Emerging Opportunities for Synthetic Biology in Agriculture
Abstract
1. Synthetic Biology and the Primary Industries, Early Adoption of Disruptive Technology
2. Biosynthesis of High Value Plant Metabolites in Microorganisms
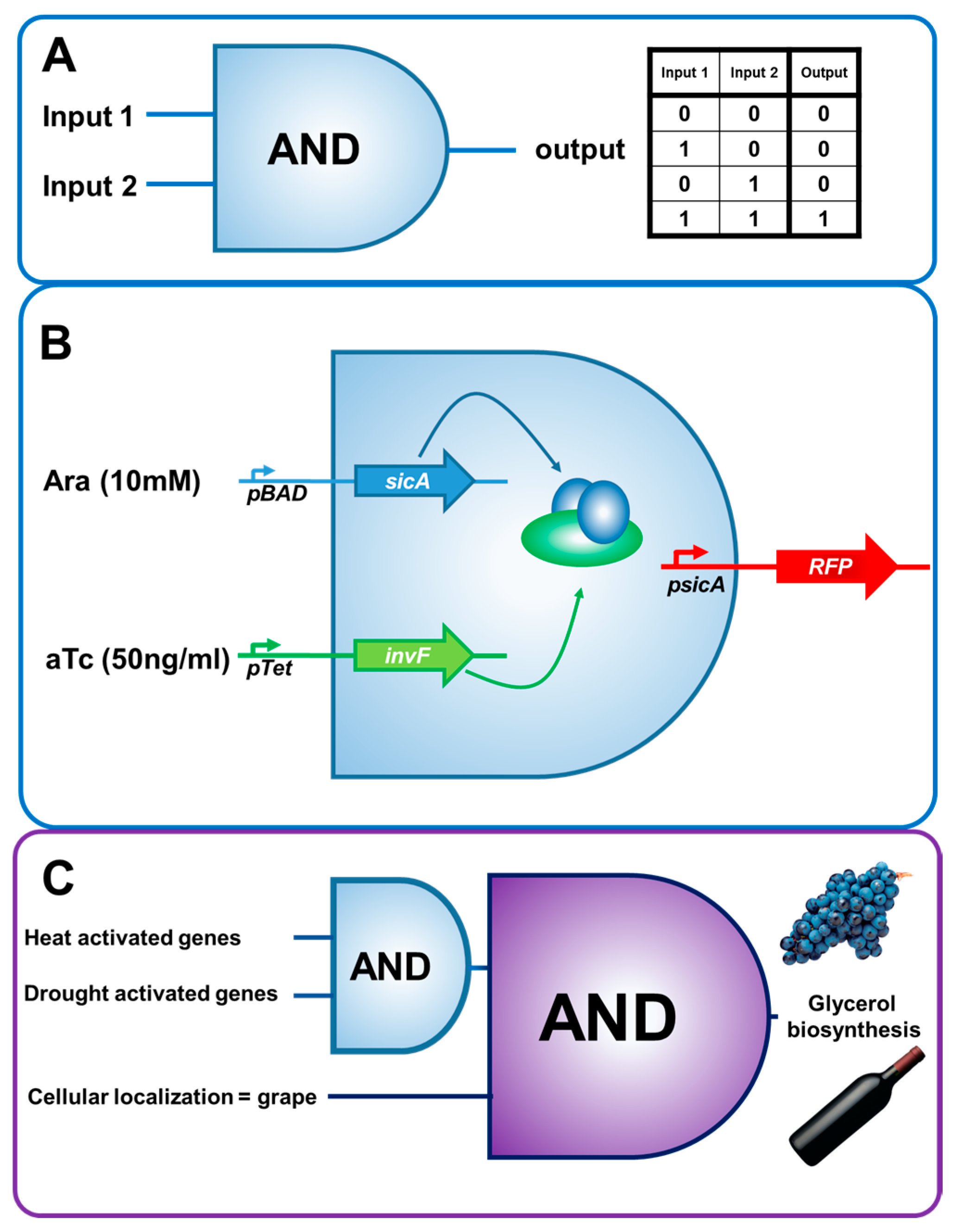
3. Biosensors and Molecular Circuitry: A Reductionist View of Biology
4. Opportunities for Plant-Based Agriculture through Innovations Drawn from Synthetic Biology
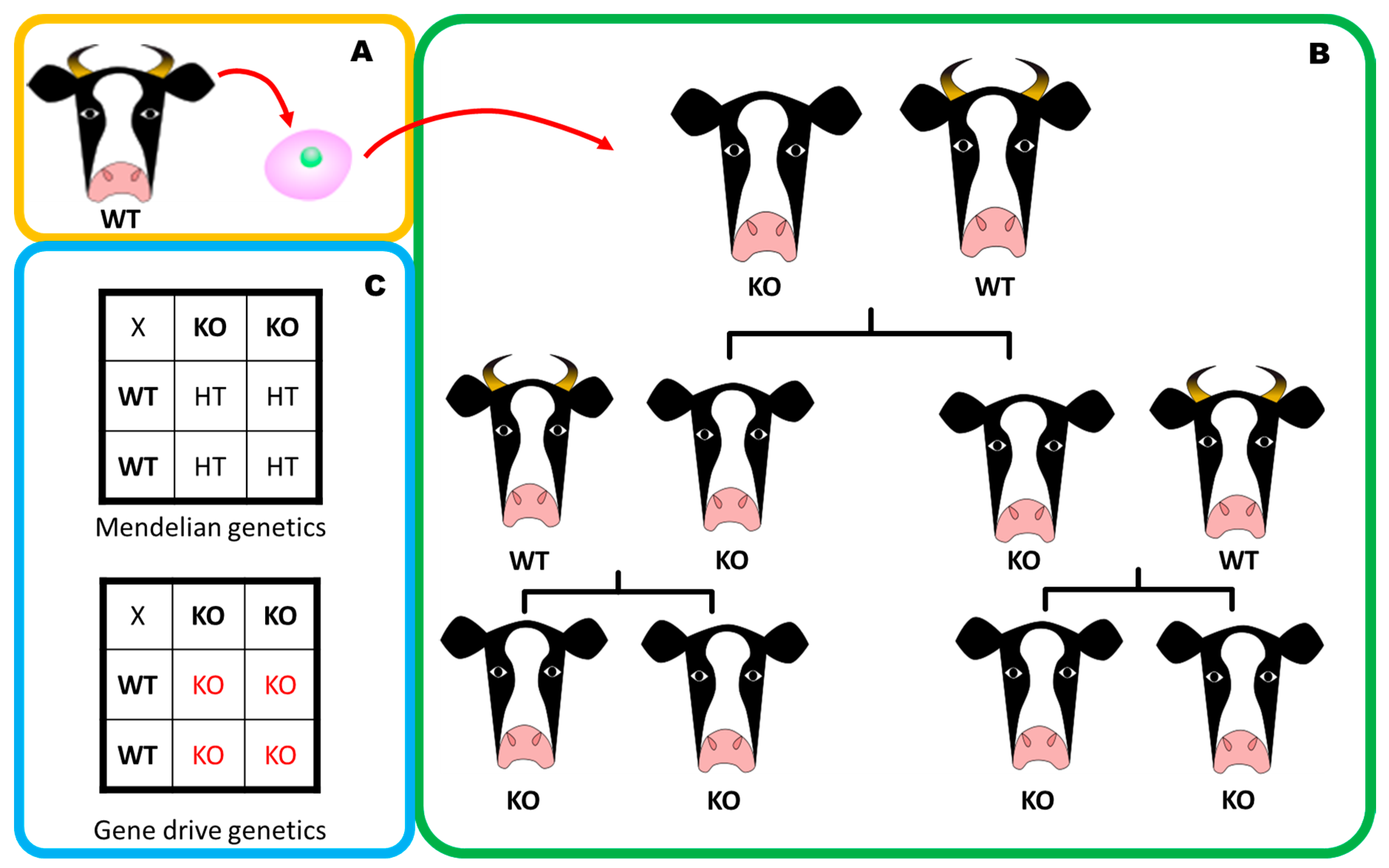
5. Gene Drives: A Powerful Technology Accelerated by Gene Editing
6. Whole Genome Approaches to Synthetic Biology: Synthetic Genomics
7. Regulation and Commercialization, the Next Challenges to Synthetic Biology
8. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Glass, J.I.; Assad-Garcia, N.; Alperovich, N.; Yooseph, S.; Lewis, M.R.; Maruf, M.; Hutchison, C.A.; Smith, H.O.; Venter, J.C. Essential genes of a minimal bacterium. Proc. Natl. Acad. Sci. USA 2006, 103, 425–430. [Google Scholar] [CrossRef] [PubMed]
- Lartigue, C.; Glass, J.I.; Alperovich, N.; Pieper, R.; Parmar, P.P.; Hutchison, C.A.; Smith, H.O.; Venter, J.C. Genome transplantation in bacteria: Changing one species to another. Science 2007, 317, 632–638. [Google Scholar] [CrossRef] [PubMed]
- Gibson, D.G.; Benders, G.A.; Andrews-pfannkoch, C.; Denisova, E.A.; Baden-tillson, H.; Zaveri, J.; Stockwell, T.B.; Brownley, A.; Thomas, D.W.; Algire, M.A.; et al. Complete checmical synthesis, assembly, and cloning of a Mycoplasma genitalium genome. Science 2008, 319, 1215–1221. [Google Scholar] [CrossRef] [PubMed]
- Lartigue, C.; Vashee, S.; Algire, M.A.; Chuang, R.; Benders, G.A.; Ma, L.; Noskov, V.N.; Denisova, E.A.; Gibson, D.G.; Assad-garcia, N.; et al. Creating Bacterial Strains from Engineered in Yeast. Science 2010, 1693, 1693–1696. [Google Scholar]
- Doudna, J.A.; Charpentier, E. The new frontier of genome engineering with CRISPR-Cas9. Science 2014, 346. [Google Scholar] [CrossRef] [PubMed]
- Jinek, M.; Chylinski, K.; Fonfara, I.; Hauer, M.; Doudna, J.A.; Charpentier, E. A Programmable Dual-RNA–Guided DNA Endonuclease in Adaptive Bacterial Immunity. Science 2012, 337, 816–822. [Google Scholar] [CrossRef] [PubMed]
- Xie, Z.-X.; Li, B.-Z.; Mitchell, L.A.; Wu, Y.; Qi, X.; Jin, Z.; Jia, B.; Wang, X.; Zeng, B.-X.; Liu, H.-M.; et al. “Perfect” designer chromosome V and behavior of a ring derivative. Science 2017, 355. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, L.A.; Wang, A.; Stracquadanio, G.; Kuang, Z.; Wang, X.; Yang, K.; Richardson, S.; Martin, J.A.; Zhao, Y.; Walker, R.; et al. Synthesis, debugging, and effects of synthetic chromosome consolidation: synVI and beyond. Science 2017, 355. [Google Scholar] [CrossRef] [PubMed]
- Shen, Y.; Wang, Y.; Chen, T.; Gao, F.; Gong, J.; Abramczyk, D.; Walker, R.; Zhao, H.; Chen, S.; Liu, W.; et al. Deep functional analysis of synII, a 770-kilobase synthetic yeast chromosome. Science 2017, 355. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Li, B.-Z.; Zhao, M.; Mitchell, L.A.; Xie, Z.-X.; Lin, Q.-H.; Wang, X.; Xiao, W.-H.; Wang, Y.; Zhou, X.; et al. Bug mapping and fitness testing of chemically synthesized chromosome X. Science 2017, 355. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Zhao, G.; Luo, Z.; Lin, Y.; Wang, L.; Guo, Y.; Wang, A.; Jiang, S.; Jiang, Q.; Gong, J.; et al. Engineering the ribosomal DNA in a megabase synthetic chromosome. Science 2017, 355. [Google Scholar] [CrossRef] [PubMed]
- Richardson, S.M.; Mitchell, L.A.; Stracquadanio, G.; Yang, K.; Dymond, J.S.; DiCarlo, J.E.; Lee, D.; Huang, C.L.V.; Chandrasegaran, S.; Cai, Y.; et al. Design of a synthetic yeast genome. Science 2017, 355, 1040–1044. [Google Scholar] [CrossRef] [PubMed]
