Characterization of the Transcriptome and Gene Expression of Tetraploid Black Locust Cuttings in Response to Etiolation
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Material and Etiolation Procedures
2.2. Plant Hormone Content Measurements
2.3. Analysis of Phenylene Oxide, Peroxidase and Indole Acetic Acid Oxidase
2.4. RNA Extraction and Deep Sequencing
2.5. Analysis of Illumina Transcriptome Sequencing Results
2.6. Differential Expression Analysis and Functional Enrichment
2.7. Statistical Analyses
3. Results
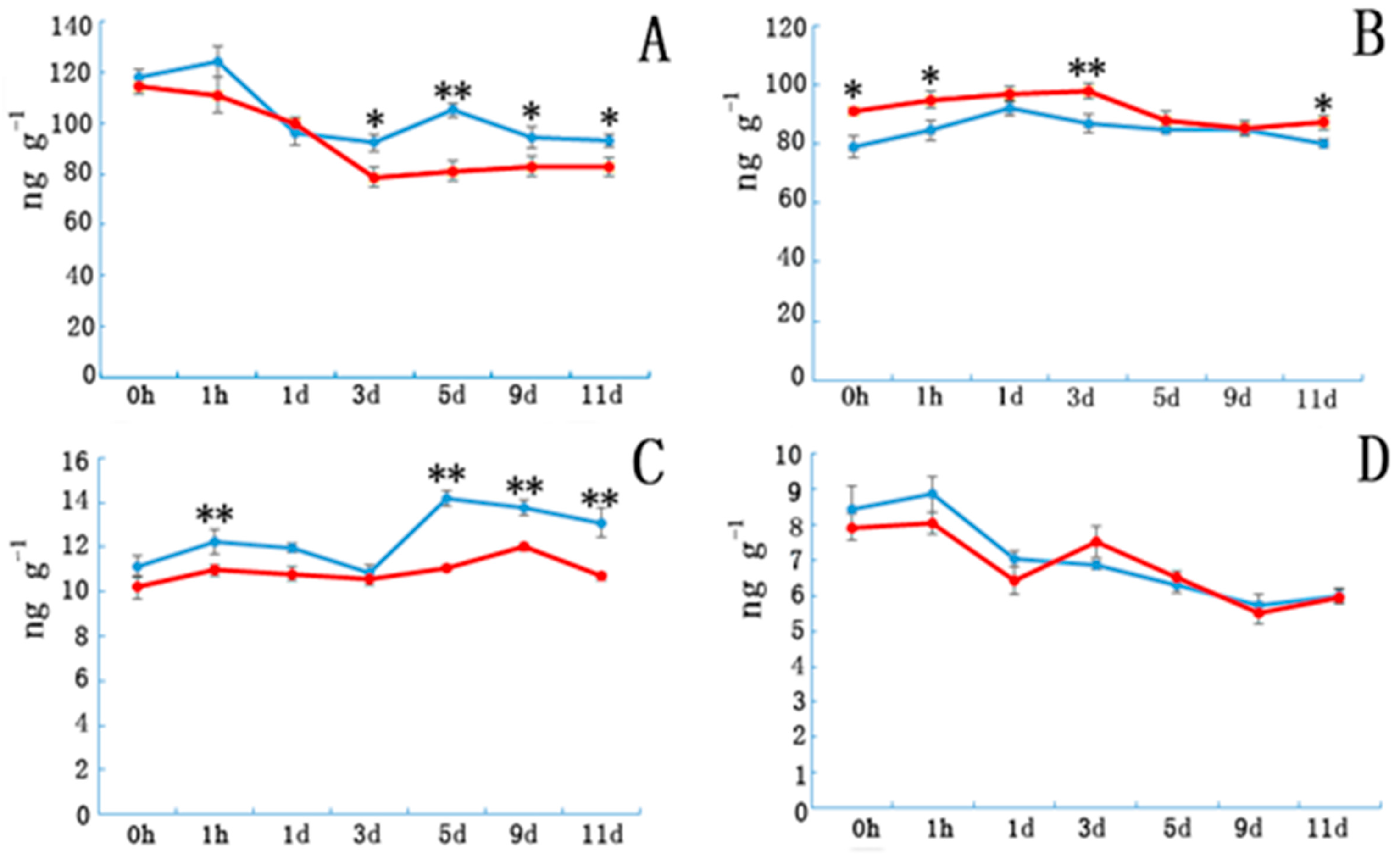
3.1. Indoleacetic Acid, Abscisic Acid, Gibberellin, and Zeatin Riboside Content
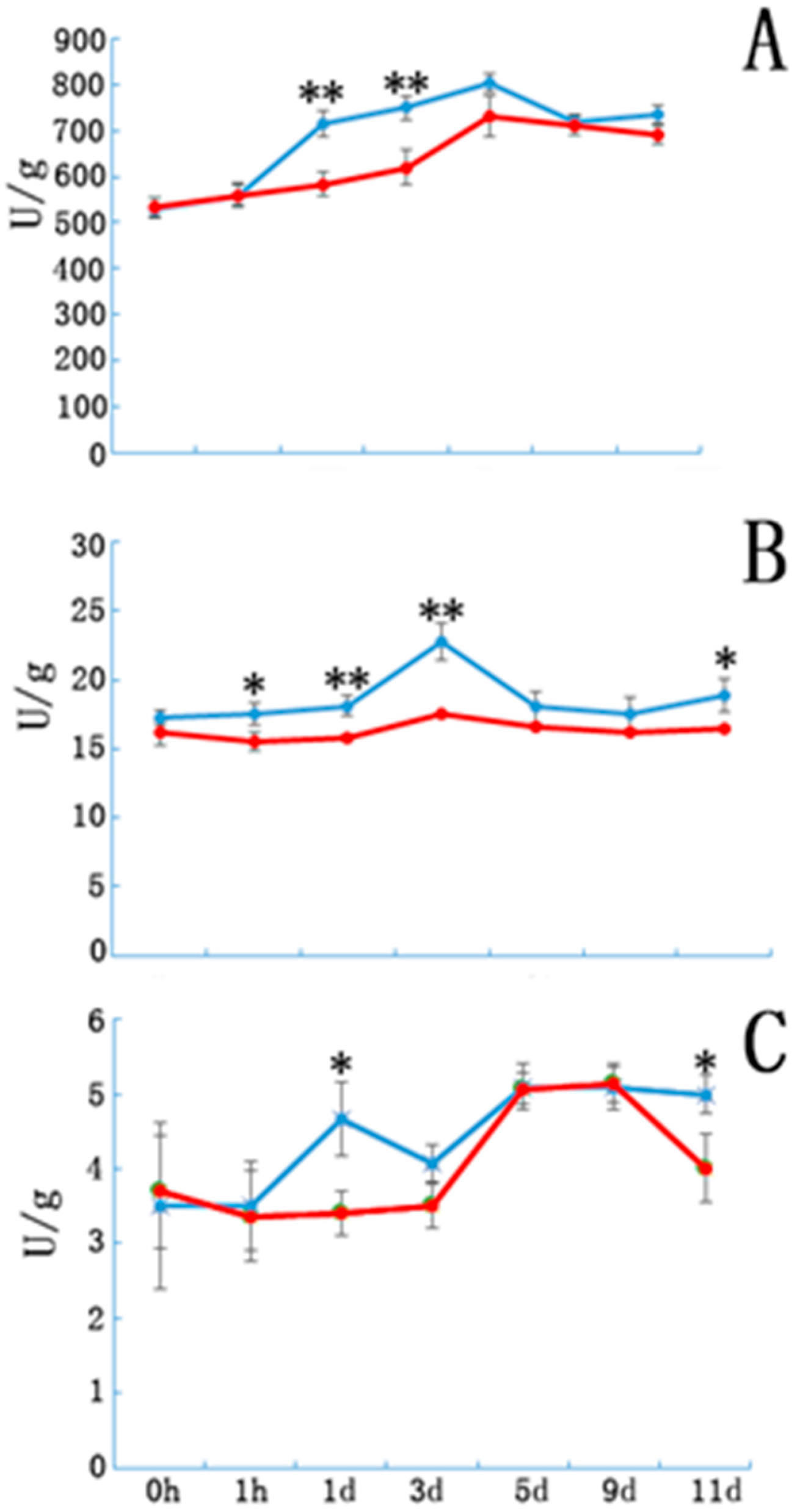
3.2. Peroxidase, Phenylene Oxide and Indole Acetic Acid Oxidase Activity
3.3. Deep Sequencing and de Novo Assembly
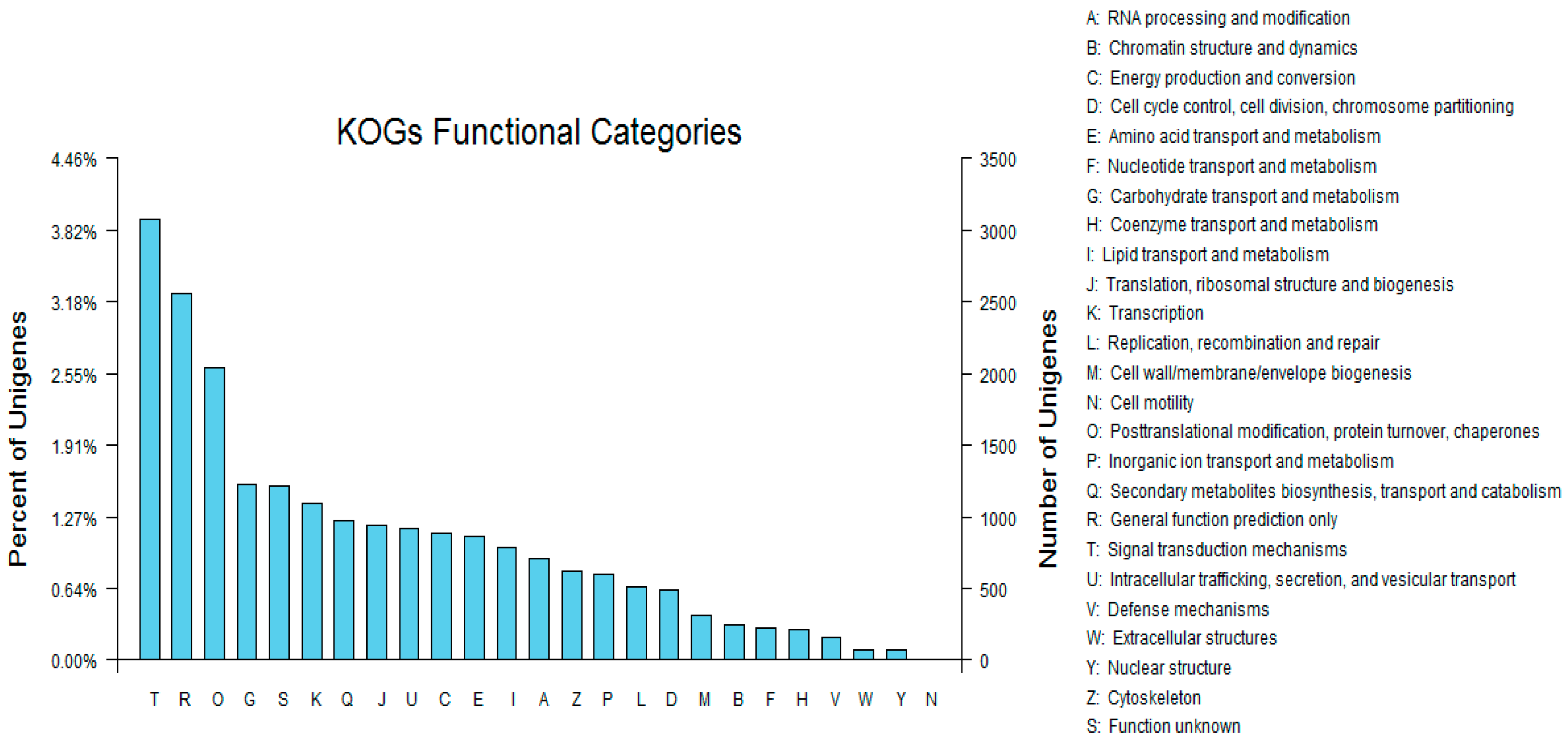
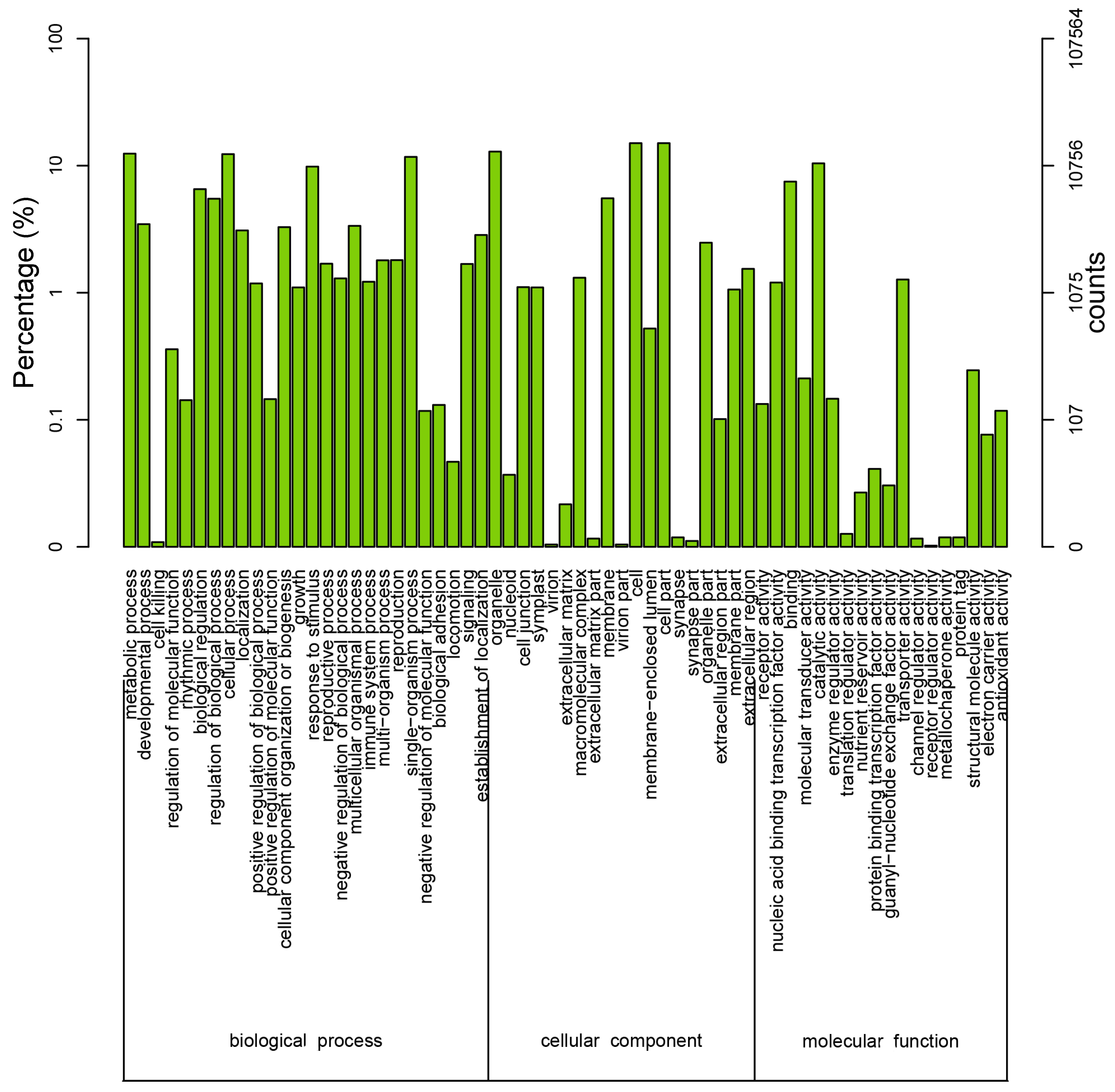
3.4. Sequence Alignment and Functional Annotation of the Transcriptome
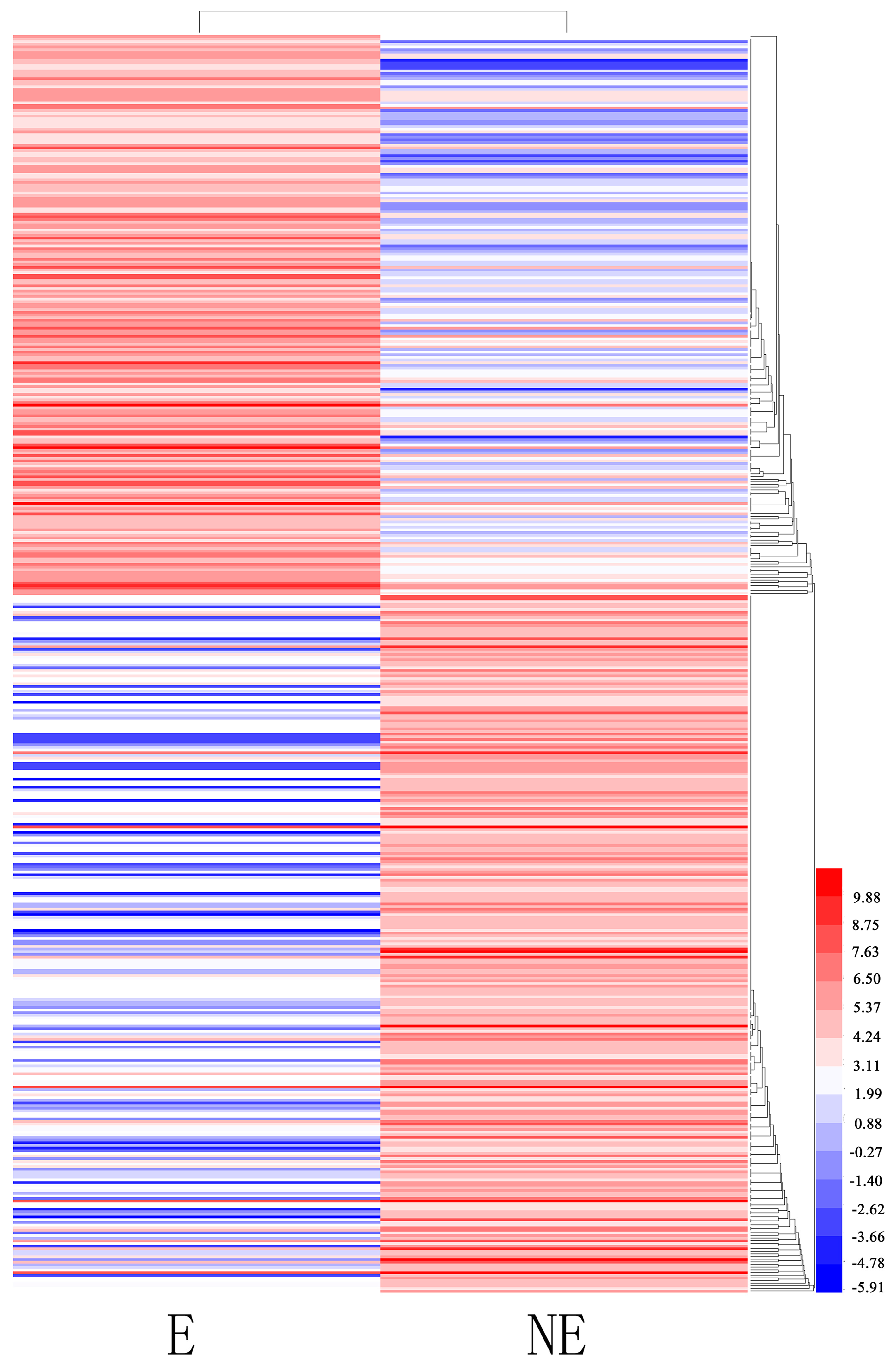
3.5. Important Differentially Expressed Genes Responses to Etiolation
4. Discussion
4.1. Gene Annotation and Enrichment Analysis of Differential Expressed Genes
4.2. Peroxidase and Polyphenol Oxidase Activity Phenylene oxide, Peroxidase and Indole Acetic Acid Oxidase
4.3. Differential Expressed Genes Related to Phytohormone Generation and Responses
4.4. Differential Expressed Genes Related to Starch and Sucrose Metabolism
4.5. Differentially Expressed Genes Related to Stress and Wound Responses
4.6. Other Transcripts with Highly Differential Expression
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Zhang, G.J.; Li, Y.; Li, F.P.; Xu, Z.H.; Sun, Y.H. Effects of root age on biomass and leaf nutrition in tetraploid Robinia pseudoacacia. J. Beijing For. Univ. 2009, 31, 37–41. [Google Scholar]
- Li, Y.; Tian, Y.T.; Qian, Y.Q.; Wang, S.Z. Effect of NAA and IBA on Rooting of tetraploid Robinia pseudoacacia in vitro and anatomical observation on adventitious root development. Sci. Silvae Sin. 2004, 40, 75–80. [Google Scholar]
- Meng, B.N.; Peng, Z.D.; Zhang, Z.L.; Xu, H.M.; Li, Y. Research on cuttage of tetraploid black locust (Robinia pseudoacacia L.) hardwood treated by low temperature sand storage and plant growth regulator. Heilongjiang Agric. Sci. 2010, 8, 85–88. [Google Scholar]
- Lu, N.; Meng, B.N.; Sun, Y.H.; Li, Y.; Wang, S.M.; Guo, Z.M.; Wang, Q.L.; Xu, H.M. Physiology and biochemistry during etiolated shoots cutting of tetraploid Robinia pseudoacacia. J. Northeast For. Univ. 2013, 41, 5–9. [Google Scholar]
