Elaborated Action of the Human Primosome
Abstract
:1. Introduction
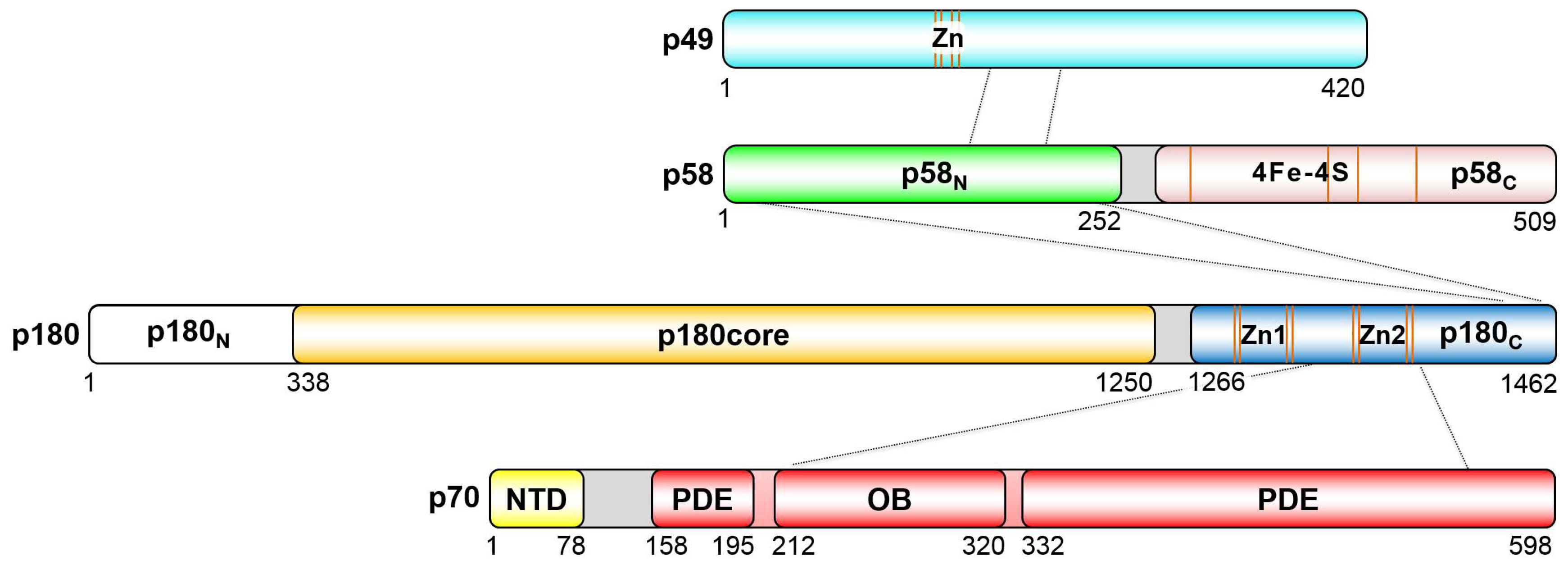
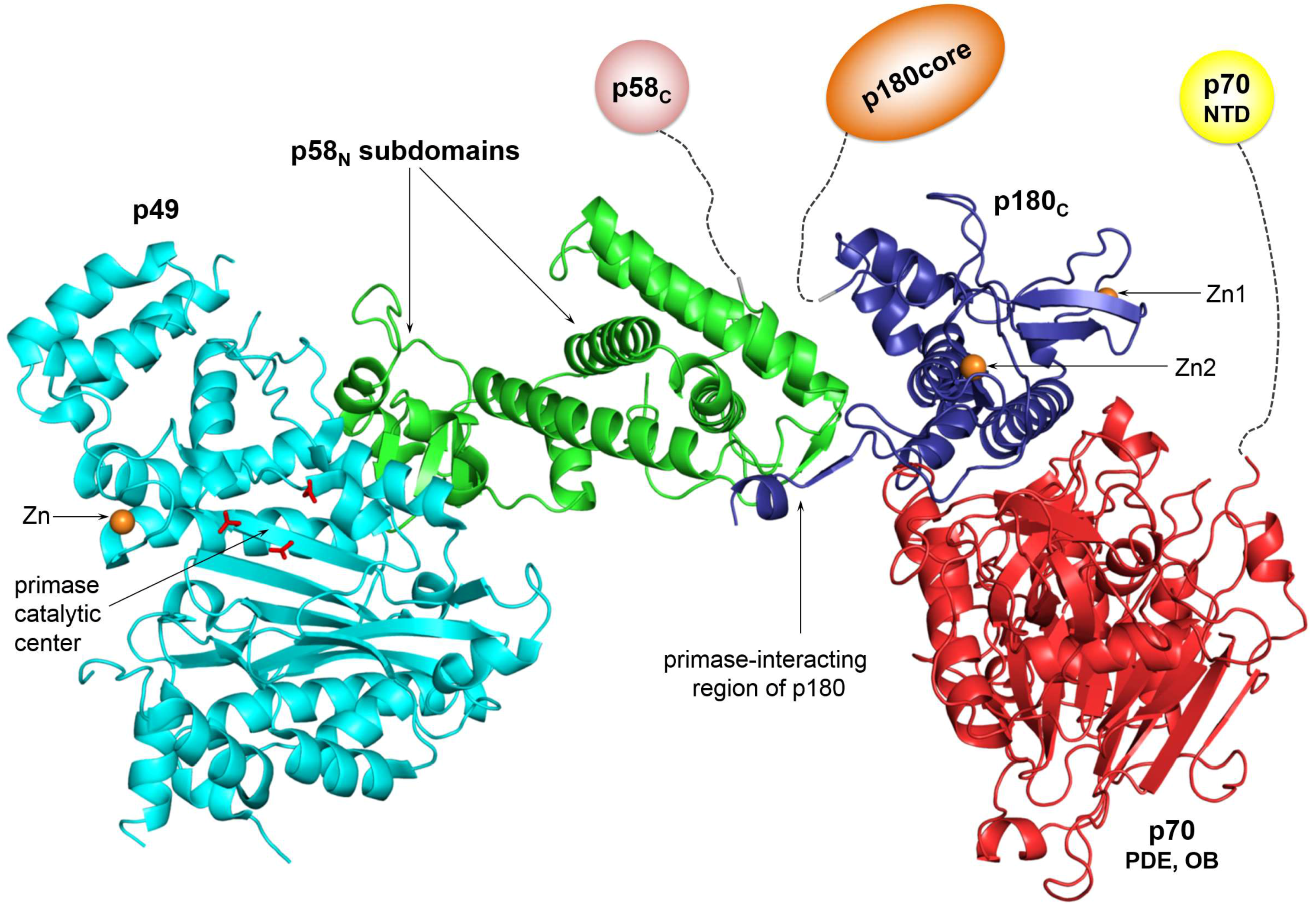
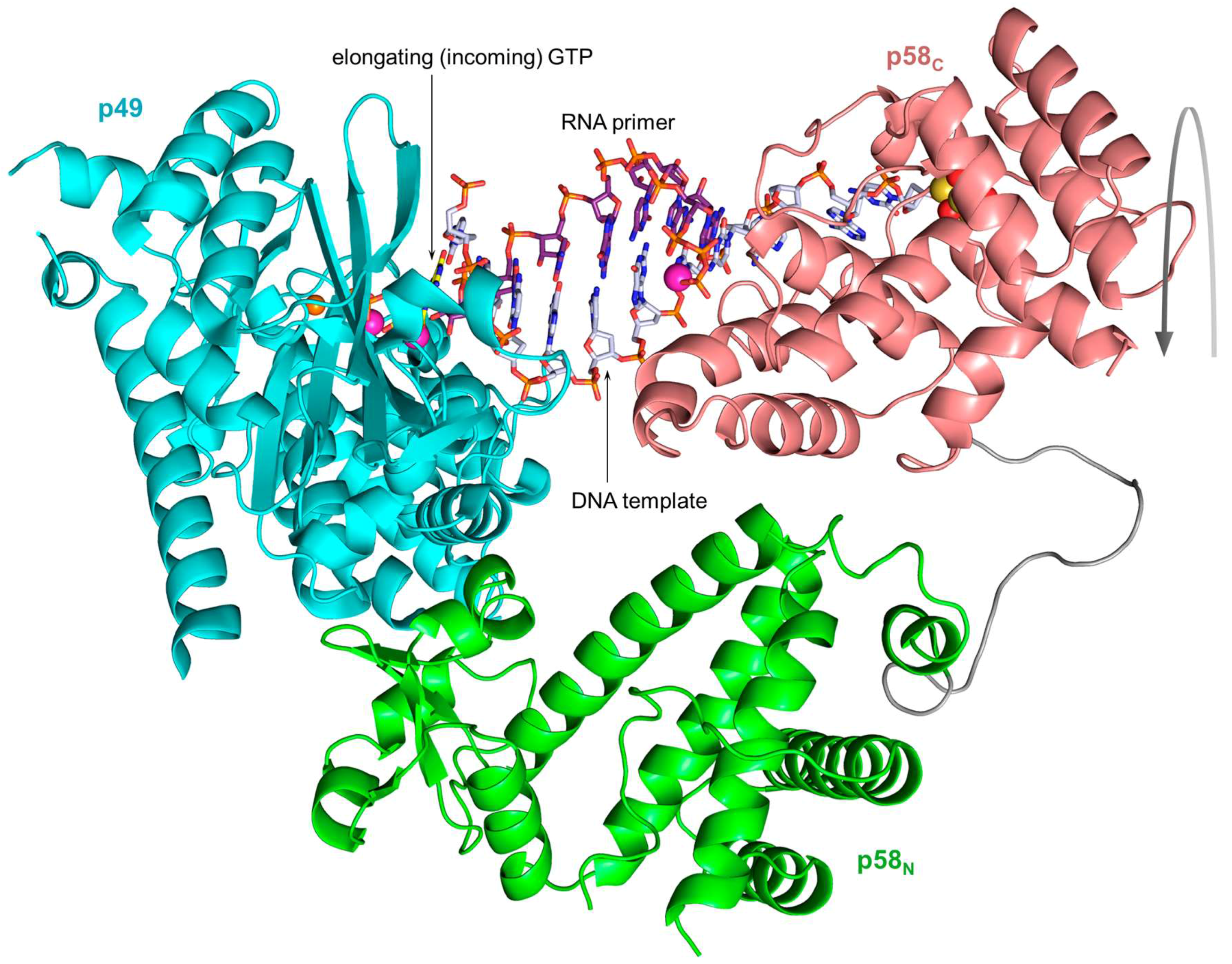
2. Organization of the Human Primosome
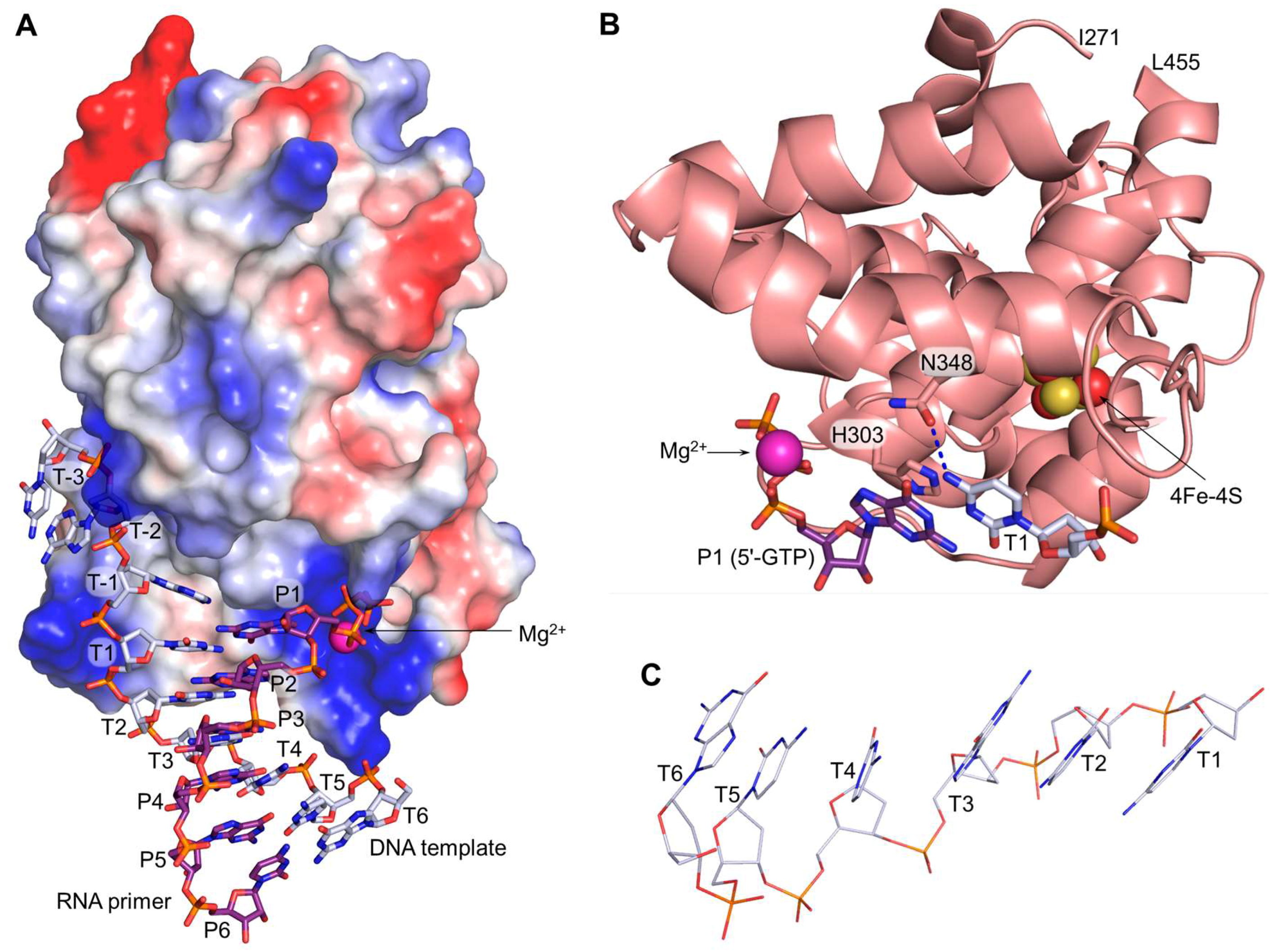
3. Interaction of Human Primase with a Template:Primer
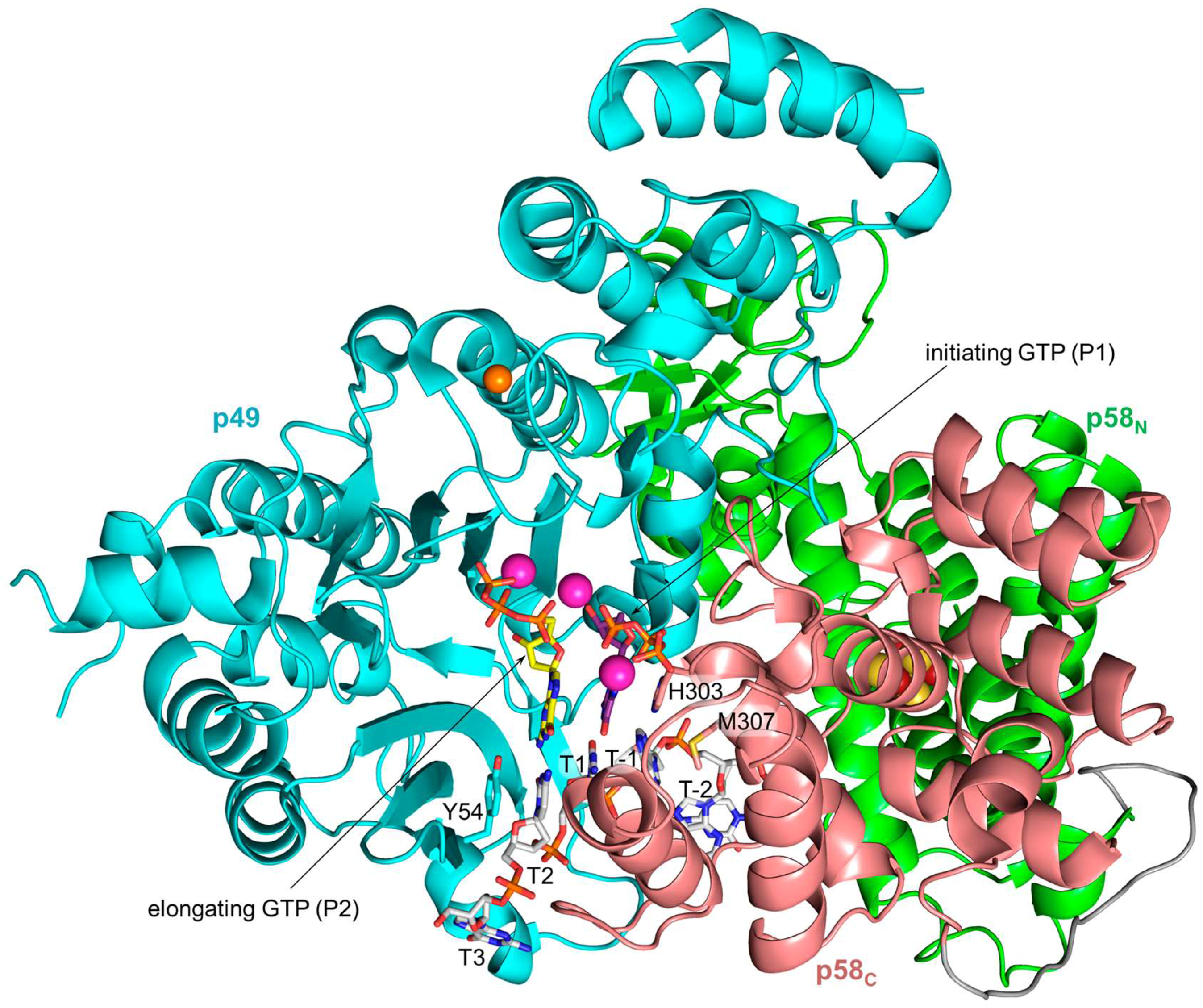
4. Mechanisms of RNA Synthesis Initiation, Elongation, and Termination
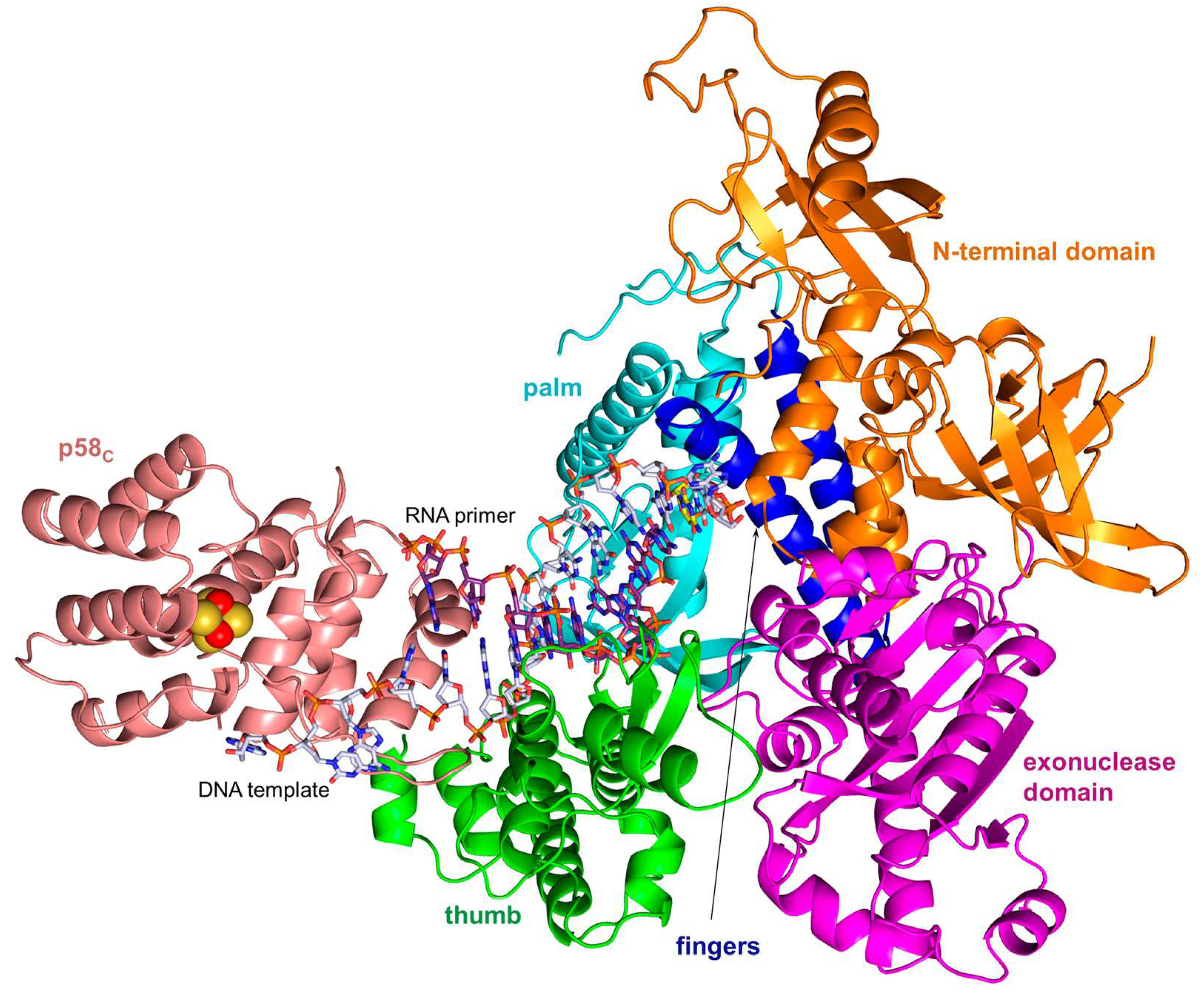
5. Mechanism of RNA Primer Transfer to Polα and Its Extension with dNTPs
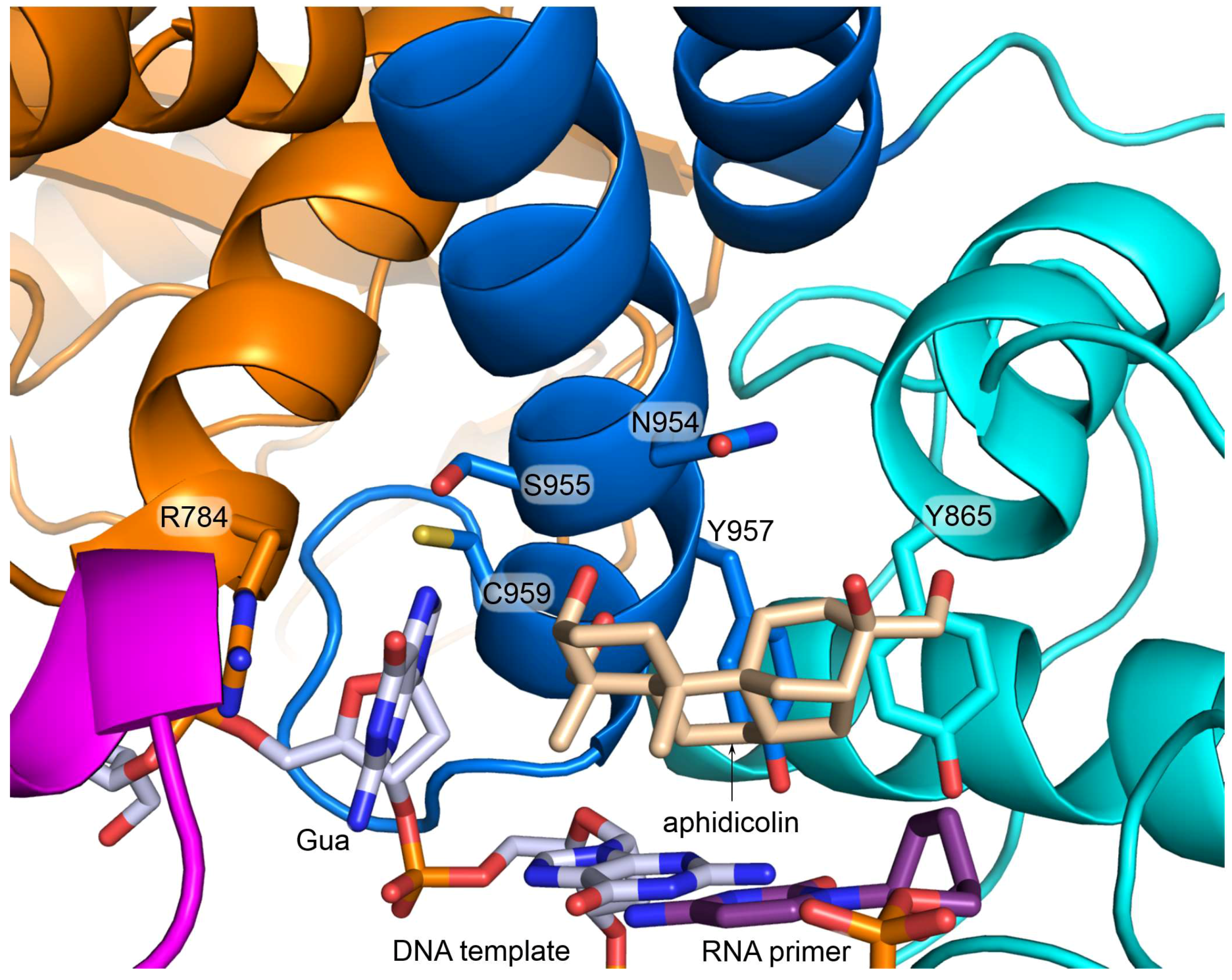
6. Polα Inhibition by Aphidicolin
7. Mechanism of Concerted RNA-DNA Primer Synthesis by the Human Primosome
8. Concluding Remarks
Acknowledgments
Conflicts of Interest
References
- Muzi-Falconi, M.; Giannattasio, M.; Foiani, M.; Plevani, P. The DNA polymerase α-primase complex: Multiple functions and interactions. Sci. World J. 2003, 3, 21–33. [Google Scholar] [CrossRef] [PubMed]
- Pavlov, Y.I.; Shcherbakova, P.V. DNA polymerases at the eukaryotic fork-20 years later. Mutat. Res. 2010, 685, 45–53. [Google Scholar] [CrossRef] [PubMed]
- Lujan, S.A.; Williams, J.S.; Kunkel, T.A. DNA polymerases divide the labor of genome replication. Trends Cell Biol. 2016, 26, 640–654. [Google Scholar] [CrossRef] [PubMed]
- Smith, D.J.; Whitehouse, I. Intrinsic coupling of lagging-strand synthesis to chromatin assembly. Nature 2012, 483, 434–438. [Google Scholar] [CrossRef] [PubMed]
- Pellegrini, L. The pol α-primase complex. Subcell. Biochem. 2012, 62, 157–169. [Google Scholar] [PubMed]
- MacNeill, S. Composition and dynamics of the eukaryotic replisome: A brief overview. Subcell. Biochem. 2012, 62, 1–17. [Google Scholar] [PubMed]
- Reijns, M.A.; Kemp, H.; Ding, J.; de Proce, S.M.; Jackson, A.P.; Taylor, M.S. Lagging-strand replication shapes the mutational landscape of the genome. Nature 2015, 518, 502–506. [Google Scholar] [CrossRef] [PubMed]
- Starokadomskyy, P.; Gemelli, T.; Rios, J.J.; Xing, C.; Wang, R.C.; Li, H.; Pokatayev, V.; Dozmorov, I.; Khan, S.; Miyata, N.; et al. DNA polymerase-α regulates the activation of type I interferons through cytosolic rna:DNA synthesis. Nat. Immunol. 2016, 17, 495–504. [Google Scholar] [CrossRef] [PubMed]
- Kuchta, R.D.; Stengel, G. Mechanism and evolution of DNA primases. Biochim. Biophys. Acta 2010, 1804, 1180–1189. [Google Scholar] [CrossRef] [PubMed]
