Microfluidic Devices for Drug Delivery Systems and Drug Screening
Abstract
1. Introduction
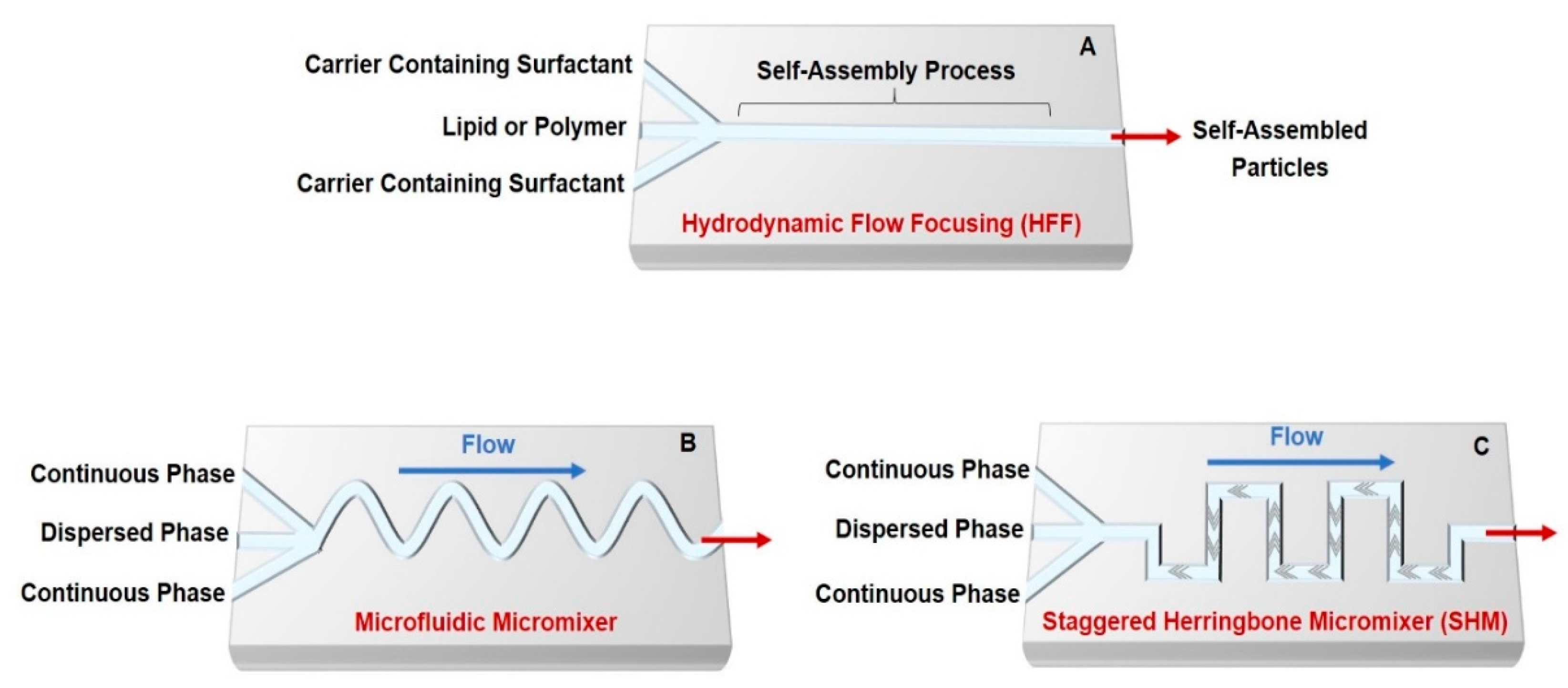
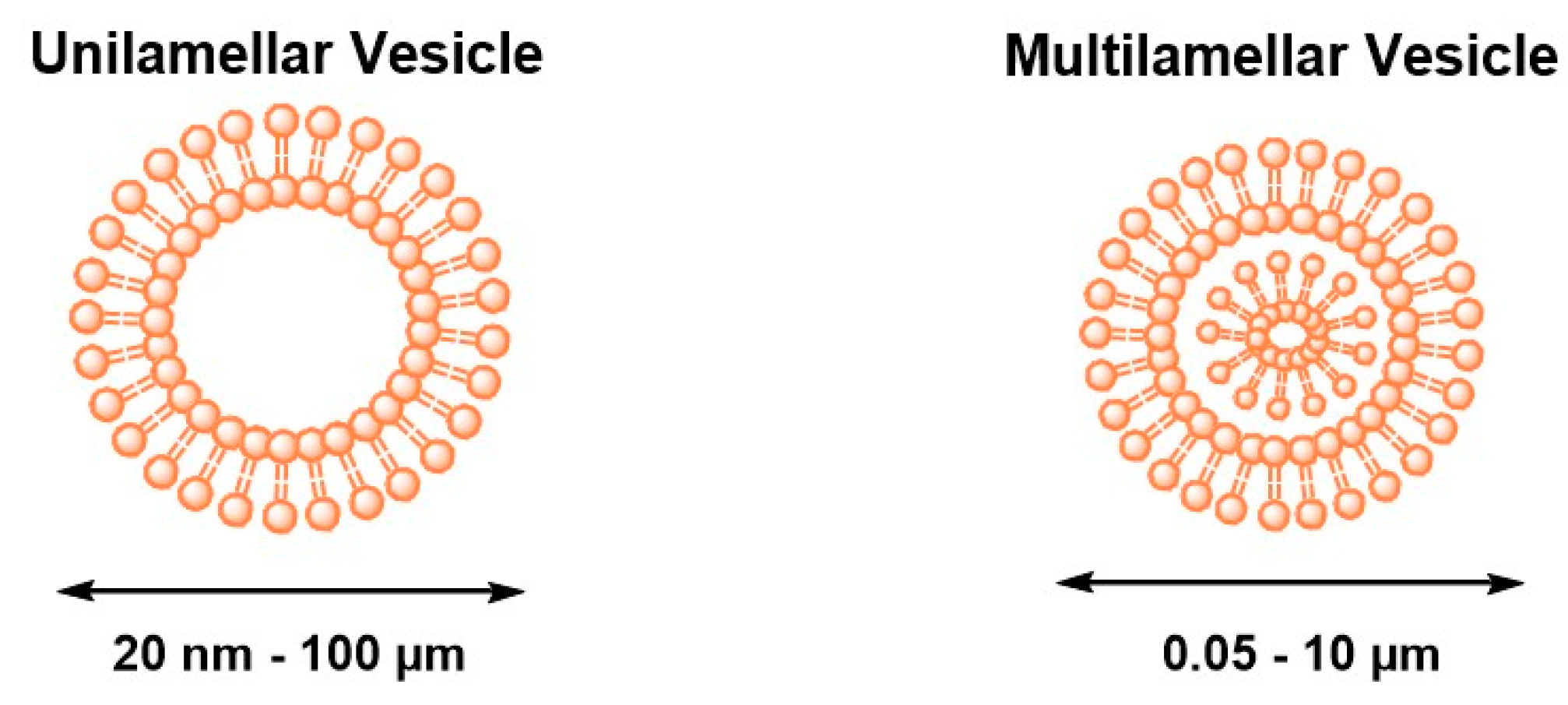
2. Self-Assembled Drug Delivery System
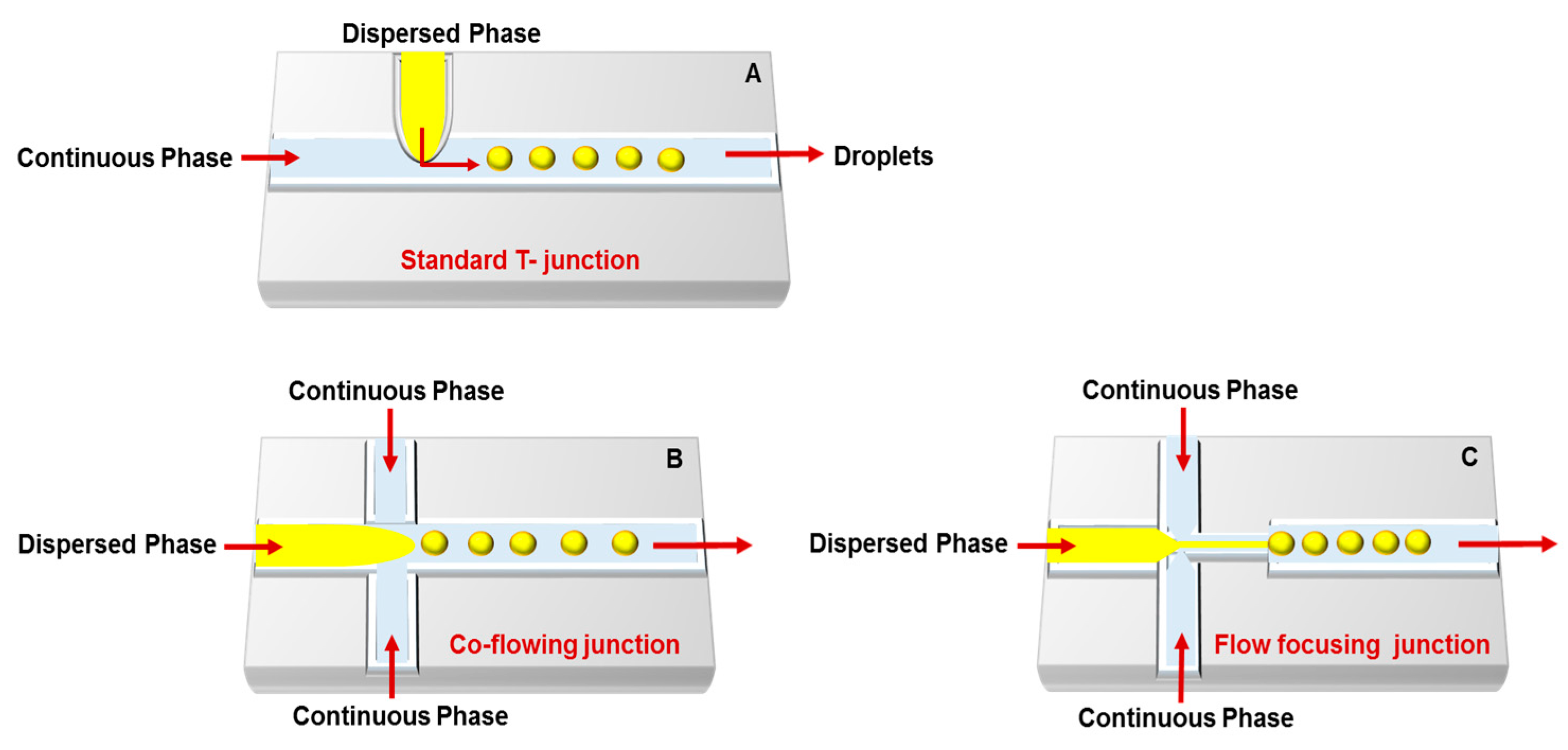
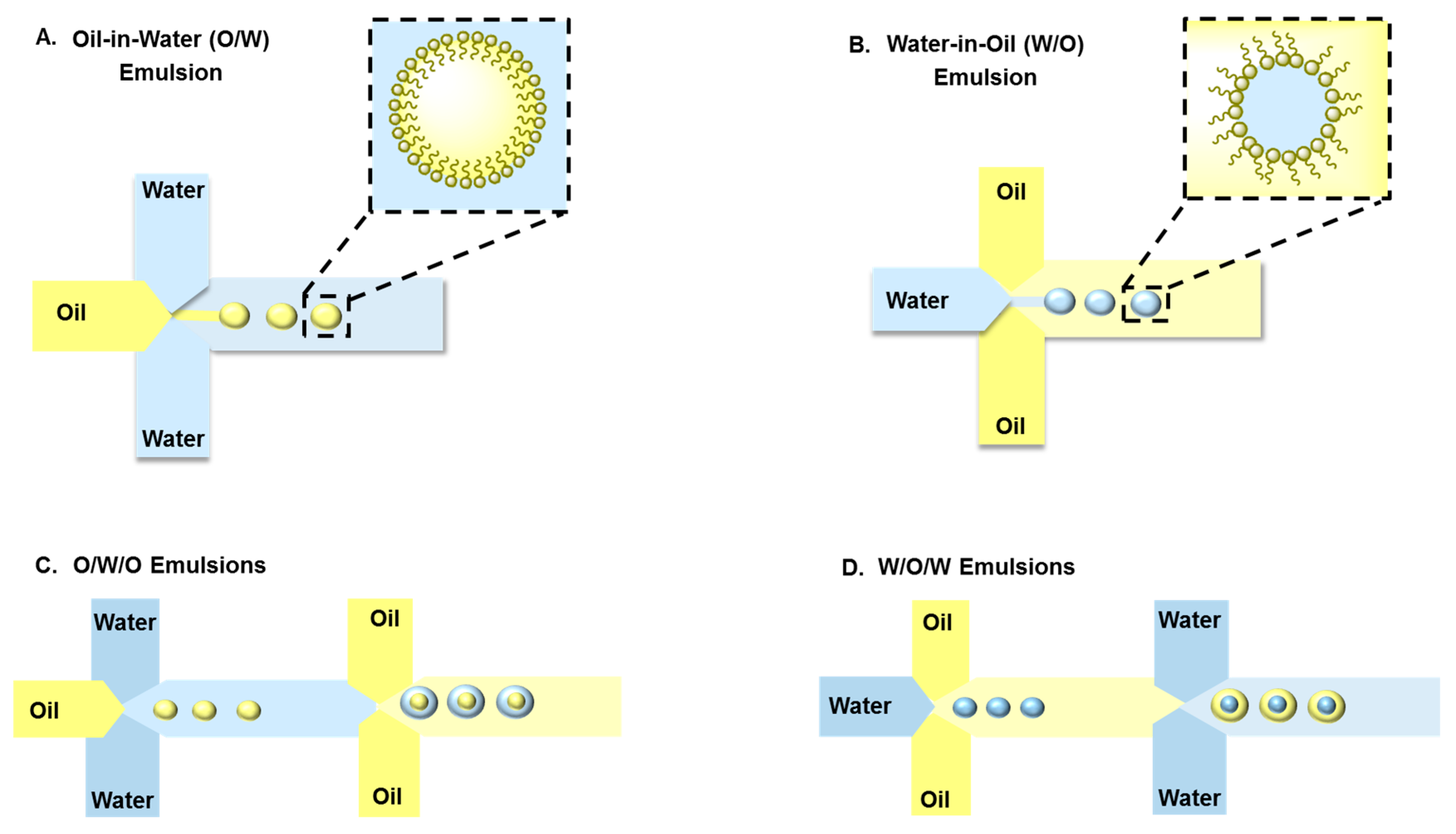
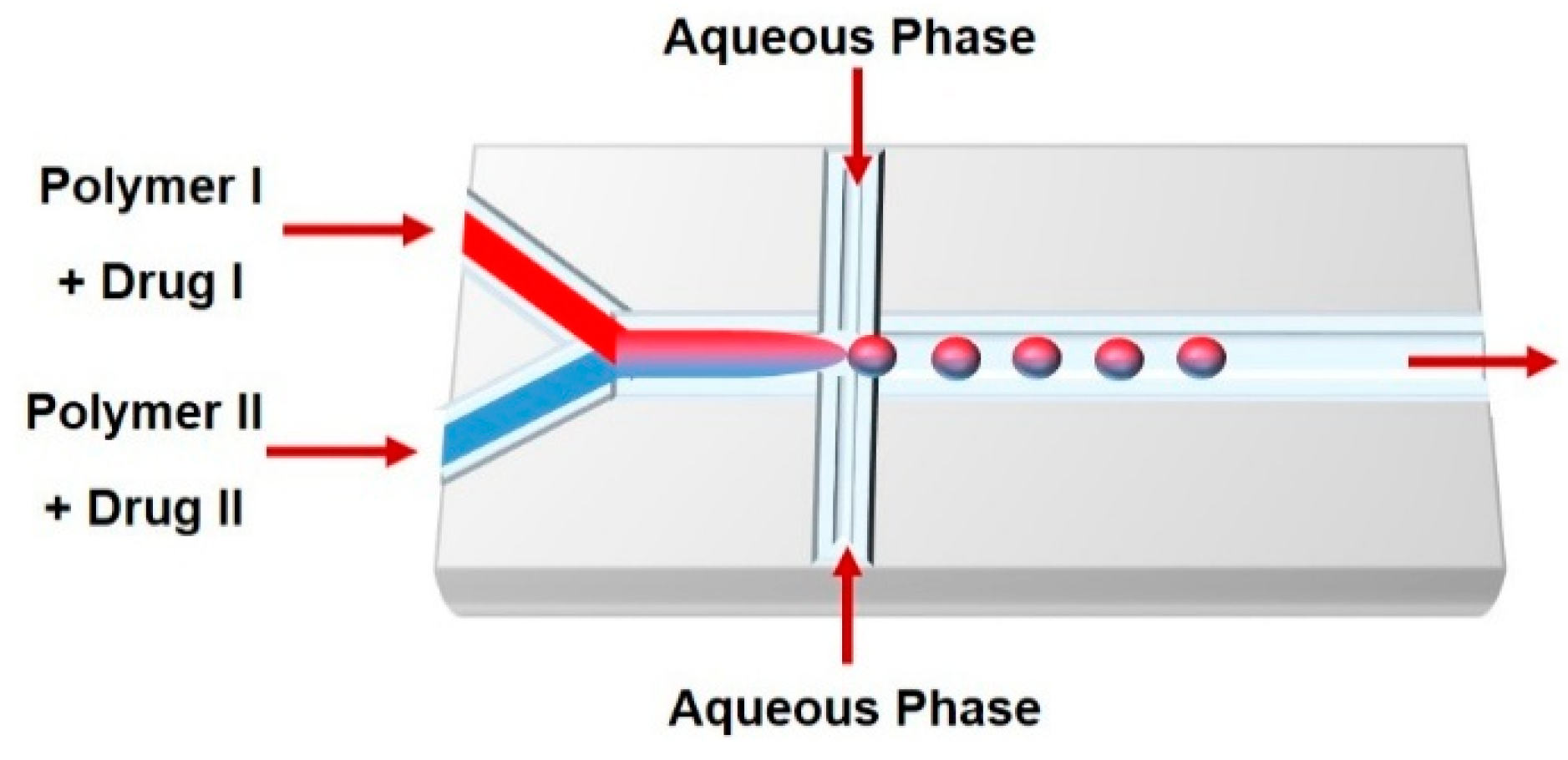
3. Droplet-Based Carriers
4. Non-Spherical Drug Delivery Systems
5. Nucleic Acid Delivery Systems
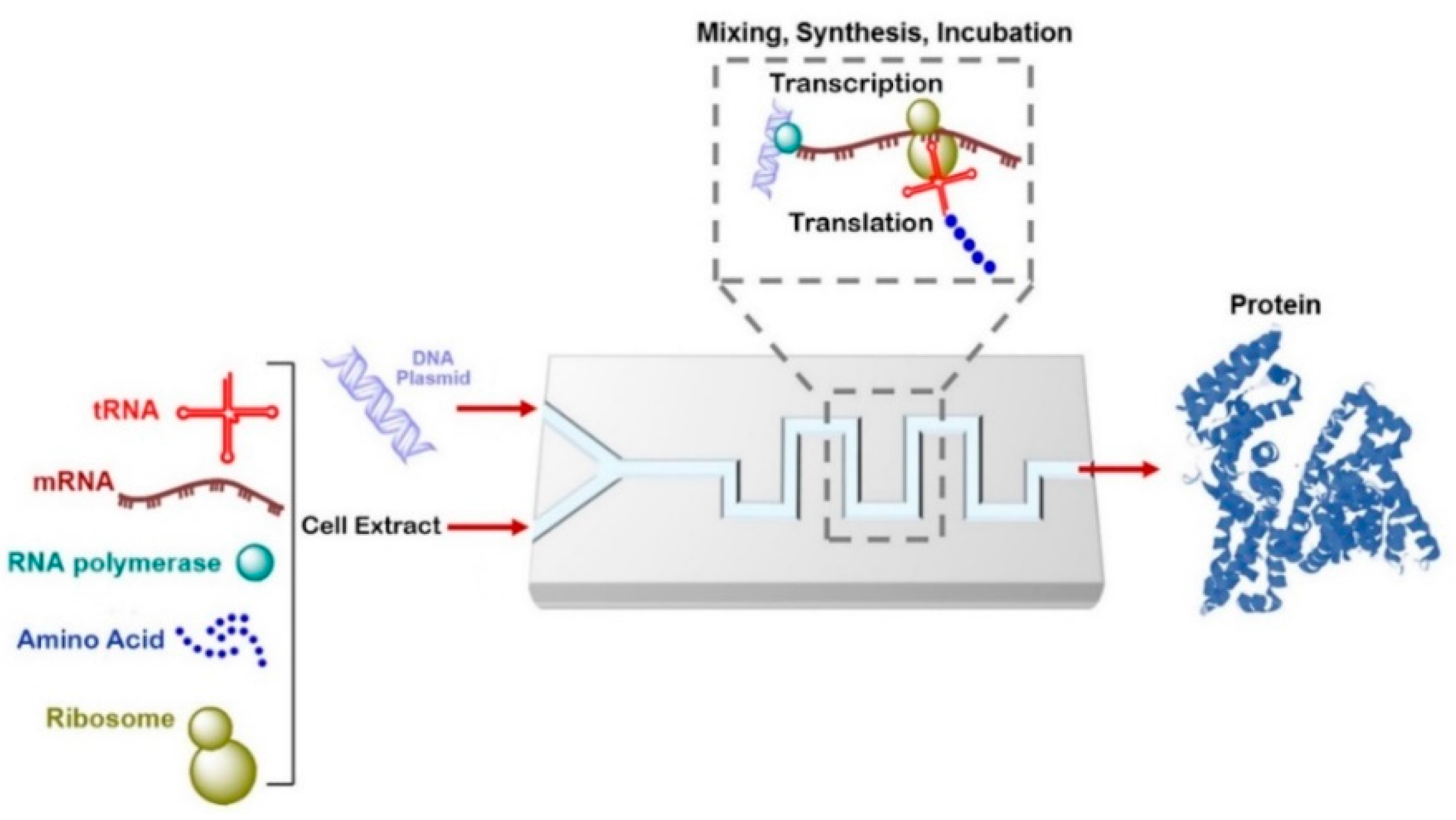
6. Application of Microfluidics for Protein Synthesis and Drug Screening
6.1. Cell-Free Protein Synthesis on a Chip
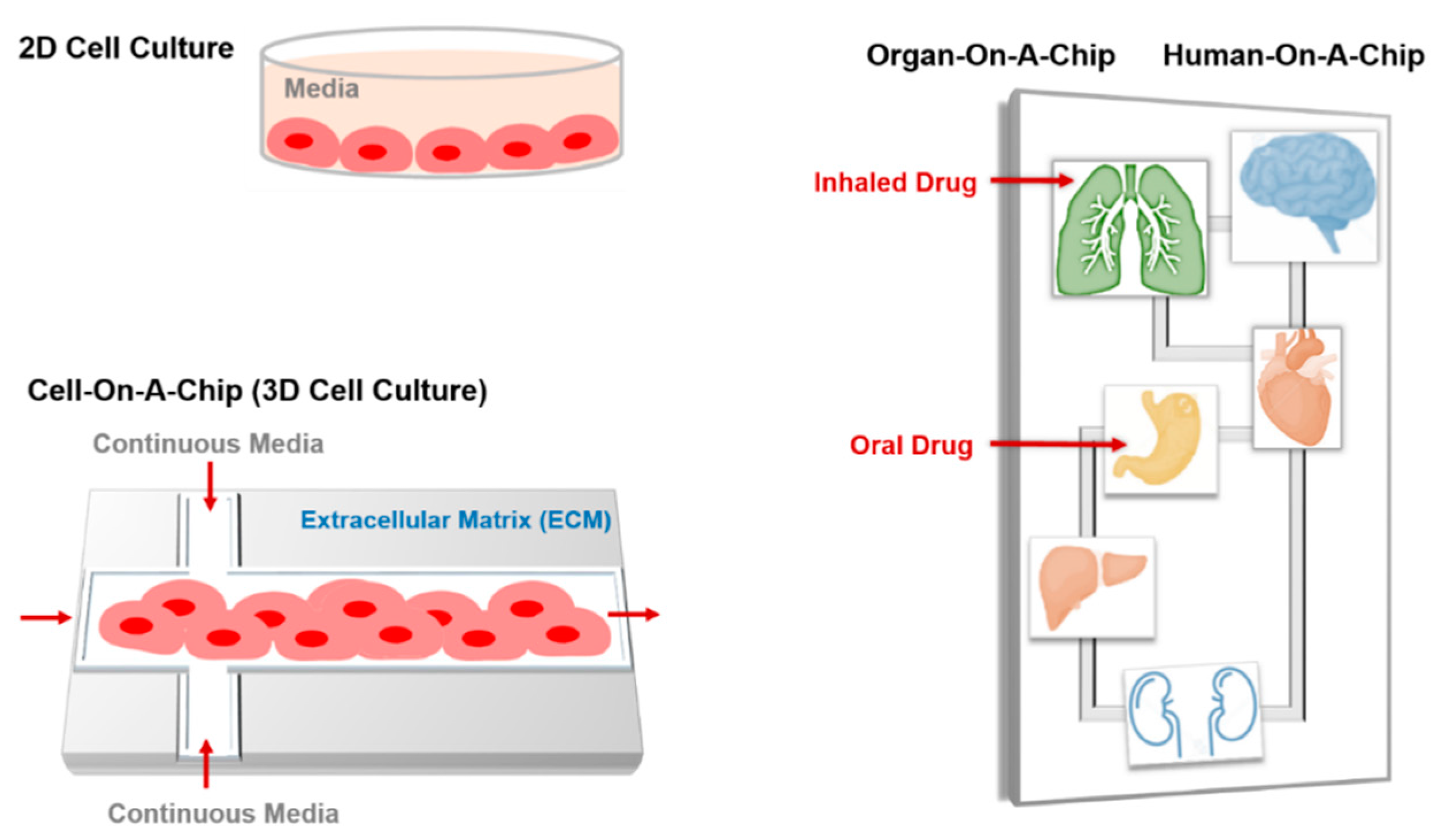
6.2. Cell-on-a-Chip
6.3. Organ-on-a-Chip
6.4. Human-On-A-Chip
7. Current and Future Perspectives
8. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Puigmartí-Luis, J. Microfluidic platforms: A mainstream technology for the preparation of crystals. Chem. Soc. Rev. 2014, 43, 2253–2271. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Fang, Q. Analytical detection techniques for droplet microfluidics—A review. Anal. Chim. Acta 2013, 787, 24–35. [Google Scholar] [CrossRef] [PubMed]
- Samiei, E.; Hoorfar, M. Systematic analysis of geometrical based unequal droplet splitting in digital microfluidics. J. Micromech. Microeng. 2015, 25, 055008. [Google Scholar] [CrossRef]
- Oliveira, K.A.; Silva, P.B.M.; de Souza, F.R.; Martins, F.T.; Coltro, W.K.T. Kinetic study of glucose oxidase on microfluidic toner-based analytical devices for clinical diagnostics with image-based detection. Anal. Methods 2014, 6, 4995–5000. [Google Scholar] [CrossRef]
- Whitesides, G.M. The origins and the future of microfluidics. Nature 2006, 442, 368–373. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Ren, L.; Li, L.; Liu, W.; Zhou, J.; Yu, W.; Tong, D.; Chen, S. Microfluidics: A new cosset for neurobiology. Lab Chip 2009, 9, 644–652. [Google Scholar] [CrossRef] [PubMed]
- Jones, C.N.; Hoang, A.N.; Dimisko, L.; Hamza, B.; Martel, J.; Irimia, D. Microfluidic platform for measuring neutrophil chemotaxis from unprocessed whole blood. J. Vis. Exp. 2014. [Google Scholar] [CrossRef] [PubMed]
- Bernacka-Wojcik, I.; Lopes, P.; Vaz, A.C.; Veigas, B.; Wojcik, P.J.; Simões, P.; Barata, D.; Fortunato, E.; Baptista, P.V.; Águas, H.; et al. Bio-microfluidic platform for gold nanoprobebased DNA detection-application to mycobacterium tuberculosis. Biosens. Bioelectron. 2013, 48, 87–93. [Google Scholar] [CrossRef] [PubMed]
- Peng, Z.; Young, B.; Baird, A.E.; Soper, S.A. Single-pair fluorescence resonance energy transfer analysis of mRNA transcripts for highly sensitive gene expression profiling in near real time. Anal. Chem. 2013, 85, 7851–7858. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Valadez, A.; Zuo, P.; Nie, Z. Microfluidic 3D cell culture: Potential application for tissue-based bioassays. Bioanalysis 2012, 4, 1509–1525. [Google Scholar] [CrossRef] [PubMed]
- Kodzius, R.; Xiao, K.; Wu, J.; Yi, X.; Gong, X.; Foulds, I.G.; Wen, W. Inhibitory effect of common microfluidic materials on PCR outcome. Sens. Actuators B Chem 2012, 161, 349–358. [Google Scholar] [CrossRef]
- Park, L.; Li, J.; Han, A. Micro-macro hybrid soft-lithography master (MMHSM) fabrication for lab-on-a-chip applications. Biomed. Microdevices 2010, 12, 345–351. [Google Scholar] [CrossRef] [PubMed]
- Pla-Roca, M.; Fernandez, J.; Mills, C.; Elena Martínez, E.; Samitier, J. Micro/nanopatterning of proteins via contact printing using high aspect ratio PMMA stamps and NanoImprint apparatus. Langmuir 2007, 23, 8614–8618. [Google Scholar] [CrossRef] [PubMed]
- Lee, W.; Jambovane, S.; Kim, D.; Hong, J. Predictive model on micro droplet generation through mechanical cutting. Microfluid. Nanofluid. 2009, 7, 431–438. [Google Scholar] [CrossRef]
- Nunes, P.; Ohlsson, P.; Ordeig, O.; Kutter, J. Cyclic olefin polymers: Emerging materials for lab-on-a-chip applications. Microfluid. Nanofluid. 2010, 9, 145–161. [Google Scholar] [CrossRef]
- Ren, K.; Dai, W.; Zhou, J.; Su, J.; Wu, H. Whole-Teflon microfluidic chips. Proc. Natl. Acad. Sci. USA 2011, 108, 8162–8166. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Kang, M.; Jensen, E.; Mathies, R. microvalves and pumps for microfluidic control. Anal. Chem. 2012, 84, 2067–2071. [Google Scholar] [CrossRef] [PubMed]
- Tsui, J.; Lee, W.; Pun, S.; Kim, J.; Kim, D. Microfluidics-assisted in vitro drug screening and carrier production. Adv. Drug Deliv. Rev. 2013, 15, 1575–1588. [Google Scholar] [CrossRef] [PubMed]
- Ligler, F.S.; Kim, J.S. The Microflow Cytometer; Pan Stanford: New York, USA, 2010; p. 106. [Google Scholar]
- Owens, D.; Peppas, N. Opsonization, biodistribution, and pharmacokinetics of polymeric nanoparticles. Int. J. Pharmaceut. 2006, 307, 93–102. [Google Scholar] [CrossRef] [PubMed]
- Thomas, A.; Tan, J.; Liu, Y. Characterization of nanoparticle delivery in microcirculation using a microfluidic device. Microvasc. Res. 2014, 94, 17–27. [Google Scholar] [CrossRef] [PubMed]
