Air Quality Effects on Human Health and Approaches for Its Assessment through Microfluidic Chips
Abstract
1. Introduction
2. The Air We Breathe
3. Air Pollution Sources
3.1. Indoor Air Pollution Origins and Prevention
3.1.1. Gaseous Pollutants
3.1.2. Organic Compounds
3.1.3. Asbestos
3.1.4. Biological Pollution Sources
3.2. Outdoor Air Pollution
3.2.1. Natural (Non-Anthropogenic) Pollution Sources
3.2.2. Human-Made (Anthropogenic) Pollution Sources
3.2.3. Aerosols and Particulate Matter
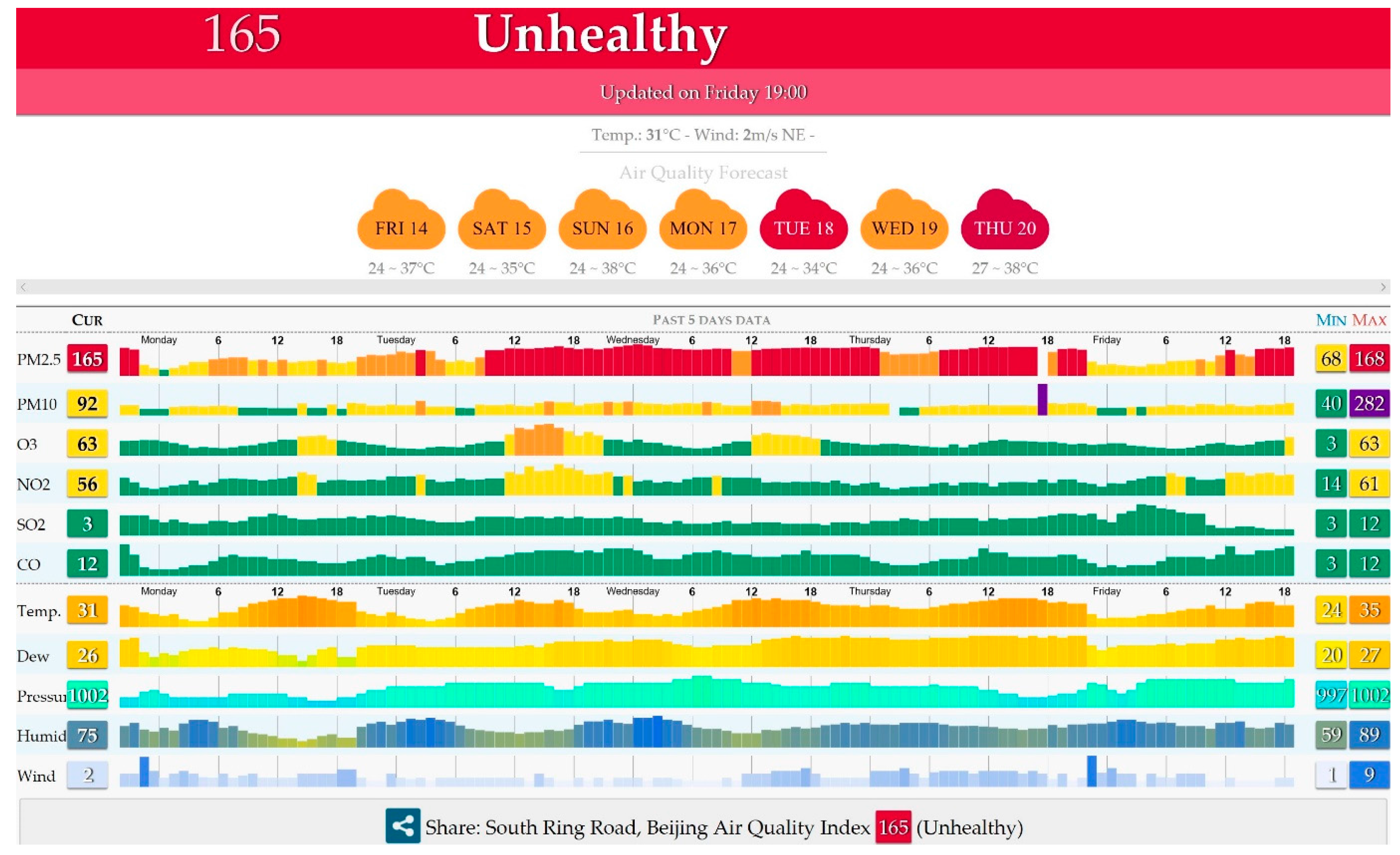
4. Emerging On-Chip Technologies for the Air Quality Measurements
5. OoC for Assessing Health Effects
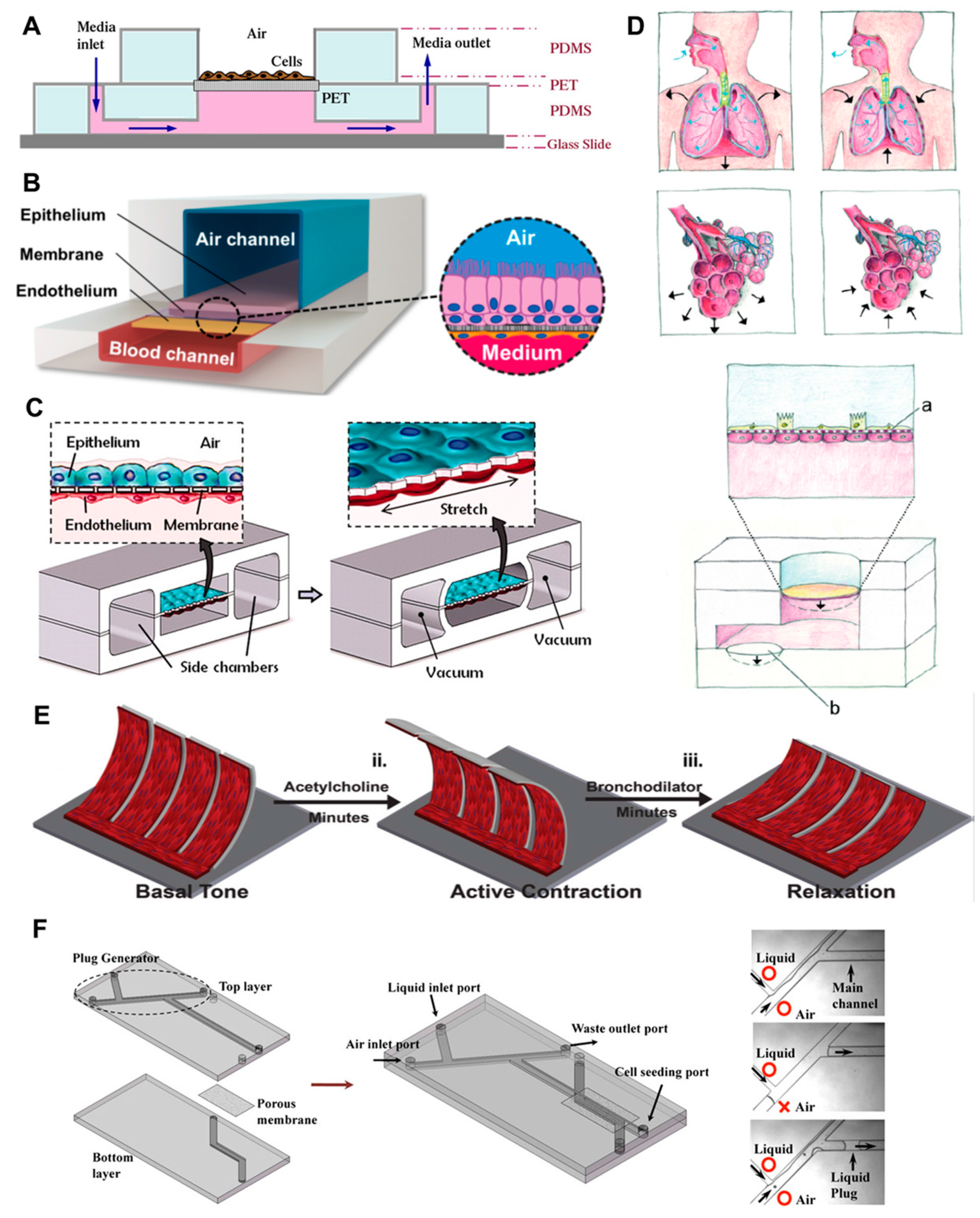
5.1. OoC Technology for Modeling Healthy and Pathological Lung Function
5.2. OoC Technology for Modeling Lung Pathologies and Pollutant Exposure
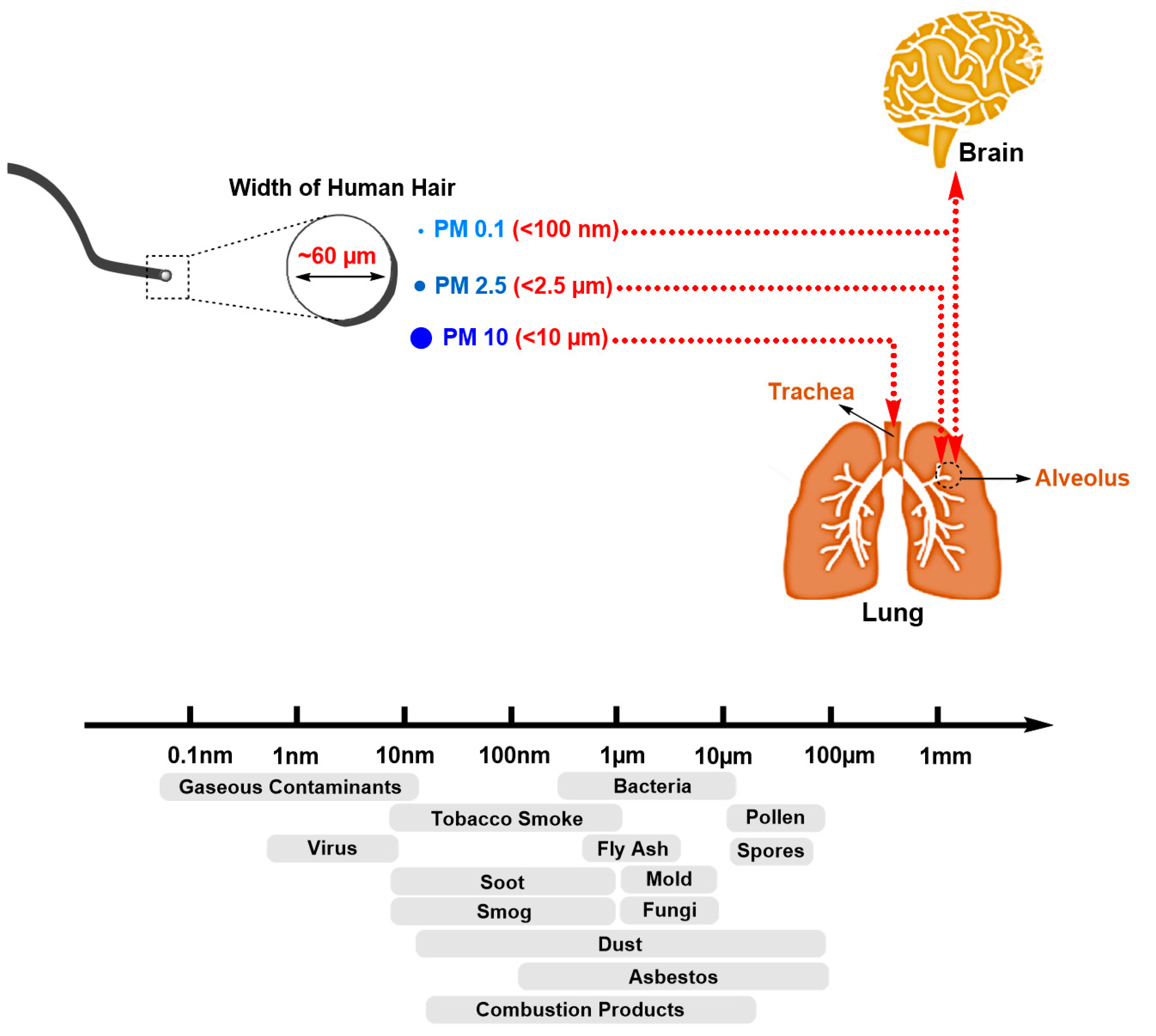