- Pretorius, I.S.; Boeke, J.D. Yeast 2.0—connecting the dots in the construction of the world’s first functional synthetic eukaryotic genome. FEMS Yeast Res. 2018, 18, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Krivoruchko, A.; Nielsen, J. Production of natural products through metabolic engineering of Saccharomyces cerevisiae. Curr. Opin. Biotechnol. 2015, 35, 7–15. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Xiao, W.; Wang, Y.; Liu, H.; Li, X.; Yuan, Y. Lycopene overproduction in Saccharomyces cerevisiae through combining pathway engineering with host engineering. Microb. Cell Fact. 2016, 15, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Chai, F.; Wang, Y.; Mei, X.; Yao, M.; Chen, Y.; Liu, H.; Xiao, W.; Yuan, Y. Heterologous biosynthesis and manipulation of crocetin in Saccharomyces cerevisiae. Microb. Cell Fact. 2017, 16, 54. [Google Scholar] [CrossRef] [PubMed]
- Gander, M.W.; Vrana, J.D.; Voje, W.E.; Carothers, J.M.; Klavins, E. Digital logic circuits in yeast with CRISPR-dCas9 NOR gates. Nat. Commun. 2017, 8, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Moon, T.S.; Lou, C.; Tamsir, A.; Stanton, B.C.; Voigt, C.A. Genetic programs constructed from layered logic gates in single cells. Nature 2012, 491, 249–253. [Google Scholar] [CrossRef] [PubMed]
- Medford, J.I.; Prasad, A. Towards programmable plant genetic circuits. Plant J. 2016, 87, 139–148. [Google Scholar] [CrossRef] [PubMed]
- Brophy, J.A.N.; Voigt, C.A. Principles of genetic circuit design. Nat. Methods 2014, 11, 508–520. [Google Scholar] [CrossRef] [PubMed]
- Evenson, R.E.; Gollin, D. Crop Variety Improvement and Its Effect on Productivity the Impact of International Agricultural Research; CABI Publishing: Wallingford, UK, 2003; ISBN 0851995497. [Google Scholar]
- Thornton, P.K. Livestock production: Recent trends, future prospects. Philos. Trans. R. Soc. B Biol. Sci. 2010, 365, 2853–2867. [Google Scholar] [CrossRef] [PubMed]
- Brookes, G.; Barfoot, P. The global income and production effects of genetically modified (GM) crops 1996-2011. GM Crops Food 2013, 4, 74–83. [Google Scholar] [CrossRef] [PubMed]
- Mittler, R.; Blumwald, E. Genetic Engineering for Modern Agriculture: Challenges and Perspectives. Annu. Rev. Plant Biol. 2010, 61, 443–462. [Google Scholar] [CrossRef] [PubMed]
- Hajkowicz, B.S.; Eady, S. Rural Industry Futures: Megatrends Impacting Australian Agriculture over the Coming Twenty Years; Rural Industries Research and Development Corporation: Barton, ACT, Australia, 2015; Publication No. 15/065, Project No. PRJ-009712. [Google Scholar]
- Flores Bueso, Y.; Tangney, M. Synthetic Biology in the Driving Seat of the Bioeconomy. Trends Biotechnol. 2017, 35, 373–378. [Google Scholar] [CrossRef] [PubMed]
- Jensen, M.K.; Keasling, J.D. Recent applications of synthetic biology tools for yeast metabolic engineering. FEMS Yeast Res. 2015, 15, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Caspeta, L.; Chen, Y.; Ghiaci, P.; Feizi, A.; Buskov, S.; Hallstrom, B.M.; Petranovic, D.; Nielsen, J. Altered sterol composition renders yeast thermotolerant. Science 2014, 346, 75–78. [Google Scholar] [CrossRef] [PubMed]
- Tsai, C.-S.; Kwak, S.; Turner, T.L.; Jin, Y.-S. Yeast synthetic biology toolbox and applications for biofuel production. FEMS Yeast Res. 2015, 15, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Awan, A.R.; Shaw, W.M.; Ellis, T. Biosynthesis of therapeutic natural products using synthetic biology. Adv. Drug Deliv. Rev. 2016, 105, 96–106. [Google Scholar] [CrossRef] [PubMed]