- Frick, D.N.; Richardson, C.C. DNA primases. Annu. Rev. Biochem. 2001, 70, 39–80. [Google Scholar] [CrossRef]
- Copeland, W.C.; Wang, T.S. Enzymatic characterization of the individual mammalian primase subunits reveals a biphasic mechanism for initiation of DNA replication. J. Biol. Chem. 1993, 268, 26179–26189. [Google Scholar] [PubMed]
- Sheaff, R.J.; Kuchta, R.D. Mechanism of calf thymus DNA primase: Slow initiation, rapid polymerization, and intelligent termination. Biochemistry 1993, 32, 3027–3037. [Google Scholar] [CrossRef] [PubMed]
- Baranovskiy, A.G.; Babayeva, N.D.; Zhang, Y.; Gu, J.; Suwa, Y.; Pavlov, Y.I.; Tahirov, T.H. Mechanism of concerted RNA-DNA primer synthesis by the human primosome. J. Biol. Chem. 2016, 291, 10006–10020. [Google Scholar] [CrossRef] [PubMed]
- Sheaff, R.J.; Kuchta, R.D.; Ilsley, D. Calf thymus DNA polymerase α-primase: “Communication” and primer-template movement between the two active sites. Biochemistry 1994, 33, 2247–2254. [Google Scholar] [CrossRef] [PubMed]
- Kuchta, R.D.; Reid, B.; Chang, L.M. DNA primase. Processivity and the primase to polymerase α activity switch. J. Biol. Chem. 1990, 265, 16158–16165. [Google Scholar] [PubMed]
- Kilkenny, M.L.; Longo, M.A.; Perera, R.L.; Pellegrini, L. Structures of human primase reveal design of nucleotide elongation site and mode of pol α tethering. Proc. Natl. Acad. Sci. USA 2013, 110, 15961–15966. [Google Scholar] [CrossRef] [PubMed]
- Baranovskiy, A.G.; Babayeva, N.D.; Suwa, Y.; Gu, J.; Pavlov, Y.I.; Tahirov, T.H. Structural basis for inhibition of DNA replication by aphidicolin. Nucleic Acids Res. 2014, 42, 14013–14021. [Google Scholar] [CrossRef] [PubMed]
- Baranovskiy, A.G.; Zhang, Y.; Suwa, Y.; Babayeva, N.D.; Gu, J.; Pavlov, Y.I.; Tahirov, T.H. Crystal structure of the human primase. J. Biol. Chem. 2015, 290, 5635–5646. [Google Scholar] [CrossRef] [PubMed]
- Suwa, Y.; Gu, J.; Baranovskiy, A.G.; Babayeva, N.D.; Pavlov, Y.I.; Tahirov, T.H. Crystal structure of the human Pol α B subunit in complex with the C-terminal domain of the catalytic subunit. J. Biol. Chem. 2015, 290, 14328–14337. [Google Scholar] [CrossRef] [PubMed]