- Frolich, E.F.; Platt, R.G. Use of the etiolation technique in rooting avocado cuttings. Calif. Avocado Soc. Yearb. 1972, 55, 97–109. [Google Scholar]
- Hausman, J.F. Changes in peroxidase activity, auxin level and ethylene production during root formation by poplar shoots raised in vitro. Plant Growth Regul. 1993, 13, 263–268. [Google Scholar] [CrossRef]
- Richards, M.R.; Rupp, L.A. Etiolation improves rooting of bigtooth maple cuttings. HortTechnology 2012, 22, 305–310. [Google Scholar]
- Batten, D.J.; Mullins, M.G. Ethylene and adventitious root formation in hypocotyl segments of etiolated mung-bean (Vigna radiata (L.) Wilczek) seedlings. Planta 1978, 138, 193–197. [Google Scholar] [CrossRef] [PubMed]
- Husen, A. Stock-plant etiolation causes drifts in total soluble sugars and anthraquinones, and promotes adventitious root formation in teak (Tectona grandis L. f.) coppice shoots. Plant Growth Regul. 2007, 54, 13–21. [Google Scholar] [CrossRef]
- Rasmussen, A.; Mason, M.G.; De Cuyper, C.; Brewer, P.B.; Herold, S.; Agusti, J.; Geelen, D.; Greb, T.; Goormachtig, S.; Beeckman, T.; et al. Strigolactones suppress adventitious rooting in Arabidopsis and pea. Plant Physiol. 2012, 158, 1976–1987. [Google Scholar] [CrossRef] [PubMed]
- Lu, N.; Xu, Z.; Meng, B.; Sun, Y.; Zhang, J.; Wang, S.; Li, Y. Proteomic analysis of etiolated juvenile tetraploid Robinia pseudoacacia branches during different cutting periods. Int. J. Mol. Sci. 2014, 15, 6674–6688. [Google Scholar] [CrossRef] [PubMed]
- Norman, S.M.; Poling, S.M.; Maier, V.P. An indirect enzyme-linked immunosorbent assay for (+)-abscisic acid in citrus, ricinus, and xanthium leaves. J. Agric. Food Chem. 1988, 36, 225–231. [Google Scholar] [CrossRef]
- Yang, J.; Zhang, J.; Wang, Z.; Zhu, Q.; Wang, W. Hormonal changes in the grains of rice subjected to water stress during grain filling. Plant Physiol. 2001, 127, 315–323. [Google Scholar] [CrossRef] [PubMed]
- Swiss-Prot Protein Database. Available online: http://www.expasy.ch/sprot (accessed on 20 April 2013).
- Gene Ontology Consortium. Available online: http://www.geneontology.org (accessed on 20 April 2013).
- Clusters of Orthologous Groups. Available online: http://www.ncbi.nlm.nih.gov/COG (accessed on 20 April 2013).
- Kyoto Encyclopaedia of Genes and Genomes Pathway. Available online: http://www.genome.jp/kegg (accessed on 20 April 2013).
- Zhang, Z.L. Plant Physiology Experiment Instruction; Higher Education: Beijing, China, 2003. [Google Scholar]
- Mortazavi, A.; Williams, B.A.; McCue, K.; Schaeffer, L.; Wold, B. Mapping and quantifying mammalian transcriptomes by RNA-Seq. Nat. Methods 2008, 5, 621–628. [Google Scholar] [CrossRef] [PubMed]
