Identification of Transposable Elements Contributing to Tissue-Specific Expression of Long Non-Coding RNAs
Abstract
1. Introduction
2. Materials and Methods
2.1. Input Data
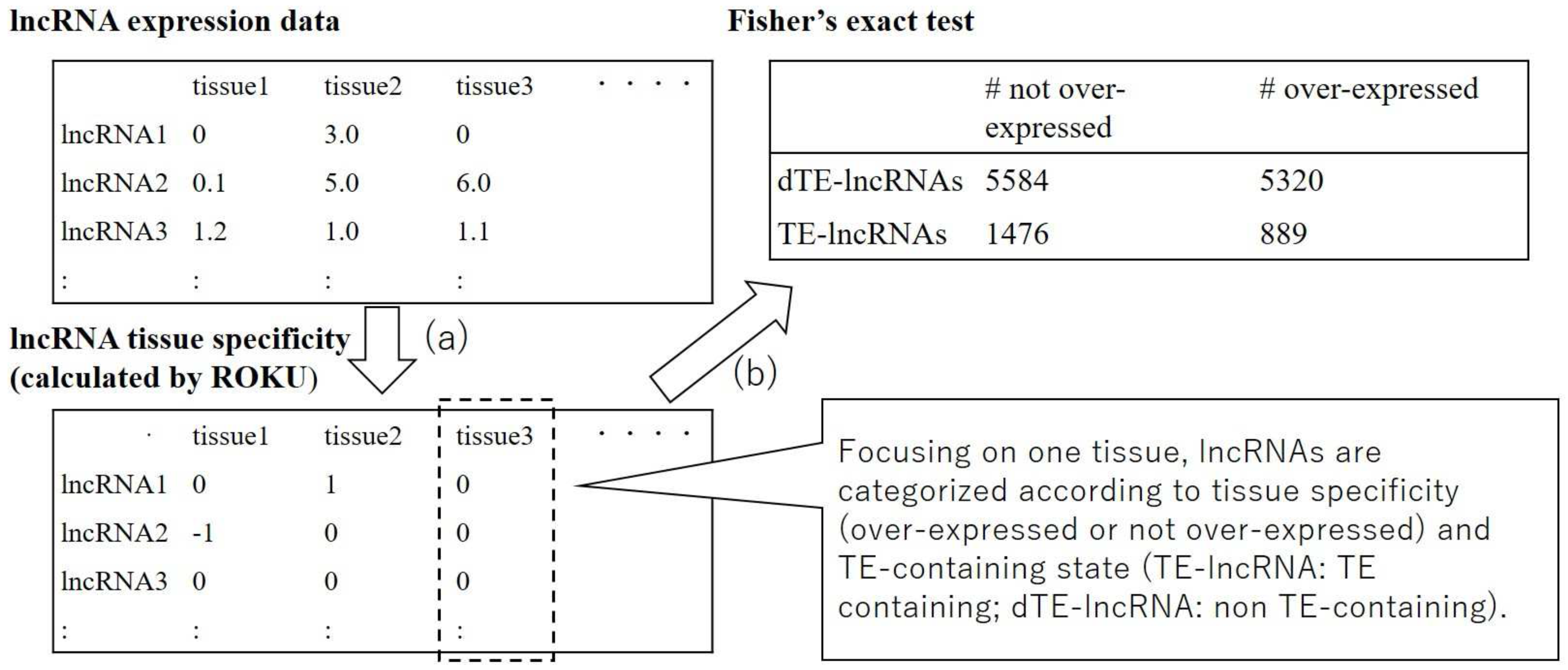
2.2. Identification of Transposable Elements that Contribute to Tissue-Specific Expression of Long Non-Coding RNAs
3. Results
3.1. Multiple Transposable Element Families Are Significantly Related to Tissue-Specific Expression of Long Non-Coding RNAs
3.1.1. Transposable Element Family-Level Analysis
3.1.2. Transposable Element Subfamily-Level Analysis
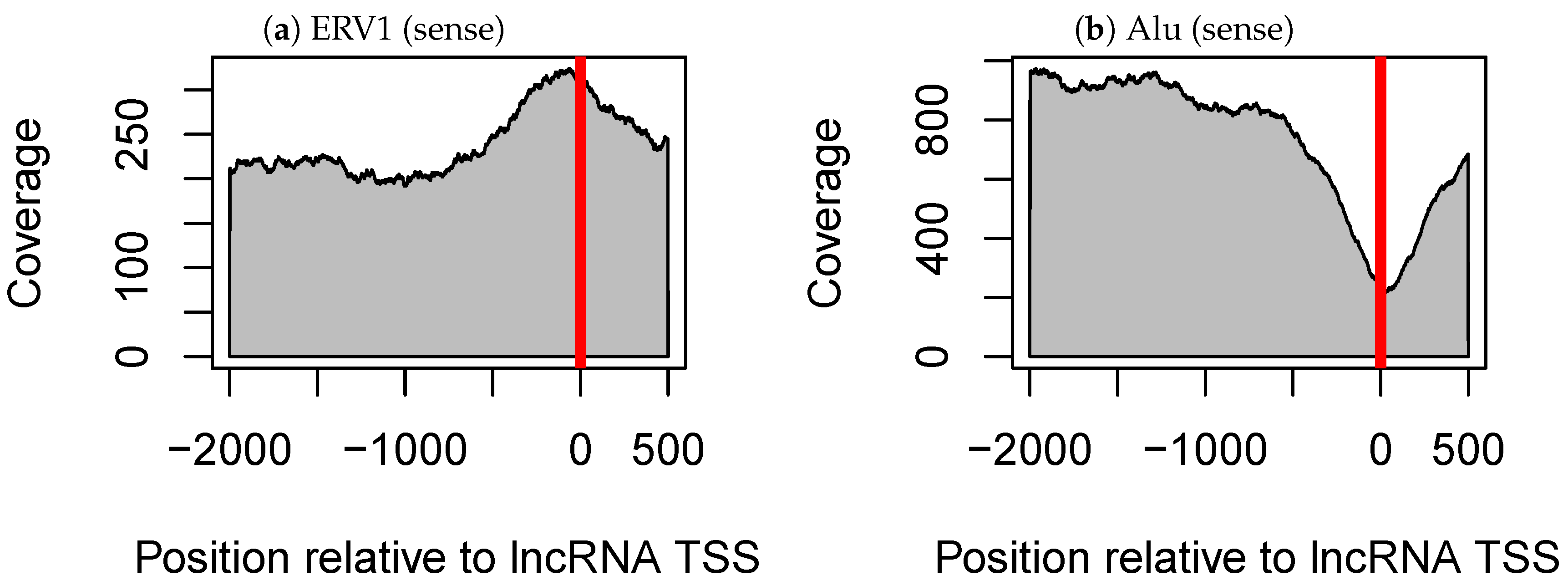
3.2. The Alu Family Is Significantly Related not only to Tissue-Specific Expression of Long Non-Coding RNAs but also to that of mRNAs
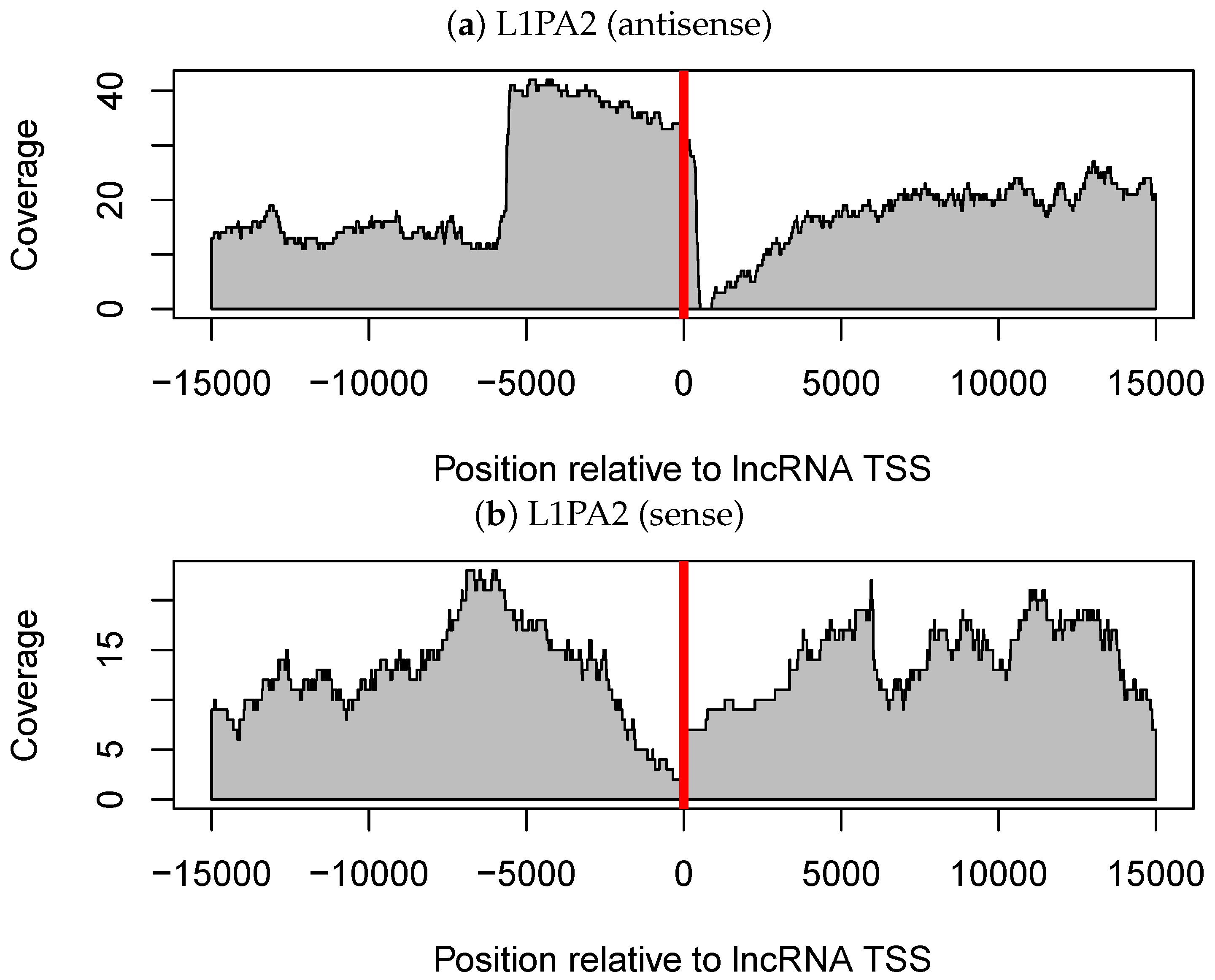
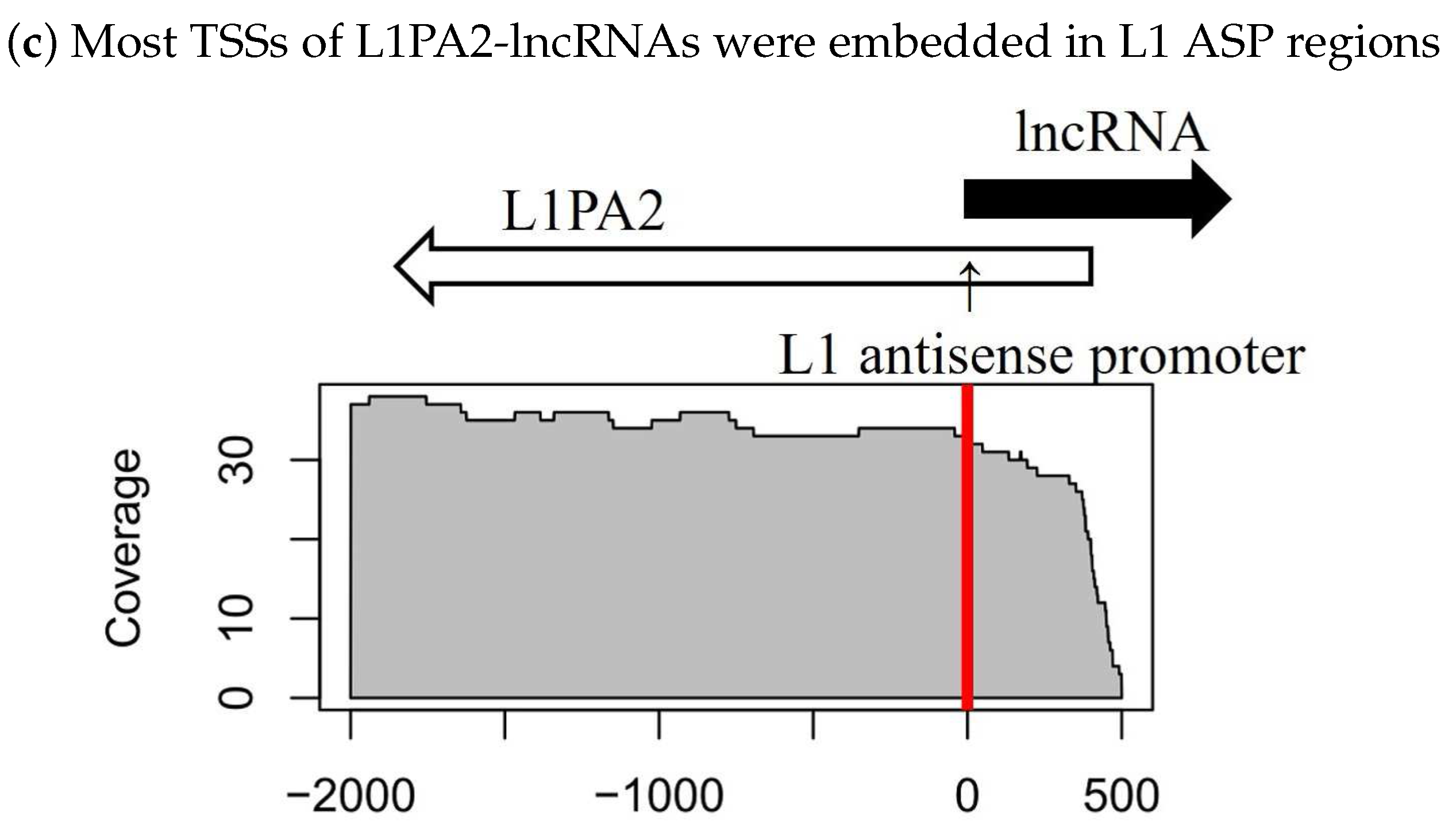
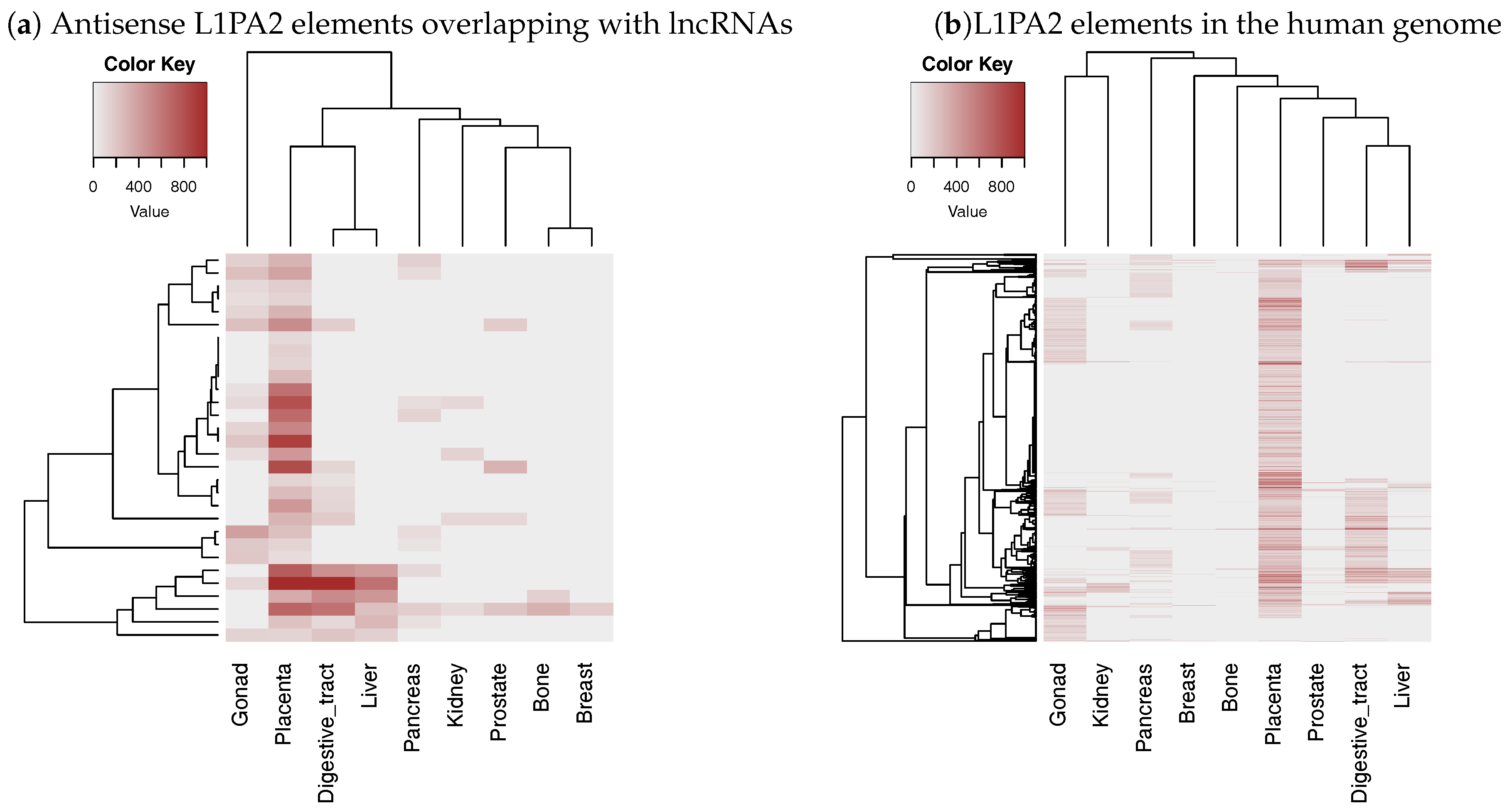
3.3. The Antisense Promoter of L1PA2 May Contribute to Placenta-Specific Transcription of Long Non-Coding RNAs
3.3.1. Antisense L1PA2 Was Enriched in Long Non-Coding RNA TSSs and Overlapped with lncRNAs by Approximately 500 nt
