Roles of CDK and DDK in Genome Duplication and Maintenance: Meiotic Singularities
Abstract
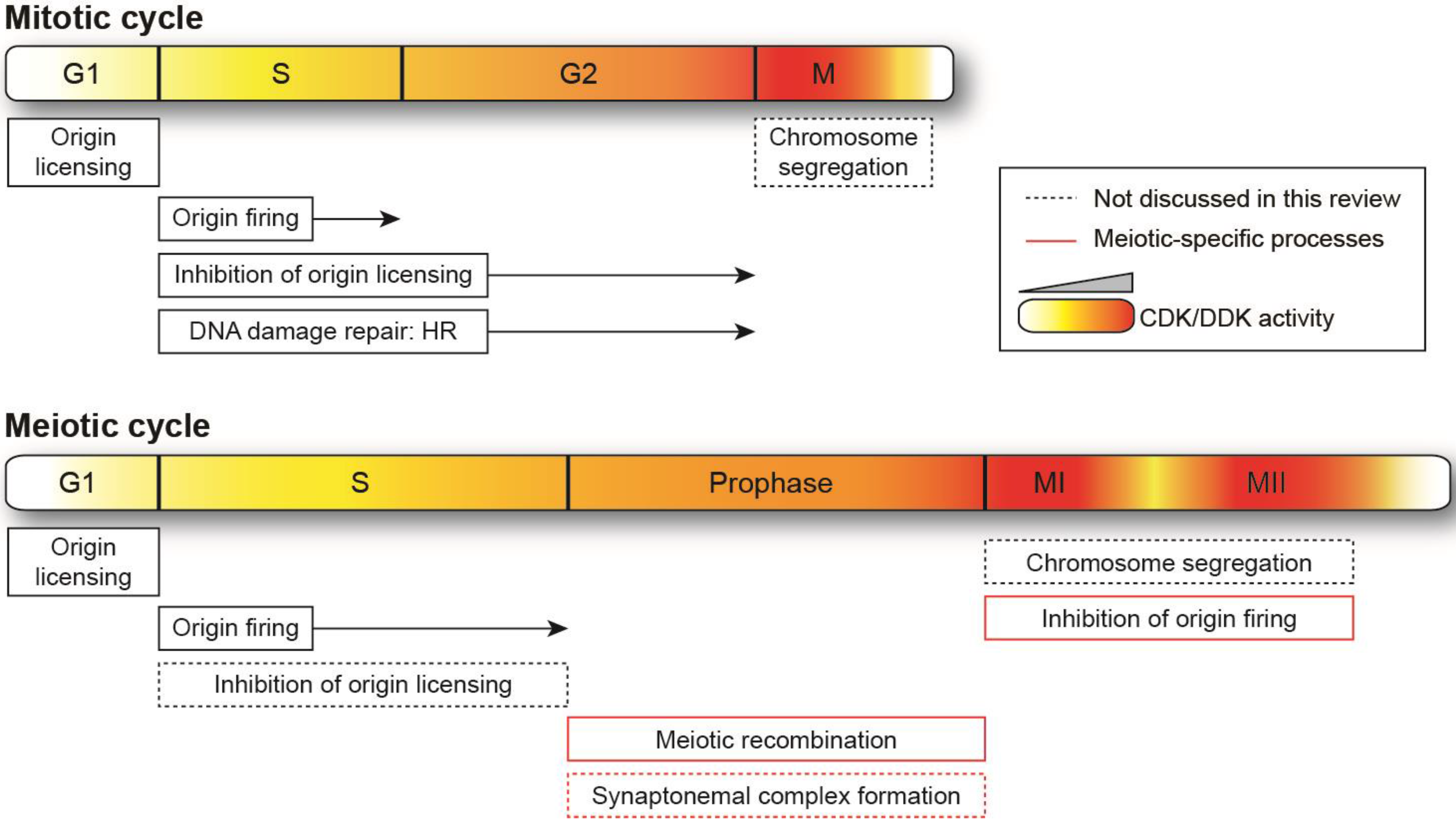
:1. Introduction
2. Regulation of the Mitotic Cycle by Quantitative Changes in Cyclin-Dependent Kinase and Dbf4-Dependent Kinase Activities
3. A Quantitative Model for Cyclin-Dependent Kinase and Dbf4-Dependent Kinase Activities in Meiosis
4. Further Specificities of Cyclin-Dependent Kinase and Dbf4-Dependent Kinase Regulation in Meiotic Cycles
5. Genome Duplication in Mitosis and Meiosis
6. Coordination between Pre-Meiotic Replication and DNA Double-Strand Break Formation
7. Repair of DNA Double-Strand Breaks in Mitotic and Meiotic Cycles
8. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Kleckner, N. Meiosis: How could it work? Proc. Natl. Acad. Sci. USA 1996, 93, 8167–8174. [Google Scholar] [CrossRef] [PubMed]
- Lam, I.; Keeney, S. Mechanism and regulation of meiotic recombination initiation. Cold Spring Harb. Perspect. Biol. 2014, 7, a016634. [Google Scholar] [CrossRef] [PubMed]
- Labib, K. How do Cdc7 and cyclin-dependent kinases trigger the initiation of chromosome replication in eukaryotic cells? Genes Dev. 2010, 24, 1208–1219. [Google Scholar] [CrossRef] [PubMed]
- Zegerman, P. Evolutionary conservation of the CDK targets in eukaryotic DNA replication initiation. Chromosoma 2015, 124, 309–321. [Google Scholar] [CrossRef] [PubMed]
- Morgan, D.O. Cyclin-dependent kinases: Engines, clocks, and microprocessors. Annu. Rev. Cell Dev. Biol. 1997, 13, 261–291. [Google Scholar] [CrossRef] [PubMed]
- Yata, K.; Esashi, F. Dual role of CDKs in DNA repair: To be, or not to be. DNA Repair 2009, 8, 6–18. [Google Scholar] [CrossRef] [PubMed]
- Lim, S.; Kaldis, P. Cdks, cyclins and CKIs: Roles beyond cell cycle regulation. Development 2013, 140, 3079–3093. [Google Scholar] [CrossRef] [PubMed]
- Bailis, J.M.; Bernard, P.; Antonelli, R.; Allshire, R.C.; Forsburg, S.L. Hsk1-Dfp1 is required for heterochromatin-mediated cohesion at centromeres. Nat. Cell Biol. 2003, 5, 1111–1116. [Google Scholar] [CrossRef] [PubMed]
- Tsuji, T.; Lau, E.; Chiang, G.G.; Jiang, W. The role of Dbf4/Drf1-dependent kinase Cdc7 in DNA-damage checkpoint control. Mol. Cell 2008, 32, 862–869. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, T.S.; Basu, A.; Bermudez, V.; Hurwitz, J.; Walter, J.C. Cdc7-Drf1 kinase links chromosome cohesion to the initiation of DNA replication in Xenopus egg extracts. Genes Dev. 2008, 22, 1894–1905. [Google Scholar] [CrossRef] [PubMed]
- Furuya, K.; Miyabe, I.; Tsutsui, Y.; Paderi, F.; Kakusho, N.; Masai, H.; Niki, H.; Carr, A.M. DDK phosphorylates checkpoint clamp component Rad9 and promotes its release from damaged chromatin. Mol. Cell 2010, 40, 606–618. [Google Scholar] [CrossRef] [PubMed]
- Lee, A.Y.-L.; Chiba, T.; Truong, L.N.; Cheng, A.N.; Do, J.; Cho, M.J.; Chen, L.; Wu, X. Dbf4 is direct downstream target of ataxia telangiectasia mutated (ATM) and ataxia telangiectasia and Rad3-related (ATR) protein to regulate intra-S-phase checkpoint. J. Biol. Chem. 2012, 287, 2531–2543. [Google Scholar] [CrossRef] [PubMed]
- Ogino, K.; Hirota, K.; Matsumoto, S.; Takeda, T.; Ohta, K.; Arai, K.-I.; Masai, H. Hsk1 kinase is required for induction of meiotic dsDNA breaks without involving checkpoint kinases in fission yeast. Proc. Natl. Acad. Sci. USA 2006, 103, 8131–8136. [Google Scholar] [CrossRef] [PubMed]
- Wan, L.; Zhang, C.; Shokat, K.M.; Hollingsworth, N.M. Chemical inactivation of cdc7 kinase in budding yeast results in a reversible arrest that allows efficient cell synchronization prior to meiotic recombination. Genetics 2006, 174, 1767–1774. [Google Scholar] [CrossRef] [PubMed]
