The Role of c-MYC in B-Cell Lymphomas: Diagnostic and Molecular Aspects
Abstract
:1. Introduction
2. c-MYC in B-Cell Development
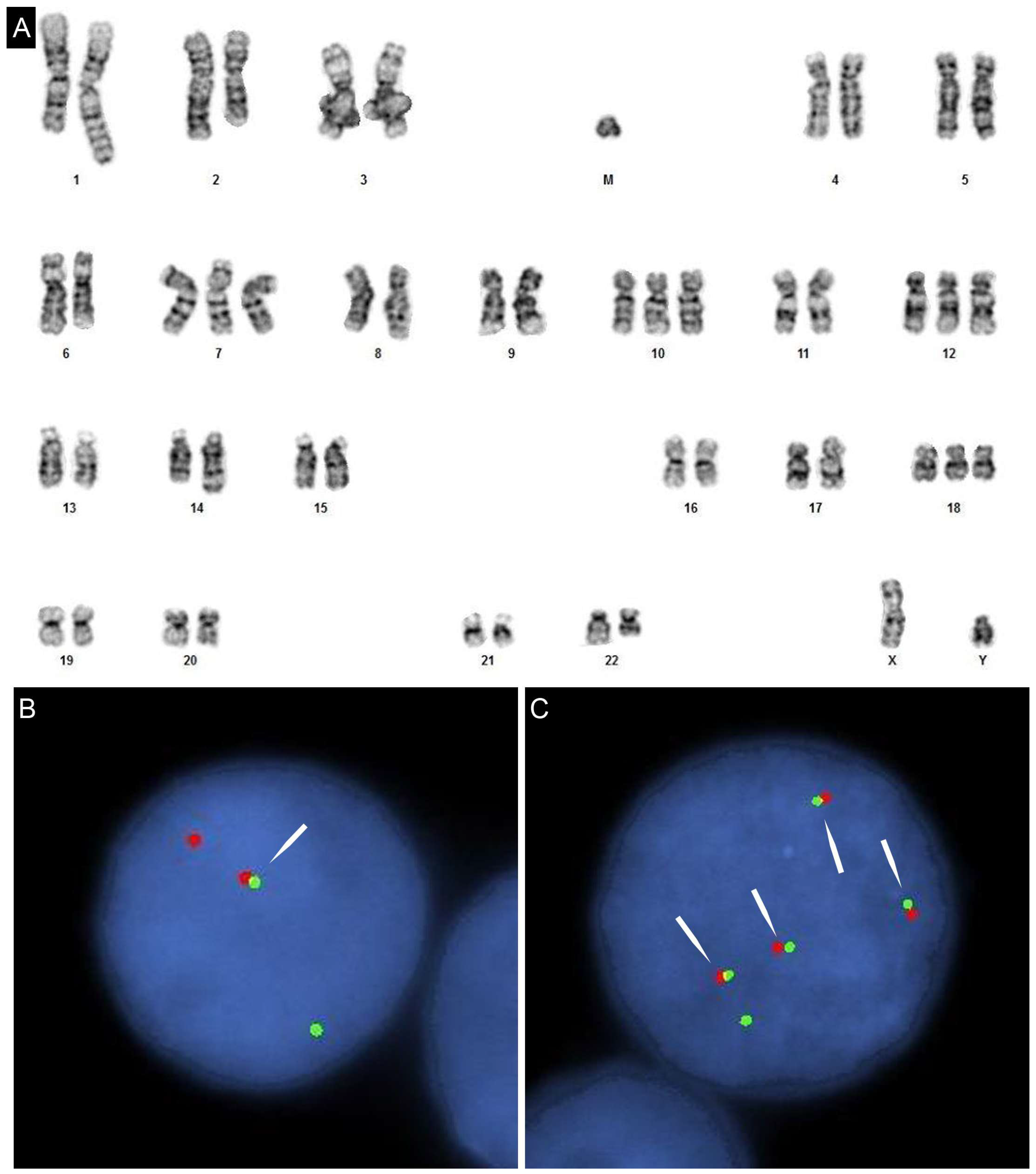
3. Clinical Detection of c-MYC Abnormalities
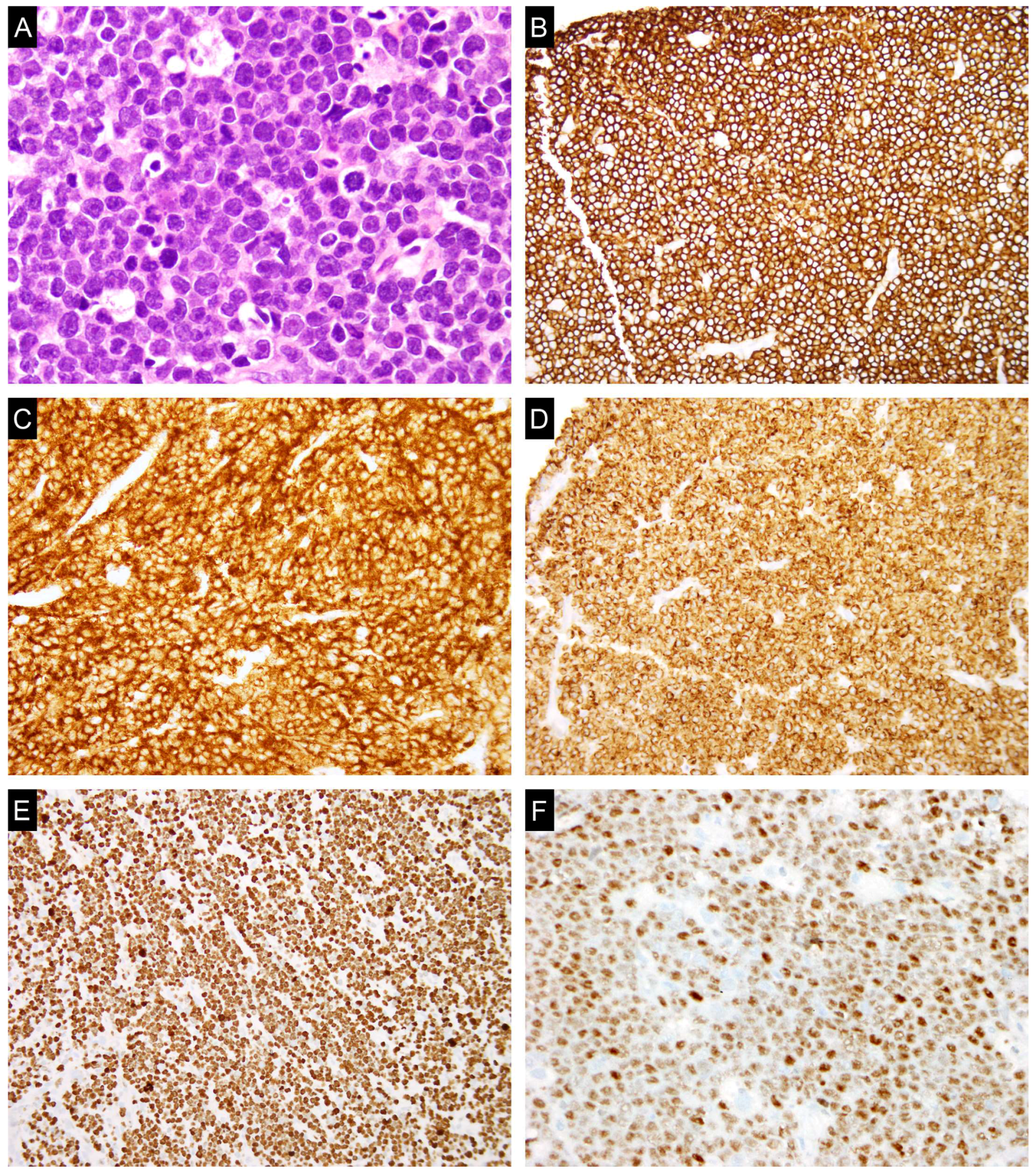
4. c-MYC in Burkitt Lymphoma
5. c-MYC in Diffuse Large B-Cell Lymphoma and Double-Hit Lymphoma
6. c-MYC in Plasmablastic Lymphoma
7. c-MYC in Mantle Cell Lymphoma
8. c-MYC in Low Grade B-Cell Lymphomas
9. Conclusions
Conflicts of Interest
References
- Sheiness, D.; Fanshier, L.; Bishop, J.M. Identification of nucleotide sequences which may encode the oncogenic capacity of avian retrovirus MC29. J. Virol. 1978, 28, 600–610. [Google Scholar] [PubMed]
- Bishop, J.M. Retroviruses and cancer genes. Adv. Cancer Res. 1982, 37, 1–32. [Google Scholar] [PubMed]
- Dalla-Favera, R.; Bregni, M.; Erikson, J.; Patterson, D.; Gallo, R.C.; Croce, C.M. Human c-myc onc gene is located on the region of chromosome 8 that is translocated in Burkitt lymphoma cells. Proc. Natl. Acad. Sci. USA 1982, 79, 7824–7827. [Google Scholar] [CrossRef] [PubMed]
- Neel, B.G.; Jhanwar, S.C.; Chaganti, R.S.; Hayward, W.S. Two human c-onc genes are located on the long arm of chromosome 8. Proc. Natl. Acad. Sci. USA 1982, 79, 7842–7846. [Google Scholar] [CrossRef] [PubMed]
- Taub, R.; Kirsch, I.; Morton, C.; Lenoir, G.; Swan, D.; Tronick, S.; Aaronson, S.; Leder, P. Translocation of the c-myc gene into the immunoglobulin heavy chain locus in human Burkitt lymphoma and murine plasmacytoma cells. Proc. Natl. Acad. Sci. USA 1982, 79, 7837–7841. [Google Scholar] [CrossRef] [PubMed]
- Adams, J.M.; Harris, A.W.; Pinkert, C.A.; Corcoran, L.M.; Alexander, W.S.; Cory, S.; Palmiter, R.D.; Brinster, R.L. The c-myc oncogene driven by immunoglobulin enhancers induces lymphoid malignancy in transgenic mice. Nature 1985, 318, 533–538. [Google Scholar] [CrossRef] [PubMed]
- Knoepfler, P.S. Myc goes global: New tricks for an old oncogene. Cancer Res. 2007, 67, 5061–5063. [Google Scholar] [CrossRef] [PubMed]
- Dang, C.V.; O’Donnell, K.A.; Zeller, K.I.; Nguyen, T.; Osthus, R.C.; Li, F. The c-Myc target gene network. Semin. Cancer Biol. 2006, 16, 253–264. [Google Scholar] [CrossRef] [PubMed]
- Spencer, C.A.; Groudine, M. Control of c-myc regulation in normal and neoplastic cells. Adv. Cancer Res. 1991, 56, 1–48. [Google Scholar] [PubMed]
- Taub, R.; Moulding, C.; Battey, J.; Murphy, W.; Vasicek, T.; Lenoir, G.M.; Leder, P. Activation and somatic mutation of the translocated c-myc gene in burkitt lymphoma cells. Cell 1984, 36, 339–348. [Google Scholar] [CrossRef]
- Herrick, D.J.; Ross, J. The half-life of c-myc mRNA in growing and serum-stimulated cells: Influence of the coding and 3’ untranslated regions and role of ribosome translocation. Mol. Cell. Biol. 1994, 14, 2119–2128. [Google Scholar] [CrossRef] [PubMed]
- Hann, S.R.; Eisenman, R.N. Proteins encoded by the human c-myc oncogene: Differential expression in neoplastic cells. Mol. Cell. Biol. 1984, 4, 2486–2497. [Google Scholar] [CrossRef] [PubMed]
- Ciechanover, A.; DiGiuseppe, J.A.; Schwartz, A.L.; Brodeur, G.M. Degradation of MYCN oncoprotein by the ubiquitin system. Prog. Clin. Biol. Res. 1991, 366, 37–43. [Google Scholar] [PubMed]
- Pelengaris, S.; Khan, M.; Evan, G. c-MYC: More than just a matter of life and death. Nat. Rev. Cancer 2002, 2, 764–776. [Google Scholar] [CrossRef] [PubMed]
- Dudley, J.P.; Mertz, J.A.; Rajan, L.; Lozano, M.; Broussard, D.R. What retroviruses teach us about the involvement of c-Myc in leukemias and lymphomas. Leukemia 2002, 16, 1086–1098. [Google Scholar] [CrossRef] [PubMed]
- Alitalo, K.; Schwab, M.; Lin, C.C.; Varmus, H.E.; Bishop, J.M. Homogeneously staining chromosomal regions contain amplified copies of an abundantly expressed cellular oncogene (c-myc) in malignant neuroendocrine cells from a human colon carcinoma. Proc. Natl. Acad. Sci. USA 1983, 80, 1707–1711. [Google Scholar] [CrossRef] [PubMed]
- Collins, S.; Groudine, M. Amplification of endogenous myc-related DNA sequences in a human myeloid leukaemia cell line. Nature 1982, 298, 679–681. [Google Scholar] [CrossRef] [PubMed]
- Little, C.D.; Nau, M.M.; Carney, D.N.; Gazdar, A.F.; Minna, J.D. Amplification and expression of the c-myc oncogene in human lung cancer cell lines. Nature 1983, 306, 194–196. [Google Scholar] [CrossRef] [PubMed]
- Mariani-Costantini, R.; Escot, C.; Theillet, C.; Gentile, A.; Merlo, G.; Lidereau, R.; Callahan, R. In situ c-myc expression and genomic status of the c-myc locus in infiltrating ductal carcinomas of the breast. Cancer Res. 1988, 48, 199–205. [Google Scholar] [PubMed]
- Munzel, P.; Marx, D.; Kochel, H.; Schauer, A.; Bock, K.W. Genomic alterations of the c-myc protooncogene in relation to the overexpression of c-erbB2 and Ki-67 in human breast and cervix carcinomas. J. Cancer Res. Clin. Oncol. 1991, 117, 603–607. [Google Scholar] [CrossRef] [PubMed]
- Augenlicht, L.H.; Wadler, S.; Corner, G.; Richards, C.; Ryan, L.; Multani, A.S.; Pathak, S.; Benson, A.; Haller, D.; Heerdt, B.G. Low-level c-myc amplification in human colonic carcinoma cell lines and tumors: A frequent, p53-independent mutation associated with improved outcome in a randomized multi-institutional trial. Cancer Res. 1997, 57, 1769–1775. [Google Scholar] [PubMed]
- Erikson, J.; ar-Rushdi, A.; Drwinga, H.L.; Nowell, P.C.; Croce, C.M. Transcriptional activation of the translocated c-myc oncogene in burkitt lymphoma. Proc. Natl. Acad. Sci. USA 1983, 80, 820–824. [Google Scholar] [CrossRef] [PubMed]
- Littlewood, T.D.; Kreuzaler, P.; Evan, G.I. All things to all people. Cell 2012, 151, 11–13. [Google Scholar] [CrossRef] [PubMed]
- Nie, Z.; Hu, G.; Wei, G.; Cui, K.; Yamane, A.; Resch, W.; Wang, R.; Green, D.R.; Tessarollo, L.; Casellas, R.; et al. c-Myc is a universal amplifier of expressed genes in lymphocytes and embryonic stem cells. Cell 2012, 151, 68–79. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.Y.; Loven, J.; Rahl, P.B.; Paranal, R.M.; Burge, C.B.; Bradner, J.E.; Lee, T.I.; Young, R.A. Transcriptional amplification in tumor cells with elevated c-Myc. Cell 2012, 151, 56–67. [Google Scholar] [CrossRef] [PubMed]