5.3. PM2.5 Particles Penetrate Deep into Tissues
5.4. Cancer Types Caused by PM
5.5. Pharmaceutical Drugs to Treat Lung Cancer
6. Summary
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Kane, A.B.; Hurt, R.H. Nanotoxicology: The asbestos analogy revisited. Nat. Nanotechnol. 2008, 3, 378–379. [Google Scholar] [CrossRef] [PubMed]
- Qu, H. CMOS MEMS Fabrication Technologies and Devices. Micromachines 2016, 7, 14. [Google Scholar] [CrossRef]
- Bhatia, S.N.; Ingber, D.E. Microfluidic organs-on-chips. Nat. Biotechnol. 2014, 32, 760–772. [Google Scholar] [CrossRef] [PubMed]
- Hajati, A.; Latev, D.; Gardner, D.; Hajati, A.; Imai, D.; Torrey, M.; Schoeppler, M. Three-dimensional micro electromechanical system piezoelectric ultrasound transducer. Appl. Phys. Lett. 2012, 101, 253101. [Google Scholar] [CrossRef]
- Hajati, A.; Latev, D.; Gardner, D.; Ottosson, M.; Imai, D.; Torrey, M.; Schoeppler, M. Monolithic ultrasonic integrated circuits based on micromachined semi-ellipsoidal piezoelectric domes. Appl Phys. Lett 2013, 103, 202906. [Google Scholar] [CrossRef]
- Louizos, L.A.; Athanasopoulos, P.G.; Varty, K. Microelectromechanical systems and nanotechnology: A platform for the next stent technological era. Vasc. Endovascular. Surg. 2012, 46, 605–609. [Google Scholar] [CrossRef] [PubMed]
- Nalayanda, D.D.; Puleo, C.; Fulton, W.B.; Sharpe, L.M.; Wang, T.H.; Abdullah, F. An open-access microfluidic model for lung-specific functional studies at an air-liquid interface. Biomed. Microdevices 2009, 11, 1081–1089. [Google Scholar] [CrossRef] [PubMed]
- Zhu, L.; Jacob, D.J.; Keutsch, F.N.; Mickley, L.J.; Scheffe, R.; Strum, M.; Abad, G.G.; Chance, K.; Yang, K.; Rappengluck, B.; et al. Formaldehyde (HCHO) As a Hazardous Air Pollutant: Mapping Surface Air Concentrations from Satellite and Inferring Cancer Risks in the United States. Environ. Sci. Technol. 2017, 51, 5650–5657. [Google Scholar] [CrossRef] [PubMed]
- Fiedziuszko, S.J. Applications of MEMS in communication satellites. In Proceedings of the 13th International Conference on Microwaves, Radar and Wireless Communications, MIKON-2000, Conference Proceedings (IEEE Cat. No.00EX428), Wrocław, Poland, 22–24 May 2000; pp. 201–211. [Google Scholar]
- Kong, L.; Xin, J.; Zhang, W.; Wang, Y. The empirical correlations between PM2.5, PM10 and AOD in the Beijing metropolitan region and the PM2.5, PM10 distributions retrieved by MODIS. Environ. Pollut. 2016, 216, 350–360. [Google Scholar] [CrossRef] [PubMed]
- World Air Quality Index Team. The World Air Quality Index Project. Available online: http://aqicn.org/contact/ (accessed on 25 September 2017).