- Lo, H.-C.; Wan, L.; Rosebrock, A.; Futcher, B.; Hollingsworth, N.M. Cdc7-Dbf4 regulates NDT80 transcription as well as reductional segregation during budding yeast meiosis. Mol. Biol. Cell 2008, 19, 4956–4967. [Google Scholar] [CrossRef] [PubMed]
- Matos, J.; Lipp, J.J.; Bogdanova, A.; Guillot, S.; Okaz, E.; Junqueira, M.; Shevchenko, A.; Zachariae, W. Dbf4-dependent CDC7 kinase links DNA replication to the segregation of homologous chromosomes in meiosis I. Cell 2008, 135, 662–678. [Google Scholar] [CrossRef] [PubMed]
- Sasanuma, H.; Hirota, K.; Fukuda, T.; Kakusho, N.; Kugou, K.; Kawasaki, Y.; Shibata, T.; Masai, H.; Ohta, K. Cdc7-dependent phosphorylation of Mer2 facilitates initiation of yeast meiotic recombination. Genes Dev. 2008, 22, 398–410. [Google Scholar] [CrossRef] [PubMed]
- Wan, L.; Niu, H.; Futcher, B.; Zhang, C.; Shokat, K.M.; Boulton, S.J.; Hollingsworth, N.M. Cdc28-Clb5 (CDK-S) and Cdc7-Dbf4 (DDK) collaborate to initiate meiotic recombination in yeast. Genes Dev. 2008, 22, 386–397. [Google Scholar] [CrossRef] [PubMed]
- Masai, H.; Matsui, E.; You, Z.; Ishimi, Y.; Tamai, K.; Arai, K. Human Cdc7-related kinase complex. In vitro phosphorylation of MCM by concerted actions of Cdks and Cdc7 and that of a criticial threonine residue of Cdc7 bY Cdks. J. Biol. Chem. 2000, 275, 29042–29052. [Google Scholar] [CrossRef] [PubMed]
- Nougarède, R.; Seta, Della, F.; Zarzov, P.; Schwob, E. Hierarchy of S-phase-promoting factors: Yeast Dbf4-Cdc7 kinase requires prior S-phase cyclin-dependent kinase activation. Mol. Cell. Biol. 2000, 20, 3795–3806. [Google Scholar]
- Devault, A.; Gueydon, E.; Schwob, E. Interplay between S-cyclin-dependent kinase and Dbf4-dependent kinase in controlling DNA replication through phosphorylation of yeast Mcm4 N-terminal domain. Mol. Biol. Cell 2008, 19, 2267–2277. [Google Scholar] [CrossRef] [PubMed]
- Sheu, Y.-J.; Stillman, B. The Dbf4-Cdc7 kinase promotes S phase by alleviating an inhibitory activity in Mcm4. Nature 2010, 463, 113–117. [Google Scholar] [CrossRef] [PubMed]
- Larasati; Duncker, B.P. Mechanisms Governing DDK Regulation of the Initiation of DNA Replication. Genes 2016, 8, 3. [Google Scholar]
- Kitada, K.; Johnston, L.H.; Sugino, T.; Sugino, A. Temperature-sensitive Cdc7 mutations of Saccharomyces cerevisiae are suppressed by the DBF4 gene, which is required for the G1/S cell cycle transition. Genetics 1992, 131, 21–29. [Google Scholar] [PubMed]
- Jackson, A.L.; Pahl, P.M.; Harrison, K.; Rosamond, J.; Sclafani, R.A. Cell cycle regulation of the yeast Cdc7 protein kinase by association with the Dbf4 protein. Mol. Cell. Biol. 1993, 13, 2899–2908. [Google Scholar] [CrossRef] [PubMed]
- Montagnoli, A.; Bosotti, R.; Villa, F.; Rialland, M.; Brotherton, D.; Mercurio, C.; Berthelsen, J.; Santocanale, C. Drf1, a novel regulatory subunit for human Cdc7 kinase. EMBO J. 2002, 21, 3171–3181. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, T.S.; Walter, J.C. Cdc7-Drf1 is a developmentally regulated protein kinase required for the initiation of vertebrate DNA replication. Genes Dev. 2005, 19, 2295–2300. [Google Scholar] [CrossRef] [PubMed]
- Fisher, D.L.; Nurse, P. A single fission yeast mitotic cyclin B p34cdc2 kinase promotes both S-phase and mitosis in the absence of G1 cyclins. EMBO J. 1996, 15, 850–860. [Google Scholar] [PubMed]
- Stern, B.; Nurse, P. A quantitative model for the cdc2 control of S phase and mitosis in fission yeast. Trends Genet. 1996, 12, 345–350. [Google Scholar] [CrossRef]