- Swerdlow, S.H.; Campo, E.; Pileri, S.A.; Harris, N.L.; Stein, H.; Siebert, R.; Advani, R.; Ghielmini, M.; Salles, G.A.; Zelenetz, A.D.; et al. The 2016 revision of the World Health Organization classification of lymphoid neoplasms. Blood 2016, 127, 2375–2390. [Google Scholar] [CrossRef] [PubMed]
- MacLennan, I.C. Germinal centers. Annu. Rev. Immunol. 1994, 12, 117–139. [Google Scholar] [CrossRef] [PubMed]
- Cattoretti, G.; Chang, C.C.; Cechova, K.; Zhang, J.; Ye, B.H.; Falini, B.; Louie, D.C.; Offit, K.; Chaganti, R.S.; Dalla-Favera, R. BCL-6 protein is expressed in germinal-center B cells. Blood 1995, 86, 45–53. [Google Scholar] [PubMed]
- Ye, B.H.; Cattoretti, G.; Shen, Q.; Zhang, J.; Hawe, N.; de Waard, R.; Leung, C.; Nouri-Shirazi, M.; Orazi, A.; Chaganti, R.S.; et al. The BCL-6 proto-oncogene controls germinal-centre formation and Th2-type inflammation. Nat. Genet. 1997, 16, 161–170. [Google Scholar] [CrossRef] [PubMed]
- Falini, B.; Fizzotti, M.; Pucciarini, A.; Bigerna, B.; Marafioti, T.; Gambacorta, M.; Pacini, R.; Alunni, C.; Natali-Tanci, L.; Ugolini, B.; et al. A monoclonal antibody (MUM1p) detects expression of the MUM1/IRF4 protein in a subset of germinal center B cells, plasma cells, and activated T cells. Blood 2000, 95, 2084–2092. [Google Scholar] [PubMed]
- Cattoretti, G.; Shaknovich, R.; Smith, P.M.; Jack, H.M.; Murty, V.V.; Alobeid, B. Stages of germinal center transit are defined by B cell transcription factor coexpression and relative abundance. J. Immunol. 2006, 177, 6930–6939. [Google Scholar] [CrossRef] [PubMed]
- Vallespinos, M.; Fernandez, D.; Rodriguez, L.; Alvaro-Blanco, J.; Baena, E.; Ortiz, M.; Dukovska, D.; Martinez, D.; Rojas, A.; Campanero, M.R.; et al. B Lymphocyte commitment program is driven by the proto-oncogene c-Myc. J. Immunol. 2011, 186, 6726–6736. [Google Scholar] [CrossRef] [PubMed]
- Klein, U.; Tu, Y.; Stolovitzky, G.A.; Keller, J.L.; Haddad, J., Jr.; Miljkovic, V.; Cattoretti, G.; Califano, A.; Dalla-Favera, R. Transcriptional analysis of the B cell germinal center reaction. Proc. Natl. Acad. Sci. USA 2003, 100, 2639–2644. [Google Scholar] [CrossRef] [PubMed]
- Calado, D.P.; Sasaki, Y.; Godinho, S.A.; Pellerin, A.; Kochert, K.; Sleckman, B.P.; de Alboran, I.M.; Janz, M.; Rodig, S.; Rajewsky, K. The cell-cycle regulator c-Myc is essential for the formation and maintenance of germinal centers. Nat. Immunol. 2012, 13, 1092–1100. [Google Scholar] [CrossRef] [PubMed]
- Dominguez-Sola, D.; Victora, G.D.; Ying, C.Y.; Phan, R.T.; Saito, M.; Nussenzweig, M.C.; Dalla-Favera, R. The proto-oncogene MYC is required for selection in the germinal center and cyclic reentry. Nat. Immunol. 2012, 13, 1083–1091. [Google Scholar] [CrossRef] [PubMed]
- Oestreich, K.J.; Mohn, S.E.; Weinmann, A.S. Molecular mechanisms that control the expression and activity of Bcl-6 in TH1 cells to regulate flexibility with a TFH-like gene profile. Nat. Immunol. 2012, 13, 405–411. [Google Scholar] [CrossRef] [PubMed]
- De Silva, N.S.; Klein, U. Dynamics of B cells in germinal centres. Nat. Rev. Immunol. 2015, 15, 137–148. [Google Scholar] [CrossRef] [PubMed]
- Basso, K.; Saito, M.; Sumazin, P.; Margolin, A.A.; Wang, K.; Lim, W.K.; Kitagawa, Y.; Schneider, C.; Alvarez, M.J.; Califano, A.; et al. Integrated biochemical and computational approach identifies BCL6 direct target genes controlling multiple pathways in normal germinal center B cells. Blood 2010, 115, 975–984. [Google Scholar] [CrossRef] [PubMed]
- Ci, W.; Polo, J.M.; Cerchietti, L.; Shaknovich, R.; Wang, L.; Yang, S.N.; Ye, K.; Farinha, P.; Horsman, D.E.; Gascoyne, R.D.; et al. The BCL6 transcriptional program features repression of multiple oncogenes in primary B cells and is deregulated in DLBCL. Blood 2009, 113, 5536–5548. [Google Scholar] [CrossRef] [PubMed]
- Nahar, R.; Ramezani-Rad, P.; Mossner, M.; Duy, C.; Cerchietti, L.; Geng, H.; Dovat, S.; Jumaa, H.; Ye, B.H.; Melnick, A.; et al. Pre-B cell receptor-mediated activation of BCL6 induces pre-B cell quiescence through transcriptional repression of MYC. Blood 2011, 118, 4174–4178. [Google Scholar] [CrossRef] [PubMed]
- Phan, R.T.; Saito, M.; Basso, K.; Niu, H.; Dalla-Favera, R. BCL6 interacts with the transcription factor Miz-1 to suppress the cyclin-dependent kinase inhibitor p21 and cell cycle arrest in germinal center B cells. Nat. Immunol. 2005, 6, 1054–1060. [Google Scholar] [CrossRef] [PubMed]
- Shaffer, A.L.; Yu, X.; He, Y.; Boldrick, J.; Chan, E.P.; Staudt, L.M. BCL-6 represses genes that function in lymphocyte differentiation, inflammation, and cell cycle control. Immunity 2000, 13, 199–212. [Google Scholar] [CrossRef]
- Oprea, M.; Perelson, A.S. Somatic mutation leads to efficient affinity maturation when centrocytes recycle back to centroblasts. J. Immunol. 1997, 158, 5155–5162. [Google Scholar] [PubMed]
- Rahl, P.B.; Lin, C.Y.; Seila, A.C.; Flynn, R.A.; McCuine, S.; Burge, C.B.; Sharp, P.A.; Young, R.A. c-Myc regulates transcriptional pause release. Cell 2010, 141, 432–445. [Google Scholar] [CrossRef] [PubMed]
- Perry, A.M.; Crockett, D.; Dave, B.J.; Althof, P.; Winkler, L.; Smith, L.M.; Aoun, P.; Chan, W.C.; Fu, K.; Greiner, T.C.; et al. B-cell lymphoma, unclassifiable, with features intermediate between diffuse large B-cell lymphoma and burkitt lymphoma: Study of 39 cases. Br. J. Haematol. 2013, 162, 40–49. [Google Scholar] [CrossRef] [PubMed]
- Haralambieva, E.; Schuuring, E.; Rosati, S.; van Noesel, C.; Jansen, P.; Appel, I.; Guikema, J.; Wabinga, H.; Bleggi-Torres, L.F.; Lam, K.; et al. Interphase fluorescence in situ hybridization for detection of 8q24/MYC breakpoints on routine histologic sections: Validation in Burkitt lymphomas from three geographic regions. Genes Chromosomes Cancer 2004, 40, 10–18. [Google Scholar] [CrossRef] [PubMed]