- Paddon, C.J.; Westfall, P.J.; Pitera, D.J.; Benjamin, K.; Fisher, K.; McPhee, D.; Leavell, M.D.; Tai, A.; Main, A.; Eng, D.; et al. High-level semi-synthetic production of the potent antimalarial artemisinin. Nature 2013, 496, 528–532. [Google Scholar] [CrossRef] [PubMed]
- Pretorius, I.S. Synthetic genome engineering forging new frontiers for wine yeast. Crit. Rev. Biotechnol. 2017, 37, 112–136. [Google Scholar] [CrossRef] [PubMed]
- Pouvreau, B.; Vanhercke, T.; Singh, S. From Plant Metabolic Engineering to Plant Synthetic Biology: The evolution of the design/build/test/learn cycle. Plant Sci. 2018, 273, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Larkum, A.W.D. Limitations and prospects of natural photosynthesis for bioenergy production. Curr. Opin. Biotechnol. 2010, 21, 271–276. [Google Scholar] [CrossRef] [PubMed]
- Scott, S.A.; Davey, M.P.; Dennis, J.S.; Horst, I.; Howe, C.J.; Lea-Smith, D.J.; Smith, A.G. Biodiesel from algae: Challenges and prospects. Curr. Opin. Biotechnol. 2010, 21, 277–286. [Google Scholar] [CrossRef] [PubMed]
- Moses, T.; Mehrshahi, P.; Smith, A.G.; Goossens, A. Synthetic biology approaches for the production of plant metabolites in unicellular organisms. J. Exp. Bot. 2017, 68, 4057–4074. [Google Scholar] [CrossRef] [PubMed]
- Zimin, A.V.; Puiu, D.; Hall, R.; Kingan, S.; Clavijo, B.J.; Salzberg, S.L. The first near-complete assembly of the hexaploid bread wheat genome, Triticum aestivum. Gigascience 2017, 6, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Li, Y.; Smolke, C.D. Strategies for microbial synthesis of high-value phytochemicals. Nat. Chem. 2018, 10, 395–404. [Google Scholar] [CrossRef] [PubMed]
- Lee, D.; Lloyd, N.D.R.; Pretorius, I.S.; Borneman, A.R. Heterologous production of raspberry ketone in the wine yeast Saccharomyces cerevisiae via pathway engineering and synthetic enzyme fusion. Microb. Cell Fact. 2016, 15, 49. [Google Scholar] [CrossRef] [PubMed]
- Carvalho, Â.; Hansen, E.H.; Kayser, O.; Carlsen, S.; Stehle, F. Designing microorganisms for heterologous biosynthesis of cannabinoids. FEMS Yeast Res. 2017, 17, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Zirpel, B.; Degenhardt, F.; Martin, C.; Kayser, O.; Stehle, F. Engineering yeasts as platform organisms for cannabinoid biosynthesis. J. Biotechnol. 2017, 259, 204–212. [Google Scholar] [CrossRef] [PubMed]
- Galanie, S.; Thodey, K.; Trenchard, I.J.; Filsinger Interrante, M.; Smolke, C.D. Complete biosynthesis of opioids in yeast. Science 2015, 349, 1095–1100. [Google Scholar] [CrossRef] [PubMed]
- Wei, Y.; Bergenholm, D.; Gossing, M.; Siewers, V.; Nielsen, J. Expression of cocoa genes in Saccharomyces cerevisiae improves cocoa butter production. Microb. Cell Fact. 2018, 17, 11. [Google Scholar] [CrossRef] [PubMed]
- Wei, Y.; Siewers, V.; Nielsen, J. Cocoa butter-like lipid production ability of non-oleaginous and oleaginous yeasts under nitrogen-limited culture conditions. Appl. Microbiol. Biotechnol. 2017, 101, 3577–3585. [Google Scholar] [CrossRef] [PubMed]
- Chao, R.; Mishra, S.; Si, T.; Zhao, H. Engineering biological systems using automated biofoundries. Metab. Eng. 2017, 42, 98–108. [Google Scholar] [CrossRef] [PubMed]
- Peplow, M. Synthetic biology’s first malaria drug meets market resistance. Nature 2016, 530, 389–390. [Google Scholar] [CrossRef] [PubMed]
- Arendt, P.; Pollier, J.; Callewaert, N.; Goossens, A. Synthetic biology for production of natural and new-to-nature terpenoids in photosynthetic organisms. Plant J. 2016, 87, 16–37. [Google Scholar] [CrossRef] [PubMed]
- Williams, T.C.; Xu, X.; Ostrowski, M.; Pretorius, I.S.; Paulsen, I.T. Positive-feedback, ratiometric biosensor expression improves high-throughput metabolite-producer screening efficiency in yeast. Synth. Biol. 2017, 2, 1–13. [Google Scholar] [CrossRef]