- Audic, S.; Claverie, J.M. The significance of digital gene expression profiles. Genome Res. 1997, 7, 986–995. [Google Scholar] [CrossRef] [PubMed]
- Todd, E.V.; Black, M.A.; Gemmell, N.J. The power and promise of RNA-seq in ecology and evolution. Mol. Ecol. 2016, 25, 1224–1241. [Google Scholar] [CrossRef] [PubMed]
- Frolich, E.F. Etiolation and the rooting of cuttings. In Proceedings of the 11th Annual Meeting Plant Propagators’ Society; 1961; pp. 277–283. [Google Scholar]
- Dai, W.; Su, Y.; Castillo, C.; Beslot, O. Plant regeneration from in vitro leaf tissues of Viburnum dentatum L. Plant Cell Tissue Organ Cult. 2011, 104, 257–262. [Google Scholar] [CrossRef]
- Herman, D.E.; Hess, C.E. The Effect of etiolation upon the rooting of cuttings. In Proceedings of the International Plant Propagators’ Society; 1963; Volume 13, pp. 42–62. [Google Scholar]
- Kariola, T. Chlorophyllase 1, a damage control enzyme, affects the balance between defense pathways in plants. Plant Cell Online 2005, 17, 282–294. [Google Scholar] [CrossRef] [PubMed]
- Weijers, D.; van Franke, D.M.; Vencken, R.J.; Quint, A.; Hooykaas, P.; Offringa, R. An Arabidopsis minute-like phenotype caused by a semi-dominant mutation in a RIBOSOMAL PROTEIN S5 gene. Development 2011, 128, 4289–4299. [Google Scholar]
- Francoz, E.; Ranocha, P.; Nguyen-Kim, H.; Jamet, E.; Burlat, V.; Dunand, C. Roles of cell wall peroxidases in plant development. Phytochemistry 2015, 112, 15–21. [Google Scholar] [CrossRef] [PubMed]
- Syros, T.; Yupsanis, T.; Zafiriadis, H.; Economou, A. Activity and isoforms of peroxidases, lignin and anatomy, during adventitious rooting in cuttings of Ebenus cretica L. J. Plant Physiol. 2004, 161, 69–77. [Google Scholar] [CrossRef] [PubMed]
- Rout, G.R.; Samantaray, S.; Das, P. In vitro rooting of Psoralea corylifolia Linn: Peroxidase activity as a marker. Plant Growth Regul. 2000, 30, 215–219. [Google Scholar] [CrossRef]
- Li, S.W.; Xue, L.; Xu, S.; Feng, H.; An, L. Hydrogen peroxide acts as a signal molecule in the adventitious root formation of mung bean seedlings. Environ. Exp. Bot. 2009, 65, 63–71. [Google Scholar] [CrossRef]
- Fahnenstich, H.; Scarpeci, T.E.; Valle, E.M.; Flügge, U.I.; Maurino, V.G. Generation of hydrogen peroxide in chloroplasts of Arabidopsis overexpressing glycolate oxidase as an inducible system to study oxidative stress. Plant Physiol. 2008, 148, 719–729. [Google Scholar] [CrossRef] [PubMed]
