk. Obstet. Gynecol. 2017, 130, e146–e149. [Google Scholar]
- Szecsi, P.B.; Andersen, M.R.; Bjorngaard, B.; Hedengran, K.K.; Stender, S. Cancer antigen 125 after delivery in women with a normal pregnancy: A prospective cohort study. Acta Obstet. Gynecol. Scand. 2014, 93, 1295–1301. [Google Scholar] [CrossRef] [PubMed]
- Mozas, J.; Castilla, J.A.; Jimena, P.; Gil, T.; Acebal, M.; Herruzo, A.J. Serum CA-125 in the diagnosis of acute pelvic inflammatory disease. Int. J. Gynaecol. Obstet. 1994, 44, 53–57. [Google Scholar] [CrossRef]
- Brun, R.; Hutmacher, J.; Fink, D.; Imesch, P. Erroneously suspected ovarian cancer in a 38-year-old woman with pelvic inflammatory disease and chlamydia. Case Rep. Obstet. Gynecol. 2017, 2017, 2514613. [Google Scholar] [CrossRef] [PubMed]
- Bai, H.; Sha, G.; Xiao, M.; Gao, H.; Cao, D.; Yang, J.; Chen, J.; Wang, Y.; Zhang, Z.; Shen, K. The prognostic value of pretreatment CA-125 levels and CA-125 normalization in ovarian clear cell carcinoma: A two-academic-institute study. Oncotarget 2016, 7, 15566–15576. [Google Scholar] [CrossRef] [PubMed]
- Deeks, E.D. Olaparib: First global approval. Drugs 2015, 75, 231–240. [Google Scholar] [CrossRef] [PubMed]
- Varga, D.; Deniz, M.; Schwentner, L.; Wiesmuller, L. Ovarian cancer: In search of better marker systems based on DNA repair defects. Int. J. Mol. Sci. 2013, 14, 640–673. [Google Scholar] [CrossRef] [PubMed]
- Tan, D.S.; Kaye, S.B. Chemotherapy for patients with BRCA1 and BRCA2-mutated ovarian cancer: Same or different? Am. Soc. Clin. Oncol. Educ. Book 2015, 35, 114–121. [Google Scholar] [CrossRef] [PubMed]
- Plummer, R. Poly(ADP-ribose) polymerase inhibition: A new direction for BRCA and triple-negative breast cancer? Breast Cancer Res. 2011, 13, 218. [Google Scholar] [CrossRef] [PubMed]
- Gottipati, P.; Vischioni, B.; Schultz, N.; Solomons, J.; Bryant, H.E.; Djureinovic, T.; Issaeva, N.; Sleeth, K.; Sharma, R.A.; Helleday, T. Poly(ADP-ribose) polymerase is hyperactivated in homologous recombination-defective cells. Cancer Res. 2010, 70, 5389–5398. [Google Scholar] [CrossRef] [PubMed]
