A Two-Cohort RNA-seq Study Reveals Changes in Endometrial and Blood miRNome in Fertile and Infertile Women
Abstract
1. Introduction
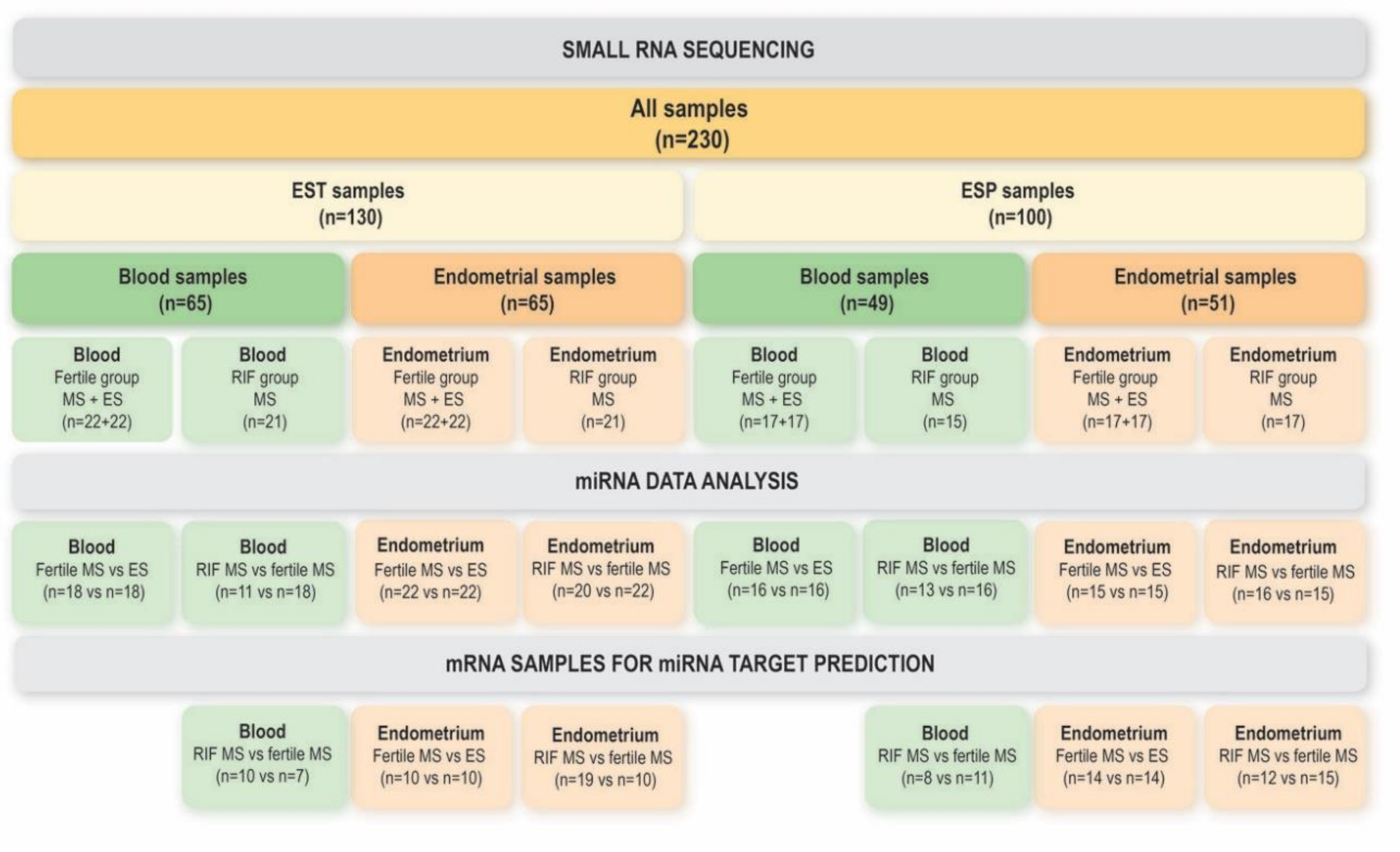
2. Materials and Methods
2.1. Study Participants
2.2. Sample Collection
2.3. RNA Extraction from Endometrial Tissue
2.4. RNA Extraction from Blood Samples
2.5. Small RNA Sequencing
2.6. SMALL RNA Sequencing Data Analysis
2.7. Novel miRNA Identification
2.8. mRNA Data Analysis for miRNA Target Prediction
2.9. Integrated Analysis of miRNA and mRNA Data
2.10. Novel miRNA Validation Using Quantitative Real-Time Polymerase Chain Reaction
2.11. Data Availability
3. Results
3.1. miRNAs in the Endometrium
3.2. miRNAs in Blood
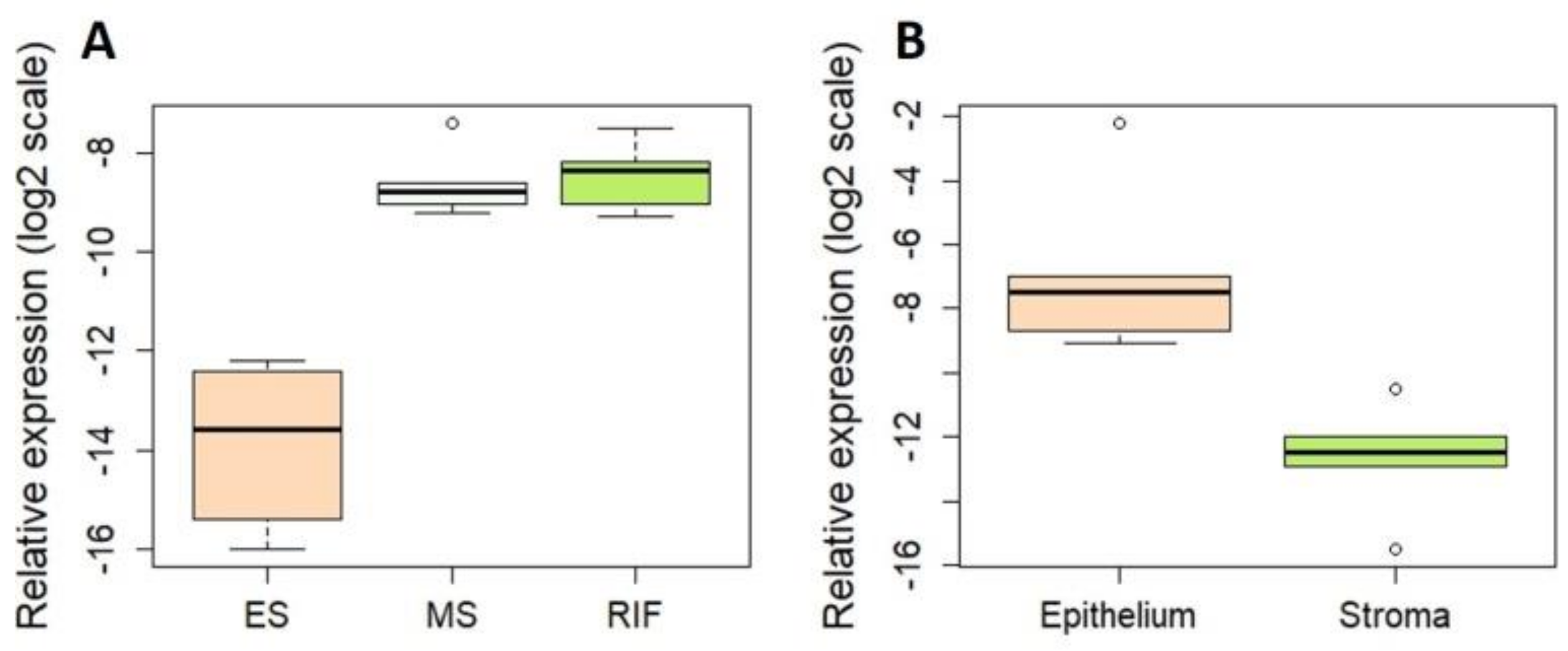
3.3. Novel miRNAs
3.4. miRNA–mRNA Interaction Prediction
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Simon, A.; Laufer, N. Repeated implantation failure: Clinical approach. Fertil. Steril. 2012, 97, 1039–1043. [Google Scholar] [CrossRef] [PubMed]
- Macklon, N.S.; Stouffer, R.L.; Giudice, L.C.; Fauser, B.C. The science behind 25 years of ovarian stimulation for in vitro fertilization. Endocr. Rev. 2006, 27, 170–207. [Google Scholar] [CrossRef] [PubMed]
- Altmäe, S.; Esteban, F.J.; Stavreus-Evers, A.; Simon, C.; Giudice, L.; Lessey, B.A.; Horcajadas, J.A.; Macklon, N.S.; D’Hooghe, T.; Campoy, C.; et al. Guidelines for the design, analysis and interpretation of ‘omics’ data: Focus on human endometrium. Hum. Reprod. Update 2014, 20, 12–28. [Google Scholar] [CrossRef] [PubMed]
- Altmäe, S.; Koel, M.; Vosa, U.; Adler, P.; Suhorutsenko, M.; Laisk-Podar, T.; Kukushkina, V.; Saare, M.; Velthut-Meikas, A.; Krjutskov, K.; et al. Meta-signature of human endometrial receptivity: A meta-analysis and validation study of transcriptomic biomarkers. Sci. Rep. 2017, 7, 10077. [Google Scholar] [CrossRef] [PubMed]
- Koler, M.; Achache, H.; Tsafrir, A.; Smith, Y.; Revel, A.; Reich, R. Disrupted gene pattern in patients with repeated in vitro fertilization (IVF) failure. Hum. Reprod. 2009, 24, 2541–2548. [Google Scholar] [CrossRef] [PubMed]
- Altmäe, S.; Martinez-Conejero, J.A.; Salumets, A.; Simon, C.; Horcajadas, J.A.; Stavreus-Evers, A. Endometrial gene expression analysis at the time of embryo implantation in women with unexplained infertility. Mol. Hum. Reprod. 2010, 16, 178–187. [Google Scholar] [CrossRef] [PubMed]
