De Novo Analysis Reveals Transcriptomic Responses in Eriobotrya japonica Fruits during Postharvest Cold Storage
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Materials
2.2. RNA Extraction, cDNA Library Construction and Illumina Sequencing
2.3. Transcriptome Assembly and Unigene Annotation
2.4. Gene Expression and Enrichment Analysis
2.5. Cold Regulation of Reversible Protein Phosphorylation and Ca2+ Signal Components
2.6. Cold Regulation of Major Sugars and Polysaccharides Metabolisms
3. Results and Discussion
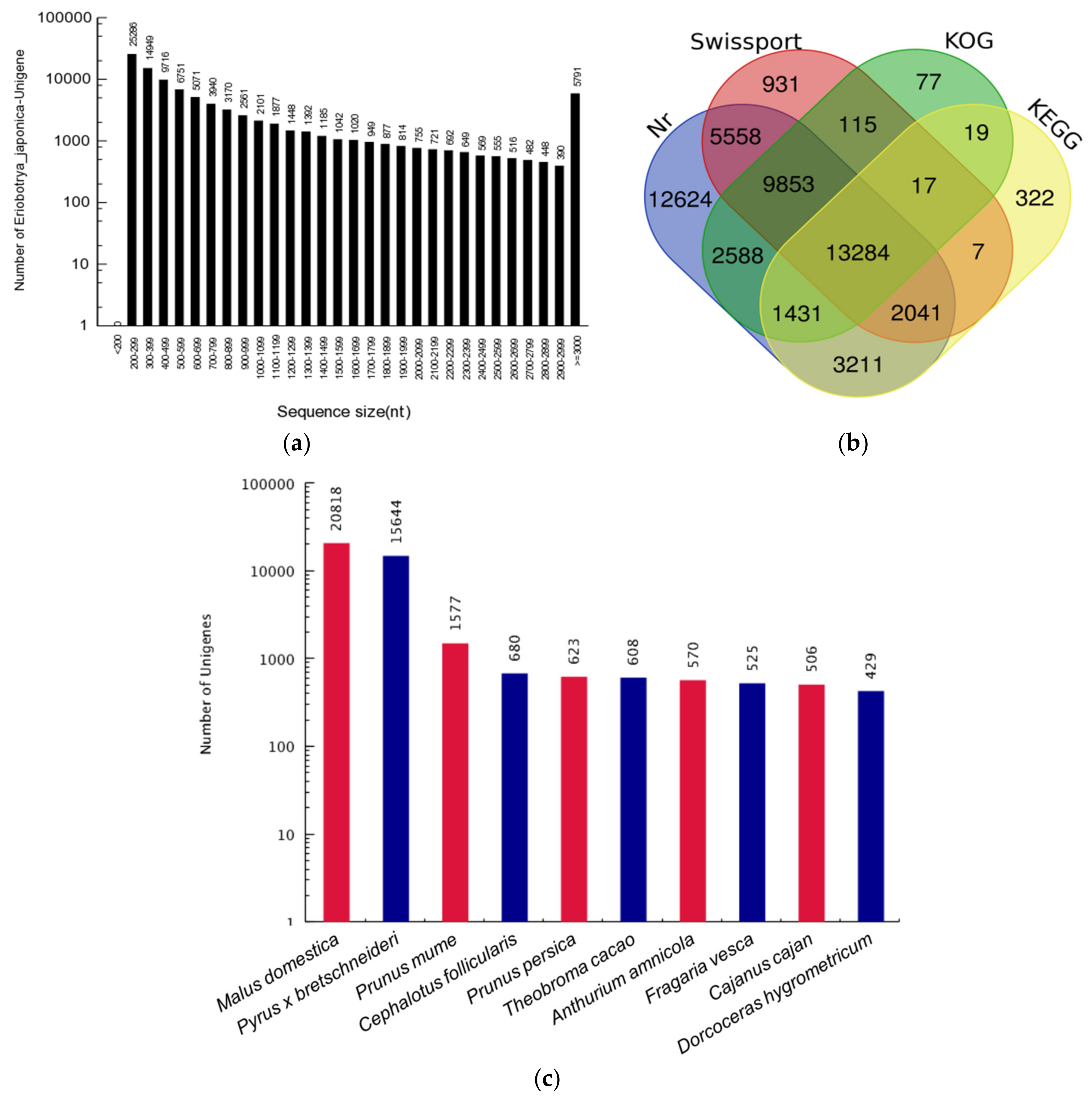
3.1. Transcriptome Assembly and Unigene Annotation
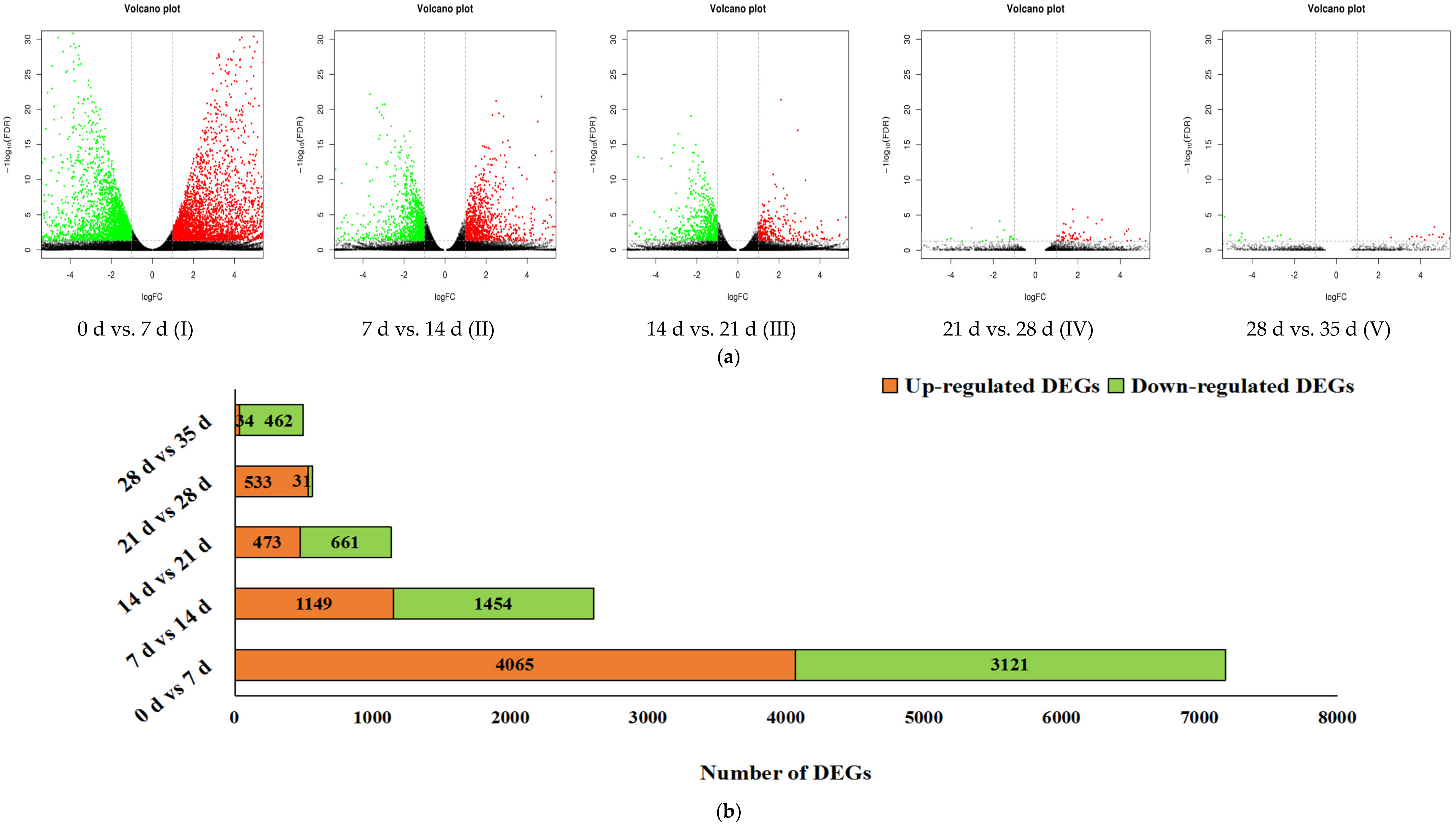
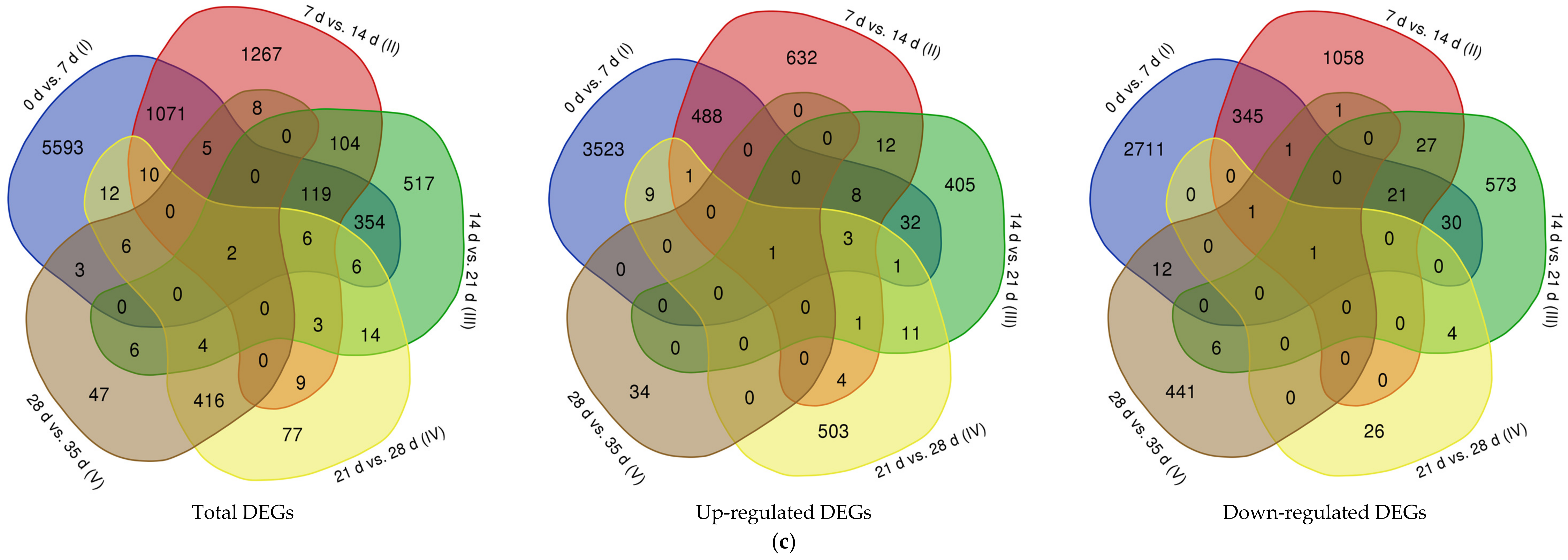
3.2. Gene Expression Analysis and Significant Differentially Expressed Genes (DEGs)
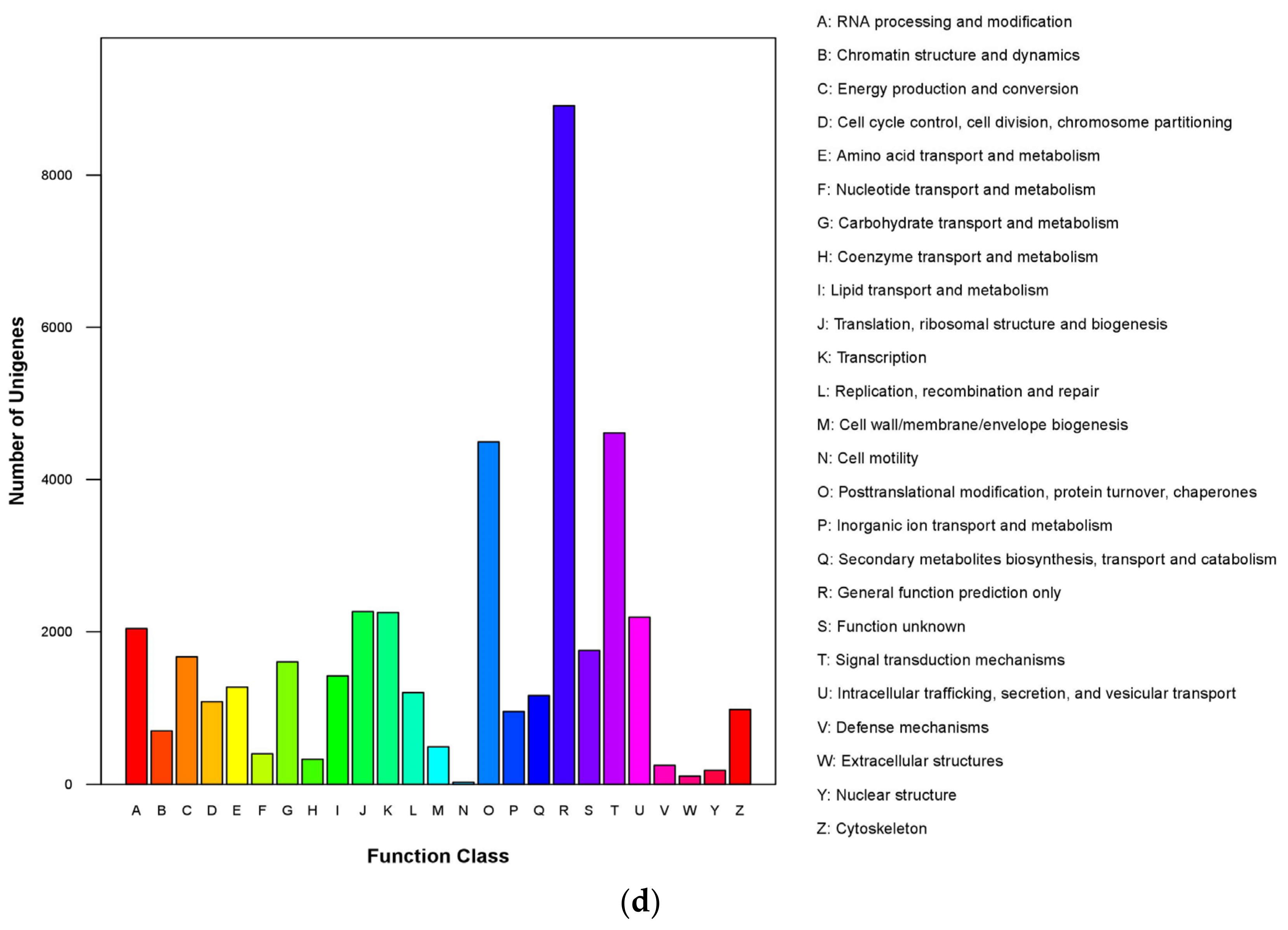
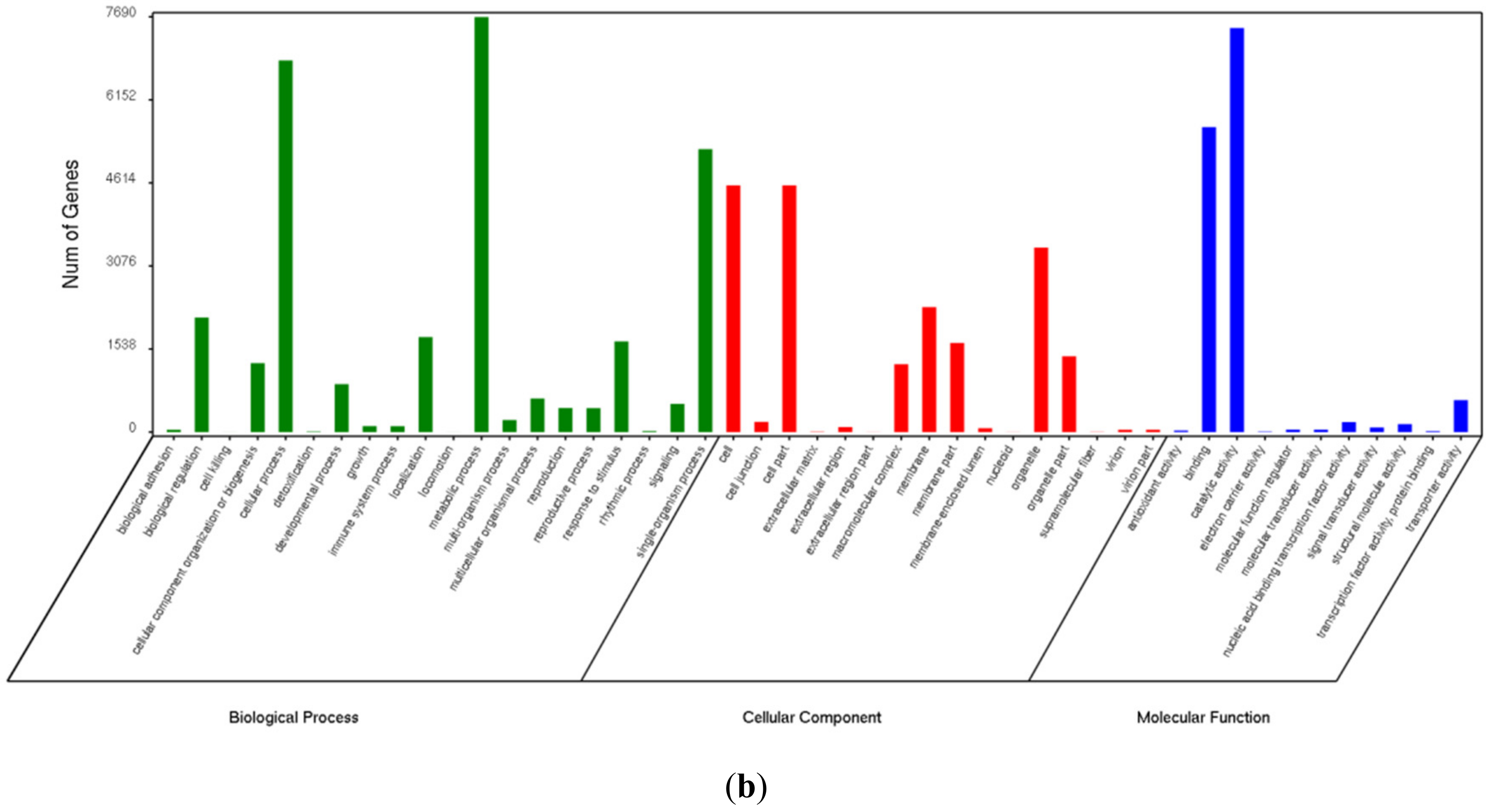
3.3. Gene Ontology Enrichment Analysis of Differentially Expressed Genes
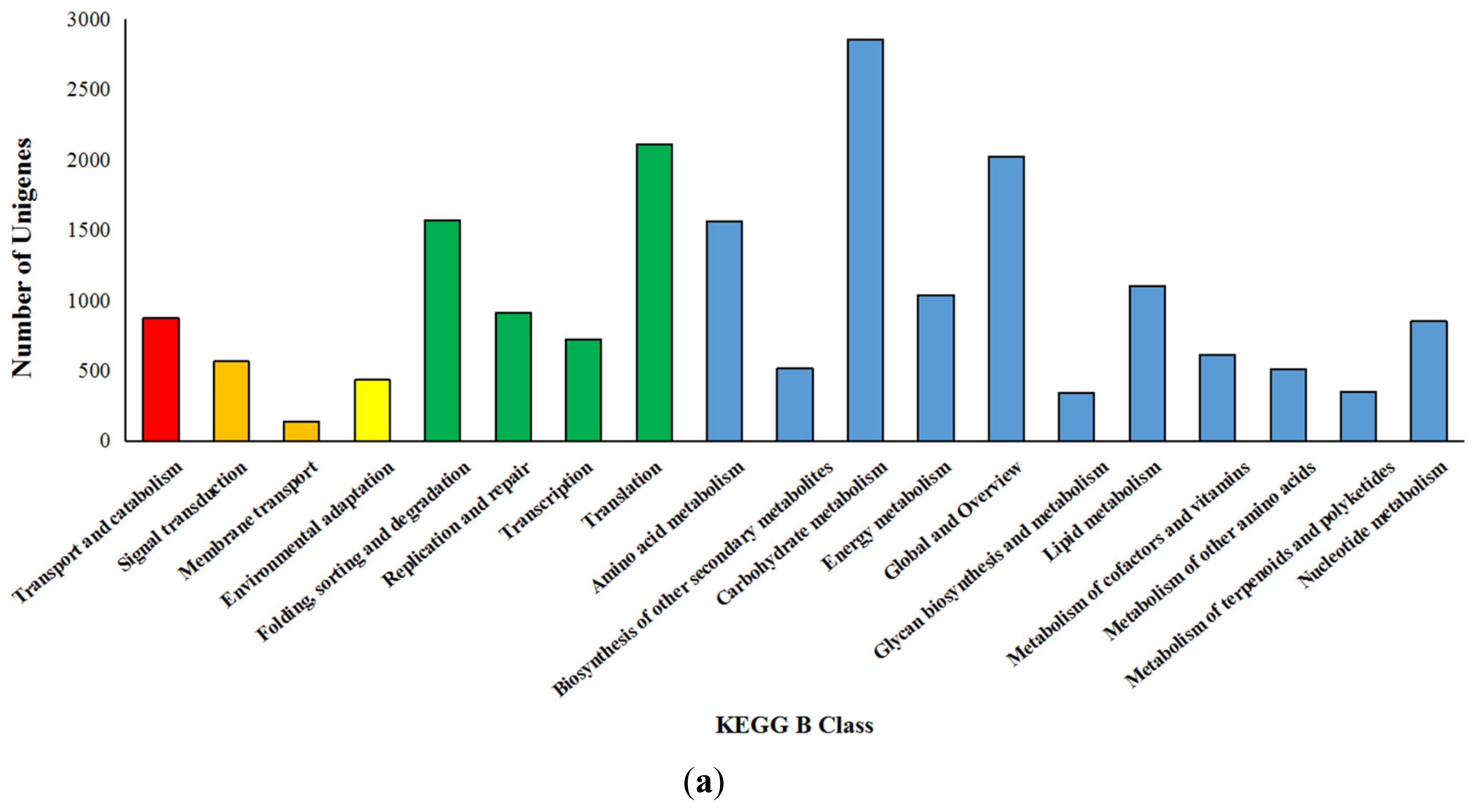
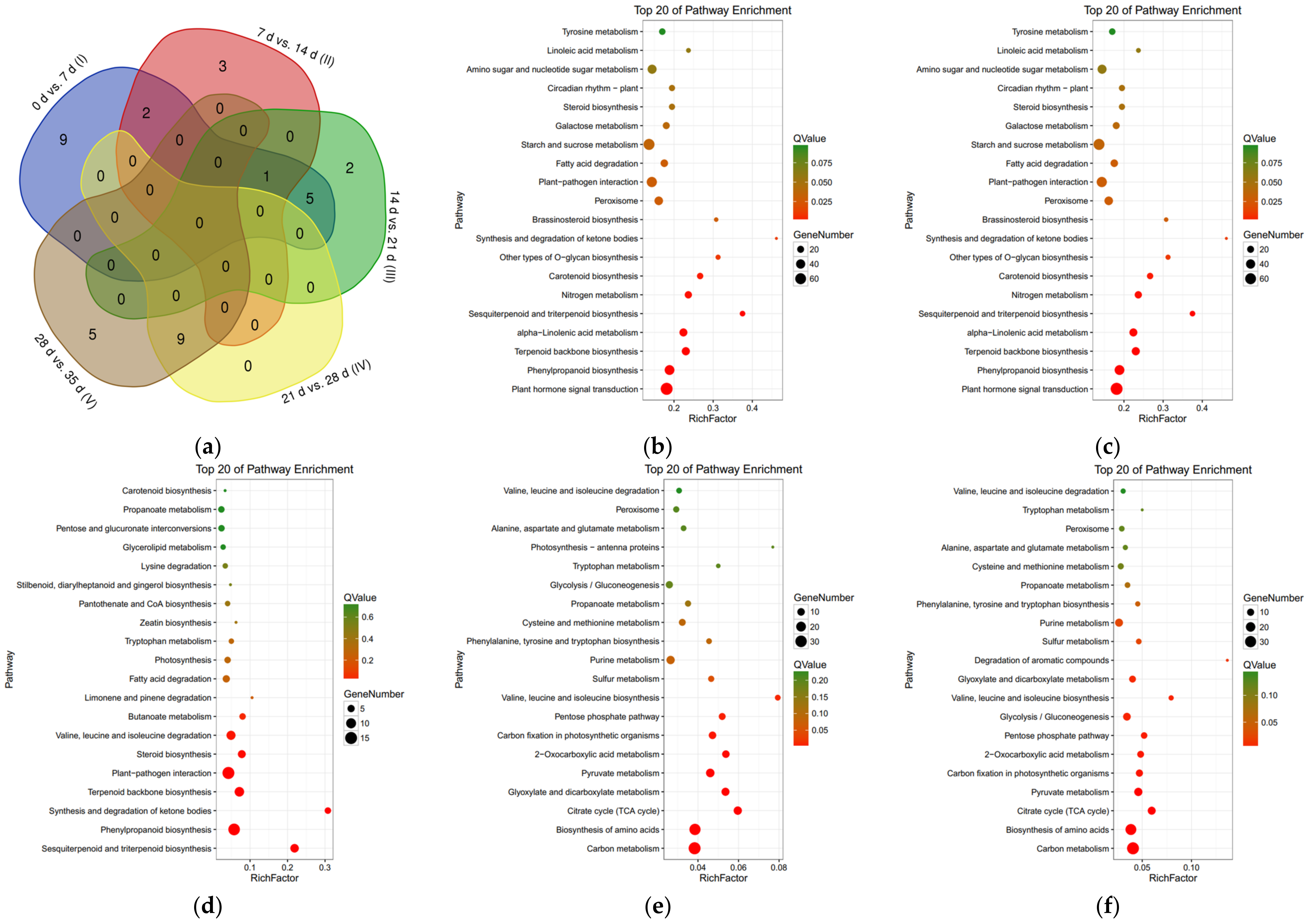
3.4. Kyoto Encyclopedia of Genes and Genomes Enrichment Analysis of Differentially Expressed Genes
3.5. Cold Regulation of Reversible Protein Phosphorylation
3.6. Cold Regulation of Ca2+ Signal Components
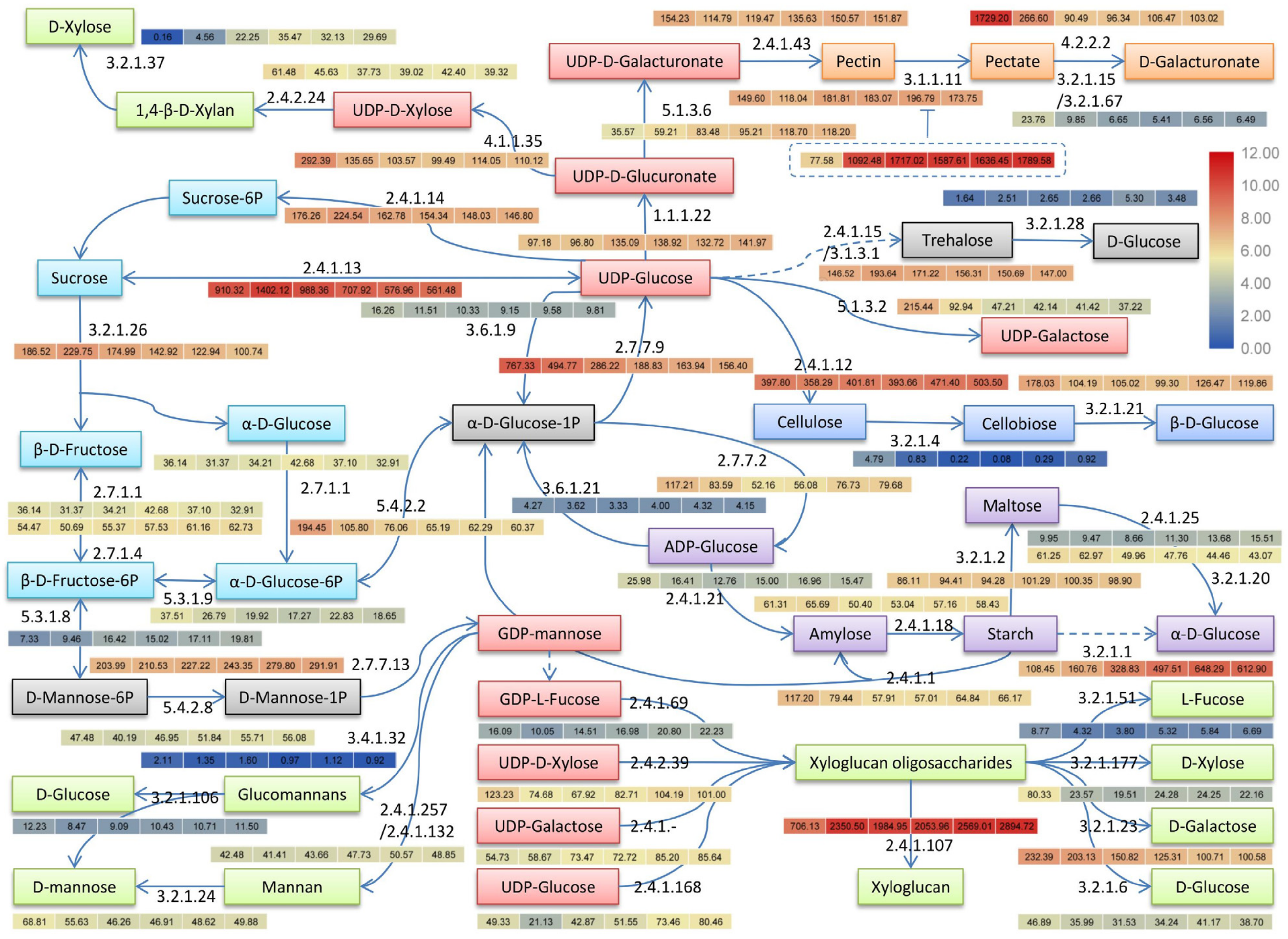
3.7. Cold Regulation of Major Sugars and Polysaccharides Metabolisms
3.7.1. Major Sugars and Starch Metabolisms
3.7.2. Cell Wall Polysaccharides Metabolisms
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Liu, Y.; Zou, D.; Wu, B.; Lin, D.; Zhang, Z.; Wu, J. Cloning and expression analysis of a CCoAOMT homolog in loquat fruit in response to low-temperature storage. Postharvest Boil. Technol. 2015, 105, 45–50. [Google Scholar] [CrossRef]
- Wu, T.; Lin, S.; Wu, B.; Chen, Y.; Lin, S.; Lin, D.; Chen, L.; Zhang, Z.; Wu, J. Cloning and in silico and expression analyses of the heterotrimeric G protein α subunit gene (EjLGA1) from loquat (Eriobotrya japonica) fruits under cold storage with different pretreatments. J. Hortic. Sci. Biotechnol. 2018, 93, 1–10. [Google Scholar] [CrossRef]
- Cai, C.; Chen, K.S.; Xu, W.P.; Zhang, W.S.; Li, X.; Ferguson, I. Effect of 1-MCP on postharvest quality of loquat fruit. Postharvest Boil. Technol. 2006, 40, 155–162. [Google Scholar] [CrossRef]
- Cao, S.F.; Wang, X.Q.; Yang, Z.F.; Ma, S.J.; Li, N.; Tang, S.S.; Wang, X.X.; Zheng, Y.H. Effects of methyl jasmonate treatment on quality and decay in cold-stored loquat fruit. Acta Hortic. 2007, 750, 425–430. [Google Scholar] [CrossRef]