- Domchek, S.M.; Aghajanian, C.; Shapira-Frommer, R.; Schmutzler, R.K.; Audeh, M.W.; Friedlander, M.; Balmana, J.; Mitchell, G.; Fried, G.; Stemmer, S.M.; et al. Efficacy and safety of olaparib monotherapy in germline BRCA1/2 mutation carriers with advanced ovarian cancer and three or more lines of prior therapy. Gynecol. Oncol. 2016, 140, 199–203. [Google Scholar] [CrossRef] [PubMed]
- Rafii, S.; Gourley, C.; Kumar, R.; Geuna, E.; Ang, J.E.; Rye, T.; Chen, L.-M.; Shapira-Frommer, R.; Friedlander, M.; Matulonis, U.; et al. Baseline clinical predictors of antitumor response to the PARP inhibitor olaparib in germline BRCA1/2 mutated patients with advanced ovarian cancer. Oncotarget 2017, 8, 47154–47160. [Google Scholar] [CrossRef] [PubMed]
- Tucker, T.; Marra, M.; Friedman, J.M. Massively parallel sequencing: The next big thing in genetic medicine. Am. J. Hum. Genet. 2009, 85, 142–154. [Google Scholar] [CrossRef] [PubMed]
- Quail, M.A.; Smith, M.; Coupland, P.; Otto, T.D.; Harris, S.R.; Connor, T.R.; Bertoni, A.; Swerdlow, H.P.; Gu, Y. A tale of three next generation sequencing platforms: Comparison of Ion Torrent, Pacific Biosciences and Illumina Miseq sequencers. BMC Genom. 2012, 13, 341. [Google Scholar]
- The Cancer Genome Atlas Research Network. Integrated genomic analyses of ovarian carcinoma. Nature 2011, 474, 609–615. [Google Scholar]
- George, A.; Kaye, S.; Banerjee, S. Delivering widespread BRCA testing and PARP inhibition to patients with ovarian cancer. Nat. Rev. Clin. Oncol. 2017, 14, 284–296. [Google Scholar] [CrossRef] [PubMed]
- Bettegowda, C.; Sausen, M.; Leary, R.J.; Kinde, I.; Wang, Y.; Agrawal, N.; Bartlett, B.R.; Wang, H.; Luber, B.; Alani, R.M.; et al. Detection of circulating tumor DNA in early- and late-stage human malignancies. Sci. Transl. Med. 2014, 6, 3007094. [Google Scholar] [CrossRef] [PubMed]
- Alix-Panabieres, C.; Pantel, K. Circulating tumor cells: Liquid biopsy of cancer. Clin. Chem. 2013, 59, 110–118. [Google Scholar] [CrossRef] [PubMed]
- Diaz, L.A., Jr.; Bardelli, A. Liquid biopsies: Genotyping circulating tumor DNA. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2014, 32, 579–586. [Google Scholar] [CrossRef] [PubMed]
- Dean, M.; Boland, J.; Yeager, M.; Im, K.M.; Garland, L.; Rodriguez-Herrera, M.; Perez, M.; Mitchell, J.; Roberson, D.; Jones, K.; et al. Addressing health disparities in Hispanic breast cancer: Accurate and inexpensive sequencing of BRCA1 and BRCA2. GigaScience 2015, 4, 50. [Google Scholar] [PubMed]