- Riahi, R.; Tamayol, A.; Shaegh, S.; Ghaemmaghami, A.; Dokmeci, M.; Khademhosseini, A. Microfluidics for advanced drug delivery systems. Curr. Opin. Chem. Eng. 2015, 7, 101–112. [Google Scholar] [CrossRef]
- Chan, H.; Ma, S.; Leong, K. Can microfluidics address biomanufacturing challenges in drug/gene/cell therapies? Regen. Biomater. 2016, 3, 87–98. [Google Scholar] [CrossRef] [PubMed]
- Ward, K.; Fan, Z. Mixing in microfluidic devices and enhancement methods. J. Micromech. Microeng. 2015, 25, 094001. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Sanche, S. Self-assembly via microfluidics. Lab Chip 2015, 7, 4383–4386. [Google Scholar] [CrossRef] [PubMed]
- DeMello, A. Control and detection of chemical reactions in microfluidic systems. Nature 2006, 442, 349–402. [Google Scholar] [CrossRef] [PubMed]
- Lo, C.; Jahn, A.; Locascio, L.; Vreeland, W.N. Controlled self-assembly of monodisperse niosomes by microfluidic hydrodynamic focusing. Langmuir 2010, 26, 8559–8566. [Google Scholar] [CrossRef] [PubMed]
- Streets, A.; Huang, Y. Microfluidics for biological measurements with single-molecule resolution. Curr. Opin. Biotechnol. 2014, 25C, 69–77. [Google Scholar] [CrossRef] [PubMed]
- Saliterman, S.S. Fundamentals of BioMEMS and Medical Microdevices; SPIE Press: Bellingham, WA, USA, 2006. [Google Scholar]
- Karnik, R.; Gu, F.; Basto, P.; Cannizzaro, C.; Dean, L.; Kyei-Manu, W.; Langer, R.; Farokhzad, O. Microfluidic platform for controlled synthesis of polymeric nanoparticles. Nano Lett. 2008, 8, 2906–2912. [Google Scholar] [CrossRef] [PubMed]
- Lim, J.; Bertrand, N.; Valencia, P.; Rhee, M.; Langer, R.; Jon, S.; Farokhzad, O.; Karnik, R. Parallel microfluidic synthesis of size-tunable polymeric nanoparticles using 3D flow focusing towards in vivo study. Nanomed. Nanotechnol. Biol. Med. 2014, 10, 401–409. [Google Scholar] [CrossRef] [PubMed]
- Kock, M.; Witt, H.; Evans, A.; Brunnschweiler, A. Improved characterization technique for micromixers. J. Micromech. Microeng. 1999, 9, 156–158. [Google Scholar] [CrossRef]
- Yaralioglu, G.; Wygant, I.; Marentis, T.; Khuri-Yakub, B. Ultrasonic mixing in microfluidic channels using integrated transducers. Anal. Chem. 2004, 76, 3694–3698. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.; Lee, S.; Kwon, T.; Ahn, C. A serpentine laminating micromixer combining splitting/recombination and advection. Lab Chip 2005, 7, 739–747. [Google Scholar] [CrossRef] [PubMed]
- Park, S.; Kim, J.; Park, J.; Chung, S.; Chung, C.; Chang, J. Rapid three-dimensional passive rotation micromixer using the breakup process. J. Micromech. Microeng. 2004, 14, 6–14. [Google Scholar] [CrossRef]
- Belliveau, N.; Huft, J.; Lin, P.; Chen, S.; Leung, A.; Leaver, T.; Wild, A.; Lee, J.; Taylor, R.; Tam, Y. Microfluidic synthesis of highly potent limit-size lipid nanoparticles for in vivo delivery of siRNA. Mol. Ther. Nucleic Acids 2012, 1, e37. [Google Scholar] [CrossRef] [PubMed]
- Zhigaltsev, I.; Belliveau, N.; Hafez, I.; Leung, A.; Huft, J.; Hansen, C.; Cullis, P. Bottom-up design and synthesis of limit size lipid nanoparticle systems with aqueous and triglyceride cores using millisecond microfluidic mixing. Langmuir 2012, 28, 3633–3640. [Google Scholar] [CrossRef] [PubMed]
- Otten, A.; Koster, S.; Struth, B.; Snigirev, A.; Pfohl, T. Microfluidics of soft matter investigated by small-angle X-ray scattering. J. Synchrotron Radiat. 2005, 12, 745–750. [Google Scholar] [CrossRef] [PubMed]
- Dootz, R.; Otten, A.; Koster, S.; Struth, B.; Pfohl, T. Evolution of DNA compaction in microchannels. J. Phys. Condes. Matter. 2006, 18, S639–S652. [Google Scholar] [CrossRef]
- Koh, C.; Zhang, X.; Liu, S.; Golan, S.; Yu, B.; Yang, X.; Guan, J.; Jin, Y.; Talmon, Y.; Muthusamy, N. Delivery of antisense oligodeoxyribonucleotide lipopolyplex nanoparticles assembled by microfluidic hydrodynamic focusing. J. Control Release 2010, 141, 62–69. [Google Scholar] [CrossRef] [PubMed]
- Tran, T.; Nguyen, C.; Kim, D.; Lee, Y.; Huh, K. Microfluidic approach for highly efficient synthesis of heparin-based bioconjugates for drug delivery. Lab Chip 2012, 12, 589–594. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Jiang, J. Droplet microfluidic technology: Mirodroplets formation and manipulation. Chin. J. Anal. Chem. 2012, 40, 1293–1300. [Google Scholar] [CrossRef]
- Park, S.; Zhang, Y.; Lin, S.; Wang, T.; Yang, S. Advances in microfluidic PCR for point-of-care infectious disease diagnostics. Biotechnol. Adv. 2011, 29, 830–839. [Google Scholar] [CrossRef] [PubMed]
- Basova, E.; Foret, F. Droplet microfluidics in (bio) chemical analysis. Analyst 2015, 140, 22–38. [Google Scholar] [CrossRef] [PubMed]
- Chou, W.; Lee, P.; Yang, C.; Huang, W.; Lin, Y. Recent advances in applications of droplet microfluidics. Micromachines 2015, 6, 1249–1271. [Google Scholar] [CrossRef]