- Donaldson, A.D.; Raghuraman, M.K.; Friedman, K.L.; Cross, F.R.; Brewer, B.J.; Fangman, W.L. CLB5-dependent activation of late replication origins in S. cerevisiae. Mol. Cell 1998, 2, 173–182. [Google Scholar] [CrossRef]
- Hu, F.; Aparicio, O.M. Swe1 regulation and transcriptional control restrict the activity of mitotic cyclins toward replication proteins in Saccharomyces cerevisiae. Proc. Natl. Acad. Sci. USA 2005, 102, 8910–8915. [Google Scholar] [CrossRef] [PubMed]
- Moore, J.D.; Kirk, J.A.; Hunt, T. Unmasking the S-phase-promoting potential of cyclin B1. Science 2003, 300, 987–990. [Google Scholar] [CrossRef] [PubMed]
- Kozar, K.; Ciemerych, M.A.; Rebel, V.I.; Shigematsu, H.; Zagozdzon, A.; Sicinska, E.; Geng, Y.; Yu, Q.; Bhattacharya, S.; Bronson, R.T.; et al. Mouse development and cell proliferation in the absence of D-cyclins. Cell 2004, 118, 477–491. [Google Scholar] [CrossRef] [PubMed]
- Santamaría, D.; Barrière, C.; Cerqueira, A.; Hunt, S.; Tardy, C.; Newton, K.; Cáceres, J.F.; Dubus, P.; Malumbres, M.; Barbacid, M. Cdk1 is sufficient to drive the mammalian cell cycle. Nature 2007, 448, 811–815. [Google Scholar] [CrossRef] [PubMed]
- Coudreuse, D.; Nurse, P. Driving the cell cycle with a minimal CDK control network. Nature 2010, 468, 1074–1079. [Google Scholar] [CrossRef] [PubMed]
- Johnson, R.T.; Rao, P.N. Mammalian cell fusion: Induction of premature chromosome condensation in interphase nuclei. Nature 1970, 226, 717–722. [Google Scholar] [CrossRef] [PubMed]
- Banyai, G.; Baïdi, F.; Coudreuse, D.; Szilagyi, Z. Cdk1 activity acts as a quantitative platform for coordinating cell cycle progression with periodic transcription. Nat. Commun. 2016, 7, 11161. [Google Scholar] [CrossRef] [PubMed]
- Rahi, S.J.; Pecani, K.; Ondracka, A.; Oikonomou, C.; Cross, F.R. The CDK-APC/C Oscillator Predominantly Entrains Periodic Cell-Cycle Transcription. Cell 2016, 165, 475–487. [Google Scholar] [CrossRef] [PubMed]
- Swaffer, M.P.; Jones, A.W.; Flynn, H.R.; Snijders, A.P.; Nurse, P. CDK Substrate Phosphorylation and Ordering the Cell Cycle. Cell 2016, 167, 1750–1761. [Google Scholar] [CrossRef] [PubMed]
- Brown, G.W.; Kelly, T.J. Cell cycle regulation of Dfp1, an activator of the Hsk1 protein kinase. Proc. Natl. Acad. Sci. USA 1999, 96, 8443–8448. [Google Scholar] [CrossRef] [PubMed]
- Oshiro, G.; Owens, J.C.; Shellman, Y.; Sclafani, R.A.; Li, J.J. Cell cycle control of Cdc7p kinase activity through regulation of Dbf4p stability. Mol. Cell. Biol. 1999, 19, 4888–4896. [Google Scholar] [CrossRef] [PubMed]
- Takeda, T.; Ogino, K.; Matsui, E.; Cho, M.K.; Kumagai, H.; Miyake, T.; Arai, K.; Masai, H. A fission yeast gene, him1+/dfp1+, encoding a regulatory subunit for Hsk1 kinase, plays essential roles in S-phase initiation as well as in S-phase checkpoint control and recovery from DNA damage. Mol. Cell. Biol. 1999, 19, 5535–5547. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, M.F.; Santocanale, C.; Drury, L.S.; Diffley, J.F. Dbf4p, an essential S phase-promoting factor, is targeted for degradation by the anaphase-promoting complex. Mol. Cell. Biol. 2000, 20, 242–248. [Google Scholar] [CrossRef] [PubMed]
- Sclafani, R.A.; Patterson, M.; Rosamond, J.; Fangman, W.L. Differential regulation of the yeast CDC7 gene during mitosis and meiosis. Mol. Cell. Biol. 1988, 8, 293–300. [Google Scholar] [CrossRef] [PubMed]
- Patel, P.K.; Kommajosyula, N.; Rosebrock, A.; Bensimon, A.; Leatherwood, J.; Bechhoefer, J.; Rhind, N. The Hsk1(Cdc7) replication kinase regulates origin efficiency. Mol. Biol. Cell 2008, 19, 5550–5558. [Google Scholar] [CrossRef] [PubMed]