- Coloma, J.; Johnson, R.E.; Prakash, L.; Prakash, S.; Aggarwal, A.K. Human DNA polymerase α in binary complex with a DNA:DNA template-primer. Sci. Rep. 2016, 6, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Vaithiyalingam, S.; Warren, E.M.; Eichman, B.F.; Chazin, W.J. Insights into eukaryotic DNA priming from the structure and functional interactions of the 4Fe-4S cluster domain of human DNA primase. Proc. Natl. Acad. Sci. USA 2010, 107, 13684–13689. [Google Scholar] [CrossRef] [PubMed]
- Agarkar, V.B.; Babayeva, N.D.; Pavlov, Y.I.; Tahirov, T.H. Crystal structure of the C-terminal domain of human DNA primase large subunit: Implications for the mechanism of the primase-polymerase α switch. Cell Cycle 2011, 10, 926–931. [Google Scholar] [CrossRef] [PubMed]
- Vaithiyalingam, S.; Arnett, D.R.; Aggarwal, A.; Eichman, B.F.; Fanning, E.; Chazin, W.J. Insights into eukaryotic primer synthesis from structures of the p48 subunit of human DNA primase. J. Mol. Biol. 2014, 426, 558–569. [Google Scholar] [CrossRef] [PubMed]
- Mizuno, T.; Yamagishi, K.; Miyazawa, H.; Hanaoka, F. Molecular architecture of the mouse DNA polymerase α-primase complex. Mol. Cell. Biol. 1999, 19, 7886–7896. [Google Scholar] [CrossRef] [PubMed]
- Simon, A.C.; Zhou, J.C.; Perera, R.L.; van Deursen, F.; Evrin, C.; Ivanova, M.E.; Kilkenny, M.L.; Renault, L.; Kjaer, S.; Matak-Vinkovic, D.; et al. A Ctf4 trimer couples the cmg helicase to DNA polymerase α in the eukaryotic replisome. Nature 2014, 510, 293–297. [Google Scholar] [CrossRef] [PubMed]
- Baranovskiy, A.G.; Babayeva, N.D.; Liston, V.G.; Rogozin, I.B.; Koonin, E.V.; Pavlov, Y.I.; Vassylyev, D.G.; Tahirov, T.H. X-ray structure of the complex of regulatory subunits of human DNA polymerase delta. Cell Cycle 2008, 7, 3026–3036. [Google Scholar] [CrossRef] [PubMed]
- Zhou, B.; Arnett, D.R.; Yu, X.; Brewster, A.; Sowd, G.A.; Xie, C.L.; Vila, S.; Gai, D.; Fanning, E.; Chen, X.S. Structural basis for the interaction of a hexameric replicative helicase with the regulatory subunit of human DNA polymerase α-primase. J. Biol. Chem. 2012, 287, 26854–26866. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.; Zhao, K.; Arnett, D.R.; Fanning, E. A specific docking site for DNA polymerase {α}-primase on the SV40 helicase is required for viral primosome activity, but helicase activity is dispensable. J. Biol. Chem. 2010, 285, 33475–33484. [Google Scholar] [CrossRef] [PubMed]