- Cao, S.; Zheng, Y.; Wang, K.; Rui, H.; Tang, S. Effect of methyl jasmonate on cell wall modification of loquat fruit in relation to chilling injury after harvest. Food Chem. 2010, 118, 641–647. [Google Scholar] [CrossRef]
- Li, X.; Zang, C.; Ge, H.; Zhang, J.; Grierson, D.; Yin, X.R.; Chen, K.S. Involvement of PAL, C4H, and 4CL in chilling injury-induced flesh lignification of loquat fruit. Hortsci. A Publ. Am. Soc. Hortic. Sci. 2017, 52, 127–131. [Google Scholar] [CrossRef]
- Cai, C.; Li, X.; Chen, K.S. Acetylsalicylic acid alleviates chilling injury of postharvest loquat (Eriobotrya japonica lindl.) fruit. Eur. Food Res. Technol. 2006, 223, 533–539. [Google Scholar] [CrossRef]
- Cai, C.; Xu, C.J.; Shan, L.L.; Li, X.; Zhou, C.H.; Zhang, W.S.; Ferguson, I.; Chen, K.S. Low temperature conditioning reduces postharvest chilling injury in loquat fruit. Postharvest Boil. Technol. 2006, 41, 252–259. [Google Scholar] [CrossRef]
- Cao, S.; Zheng, Y.; Yang, Z.; Wang, K.; Rui, H. Effect of methyl jasmonate on quality and antioxidant activity of postharvest loquat fruit. J. Sci. Food Agric. 2009, 89, 2064–2070. [Google Scholar] [CrossRef]
- Cao, S.; Yang, Z.; Zheng, Y. Sugar metabolism in relation to chilling tolerance of loquat fruit. Food Chem. 2013, 136, 139–143. [Google Scholar] [CrossRef]
- Shan, L.L.; Li, X.; Wang, P.; Cai, C.; Zhang, B.; Sun, C.D.; Zhang, W.S.; Xu, C.J.; Ferguson, I.; Chen, K.S. Characterization of cDNAs associated with lignification and their expression profiles in loquat fruit with different lignin accumulation. Planta 2008, 227, 1243–1254. [Google Scholar] [CrossRef] [PubMed]
- Xu, Q.; Yin, X.R.; Zeng, J.K.; Ge, H.; Song, M.; Xu, C.J.; Li, X.; Ferguson, I.B.; Chen, K.S. Activator- and repressor-type MYB transcription factors are involved in chilling injury induced flesh lignification in loquat via their interactions with the phenylpropanoid pathway. J. Exp. Bot. 2014, 65, 4349–4359. [Google Scholar] [CrossRef]
- Xu, Q.; Wang, W.; Zeng, J.; Zhang, J.; Grierson, D.; Li, X.; Yin, X.; Chen, K. A NAC transcription factor, EjNAC1, affects lignification of loquat fruit by regulating lignin. Postharvest Boil. Technol. 2015, 102, 25–31. [Google Scholar] [CrossRef]
- Wang, W.Q.; Jing, Z.; Hang, G.; Li, S.J.; Xian, L.; Yin, X.R.; Donald, G.; Chen, K.S. EjMYB8 transcriptionally regulates flesh lignification in loquat fruit. PLoS ONE 2016, 11, e0154399. [Google Scholar] [CrossRef] [PubMed]