- Wu, P.-Y.J.; Nurse, P. Establishing the program of origin firing during S phase in fission Yeast. Cell 2009, 136, 852–864. [Google Scholar] [CrossRef] [PubMed]
- Mantiero, D.; Mackenzie, A.; Donaldson, A.; Zegerman, P. Limiting replication initiation factors execute the temporal programme of origin firing in budding yeast. EMBO J. 2011, 30, 4805–4814. [Google Scholar] [CrossRef] [PubMed]
- Grandin, N.; Reed, S.I. Differential function and expression of Saccharomyces cerevisiae B-type cyclins in mitosis and meiosis. Mol. Cell. Biol. 1993, 13, 2113–2125. [Google Scholar] [CrossRef] [PubMed]
- Chu, S.; Herskowitz, I. Gametogenesis in yeast is regulated by a transcriptional cascade dependent on Ndt80. Mol. Cell 1998, 1, 685–696. [Google Scholar] [CrossRef]
- Dahmann, C.; Futcher, B. Specialization of B-type cyclins for mitosis or meiosis in S. cerevisiae. Genetics 1995, 140, 957–963. [Google Scholar] [PubMed]
- Carlile, T.M.; Amon, A. Meiosis I is established through division-specific translational control of a cyclin. Cell 2008, 133, 280–291. [Google Scholar] [CrossRef] [PubMed]
- Schwob, E.; Nasmyth, K. CLB5 and CLB6, a new pair of B cyclins involved in DNA replication in Saccharomyces cerevisiae. Genes Dev. 1993, 7, 1160–1175. [Google Scholar] [CrossRef] [PubMed]
- Dirick, L.; Goetsch, L.; Ammerer, G.; Byers, B. Regulation of meiotic S phase by Ime2 and a Clb5,6-associated kinase in Saccharomyces cerevisiae. Science 1998, 281, 1854–1857. [Google Scholar] [CrossRef] [PubMed]
- Stuart, D.; Wittenberg, C. CLB5 and CLB6 are required for premeiotic DNA replication and activation of the meiotic S/M checkpoint. Genes Dev. 1998, 12, 2698–2710. [Google Scholar] [CrossRef] [PubMed]
- DeCesare, J.M.; Stuart, D.T. Among B-type cyclins only CLB5 and CLB6 promote premeiotic S phase in Saccharomyces cerevisiae. Genetics 2012, 190, 1001–1016. [Google Scholar] [CrossRef] [PubMed]
- Smith, K.N.; Penkner, A.; Ohta, K.; Klein, F.; Nicolas, A. B-type cyclins CLB5 and CLB6 control the initiation of recombination and synaptonemal complex formation in yeast meiosis. Current Biology 2001, 11, 88–97. [Google Scholar] [CrossRef]
- Gutiérrez-Escribano, P.; Nurse, P. A single cyclin-CDK complex is sufficient for both mitotic and meiotic progression in fission yeast. Nat. Commun. 2015, 6, 6871. [Google Scholar] [CrossRef] [PubMed]
- Malapeira, J.; Moldón, A.; Hidalgo, E.; Smith, G.R.; Nurse, P.; Ayté, J. A meiosis-specific cyclin regulated by splicing is required for proper progression through meiosis. Mol. Cell. Biol. 2005, 25, 6330–6337. [Google Scholar] [CrossRef] [PubMed]
- Geng, Y.; Yu, Q.; Sicinska, E.; Das, M.; Schneider, J.E.; Bhattacharya, S.; Rideout, W.M.; Bronson, R.T.; Gardner, H.; Sicinski, P. Cyclin E ablation in the mouse. Cell 2003, 114, 431–443. [Google Scholar] [CrossRef]
- Liu, D.; Matzuk, M.M.; Sung, W.K.; Guo, Q.; Wang, P.; Wolgemuth, D.J. Cyclin A1 is required for meiosis in the male mouse. Nat. Genet. 1998, 20, 377–380. [Google Scholar] [PubMed]
- Benjamin, K.R.; Benjamin, K.R.; Zhang, C.; Zhang, C.; Shokat, K.M.; Shokat, K.M.; Herskowitz, I.; Herskowitz, I. Control of landmark events in meiosis by the CDK Cdc28 and the meiosis-specific kinase Ime2. Genes Dev. 2003, 17, 1524–1539. [Google Scholar] [CrossRef] [PubMed]
- Schindler, K.; Winter, E. Phosphorylation of Ime2 regulates meiotic progression in Saccharomyces cerevisiae. J. Biol. Chem. 2006, 281, 18307–18316. [Google Scholar] [CrossRef] [PubMed]