- Castell, N.; Dauge, F.R.; Schneider, P.; Vogt, M.; Lerner, U.; Fishbain, B.; Broday, D.; Bartonova, A. Can commercial low-cost sensor platforms contribute to air quality monitoring and exposure estimates? Environ. Int. 2017, 99, 293–302. [Google Scholar] [CrossRef] [PubMed]
- Knight, A. Systematic reviews of animal experiments demonstrate poor human clinical and toxicological utility. Altern. Lab. Anim. 2007, 35, 641–659. [Google Scholar] [CrossRef] [PubMed]
- Akhtar, A. The flaws and human harms of animal experimentation. Camb. Q. Healthc. Ethics 2015, 24, 407–419. [Google Scholar] [CrossRef] [PubMed]
- Zheng, F.; Fu, F.; Cheng, Y.; Wang, C.; Zhao, Y.; Gu, Z. Organ-on-a-Chip Systems: Microengineering to Biomimic Living Systems. Small 2016, 12, 2253–2282. [Google Scholar] [CrossRef] [PubMed]
- Capulli, A.K.; Tian, K.; Mehandru, N.; Bukhta, A.; Choudhury, S.F.; Suchyta, M.; Parker, K.K. Approaching the in vitro clinical trial: Engineering organs on chips. Lab Chip 2014, 14, 3181–3186. [Google Scholar] [CrossRef] [PubMed]
- Konar, D.; Devarasetty, M.; Yildiz, D.V.; Atala, A.; Murphy, S.V. Lung-On-A-Chip Technologies for Disease Modeling and Drug Development. Biomed. Eng. Comput. Biol. 2016, 7, 17–27. [Google Scholar] [CrossRef] [PubMed]
- Benam, K.H.; Novak, R.; Nawroth, J.; Hirano-Kobayashi, M.; Ferrante, T.C.; Choe, Y.; Prantil-Baun, R.; Weaver, J.C.; Bahinski, A.; Parker, K.K.; et al. Matched-Comparative Modeling of Normal and Diseased Human Airway Responses Using a Microengineered Breathing Lung Chip. Cell Syst. 2016, 3, 456–466. [Google Scholar] [CrossRef] [PubMed]
- Holland, H.D. The oxygenation of the atmosphere and oceans. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2006, 361, 903–915. [Google Scholar] [CrossRef] [PubMed]
- Berner, R.A. Atmospheric oxygen over Phanerozoic time. Proc. Natl. Acad. Sci. USA 1999, 96, 10955–10957. [Google Scholar] [CrossRef] [PubMed]
- Ward, P.D. Out of Thin Air: Dinosaurs, Birds, and Earth’s Ancient Atmosphere; The National Academies Press: Washington, DC, USA, 2006; p. 296. [Google Scholar] [CrossRef]
- Crowe, S.A.; Dossing, L.N.; Beukes, N.J.; Bau, M.; Kruger, S.J.; Frei, R.; Canfield, D.E. Atmospheric oxygenation three billion years ago. Nature 2013, 501, 535–538. [Google Scholar] [CrossRef] [PubMed]
- Fischer, W.W. Breathing Life into Oxygen. Science 2014, 343, 840. [Google Scholar] [CrossRef]
- Chen, Y.; Mills, S.; Street, J.; Golan, D.; Post, A.; Jacobson, M.; Paytan, A. Estimates of atmospheric dry deposition and associated input of nutrients to Gulf of Aqaba seawater. J. Geophys. Res. 2007, 112, D04309. [Google Scholar] [CrossRef]
- Walsh, J.J.; Steidinger, K.A. Saharan dust and Florida red tides: The cyanophyte connection. J. Geophys. Res. Oceans 2001, 106, 11597–11612. [Google Scholar] [CrossRef]
- Jish Prakash, P.; Stenchikov, G.; Kalenderski, S.; Osipov, S.; Bangalath, H. The impact of dust storms on the Arabian Peninsula and the Red Sea. Atmos. Chem. Phys. 2015, 15, 199–222. [Google Scholar] [CrossRef]
- Klepeis, N.E.; Nelson, W.C.; Ott, W.R.; Robinson, J.P.; Tsang, A.M.; Switzer, P.; Behar, J.V.; Hern, S.C.; Engelmann, W.H. The National Human Activity Pattern Survey (NHAPS): A resource for assessing exposure to environmental pollutants. J. Expo. Anal. Environ. Epidemiol. 2001, 11, 231–252. [Google Scholar] [CrossRef] [PubMed]
- Ezzati, M. Indoor air pollution and health in developing countries. Lancet 2005, 366, 104–106. [Google Scholar] [CrossRef]
- Guan, W.J.; Zheng, X.Y.; Chung, K.F.; Zhong, N.S. Impact of air pollution on the burden of chronic respiratory diseases in China: Time for urgent action. Lancet 2016, 388, 1939–1951. [Google Scholar] [CrossRef]
- The, L. Improving air quality starts at home. Lancet 2014, 384, 1821. [Google Scholar] [CrossRef]
- Duflo, E.; Greenstone, M.; Hanna, R. Indoor air pollution, health and economic well-being. SAPIENS [Online] 2008, 1. [Google Scholar] [CrossRef]
- Bruce, N.; Perez-Padilla, R.; Albalak, R. Indoor air pollution in developing countries: A major environmental and public health challenge. Bull. World Health Organ. 2000, 78, 1078–1092. [Google Scholar] [PubMed]
- Kilpatrick, K. Sick Classrooms Caused by Rising CO2 Levels. Available online: http://energyalliancegroup.org/sick-classrooms-require-energy-efficient-solutions-2/ (accessed on 25 September 2017).