- Robinson, J.T.; Thorvaldsdottir, H.; Winckler, W.; Guttman, M.; Lander, E.S.; Getz, G.; Mesirov, J.P. Integrative genomics viewer. Nat. Biotechnol. 2011, 29, 24–26. [Google Scholar] [CrossRef] [PubMed]
- Gallardo-Rincon, D.; Cantu-de-Leon, D.; Alanis-Lopez, P.; Alvarez-Avitia, M.A.; Banuelos-Flores, J.; Herbert-Nunez, G.S.; Onate-Ocana, L.F.; Perez-Montiel, M.D.; Rodriguez-Trejo, A.; Ruvalcaba-Limon, E.; et al. Third national ovarian consensus. 2011. Grupo de investigacion en cancer de ovario y tumores ginecologicos de Mexico “GICOM”. Rev. Investig. Clin. 2011, 63, 665–702. [Google Scholar]
- Lazcano-Ponce, E.; Palacio-Mejia, L.S.; Allen-Leigh, B.; Yunes-Diaz, E.; Alonso, P.; Schiavon, R.; Hernandez-Avila, M. Decreasing cervical cancer mortality in Mexico: Effect of papanicolaou coverage, birthrate, and the importance of diagnostic validity of cytology. Cancer Epidemiol. Biomark. Prev. 2008, 17, 2808–2817. [Google Scholar] [CrossRef] [PubMed]
- Chavarri-Guerra, Y.; Villarreal-Garza, C.; Liedke, P.E.; Knaul, F.; Mohar, A.; Finkelstein, D.M.; Goss, P.E. Breast cancer in Mexico: A growing challenge to health and the health system. Lancet Oncol. 2012, 13, e335–e343. [Google Scholar] [CrossRef]
- Landrum, M.J.; Lee, J.M.; Riley, G.R.; Jang, W.; Rubinstein, W.S.; Church, D.M.; Maglott, D.R. Clinvar: Public archive of relationships among sequence variation and human phenotype. Nucleic Acids Res. 2014, 42, D980–D985. [Google Scholar] [CrossRef] [PubMed]
- Forbes, S.A.; Bindal, N.; Bamford, S.; Cole, C.; Kok, C.Y.; Beare, D.; Jia, M.; Shepherd, R.; Leung, K.; Menzies, A.; et al. COSMIC: Mining complete cancer genomes in the Catalogue of Somatic Mutations in Cancer. Nucleic Acids Res. 2011, 39, D945–D950. [Google Scholar] [CrossRef] [PubMed]
- Ozols, R.F. Paclitaxel (Taxol)/carboplatin combination chemotherapy in the treatment of advanced ovarian cancer. Semin. Oncol. 2000, 27, 3–7. [Google Scholar] [PubMed]
- Cristea, M.; Han, E.; Salmon, L.; Morgan, R.J. Practical considerations in ovarian cancer chemotherapy. Ther. Adv. Med. Oncol. 2010, 2, 175–187. [Google Scholar] [CrossRef] [PubMed]