- Szwarcwort-Cohen, M.; Kasulin-Boneh, Z.; Sagee, S.; Kassir, Y. Human Cdk2 is a functional homolog of budding yeast Ime2, the meiosis-specific Cdk-like kinase. Cell Cycle 2009, 8, 647–654. [Google Scholar] [CrossRef] [PubMed]
- Honigberg, S.M. Ime2p and Cdc28p: Co-pilots driving meiotic development. J. Cell. Biochem. 2004, 92, 1025–1033. [Google Scholar] [CrossRef] [PubMed]
- Averbeck, N.; Sunder, S.; Sample, N.; Wise, J.A.; Leatherwood, J. Negative control contributes to an extensive program of meiotic splicing in fission yeast. Mol. Cell 2005, 18, 491–498. [Google Scholar] [CrossRef] [PubMed]
- Borgne, A.; Murakami, H.; Ayté, J.; Nurse, P. The G1/S Cyclin Cig2p during Meiosis in Fission Yeast. Mol. Biol. Cell 2002, 13, 2080–2090. [Google Scholar] [CrossRef] [PubMed]
- Schild, D.; Byers, B. Meiotic effects of DNA-defective cell division cycle mutations of Saccharomyces cerevisiae. Chromosoma 1978, 70, 109–130. [Google Scholar] [CrossRef] [PubMed]
- Hollingsworth, R.E.; Sclafani, R.A. Yeast pre-meiotic DNA replication utilizes mitotic origin ARS1 independently of CDC7 function. Chromosoma 1993, 102, 415–420. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, T.; Nakamura-Kubo, M.; Nakamura, T.; Shimoda, C. Novel Fission Yeast Cdc7-Dbf4-Like Kinase Complex Required for the Initiation and Progression of Meiotic Second Division. Mol. Cell. Biol. 2002, 22, 309–320. [Google Scholar] [CrossRef] [PubMed]
- Kovacikova, I.; Polakova, S.; Benko, Z.; Cipak, L.; Zhang, L.; Rumpf, C.; Miadokova, E.; Gregan, J. A knockout screen for protein kinases required for the proper meiotic segregation of chromosomes in the fission yeast Schizosaccharomyces pombe. Cell Cycle 2013, 12, 618–624. [Google Scholar] [CrossRef] [PubMed]
- Donaldson, A.D.; Cross, F.R.; Raghuraman, M.K.; Levine, K.; Friedman, K.L.; Cross, F.R.; Brewer, B.J.; Fangman, W.L. Molecular evolution allows bypass of the requirement for activation loop phosphorylation of the Cdc28 cyclin-dependent kinase. Mol. Cell. Biol. 1998, 18, 2923–2931. [Google Scholar]
- Kaldis, P.; Pitluk, Z.W.; Bany, I.A.; Enke, D.A.; Wagner, M.; Winter, E.; Solomon, M.J. Localization and regulation of the cdk-activating kinase (Cak1p) from budding yeast. J. Cell Sci. 1998, 111, 3585–3596. [Google Scholar] [PubMed]
- Schwob, E.; Böhm, T.; Mendenhall, M.D.; Nasmyth, K. The B-type cyclin kinase inhibitor p40SIC1 controls the G1 to S transition in S. cerevisiae. Cell 1994, 79, 233–244. [Google Scholar] [CrossRef]
- Verma, R.; Annan, R.S.; Huddleston, M.J.; Carr, S.A.; Reynard, G.; Deshaies, R.J. Phosphorylation of Sic1p by G1 Cdk required for its degradation and entry into S phase. Science 1997, 278, 455–460. [Google Scholar] [CrossRef] [PubMed]
- Schindler, K.; Benjamin, K.R.; Martin, A.; Boglioli, A.; Herskowitz, I.; Winter, E. The Cdk-Activating Kinase Cak1p Promotes Meiotic S Phase through Ime2p. Mol. Cell. Biol. 2003, 23, 8718–8728. [Google Scholar] [CrossRef] [PubMed]
- Murakami, H.; Nurse, P. Regulation of premeiotic S phase and recombination-related double-strand DNA breaks during meiosis in fission yeast. Nat. Genet. 2001, 28, 290–293. [Google Scholar] [CrossRef] [PubMed]
- Lindner, K.; Gregan, J.; Montgomery, S.; Kearsey, S.E. Essential role of MCM proteins in premeiotic DNA replication. Mol. Biol. Cell 2002, 13, 435–444. [Google Scholar] [CrossRef] [PubMed]
- Ofir, Y.; Sagee, S.; Guttmann-Raviv, N.; Pnueli, L.; Kassir, Y. The role and regulation of the preRC component Cdc6 in the initiation of premeiotic DNA replication. Mol. Biol. Cell 2004, 15, 2230–2242. [Google Scholar] [CrossRef] [PubMed]