- Le, N.C.H.; Gel, M.; Zhu, Y.; Dacres, H.; Anderson, A.; Trowell, S.C. Real-time, continuous detection of maltose using bioluminescence resonance energy transfer (BRET) on a microfluidic system. Biosens. Bioelectron. 2014, 62, 177–181. [Google Scholar] [CrossRef] [PubMed]
- Fowler, C.C.; Brown, E.D.; Li, Y. Using a riboswitch sensor to examine coenzyme B12 metabolism and transport in E. coli. Chem. Biol. 2010, 17, 756–765. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.J.; Jeong, H.; Lee, S.J. Synthetic biology for microbial heavy metal biosensors. Anal. Bioanal. Chem. 2018, 410, 1191–1203. [Google Scholar] [CrossRef] [PubMed]
- Mustafi, N.; Grünberger, A.; Kohlheyer, D.; Bott, M.; Frunzke, J. The development and application of a single-cell biosensor for the detection of l-methionine and branched-chain amino acids. Metab. Eng. 2012, 14, 449–457. [Google Scholar] [CrossRef] [PubMed]
- Levskaya, A.; Chevalier, A.A.; Tabor, J.J.; Simpson, Z.B.; Lavery, L.A.; Levy, M.; Davidson, E.A.; Scouras, A.; Ellignton, A.D.; Marcotte, E.M.; et al. Engineering Escherichia coli to see light. Nature 2005, 438, 442. [Google Scholar] [CrossRef] [PubMed]
- Lubkowicz, D.; Ho, C.L.; Hwang, I.Y.; Yew, W.S.; Lee, Y.S.; Chang, M.W. Reprogramming Probiotic Lactobacillus reuteri as a Biosensor for Staphylococcus aureus Derived AIP-I Detection. ACS Synth. Biol. 2018, 7, 1229–1237. [Google Scholar] [CrossRef] [PubMed]
- Wend, S.; Dal Bosco, C.; Kämpf, M.M.; Ren, F.; Palme, K.; Weber, W.; Dovzhenko, A.; Zurbriggen, M.D. A quantitative ratiometric sensor for time-resolved analysis of auxin dynamics. Sci. Rep. 2013, 3, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Goold, H.D.; Kroukamp, H.; Williams, T.C.; Paulsen, I.T.; Varela, C.; Pretorius, I.S. Yeast’s balancing act between ethanol and glycerol production in low-alcohol wines. Microb. Biotechnol. 2017, 10, 264–278. [Google Scholar] [CrossRef] [PubMed]
- Longo, R.; Blackman, J.W.; Antalick, G.; Torley, P.J.; Rogiers, S.Y.; Schmidtke, L.M. Harvesting and blending options for lower alcohol wines: A sensory and chemical investigation. J. Sci. Food Agric. 2018, 98, 33–42. [Google Scholar] [CrossRef] [PubMed]
- Din, M.O.; Danino, T.; Prindle, A.; Skalak, M.; Selimkhanov, J.; Allen, K.; Julio, E.; Atolia, E.; Tsimring, L.S.; Bhatia, S.N.; Hasty, J. Synchronized cycles of bacterial lysis for in vivo delivery. Nature 2016, 536, 81–85. [Google Scholar] [CrossRef] [PubMed]
- Williams, T.C.; Pretorius, I.S.; Paulsen, I.T. Synthetic Evolution of Metabolic Productivity Using Biosensors. Trends Biotechnol. 2016, 34, 371–381. [Google Scholar] [CrossRef] [PubMed]
- Dacres, H.; Wang, J.; Leitch, V.; Horne, I.; Anderson, A.R.; Trowell, S.C. Greatly enhanced detection of a volatile ligand at femtomolar levels using bioluminescence resonance energy transfer (BRET). Biosens. Bioelectron. 2011, 29, 119–124. [Google Scholar] [CrossRef] [PubMed]
- Swiegers, J.H.; Chambers, P.J.; Pretorius, I.S. Olfaction and taste: Human perception, physiology and genetics. Aust. J. Grape Wine Res. 2005, 11, 109–113. [Google Scholar] [CrossRef]
- Mukherjee, K.; Bhattacharyya, S.; Peralta-Yahya, P. GPCR-Based Chemical Biosensors for Medium-Chain Fatty Acids. ACS Synth. Biol. 2015, 4, 1261–1269. [Google Scholar] [CrossRef] [PubMed]
- Antunes, M.S.; Morey, K.J.; Jeff Smith, J.; Albrecht, K.D.; Bowen, T.A.; Zdunek, J.K.; Troupe, J.F.; Cuneo, M.J.; Webb, C.T.; Hellinga, H.W.; et al. Programmable ligand detection system in plants through a synthetic signal transduction pathway. PLoS ONE 2011, 6, e16292. [Google Scholar] [CrossRef] [PubMed]