- The National Institute for Occupational Safety and Health (NIOSH). Indoor Environmental Quality. Available online: https://www.cdc.gov/niosh/topics/indoorenv/BuildingVentilation.html (accessed on 25 September 2017).
- (EPD), T.E.B.a.t.E.P.D. Hong Kong Government Initiatives to Improve Indoor Air Quality. Available online: https://web.archive.org/web/20160108175015/http:/zcada.net/hk-government-initiatives-improve-iaq/ (accessed on 25 September 2017).
- Arc Suppression Technologies. Environmental Impact—Of Arc Suppression, Lab. Note #106; Arc Suppression Technologies: Bloomington, IN, USA, 2013.
- United States Environmental Protection Agency (EPA). Ozone Generators that Are Sold as Air Cleaners. Available online: https://www.epa.gov/indoor-air-quality-iaq/ozone-generators-are-sold-air-cleaners (accessed on 25 September 2017).
- Apte, M.G.; Buchanan, I.S.; Mendell, M.J. Outdoor ozone and building-related symptoms in the BASE study. Indoor Air 2008, 18, 156–170. [Google Scholar] [CrossRef] [PubMed]
- Talk of the Nation. Study: Bad In-Flight Air Exacerbated by Passengers; Research News: Beijing, China, 2007. [Google Scholar]
- Kusky, T.M. Geological Hazards: A Sourcebook; Greenwood Publishing Group: London, UK, 2003. [Google Scholar]
- Godish, T. Indoor Environment Quality; CRC Press: Boca Raton, FL, USA, 2000. [Google Scholar]
- Ubysz, A.; Maj, M.; Musiał, M.; Ubysz, J. Radon—Occurrence and Health Risks in Civil Engineering. Proc. Eng. 2017, 172, 1184–1189. [Google Scholar] [CrossRef]
- United States Environmental Protection Agency. A Citizen’s Guide to Radon: The Guide to Protecting Yourself and Your Family from Radon. Available online: https://www.epa.gov/radon/citizens-guide-radon-guide-protecting-yourself-and-your-family-radon (accessed on 25 September 2017).
- WHO. Radon and Health, Fact Sheet. Available online: http://www.who.int/mediacentre/factsheets/fs291/en/ (accessed on 25 September 2017).
- American Cancer Society (ACS). Known and Probable Human Carcinogens. Available online: https://www.cancer.org/cancer/cancer-causes/general-info/known-and-probable-human-carcinogens.html#additional_resources (accessed on 25 September 2017).
- Roscoe, R.J. Lung Cancer Mortality Among Nonsmoking Uranium Miners Exposed to Radon Daughters. JAMA J. Am. Med. Assoc. 1989, 262, 629–633. [Google Scholar] [CrossRef]
- Tirmarche, M.; Laurier, D.; Mitton, N.; Gelas, J. Lung cancer risk associated with low chronic radon exposure. Results from the French uranium miners cohort and the European project. In Proceedings of the 10th International Congress of the International Radiation Protection/Association on Harmonization of Radiation, Human Life and the Ecosystem, Tokyo, Japan, 14–19 May 2000. [Google Scholar]
- Darby, S.; Hill, D.; Doll, R. Radon: A likely carcinogen at all exposures. Ann. Oncol. 2001, 12, 1341–1351. [Google Scholar] [CrossRef] [PubMed]
- Catelinois, O.; Rogel, A.; Laurier, D.; Billon, S.; Hemon, D.; Verger, P.; Tirmarche, M. Lung cancer attributable to indoor radon exposure in france: Impact of the risk models and uncertainty analysis. Environ. Health Perspect. 2006, 114, 1361–1366. [Google Scholar] [CrossRef] [PubMed]
- Darby, S.; Hill, D.; Auvinen, A.; Barros-Dios, J.M.; Baysson, H.; Bochicchio, F.; Deo, H.; Falk, R.; Forastiere, F.; Hakama, M.; et al. Radon in homes and risk of lung cancer: Collaborative analysis of individual data from 13 European case-control studies. BMJ 2005, 330, 223. [Google Scholar] [CrossRef] [PubMed]
- Rericha, V.; Kulich, M.; Rericha, R.; Shore, D.L.; Sandler, D.P. Incidence of leukemia, lymphoma, and multiple myeloma in Czech uranium miners: A case-cohort study. Environ. Health Perspect. 2006, 114, 818–822. [Google Scholar] [CrossRef] [PubMed]
- Wolkoff, P.; Kjaergaard, S.K. The dichotomy of relative humidity on indoor air quality. Environ. Int. 2007, 33, 850–857. [Google Scholar] [CrossRef] [PubMed]
- Cosgrove-Mather, B. FDA: Too Much Benzene in Some Drinks. Available online: http://www.cbsnews.com/news/fda-too-much-benzene-in-some-drinks/ (accessed on 25 September 2017).