- Altmäe, S.; Tamm-Rosenstein, K.; Esteban, F.J.; Simm, J.; Kolberg, L.; Peterson, H.; Metsis, M.; Haldre, K.; Horcajadas, J.A.; Salumets, A.; et al. Endometrial transcriptome analysis indicates superiority of natural over artificial cycles in recurrent implantation failure patients undergoing frozen embryo transfer. Reprod. Biomed. Online 2016, 32, 597–613. [Google Scholar] [CrossRef] [PubMed]
- Ledee, N.; Munaut, C.; Aubert, J.; Serazin, V.; Rahmati, M.; Chaouat, G.; Sandra, O.; Foidart, J.M. Specific and extensive endometrial deregulation is present before conception in IVF/ICSI repeated implantation failures (IF) or recurrent miscarriages. J. Pathol. 2011, 225, 554–564. [Google Scholar] [CrossRef] [PubMed]
- Koot, Y.E.; van Hooff, S.R.; Boomsma, C.M.; van Leenen, D.; Koerkamp, M.J.G.; Goddijn, M.; Eijkemans, M.J.; Fauser, B.C.; Holstege, F.C.; Macklon, N.S. An endometrial gene expression signature accurately predicts recurrent implantation failure after IVF. Sci. Rep. 2016, 6, 19411. [Google Scholar] [CrossRef] [PubMed]
- Fan, L.J.; Han, H.J.; Guan, J.; Zhang, X.W.; Cui, Q.H.; Shen, H.; Shi, C. Aberrantly expressed long noncoding RNAs in recurrent implantation failure: A microarray related study. Syst. Biol. Reprod. Med. 2017, 63, 269–278. [Google Scholar] [CrossRef] [PubMed]
- Carthew, R.W.; Sontheimer, E.J. Origins and mechanisms of miRNas and siRNAs. Cell 2009, 136, 642–655. [Google Scholar] [CrossRef] [PubMed]
- Lewis, B.P.; Burge, C.B.; Bartel, D.P. Conserved seed pairing, often flanked by adenosines, indicates that thousands of human genes are microRNA targets. Cell 2005, 120, 15–20. [Google Scholar] [CrossRef] [PubMed]
- Lam, E.W.; Shah, K.; Brosens, J.J. The diversity of sex steroid action: The role of micro-RNAs and FOXO transcription factors in cycling endometrium and cancer. J. Endocrinol. 2012, 212, 13–25. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Niu, Z.; Li, Q.; Pang, R.T.; Chiu, P.C.; Yeung, W.S. MicroRNA and embryo implantation. Am. J. Reprod. Immunol. 2016, 75, 263–271. [Google Scholar] [CrossRef] [PubMed]
- Altmäe, S.; Martinez-Conejero, J.A.; Esteban, F.J.; Ruiz-Alonso, M.; Stavreus-Evers, A.; Horcajadas, J.A.; Salumets, A. MicroRNAs miR-30b, miR-30d, and miR-494 regulate human endometrial receptivity. Reprod. Sci. 2013, 20, 308–317. [Google Scholar] [CrossRef] [PubMed]
- Vilella, F.; Moreno-Moya, J.M.; Balaguer, N.; Grasso, A.; Herrero, M.; Martinez, S.; Marcilla, A.; Simon, C. Hsa-miR-30d, secreted by the human endometrium, is taken up by the pre-implantation embryo and might modify its transcriptome. Development 2015, 142, 3210–3221. [Google Scholar] [CrossRef] [PubMed]