- Peng, Y.; Allen, S.; Millwood, R.J.; Stewart, C.N. “Fukusensor:” A genetically engineered plant for reporting DNA damage in response to gamma radiation. Plant Biotechnol. J. 2014, 12, 1329–1332. [Google Scholar] [CrossRef] [PubMed]
- Jez, J.M.; Lee, S.G.; Sherp, A.M. The next green movement: Plant biology for the environment and sustainability. Science 2016, 353, 1241–1244. [Google Scholar] [CrossRef] [PubMed]
- Hammond, J.P.; Bennett, M.J.; Bowen, H.C.; Broadley, M.R.; Eastwood, D.C.; May, S.T.; Rahn, C.; Swarup, R.; Woolaway, K.E.; White, P.J. Changes in Gene Expression in Arabidopsis Shoot during Phosphate Starvation and the Potential for Developing Smart Plants. Society 2003, 132, 578–596. [Google Scholar] [CrossRef] [PubMed]
- Vanhercke, T.; Divi, U.K.; El Tahchy, A.; Liu, Q.; Mitchell, M.; Taylor, M.C.; Eastmond, P.J.; Bryant, F.; Mechanicos, A.; Blundell, C.; et al. Step changes in leaf oil accumulation via iterative metabolic engineering. Metab. Eng. 2017, 39, 237–246. [Google Scholar] [CrossRef] [PubMed]
- Way, J.C.; Collins, J.J.; Keasling, J.D.; Silver, P.A. Integrating biological redesign: Where synthetic biology came from and where it needs to go. Cell 2014, 157, 151–161. [Google Scholar] [CrossRef] [PubMed]
- Toussaint, M.; Bontemps, C.; Besserer, A.; Hotel, L.; Gérardin, P.; Leblond, P. Whole-cell biosensor of cellobiose and application to wood decay detection. J. Biotechnol. 2016, 239, 39–46. [Google Scholar] [CrossRef] [PubMed]
- Scognamiglio, V.; Arduini, F.; Palleschi, G.; Rea, G. Biosensing technology for sustainable food safety. TrAC Trends Anal. Chem. 2014, 62, 1–10. [Google Scholar] [CrossRef]
- Puchta, H. Using CRISPR/Cas in three dimensions: Towards synthetic plant genomes, transcriptomes and epigenomes. Plant J. 2016, 87, 5–15. [Google Scholar] [CrossRef] [PubMed]
- Watson, A.; Ghosh, S.; Williams, M.J.; Cuddy, W.S.; Simmonds, J.; Rey, M.D.; Asyraf Md Hatta, M.; Hinchliffe, A.; Steed, A.; Reynolds, D.; et al. Speed breeding is a powerful tool to accelerate crop research and breeding. Nat. Plants 2018, 4, 23–29. [Google Scholar] [CrossRef] [PubMed]
- Jarvis, D.E.; Ho, Y.S.; Lightfoot, D.J.; Schmöckel, S.M.; Li, B.; Borm, T.J.A.; Ohyanagi, H.; Mineta, K.; Michell, C.T.; Saber, N.; et al. The genome of Chenopodium quinoa. Nature 2017, 542, 307–312. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Stewart, C.N. Plant synthetic biology. Trends Plant Sci. 2015, 20, 309–317. [Google Scholar] [CrossRef] [PubMed]
- Gao, C. The future of CRISPR technologies in agriculture. Nat. Rev. Mol. Cell Biol. 2018, 19, 275–276. [Google Scholar] [CrossRef] [PubMed]
- Barton, K.A.; Brill, W.J. Prospects in Plant Genetic Engineering. Science 1983, 219, 671–676. [Google Scholar] [CrossRef] [PubMed]
- Rogers, C.; Oldroyd, G.E.D. Synthetic biology approaches to engineering the nitrogen symbiosis in cereals. J. Exp. Bot. 2014, 65, 1939–1946. [Google Scholar] [CrossRef] [PubMed]
- Pikaar, I.; Matassa, S.; Rabaey, K.; Bodirsky, B.L.; Popp, A.; Herrero, M.; Verstraete, W. Microbes and the Next Nitrogen Revolution. Environ. Sci. Technol. 2017, 51, 7297–7303. [Google Scholar] [CrossRef] [PubMed]
- Waltz, E. A new crop of microbe startups raises big bucks, takes on the establishment. Nat. Biotechnol. 2017, 35, 1120–1122. [Google Scholar] [CrossRef] [PubMed]
- Allen, R.S.; Tilbrook, K.; Warden, A.C.; Campbell, P.C.; Rolland, V.; Singh, S.P.; Wood, C.C. Expression of 16 Nitrogenase Proteins within the Plant Mitochondrial Matrix. Front. Plant Sci. 2017, 8, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Ort, D.R.; Merchant, S.S.; Alric, J.; Barkan, A.; Blankenship, R.E.; Bock, R.; Croce, R.; Hanson, M.R.; Hibberd, J.M.; Long, S.P.; et al. Redesigning photosynthesis to sustainably meet global food and bioenergy demand. Proc. Natl. Acad. Sci. USA 2015, 112, 8529–8536. [Google Scholar] [CrossRef] [PubMed]