- Gelmon, K.A.; Tischkowitz, M.; Mackay, H.; Swenerton, K.; Robidoux, A.; Tonkin, K.; Hirte, H.; Huntsman, D.; Clemons, M.; Gilks, B.; et al. Olaparib in patients with recurrent high-grade serous or poorly differentiated ovarian carcinoma or triple-negative breast cancer: A phase 2, multicentre, open-label, non-randomised study. Lancet Oncol. 2011, 12, 852–861. [Google Scholar] [CrossRef]
- Ledermann, J.; Harter, P.; Gourley, C.; Friedlander, M.; Vergote, I.; Rustin, G.; Scott, C.; Meier, W.; Shapira-Frommer, R.; Safra, T.; et al. Olaparib maintenance therapy in platinum-sensitive relapsed ovarian cancer. N. Engl. J. Med. 2012, 366, 1382–1392. [Google Scholar] [CrossRef] [PubMed]
- Vaca-Paniagua, F.; Alvarez-Gomez, R.M.; Fragoso-Ontiveros, V.; Vidal-Millan, S.; Herrera, L.A.; Cantu, D.; Bargallo-Rocha, E.; Mohar, A.; Lopez-Camarillo, C.; Perez-Plasencia, C. Full-exon pyrosequencing screening of BRCA germline mutations in mexican women with inherited breast and ovarian cancer. PLoS ONE 2012, 7, 24. [Google Scholar] [CrossRef] [PubMed]
- Villarreal-Garza, C.; Alvarez-Gomez, R.M.; Perez-Plasencia, C.; Herrera, L.A.; Herzog, J.; Castillo, D.; Mohar, A.; Castro, C.; Gallardo, L.N.; Gallardo, D.; et al. Significant clinical impact of recurrent BRCA1 and BRCA2 mutations in Mexico. Cancer 2015, 121, 372–378. [Google Scholar] [CrossRef] [PubMed]
- Weitzel, J.N.; Lagos, V.I.; Herzog, J.S.; Judkins, T.; Hendrickson, B.; Ho, J.S.; Ricker, C.N.; Lowstuter, K.J.; Blazer, K.R.; Tomlinson, G.; et al. Evidence for common ancestral origin of a recurring BRCA1 genomic rearrangement identified in high-risk Hispanic families. Cancer Epidemiol. Biomark. Prev. 2007, 16, 1615–1620. [Google Scholar] [CrossRef] [PubMed]
- Weitzel, J.N.; Clague, J.; Martir-Negron, A.; Ogaz, R.; Herzog, J.; Ricker, C.; Jungbluth, C.; Cina, C.; Duncan, P.; Unzeitig, G.; et al. Prevalence and type of BRCA mutations in Hispanics undergoing genetic cancer risk assessment in the southwestern United States: A report from the clinical cancer genetics community research network. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2013, 31, 210–216. [Google Scholar] [CrossRef] [PubMed]
- Wallbillich, J.J.; Forde, B.; Havrilesky, L.J.; Cohn, D.E. A personalized paradigm in the treatment of platinum-resistant ovarian cancer—A cost utility analysis of genomic-based versus cytotoxic therapy. Gynecol. Oncol. 2016, 142, 144–149. [Google Scholar] [CrossRef] [PubMed]