- Kim, S.; Sukovich, D.; Abate, A. Patterning microfluidic device wettability with spatially-controlled plasma oxidation. Lab Chip 2015, 15, 3163–3169. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Li, S.; Tan, J.; Wang, Y.; Luo, G. Controllable preparation of monodisperse O/W and W/O emulsions in the same microfluidic device. Langmuir 2006, 22, 7943–7946. [Google Scholar] [CrossRef] [PubMed]
- Zhao, C.; Middelberg, A. Two-phase microfluidic flows. Chem. Eng. Sci. 2011, 66, 1394–1411. [Google Scholar] [CrossRef]
- De Menech, M.; Garstecki, P.; Jousse, F.; Stone, H. Transition from squeezing to dripping in a microfluidic T-shaped junction. J. Fluid Mech. 2008, 595, 141–161. [Google Scholar] [CrossRef]
- Teh, S.; Lin, R.; Hung, L.; Lee, A. Droplet microfluidics. Lab Chip 2008, 8, 198–220. [Google Scholar] [CrossRef] [PubMed]
- Murshed, S.; Tan, S.; Nguyen, N.; Wong, T.; Yobas, L. Microdroplet formation of water and nanofluids in heat-induced microfluidic T-junction. Microfluid. Nanofluid. 2009, 6, 253–259. [Google Scholar] [CrossRef]
- Lin, B.; Su, Y. On-demand liquid-in-liquid droplet metering and fusion utilizing pneumatically actuated membrane valves. J. Microelectromech. Syst. 2008, 18, 115005. [Google Scholar] [CrossRef]
- Utada, A.; Fernandez-Nieves, A.; Stone, H.; Weitz, D. Dripping to jetting transitions in coflowing liquid streams. Phys. Rev. Lett. 2007, 99, 094502. [Google Scholar] [CrossRef] [PubMed]
- Anna, S.; Bontoux, N.; Stone, H. Formation of dispersions using “flow focusing” in microchannels. Appl. Phys. Lett. 2003, 82, 364–366. [Google Scholar] [CrossRef]
- Nie, Z.; Xu, S.; Seo, M.; Lewis, P.; Kumacheva, E. Polymer particles with various shapes and morphologies produced in continuous microfluidic reactors. J. Am. Chem. Soc. 2005, 127, 8058–8063. [Google Scholar] [CrossRef] [PubMed]
- Garstecki, P.; Gañán-Calvo, A.; Whitesides, G. Formation of bubbles and droplets in microfluidic systems. Bull. Polish Acad. Sci. Tech. Sci. 2005, 53, 361–372. [Google Scholar]
- Schneider, T.; Chapman, G.; Häfeli, U. Effects of chemical and physical parameters in the generation of microspheres by hydrodynamic flow focusing. Colloids Surf. B 2011, 87, 361–368. [Google Scholar] [CrossRef] [PubMed]
- Tan, S.H.; Maes, F.; Semin, B.; Vrignon, J.; Baret, J. The Microfluidic Jukebox. Sci. Rep. 2014, 4, 597–600. [Google Scholar] [CrossRef] [PubMed]
- Tan, S.H.; Nguyen, N.T. Generation and manipulation of monodispersed ferrofluid emulsions: The effect of a uniform magnetic field in flow-focusing and T-junction configurations. Phys. Rev. E 2011, 84, 2299–2304. [Google Scholar] [CrossRef] [PubMed]
- Zeng, S.; Li, B.; Su, X.; Qin, J.; Lin, B. Microvalve-actuated precise control of individual droplets in microfluidic devices. Lab Chip 2009, 9, 1340–1343. [Google Scholar] [CrossRef] [PubMed]
- Ma, Z.; Teo, A.; Tan, S.; Ai, Y.; Nguyen, N.T. Self-aligned interdigitated transducers for acoustofluidics. Micromachines 2016, 7, 216. [Google Scholar] [CrossRef]
- Tan, S.; Sohel Murched, S.; Nguyen, N.; Neng, T.; Wong, T.; Yobas, L. Thermally controlled droplet formation in flow focusing geometry: Formation regimes and effect of nanoparticle suspension. J. Phys. D Appl. Phys. 2008, 41, 165501. [Google Scholar] [CrossRef]
- Othman, R.; Vladisavljević, G.; Bandulasena, H.; Nagy, Z. Production of polymeric nanoparticles by micromixing in a co-flow microfluidic glass capillary device. Chem. Eng. J. 2015, 280, 316–329. [Google Scholar] [CrossRef]
- Dang, T.; Kim, Y.; Kim, H.; Kim, G. Preparation of monodisperse PEG hydrogel microparticles using a microfluidic flow-focusing device. J. Ind. Eng. Chem. 2012, 18, 1308–1313. [Google Scholar] [CrossRef]
- Cohen, C.; Giles, R.; Sergeyeva, V.; Mittal, N.; Tabeling, P.; Zerrouki, D.; Baudry, J.; Bibette, J.; Bremond, N. Parallelised production of fine and calibrated emulsions by coupling flow-focusing technique and partial wetting phenomenon. Microfluid. Nanofluid. 2016, 17, 959–966. [Google Scholar] [CrossRef]
- Garstecki, P.; Fuerstman, M.; Stone, H.; Whitesides, G. Formation of droplets and bubbles in a microfluidic T-junction-scaling and mechanism of break-up. Lab Chip 2006, 6, 437–446. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Fu, T.; Ma, Y.; Li, H. Active control of ferrofluid droplet breakup dynamics in a microfluidic T-junction. Microfluid. Nanofluid. 2015, 18, 19–27. [Google Scholar] [CrossRef]
- Chokkalingam, V.; Herminghaus, S.; Seemann, R. Self-synchronizing pairwise production of monodisperse droplets by microfluidic step emulsification. Appl. Phys. Lett. 2008, 93, 254101. [Google Scholar] [CrossRef]
- Lucas Frenz, L.; El Harrak, A.; Pauly, M.; Bégin-Colin, S.; Griffiths, A.; Baret, J. Droplet-based microreactors for the synthesis of magnetic iron oxide nanoparticles. Angew. Chem. Int. 2008, 47, 6817–6820. [Google Scholar] [CrossRef] [PubMed]
- Sammer-ul, H. Droplet-based microfluidics as a potential platform for point-of-care diagnostics. Austin J. Biosens. Bioelectron. 2015, 1, 1013. [Google Scholar]