- Zeng, J.K.; Li, X.; Xu, Q.; Chen, J.Y.; Yin, X.R.; Ferguson, I.B.; Chen, K.S. EjAP2-1, an AP2/ERF gene, is a novel regulator of fruit lignification induced by chilling injury, via interaction with EjMYB transcription factors. Plant Biotechnol. J. 2015, 13, 1325–1334. [Google Scholar] [CrossRef] [PubMed]
- Ge, H.; Zhang, J.; Zhang, Y.J.; Li, X.; Yin, X.R.; Grierson, D.; Chen, K.S. EjNAC3 transcriptionally regulates chilling-induced lignification of loquat fruit via physical interaction with an atypical CAD-like gene. J. Exp. Bot. 2017, 68, 5129–5136. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Ge, H.; Zang, C.; Li, X.; Donald, G.; Chen, K.S.; Yin, X.R. EjODO1, a MYB transcription factor, regulating lignin biosynthesis in developing loquat (Eriobotrya japonica) fruit. Front. Plant Sci. 2016, 7, 1360. [Google Scholar] [CrossRef]
- Zeng, J.K.; Li, X.; Zhang, J.; Ge, H.; Yin, X.R.; Chen, K.S. Regulation of loquat fruit low temperature response and lignification involves interaction of heat shock factors and genes associated with lignin biosynthesis. Plant Cell Environ. 2016, 39, 1780–1789. [Google Scholar] [CrossRef]
- Ping, W.; Bo, Z.; Xian, L.; Xu, C.; Yin, X.; Shan, L.; Ferguson, I.; Chen, K. Ethylene signal transduction elements involved in chilling injury in non-climacteric loquat fruit. J. Exp. Bot. 2010, 61, 179. [Google Scholar]
- Jin, W.; Xu, C.; Li, X.; Zhang, B.; Wang, P.; Allan, A.C.; Chen, K. Expression of ROP/RAC GTPase genes in postharvest loquat fruit in association with senescence and cold regulated lignification. Postharvest Boil. Technol. 2009, 54, 9–14. [Google Scholar] [CrossRef]
- Grabherr, M.G.; Haas, B.J.; Yassour, M.; Levin, J.Z.; Thompson, D.A.; Amit, I.; Xian, A.; Fan, L.; Raychowdhury, R.; Zeng, Q. Trinity: Reconstructing a full-length transcriptome without a genome from RNA-seq data. Nat. Biotechnol. 2011, 29, 644. [Google Scholar] [CrossRef] [PubMed]
- Mellidou, I.; Buts, K.; Hatoum, D.; Ho, Q.T.; Johnston, J.W.; Watkins, C.B.; Schaffer, R.J.; Gapper, N.E.; Giovannoni, J.J.; Rudell, D.R. Transcriptomic events associated with internal browning of apple during postharvest storage. BMC Plant Boil. 2014, 14, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Dagar, A.; Puig, C.P.; Ibanez, C.M.; Ziliotto, F.; Bonghi, C.; Crisosto, C.H.; Friedman, H.; Lurie, S.; Granell, A. Comparative transcript profiling of a peach and its nectarine mutant at harvest reveals differences in gene expression related to storability. Tree Genet. Genomes 2013, 9, 223–235. [Google Scholar] [CrossRef]