3.3.2. The Level of H3K4me3 in L1PA2- Long Non-Coding RNAs Regions Increased Specifically in Placenta Tissue
4. Discussion
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Iyer, M.K.; Niknafs, Y.S.; Malik, R.; Singhal, U.; Sahu, A.; Hosono, Y.; Barrette, T.R.; Prensner, J.R.; Evans, J.R.; Zhao, S.; et al. The landscape of long noncoding RNAs in the human transcriptome. Nat. Genet. 2015, 47, 199–208. [Google Scholar] [CrossRef] [PubMed]
- Hon, C.C.; Ramilowski, J.A.; Harshbarger, J.; Bertin, N.; Rackham, O.J.; Gough, J.; Denisenko, E.; Schmeier, S.; Poulsen, T.M.; Severin, J.; et al. An atlas of human long non-coding RNAs with accurate 5’ ends. Nature 2017, 543, 199–204. [Google Scholar] [CrossRef] [PubMed]
- You, B.H.; Yoon, S.H.; Nam, J.W. High-confidence coding and noncoding transcriptome maps. Genome Res. 2017, 27, 1050–1062. [Google Scholar] [CrossRef] [PubMed]
- Mercer, T.R.; Dinger, M.E.; Mattick, J.S. Long non-coding RNAs: Insights into functions. Nat. Rev. Genet. 2009, 10, 155–159. [Google Scholar] [CrossRef] [PubMed]