- Moreno-Moya, J.M.; Vilella, F.; Martinez, S.; Pellicer, A.; Simon, C. The transcriptomic and proteomic effects of ectopic overexpression of miR-30d in human endometrial epithelial cells. Mol. Hum. Reprod. 2014, 20, 550–566. [Google Scholar] [CrossRef] [PubMed]
- Choi, Y.; Kim, H.R.; Lim, E.J.; Park, M.; Yoon, J.A.; Kim, Y.S.; Kim, E.K.; Shin, J.E.; Kim, J.H.; Kwon, H.; et al. Integrative analyses of uterine transcriptome and microRNAome reveal compromised LIF-STAT3 signaling and progesterone response in the endometrium of patients with recurrent/repeated implantation failure (RIF). PLoS ONE 2016, 11, e0157696. [Google Scholar] [CrossRef] [PubMed]
- Li, R.; Qiao, J.; Wang, L.; Li, L.; Zhen, X.; Liu, P.; Zheng, X. microRNA Array and microarray evaluation of endometrial receptivity in patients with high serum progesterone levels on the day of hCG administration. Reprod. Biol. Endocrinol. 2011, 9, 29. [Google Scholar] [CrossRef] [PubMed]
- Revel, A.; Achache, H.; Stevens, J.; Smith, Y.; Reich, R. microRNAs are associated with human embryo implantation defects. Hum. Reprod. 2011, 26, 2830–2840. [Google Scholar] [CrossRef] [PubMed]
- Shi, C.; Shen, H.; Fan, L.J.; Guan, J.; Zheng, X.B.; Chen, X.; Liang, R.; Zhang, X.W.; Cui, Q.H.; Sun, K.K.; et al. Endometrial microRNA signature during the window of implantation changed in patients with repeated implantation failure. Chin. Med. J. (Engl.) 2017, 130, 566–573. [Google Scholar] [PubMed]
- Sha, A.G.; Liu, J.L.; Jiang, X.M.; Ren, J.Z.; Ma, C.H.; Lei, W.; Su, R.W.; Yang, Z.M. Genome-wide identification of micro-ribonucleic acids associated with human endometrial receptivity in natural and stimulated cycles by deep sequencing. Fertil. Steril. 2011, 96, 150–155. [Google Scholar] [CrossRef] [PubMed]
- Sigurgeirsson, B.; Amark, H.; Jemt, A.; Ujvari, D.; Westgren, M.; Lundeberg, J.; Gidlof, S. Comprehensive RNA sequencing of healthy human endometrium at two time points of the menstrual cycle. Biol. Reprod. 2017, 96, 24–33. [Google Scholar] [PubMed]
- Suhorutshenko, M.; Kukushkina, V.; Velthut-Meikas, A.; Altmae, S.; Peters, M.; Magi, R.; Krjutskov, K.; Koel, M.; Codoner, F.M.; Martinez-Blanch, J.F.; et al. Endometrial receptivity revisited: Endometrial transcriptome adjusted for tissue cellular heterogeneity. Hum. Reprod. 2018, 33, 2074–2086. [Google Scholar] [CrossRef] [PubMed]
- Bolger, A.M.; Lohse, M.; Usadel, B. Trimmomatic: A flexible trimmer for Illumina sequence data. Bioinformatics 2014, 30, 2114–2120. [Google Scholar] [CrossRef] [PubMed]