- León, J.; Rojo, E.; Sánchez-Serrano, J.J. Wound signalling in plants. J. Exp. Bot. 2001, 52, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Tani, T.; Sobajima, H.; Okada, K.; Chujo, T.; Arimura, S.; Tsutsumi, N.; Nishimura, M.; Seto, H.; Nojiri, H.; Yamane, H. Identification of the OsOPR7 gene encoding 12-oxophytodienoate reductase involved in the biosynthesis of jasmonic acid in rice. Planta 2008, 227, 517–526. [Google Scholar] [CrossRef] [PubMed]
- Vellosillo, T.; Martínez, M.; López, M.A.; Vicente, J.; Cascón, T.; Dolan, L.; Castresana, C. Oxylipins produced by the 9-lipoxygenase pathway in Arabidopsis regulate lateral root development and defense responses through a specific signaling cascade. Plant Cell 2007, 19, 831–846. [Google Scholar] [CrossRef] [PubMed]
- Da Costa, C.T.; de Almeida, M.R.; Ruedell, C.M.; Schwambach, J.; Maraschin, F.S.; Fett-Neto, A.G. When stress and development go hand in hand: Main hormonal controls of adventitious rooting in cuttings. Front. Plant Sci. 2013, 4, 33. [Google Scholar] [CrossRef] [PubMed]
- Mergemann, H.; Sauter, M. Ethylene induces epidermal cell death at the site of adventitious root emergence in rice. Plant Physiol. 2000, 124, 609–614. [Google Scholar] [CrossRef] [PubMed]
- Vriezen, W.H.; Hulzink, R.; Mariani, C.; Voesenek, L.A. 1-Aminocyclopropane-1-carboxylate oxidase activity limits ethylene biosynthesis in Rumex palustrisduring submergence. Plant Physiol. 1999, 121, 189–196. [Google Scholar] [CrossRef] [PubMed]
- Purvis, A.C.; Barmore, C.R. Involvement of ethylene in chlorophyll degradation in peel of citrus fruits. Plant Physiol. 1981, 68, 854–856. [Google Scholar] [CrossRef] [PubMed]
- Sitbon, F.; Hennion, S.; Sundberg, B.; Little, C.H.; Olsson, O.; Sandberg, G. Transgenic tobacco plants coexpressing the Agrobacterium tumefaciens iaaM and iaaH genes display altered growth and indoleacetic acid metabolism. Plant Physiol. 1992, 99, 1062–1069. [Google Scholar] [CrossRef] [PubMed]
- Boerjan, W.; Cervera, M.T.; Delarue, M.; Beeckman, T.; Dewitte, W.; Bellini, C.; Caboche, M.; Van Onckelen, H.; Van Montagu, M.; Inzé, D. Superroot, a recessive mutation in Arabidopsis, confers auxin overproduction. Plant Cell 1995, 7, 1405–1419. [Google Scholar] [CrossRef] [PubMed]
- Xiong, J.; Liang, J.; Chen, X.Y.; Li, W.; Li, H.; Liu, Y. Improving the rooting ability of Populus tomentosa by introduction of the rolB gene. J. Beijing For. Univ. 2005, 27, 54–58. [Google Scholar]
- Gutierrez, L.; Mongelard, G.; Floková, K.; Păcurar, D.I.; Novák, O.; Staswick, P.; Kowalczyk, M.; Păcurar, M.; Demailly, H.; Geiss, G.; et al. Auxin controls Arabidopsis adventitious root initiation by regulating jasmonic acid homeostasis. Plant Cell 2012, 24, 2515–2527. [Google Scholar] [CrossRef] [PubMed]