- Sanhueza, D.; Vizoso, P.; Balic, I.; Camposvargas, R.; Meneses, C. Transcriptomic analysis of fruit stored under cold conditions using controlled atmosphere in prunus persica cv. “Red pearl”. Front. Plant Sci. 2015, 6, 788. [Google Scholar] [CrossRef] [PubMed]
- Lafuente, M.T.; Establésortíz, B.; Gonzálezcandelas, L. Insights into the molecular events that regulate heat-induced chilling tolerance in citrus fruits. Front. Plant Sci. 2017, 8. [Google Scholar] [CrossRef] [PubMed]
- Sivankalyani, V.; Sela, N.; Feygenberg, O.; Zemach, H.; Maurer, D.; Alkan, N. Transcriptome dynamics in mango fruit peel reveals mechanisms of chilling stress. Front. Plant Sci. 2016, 7, 1579. [Google Scholar] [CrossRef] [PubMed]
- Altschul, S.F.; Gish, W.; Miller, W.; Myers, E.W.; Lipman, D.J. Basic local alignment search tool. J. Mol. Biol. 1990, 215, 403–410. [Google Scholar] [CrossRef]
- Bairoch, A.; Apweiler, R. The SWISS-PROT protein sequence database and its supplement TrEMBL in 2000. Nucleic Acids Res. 2000, 28, 45. [Google Scholar] [CrossRef]
- Kanehisa, M.; Goto, S. KEGG: Kyoto encyclopedia of genes and genomes. Nucleic Acids Res. 2000, 28, 27–30. [Google Scholar] [CrossRef]
- Koonin, E.V.; Fedorova, N.D.; Jackson, J.D.; Jacobs, A.R.; Krylov, D.M.; Makarova, K.S.; Mazumder, R.; Mekhedov, S.L.; Nikolskaya, A.N.; Rao, B.S. A comprehensive evolutionary classification of proteins encoded in complete eukaryotic genomes. Genome Biol. 2004, 5, R7. [Google Scholar] [CrossRef]
- Valero-Mora, P.M. Ggplot2: Elegant graphics for data analysis. J. Stat. Softw. 2010, 35. [Google Scholar] [CrossRef]
- Conesa, A.; Götz, S.; Garcíagómez, J.M.; Terol, J.; Talón, M.; Robles, M. Blast2GO: A universal tool for annotation, visualization and analysis in functional genomics research. Bioinformatics 2005, 21, 3674–3676. [Google Scholar] [CrossRef] [PubMed]
- Ye, J.; Fang, L.; Zheng, H.; Zhang, Y.; Chen, J.; Zhang, Z.; Wang, J.; Li, S.; Li, R.; Bolund, L. WEGO: A web tool for plotting GO annotations. Nucleic Acids Res. 2006, 34, W293–W297. [Google Scholar] [CrossRef] [PubMed]
- Langmead, B.; Salzberg, S.L. Fast gapped-read alignment with Bowtie 2. Nat. Methods 2012, 9, 357–359. [Google Scholar] [CrossRef] [PubMed]
- Li, B.; Dewey, C.N. RSEM: Accurate transcript quantification from RNA-seq data with or without a reference genome. BMC Bioinform. 2011, 12, 323. [Google Scholar] [CrossRef] [PubMed]
- Robinson, M.; Mccarthy, D.; Smyth, G.K. EdgeR: Differential expression analysis of digital gene expression data. J. Hosp. Palliat. Nurs. 2010, 4, 206–207. [Google Scholar]