- Fan, A.M. Encyclopedia of Toxicology, “Chloroform”, 2nd ed.; Wexler, P., Ed.; Elsevier: Amsterdam, The Netherlands, 2005. [Google Scholar]
- United States Environmental Protection Agency. Organic Chemicals, Plastics and Synthetic Fibers Effluent Guidelines. Available online: https://www.epa.gov/eg/organic-chemicals-plastics-and-synthetic-fibers-effluent-guidelines (accessed on 25 September 2017).
- Meruva, N.K.; Penn, J.M.; Farthing, D.E. Rapid identification of microbial VOCs from tobacco molds using closed-loop stripping and gas chromatography/time-of-flight mass spectrometry. J. Ind. Microbiol. Biotechnol. 2004, 31, 482–488. [Google Scholar] [CrossRef] [PubMed]
- Bioidea.net, a.B.C. Smelly, Musty, Moldy Houses. Available online: https://www.bioidea.net/resources/smelly-musty-moldy-houses/ (accessed on 25 September 2017).
- Buszewski, B.; Kesy, M.; Ligor, T.; Amann, A. Human exhaled air analytics: Biomarkers of diseases. Biomed. Chromatogr. BMC 2007, 21, 553–566. [Google Scholar] [CrossRef] [PubMed]
- Renner, R. US EPA to revisit asbestos toxicity. Environ. Sci. Technol. 2007, 41, 1808–1809. [Google Scholar] [PubMed]
- Alleman, J.E.; Mossman, B.T. Asbestos revisited. Sci. Am. 1997, 277, 70–75. [Google Scholar] [CrossRef]
- Cooke, W.E. Fibrosis of the Lungs Due to the Inhalation of Asbestos Dust. BMJ 1924, 2, 147. [Google Scholar] [CrossRef] [PubMed]
- Selikoff, I.J.; Greenberg, M. A landmark case in asbestosis. JAMA 1991, 265, 898–901. [Google Scholar] [CrossRef] [PubMed]
- Burke, B. Shipbuilding’s Deadly Legacy: Introduction: Horrible Toll Could Have Been Avoided; Virginian-Pilot Norfolk: Virginia Beach, VA, USA, 2001. [Google Scholar]
- Gualtieri, A.F.; Tartaglia, A. Thermal decomposition of asbestos and recycling in traditional ceramics. J. Eur. Ceram. Soc. 2000, 20, 1409–1418. [Google Scholar] [CrossRef]
- McFall-Ngai, M. Adaptive immunity: Care for the community. Nature 2007, 445, 153. [Google Scholar] [CrossRef] [PubMed]
- Schleibinger, H.; Keller, R.; Rüden, H. Indoor Air Pollution by Microorganisms and Their Metabolites. In Air Pollution: Indoor Air Pollution; Pluschke, P., Ed.; Springer: Berlin/Heidelberg, Germany, 2004; pp. 149–177. [Google Scholar] [CrossRef]
- Levin, H. Can House Plants Solve IAQ Problems; Building Ecology Research Group: Santa Cruz, CA, USA, 1992. [Google Scholar]
- Wolverton, B.C.; Johnson, A.; Bounds, K. Interior Landscape Plants for Indoor Air Pollution Abatement; NASA Stennis Space Center: New York, NY, USA, 1989. [Google Scholar]
- Down, S. Houseplants as Air Fresheners. Available online: http://www.spectroscopynow.com/details/ezine/sepspec22493ezine/Houseplants-as-air-fresheners.html (accessed on 25 September 2017).