- Reed, R.C.; Brady, S.R.; Muday, G.K. Inhibition of auxin movement from the shoot into the root inhibits lateral root development in Arabidopsis. Plant Physiol. 1998, 118, 1369–1378. [Google Scholar] [CrossRef] [PubMed]
- Casimiro, I.; Marchant, A.; Bhalerao, R.P.; Beeckman, T.; Dhooge, S.; Swarup, R.; Graham, N.; Inzé, D.; Sandberg, G.; Casero, P.J.; et al. Auxin transport promotes Arabidopsis lateral root initiation. Plant Cell 2001, 13, 843–852. [Google Scholar] [CrossRef] [PubMed]
- Ludwig-Müller, J.; Vertocnik, A.; Town, C.D. Analysis of indole-3-butyric acid-induced adventitious root formation on Arabidopsis stem segments. J. Exp. Bot. 2005, 56, 2095–2105. [Google Scholar] [CrossRef] [PubMed]
- Komeda, Y.; Burtin, D.; Hanzawa, Y.; Long, D.; Takahashi, T.; Pineiro, M.; Coupland, G.; Michael, A.J. ACAULIS5, an Arabidopsis gene required for stem elongation, encodes a spermine synthase. EMBO J. 2000, 19, 4248–4256. [Google Scholar]
- Muñiz, L.; Minguet, E.G.; Singh, S.K.; Pesquet, E.; Vera-Sirera, F.; Moreau-Courtois, C.L.; Carbonell, J.; Blázquez, M.A.; Tuominen, H. ACAULIS5 controls Arabidopsis xylem specification through the prevention of premature cell death. Development 2008, 135, 2573–2582. [Google Scholar] [CrossRef] [PubMed]
- Vera-Sirera, F.; Minguet, E.G.; Singh, S.K.; Ljung, K.; Tuominen, H.; Blázquez, M.A.; Carbonell, J. Role of polyamines in plant vascular development. Plant Physiol. Biochem. 2010, 48, 534–539. [Google Scholar] [CrossRef] [PubMed]
- Moubayidin, L.; Perilli, S.; Ioio, R.D.; Di Mambro, R.; Costantino, P.; Sabatini, S. The rate of cell differentiation controls the Arabidopsis root meristem growth phase. Curr. Biol. 2010, 20, 1138–1143. [Google Scholar] [CrossRef] [PubMed]
- Meng, L.; Buchanan, B.B.; Feldman, L.J.; Luan, S. CLE-like (CLEL) peptides control the pattern of root growth and lateral root development in Arabidopsis. Proc. Natl. Acad. Sci. USA 2012, 109, 1760–1765. [Google Scholar] [CrossRef] [PubMed]
- Cluis, C.P.; Mouchel, C.F.; Hardtke, C.S. The Arabidopsis transcription factor HY5 integrates light and hormone signaling pathways. Plant J. 2004, 38, 332–347. [Google Scholar] [CrossRef] [PubMed]
- Malinova, I.; Kunz, H.H.; Alseekh, S.; Herbst, K.; Fernie, A.R.; Gierth, M.; Fettke, J. Reduction of the cytosolic phosphoglucomutase in Arabidopsis reveals impact on plant growth, seed and root development, and carbohydrate partitioning. PLoS ONE 2014, 9, e112468. [Google Scholar] [CrossRef] [PubMed]