- Leipe, D.D.; Aravind, L.; Koonin, E.V. Did DNA replication evolve twice independently? Nucleic Acids Res. 1999, 27, 3389–3401. [Google Scholar] [CrossRef] [PubMed]
- Ito, N.; Nureki, O.; Shirouzu, M.; Yokoyama, S.; Hanaoka, F. Crystal structure of the Pyrococcus horikoshii DNA primase-UTP complex: Implications for the mechanism of primer synthesis. Genes Cells 2003, 8, 913–923. [Google Scholar] [CrossRef] [PubMed]
- Lao-Sirieix, S.H.; Nookala, R.K.; Roversi, P.; Bell, S.D.; Pellegrini, L. Structure of the heterodimeric core primase. Nat. Struct. Mol. Biol. 2005, 12, 1137–1144. [Google Scholar] [CrossRef] [PubMed]
- Augustin, M.A.; Huber, R.; Kaiser, J.T. Crystal structure of a DNA-dependent RNA polymerase (DNA primase). Nat. Struct. Biol. 2001, 8, 57–61. [Google Scholar] [CrossRef] [PubMed]
- Klinge, S.; Hirst, J.; Maman, J.D.; Krude, T.; Pellegrini, L. An iron-sulfur domain of the eukaryotic primase is essential for RNA primer synthesis. Nat. Struct. Mol. Biol. 2007, 14, 875–877. [Google Scholar] [CrossRef] [PubMed]
- Francesconi, S.; Longhese, M.P.; Piseri, A.; Santocanale, C.; Lucchini, G.; Plevani, P. Mutations in conserved yeast DNA primase domains impair DNA replication in vivo. Proc. Natl. Acad. Sci. USA 1991, 88, 3877–3881. [Google Scholar] [CrossRef] [PubMed]
- Weiner, B.E.; Huang, H.; Dattilo, B.M.; Nilges, M.J.; Fanning, E.; Chazin, W.J. An iron-sulfur cluster in the C-terminal domain of the p58 subunit of human DNA primase. J. Biol. Chem. 2007, 282, 33444–33451. [Google Scholar] [CrossRef] [PubMed]
- Netz, D.J.; Stith, C.M.; Stumpfig, M.; Kopf, G.; Vogel, D.; Genau, H.M.; Stodola, J.L.; Lill, R.; Burgers, P.M.; Pierik, A.J. Eukaryotic DNA polymerases require an iron-sulfur cluster for the formation of active complexes. Nat. Chem. Biol. 2011, 8, 125–132. [Google Scholar] [CrossRef] [PubMed]
- Baranovskiy, A.G.; Lada, A.G.; Siebler, H.M.; Zhang, Y.; Pavlov, Y.I.; Tahirov, T.H. DNA polymerase delta and zeta switch by sharing accessory subunits of DNA polymerase delta. J. Biol. Chem. 2012, 287, 17281–17287. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Baranovskiy, A.G.; Tahirov, T.H.; Pavlov, Y.I. The C-terminal domain of the DNA polymerase catalytic subunit regulates the primase and polymerase activities of the human DNA polymerase α-primase complex. J. Biol. Chem. 2014, 289, 22021–22034. [Google Scholar] [CrossRef]