- Mao, X.; Cai, T.; Olyarchuk, J.G.; Wei, L. Automated genome annotation and pathway identification using the KEGG orthology (KO) as a controlled vocabulary. Bioinformatics 2005, 21, 3787–3793. [Google Scholar] [CrossRef]
- Deng, W.; Wang, Y.; Liu, Z.; Cheng, H.; Xue, Y. Hemi: A toolkit for illustrating heatmaps. PloS ONE 2014, 9, e111988. [Google Scholar] [CrossRef]
- Chen, C.; Xia, R.; Chen, H.; He, Y. TBtools, a toolkit for biologists integrating various HTS-data handling tools with a user-friendly interface. BioRxiv 2018. [Google Scholar]
- Kim, J. Perception, transduction, and networks in cold signaling. J. Plant Boil. 2007, 50, 139–147. [Google Scholar] [CrossRef]
- Yamaguchi-Shinozaki, K.; Shinozaki, K. Transcriptional regulatory networks in cellular responses and tolerance to dehydration and cold stresses. Annu. Rev. Plant Boil. 2006, 57, 781–803. [Google Scholar] [CrossRef]
- Huang, G.T.; Ma, S.L.; Bai, L.P.; Zhang, L.; Ma, H.; Jia, P.; Liu, J.; Zhong, M.; Guo, Z.F. Signal transduction during cold, salt, and drought stresses in plants. Mol. Boil. Rep. 2012, 39, 969–987. [Google Scholar] [CrossRef] [PubMed]
- Nardozzi, J.D.; Lott, K.; Cingolani, G. Phosphorylation meets nuclear import: A review. Cell Commun. Signal. 2010, 8, 32. [Google Scholar] [CrossRef] [PubMed]
- Solanke, A.U.; Sharma, A.K. Signal transduction during cold stress in plants. Physiol. Mol. Boil. Plants 2008, 14, 69–79. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Li, J.; Li, F.; Liu, H.; Yang, W.; Chong, K.; Xu, Y. OsMAPK3 phosphorylates OsBHLH002/OsICE1 and inhibits its ubiquitination to activate OsTPP1 and enhances rice chilling tolerance. Dev. Cell 2017, 43, 731. [Google Scholar] [CrossRef] [PubMed]
- Zhao, C.; Wang, P.; Si, T.; Hsu, C.C.; Wang, L.; Zayed, O.; Yu, Z.; Zhu, Y.; Dong, J.; Tao, W.A. MAP kinase cascades regulate the cold response by modulating ICE1 protein stability. Dev. Cell 2017, 43, 618. [Google Scholar] [CrossRef] [PubMed]
- Zhao, C.; Zhang, Z.; Xie, S.; Si, T.; Li, Y.; Zhu, J.K. Mutational evidence for the critical role of CBF transcription factors in cold acclimation in Arabidopsis. Plant Physiol. 2016, 171, 2744. [Google Scholar]
- Tang, N.; Deng, W.; Hu, N.; Chen, N.; Li, Z. Metabolite and transcriptomic analysis reveals metabolic and regulatory features associated with powell orange pulp deterioration during room temperature and cold storage. Postharvest Boil. Technol. 2015, 112, 75–86. [Google Scholar] [CrossRef]
- Vandesteene, L.; Dijck, P.V. Expansive evolution of the trehalose-6-phosphate phosphatase gene family in Arabidopsis. Plant Physiol. 2012, 160, 884. [Google Scholar] [CrossRef]