- Niu, S.; Li, Z.; Yuan, H.; Fang, P.; Chen, X.; Li, W. Proper gibberellin localization in vascular tissue is required to regulate adventitious root development in tobacco. J. Exp. Bot. 2013, 64, 3411–3424. [Google Scholar] [CrossRef] [PubMed]
- Sun, T.; Gubler, F. Molecular mechanism of gibberellin signaling in plants. Annu. Rev. Plant Biol. 2004, 55, 197–223. [Google Scholar] [CrossRef] [PubMed]
- Wiesman, Z.; Lavee, S. Relationship of carbohydrate sources and indole-3-butyric acid in olive cuttings. Funct. Plant Biol. 1995, 22, 811–816. [Google Scholar] [CrossRef]
- OuYang, F.; Wang, J.; Li, Y. Effects of cutting size and exogenous hormone treatment on rooting of shoot cuttings in Norway spruce [Picea abies (L.) Karst.]. New For. 2015, 46, 91–105. [Google Scholar] [CrossRef]
- Thalmann, M.R.; Pazmino, D.; Seung, D.; Horrer, D.; Nigro, A.; Meier, T.; Kölling, K.; Pfeifhofer, H.; Zeeman, S.; Santelia, D. Regulation of leaf starch degradation by abscisic acid is important for osmotic stress tolerance in plants. Plant Cell 2016, 28, 1860–1878. [Google Scholar] [CrossRef] [PubMed]
- Gómez, L.D.; Baud, S.; Gilday, A.; Li, Y.; Graham, I.A. Delayed embryo development in the Arabidopsis TREHALOSE-6-PHOSPHATE SYNTHASE 1 mutant is associated with altered cell wall structure, decreased cell division and starch accumulation. Plant J. 2006, 46, 69–84. [Google Scholar] [CrossRef] [PubMed]
- Haruta, M.; Major, I.T.; Christopher, M.E.; Patton, J.J.; Constabel, C.P. A Kunitz trypsin inhibitor gene family from trembling aspen (Populus tremuloides Michx.): Cloning, functional expression, and induction by wounding and herbivory. Plant Mol. Biol. 2001, 46, 347–359. [Google Scholar] [CrossRef] [PubMed]
- Christopher, M.E.; Miranda, M.; Major, I.T.; Constabel, C.P. Gene expression profiling of systemically wound-induced defenses in hybrid poplar. Planta 2004, 219, 936–947. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Zhang, Y.; Bian, X.; Lei, J.; Sun, J.; Guo, N.; Gai, J.; Xing, H. A comparative proteomics analysis of soybean leaves under biotic and abiotic treatments. Mol. Biol. Rep. 2013, 40, 1553–1562. [Google Scholar] [CrossRef] [PubMed]
- Irshad, M.; Canut, H.; Borderies, G.; Pont-Lezica, R.; Jamet, E. A new picture of cell wall protein dynamics in elongating cells of Arabidopsis thaliana: Confirmed actors and newcomers. BMC Plant Biol. 2008, 8, 1. [Google Scholar] [CrossRef] [PubMed]