- Klinge, S.; Nunez-Ramirez, R.; Llorca, O.; Pellegrini, L. 3D architecture of DNA pol α reveals the functional core of multi-subunit replicative polymerases. EMBO J. 2009, 28, 1978–1987. [Google Scholar] [CrossRef] [PubMed]
- Copeland, W.C.; Wang, T.S. Mutational analysis of the human DNA polymerase α. The most conserved region in α-like DNA polymerases is involved in metal-specific catalysis. J. Biol. Chem. 1993, 268, 11028–11040. [Google Scholar] [PubMed]
- Copeland, W.C.; Tan, X. Active site mapping of the catalytic mouse primase subunit by alanine scanning mutagenesis. J. Biol. Chem. 1995, 270, 3905–3913. [Google Scholar] [PubMed]
- Zerbe, L.K.; Kuchta, R.D. The p58 subunit of human DNA primase is important for primer initiation, elongation, and counting. Biochemistry 2002, 41, 4891–4900. [Google Scholar] [CrossRef] [PubMed]
- Baranovskiy, A.G.; Zhang, Y.; Suwa, Y.; Gu, J.; Babayeva, N.D.; Pavlov, Y.I.; Tahirov, T.H. Insight into the human DNA primase interaction with template-primer. J. Biol. Chem. 2016, 291, 4793–4802. [Google Scholar] [CrossRef] [PubMed]
- Podust, V.N.; Vladimirova, O.V.; Manakova, E.N.; Lavrik, O.I. Eukaryotic DNA primase appears to act as oligomer in DNA-polymerase-α--primase complex. Eur. J. Biochem. 1992, 206, 7–13. [Google Scholar] [CrossRef] [PubMed]
- Kirk, B.W.; Kuchta, R.D. Arg304 of human DNA primase is a key contributor to catalysis and NTP binding: Primase and the family X polymerases share significant sequence homology. Biochemistry 1999, 38, 7727–7736. [Google Scholar] [CrossRef] [PubMed]
- Grosse, F.; Krauss, G. The primase activity of DNA polymerase α from calf thymus. J. Biol. Chem. 1985, 260, 1881–1888. [Google Scholar] [PubMed]
- Zahurancik, W.J.; Klein, S.J.; Suo, Z. Kinetic mechanism of DNA polymerization catalyzed by human DNA polymerase epsilon. Biochemistry 2013, 52, 7041–7049. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.; Lee, J.Y.; Nowotny, M. Making and breaking nucleic acids: Two-Mg2+-ion catalysis and substrate specificity. Mol. Cell 2006, 22, 5–13. [Google Scholar] [CrossRef] [PubMed]
- Kuchta, R.D.; Ilsley, D.; Kravig, K.D.; Schubert, S.; Harris, B. Inhibition of DNA primase and polymerase α by arabinofuranosylnucleoside triphosphates and related compounds. Biochemistry 1992, 31, 4720–4728. [Google Scholar] [CrossRef] [PubMed]