- Friedlander, M.R.; Mackowiak, S.D.; Li, N.; Chen, W.; Rajewsky, N. miRDeep2 accurately identifies known and hundreds of novel microRNA genes in seven animal clades. Nucl. Acids Res. 2012, 40, 37–52. [Google Scholar] [CrossRef] [PubMed]
- Griffiths-Jones, S. The microRNA registry. Nucl. Acids Res. 2004, 32, D109–D111. [Google Scholar] [CrossRef] [PubMed]
- Robinson, M.D.; McCarthy, D.J.; Smyth, G.K. edgeR: A Bioconductor package for differential expression analysis of digital gene expression data. Bioinformatics 2010, 26, 139–140. [Google Scholar] [CrossRef] [PubMed]
- McCarthy, D.J.; Chen, Y.; Smyth, G.K. Differential expression analysis of multifactor RNA-Seq experiments with respect to biological variation. Nucl. Acids Res. 2012, 40, 4288–4297. [Google Scholar] [CrossRef] [PubMed]
- Robinson, M.D.; Oshlack, A. A scaling normalization method for differential expression analysis of RNA-seq data. Genom. Biol. 2010, 11, R25. [Google Scholar] [CrossRef] [PubMed]
- Friedlander, M.R.; Chen, W.; Adamidi, C.; Maaskola, J.; Einspanier, R.; Knespel, S.; Rajewsky, N. Discovering microRNAs from deep sequencing data using miRDeep. Nat. Biotechnol. 2008, 26, 407–415. [Google Scholar] [CrossRef] [PubMed]
- Wong, N.; Wang, X. miRDB: An online resource for microRNA target prediction and functional annotations. Nucl. Acids Res. 2015, 43, D146–D152. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.; Pertea, G.; Trapnell, C.; Pimentel, H.; Kelley, R.; Salzberg, S.L. TopHat2: Accurate alignment of transcriptomes in the presence of insertions, deletions and gene fusions. Genom. Biol. 2013, 14, R36. [Google Scholar] [CrossRef] [PubMed]
- Anders, S.; Pyl, P.T.; Huber, W. HTSeq—A Python framework to work with high-throughput sequencing data. Bioinformatics 2015, 31, 166–169. [Google Scholar] [CrossRef] [PubMed]
- Walter, W.; Sanchez-Cabo, F.; Ricote, M. GOplot: An R package for visually combining expression data with functional analysis. Bioinformatics 2015, 31, 2912–2914. [Google Scholar] [CrossRef] [PubMed]
- Saare, M.; Rekker, K.; Laisk-Podar, T.; Soritsa, D.; Roost, A.M.; Simm, J.; Velthut-Meikas, A.; Samuel, K.; Metsalu, T.; Karro, H.; et al. High-throughput sequencing approach uncovers the miRNome of peritoneal endometriotic lesions and adjacent healthy tissues. PLoS ONE 2014, 9, e112630. [Google Scholar] [CrossRef] [PubMed]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCt method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef] [PubMed]