- Kwon, H.K.; Yokoyama, R.; Nishitani, K. A proteomic approach to apoplastic proteins involved in cell wall regeneration in protoplasts of Arabidopsis suspension-cultured cells. Plant Cell Physiol. 2005, 46, 843–857. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.; Hrmova, M.; Wan, C.H.; Wu, C.; Balzen, J.; Cai, W.; Wang, J.; Densmore, L.D.; Fincher, G.B.; Zhang, H.; et al. Members of a new group of chitinase-like genes are expressed preferentially in cotton cells with secondary walls. Plant Mol. Biol. 2004, 54, 353–372. [Google Scholar] [CrossRef] [PubMed]
- Hossain, M.A.; Noh, H.N.; Kim, K.I.; Koh, E.J.; Wi, S.G.; Bae, H.J.; Lee, H.J.; Hong, S.W. Mutation of the chitinase-like protein-encoding AtCTL2 gene enhances lignin accumulation in dark-grown Arabidopsis seedlings. Plant Physiol. 2010, 167, 650–658. [Google Scholar] [CrossRef] [PubMed]
- Hong, Z.; Lakkineni, K.; Zhang, Z.; Verma, D.P.S. Removal of feedback inhibition of Δ1-pyrroline-5-carboxylate synthetase results in increased proline accumulation and protection of plants from osmotic stress. Plant Physiol. 2000, 122, 1129–1136. [Google Scholar] [CrossRef] [PubMed]
- Lehmann, S.; Funck, D.; Szabados, L.; Rentsch, D. Proline metabolism and transport in plant development. Amino Acids 2010, 39, 949–962. [Google Scholar] [CrossRef] [PubMed]
- López-Carrión, A.I.; Castellano, R.; Rosales, M.A.; Ruiz, J.M.; Romero, L. Role of nitric oxide under saline stress: Implications on proline metabolism. Biol. Plant. 2008, 52, 587–591. [Google Scholar] [CrossRef]
- Funck, D.; Eckard, S.; Muller, G. Non-redundant functions of two proline dehydrogenase isoforms in Arabidopsis. BMC Plant Biol. 2010, 10, 70. [Google Scholar] [CrossRef] [PubMed]
- Schwambach, J.; Fadanelli, C.; Fett-Neto, A.G. Mineral nutrition and adventitious rooting in microcuttings of Eucalyptus globulus. Tree Physiol. 2005, 25, 487–494. [Google Scholar] [CrossRef] [PubMed]
- Zerche, S.; Haensch, K.; Druege, U.; Hajirezaei, M. Nitrogen remobilisation facilitates adventitious root formation on reversible dark-induced carbohydrate depletion in Petunia hybrida. BMC Plant Biol. 2016, 16, 219. [Google Scholar] [CrossRef] [PubMed]
- Zerche, S.; Druege, U. Nitrogen content determines adventitious rooting in Euphorbia pulcherrima under adequate light independently of pre-rooting carbohydrate depletion of cuttings. Sci. Horticult. 2009, 121, 340–347. [Google Scholar] [CrossRef]
- Fernandez, A.; Drozdzecki, A.; Hoogewijs, K.; Nguyen, A.; Beeckman, T.; Madder, A.; Hilson, P. Transcriptional and functional classification of the GOLVEN/ROOT GROWTH FACTOR/CLE-like signaling peptides reveals their role in lateral root and hair formation. Plant Physiol. 2013, 161, 954–970. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Wu, F.; Shi, J.; Zhu, Y.; Zhu, Z.; Gong, Q.; Hu, J. ROOT HAIR DEFECTIVE3 family of dynamin-like GTPases mediates homotypic endoplasmic reticulum fusion and is essential for Arabidopsis development. Plant Physiol. 2013, 163, 713–720. [Google Scholar] [CrossRef] [PubMed]