- Guttman, M.; Amit, I.; Garber, M.; French, C.; Lin, M.F.; Feldser, D.; Huarte, M.; Zuk, O.; Carey, B.W.; Cassady, J.P.; et al. Chromatin signature reveals over a thousand highly conserved large non-coding RNAs in mammals. Nature 2009, 458, 223–227. [Google Scholar] [CrossRef] [PubMed]
- Derrien, T.; Johnson, R.; Bussotti, G.; Tanzer, A.; Djebali, S.; Tilgner, H.; Guernec, G.; Martin, D.; Merkel, A.; Knowles, D.G.; et al. The GENCODE v7 catalog of human long noncoding RNAs: Analysis of their gene structure, evolution, and expression. Genome Res. 2012, 22, 1775–1789. [Google Scholar] [CrossRef] [PubMed]
- Hirose, T.; Mishima, Y.; Tomari, Y. Elements and machinery of non-coding RNAs: Toward their taxonomy. EMBO Rep. 2014, 15, 489–507. [Google Scholar] [CrossRef] [PubMed]
- Signal, B.; Gloss, B.S.; Dinger, M.E. Computational Approaches for Functional Prediction and Characterisation of Long Noncoding RNAs. Trends Genet. 2016, 32, 620–637. [Google Scholar] [CrossRef] [PubMed]
- De Hoon, M.; Shin, J.W.; Carninci, P. Paradigm shifts in genomics through the FANTOM projects. Mamm. Genome 2015, 26, 391–402. [Google Scholar] [CrossRef] [PubMed]