- Williamson, D.H.; Johnston, L.H.; Fennell, D.J.; Simchen, G. The timing of the S phase and other nuclear events in yeast meiosis. Exp. Cell Res. 1983, 145, 209–217. [Google Scholar] [CrossRef]
- Cha, R.S.; Weiner, B.M.; Keeney, S.; Dekker, J.; Kleckner, N. Progression of meiotic DNA replication is modulated by interchromosomal interaction proteins, negatively by Spo11p and positively by Rec8p. Genes Dev. 2000, 14, 493–503. [Google Scholar] [PubMed]
- Heichinger, C.; Penkett, C.J.; Bähler, J.; Nurse, P. Genome-wide characterization of fission yeast DNA replication origins. EMBO J. 2006, 25, 5171–5179. [Google Scholar] [CrossRef] [PubMed]
- Blitzblau, H.G.; Chan, C.S.; Hochwagen, A.; Bell, S.P. Separation of DNA replication from the assembly of break-competent meiotic chromosomes. PLoS Genet. 2012, 8, e1002643. [Google Scholar] [CrossRef] [PubMed]
- Wu, P.-Y.J.; Nurse, P. Replication origin selection regulates the distribution of meiotic recombination. Mol. Cell 2014, 53, 655–662. [Google Scholar] [CrossRef] [PubMed]
- Holm, P.B. The premeiotic DNA replication of euchromatin and heterochromatin in Lilium longiflorum (Thunb.). Carlsberg Res. Commun. 1977, 42, 249–281. [Google Scholar] [CrossRef]
- Cho, W.-H.; Lee, Y.-J.; Kong, S.-I.; Hurwitz, J.; Lee, J.-K. CDC7 kinase phosphorylates serine residues adjacent to acidic amino acids in the minichromosome maintenance 2 protein. Proc. Natl. Acad. Sci. USA 2006, 103, 11521–11526. [Google Scholar] [CrossRef] [PubMed]
- Zegerman, P.; Diffley, J.F.X. Phosphorylation of Sld2 and Sld3 by cyclin-dependent kinases promotes DNA replication in budding yeast. Nature 2007, 445, 281–285. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, S.; Umemori, T.; Hirai, K.; Muramatsu, S.; Kamimura, Y.; Araki, H. CDK-dependent phosphorylation of Sld2 and Sld3 initiates DNA replication in budding yeast. Nature 2007, 445, 328–332. [Google Scholar] [CrossRef] [PubMed]
- Heller, R.C.; Kang, S.; Lam, W.M.; Chen, S.; Chan, C.S.; Bell, S.P. Eukaryotic origin-dependent DNA replication in vitro reveals sequential action of DDK and S-CDK kinases. Cell 2011, 146, 80–91. [Google Scholar] [CrossRef] [PubMed]
- Yeeles, J.T.P.; Deegan, T.D.; Janska, A.; Early, A.; Diffley, J.F.X. Regulated eukaryotic DNA replication origin firing with purified proteins. Nature 2015, 519, 431–435. [Google Scholar] [CrossRef] [PubMed]
- Fragkos, M.; Ganier, O.; Coulombe, P.; Méchali, M. DNA replication origin activation in space and time. Nat. Rev. Mol. Cell Biol. 2015, 16, 360–374. [Google Scholar] [CrossRef] [PubMed]
- Picard, A.; Galas, S.; Peaucellier, G.; Dorée, M. Newly assembled cyclin B-cdc2 kinase is required to suppress DNA replication between meiosis I and meiosis II in starfish oocytes. EMBO J. 1996, 15, 3590–3598. [Google Scholar] [PubMed]
- Iwabuchi, M. Residual Cdc2 activity remaining at meiosis I exit is essential for meiotic M-M transition in Xenopus oocyte extracts. EMBO J. 2000, 19, 4513–4523. [Google Scholar] [CrossRef] [PubMed]
- Nakajo, N.; Yoshitome, S.; Iwashita, J.; Iida, M.; Uto, K.; Ueno, S.; Okamoto, K.; Sagata, N. Absence of Wee1 ensures the meiotic cell cycle in Xenopus oocytes. Genes Dev. 2000, 14, 328–338. [Google Scholar] [PubMed]
- Pesin, J.A.; Orr-Weaver, T.L. Regulation of APC/C activators in mitosis and meiosis. Annu. Rev. Cell Dev. Biol. 2008, 24, 475–499. [Google Scholar] [CrossRef] [PubMed]
- Cooper, K.F.; Strich, R. Meiotic control of the APC/C: Similarities & differences from mitosis. Cell Div 2011, 6, 16. [Google Scholar] [PubMed]