- Belda-Palazón, B.; Almendáriz, C.; Martí, E.; Carbonell, J.; Ferrando, A. Relevance of the axis spermidine/eIF5A for plant growth and development. Front. Plant Sci. 2016, 7, 245. [Google Scholar]
- Toscano-Morales, R.; Xoconostle-Cázares, B.; Martínez-Navarro, A.C.; Ruiz-Medrano, R. AtTCTP2 mRNA and protein movement correlates with formation of adventitious roots in tobacco. Plant Signal. Behav. 2016, 11, e1071003. [Google Scholar] [CrossRef] [PubMed]
- Roger, R.B. Physiology of vegetative reproduction. Agrofor. Novel Crops Unit Sch. Trop. Biol. 2004, 1655–1668. [Google Scholar] [CrossRef]
- Baucher, M.; Bernard-Vailhe, M.A.; Chabbert, B.; Besle, J.M.; Opsomer, C.; Van Montagu, M.; Botterman, J. Down-regulation of cinnamyl alcohol dehydrogenase in transgenic alfalfa (Medicago sativa L.) and the effect on lignin composition and digestibility. Plant Mol. Biol. 1999, 39, 437–447. [Google Scholar] [CrossRef] [PubMed]
- Quan, J.E.; Zhang, C.X.; Zhang, X.P.; Zhu, H.L.; Zhang, S.; Zhao, Z. Effect of cuttings with different lingin contents on cutting rooting of tetraploid Robinia pseudoacacia. Acta Bot. Boreal. 2014, 34, 1179–1186. [Google Scholar]

© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lu, N.; Dai, L.; Luo, Z.; Wang, S.; Wen, Y.; Duan, H.; Hou, R.; Sun, Y.; Li, Y. Characterization of the Transcriptome and Gene Expression of Tetraploid Black Locust Cuttings in Response to Etiolation. Genes 2017, 8, 345. https://doi.org/10.3390/genes8120345
Lu N, Dai L, Luo Z, Wang S, Wen Y, Duan H, Hou R, Sun Y, Li Y. Characterization of the Transcriptome and Gene Expression of Tetraploid Black Locust Cuttings in Response to Etiolation. Genes. 2017; 8(12):345. https://doi.org/10.3390/genes8120345
Chicago/Turabian StyleLu, Nan, Li Dai, Zijing Luo, Shaoming Wang, Yanzhong Wen, Hongjing Duan, Rongxuan Hou, Yuhan Sun, and Yun Li. 2017. "Characterization of the Transcriptome and Gene Expression of Tetraploid Black Locust Cuttings in Response to Etiolation" Genes 8, no. 12: 345. https://doi.org/10.3390/genes8120345
APA StyleLu, N., Dai, L., Luo, Z., Wang, S., Wen, Y., Duan, H., Hou, R., Sun, Y., & Li, Y. (2017). Characterization of the Transcriptome and Gene Expression of Tetraploid Black Locust Cuttings in Response to Etiolation. Genes, 8(12), 345. https://doi.org/10.3390/genes8120345