- Wutz, A.; Rasmussen, T.P.; Jaenisch, R. Chromosomal silencing and localization are mediated by different domains of Xist RNA. Nat. Genet. 2002, 30, 167–174. [Google Scholar] [CrossRef] [PubMed]
- Lander, E.S.; Linton, L.M.; Birren, B.; Nusbaum, C.; Zody, M.C.; Baldwin, J.; Devon, K.; Dewar, K.; Doyle, M.; FitzHugh, W.; et al. Initial sequencing and analysis of the human genome. Nature 2001, 409, 860–921. [Google Scholar] [CrossRef] [PubMed]
- Mi, S.; Lee, X.; Li, X.; Veldman, G.M.; Finnerty, H.; Racie, L.; LaVallie, E.; Tang, X.Y.; Edouard, P.; Howes, S.; et al. Syncytin is a captive retroviral envelope protein involved in human placental morphogenesis. Nature 2000, 403, 785–789. [Google Scholar] [CrossRef] [PubMed]
- Conley, A.B.; Piriyapongsa, J.; Jordan, I.K. Retroviral promoters in the human genome. Bioinformatics 2008, 24, 1563–1567. [Google Scholar] [CrossRef] [PubMed]
- Faulkner, G.J.; Kimura, Y.; Daub, C.O.; Wani, S.; Plessy, C.; Irvine, K.M.; Schroder, K.; Cloonan, N.; Steptoe, A.L.; Lassmann, T.; et al. The regulated retrotransposon transcriptome of mammalian cells. Nat. Genet. 2009, 41, 563–571. [Google Scholar] [CrossRef] [PubMed]