- Giessen, T.W.; Silver, P.A. Engineering carbon fixation with artificial protein organelles. Curr. Opin. Biotechnol. 2017, 46, 42–50. [Google Scholar] [CrossRef] [PubMed]
- Lin, M.T.; Occhialini, A.; Andralojc, P.J.; Devonshire, J.; Hines, K.M.; Parry, M.A.J.; Hanson, M.R. β-Carboxysomal proteins assemble into highly organized structures in Nicotiana chloroplasts. Plant J. 2014, 79, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Majer, E.; Llorente, B.; Rodríguez-Concepción, M.; Daròs, J.-A. Rewiring carotenoid biosynthesis in plants using a viral vector. Sci. Rep. 2017, 7, 41645. [Google Scholar] [CrossRef] [PubMed]
- D’Andrea, L.; Simon-Moya, M.; Llorente, B.; Llamas, E.; Marro, M.; Loza-Alvarez, P.; Li, L.; Rodriguez-Concepcion, M. Interference with Clp protease impairs carotenoid accumulation during tomato fruit ripening. J. Exp. Bot. 2018, 69, 1557–1567. [Google Scholar] [CrossRef] [PubMed]
- Llorente, B.; Martinez-Garcia, J.F.; Stange, C.; Rodriguez-Concepcion, M. Illuminating colors: Regulation of carotenoid biosynthesis and accumulation by light. Curr. Opin. Plant Biol. 2017, 37, 49–55. [Google Scholar] [CrossRef] [PubMed]
- Petrie, J.R.; Shrestha, P.; Zhou, X.R.; Mansour, M.P.; Liu, Q.; Belide, S.; Nichols, P.D.; Singh, S.P. Metabolic Engineering Plant Seeds with Fish Oil-Like Levels of DHA. PLoS ONE 2012, 7, e49165. [Google Scholar] [CrossRef] [PubMed]
- Petrie, J.R.; Shrestha, P.; Belide, S.; Kennedy, Y.; Lester, G.; Liu, Q.; Divi, U.K.; Mulder, R.J.; Mansour, M.P.; Nichols, P.D.; et al. Metabolic engineering Camelina sativa with fish oil-like levels of DHA. PLoS ONE 2014, 9, e85061. [Google Scholar] [CrossRef] [PubMed]
- Jugder, B.E.; Ertan, H.; Bohl, S.; Lee, M.; Marquis, C.P.; Manefield, M. Organohalide respiring bacteria and reductive dehalogenases: Key tools in organohalide bioremediation. Front. Microbiol. 2016, 7, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Ravikumar, S.; Baylon, M.G.; Park, S.J.; Choi, J. il Engineered microbial biosensors based on bacterial two-component systems as synthetic biotechnology platforms in bioremediation and biorefinery. Microb. Cell Fact. 2017, 16, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Aracic, S.; Manna, S.; Petrovski, S.; Wiltshire, J.L.; Mann, G.; Franks, A.E. Innovative biological approaches for monitoring and improving water quality. Front. Microbiol. 2015, 6, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Hu, H.; Xiong, L. Genetic Engineering and Breeding of Drought-Resistant Crops. Annu. Rev. Plant Biol. 2014, 65, 715–741. [Google Scholar] [CrossRef] [PubMed]
- Friedel, S.; Usadel, B.; von Wirén, N.; Sreenivasulu, N. Reverse Engineering: A Key Component of Systems Biology to Unravel Global Abiotic Stress Cross-Talk. Front. Plant Sci. 2012, 3, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Olsen, J.L.; Rouzé, P.; Verhelst, B.; Lin, Y.C.; Bayer, T.; Collen, J.; Dattolo, E.; De Paoli, E.; Dittami, S.; Maumus, F.; et al. The genome of the seagrass Zostera marina reveals angiosperm adaptation to the sea. Nature 2016, 530, 331–335. [Google Scholar] [CrossRef] [PubMed]
- Pilon-Smits, E.; Pilon, M. Phytoremediation of metals using transgenic plants. CRC. Crit. Rev. Plant Sci. 2002, 21, 439–456. [Google Scholar] [CrossRef]
- Lhermie, G.; Gröhn, Y.T.; Raboisson, D. Addressing Antimicrobial Resistance: An Overview of Priority Actions to Prevent Suboptimal Antimicrobial Use in Food-Animal Production. Front. Microbiol. 2017, 7, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Bikard, D.; Euler, C.W.; Jiang, W.; Nussenzweig, P.M.; Goldberg, G.W.; Duportet, X.; Fischetti, V.A.; Marraffini, L.A. Exploiting CRISPR-Cas nucleases to produce sequence-specific antimicrobials. Nat. Biotechnol. 2014, 32, 1146–1150. [Google Scholar] [CrossRef] [PubMed]