- Traut, T.W. Physiological concentrations of purines and pyrimidines. Mol. Cell. Biochem. 1994, 140, 1–22. [Google Scholar] [CrossRef] [PubMed]
- Urban, M.; Joubert, N.; Purse, B.W.; Hocek, M.; Kuchta, R.D. Mechanisms by which human DNA primase chooses to polymerize a nucleoside triphosphate. Biochemistry 2010, 49, 727–735. [Google Scholar] [CrossRef] [PubMed]
- Jozwiakowski, S.K.; Borazjani Gholami, F.; Doherty, A.J. Archaeal replicative primases can perform translesion DNA synthesis. Proc. Natl. Acad. Sci. USA 2015, 112, E633–E638. [Google Scholar] [CrossRef] [PubMed]
- Ogawa, T.; Okazaki, T. Discontinuous DNA replication. Ann. Rev. Biochem. 1980, 19, 421–457. [Google Scholar] [CrossRef] [PubMed]
- Pavlov, Y.I.; Shcherbakova, P.V.; Rogozin, I.B. Roles of DNA polymerases in replication, repair, and recombination in eukaryotes. Int. Rev. Cytol. 2006, 255, 41–132. [Google Scholar]
- Thompson, H.C.; Sheaff, R.J.; Kuchta, R.D. Interactions of calf thymus DNA polymerase α with primer/templates. Nucleic Acids Res. 1995, 23, 4109–4115. [Google Scholar] [CrossRef] [PubMed]
- Perera, R.L.; Torella, R.; Klinge, S.; Kilkenny, M.L.; Maman, J.D.; Pellegrini, L. Mechanism for priming DNA synthesis by yeast DNA polymerase α. Elife 2013, 2, e00482. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Baranovskiy, A.G.; Tahirov, E.T.; Tahirov, T.H.; Pavlov, Y.I. Divalent ions attenuate DNA synthesis by human DNA polymerase α by changing the structure of the template/primer or by perturbing the polymerase reaction. DNA Repair 2016, 43, 24–33. [Google Scholar] [CrossRef] [PubMed]
- Nadeau, J.G.; Crothers, D.M. Structural basis for DNA bending. Proc. Natl. Acad. Sci. USA 1989, 86, 2622–2626. [Google Scholar] [CrossRef] [PubMed]
- Ikegami, S.; Taguchi, T.; Ohashi, M.; Oguro, M.; Nagano, H.; Mano, Y. Aphidicolin prevents mitotic cell division by interfering with the activity of DNA polymerase-α. Nature 1978, 275, 458–460. [Google Scholar] [CrossRef] [PubMed]
- Huberman, J.A. New views of the biochemistry of eucaryotic DNA replication revealed by aphidicolin, an unusual inhibitor of DNA polymerase α. Cell 1981, 23, 647–648. [Google Scholar] [CrossRef]
- Sheaff, R.; Ilsley, D.; Kuchta, R. Mechanism of DNA polymerase α inhibition by aphidicolin. Biochemistry 1991, 30, 8590–8597. [Google Scholar] [CrossRef] [PubMed]