- Kelley, D.; Rinn, J. Transposable elements reveal a stem cell-specific class of long noncoding RNAs. Genome Biol. 2012, 13, R107. [Google Scholar] [CrossRef] [PubMed]
- Kannan, S.; Chernikova, D.; Rogozin, I.B.; Poliakov, E.; Managadze, D.; Koonin, E.V.; Milanesi, L. Transposable Element Insertions in Long Intergenic Non-Coding RNA Genes. Front. Bioeng. Biotechnol. 2015, 3, 71. [Google Scholar] [CrossRef] [PubMed]
- Johnson, R.; Guigo, R. The RIDL hypothesis: Transposable elements as functional domains of long noncoding RNAs. RNA 2014, 20, 959–976. [Google Scholar] [CrossRef] [PubMed]
- Gong, C.; Maquat, L.E. lncRNAs transactivate STAU1-mediated mRNA decay by duplexing with 3’ UTRs via Alu elements. Nature 2011, 470, 284–288. [Google Scholar] [CrossRef] [PubMed]
- Lu, X.; Sachs, F.; Ramsay, L.; Jacques, P.E.; Goke, J.; Bourque, G.; Ng, H.H. The retrovirus HERVH is a long noncoding RNA required for human embryonic stem cell identity. Nat. Struct. Mol. Biol. 2014, 21, 423–425. [Google Scholar] [CrossRef] [PubMed]
- RepeatMasker Open-4.0. 2013–2015. Available online: http://www.repeatmasker.org (accessed on 5 January 2018).