- Furuno, N.; Nishizawa, M.; Okazaki, K.; Tanaka, H.; Iwashita, J.; Nakajo, N.; Ogawa, Y.; Sagata, N. Suppression of DNA replication via Mos function during meiotic divisions in Xenopus oocytes. EMBO J. 1994, 13, 2399–2410. [Google Scholar] [PubMed]
- Tachibana, K.; Tanaka, D.; Isobe, T.; Kishimoto, T. c-Mos forces the mitotic cell cycle to undergo meiosis II to produce haploid gametes. Proc. Natl. Acad. Sci. USA 2000, 97, 14301–14306. [Google Scholar] [CrossRef] [PubMed]
- Hua, H.; Namdar, M.; Ganier, O.; Gregan, J.; Méchali, M.; Kearsey, S.E. Sequential steps in DNA replication are inhibited to ensure reduction of ploidy in meiosis. Mol. Biol. Cell 2013, 24, 578–587. [Google Scholar] [CrossRef] [PubMed]
- Knockleby, J.; Kim, B.J.; Mehta, A.; Lee, H. Cdk1-mediated phosphorylation of Cdc7 suppresses DNA re-replication. Cell Cycle 2016, 15, 1494–1505. [Google Scholar] [CrossRef] [PubMed]
- Whitmire, E.; Khan, B.; Coué, M. Cdc6 synthesis regulates replication competence in Xenopus oocytes. Nature 2002, 419, 722–725. [Google Scholar] [CrossRef] [PubMed]
- Murakami, H.; Keeney, S. Regulating the formation of DNA double-strand breaks in meiosis. Genes Dev. 2008, 22, 286–292. [Google Scholar] [CrossRef] [PubMed]
- Hochwagen, A.; Tham, W.-H.; Brar, G.A.; Amon, A. The FK506 binding protein Fpr3 counteracts protein phosphatase 1 to maintain meiotic recombination checkpoint activity. Cell 2005, 122, 861–873. [Google Scholar] [CrossRef] [PubMed]
- Tonami, Y.; Murakami, H.; Shirahige, K.; Nakanishi, M. A checkpoint control linking meiotic S phase and recombination initiation in fission yeast. Proc. Natl. Acad. Sci. USA 2005, 102, 5797–5801. [Google Scholar] [CrossRef] [PubMed]
- Ogino, K.; Masai, H. Rad3-Cds1 mediates coupling of initiation of meiotic recombination with DNA replication. Mei4-dependent transcription as a potential target of meiotic checkpoint. J. Biol. Chem. 2006, 281, 1338–1344. [Google Scholar] [CrossRef] [PubMed]
- Borde, V.; Goldman, A.S.; Lichten, M. Direct coupling between meiotic DNA replication and recombination initiation. Science 2000, 290, 806–809. [Google Scholar] [CrossRef] [PubMed]
- Murakami, H.; Borde, V.; Shibata, T.; Lichten, M.; Ohta, K. Correlation between premeiotic DNA replication and chromatin transition at yeast recombination initiation sites. Nucleic Acids Res. 2003, 31, 4085–4090. [Google Scholar] [CrossRef] [PubMed]
- Henderson, K.A.; Kee, K.; Maleki, S.; Santini, P.A.; Keeney, S. Cyclin-dependent kinase directly regulates initiation of meiotic recombination. Cell 2006, 125, 1321–1332. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, S.; Ogino, K.; Noguchi, E.; Russell, P.; Masai, H. Hsk1-Dfp1/Him1, the Cdc7-Dbf4 kinase in Schizosaccharomyces pombe, associates with Swi1, a component of the replication fork protection complex. J. Biol. Chem. 2005, 280, 42536–42542. [Google Scholar] [CrossRef] [PubMed]
- Murakami, H.; Keeney, S. Temporospatial coordination of meiotic DNA replication and recombination via DDK recruitment to replisomes. Cell 2014, 158, 861–873. [Google Scholar] [CrossRef] [PubMed]
- Ceccaldi, R.; Rondinelli, B.; D’Andrea, A.D. Repair Pathway Choices and Consequences at the Double-Strand Break. Trends Cell Biol. 2016, 26, 52–64. [Google Scholar] [CrossRef] [PubMed]
- Lindner, K.; Sasaki, M.; Gregan, J.; Lange, J.; Montgomery, S.; Keeney, S.; Kearsey, S.E. Genome destabilization by homologous recombination in the germ line. Nat. Rev. Mol. Cell Biol. 2010, 11, 182–195. [Google Scholar]
- Aylon, Y.; Liefshitz, B.; Kupiec, M. The CDK regulates repair of double-strand breaks by homologous recombination during the cell cycle. EMBO J. 2004, 23, 4868–4875. [Google Scholar] [CrossRef] [PubMed]