- Doran, T.; Challagulla, A.; Cooper, C.; Tizard, M.; Jenkins, K. Genome editing in poultry-opportunities and impacts. Natl. Inst. Biosci. J. 2017, 1. [Google Scholar] [CrossRef]
- Marhsall Graves, J.A. Birds do it with a Z gene. Nature 2010, 42, 472–477. [Google Scholar]
- Oishi, I.; Yoshii, K.; Miyahara, D.; Kagami, H.; Tagami, T. Targeted mutagenesis in chicken using CRISPR/Cas9 system. Sci. Rep. 2016, 6, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Alphey, L. Can CRISPR-Cas9 gene drives curb malaria? Nat. Biotechnol. 2016, 34, 149–150. [Google Scholar] [CrossRef] [PubMed]
- Akbari, O.S.; Matzen, K.D.; Marshall, J.M.; Huang, H.; Ward, C.M.; Hay, B.A. A synthetic gene drive system for local, reversible modification and suppression of insect populations. Curr. Biol. 2013, 23, 671–677. [Google Scholar] [CrossRef] [PubMed]
- Moro, D.; Byrne, M.; Kennedy, M.; Campbell, S.; Tizard, M. Identifying knowledge gaps for gene drive research to control invasive animal species: The next CRISPR step. Glob. Ecol. Conserv. 2018, 13, e00363. [Google Scholar] [CrossRef]
- Carlson, D.F.; Lancto, C.A.; Zang, B.; Kim, E.-S.; Walton, M.; Oldeschulte, D.; Seabury, C.; Sonstegard, T.S.; Fahrenkrug, S.C. Production of hornless dairy cattle from genome-edited cell lines. Nat. Biotechnol. 2016, 34, 479–481. [Google Scholar] [CrossRef] [PubMed]
- Gonen, S.; Jenko, J.; Gorjanc, G.; Mileham, A.J.; Whitelaw, C.B.A.; Hickey, J.M. Potential of gene drives with genome editing to increase genetic gain in livestock breeding programs. Genet. Sel. Evol. 2017, 49, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Chu, S.; Majumdar, A. Opportunities and challenges for a sustainable energy future. Nature 2012, 488, 294–303. [Google Scholar] [CrossRef] [PubMed]
- De Lorenzo, V. Seven microbial bio-processes to help the planet. Microb. Biotechnol. 2017, 10, 995–998. [Google Scholar] [CrossRef] [PubMed]
- Hutchison, C.A.; Chuang, R.-Y.; Noskov, V.N.; Assad-Garcia, N.; Deerinck, T.J.; Ellisman, M.H.; Gill, J.; Kannan, K.; Karas, B.J.; Ma, L.; et al. Design and synthesis of a minimal bacterial genome. Science 2016, 351, aad6253. [Google Scholar] [CrossRef] [PubMed]
- Goold, H.D.; Nguyen, H.M.; Kong, F.; Beyly-Adriano, A.; Légeret, B.; Billon, E.; Cuiné, S.; Beisson, F.; Peltier, G.; Li-Beisson, Y. Whole Genome Re-Sequencing Identifies a Quantitative Trait Locus Repressing Carbon Reserve Accumulation during Optimal Growth in Chlamydomonas reinhardtii. Sci. Rep. 2016, 6, 25209. [Google Scholar] [CrossRef] [PubMed]
- Niu, D.; Wei, H.-J.; Lin, L.; George, H.; Wang, T.; Lee, I.-H.; Zhao, H.-Y.; Wang, Y.; Kan, Y.; Shrock, E.; et al. Inactivation of porcine endogenous retrovirus in pigs using CRISPR-Cas9. Science 2017, 357, 1303–1307. [Google Scholar] [CrossRef] [PubMed]
- Jovicevic, D.; Blount, B.A.; Ellis, T. Total synthesis of a eukaryotic chromosome: Redesigning and SCRaMbLE-ing yeast. BioEssays 2014, 36, 855–860. [Google Scholar] [CrossRef] [PubMed]
- Shen, Y.; Stracquadanio, G.; Wang, Y.; Yang, K.; Mitchell, L.A.; Xue, Y.; Cai, Y.; Chen, T.; Dymond, J.S.; Kang, K.; et al. SCRaMbLE generates designed combinatorial stochastic diversity in synthetic chromosomes. Genome Res. 2016, 26, 36–49. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Luo, Z.; Wang, Y.; Pham, N.T.; Tuck, L.; Pérez-Pi, I.; Liu, L.; Shen, Y.; French, C.; Auer, M.; et al. Rapid pathway prototyping and engineering using in vitro and in vivo synthetic genome SCRaMbLE-in methods. Nat. Commun. 2018, 9, 1936. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Zhu, R.; Mitchell, L.A.; Ma, L.; Liu, R.; Zhao, M.; Jia, B.; Xu, H.; Li, Y.-X.; Yang, Z.-M.; et al. In vitro DNA SCRaMbLE. Nat. Commun. 2018, 9, 1935. [Google Scholar] [CrossRef] [PubMed]