- Evan, G.I.; Lewis, G.K.; Ramsay, G.; Bishop, J.M. Isolation of monoclonal antibodies specific for human c-myc proto-oncogene product. Mol. Cell. Biol. 1985, 5, 3610–3616. [Google Scholar] [CrossRef] [PubMed]
- Cattoretti, G. MYC expression and distribution in normal mature lymphoid cells. J. Pathol. 2013, 229, 430–440. [Google Scholar] [CrossRef] [PubMed]
- Hu, S.; Xu-Monette, Z.Y.; Tzankov, A.; Green, T.; Wu, L.; Balasubramanyam, A.; Liu, W.M.; Visco, C.; Li, Y.; Miranda, R.N.; et al. MYC/BCL2 protein coexpression contributes to the inferior survival of activated B-cell subtype of diffuse large B-cell lymphoma and demonstrates high-risk gene expression signatures: A report from The International DLBCL Rituximab-CHOP Consortium Program. Blood 2013, 121, 4021–4031, quiz 4250. [Google Scholar] [CrossRef] [PubMed]
- Green, T.M.; Young, K.H.; Visco, C.; Xu-Monette, Z.Y.; Orazi, A.; Go, R.S.; Nielsen, O.; Gadeberg, O.V.; Mourits-Andersen, T.; Frederiksen, M.; et al. Immunohistochemical double-hit score is a strong predictor of outcome in patients with diffuse large B-cell lymphoma treated with rituximab plus cyclophosphamide, doxorubicin, vincristine, and prednisone. J. Clin. Oncol. 2012, 30, 3460–3467. [Google Scholar] [CrossRef] [PubMed]
- Johnson, N.A.; Slack, G.W.; Savage, K.J.; Connors, J.M.; Ben-Neriah, S.; Rogic, S.; Scott, D.W.; Tan, K.L.; Steidl, C.; Sehn, L.H.; et al. Concurrent expression of MYC and BCL2 in diffuse large B-cell lymphoma treated with rituximab plus cyclophosphamide, doxorubicin, vincristine, and prednisone. J. Clin. Oncol. 2012, 30, 3452–3459. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.J.; Medeiros, L.J.; Lin, P.; Yin, C.C.; Hu, S.; Thompson, M.A.; Li, S. MYC cytogenetic status correlates with expression and has prognostic significance in patients with MYC/BCL2 protein double-positive diffuse large B-cell lymphoma. Am. J. Surg. Pathol. 2015, 39, 1250–1258. [Google Scholar] [CrossRef] [PubMed]
- Chisholm, K.M.; Bangs, C.D.; Bacchi, C.E.; Molina-Kirsch, H.; Cherry, A.; Natkunam, Y. Expression profiles of MYC protein and MYC gene rearrangement in lymphomas. Am. J. Surg. Pathol. 2015, 39, 294–303. [Google Scholar] [CrossRef] [PubMed]
- Ruzinova, M.B.; Caron, T.; Rodig, S.J. Altered subcellular localization of c-Myc protein identifies aggressive B-cell lymphomas harboring a c-MYC translocation. Am. J. Surg. Pathol. 2010, 34, 882–891. [Google Scholar] [CrossRef] [PubMed]
- Tapia, G.; Lopez, R.; Munoz-Marmol, A.M.; Mate, J.L.; Sanz, C.; Marginet, R.; Navarro, J.T.; Ribera, J.M.; Ariza, A. Immunohistochemical detection of MYC protein correlates with MYC gene status in aggressive B cell lymphomas. Histopathology 2011, 59, 672–678. [Google Scholar] [CrossRef] [PubMed]
- Kluk, M.J.; Chapuy, B.; Sinha, P.; Roy, A.; Dal Cin, P.; Neuberg, D.S.; Monti, S.; Pinkus, G.S.; Shipp, M.A.; Rodig, S.J. Immunohistochemical detection of MYC-driven diffuse large B-cell lymphomas. PLoS ONE 2012, 7, e33813. [Google Scholar] [CrossRef] [PubMed]
- Green, T.M.; Nielsen, O.; de Stricker, K.; Xu-Monette, Z.Y.; Young, K.H.; Moller, M.B. High levels of nuclear MYC protein predict the presence of MYC rearrangement in diffuse large B-cell lymphoma. Am. J. Surg. Pathol. 2012, 36, 612–619. [Google Scholar] [CrossRef] [PubMed]
- Swerdlow, S.H.; Campo, E.; Harris, N.L.; Jaffe, E.S.; Pileri, S.A.; Stein, H.; Thiele, J.; Vardiman, J.W. WHO Classification of Tumors of Haematopoietic and Lymphoid Tissues, 4th ed.; IARC Press: Lyon, France, 2008. [Google Scholar]
- Seegmiller, A.C.; Garcia, R.; Huang, R.; Maleki, A.; Karandikar, N.J.; Chen, W. Simple karyotype and bcl-6 expression predict a diagnosis of Burkitt lymphoma and better survival in IG-MYC rearranged high-grade B-cell lymphomas. Mod. Pathol. 2010, 23, 909–920. [Google Scholar] [CrossRef] [PubMed]
- Cory, S. Activation of cellular oncogenes in hemopoietic cells by chromosome translocation. Adv. Cancer Res. 1986, 47, 189–234. [Google Scholar] [PubMed]
- Shiramizu, B.; Barriga, F.; Neequaye, J.; Jafri, A.; Dalla-Favera, R.; Neri, A.; Guttierez, M.; Levine, P.; Magrath, I. Patterns of chromosomal breakpoint locations in Burkitt’s lymphoma: Relevance to geography and Epstein-Barr virus association. Blood 1991, 77, 1516–1526. [Google Scholar] [PubMed]
- Magrath, I. The pathogenesis of Burkitt's lymphoma. Adv. Cancer Res. 1990, 55, 133–270. [Google Scholar] [PubMed]
- Pelicci, P.G.; Knowles, D.M., II; Magrath, I.; Dalla-Favera, R. Chromosomal breakpoints and structural alterations of the c-myc locus differ in endemic and sporadic forms of Burkitt lymphoma. Proc. Natl. Acad. Sci. USA 1986, 83, 2984–2988. [Google Scholar] [CrossRef] [PubMed]
- Polack, A.; Feederle, R.; Klobeck, G.; Hortnagel, K. Regulatory elements in the immunoglobulin kappa locus induce c-myc activation and the promoter shift in Burkitt’s lymphoma cells. EMBO J. 1993, 12, 3913–3920. [Google Scholar] [PubMed]
- Bemark, M.; Neuberger, M.S. The c-MYC allele that is translocated into the IgH locus undergoes constitutive hypermutation in a Burkitt’s lymphoma line. Oncogene 2000, 19, 3404–3410. [Google Scholar] [CrossRef] [PubMed]
- Cesarman, E.; Dalla-Favera, R.; Bentley, D.; Groudine, M. Mutations in the first exon are associated with altered transcription of c-myc in Burkitt lymphoma. Science 1987, 238, 1272–1275. [Google Scholar] [CrossRef] [PubMed]
- Rabbitts, T.H.; Forster, A.; Hamlyn, P.; Baer, R. Effect of somatic mutation within translocated c-myc genes in Burkitt’s lymphoma. Nature 1984, 309, 592–597. [Google Scholar] [CrossRef] [PubMed]
- Yu, B.W.; Ichinose, I.; Bonham, M.A.; Zajac-Kaye, M. Somatic mutations in c-myc intron I cluster in discrete domains that define protein binding sequences. J. Biol. Chem. 1993, 268, 19586–19592. [Google Scholar] [PubMed]