- Silva-Zolezzi, I.; Hidalgo-Miranda, A.; Estrada-Gil, J.; Fernandez-Lopez, J.C.; Uribe-Figueroa, L.; Contreras, A.; Balam-Ortiz, E.; del Bosque-Plata, L.; Velazquez-Fernandez, D.; Lara, C.; et al. Analysis of genomic diversity in Mexican Mestizo populations to develop genomic medicine in Mexico. Proc. Natl. Acad. Sci. USA 2009, 106, 8611–8616. [Google Scholar] [CrossRef] [PubMed]
- Alcazar-Gonzalez, G.A.; Calderon-Garciduenas, A.L.; Garza-Rodriguez, M.L.; Rubio-Hernandez, G.; Escorza-Trevino, S.; Olano-Martin, E.; Cerda-Flores, R.M.; Castruita-Avila, A.L.; Gonzalez-Guerrero, J.F.; le Brun, S.; et al. Comparative study of polymorphism frequencies of the CYP2D6, CYP3A5, CYP2C8 and IL-10 genes in mexican and spanish women with breast cancer. Pharmacogenomics 2013, 14, 1583–1592. [Google Scholar] [CrossRef] [PubMed]
- Leon-Cachon, R.B.; Ascacio-Martinez, J.A.; Gamino-Pena, M.E.; Cerda-Flores, R.M.; Meester, I.; Gallardo-Blanco, H.L.; Gomez-Silva, M.; Pineyro-Garza, E.; Barrera-Saldana, H.A. A pharmacogenetic pilot study reveals MTHFR, DRD3, and MDR1 polymorphisms as biomarker candidates for slow atorvastatin metabolizers. BMC Cancer 2016, 16, 74. [Google Scholar] [CrossRef] [PubMed]
- Kanchi, K.L.; Johnson, K.J.; Lu, C.; McLellan, M.D.; Leiserson, M.D.; Wendl, M.C.; Zhang, Q.; Koboldt, D.C.; Xie, M.; Kandoth, C.; et al. Integrated analysis of germline and somatic variants in ovarian cancer. Nat. Commun. 2014, 5, 3156. [Google Scholar] [CrossRef] [PubMed]
- Gomez-Rueda, H.; Martinez-Ledesma, E.; Martinez-Torteya, A.; Palacios-Corona, R.; Trevino, V. Integration and comparison of different genomic data for outcome prediction in cancer. BioData Min. 2015, 8, 32. [Google Scholar] [CrossRef] [PubMed]

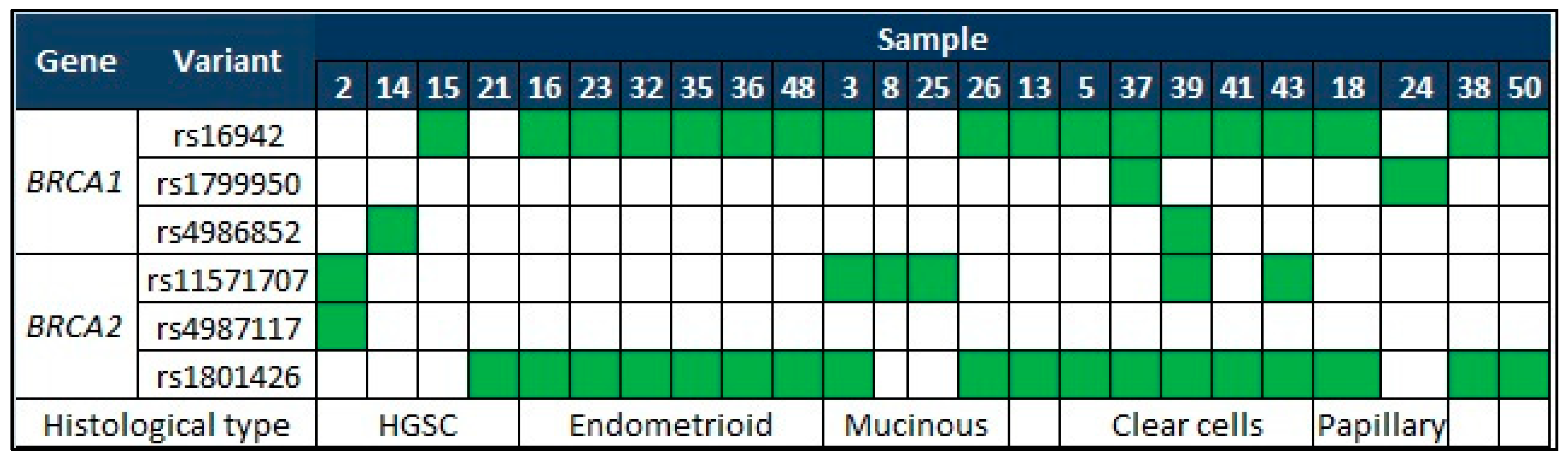
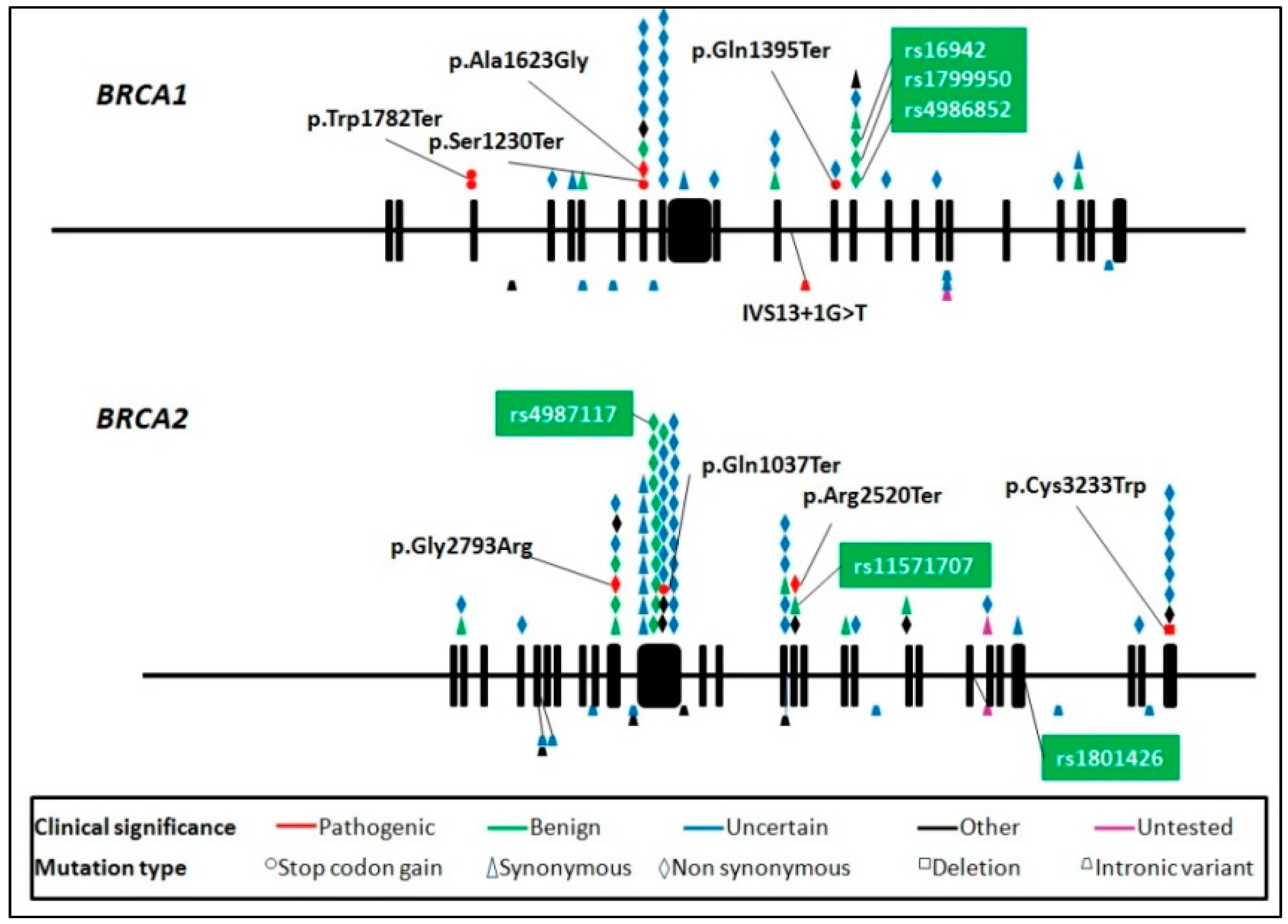
| Gene | Variants | HGVS | Amino Acid Change | Genomic Location (GHCh38) | Type |
|---|---|---|---|---|---|
| BRCA1 | rs80356862 | 4868C>G | A1623G | Chr17:43071046 | Missense variant |
| rs80358027 | c.4357+1G>A | IVS13+1G>A | Chr17:43082403 | Splice donor variant | |
| rs80357260 | c.4183C>T | Q1395* | Chr17:43090946 | Nonsense variant | |
| rs80357284 | c.5346G>A | W1782* | Chr17:43049181 | Nonsense variant | |