- Ira, G.; Pellicioli, A.; Balijja, A.; Wang, X.; Fiorani, S.; Carotenuto, W.; Liberi, G.; Bressan, D.; Wan, L.; Hollingsworth, N.M.; et al. DNA end resection, homologous recombination and DNA damage checkpoint activation require CDK1. Nature 2004, 431, 1011–1017. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, M.G.; Cooper, J.P. Two modes of DNA double-strand break repair are reciprocally regulated through the fission yeast cell cycle. Genes Dev. 2004, 18, 2249–2254. [Google Scholar] [CrossRef] [PubMed]
- Hentges, P.; Waller, H.; Reis, C.C.; Ferreira, M.G.; Doherty, A.J. Cdk1 restrains NHEJ through phosphorylation of XRCC4-like factor Xlf1. Cell Rep. 2014, 9, 2011–2017. [Google Scholar] [CrossRef] [PubMed]
- Heyer, W.-D.; Ehmsen, K.T.; Liu, J. Regulation of homologous recombination in eukaryotes. Annu. Rev. Genet. 2010, 44, 113–139. [Google Scholar] [CrossRef] [PubMed]
- Ferretti, L.P.; Lafranchi, L.; Sartori, A.A. Controlling DNA-end resection: A new task for CDKs. Front. Genet. 2013, 4, 99. [Google Scholar] [CrossRef] [PubMed]
- Huertas, P.; Cortés-Ledesma, F.; Sartori, A.A.; Aguilera, A.; Jackson, S.P. CDK targets Sae2 to control DNA-end resection and homologous recombination. Nature 2008, 455, 689–692. [Google Scholar] [CrossRef] [PubMed]
- Huertas, P.; Jackson, S.P. Human CtIP mediates cell cycle control of DNA end resection and double strand break repair. J. Biol. Chem. 2009, 284, 9558–9565. [Google Scholar] [CrossRef] [PubMed]
- Yun, M.H.; Hiom, K. CtIP-BRCA1 modulates the choice of DNA double-strand-break repair pathway throughout the cell cycle. Nature 2009, 459, 460–463. [Google Scholar] [CrossRef] [PubMed]
- Matos, J.; Blanco, M.G.; Maslen, S.; Skehel, J.M.; West, S.C. Regulatory control of the resolution of DNA recombination intermediates during meiosis and mitosis. Cell 2011, 147, 158–172. [Google Scholar] [CrossRef] [PubMed]
- Gallo-Fernández, M.; Saugar, I.; Ortiz-Bazán, M.Á.; Vázquez, M.V.; Tercero, J.A. Cell cycle-dependent regulation of the nuclease activity of Mus81-Eme1/Mms4. Nucleic Acids Res. 2012, 40, 8325–8335. [Google Scholar] [CrossRef]
- Szakal, B.; Branzei, D. Premature Cdk1/Cdc5/Mus81 pathway activation induces aberrant replication and deleterious crossover. EMBO J. 2013, 32, 1155–1167. [Google Scholar] [CrossRef] [PubMed]
- Clerici, M.; Mantiero, D.; Lucchini, G.; Longhese, M.P. The Saccharomyces cerevisiae Sae2 protein promotes resection and bridging of double strand break ends. J. Biol. Chem. 2005, 280, 38631–38638. [Google Scholar] [CrossRef] [PubMed]
- Princz, L.N.; Wild, P.; Bittmann, J.; Aguado, F.J.; Blanco, M.G.; Matos, J.; Pfander, B. Dbf4-dependent kinase and the Rtt107 scaffold promote Mus81-Mms4 resolvase activation during mitosis. EMBO J. 2017, 36, 664–678. [Google Scholar] [CrossRef] [PubMed]
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gómez-Escoda, B.; Wu, P.-Y.J.
Roles of CDK and DDK in Genome Duplication and Maintenance: Meiotic Singularities
. Genes 2017, 8, 105.
https://doi.org/10.3390/genes8030105
Gómez-Escoda B, Wu P-YJ.
Roles of CDK and DDK in Genome Duplication and Maintenance: Meiotic Singularities
. Genes. 2017; 8(3):105.
https://doi.org/10.3390/genes8030105
Gómez-Escoda, Blanca, and Pei-Yun Jenny Wu.
2017. "Roles of CDK and DDK in Genome Duplication and Maintenance: Meiotic Singularities
" Genes 8, no. 3: 105.
https://doi.org/10.3390/genes8030105
Gómez-Escoda, B., & Wu, P.-Y. J.
(2017). Roles of CDK and DDK in Genome Duplication and Maintenance: Meiotic Singularities
. Genes, 8(3), 105.
https://doi.org/10.3390/genes8030105