- Iwakiri, J.; Terai, G.; Hamada, M. Computational prediction of lncRNA-mRNA interactionsby integrating tissue specificity in human transcriptome. Biol. Direct 2017, 12, 15. [Google Scholar] [CrossRef] [PubMed]
- Petryszak, R.; Burdett, T.; Fiorelli, B.; Fonseca, N.A.; Gonzalez-Porta, M.; Hastings, E.; Huber, W.; Jupp, S.; Keays, M.; Kryvych, N.; et al. Expression Atlas update—A database of gene and transcript expression from microarray- and sequencing-based functional genomics experiments. Nucleic Acids Res. 2014, 42, D926–D932. [Google Scholar] [CrossRef] [PubMed]
- Cabili, M.N.; Trapnell, C.; Goff, L.; Koziol, M.; Tazon-Vega, B.; Regev, A.; Rinn, J.L. Integrative annotation of human large intergenic noncoding RNAs reveals global properties and specific subclasses. Genes Dev. 2011, 25, 1915–1927. [Google Scholar] [CrossRef] [PubMed]
- Uhlen, M.; Fagerberg, L.; Hallstrom, B.M.; Lindskog, C.; Oksvold, P.; Mardinoglu, A.; Sivertsson, Å.; Kampf, C.; Sjöstedt, E.; Asplund, A.; et al. Proteomics. Tissue-based map of the human proteome. Science 2015, 347. [Google Scholar] [CrossRef] [PubMed]
- Ardlie, K.G.; Deluca, D.S.; Segre, A.V.; Sullivan, T.J.; Young, T.R.; Gelfand, E.T.; Trowbridge, C.A.; Maller, J.B.; Tukiainen, T.; Lek, M.; et al. Human genomics. The Genotype-Tissue Expression (GTEx) pilot analysis: Multitissue gene regulation in humans. Science 2015, 348, 648–660. [Google Scholar]
- Kadota, K.; Ye, J.; Nakai, Y.; Terada, T.; Shimizu, K. ROKU: A novel method for identification of tissue-specific genes. BMC Bioinform. 2006, 7, 294. [Google Scholar] [CrossRef] [PubMed]
- Benjamini, Y.; Hochberg, Y. Controlling the False Discovery Rate: A Practical and Powerful Approach to Multiple Testing. J. R. Stat. Soc. Ser. B (Methodol.) 1995, 57, 289–300. [Google Scholar]
- Speek, M. Antisense promoter of human L1 retrotransposon drives transcription of adjacent cellular genes. Mol. Cell. Biol. 2001, 21, 1973–1985. [Google Scholar] [CrossRef] [PubMed]
- Nigumann, P.; Redik, K.; Matlik, K.; Speek, M. Many human genes are transcribed from the antisense promoter of L1 retrotransposon. Genomics 2002, 79, 628–634. [Google Scholar] [CrossRef] [PubMed]
- Matlik, K.; Redik, K.; Speek, M. L1 antisense promoter drives tissue-specific transcription of human genes. J. Biomed. Biotechnol. 2006, 2006, 71753. [Google Scholar] [CrossRef] [PubMed]
- Criscione, S.W.; Theodosakis, N.; Micevic, G.; Cornish, T.C.; Burns, K.H.; Neretti, N.; Rodic, N. Genome-wide characterization of human L1 antisense promoter-driven transcripts. BMC Genom. 2016, 17, 463. [Google Scholar] [CrossRef] [PubMed]
- Sundaram, V.; Cheng, Y.; Ma, Z.; Li, D.; Xing, X.; Edge, P.; Snyder, M.P.; Wang, T. Widespread contribution of transposable elements to the innovation of gene regulatory networks. Genome Res. 2014, 24, 1963–1976. [Google Scholar] [CrossRef] [PubMed]
- Nishikura, K. A-to-I editing of coding and non-coding RNAs by ADARs. Nat. Rev. Mol. Cell Biol. 2016, 17, 83–96. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Zhou, X.; Jin, Y. ADAR-mediated RNA editing in non-coding RNA sequences. Sci. China Life Sci. 2013, 56, 944–952. [Google Scholar] [CrossRef] [PubMed]
- Girard, A.; Sachidanandam, R.; Hannon, G.J.; Carmell, M.A. A germline-specific class of small RNAs binds mammalian Piwi proteins. Nature 2006, 442, 199–202. [Google Scholar] [CrossRef] [PubMed]