- Link, D.; Grasland-Mongrain, E.; Duri, A.; Sarrazin, F.; Cheng, Z.; Cristobal, G. Electric control of droplets in microfluidic devices. Angew. Chem. Int. Ed. Engl. 2006, 45, 2556–2560. [Google Scholar] [CrossRef] [PubMed]
- Berthier, J.; Brakke, K. The Physics of Microdroplets; John Wiley: Hoboken, NJ, USA, 2012. [Google Scholar]
- Choi, K.; Ng, A.H.C.; Fobel, R. Digital microfluidics. Annu. Rev. Anal. Chem. 2012, 5, 413–440. [Google Scholar] [CrossRef] [PubMed]
- Song, H.; Bringer, M.R.; Tice, J.D. Experimental test of scaling of mixing by chaotic advection in droplets moving through microfluidic channels. Appl. Phys. Lett. 2003, 83, 4664–4666. [Google Scholar] [CrossRef] [PubMed]
- Dressler, O.; Maceiczyk, R.; Chang, S.; de Mello, A. Droplet-based microfluidics: Enabling impact on drug discovery. J. Biomol. Screen. 2014, 19, 483–496. [Google Scholar] [CrossRef] [PubMed]
- Song, H.; Chen, D.; Ismagilov, R. Reactions in droplets in microfluidic channels. Angew. Chem. Int. Ed. Engl. 2006, 45, 7336–7356. [Google Scholar] [CrossRef] [PubMed]
- Sung, J.; Kam, C.; Shuler, M. A microfluidic device for a pharmacokinetic-pharmacodynamic (PK-PD) model on a chip. Lab Chip 2010, 10, 446–455. [Google Scholar] [CrossRef] [PubMed]
- Champion, J.; Katare, Y.; Mitragotri, S. Particle shape: A new design parameter for micro- and nanoscale drug delivery carriers. J. Control Release 2007, 121, 3–9. [Google Scholar] [CrossRef] [PubMed]
- Capretto, L.; Mazzitelli, S.; Nastruzzi, C. Design, production and optimization of solid lipid microparticles (SLM) by a coaxial microfluidic device. J. Control Release 2012, 160, 409–417. [Google Scholar] [CrossRef] [PubMed]
- Chen, P.; Erb, R.; Studart, A. Designer polymer-based microcapsules made using microfluidics. Langmuir 2011, 28, 144–152. [Google Scholar] [CrossRef] [PubMed]
- Priest, C.; Quinn, A.; Postma, A.; Zelikin, A.; Ralston, J.; Caruso, F. Microfluidic polymer multilayer adsorption on liquid crystal droplets for microcapsule synthesis. Lab Chip 2008, 8, 2182–2187. [Google Scholar] [CrossRef] [PubMed]
- Tumarkin, E.; Kumacheva, E. Microfluidic generation of microgels from synthetic and natural polymers. Chem. Soc. Rev. 2009, 38, 2161–2168. [Google Scholar] [CrossRef] [PubMed]
- Rosiak, J.M.; Yoshii, F. Hydrogels and their medical applications. Nucl. Instrum. Methods Phys. Res. Sec. B Beam Interact. Mater. Atoms 1999, 151, 56–64. [Google Scholar] [CrossRef]
- Morrison, D.R.; Mosier, B. Externally triggered microcapsules. [NASA/Johnson Space Center]. PCT Int. Appl. 1999, 99, 59556. [Google Scholar]
- Sirsi, S.; Borden, M. Microbubble compositions, properties and biomedical applications. Bubble Sci. Eng. Technol. 2009, 1, 3–17. [Google Scholar] [CrossRef] [PubMed]
- Plamper, F.A.; Richtering, W. Functional microgels and microgel systems. Acc. Chem. Res. 2017, 50, 131–140. [Google Scholar] [CrossRef] [PubMed]
- Chung, T.; Huang, Y.; Liu, Y. Effects of the rate of solvent evaporation on the characteristics of drug loaded PLLA and PDLLA microspheres. Int. J. Pharm. 2001, 212, 161–169. [Google Scholar] [CrossRef]
- Özkan, A.; Nikdoost, A.; Keita, H.; Tuncel, D.; Erdem, Y. Synthesis of conjugated polymer nanoparticles in droplet-based microreactor. In Proceedings of the 11th Nanoscience and Nanotechnology Conference (NanoTR-11), Ankara, Turkey, 22–25 June 2015. [Google Scholar]
- Jelinek, R.; Silbert, L. Biomimetic approaches for studying membrane processes. Mol. BioSyst. 2009, 5, 811–818. [Google Scholar] [CrossRef] [PubMed]
- Seddon, A.; Curnow, P.; Booth, P. Membrane proteins, lipids and detergents: Not just a soap opera. Biochim. Biophys. Acta 2004, 1666, 105–117. [Google Scholar] [CrossRef] [PubMed]
- Perrin, P. Amphiphilic Copolymers: A new route to prepare ordered monodisperse emulsions. Langmuir 1998, 14, 5977–5979. [Google Scholar] [CrossRef]
- Herbert, A.; Martin, M.; Gilbert, S. Pharmaceutical Emulsions and Microemulsions, Pharmaceutical Dosage Forms Disperse Systems; Marcel Dekker, Inc.: New York, NY, USA; Basel, Switzerland, 1996; pp. 67–95. [Google Scholar]
- Christopher, A.; Dawn, B. Pharmaceutical Compounding and DispenSing; RPS: Cambridge, UK, 2008; pp. 65–70. [Google Scholar]
- Leon, S.; Alan, H.; Paul, F.; Larry, N. Comprehensive Pharmacy Review; Lipponcott Williams and Wilkins: Philadelphia, PA, USA, 2004; pp. 47–51. [Google Scholar]
- Nita, A.; Rouzhou, H.; Richard, A. Performance of a rotating membrane emulsifier for production of coarse droplets. J. Membrane. Sci. 2009, 326, 9–18. [Google Scholar]
- Utada, A.; Lorenceau, E.; Link, D.; Kaplan, P.; Stone, H.; Weitz, D. Monodisperse double emulsions generated from a microcapillary device. Science 2005, 308, 537–541. [Google Scholar] [CrossRef] [PubMed]
- Chu, L.; Utada, A.; Shah, R.; Kim, J.; Weitz, D. Controllable monodisperse multiple emulsions. Angew. Chem. Int. Ed. 2007, 46, 8970–8974. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Shum, H.; Weitz, D. Fabrication of monodisperse toroidal particles by polymer solidification in microfluidics. Chem. Phys. Chem. 2009, 10, 641–645. [Google Scholar] [CrossRef] [PubMed]