- Kuokkanen, S.; Chen, B.; Ojalvo, L.; Benard, L.; Santoro, N.; Pollard, J.W. Genomic profiling of microRNAs and messenger RNAs reveals hormonal regulation in microRNA expression in human endometrium. Biol. Reprod. 2010, 82, 791–801. [Google Scholar] [CrossRef] [PubMed]
- Panda, H.; Pelakh, L.; Chuang, T.D.; Luo, X.; Bukulmez, O.; Chegini, N. Endometrial miR-200c is altered during transformation into cancerous states and targets the expression of ZEBs, VEGFA, FLT1, IKKβ, KLF9, and FBLN5. Reprod. Sci. 2012, 19, 786–796. [Google Scholar] [CrossRef] [PubMed]
- Sontakke, S.D.; Mohammed, B.T.; McNeilly, A.S.; Donadeu, F.X. Characterization of microRNAs differentially expressed during bovine follicle development. Reproduction 2014, 148, 271–283. [Google Scholar] [CrossRef] [PubMed]
- Aghajanova, L.; Altmae, S.; Bjuresten, K.; Hovatta, O.; Landgren, B.M.; Stavreus-Evers, A. Disturbances in the LIF pathway in the endometrium among women with unexplained infertility. Fertil. Steril. 2009, 91, 2602–2610. [Google Scholar] [CrossRef] [PubMed]
- Pawar, S.; Starosvetsky, E.; Orvis, G.D.; Behringer, R.R.; Bagchi, I.C.; Bagchi, M.K. STAT3 regulates uterine epithelial remodeling and epithelial-stromal crosstalk during implantation. Mol. Endocrinol. 2013, 27, 1996–2012. [Google Scholar] [CrossRef] [PubMed]
- Altmäe, S.; Reimand, J.; Hovatta, O.; Zhang, P.; Kere, J.; Laisk, T.; Saare, M.; Peters, M.; Vilo, J.; Stavreus-Evers, A.; et al. Research resource: Interactome of human embryo implantation: Identification of gene expression pathways, regulation, and integrated regulatory networks. Mol. Endocrinol. 2012, 26, 203–217. [Google Scholar] [CrossRef] [PubMed]
- Aghajanova, L. Update on the role of leukemia inhibitory factor in assisted reproduction. Curr. Opin. Obstet. Gynecol. 2010, 22, 213–219. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez, R.R.; Simon, C.; Caballero-Campo, P.; Norman, R.; Chardonnens, D.; Devoto, L.; Bischof, P. Leptin and reproduction. Hum. Reprod. Update 2000, 6, 290–300. [Google Scholar] [CrossRef] [PubMed]
- Margetic, S.; Gazzola, C.; Pegg, G.G.; Hill, R.A. Leptin: A review of its peripheral actions and interactions. Int. J. Obes. Relat. Metab. Disord. 2002, 26, 1407–1433. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.-M.; Hong, J.; Zhang, W.-X.; Li, Y. The effects of growth hormone on clinical outcomes after frozen-thawed embryo transfer. Int. J. Gynaecol. Obstet. 2016, 133, 347–350. [Google Scholar]