- Ishizu, H.; Siomi, H.; Siomi, M.C. Biology of PIWI-interacting RNAs: New insights into biogenesis and function inside and outside of germlines. Genes Dev. 2012, 26, 2361–2373. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, T.; Cheng, E.C.; Zhong, M.; Lin, H. Retrotransposons and pseudogenes regulate mRNAs and lncRNAs via the piRNA pathway in the germline. Genome Res. 2015, 25, 368–380. [Google Scholar] [CrossRef] [PubMed]
- Chakraborty, S.; Deb, A.; Maji, R.K.; Saha, S.; Ghosh, Z. LncRBase: An enriched resource for lncRNA information. PLoS ONE 2014, 9, e108010. [Google Scholar] [CrossRef] [PubMed]
- Ha, H.; Song, J.; Wang, S.; Kapusta, A.; Feschotte, C.; Chen, K.C.; Xing, J. A comprehensive analysis of piRNAs from adult human testis and their relationship with genes and mobile elements. BMC Genom. 2014, 15, 545. [Google Scholar] [CrossRef] [PubMed]
- Lubelsky, Y.; Ulitsky, I. Sequences enriched in Alu repeats drive nuclear localization of long RNAs in human cells. bioRxiv 2017. [Google Scholar] [CrossRef]
- Carlevaro-Fita, J.; Das, M.; Polidori, T.; Navarro, C.; Johnson, R. Ancient exapted transposable elements promote nuclear enrichment of long noncoding RNAs. bioRxiv 2017. [Google Scholar] [CrossRef]
- Kelley, D.R.; Hendrickson, D.G.; Tenen, D.; Rinn, J.L. Transposable elements modulate human RNA abundance and splicing via specific RNA-protein interactions. Genome Biol. 2014, 15, 537. [Google Scholar] [CrossRef] [PubMed]
| 1. | This bed file was obtained from the following url: http://dbarchive.biosciencedbc.jp/kyushu-u/hg19/assembled/His.ALL.05.H3K4me3.AllCell.bed (ChIP-Atlas v7, accessed on Jul 31st, 2017). |
| ID | Expression Atlas ID | Data Provider | # Tissues | # Samples | Reference |
|---|---|---|---|---|---|
| 1 | E-MTAB-513 | Illumina Body Map | 16 | 19 | [23] |
| 2 | E-MTAB-2836 | Human Protein Atlas | 32 | 122 | [24] |
| 3 | E-MTAB-2919 | Genotype-Tissue Expression (GTEx) | 53 | 3282 | [25] |
| No. | TE Family | Tissue | Strand | Effect | Data ID |
|---|---|---|---|---|---|
| 1 | LINE.L1 | Brain | − | more specific | 1 |
| 2 | LINE.L1 | Cerebral_cortex | − | more specific | 2 |
| 3 | LTR.ERV1 | Leukocyte | + | more specific | 1 |
| 4 | LTR.ERV1 | Placenta | + | more specific | 2 |
| 5 | LTR.ERV1 | Testis | + | more specific | 1 |
| 6 | LTR.ERV1 | Testis | + | more specific | 2 |
| 7 | LTR.ERV1 | Testis | + | more specific | 3 |
| 8 | LTR.ERVL | Bone_marrow | + | less specific | 2 |
| 9 | LTR.ERVL.MaLR | Bone_marrow | +/− | less specific | 2 |
| 10 | SINE.Alu | Adrenal | +/− | more specific | 1 |
| 11 | SINE.Alu | Bone_marrow | + | more specific | 2 |
| 12 | SINE.Alu | Brain | + | more specific | 3 |
| 13 | SINE.Alu | Lymph_node | − | more specific | 1 |
| 14 | SINE.Alu | Skin | +/− | more specific | 2 |
| 15 | SINE.Alu | Testis | +/− | less specific | 1 |
| 16 | SINE.Alu | Testis | +/− | less specific | 2 |
| 17 | SINE.Alu | Testis | +/− | less specific | 3 |
| No. | TE Subfamily | Tissue | Strand | Effect | Data ID |
|---|---|---|---|---|---|
| 1 | AluJb | Adrenal | − | more specific | 1 |
| 2 | AluSc | Adrenal | +/− | more specific | 1 |
| 3 | AluSg | Adrenal | − | more specific | 1 |
| 4 | AluSp | Adrenal | +/− | more specific | 1 |
| 5 | AluSq2 | Adrenal | − | more specific | 1 |
| 6 | AluSx | Adrenal | +/− | more specific | 1 |
| 7 | AluSx | Testis | +/− | less specific | 1 |
| 8 | AluSx1 | Adrenal | +/− | more specific | 1 |