- Williams, G.J.; Thorson, J.S. Natural product glycosyltransferases: Properties and applications. Adv. Enzym. Relat. Areas Mol. Biol. 2009, 76, 55–119. [Google Scholar]
- Jin, P.; Duan, Y.; Wang, L.; Wang, J.; Zheng, Y. Reducing chilling injury of loquat fruit by combined treatment with hot air and methyl jasmonate. Food Bioprocess Technol. 2014, 7, 2259–2266. [Google Scholar] [CrossRef]
- Somerville, C. Cellulose synthesis in higher plants. Annu. Rev. Cell Dev. Biol. 2006, 22, 53–78. [Google Scholar] [CrossRef] [PubMed]
- Marcus, S.E.; Verhertbruggen, Y.; Farkas, V.; Pedersen, H.L.; Willats, W.G.; Knox, J.P. Pectic homogalacturonan masks abundant sets of xyloglucan epitopes in plant cell walls. BMC Plant Boil. 2008, 8, 60. [Google Scholar] [CrossRef]
- Braidwood, L.; Breuer, C.; Sugimoto, K. My body is a cage: Mechanisms and modulation of plant cell growth. New Phytol. 2013, 201, 388–402. [Google Scholar] [CrossRef] [PubMed]
- Rockel, N.; Wolf, S.; Kost, B.; Rausch, T.; Greiner, S. Elaborate spatial patterning of cell-wall pme and pmei at the pollen tube tip involves pmei endocytosis, and reflects the distribution of esterified and de-esterified pectins. Plant J. 2008, 53, 133–143. [Google Scholar] [CrossRef] [PubMed]
- Scheller, H.V.; Ulvskov, P. Hemicelluloses. Annu. Rev. Plant Biol. 2010, 61, 263–289. [Google Scholar] [CrossRef] [PubMed]
- Brown, D.M.; Goubet, F.; Wong, V.W.; Goodacre, R.; Stephens, E.; Dupree, P.; Turner, S.R. Comparison of five xylan synthesis mutants reveals new insight into the mechanisms of xylan synthesis. Plant J. 2010, 52, 1154–1168. [Google Scholar] [CrossRef]
- Bem, L.E.V.D.; Vincentz, M.G. Evolution of xyloglucan-related genes in green plants. BMC Evol. Biol. 2010, 10, 1–17. [Google Scholar]
- Hayashi, T. Xyloglucans in the primary cell wall. Annu. Rev. Plant Physiol. Plant Mol. Boil. 1989, 40, 139–168. [Google Scholar] [CrossRef]
- Sulová, Z.; Baran, R.; Farkaš, V. Release of complexed xyloglucan endotransglycosylase (XET) from plant cell walls by a transglycosylation reaction with xyloglucan-derived oligosaccharides. Plant Physiol. Biochem. 2001, 39, 927–932. [Google Scholar] [CrossRef]
- Marcus, S.E.; Blake, A.W.; Benians, T.A.S.; Lee, K.J.D.; Poyser, C.; Donaldson, L.; Leroux, O.; Rogowski, A.; Petersen, H.L.; Boraston, A. Restricted access of proteins to mannan polysaccharides in intact plant cell walls. Plant J. Cell Mol. Boil. 2010, 64, 191. [Google Scholar] [CrossRef] [PubMed]