| rs80357219 | c.5345G>A | W1782* | Chr17:43049182 | Nonsense variant | |
| BRCA2 | rs80359082 | c.8377G>A | G2793R | Chr13:32370447 | Missense variant |
| rs80358557 | c.3109C>T | Q1037* | Chr13:32337464 | Nonsense variant | |
| rs80359775 | c.9699_9702delTATG | C3233Wfs | Chr13:32398212 - 32398215 | Frameshift variant | |
| rs80358981 | c.7558C>T | R2520* | Chr13:32356550 | Nonsense variant |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Delgado-Balderas, J.R.; Garza-Rodriguez, M.L.; Gomez-Macias, G.S.; Barboza-Quintana, A.; Barboza-Quintana, O.; Cerda-Flores, R.M.; Miranda-Maldonado, I.; Vazquez-Garcia, H.M.; Valdez-Chapa, L.D.; Antonio-Macedo, M.; et al. Description of Genetic Variants in BRCA Genes in Mexican Patients with Ovarian Cancer: A First Step towards Implementing Personalized Medicine. Genes 2018, 9, 349. https://doi.org/10.3390/genes9070349
Delgado-Balderas JR, Garza-Rodriguez ML, Gomez-Macias GS, Barboza-Quintana A, Barboza-Quintana O, Cerda-Flores RM, Miranda-Maldonado I, Vazquez-Garcia HM, Valdez-Chapa LD, Antonio-Macedo M, et al. Description of Genetic Variants in BRCA Genes in Mexican Patients with Ovarian Cancer: A First Step towards Implementing Personalized Medicine. Genes. 2018; 9(7):349. https://doi.org/10.3390/genes9070349
Chicago/Turabian StyleDelgado-Balderas, Jesus Rolando, Maria Lourdes Garza-Rodriguez, Gabriela Sofia Gomez-Macias, Alvaro Barboza-Quintana, Oralia Barboza-Quintana, Ricardo M. Cerda-Flores, Ivett Miranda-Maldonado, Hugo Mauricio Vazquez-Garcia, Lezmes Dionicio Valdez-Chapa, Mauro Antonio-Macedo, and et al. 2018. "Description of Genetic Variants in BRCA Genes in Mexican Patients with Ovarian Cancer: A First Step towards Implementing Personalized Medicine" Genes 9, no. 7: 349. https://doi.org/10.3390/genes9070349
APA StyleDelgado-Balderas, J. R., Garza-Rodriguez, M. L., Gomez-Macias, G. S., Barboza-Quintana, A., Barboza-Quintana, O., Cerda-Flores, R. M., Miranda-Maldonado, I., Vazquez-Garcia, H. M., Valdez-Chapa, L. D., Antonio-Macedo, M., Dean, M., & Barrera-Saldaña, H. A. (2018). Description of Genetic Variants in BRCA Genes in Mexican Patients with Ovarian Cancer: A First Step towards Implementing Personalized Medicine. Genes, 9(7), 349. https://doi.org/10.3390/genes9070349