- Prasad, G.; Edelson, R.A.; Gorycki, P.D.; Macdonald, T.L. Structure-activity relationships for the inhibition of DNA polymerase α by aphidicolin derivatives. Nucleic Acids Res. 1989, 17, 6339–6348. [Google Scholar] [CrossRef] [PubMed]
- Pedrali-Noy, G.; Belvedere, M.; Crepaldi, T.; Focher, F.; Spadari, S. Inhibition of DNA replication and growth of several human and murine neoplastic cells by aphidicolin without detectable effect upon synthesis of immunoglobulins and HLA antigens. Cancer Res. 1982, 42, 3810–3813. [Google Scholar] [PubMed]
- Cinatl, J., Jr.; Cinatl, J.; Kotchetkov, R.; Driever, P.H.; Bertels, S.; Siems, K.; Jas, G.; Bindseil, K.; Rabenau, H.F.; Pouckova, P.; et al. Aphidicolin glycinate inhibits human neuroblastoma cell growth in vivo. Oncol. Rep. 1999, 6, 563–568. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Huang, M. Essential role of the iron-sulfur cluster binding domain of the primase regulatory subunit Pri2 in DNA replication initiation. Protein Cell 2015, 6, 194–210. [Google Scholar] [CrossRef] [PubMed]
- Conaway, R.C.; Lehman, I.R. A DNA primase activity associated with DNA polymerase α from Drosophila melanogaster embryos. Proc. Natl. Acad. Sci. USA 1982, 79, 2523–2527. [Google Scholar] [CrossRef] [PubMed]
- Hubscher, U. The mammalian primase is part of a high molecular weight DNA polymerase α polypeptide. EMBO J. 1983, 2, 133–136. [Google Scholar] [PubMed]
- Plevani, P.; Badaracco, G.; Augl, C.; Chang, L.M. DNA polymerase I and DNA primase complex in yeast. J. Biol. Chem. 1984, 259, 7532–7539. [Google Scholar]
- Villa, F.; Simon, A.C.; Ortiz Bazan, M.A.; Kilkenny, M.L.; Wirthensohn, D.; Wightman, M.; Matak-Vinkovic, D.; Pellegrini, L.; Labib, K. Ctf4 is a hub in the eukaryotic replisome that links multiple CIP-Box proteins to the CMG helicase. Mol. Cell 2016, 63, 385–396. [Google Scholar] [CrossRef] [PubMed]
- Sengupta, S.; van Deursen, F.; de Piccoli, G.; Labib, K. Dpb2 integrates the leading-strand DNA polymerase into the eukaryotic replisome. Curr. Biol. 2013, 23, 543–552. [Google Scholar] [CrossRef] [PubMed]