- Bahram, F.; von der Lehr, N.; Cetinkaya, C.; Larsson, L.G. c-Myc hot spot mutations in lymphomas result in inefficient ubiquitination and decreased proteasome-mediated turnover. Blood 2000, 95, 2104–2110. [Google Scholar] [PubMed]
- Gregory, M.A.; Hann, S.R. c-Myc proteolysis by the ubiquitin-proteasome pathway: Stabilization of c-Myc in Burkitt’s lymphoma cells. Mol. Cell. Biol. 2000, 20, 2423–2435. [Google Scholar] [CrossRef] [PubMed]
- Sears, R.; Leone, G.; DeGregori, J.; Nevins, J.R. Ras enhances Myc protein stability. Mol. Cell 1999, 3, 169–179. [Google Scholar] [CrossRef]
- Hummel, M.; Bentink, S.; Berger, H.; Klapper, W.; Wessendorf, S.; Barth, T.F.; Bernd, H.W.; Cogliatti, S.B.; Dierlamm, J.; Feller, A.C.; et al. A biologic definition of Burkitt’s lymphoma from transcriptional and genomic profiling. N. Engl. J. Med. 2006, 354, 2419–2430. [Google Scholar] [CrossRef] [PubMed]
- Dave, S.S.; Fu, K.; Wright, G.W.; Lam, L.T.; Kluin, P.; Boerma, E.J.; Greiner, T.C.; Weisenburger, D.D.; Rosenwald, A.; Ott, G.; et al. Molecular diagnosis of Burkitt’s lymphoma. N. Engl. J. Med. 2006, 354, 2431–2442. [Google Scholar] [CrossRef] [PubMed]
- Ferreiro, J.F.; Morscio, J.; Dierickx, D.; Marcelis, L.; Verhoef, G.; Vandenberghe, P.; Tousseyn, T.; Wlodarska, I. Post-transplant molecularly defined Burkitt lymphomas are frequently MYC-negative and characterized by the 11q-gain/loss pattern. Haematologica 2015, 100, e275–e279. [Google Scholar] [CrossRef] [PubMed]
- Salaverria, I.; Martin-Guerrero, I.; Wagener, R.; Kreuz, M.; Kohler, C.W.; Richter, J.; Pienkowska-Grela, B.; Adam, P.; Burkhardt, B.; Claviez, A.; et al. A recurrent 11q aberration pattern characterizes a subset of MYC-negative high-grade B-cell lymphomas resembling Burkitt lymphoma. Blood 2014, 123, 1187–1198. [Google Scholar] [CrossRef] [PubMed]
- Love, C.; Sun, Z.; Jima, D.; Li, G.; Zhang, J.; Miles, R.; Richards, K.L.; Dunphy, C.H.; Choi, W.W.; Srivastava, G.; et al. The genetic landscape of mutations in Burkitt lymphoma. Nat. Genet. 2012, 44, 1321–1325. [Google Scholar] [CrossRef] [PubMed]
- Richter, J.; Schlesner, M.; Hoffmann, S.; Kreuz, M.; Leich, E.; Burkhardt, B.; Rosolowski, M.; Ammerpohl, O.; Wagener, R.; Bernhart, S.H.; et al. Recurrent mutation of the ID3 gene in Burkitt lymphoma identified by integrated genome, exome and transcriptome sequencing. Nat. Genet. 2012, 44, 1316–1320. [Google Scholar] [CrossRef] [PubMed]
- Sander, S.; Calado, D.P.; Srinivasan, L.; Kochert, K.; Zhang, B.; Rosolowski, M.; Rodig, S.J.; Holzmann, K.; Stilgenbauer, S.; Siebert, R.; et al. Synergy between PI3K signaling and MYC in Burkitt lymphomagenesis. Cancer Cell 2012, 22, 167–179. [Google Scholar] [CrossRef] [PubMed]
- Schmitz, R.; Ceribelli, M.; Pittaluga, S.; Wright, G.; Staudt, L.M. Oncogenic mechanisms in Burkitt lymphoma. Cold Spring Harb. Perspect. Med. 2014, 4, a014282. [Google Scholar] [CrossRef] [PubMed]
- God, J.M.; Cameron, C.; Figueroa, J.; Amria, S.; Hossain, A.; Kempkes, B.; Bornkamm, G.W.; Stuart, R.K.; Blum, J.S.; Haque, A. Elevation of c-MYC disrupts HLA class II-mediated immune recognition of human B cell tumors. J. Immunol. 2015, 194, 1434–1445. [Google Scholar] [CrossRef] [PubMed]
- Alizadeh, A.A.; Eisen, M.B.; Davis, R.E.; Ma, C.; Lossos, I.S.; Rosenwald, A.; Boldrick, J.C.; Sabet, H.; Tran, T.; Yu, X.; et al. Distinct types of diffuse large B-cell lymphoma identified by gene expression profiling. Nature 2000, 403, 503–511. [Google Scholar] [CrossRef] [PubMed]
- Hans, C.P.; Weisenburger, D.D.; Greiner, T.C.; Gascoyne, R.D.; Delabie, J.; Ott, G.; Muller-Hermelink, H.K.; Campo, E.; Braziel, R.M.; Jaffe, E.S.; et al. Confirmation of the molecular classification of diffuse large B-cell lymphoma by immunohistochemistry using a tissue microarray. Blood 2004, 103, 275–282. [Google Scholar] [CrossRef] [PubMed]
- Chang, C.C.; McClintock, S.; Cleveland, R.P.; Trzpuc, T.; Vesole, D.H.; Logan, B.; Kajdacsy-Balla, A.; Perkins, S.L. Immunohistochemical expression patterns of germinal center and activation B-cell markers correlate with prognosis in diffuse large B-cell lymphoma. Am. J. Surg. Pathol. 2004, 28, 464–470. [Google Scholar] [CrossRef] [PubMed]
- Shipp, M.A.; Harrington, D.P.; Anderson, J.R.; Armitage, J.O.; Bonadonna, G.; Brittinger, G.; Cabanillas, F.; Canellos, G.P.; Coiffier, B.; Connors, J.M.; et al. A predictive model for aggressive non-Hodgkin’s lymphoma. The International Non-Hodgkin’s Lymphoma Prognostic Factors Project. N. Engl. J. Med. 1993, 329, 987–994. [Google Scholar]
- Sehn, L.H.; Berry, B.; Chhanabhai, M.; Fitzgerald, C.; Gill, K.; Hoskins, P.; Klasa, R.; Savage, K.J.; Shenkier, T.; Sutherland, J.; et al. The revised International Prognostic Index (R-IPI) is a better predictor of outcome than the standard IPI for patients with diffuse large B-cell lymphoma treated with R-CHOP. Blood 2007, 109, 1857–1861. [Google Scholar] [CrossRef] [PubMed]
- Gascoyne, R.D. Pathologic prognostic factors in diffuse aggressive non-Hodgkin’s lymphoma. Hematol. Oncol. Clin. N. Am. 1997, 11, 847–862. [Google Scholar] [CrossRef]
- Barrans, S.L.; Evans, P.A.; O’Connor, S.J.; Kendall, S.J.; Owen, R.G.; Haynes, A.P.; Morgan, G.J.; Jack, A.S. The t(14;18) is associated with germinal center-derived diffuse large B-cell lymphoma and is a strong predictor of outcome. Clin. Cancer Res. 2003, 9, 2133–2139. [Google Scholar] [PubMed]
- Cigudosa, J.C.; Parsa, N.Z.; Louie, D.C.; Filippa, D.A.; Jhanwar, S.C.; Johansson, B.; Mitelman, F.; Chaganti, R.S. Cytogenetic analysis of 363 consecutively ascertained diffuse large B-cell lymphomas. Genes Chromosomes Cancer 1999, 25, 123–133. [Google Scholar] [CrossRef]
- Lo Coco, F.; Ye, B.H.; Lista, F.; Corradini, P.; Offit, K.; Knowles, D.M.; Chaganti, R.S.; Dalla-Favera, R. Rearrangements of the BCL6 gene in diffuse large cell non-Hodgkin’s lymphoma. Blood 1994, 83, 1757–1759. [Google Scholar] [PubMed]
- Otsuki, T.; Yano, T.; Clark, H.M.; Bastard, C.; Kerckaert, J.P.; Jaffe, E.S.; Raffeld, M. Analysis of LAZ3 (BCL-6) status in B-cell non-Hodgkin’s lymphomas: Results of rearrangement and gene expression studies and a mutational analysis of coding region sequences. Blood 1995, 85, 2877–2884. [Google Scholar] [PubMed]