- Altmäe, S.; Mendoza-Tesarik, R.; Mendoza, C.; Mendoza, N.; Cucinelli, F.; Tesarik, J. Effect of growth hormone on uterine receptivity in women with repeated implantation failure in an oocyte donation program: A randomized controlled trial. J. Endocr. Soc. 2017, 2, 96–105. [Google Scholar] [CrossRef] [PubMed]
- Sonderegger, S.; Pollheimer, J.; Knofler, M. Wnt signalling in implantation, decidualisation and placental differentiation—Review. Placenta 2010, 31, 839–847. [Google Scholar] [CrossRef] [PubMed]
- Evans, G.E.; Phillipson, G.T.M.; Sykes, P.H.; McNoe, L.A.; Print, C.G.; Evans, J.J. Does the endometrial gene expression of fertile women vary within and between cycles? Hum. Reprod. 2018, 33, 452–463. [Google Scholar] [CrossRef] [PubMed]
- Rekker, K.; Saare, M.; Roost, A.M.; Salumets, A.; Peters, M. Circulating microRNA profile throughout the menstrual cycle. PLoS ONE 2013, 8, e81166. [Google Scholar] [CrossRef] [PubMed]
- Johnson, G.A.; Burghardt, R.C.; Bazer, F.W. Osteopontin: A leading candidate adhesion molecule for implantation in pigs and sheep. J. Anim. Sci. Biotechnol. 2014, 5, 56. [Google Scholar] [CrossRef] [PubMed]
- Kang, Y.J.; Forbes, K.; Carver, J.; Aplin, J.D. The role of the osteopontin-integrin αvβ3 interaction at implantation: Functional analysis using three different in vitro models. Hum. Reprod. 2014, 29, 739–749. [Google Scholar] [CrossRef] [PubMed]
- Casals, G.; Ordi, J.; Creus, M.; Fabregues, F.; Carmona, F.; Casamitjana, R.; Balasch, J. Osteopontin and αvβ3 integrin as markers of endometrial receptivity: The effect of different hormone therapies. Reprod. Biomed. Online 2010, 21, 349–359. [Google Scholar] [CrossRef] [PubMed]
- Haller-Kikkatalo, K.; Altmae, S.; Tagoma, A.; Uibo, R.; Salumets, A. Autoimmune activation toward embryo implantation is rare in immune-privileged human endometrium. Semin. Reprod. Med. 2014, 32, 376–384. [Google Scholar] [CrossRef] [PubMed]
- Pathare, A.D.S.; Zaveri, K.; Hinduja, I. Downregulation of genes related to immune and inflammatory response in IVF implantation failure cases under controlled ovarian stimulation. Am. J. Reprod. Immunol. 2017, 78. [Google Scholar] [CrossRef] [PubMed]
- Gamper, N.; Fillon, S.; Feng, Y.; Friedrich, B.; Lang, P.A.; Henke, G.; Huber, S.M.; Kobayashi, T.; Cohen, P.; Lang, F. K+ channel activation by all three isoforms of serum- and glucocorticoid-dependent protein kinase SGK. Pflugers Arch. 2002, 445, 60–66. [Google Scholar] [PubMed]
- Ruan, Y.C.; Chen, H.; Chan, H.C. Ion channels in the endometrium: Regulation of endometrial receptivity and embryo implantation. Hum. Reprod. Update 2014, 20, 517–529. [Google Scholar] [CrossRef] [PubMed]