- Langston, L.D.; Zhang, D.; Yurieva, O.; Georgescu, R.E.; Finkelstein, J.; Yao, N.Y.; Indiani, C.; O’Donnell, M.E. Cmg helicase and DNA polymerase epsilon form a functional 15-subunit holoenzyme for eukaryotic leading-strand DNA replication. Proc. Natl. Acad. Sci. USA 2014, 111, 15390–15395. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; Shi, Y.; Georgescu, R.E.; Yuan, Z.; Chait, B.T.; Li, H.; O’Donnell, M.E. The architecture of a eukaryotic replisome. Nat. Struct. Mol. Biol. 2015, 22, 976–982. [Google Scholar] [CrossRef] [PubMed]


| PDB ID | Resolution, (Å) | Protein Construct | Structural Metals | Cofactors | Deposition Date | Reference |
|---|---|---|---|---|---|---|
| 3L9Q | 1.7 | p58(272–464) | 4Fe-4S | 5 January 2010 | [21] | |
| 3Q36 | 2.5 | p58(266–457) | 4Fe-4S | 21 December 2010 | [22] | |
| 4BPU | 2.7 | p49 a/p58(1–253) | Zn | 28 May 2013 | [16] | |
| 4BPW | 3.0 | p49 a/p58(1–253) | Zn | UTP, Mg | 28 May 2013 | [16] |
| 4BPX | 3.4 | p49 a/p58(19–253) b/p180(1445–1462) | Zn | 28 May 2013 | [16] | |
| 4LIK | 1.7 | p49(1–390) c | Zn | 2 July 2013 | [23] | |
| 4LIL | 2.6 | p49(1–390) c | Zn | UTP, Mn | 2 July 2013 | [23] |
| 4MHQ | 2.2 | p49 | Zn | 30 August 2013 | ||
| 4QCL | 2.2 | p180(335–1257) d | Zn | DNA:RNA, dCTP, Mg | 12 May 2014 | [17] |
| 4Q5V | 2.52 | p180(335–1257) d | DNA:RNA, aphidicolin | 17 April 2014 | [17] | |
| 4RR2 | 2.65 | p49/p58 | Zn, 4Fe-4S | 5 November 2014 | [18] | |
| 4Y97 | 2.51 | p180(1265–1444)/p70 | Zn | 17 February 2015 | [19] | |
| 5DQO | 2.3 | p58(272–464) e | 4Fe-4S | 15 September 2015 | ||
| 5EXR | 3.6 | p49/p58/p180 d/p70 | Zn, 4Fe-4S | 24 November 2015 | [13] | |
| 5F0Q | 2.2 | p58(266–456) | 4Fe-4S | DNA:RNA, Mg | 28 November 2015 | [13] |
| 5F0S | 3.0 | p58(266–456) | 4Fe-4S | DNA:RNA, Mn | 28 November 2015 | [13] |
| 5IUD | 3.3 | p180(338–1255) | DNA:DNA | 17 March 2016 | [20] |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Baranovskiy, A.G.; Tahirov, T.H. Elaborated Action of the Human Primosome. Genes 2017, 8, 62. https://doi.org/10.3390/genes8020062
Baranovskiy AG, Tahirov TH. Elaborated Action of the Human Primosome. Genes. 2017; 8(2):62. https://doi.org/10.3390/genes8020062
Chicago/Turabian StyleBaranovskiy, Andrey G., and Tahir H. Tahirov. 2017. "Elaborated Action of the Human Primosome" Genes 8, no. 2: 62. https://doi.org/10.3390/genes8020062
APA StyleBaranovskiy, A. G., & Tahirov, T. H. (2017). Elaborated Action of the Human Primosome. Genes, 8(2), 62. https://doi.org/10.3390/genes8020062