| 9 | AluSx1 | Testis | + | less specific | 1 |
| 10 | AluSz | Adrenal | +/− | more specific | 1 |
| 11 | AluY | Adrenal | + | more specific | 1 |
| 12 | L1PA2 | Placenta | − | more specific | 2 |
| No. | TE Family | Tissue | Strand | Effect | Data ID |
|---|---|---|---|---|---|
| 1 | DNA | Brain | − | more specific | 1 |
| 2 | DNA.TcMar.Tigger | Testis | − | less specific | 3 |
| 3 | DNA.hAT.Blackjack | Lung | − | more specific | 3 |
| 4 | DNA.hAT.Charlie | Brain | − | more specific | 1 |
| 5 | DNA.hAT.Charlie | Testis | + | less specific | 1 |
| 6 | DNA.hAT.Charlie | Testis | + | less specific | 2 |
| 7 | DNA.hAT.Charlie | Thyroid | + | more specific | 1 |
| 8 | LINE.CR1 | Brain | +/− | more specific | 1 |
| 9 | LINE.CR1 | Cerebral_cortex | +/− | more specific | 2 |
| 10 | LINE.CR1 | Kidney | + | more specific | 1 |
| 11 | LINE.L2 | Brain | − | more specific | 1 |
| 12 | LINE.L2 | Gall_bladder | + | more specific | 2 |
| 13 | LINE.L2 | Ovary | + | more specific | 1 |
| 14 | LTR.ERV1 | Testis | + | less specific | 1 |
| 15 | LTR.ERVK | Liver | − | more specific | 1 |
| 16 | LTR.ERVL | Skeletal_muscle | + | less specific | 1 |
| 17 | LTR.Gypsy | Brain | + | more specific | 1 |
| 18 | RC..Helitron. | Heart | + | more specific | 1 |
| 19 | SINE.Alu | Esophagus | − | less specific | 2 |
| 20 | SINE.Alu | Lung | +/− | less specific | 1 |
| 21 | SINE.Alu | Lymph_node | − | less specific | 1 |
| 22 | SINE.Alu | Minor_salivary_gland | + | less specific | 3 |
| 23 | SINE.Alu | Salivary_gland | + | less specific | 2 |
| 24 | SINE.Alu | Stomach | + | less specific | 2 |
| 25 | SINE.Alu | Testis | +/− | less specific | 1 |
| 26 | SINE.Alu | Testis | +/− | less specific | 2 |
| 27 | SINE.MIR | Brain | +/− | more specific | 1 |
| 28 | SINE.MIR | Brain | +/− | more specific | 3 |
| 29 | SINE.MIR | Cerebral_cortex | +/− | more specific | 2 |
| 30 | SINE.MIR | Ovary | +/− | more specific | 1 |
| 31 | SINE.MIR | Prostate | + | more specific | 1 |
| 32 | SINE.MIR | Testis | +/− | less specific | 1 |
| 33 | SINE.MIR | Testis | − | less specific | 2 |
| No. | TE Subfamily | Tissue | Strand | Effect | Data ID |
|---|---|---|---|---|---|
| 1 | MIR3 | Brain | +/− | more specific | 1 |
| 2 | MIR3 | Testis | − | less specific | 2 |
| 3 | MIRc | Brain | +/− | more specific | 1 |
| 4 | MIRc | Cerebral_cortex | − | more specific | 2 |
| 5 | MIRc | Ovary | − | more specific | 1 |
| 6 | MamGyp.int | Brain | + | more specific | 1 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chishima, T.; Iwakiri, J.; Hamada, M. Identification of Transposable Elements Contributing to Tissue-Specific Expression of Long Non-Coding RNAs. Genes 2018, 9, 23. https://doi.org/10.3390/genes9010023
Chishima T, Iwakiri J, Hamada M. Identification of Transposable Elements Contributing to Tissue-Specific Expression of Long Non-Coding RNAs. Genes. 2018; 9(1):23. https://doi.org/10.3390/genes9010023
Chicago/Turabian StyleChishima, Takafumi, Junichi Iwakiri, and Michiaki Hamada. 2018. "Identification of Transposable Elements Contributing to Tissue-Specific Expression of Long Non-Coding RNAs" Genes 9, no. 1: 23. https://doi.org/10.3390/genes9010023
APA StyleChishima, T., Iwakiri, J., & Hamada, M. (2018). Identification of Transposable Elements Contributing to Tissue-Specific Expression of Long Non-Coding RNAs. Genes, 9(1), 23. https://doi.org/10.3390/genes9010023