- Shupp, A.; Casimiro, M.C.; Pestell, R.G. Biological functions of CDK5 and potential CDK5 targeted clinical treatments. Oncotarget 2017, 8, 17373–17382. [Google Scholar] [CrossRef] [PubMed]
- Seshadri, S.; Sunkara, S.K. Natural killer cells in female infertility and recurrent miscarriage: A systematic review and meta-analysis. Hum. Reprod. Update 2014, 20, 429–438. [Google Scholar] [CrossRef] [PubMed]
- Ortega, M.; Bhatnagar, H.; Lin, A.P.; Wang, L.; Aster, J.C.; Sill, H.; Aguiar, R.C. A microRNA-mediated regulatory loop modulates NOTCH and MYC oncogenic signals in B- and T-cell malignancies. Leukemia 2015, 29, 968–976. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Liu, Q.; Zhang, Q.; Liu, L. microRNA-30a-5p suppresses proliferation, invasion and tumor growth of hepatocellular cancer cells via targeting FOXA1. Oncol. Lett. 2017, 14, 5018–5026. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Ji, Q.; Zhang, C.; Liu, Y.; Liu, N.; Sui, H.; Zhou, L.; Wang, S.; Li, Q. miR-30a acts as a tumor suppressor by double-targeting COX-2 and BCL9 in H. pylori gastric cancer models. Sci. Rep. 2017, 7, 7113. [Google Scholar] [CrossRef] [PubMed]
- Rouzbeh, S.; Kobari, L.; Cambot, M.; Mazurier, C.; Hebert, N.; Faussat, A.M.; Durand, C.; Douay, L.; Lapillonne, H. Molecular signature of erythroblast enucleation in human embryonic stem cells. Stem Cells 2015, 33, 2431–2441. [Google Scholar] [CrossRef] [PubMed]
- de Rie, D.; Abugessaisa, I.; Alam, T.; Arner, E.; Arner, P.; Ashoor, H.; Astrom, G.; Babina, M.; Bertin, N.; Burroughs, A.M.; et al.; et al.; et al. An integrated expression atlas of miRNAs and their promoters in human and mouse. Nat. Biotechnol. 2017, 35, 872–878. [Google Scholar] [CrossRef] [PubMed]



| Novel miRNA Provisional ID | Sample Type | Consensus Mature Sequence | Endometrium | Blood | ||||
|---|---|---|---|---|---|---|---|---|
| Fertile Women | Recurrent Implantation Failure (RIF) Patients | Fertile Women | RIF Patients | |||||
| Detected from Early Secretory (ES) Samples (n = 37) | Detected from Mid-Secretory (MS) Samples (n = 37) | Detected from MS Samples (n = 36) | Detected from ES Samples (n = 34) | Detected from MS Samples (n = 34) | Detected from MS Samples (n = 24) | |||
| chr2_1900 | Endometrium | aucugaaauuugaaauggucc | - | 3 | 4 | - | - | - |
| chr2_4401 | Endometrium | gaacacugaaguuaauggcug | 1 | 26 | 29 | - | - | - |
| chr2_2219 | Blood | cugagaagacagucgaacuugac | - | - | - | 11 | 7 | 5 |
| chr3_7058 | Endometrium, blood | aaaaguaaucgcggucuuugcc | - | 2 | - | 3 | 4 | 5 |