- Maeda, Y.; Awano, T.; Takabe, K.; Fujita, M. Immunolocalization of glucomannans in the cell wall of differentiating tracheids in Chamaecyparis obtusa. Protoplasma 2000, 213, 148–156. [Google Scholar] [CrossRef]

| GO Term | 0 d vs. 7 d (I) | 7 d vs. 14 d (II) | 14 d vs. 21 d (III) | 21 d vs. 28 d (IV) | 28 d vs. 35 d (V) | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| Num. | Hits | Num. | Hits | Num. | Hits | Num. | Hits | Num. | Hits | |
| Cellular Component | 5 | 973 | 5 | 332 | 6 | 125 | 7 | 253 | 11 | 420 |
| Molecular Function | 18 | 1114 | 2 | 12 | 6 | 73 | 1 | 24 | 2 | 23 |
| Biological Process | 1 | 9 | 0 | 0 | 0 | 0 | 0 | 0 | 75 | 6 |
| Total | 24 | 2096 | 7 | 344 | 12 | 198 | 8 | 277 | 88 | 449 |
| Category | 0 d vs. 7 d (I) | 7 d vs. 14 d (II) | 14 d vs. 21 d (III) | 21 d vs. 28 d (IV) | 28 d vs. 35 d (V) | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| Up | Down | Up | Down | Up | Down | Up | Down | Up | Down | |
| Protein Kinase | 282 | 130 | 103 | 36 | 31 | 21 | 10 | 0 | 1 | 10 |
| Protein Phosphatase | 35 | 50 | 9 | 16 | 5 | 2 | 3 | 0 | 0 | 2 |
| EC 1 ID | Description | KO 2 ID | Gene Number |
|---|---|---|---|
| EC:1.1.1.22 | UDP-glucose 6-dehydrogenase | K00012 | 6 |
| EC:2.4.1.? | Xyloglucan galactosyltransferase | Nr 3 | 19 |
| EC:2.4.1.1 | Starch phosphorylase | K00688 | 12 |
| EC:2.4.1.12 | Cellulose synthase | Nr | 42 |
| EC:2.4.1.13 | Sucrose synthase | K00695 | 21 |
| EC:2.4.1.14 | Sucrose-phosphate synthase | K00696 | 10 |
| EC:2.4.1.15/3.1.3.12 | Trehalose 6-phosphate synthase/phosphatase | K16055/K01087 | 29 |
| EC:2.4.1.18 | 1,4-α-glucan branching enzyme | K00700 | 15 |
| EC:2.4.1.21 | Starch synthase | K00703 | 7 |
| EC:2.4.2.24 | 1,4-β-d-xylan synthase | Nr | 6 |
| EC:2.4.1.25 | 4-α-glucanotransferase | K00705 | 5 |
| EC:2.4.1.257/2.4.1.132 | α-1,3/1,6-mannosyltransferase | Nr | 6 |
| EC:2.4.1.43 | α-1,4-galacturonosyltransferase | K13648 | 18 |
| EC:2.4.1.69 | galactoside 2-α-l-fucosyltransferase | Nr | 3 |
| EC:2.4.1.168 | Xyloglucan glycosyltransferase | Nr | 7 |
| EC:2.4.1.207 | Xyloglucan endotransglucosylase/hydrolase | Nr | 20 |
| EC:2.4.2.39 | Xyloglucan 6-xylosyltransferase | Nr | 3 |
| EC:2.7.1.1 | Hexokinase | K00844 | 10 |
| EC:2.7.1.4 | Fructokinase | K00847 | 11 |
| EC:2.7.7.13 | Mannose-1-phosphate guanylyltransferase | K00966 | 13 |
| EC:2.7.7.27 | Glucose-1-phosphate adenylyltransferase | K00975 | 18 |
| EC:2.7.7.9 | UTP-glucose-1-phosphate uridylyltransferase | K00963 | 6 |
| EC:3.1.1.11 | Pectin methylesterase | K01051 | 45 |
| EC:3.2.1.1 | α-amylase | K01176 | 16 |