- Teh, S.; Khnouf, R.; Fan, H.; Lee, A. Stable, biocompatible lipid vesicle generation by solvent extraction-based droplet microfluidics. Biomicrofluidics 2011, 5, 044113. [Google Scholar] [CrossRef] [PubMed]
- Herranz-Blanco, B.; Arriaga, L.; Mäkilä, E.; Correia, A.; Shrestha, N.; Mirza, S.; Weitz, S.; Salonen, J.; Hirvonen, J.; Santos, H. Microfluidic assembly of multistage porous silicon-lipid vesicles for controlled drug release. Lab Chip 2014, 14, 1083–1086. [Google Scholar] [CrossRef] [PubMed]
- Pessi, J.; Santos, H.; Miroshnyk, I.; Yliruusi, J.; Weitz, D.; Mirza, S. Microfluidics-assisted engineering of polymeric microcapsules with high encapsulation efficiency for protein drug delivery. Int. J. Pharm. 2014, 472, 82–87. [Google Scholar] [CrossRef] [PubMed]
- Imura, T.; Gotoh, T.; Otaka, K.; Yoda, S.; Takebayashi, Y.; Yokoyama, S.; Takebayashi, H.; Sakai, H.; Abe, M. Control of physicochemical properties of liposomes using a supercritical reverse phase evaporation method. Langmuir 2003, 19, 2021–2025. [Google Scholar] [CrossRef]
- Nguyen, T.; Tang, Q.; Doan, D.; Dang, D. Micro and nano liposome vesicles containing curcumin for a drug delivery system. Adv. Nat. Sci. Nanosci. Nanotechnol. 2016, 7, 035003. [Google Scholar] [CrossRef]
- Meure, L.; Foster, N.; Dehghani, F. Conventional and dense gas techniques for the production of liposomes: A review. AAPS PharmSciTech 2008, 9, 798–809. [Google Scholar] [CrossRef] [PubMed]
- Martz, T.; Bardin, D.; Sheeran, P.; Lee, A.; Dayton, P. Microfluidic generation of acoustically active nanodroplets. Small 2012, 8, 1876–1879. [Google Scholar] [CrossRef] [PubMed]
- Davies, R.; Kim, D.; Park, J. Formation of liposomes using a 3D flow focusing microfluidic device with spatially patterned wettability by corona discharge. J. Micromech. Microeng. 2012, 22, 5. [Google Scholar] [CrossRef]
- Jahn, A.; Vreeland, W.; Gaitan, M.; Locascio, L. Controlled vesicle self-assembly in microfluidic channels with hydrodynamic focusing. J. Am. Chem. Soc. 2004, 126, 2674–2675. [Google Scholar] [CrossRef] [PubMed]
- Jahn, A.; Vreeland, W.; DeVoe, D.; Locascio, L.; Gaitan, M. Microfluidic directed formation of liposomes of controlled size. Langmuir 2007, 23, 6289–6293. [Google Scholar] [CrossRef] [PubMed]
- Hood, R.; Vreeland, W.; DeVoe, D. Microfluidic remote loading for rapid single-step liposomal drug preparation. Lab Chip 2014, 14, 3359–3367. [Google Scholar] [CrossRef] [PubMed]
- Drummond, D.; Hong, O.; Kirpotin, D.; Papahadjopoulos, D. Optimizing liposomes for delivery of chemotherapeutic agents to solid tumors. Pharmacol. Rev. 1999, 51, 691–743. [Google Scholar] [PubMed]
- Andar, A.; Hood, R.; Vreeland, W.; DeVoe, D.; Swaan, P. Microfluidic preparation of liposomes to determine particle size influence on cellular uptake mechanisms. Pharmaceut. Res. 2014, 31, 401–413. [Google Scholar] [CrossRef] [PubMed]
- Fan, Y.; Zhang, Q. Development of liposomal formulations: From concept to clinical investigations. Asian J. Pharm. Sci. 2013, 8, 79–90. [Google Scholar] [CrossRef]
- Felgner, J.; Kumar, R.; Sridhar, C.N.; Wheeler, C.; Tsai, Y.; Border, R.; Ramsey, P.; Martin, M.; Felgner, P. Enhanced gene delivery and mechanism studies with a novel series of cationic lipid formulations. J. Biol. Chem. 1994, 269, 2550–2561. [Google Scholar] [PubMed]
- Herweijer, H.; Wolff, J. Gene therapy progress and prospects: Hydrodynamic gene delivery. Gene Ther. 2006, 14, 99–107. [Google Scholar] [CrossRef] [PubMed]
- Studart, A.; Shum, H.; Weitz, D. Arrested coalescence of particle-coated droplets into nonspherical supracolloidal structures. J. Phys. Chem. B 2009, 113, 3914–3919. [Google Scholar] [CrossRef] [PubMed]
- Annabi, N.; Tamayol, A.; Uquillas, J.; Akbari, M.; Bertassoni, L.; Cha, C.; Camci-Unal, G.; Dokmeci, M.; Peppas, N.; Khademhosseini, A. Rational design and applications of hydrogels in regenerative medicine. Adv. Mater. 2014, 26, 85–124. [Google Scholar] [CrossRef] [PubMed]
- Xu, S.; Nie, Z.; Seo, M.; Lewis, P.; Kumacheva, E.; Stone, H.A.; Garstecki, P.; Weibel, D.B.; Gitlin, I.; Whitesides, G.M. Generation of monodisperse particles by using microfluidics: Control over size, shape, and composition. Angew. Chem. 2005, 117, 734–738. [Google Scholar] [CrossRef]
- Dendukuri, D.; Tsoi, K.; Hatton, T.A.; Doyle, P.S. Controlled synthesis of nonspherical microparticles using microfluidics. Langmuir 2005, 21, 2113–2116. [Google Scholar] [CrossRef] [PubMed]
- Mathaes, R.; Winter, G.; Besheer, A.; Engert, J. Non-spherical micro- and nanoparticles: Fabrication, characterization and drug delivery applications. Expert Opin. Drug Deliv. 2015, 12, 481–492. [Google Scholar] [CrossRef] [PubMed]