| chr3_1133 | Endometrium, blood | caacaggccuugcucugcucacaga | 1 | - | 2 | 2 | - | 2 |
| chr3_2060 | Endometrium | cacaggcaggagaccccacagc | 7 | 2 | 1 | - | - | - |
| chr4_6331 | Endometrium | aacugggcauguuggaacuaagc | 2 | 1 | - | - | - | - |
| chr4_10018 | Endometrium | aucagagaaacuuucuugaac | 3 | 1 | 9 | - | - | - |
| chr8_10314 | Endometrium, blood | uuauccuccaguagacuaggga | 18 | 8 | 14 | 3 | 2 | 4 |
| chr8_8702 | Endometrium, blood | acaccggggguuagagcuucaacc | 2 | - | - | - | 1 | 2 |
| chr10_9545 | Endometrium, blood | aaaaguuauugcgguuuuugc | 1 | - | 1 | 1 | 2 | 2 |
| chr12_27523 | Endometrium, blood | ucuggcuccuuucuaaucac | - | 2 | - | 3 | 2 | 6 |
| chr13_23821 | Endometrium, blood | augugccuaguggcugcuguc | 5 | 5 | 4 | 6 | 6 | 7 |
| chr14_10307 | Blood | ucugagcccuguucucccuagg | - | - | - | - | - | 4 |
| chr14_3458 | Endometrium | aaaagucaucucgguucuugcc | 3 | 1 | 3 | - | - | - |
| chr15_26374 | Endometrium | aaacguaauuguggauuuugc | 2 | 1 | 3 | - | - | - |
| chr16_22077 | Endometrium | cugacugcccuggccuggccag | - | 2 | 2 | - | - | - |
| chr17_11615 | Endometrium, blood | uaacucuuagaauccccaaag | 1 | - | - | 18 | 16 | 10 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rekker, K.; Altmäe, S.; Suhorutshenko, M.; Peters, M.; Martinez-Blanch, J.F.; Codoñer, F.M.; Vilella, F.; Simón, C.; Salumets, A.; Velthut-Meikas, A. A Two-Cohort RNA-seq Study Reveals Changes in Endometrial and Blood miRNome in Fertile and Infertile Women. Genes 2018, 9, 574. https://doi.org/10.3390/genes9120574
Rekker K, Altmäe S, Suhorutshenko M, Peters M, Martinez-Blanch JF, Codoñer FM, Vilella F, Simón C, Salumets A, Velthut-Meikas A. A Two-Cohort RNA-seq Study Reveals Changes in Endometrial and Blood miRNome in Fertile and Infertile Women. Genes. 2018; 9(12):574. https://doi.org/10.3390/genes9120574
Chicago/Turabian StyleRekker, Kadri, Signe Altmäe, Marina Suhorutshenko, Maire Peters, Juan F. Martinez-Blanch, Francisco M. Codoñer, Felipe Vilella, Carlos Simón, Andres Salumets, and Agne Velthut-Meikas. 2018. "A Two-Cohort RNA-seq Study Reveals Changes in Endometrial and Blood miRNome in Fertile and Infertile Women" Genes 9, no. 12: 574. https://doi.org/10.3390/genes9120574
APA StyleRekker, K., Altmäe, S., Suhorutshenko, M., Peters, M., Martinez-Blanch, J. F., Codoñer, F. M., Vilella, F., Simón, C., Salumets, A., & Velthut-Meikas, A. (2018). A Two-Cohort RNA-seq Study Reveals Changes in Endometrial and Blood miRNome in Fertile and Infertile Women. Genes, 9(12), 574. https://doi.org/10.3390/genes9120574