- Akasaka, T.; Akasaka, H.; Ueda, C.; Yonetani, N.; Maesako, Y.; Shimizu, A.; Yamabe, H.; Fukuhara, S.; Uchiyama, T.; Ohno, H. Molecular and clinical features of non-Burkitt’s, diffuse large-cell lymphoma of B-cell type associated with the c-MYC/immunoglobulin heavy-chain fusion gene. J. Clin. Oncol. 2000, 18, 510–518. [Google Scholar] [CrossRef] [PubMed]
- Ladanyi, M.; Offit, K.; Jhanwar, S.C.; Filippa, D.A.; Chaganti, R.S. MYC rearrangement and translocations involving band 8q24 in diffuse large cell lymphomas. Blood 1991, 77, 1057–1063. [Google Scholar] [PubMed]
- Kramer, M.H.; Hermans, J.; Wijburg, E.; Philippo, K.; Geelen, E.; van Krieken, J.H.; de Jong, D.; Maartense, E.; Schuuring, E.; Kluin, P.M. Clinical relevance of BCL2, BCL6, and MYC rearrangements in diffuse large B-cell lymphoma. Blood 1998, 92, 3152–3162. [Google Scholar] [PubMed]
- Vitolo, U.; Gaidano, G.; Botto, B.; Volpe, G.; Audisio, E.; Bertini, M.; Calvi, R.; Freilone, R.; Novero, D.; Orsucci, L.; et al. Rearrangements of bcl-6, bcl-2, c-myc and 6q deletion in B-diffuse large-cell lymphoma: Clinical relevance in 71 patients. Ann. Oncol. 1998, 9, 55–61. [Google Scholar] [CrossRef] [PubMed]
- Kawasaki, C.; Ohshim, K.; Suzumiya, J.; Kanda, M.; Tsuchiya, T.; Tamura, K.; Kikuchi, M. Rearrangements of bcl-1, bcl-2, bcl-6, and c-myc in diffuse large B-cell lymphomas. Leuk Lymphoma 2001, 42, 1099–1106. [Google Scholar] [CrossRef] [PubMed]
- Karube, K.; Campo, E. MYC alterations in diffuse large B-cell lymphomas. Semin. Hematol. 2015, 52, 97–106. [Google Scholar] [CrossRef] [PubMed]
- Savage, K.J.; Johnson, N.A.; Ben-Neriah, S.; Connors, J.M.; Sehn, L.H.; Farinha, P.; Horsman, D.E.; Gascoyne, R.D. MYC gene rearrangements are associated with a poor prognosis in diffuse large B-cell lymphoma patients treated with R-CHOP chemotherapy. Blood 2009, 114, 3533–3537. [Google Scholar] [CrossRef] [PubMed]
- Valera, A.; Lopez-Guillermo, A.; Cardesa-Salzmann, T.; Climent, F.; Gonzalez-Barca, E.; Mercadal, S.; Espinosa, I.; Novelli, S.; Briones, J.; Mate, J.L.; et al. MYC protein expression and genetic alterations have prognostic impact in patients with diffuse large B-cell lymphoma treated with immunochemotherapy. Haematologica 2013, 98, 1554–1562. [Google Scholar] [CrossRef] [PubMed]
- Boerma, E.G.; Siebert, R.; Kluin, P.M.; Baudis, M. Translocations involving 8q24 in Burkitt lymphoma and other malignant lymphomas: A historical review of cytogenetics in the light of todays knowledge. Leukemia 2009, 23, 225–234. [Google Scholar] [CrossRef] [PubMed]
- Smith, S.M.; Anastasi, J.; Cohen, K.S.; Godley, L.A. The impact of MYC expression in lymphoma biology: Beyond Burkitt lymphoma. Blood Cells Mol. Dis. 2010, 45, 317–323. [Google Scholar] [CrossRef] [PubMed]
- Johnson, N.A.; Savage, K.J.; Ludkovski, O.; Ben-Neriah, S.; Woods, R.; Steidl, C.; Dyer, M.J.; Siebert, R.; Kuruvilla, J.; Klasa, R.; et al. Lymphomas with concurrent BCL2 and MYC translocations: The critical factors associated with survival. Blood 2009, 114, 2273–2279. [Google Scholar] [CrossRef] [PubMed]
- Pedersen, M.O.; Gang, A.O.; Poulsen, T.S.; Knudsen, H.; Lauritzen, A.F.; Nielsen, S.L.; Gang, U.O.; Norgaard, P. Double-hit BCL2/MYC translocations in a consecutive cohort of patients with large B-cell lymphoma - a single centre’s experience. Eur. J. Haematol. 2012, 89, 63–71. [Google Scholar] [CrossRef] [PubMed]
- Pillai, R.K.; Sathanoori, M.; Van Oss, S.B.; Swerdlow, S.H. Double-hit B-cell lymphomas with BCL6 and MYC translocations are aggressive, frequently extranodal lymphomas distinct from BCL2 double-hit B-cell lymphomas. Am. J. Surg. Pathol. 2013, 37, 323–332. [Google Scholar] [CrossRef] [PubMed]
- Petrich, A.M.; Gandhi, M.; Jovanovic, B.; Castillo, J.J.; Rajguru, S.; Yang, D.T.; Shah, K.A.; Whyman, J.D.; Lansigan, F.; Hernandez-Ilizaliturri, F.J.; et al. Impact of induction regimen and stem cell transplantation on outcomes in double-hit lymphoma: A multicenter retrospective analysis. Blood 2014, 124, 2354–2361. [Google Scholar] [CrossRef] [PubMed]
- Oki, Y.; Noorani, M.; Lin, P.; Davis, R.E.; Neelapu, S.S.; Ma, L.; Ahmed, M.; Rodriguez, M.A.; Hagemeister, F.B.; Fowler, N.; et al. Double hit lymphoma: The MD Anderson Cancer Center clinical experience. Br. J. Haematol. 2014, 166, 891–901. [Google Scholar] [CrossRef] [PubMed]
- Snuderl, M.; Kolman, O.K.; Chen, Y.B.; Hsu, J.J.; Ackerman, A.M.; Dal Cin, P.; Ferry, J.A.; Harris, N.L.; Hasserjian, R.P.; Zukerberg, L.R.; et al. B-cell lymphomas with concurrent IGH-BCL2 and MYC rearrangements are aggressive neoplasms with clinical and pathologic features distinct from Burkitt lymphoma and diffuse large B-cell lymphoma. Am. J. Surg. Pathol. 2010, 34, 327–340. [Google Scholar] [CrossRef] [PubMed]
- Aukema, S.M.; Siebert, R.; Schuuring, E.; van Imhoff, G.W.; Kluin-Nelemans, H.C.; Boerma, E.J.; Kluin, P.M. Double-hit B-cell lymphomas. Blood 2011, 117, 2319–2331. [Google Scholar] [CrossRef] [PubMed]
- Barrans, S.; Crouch, S.; Smith, A.; Turner, K.; Owen, R.; Patmore, R.; Roman, E.; Jack, A. Rearrangement of MYC is associated with poor prognosis in patients with diffuse large B-cell lymphoma treated in the era of rituximab. J. Clin. Oncol. 2010, 28, 3360–3365. [Google Scholar] [CrossRef] [PubMed]
- Le Gouill, S.; Talmant, P.; Touzeau, C.; Moreau, A.; Garand, R.; Juge-Morineau, N.; Gaillard, F.; Gastinne, T.; Milpied, N.; Moreau, P.; et al. The clinical presentation and prognosis of diffuse large B-cell lymphoma with t(14;18) and 8q24/c-MYC rearrangement. Haematologica 2007, 92, 1335–1342. [Google Scholar] [CrossRef] [PubMed]
- Landsburg, D.J.; Nasta, S.D.; Svoboda, J.; Morrissette, J.J.; Schuster, S.J. ‘Double-Hit’ cytogenetic status may not be predicted by baseline clinicopathological characteristics and is highly associated with overall survival in B cell lymphoma patients. Br. J. Haematol. 2014, 166, 369–374. [Google Scholar] [CrossRef] [PubMed]