- Geng, Y.; Dalhaimer, P.; Cai, S.; Tsai, R.; Tewari, M.; Minko, T.; Discher, D. Shape effects of filaments versus spherical particles in flow and drug delivery. Nat. Nanotechnol. 2007, 2, 249–255. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Cai, W.; He, L.; Nakayama, N.; Chen, K.; Sun, X.; Chen, X.; Dai, H. In vivo biodistribution and highly efficient tumour targeting of carbon nanotubes in mice. Nat. Nanotechnol. 2007, 2, 47–52. [Google Scholar] [CrossRef] [PubMed]
- Kolhar, P.; Anselmo, A.; Gupta, V.; Pant, K.; Prabhakarpandian, B.; Ruoslahti, E.; Mitragotri, S. Using shape effects to target antibody-coated nanoparticles to lung and brain endothelium. Proc. Natl. Acad. Sci. USA 2013, 110, 10753–10758. [Google Scholar] [CrossRef] [PubMed]
- Dendukuri, D.; Pregibon, D.C.; Collins, J.; Hatton, T.A.; Doyle, P.S. Continuous-flow lithography for high-throughput microparticle synthesis. Nature Mat. 2006, 5, 365–369. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Li, Y.; Wang, X.; Wang, J.; Tian, H.; Zhao, P.; Tian, Y.; Gu, Y.; Wang, L.; Wang, C. Droplet microfluidics for the production of microparticles and nanoparticles. Micromachines 2017, 8, 22. [Google Scholar] [CrossRef]
- Koh, C.; Kang, X.; Xie, Y.; Fei, Z.; Guan, J.; Yu, B.; Zhang, X.; Lee, L. Delivery of polyethylenimine/ DNA complexes assembled in a microfluidics device. Mol. Pharm. 2009, 6, 1333–1342. [Google Scholar] [CrossRef] [PubMed]
- Kuramoto, H.; Park, Y.; Kaji, N.; Tokeshi, M.; Kogure, K.; Shinohara, Y.; Harashima, H.; Baba, Y. On chip fabrication of mutifunctional envelope-type nanodevices for gene delivery. Anal. Bioanal. Chem. 2008, 391, 2729–2733. [Google Scholar] [CrossRef] [PubMed]
- Balbino, T.; Aoki, N.; Gasperini, A.; Oliveira, C.; Azzoni, A.; Cavalcanti, L.; Torre, L. Continuous flow production of cationic liposomes at high lipid concentration in microfluidic devices for gene delivery applications. Chem. Eng. J. 2013, 226, 423–433. [Google Scholar] [CrossRef]
- Hsieh, A.; Hori, N.; Massoudi, R.; Pan, P.; Sasaki, H.; Lin, Y.; Lee, A. Nonviral gene vector formation in monodispersed picolitre incubator for consistent gene delivery. Lab Chip 2009, 9, 2638–2643. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Dong, X.; Qin, J.; Lin, B. Rapid nanoliter DNA hybridization based on reciprocating flow on a compact disk microfluidic device. Anal. Chim. Acta 2009, 640, 93–99. [Google Scholar] [CrossRef] [PubMed]
- Georgi, V.; Georgi, L.; Blechert, M.; Bergmeister, M.; Zwanzig, M.; Wüstenhagen, D.; Bier, F.; Jung, E.; Kubick, S. On-chip automation of cell-free protein synthesis: New opportunities due to a novel reaction mode. Lab Chip 2016, 16, 269–281. [Google Scholar] [CrossRef] [PubMed]
- Orth, J.; Schorch, B.; Boundy, S.; Ffrench-Constant, R.; Kubick, S.; Aktories, K. Cell-free synthesis and characterization of a novel cytotoxic pierisin-like protein from the cabbage butterfly Pieris rapae. Toxicon 2011, 57, 199–207. [Google Scholar] [CrossRef] [PubMed]
- Goshima, N.; Kawamura, Y.; Fukumoto, A.; Miura, A.; Honma, R.; Satoh, R.; Wakamatsu, A.; Yamamoto, J.; Kimura, K.; Nishikawa, T.; et al. Human protein factory for converting the transcriptome into an in vitro-expressed proteome. Nat. Methods 2008, 5, 1011–1017. [Google Scholar] [CrossRef] [PubMed]
- Fenz, S.F.; Sachse, R.; Schmidt, T.; Kubick, S. Cell-free synthesis of membrane proteins: Tailored cell models out of microsomes. Biochim. Biophys. Acta 2014, 1838, 1382–1388. [Google Scholar] [CrossRef] [PubMed]
- Sachse, S.; Dondapati, S.; Fenz, S.; Schmidt, T.; Kubick, S. Membrane protein synthesis in cell-free systems: From bio-mimetic systems to bio-membranes. FEBS Lett. 2014, 588, 2774–2781. [Google Scholar] [CrossRef] [PubMed]
- Mei, Q.; Fredrickson, C.; Simon, A.; Khnouf, R.; Fa, Z. Cell-free protein synthesis in microfluidic array devices. Biotechnol. Progress 2007, 23, 1305–1311. [Google Scholar] [CrossRef] [PubMed]
- Jackson, K.; Khnouf, R.; Fan, Z. Cell-free protein synthesis in microfluidic 96-well plates. In Cell-Free Protein Synthesis; Methods in Molecular Biology (Methods and Protocols); Alexandrov, K., Johnston, W., Eds.; Humana Press: Totowa, NJ, USA, 2014; Volume 1118. [Google Scholar]
- Mazutis, L.; Baret, J.-C.; Treacy, P. MultiStep microfluidic droplet processing: Kinetic analysis of an in vitro translated enzyme. Lab Chip 2009, 9, 2902–2908. [Google Scholar] [CrossRef] [PubMed]
- Fallah-Araghi, A.; Baret, J.; Ryckelynck, M.; Griffiths, A. A completely in vitro ultrahigh-throughput droplet-based microfluidic screening system for protein engineering and directed evolution. Lab Chip 2012, 12, 882–891. [Google Scholar] [CrossRef] [PubMed]
- Timm, A.; Shankles, P.; Foster, C.; Doktycz, M.; Rettere, S. Toward microfluidic reactors for cell-free protein synthesis at the point-of-care. Small 2016, 12, 810–817. [Google Scholar] [CrossRef] [PubMed]
- Gottesman, M. Mechanisms of cancer drug resistance. Annu. Rev. Med. 2002, 53, 615–627. [Google Scholar] [CrossRef] [PubMed]