| EC:3.2.1.15/3.2.1.67 | Endo-/Exo-polygalacturonase | K01184/K01213 | 15 |
| EC:3.2.1.2 | β-amylase | K01177 | 49 |
| EC:3.2.1.4 | Endoglucanase/cellulase | K01179/K19356 | 6 |
| EC:3.2.1.6 | Endo-1,3;1,4-β-d-glucanase | Nr | 14 |
| EC:3.2.1.20 | α-glucosidase | K01187 | 6 |
| EC:3.2.1.21 | β-glucosidase | K01188/K05349 | 68 |
| EC:3.2.1.23 | β-galactosidase | K12309 | 8 |
| EC:3.2.1.24 | α-mannosidase | K01191 | 5 |
| EC:3.2.1.26 | Invertase | K01193 | 7 |
| EC:3.2.1.28 | α,α-trehalase | K01194 | 5 |
| EC:3.2.1.37 | β-d-xylosidase | K15920 | 3 |
| EC:3.2.1.51 | α-l-fucosidase | K01206 | 8 |
| EC:3.2.1.106 | Mannosyl-oligosaccharide glucosidase | Nr | 9 |
| EC:3.2.1.177 | α-xylosidase | Nr | 3 |
| EC:3.4.1.32 | Glucomannan 4-β-mannosyltransferase | Nr | 6 |
| EC:3.6.1.9 | Ectonucleotide pyrophosphatase | K01513 | 2 |
| EC:3.6.1.21 | ADP-sugar diphosphatase | K18447 | 1 |
| EC:4.1.1.35 | UDP-glucuronate decarboxylase | K08678 | 14 |
| EC:4.2.2.2 | Pectate lyase | Nr | 26 |
| EC:5.1.3.2 | UDP-glucose 4-epimerase | K01784 | 7 |
| EC:5.1.3.6 | UDP-glucuronate 4-epimerase | K08679 | 8 |
| EC:5.3.1.8 | Mannose-6-phosphate isomerase | K01809 | 7 |
| EC:5.3.1.9 | Glucose-6-phosphate isomerase | K01810 | 12 |
| EC:5.4.2.2 | Phosphoglucomutase | K01835 | 8 |
| EC:5.4.2.8 | Phosphomannomutase | K17497 | 4 |
| Total | 49 | 649 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lin, S.; Wu, T.; Lin, H.; Zhang, Y.; Xu, S.; Wang, J.; Wu, B.; Chen, Y.; Lin, S.; Lin, D.; et al. De Novo Analysis Reveals Transcriptomic Responses in Eriobotrya japonica Fruits during Postharvest Cold Storage. Genes 2018, 9, 639. https://doi.org/10.3390/genes9120639
Lin S, Wu T, Lin H, Zhang Y, Xu S, Wang J, Wu B, Chen Y, Lin S, Lin D, et al. De Novo Analysis Reveals Transcriptomic Responses in Eriobotrya japonica Fruits during Postharvest Cold Storage. Genes. 2018; 9(12):639. https://doi.org/10.3390/genes9120639
Chicago/Turabian StyleLin, Shoukai, Ti Wu, Hailan Lin, Yanqing Zhang, Shichang Xu, Jinge Wang, Bisha Wu, Yu Chen, Suying Lin, Dahe Lin, and et al. 2018. "De Novo Analysis Reveals Transcriptomic Responses in Eriobotrya japonica Fruits during Postharvest Cold Storage" Genes 9, no. 12: 639. https://doi.org/10.3390/genes9120639
APA StyleLin, S., Wu, T., Lin, H., Zhang, Y., Xu, S., Wang, J., Wu, B., Chen, Y., Lin, S., Lin, D., Wang, X., Zhao, X., & Wu, J. (2018). De Novo Analysis Reveals Transcriptomic Responses in Eriobotrya japonica Fruits during Postharvest Cold Storage. Genes, 9(12), 639. https://doi.org/10.3390/genes9120639