- Momose, S.; Weissbach, S.; Pischimarov, J.; Nedeva, T.; Bach, E.; Rudelius, M.; Geissinger, E.; Staiger, A.M.; Ott, G.; Rosenwald, A. The diagnostic gray zone between Burkitt lymphoma and diffuse large B-cell lymphoma is also a gray zone of the mutational spectrum. Leukemia 2015, 29, 1789–1791. [Google Scholar] [CrossRef] [PubMed]
- Gebauer, N.; Bernard, V.; Gebauer, W.; Thorns, C.; Feller, A.C.; Merz, H. TP53 mutations are frequent events in double-hit B-cell lymphomas with MYC and BCL2 but not MYC and BCL6 translocations. Leuk Lymphoma 2015, 56, 179–185. [Google Scholar] [CrossRef] [PubMed]
- Ramiro, A.R.; Jankovic, M.; Eisenreich, T.; Difilippantonio, S.; Chen-Kiang, S.; Muramatsu, M.; Honjo, T.; Nussenzweig, A.; Nussenzweig, M.C. AID is required for c-myc/IgH chromosome translocations in vivo. Cell 2004, 118, 431–438. [Google Scholar] [CrossRef] [PubMed]
- De Jong, D.; Voetdijk, B.M.; Beverstock, G.C.; van Ommen, G.J.; Willemze, R.; Kluin, P.M. Activation of the c-myc oncogene in a precursor-B-cell blast crisis of follicular lymphoma, presenting as composite lymphoma. N. Engl. J. Med. 1988, 318, 1373–1378. [Google Scholar] [CrossRef] [PubMed]
- Hoffman, B.; Liebermann, D.A. Apoptotic signaling by c-MYC. Oncogene 2008, 27, 6462–6472. [Google Scholar] [CrossRef] [PubMed]
- Fanidi, A.; Harrington, E.A.; Evan, G.I. Cooperative interaction between c-myc and bcl-2 proto-oncogenes. Nature 1992, 359, 554–556. [Google Scholar] [CrossRef] [PubMed]
- Gaidano, G.; Cerri, M.; Capello, D.; Berra, E.; Deambrogi, C.; Rossi, D.; Larocca, L.M.; Campo, E.; Gloghini, A.; Tirelli, U.; et al. Molecular histogenesis of plasmablastic lymphoma of the oral cavity. Br. J. Haematol. 2002, 119, 622–628. [Google Scholar] [CrossRef] [PubMed]
- Shapiro-Shelef, M.; Lin, K.I.; McHeyzer-Williams, L.J.; Liao, J.; McHeyzer-Williams, M.G.; Calame, K. Blimp-1 is required for the formation of immunoglobulin secreting plasma cells and pre-plasma memory B cells. Immunity 2003, 19, 607–620. [Google Scholar] [CrossRef]
- Lin, Y.; Wong, K.; Calame, K. Repression of c-myc transcription by Blimp-1, an inducer of terminal B cell differentiation. Science 1997, 276, 596–599. [Google Scholar] [CrossRef] [PubMed]
- Valera, A.; Balague, O.; Colomo, L.; Martinez, A.; Delabie, J.; Taddesse-Heath, L.; Jaffe, E.S.; Campo, E. IG/MYC rearrangements are the main cytogenetic alteration in plasmablastic lymphomas. Am. J. Surg. Pathol. 2010, 34, 1686–1694. [Google Scholar] [CrossRef] [PubMed]
- Montes-Moreno, S.; Martinez-Magunacelaya, N.; Zecchini-Barrese, T.; Villambrosia, S.G.; Linares, E.; Ranchal, T.; Rodriguez-Pinilla, M.; Batlle, A.; Cereceda-Company, L.; Revert-Arce, J.B.; et al. Plasmablastic lymphoma phenotype is determined by genetic alterations in MYC and PRDM1. Mod. Pathol. 2017, 30, 85–94. [Google Scholar] [CrossRef] [PubMed]
- Chiecchio, L.; Dagrada, G.P.; White, H.E.; Towsend, M.R.; Protheroe, R.K.; Cheung, K.L.; Stockley, D.M.; Orchard, K.H.; Cross, N.C.; Harrison, C.J.; et al. Frequent upregulation of MYC in plasma cell leukemia. Genes Chromosomes Cancer 2009, 48, 624–636. [Google Scholar] [CrossRef] [PubMed]
- Shou, Y.; Martelli, M.L.; Gabrea, A.; Qi, Y.; Brents, L.A.; Roschke, A.; Dewald, G.; Kirsch, I.R.; Bergsagel, P.L.; Kuehl, W.M. Diverse karyotypic abnormalities of the c-myc locus associated with c-myc dysregulation and tumor progression in multiple myeloma. Proc. Natl. Acad. Sci. USA 2000, 97, 228–233. [Google Scholar] [CrossRef] [PubMed]
- Taddesse-Heath, L.; Meloni-Ehrig, A.; Scheerle, J.; Kelly, J.C.; Jaffe, E.S. Plasmablastic lymphoma with MYC translocation: Evidence for a common pathway in the generation of plasmablastic features. Mod. Pathol. 2010, 23, 991–999. [Google Scholar] [CrossRef] [PubMed]
- Pileri, S.A.; Falini, B. Mantle cell lymphoma. Haematologica 2009, 94, 1488–1492. [Google Scholar] [CrossRef] [PubMed]
- Furtado, M.; Rule, S. Indolent mantle cell lymphoma. Haematologica 2011, 96, 1086–1088. [Google Scholar] [CrossRef] [PubMed]
- Ondrejka, S.L.; Lai, R.; Smith, S.D.; Hsi, E.D. Indolent mantle cell leukemia: A clinicopathological variant characterized by isolated lymphocytosis, interstitial bone marrow involvement, kappa light chain restriction, and good prognosis. Haematologica 2011, 96, 1121–1127. [Google Scholar] [CrossRef] [PubMed]
- Sander, B.; Quintanilla-Martinez, L.; Ott, G.; Xerri, L.; Kuzu, I.; Chan, J.K.; Swerdlow, S.H.; Campo, E. Mantle cell lymphoma--a spectrum from indolent to aggressive disease. Virchows Arch. 2016, 468, 245–257. [Google Scholar] [CrossRef] [PubMed]
- Royo, C.; Salaverria, I.; Hartmann, E.M.; Rosenwald, A.; Campo, E.; Bea, S. The complex landscape of genetic alterations in mantle cell lymphoma. Semin. Cancer Biol. 2011, 21, 322–334. [Google Scholar] [CrossRef] [PubMed]
- Salaverria, I.; Zettl, A.; Bea, S.; Moreno, V.; Valls, J.; Hartmann, E.; Ott, G.; Wright, G.; Lopez-Guillermo, A.; Chan, W.C.; et al. Specific secondary genetic alterations in mantle cell lymphoma provide prognostic information independent of the gene expression-based proliferation signature. J. Clin. Oncol. 2007, 25, 1216–1222. [Google Scholar] [CrossRef] [PubMed]
- Lovec, H.; Grzeschiczek, A.; Kowalski, M.B.; Moroy, T. Cyclin D1/bcl-1 cooperates with myc genes in the generation of B-cell lymphoma in transgenic mice. EMBO J. 1994, 13, 3487–3495. [Google Scholar] [PubMed]
- Saba, N.S.; Liu, D.; Herman, S.E.; Underbayev, C.; Tian, X.; Behrend, D.; Weniger, M.A.; Skarzynski, M.; Gyamfi, J.; Fontan, L.; et al. Pathogenic role of B-cell receptor signaling and canonical NF-kappaB activation in mantle cell lymphoma. Blood 2016, 128, 82–92. [Google Scholar] [CrossRef] [PubMed]
- Gaide, O.; Favier, B.; Legler, D.F.; Bonnet, D.; Brissoni, B.; Valitutti, S.; Bron, C.; Tschopp, J.; Thome, M. CARMA1 is a critical lipid raft-associated regulator of TCR-induced NF-kappa B activation. Nat. Immunol. 2002, 3, 836–843. [Google Scholar] [CrossRef] [PubMed]