- Bhadriraju, K.; Chen, C. Engineering cellular microenvironments to improve cell-based drug testing. Drug Discov. Today 2002, 7, 612–620. [Google Scholar] [CrossRef]
- Miller, O.; El Harrak, A.; Mangeat, T.; Baret, J.; Frenz, L.; El Debs, B.; Mayot, E.; Samuels, M.; Rooney, E.; Dieu, P.; et al. High-resolution dose–response screening using droplet-based microfluidics. Proc. Natl. Acad. Sci. USA 2012, 109, 378–383. [Google Scholar] [CrossRef] [PubMed]
- Sugiura, S.; Edahiro, J.; Kikuchi, K.; Sumaru, K.; Kanamori, T. Pressure-driven perfusion culture microchamber array for a parallel drug cytotoxicity assay. Biotechnol. Bioeng. 2008, 100, 1156–1165. [Google Scholar] [CrossRef] [PubMed]
- Wu, M.; Huang, S.; Lee, G. Microfluidic cell culture systems for drug research. Lab Chip 2010, 10, 939–956. [Google Scholar] [CrossRef] [PubMed]
- Sundberg, S. High-throughput and ultra-high-throughput screening: Solution- and cell-based approaches. Curr. Opin. Biotechnol. 2000, 11, 47–53. [Google Scholar] [CrossRef]
- Sarkar, S.; Cohen, N.; Sabhachandani, P.; Konry, T. Phenotypic drug profiling in droplet microfluidics for better targeting of drug-resistant tumors. Lab Chip 2015, 15, 4441–4450. [Google Scholar] [CrossRef] [PubMed]
- Lee, P.; Ghorashian, N.; Gaige, T.; Hung, P. Microfluidic system for automated cell-based assays. J. Lab. Autom. 2007, 12, 363–367. [Google Scholar] [CrossRef] [PubMed]
- Ziolkowska, K.; Stelmachowska, A.; Kwapiszewski, R.; Chudy, M.; Dybko, A.; Brzozka, Z. Long-term three-dimensional cell culture and anticancer drug activity evaluation in a microfluidic chip. Biosens. Bioelectron. 2013, 40, 68–74. [Google Scholar] [CrossRef] [PubMed]
- Shin, Y.; Han, S.; Jeon, J.; Yamamoto, K.; Zervantonakis, I.; Sudo, R.; Kamm, R.; Chung, S. Microfluidic assay for simultaneous culture of multiple cell types on surfaces or within hydrogels. Nat. Protoc. 2012, 7, 1247–1259. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.; Yeon, J.; Park, J. A microfluidic platform for 3-dimensional cell culture and cellbased assays. Biomed. Microdevices 2007, 9, 25–34. [Google Scholar] [CrossRef] [PubMed]
- Jeong, G.; Kwon, G.; Kang, A.; Jung, B.; Park, Y.; Chung, S.; Lee, S. Microfluidic assay of endothelial cell migration in 3D interpenetrating polymer semi-network HA-Collagen hydrogel. Biomed. Microdevices. 2011, 13, 717–723. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.; Gupta, M.; Cheung, K. Alginate-based microfluidic system for tumor spheroid formation and anticancer agent screening. Biomed. Microdevices 2010, 12, 647–654. [Google Scholar] [CrossRef] [PubMed]
- Bhatia, S.; Ingber, D. Microfluidic organs-on-chips. Nat. Biotechnol. 2014, 32, 760–772. [Google Scholar] [CrossRef] [PubMed]
- Joshi, P. Cells and organs on chip—A revolutionary platform for biomedicine. In Lab-On-A-Chip Fabrication and Application; Stoytcheva, M., Ed.; InTech: Rijeka, Croatia, 2016. [Google Scholar]
- Kodzius, R.; Schulze, F.; Gao, Z.; Schneider, M. Organ-on-chip technology: Current state and future developments. Genes 2017, 8, 266. [Google Scholar] [CrossRef] [PubMed]
- Schulze, F.; Gao, X.; Virzonis, D.; Damiati, S.; Schneider, M.R.; Kodzius, R. Air quality effects on human health and approaches for its assessment through microfluidic chips. Genes 2017, 8, 244. [Google Scholar] [CrossRef] [PubMed]
- Nalayanda, D.; Puleo, C.; Fulton, W.; Sharpe, L.; Wang, T.; Abdullah, F. An open-access microfluidic model for lung-specific functional studies at an air-liquid interface. Biomed. Microdevices. 2009, 11, 1081–1089. [Google Scholar] [CrossRef] [PubMed]
- Huh, D.; Matthews, B.; Mammoto, A.; Montoya-Zavala, M.; Hsin, H.; Ingber, D. Reconstituting organ-level lung functions on a chip. Science 2010, 328, 1662–1668. [Google Scholar] [CrossRef] [PubMed]
- Douville, N.; Zamankhan, P.; Tung, Y.; Li, R.; Vaughan, B.; Tai, C.; White, J.; Christensen, P.; Grotberg, J.; Takayama, S. Combination of fluid and solid mechanical stresses contribute to cell death and detachment in a microfluidic alveolar model. Lab Chip 2011, 11, 609–619. [Google Scholar] [CrossRef] [PubMed]
- Arany, I.; Safirstein, R. Cisplatin nephrotoxicity. Semin. Nephrol. 2003, 23, 460–464. [Google Scholar] [CrossRef]
- Jang, K.; Suh, K. A multi-layer microfluidic device for efficient culture and analysis of renal tubular cells. Lab Chip 2010, 10, 36–42. [Google Scholar] [CrossRef] [PubMed]
- Lee, P.; Hung, P.; Lee, L. An artificial liver sinusoid with a microfluidic endothelial-like barrier for primary hepatocyte culture. Biotechnol. Bioeng. 2007, 97, 1340–1346. [Google Scholar] [CrossRef] [PubMed]
- Sung, K.; Vanapalli, S.; Mukhija, D.; McKay, H.; Millunchick, J.; Burns, M.; Solomon, M. Programmable fluidic production of microparticles with configurable anisotropy. J. Am. Chem. Soc. 2008, 130, 1335–1340. [Google Scholar] [CrossRef] [PubMed]
- Khetani, S.; Bhatia, S. Microscale culture of human liver cells for drug development. Nat. Biotechnol. 2008, 26, 120–126. [Google Scholar] [CrossRef] [PubMed]