- Wang, K.; Fredens, J.; Brunner, S.F.; Kim, S.H.; Chia, T.; Chin, J.W. Defining synonymous codon compression schemes by genome recoding. Nature 2016, 539, 59–64. [Google Scholar] [CrossRef] [PubMed]
- Neumann-Staubitz, P.; Neumann, H. The use of unnatural amino acids to study and engineer protein function. Curr. Opin. Struct. Biol. 2016, 38, 119–128. [Google Scholar] [CrossRef] [PubMed]
- Lau, Y.H.; Stirling, F.; Kuo, J.; Karrenbelt, M.A.P.; Chan, Y.A.; Riesselman, A.; Horton, C.A.; Schäfer, E.; Lips, D.; Weinstock, M.T.; et al. Large-scale recoding of a bacterial genome by iterative recombineering of synthetic DNA. Nucleic Acids Res. 2017, 45, 6971–6980. [Google Scholar] [CrossRef] [PubMed]
- Ma, N.J.; Isaacs, F.J. Genomic Recoding Broadly Obstructs the Propagation of Horizontally Transferred Genetic Elements. Cell Syst. 2016, 3, 199–207. [Google Scholar] [CrossRef] [PubMed]
- Maselko, M.; Heinsch, S.C.; Chacón, J.M.; Harcombe, W.R.; Smanski, M.J. Engineering species-like barriers to sexual reproduction. Nat. Commun. 2017, 8, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Cai, Y.; Agmon, N.; Choi, W.J.; Ubide, A.; Stracquadanio, G.; Caravelli, K.; Hao, H.; Bader, J.S.; Boeke, J.D. Intrinsic biocontainment: Multiplex genome safeguards combine transcriptional and recombinational control of essential yeast genes. Proc. Natl. Acad. Sci. USA 2015, 112, 1803–1808. [Google Scholar] [CrossRef] [PubMed]
- Boeke, J.D.; Church, G.; Hessel, A.; Kelley, N.J.; Arkin, A.; Cai, Y.; Carlson, R.; Chakravarti, A.; Cornish, V.W.; Holt, L.; et al. The Genome Project-Write. Science 2016, 353, 126–127. [Google Scholar] [CrossRef] [PubMed]
- Wintle, B.C.; Boehm, C.R.; Rhodes, C.; Molloy, J.C.; Millett, P.; Adam, L.; Breitling, R.; Carlson, R.; Casagrande, R.; Dando, M.; et al. A transatlantic perspective on 20 emerging issues in biological engineering. Elife 2017, 6, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Oye, K.A.; Esvelt, K.; Appleton, E.; Catteruccia, F.; Church, G.; Kuiken, T.; Lightfoot, S.B.-Y.; McNamara, J.; Smidler, A.; Collins, J.P. Regulating gene drives. Science 2014, 345, 626–628. [Google Scholar] [CrossRef] [PubMed]
- Sliva, A.; Yang, H.; Boeke, J.D.; Mathews, D.J.H. Freedom and responsibility in synthetic genomics: The synthetic yeast project. Genetics 2015, 200, 1021–1028. [Google Scholar] [CrossRef] [PubMed]
- Vivier, M.A.; Pretorius, I.S. Genetically tailored grapevines for the wine industry. Trends Biotechnol. 2002, 20, 472–478. [Google Scholar] [CrossRef]
- Kearney, S.M.; Gibbons, S.M. Designing synbiotics for improved human health. Microb. Biotechnol. 2018, 11, 141–144. [Google Scholar] [CrossRef] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Goold, H.D.; Wright, P.; Hailstones, D. Emerging Opportunities for Synthetic Biology in Agriculture. Genes 2018, 9, 341. https://doi.org/10.3390/genes9070341
Goold HD, Wright P, Hailstones D. Emerging Opportunities for Synthetic Biology in Agriculture. Genes. 2018; 9(7):341. https://doi.org/10.3390/genes9070341
Chicago/Turabian StyleGoold, Hugh Douglas, Philip Wright, and Deborah Hailstones. 2018. "Emerging Opportunities for Synthetic Biology in Agriculture" Genes 9, no. 7: 341. https://doi.org/10.3390/genes9070341
APA StyleGoold, H. D., Wright, P., & Hailstones, D. (2018). Emerging Opportunities for Synthetic Biology in Agriculture. Genes, 9(7), 341. https://doi.org/10.3390/genes9070341