- Che, T.; You, Y.; Wang, D.; Tanner, M.J.; Dixit, V.M.; Lin, X. MALT1/paracaspase is a signaling component downstream of CARMA1 and mediates T cell receptor-induced NF-kappaB activation. J. Biol. Chem. 2004, 279, 15870–15876. [Google Scholar] [CrossRef] [PubMed]
- Dai, B.; Grau, M.; Juilland, M.; Klener, P.; Horing, E.; Molinsky, J.; Schimmack, G.; Aukema, S.M.; Hoster, E.; Vogt, N.; et al. B-cell receptor driven MALT1 activity regulates MYC signaling in mantle cell lymphoma. Blood 2016. [Google Scholar] [CrossRef] [PubMed]
- Hao, S.; Sanger, W.; Onciu, M.; Lai, R.; Schlette, E.J.; Medeiros, L.J. Mantle cell lymphoma with 8q24 chromosomal abnormalities: A report of 5 cases with blastoid features. Mod. Pathol. 2002, 15, 1266–1272. [Google Scholar] [CrossRef] [PubMed]
- Felten, C.L.; Stephenson, C.F.; Ortiz, R.O.; Hertzberg, L. Burkitt transformation of mantle cell lymphoma. Leuk Lymphoma 2004, 45, 2143–2147. [Google Scholar] [CrossRef] [PubMed]
- Vaishampayan, U.N.; Mohamed, A.N.; Dugan, M.C.; Bloom, R.E.; Palutke, M. Blastic mantle cell lymphoma associated with Burkitt-type translocation and hypodiploidy. Br. J. Haematol. 2001, 115, 66–68. [Google Scholar] [CrossRef] [PubMed]
- Reddy, K.; Ansari-Lari, M.; Dipasquale, B. Blastic mantle cell lymphoma with a Burkitt translocation. Leuk Lymphoma 2008, 49, 740–750. [Google Scholar] [CrossRef] [PubMed]
- Au, W.Y.; Horsman, D.E.; Viswanatha, D.S.; Connors, J.M.; Klasa, R.J.; Gascoyne, R.D. 8q24 translocations in blastic transformation of mantle cell lymphoma. Haematologica 2000, 85, 1225–1227. [Google Scholar] [PubMed]
- Michaux, L.; Wlodarska, I.; Theate, I.; Stul, M.; Scheiff, J.M.; Deneys, V.; Ferrant, A.; Hagemeijer, A. Coexistence of BCL1/CCND1 and CMYC aberrations in blastoid mantle cell lymphoma: A rare finding associated with very poor outcome. Ann. Hematol. 2004, 83, 578–583. [Google Scholar] [CrossRef] [PubMed]
- Tirier, C.; Zhang, Y.; Plendl, H.; Weber-Matthiesen, K.; Langer, W.; Heit, W.; Schlegelberger, B. Simultaneous presence of t(11;14) and a variant Burkitt’s translocation in the terminal phase of a mantle cell lymphoma. Leukemia 1996, 10, 346–350. [Google Scholar] [PubMed]
- Oliveira, F.M.; Tone, L.G.; Simoes, B.P.; Rego, E.M.; Araujo, A.G.; Falcao, R.P. Blastoid mantle cell lymphoma with t(2;8) (p12;q24). Leuk Lymphoma 2007, 48, 2079–2082. [Google Scholar] [CrossRef] [PubMed]
- Hu, Z.; Medeiros, L.J.; Chen, Z.; Chen, W.; Li, S.; Konoplev, S.N.; Lu, X.; Pham, L.V.; Young, K.H.; Wang, W.; et al. Mantle Cell Lymphoma With MYC Rearrangement: A Report of 17 Patients. Am. J. Surg. Pathol. 2016, 41, 216–224. [Google Scholar] [CrossRef] [PubMed]
- Setoodeh, R.; Schwartz, S.; Papenhausen, P.; Zhang, L.; Sagatys, E.M.; Moscinski, L.C.; Shao, H. Double-hit mantle cell lymphoma with MYC gene rearrangement or amplification: A report of four cases and review of the literature. Int. J. Clin. Exp. Pathol. 2013, 6, 155–167. [Google Scholar] [PubMed]
- Vincent-Fabert, C.; Fiancette, R.; Rouaud, P.; Baudet, C.; Truffinet, V.; Magnone, V.; Guillaudeau, A.; Cogne, M.; Dubus, P.; Denizot, Y. A defect of the INK4-Cdk4 checkpoint and Myc collaborate in blastoid mantle cell lymphoma-like lymphoma formation in mice. Am. J. Pathol. 2012, 180, 1688–1701. [Google Scholar] [CrossRef] [PubMed]
- Tagawa, H.; Karnan, S.; Suzuki, R.; Matsuo, K.; Zhang, X.; Ota, A.; Morishima, Y.; Nakamura, S.; Seto, M. Genome-wide array-based CGH for mantle cell lymphoma: Identification of homozygous deletions of the proapoptotic gene BIM. Oncogene 2005, 24, 1348–1358. [Google Scholar] [CrossRef] [PubMed]
- Rubio-Moscardo, F.; Climent, J.; Siebert, R.; Piris, M.A.; Martin-Subero, J.I.; Nielander, I.; Garcia-Conde, J.; Dyer, M.J.; Terol, M.J.; Pinkel, D.; et al. Mantle-cell lymphoma genotypes identified with CGH to BAC microarrays define a leukemic subgroup of disease and predict patient outcome. Blood 2005, 105, 4445–4454. [Google Scholar] [CrossRef] [PubMed]
- Bea, S.; Ribas, M.; Hernandez, J.M.; Bosch, F.; Pinyol, M.; Hernandez, L.; Garcia, J.L.; Flores, T.; Gonzalez, M.; Lopez-Guillermo, A.; et al. Increased number of chromosomal imbalances and high-level DNA amplifications in mantle cell lymphoma are associated with blastoid variants. Blood 1999, 93, 4365–4374. [Google Scholar] [PubMed]
- Huang, W.; Guo, L.; Liu, H.; Zheng, B.; Ying, J.; Lv, N. C-MYC overexpression predicts aggressive transformation and a poor outcome in mucosa-associated lymphoid tissue lymphomas. Int. J. Clin. Exp. Pathol. 2014, 7, 5634–5644. [Google Scholar] [PubMed]
- Craig, V.J.; Cogliatti, S.B.; Imig, J.; Renner, C.; Neuenschwander, S.; Rehrauer, H.; Schlapbach, R.; Dirnhofer, S.; Tzankov, A.; Muller, A. Myc-mediated repression of microRNA-34a promotes high-grade transformation of B-cell lymphoma by dysregulation of FoxP1. Blood 2011, 117, 6227–6236. [Google Scholar] [CrossRef] [PubMed]
- Stevenson, F.K.; Krysov, S.; Davies, A.J.; Steele, A.J.; Packham, G. B-cell receptor signaling in chronic lymphocytic leukemia. Blood 2011, 118, 4313–4320. [Google Scholar] [CrossRef] [PubMed]
- Gibson, S.E.; Leeman-Neill, R.J.; Jain, S.; Piao, W.; Cieply, K.M.; Swerdlow, S.H. Proliferation centres of chronic lymphocytic leukaemia/small lymphocytic lymphoma have enhanced expression of MYC protein, which does not result from rearrangement or gain of the MYC gene. Br. J. Haematol. 2016, 175, 173–175. [Google Scholar] [CrossRef] [PubMed]
- Krysov, S.; Dias, S.; Paterson, A.; Mockridge, C.I.; Potter, K.N.; Smith, K.A.; Ashton-Key, M.; Stevenson, F.K.; Packham, G. Surface IgM stimulation induces MEK1/2-dependent MYC expression in chronic lymphocytic leukemia cells. Blood 2012, 119, 170–179. [Google Scholar] [CrossRef] [PubMed]