- Grosberg, A.; Alford, P.; McCain, M.; Parker, K. Ensembles of engineered cardiac tissue for physiological and pharmacological study: Heart on a chip. Lab Chip 2011, 11, 4165–4173. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.; Huh, D.; Hamilton, G.; Ingber, D. Human gut-on-a-chip inhabited by microbial flora that experiences intestinal peristalsis-like motions and flow. Lab Chip 2012, 12, 2165–2174. [Google Scholar] [CrossRef] [PubMed]
- Deosarkar, S.; Prabhakarpandian, B.; Wang, B.; Sheffield, J.; Krynska, B.; Kiani, M. A novel dynamic neonatal blood-brain barrier on a chip. PLoS ONE 2015, 10, e0142725. [Google Scholar] [CrossRef] [PubMed]
- Huh, D.; Hamilton, G.; Ingber, D. From 3D cell culture to organs-on-chips. Trends Cell Biol. 2011, 21, 745–754. [Google Scholar] [CrossRef] [PubMed]
- Esch, M.; King, T.; Shuler, M. The role of body-on-a-chip devices in drug and toxicity studies. Ann. Rev. Biomed. Eng. 2011, 13, 55–72. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Zhao, Z.; Rahim, N.; van Noort, D.; Yu, H. Towards a human-on-chip: Culturing multiple cell types on a chip with compartmentalized microenvironments. Lab Chip 2009, 9, 3185–3192. [Google Scholar] [CrossRef] [PubMed]
- Liu, D.; Zhang, H.; Fontana, F.; Hirvonen, J.T.; Santos, H. Microfluidic-assisted fabrication of carriers for controlled drug delivery. Lab Chip 2017, 17, 1856–1883. [Google Scholar] [CrossRef] [PubMed]
- Paguirigan, A.L.; Beebe, D.J. Microfluidics meet cell biology: Bridging the gap by validation and application of microscale techniques for cell biological assays. Bioessays 2008, 30, 811–821. [Google Scholar] [CrossRef] [PubMed]
- Marco, L.; Alan, H.T. Synthesis, properties and applications of Janus nanoparticles. Nano Today 2011, 6, 286–308. [Google Scholar]
- Nisisako, T. Recent advances in microfluidic production of Janus droplets and particles. Curr. Opin. Colloid Interface Sci. 2016, 25, 1–12. [Google Scholar] [CrossRef]
- Xie, U.; She, Z.G.; Wang, S.; Sharma, G.; Smith, J.W. One-step fabrication of polymeric janus nanoparticles for drug delivery. Langmuir 2012, 28, 4459–4463. [Google Scholar] [CrossRef] [PubMed]
- Lone, S.; Kim, S.H.; Nam, S.W.; Park, S.; Joo, J.; Cheong, I.W. Microfluidic synthesis of Janus particles by UV-directed phase separation. Chem. Commun. 2011, 47, 2634–2636. [Google Scholar] [CrossRef] [PubMed]
- Fontana, F.; Ferreira, M.P.A.; Correia, A.; Hirvonen, J.T.; Santos, H.A. Microfluidics as a cutting-edge technique for drug delivery applications. Drug Delivery Sci. Technol. 2016, 34, 76–87. [Google Scholar] [CrossRef]
- Cressey, D. Drug-maker plans to cut jobs and spending as industry shies away from drug discovery. Pfizer slashes R&D. Nature 2011, 470, 154. [Google Scholar] [PubMed]
- Lim, J.M.; Swami, A.; Gilson, L.M.; Chopra, S.; Choi, S.; Wu, J.; Langer, R.; Karnik, R.; Farokhzad, O.C. Ultra-high throughput synthesis of nanoparticles with homogeneous size distribution using a coaxial turbulent jet mixer. ACS Nano 2014, 8, 6056–6065. [Google Scholar] [CrossRef] [PubMed]
- Eggersdorfer, M.L.; Zheng, W.; Nawar, S.; Mercandetti, C.; Ofner, A.; Leibacher, I.; Koehler, S.; Weitz, D.A. Tandem emulsification for high-throughput production of double emulsions. Lab Chip 2017, 17, 936–942. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, N.-T.; Hejazian, M.; Ooi, C.H.; Kashaninejad, N. Recent advances and future perspectives on microfluidic liquid handling. Micromachines 2017, 8, 186. [Google Scholar] [CrossRef]
- Sackmann, E.; Fulton, A.; Beebe, D.J. The present and future role of microfluidics in biomedical research. Nature 2014, 507, 181–189. [Google Scholar] [CrossRef] [PubMed]
- Aziz, A.U.R.; Geng, C.; Fu, M.; Yu, X.; Qin, K.; Liu, B. The role of microfluidics for organ on chip simulations. Bioengineering 2017, 4, 39. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.C.; Zhang, Y.S.; Akpek, A.; Shin, S.R.; Khademhosseini, A. 4D bioprinting: The next-generation technology for biofabrication enabled by stimuli-responsive materials. Biofabrication 2017, 9, 012001. [Google Scholar] [CrossRef] [PubMed]

© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Damiati, S.; Kompella, U.B.; Damiati, S.A.; Kodzius, R. Microfluidic Devices for Drug Delivery Systems and Drug Screening. Genes 2018, 9, 103. https://doi.org/10.3390/genes9020103
Damiati S, Kompella UB, Damiati SA, Kodzius R. Microfluidic Devices for Drug Delivery Systems and Drug Screening. Genes. 2018; 9(2):103. https://doi.org/10.3390/genes9020103
Chicago/Turabian StyleDamiati, Samar, Uday B. Kompella, Safa A. Damiati, and Rimantas Kodzius. 2018. "Microfluidic Devices for Drug Delivery Systems and Drug Screening" Genes 9, no. 2: 103. https://doi.org/10.3390/genes9020103
APA StyleDamiati, S., Kompella, U. B., Damiati, S. A., & Kodzius, R. (2018). Microfluidic Devices for Drug Delivery Systems and Drug Screening. Genes, 9(2), 103. https://doi.org/10.3390/genes9020103