- Yeomans, A.; Thirdborough, S.M.; Valle-Argos, B.; Linley, A.; Krysov, S.; Hidalgo, M.S.; Leonard, E.; Ishfaq, M.; Wagner, S.D.; Willis, A.E.; et al. Engagement of the B-cell receptor of chronic lymphocytic leukemia cells drives global and MYC-specific mRNA translation. Blood 2016, 127, 449–457. [Google Scholar] [CrossRef] [PubMed]
- Nelson, B.P.; Gupta, R.; Dewald, G.W.; Paternoster, S.F.; Rosen, S.T.; Peterson, L.C. Chronic lymphocytic leukemia FISH panel: Impact on diagnosis. Am. J. Clin. Pathol. 2007, 128, 323–332. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Hu, S.; Wang, S.A.; Li, S.; Huh, Y.O.; Tang, Z.; Medeiros, L.J.; Tang, G. The clinical significance of 8q24/MYC rearrangement in chronic lymphocytic leukemia. Mod. Pathol. 2016, 29, 444–451. [Google Scholar] [CrossRef] [PubMed]
- Huh, Y.O.; Lin, K.I.; Vega, F.; Schlette, E.; Yin, C.C.; Keating, M.J.; Luthra, R.; Medeiros, L.J.; Abruzzo, L.V. MYC translocation in chronic lymphocytic leukaemia is associated with increased prolymphocytes and a poor prognosis. Br. J. Haematol. 2008, 142, 36–44. [Google Scholar] [CrossRef] [PubMed]
- Put, N.; Van Roosbroeck, K.; Konings, P.; Meeus, P.; Brusselmans, C.; Rack, K.; Gervais, C.; Nguyen-Khac, F.; Chapiro, E.; Radford-Weiss, I.; et al. Chronic lymphocytic leukemia and prolymphocytic leukemia with MYC translocations: A subgroup with an aggressive disease course. Ann. Hematol. 2012, 91, 863–873. [Google Scholar] [CrossRef] [PubMed]
- Haberl, S.; Haferlach, T.; Stengel, A.; Jeromin, S.; Kern, W.; Haferlach, C. MYC rearranged B-cell neoplasms: Impact of genetics on classification. Cancer Genet. 2016, 209, 431–439. [Google Scholar] [CrossRef] [PubMed]
- Young, K.H.; Xie, Q.; Zhou, G.; Eickhoff, J.C.; Sanger, W.G.; Aoun, P.; Chan, W.C. Transformation of follicular lymphoma to precursor B-cell lymphoblastic lymphoma with c-myc gene rearrangement as a critical event. Am. J. Clin. Pathol. 2008, 129, 157–166. [Google Scholar] [CrossRef] [PubMed]
- Ouansafi, I.; He, B.; Fraser, C.; Nie, K.; Mathew, S.; Bhanji, R.; Hoda, R.; Arabadjief, M.; Knowles, D.; Cerutti, A.; et al. Transformation of follicular lymphoma to plasmablastic lymphoma with c-myc gene rearrangement. Am. J. Clin. Pathol. 2010, 134, 972–981. [Google Scholar] [CrossRef] [PubMed]
- Yano, T.; Jaffe, E.S.; Longo, D.L.; Raffeld, M. MYC rearrangements in histologically progressed follicular lymphomas. Blood 1992, 80, 758–767. [Google Scholar] [PubMed]
- Martinez-Climent, J.A.; Alizadeh, A.A.; Segraves, R.; Blesa, D.; Rubio-Moscardo, F.; Albertson, D.G.; Garcia-Conde, J.; Dyer, M.J.; Levy, R.; Pinkel, D.; et al. Transformation of follicular lymphoma to diffuse large cell lymphoma is associated with a heterogeneous set of DNA copy number and gene expression alterations. Blood 2003, 101, 3109–3117. [Google Scholar] [CrossRef] [PubMed]
- Lossos, I.S.; Alizadeh, A.A.; Diehn, M.; Warnke, R.; Thorstenson, Y.; Oefner, P.J.; Brown, P.O.; Botstein, D.; Levy, R. Transformation of follicular lymphoma to diffuse large-cell lymphoma: Alternative patterns with increased or decreased expression of c-myc and its regulated genes. Proc. Natl. Acad. Sci. USA 2002, 99, 8886–8891. [Google Scholar] [CrossRef] [PubMed]
- Davies, A.J.; Rosenwald, A.; Wright, G.; Lee, A.; Last, K.W.; Weisenburger, D.D.; Chan, W.C.; Delabie, J.; Braziel, R.M.; Campo, E.; et al. Transformation of follicular lymphoma to diffuse large B-cell lymphoma proceeds by distinct oncogenic mechanisms. Br. J. Haematol. 2007, 136, 286–293. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Lin, P.; Fayad, L.E.; Lennon, P.A.; Miranda, R.N.; Yin, C.C.; Lin, E.; Medeiros, L.J. B-cell lymphomas with MYC/8q24 rearrangements and IGH BCL2/t(14;18)(q32;q21): An aggressive disease with heterogeneous histology, germinal center B-cell immunophenotype and poor outcome. Mod. Pathol. 2012, 25, 145–156. [Google Scholar] [CrossRef] [PubMed]
- Christie, L.; Kernohan, N.; Levison, D.; Sales, M.; Cunningham, J.; Gillespie, K.; Batstone, P.; Meiklejohn, D.; Goodlad, J. C-MYC translocation in t(14;18) positive follicular lymphoma at presentation: An adverse prognostic indicator? Leuk Lymphoma 2008, 49, 470–476. [Google Scholar] [CrossRef] [PubMed]
- Kanda-Akano, Y.; Nomura, K.; Fujita, Y.; Horiike, S.; Nishida, K.; Nagai, M.; Miura, I.; Nakamura, S.; Seto, M.; Iida, S.; et al. Molecular-cytogenetic characterization of non-Hodgkin’s lymphoma with double and cryptic translocations of the immunoglobulin heavy chain gene. Leuk Lymphoma 2004, 45, 1559–1567. [Google Scholar] [CrossRef] [PubMed]
- Voorhees, P.M.; Carder, K.A.; Smith, S.V.; Ayscue, L.H.; Rao, K.W.; Dunphy, C.H. Follicular lymphoma with a burkitt translocation—Predictor of an aggressive clinical course: A case report and review of the literature. Arch. Pathol. Lab. Med. 2004, 128, 210–213. [Google Scholar] [PubMed]
- Kanungo, A.; Medeiros, L.J.; Abruzzo, L.V.; Lin, P. Lymphoid neoplasms associated with concurrent t(14;18) and 8q24/c-MYC translocation generally have a poor prognosis. Mod. Pathol. 2006, 19, 25–33. [Google Scholar] [CrossRef] [PubMed]
- Miao, Y.; Hu, S.; Lu, X.; Li, S.; Wang, W.; Medeiros, L.J.; Lin, P. Double-hit follicular lymphoma with MYC and BCL2 translocations: A study of 7 cases with a review of literature. Hum. Pathol. 2016, 58, 72–77. [Google Scholar] [CrossRef] [PubMed]
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nguyen, L.; Papenhausen, P.; Shao, H. The Role of c-MYC in B-Cell Lymphomas: Diagnostic and Molecular Aspects. Genes 2017, 8, 116. https://doi.org/10.3390/genes8040116
Nguyen L, Papenhausen P, Shao H. The Role of c-MYC in B-Cell Lymphomas: Diagnostic and Molecular Aspects. Genes. 2017; 8(4):116. https://doi.org/10.3390/genes8040116
Chicago/Turabian StyleNguyen, Lynh, Peter Papenhausen, and Haipeng Shao. 2017. "The Role of c-MYC in B-Cell Lymphomas: Diagnostic and Molecular Aspects" Genes 8, no. 4: 116. https://doi.org/10.3390/genes8040116
APA StyleNguyen, L., Papenhausen, P., & Shao, H. (2017). The Role of c-MYC in B-Cell Lymphomas: Diagnostic and Molecular Aspects. Genes, 8(4), 116. https://doi.org/10.3390/genes8040116