- Girman, J.; Phillips, T.; Levin, H. Critical Review: How Well Do House Plants Perform as Indoor Air Cleaners? Available online: http://www.buildingecology.com/articles/critical-review-how-well-do-house-plants-perform-as-indoor-air-cleaners/ (accessed on 25 September 2017).
- Institute of Medicine (US) Committee on Damp Indoor Spaces and Health. Damp Indoor Spaces and Health; The National Academies Press: Washington, DC, USA, 2004; p. 368. [Google Scholar] [CrossRef]
- Johnson, K. How Carbon Dioxide Became a ‘Pollutant’. Available online: https://www.wsj.com/articles/SB124001537515830975 (accessed on 25 September 2017).
- Goldstein, A.H.; Koven, C.D.; Heald, C.L.; Fung, I.Y. Biogenic carbon and anthropogenic pollutants combine to form a cooling haze over the southeastern United States. Proc. Natl. Acad. Sci. USA 2009, 106, 8835–8840. [Google Scholar] [CrossRef] [PubMed]
- Fischetti, M. The Paradox of Pollution-Producing Trees—Why some greenery can make smog worse. Sci. Am. 2014, 310. [Google Scholar]
- Tomasi, C.; Lupi, A. Primary and Secondary Sources of Atmospheric Aerosol. In Atmospheric Aerosols; Wiley-VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 2017. [Google Scholar]
- Penner, J.E. Primary and Secondary Sources of Aerosols; The Intergovernmental Panel on Climate Change (IPCC): Geneva, Switzerland, 2001. [Google Scholar]
- Rosenfeld, D.; Lahav, R.; Khain, A.; Pinsky, M. The role of sea spray in cleansing air pollution over ocean via cloud processes. Science 2002, 297, 1667–1670. [Google Scholar] [CrossRef] [PubMed]
- Von Glasow, R. Atmospheric chemistry: Pollution meets sea salt. Nat. Geosci. 2008, 1, 292–293. [Google Scholar] [CrossRef]
- Perraud, V.; Bruns, E.A.; Ezell, M.J.; Johnson, S.N.; Yu, Y.; Alexander, M.L.; Zelenyuk, A.; Imre, D.; Chang, W.L.; Dabdub, D.; et al. Nonequilibrium atmospheric secondary organic aerosol formation and growth. Proc. Natl. Acad. Sci. USA 2012, 109, 2836–2841. [Google Scholar] [CrossRef] [PubMed]
- Ilan, K.; Yoram, J.K.; Richard, W.; Martin, C.T.; Yinon, R.; Martins, J.V.; Daniel, R. The Bodélé depression: A single spot in the Sahara that provides most of the mineral dust to the Amazon forest. Environ. Res. Lett. 2006, 1, 014005. [Google Scholar]
- Sissakian, V.K.; Al-Ansari, N.; Knutsson, S. Sand and dust storm events in Iraq. Nat. Sci. 2013, 5, 1084–1094. [Google Scholar] [CrossRef]
- Lee, H.; Kim, H.; Honda, Y.; Lim, Y.-H.; Yi, S. Effect of Asian dust storms on daily mortality in seven metropolitan cities of Korea. Atmos. Environ. 2013, 79, 510–517. [Google Scholar] [CrossRef]
- Park, J.W.; Lim, Y.H.; Kyung, S.Y.; An, C.H.; Lee, S.P.; Jeong, S.H.; Ju, Y.S. Effects of ambient particulate matter on peak expiratory flow rates and respiratory symptoms of asthmatics during Asian dust periods in Korea. Respirology 2005, 10, 470–476. [Google Scholar] [CrossRef] [PubMed]
- Sandstrom, T.; Forsberg, B. Desert dust: An unrecognized source of dangerous air pollution? Epidemiology 2008, 19, 808–809. [Google Scholar] [CrossRef] [PubMed]
- Raloff, J. Ill Winds. Sci. News 2001, 160, 218. [Google Scholar] [CrossRef]
- Buzea, C.; Pacheco, I.I.; Robbie, K. Nanomaterials and nanoparticles: Sources and toxicity. Biointerphases 2007, 2, MR17-71. [Google Scholar] [CrossRef] [PubMed]
- Marques, J.T.; Carthew, R.W. A call to arms: Coevolution of animal viruses and host innate immune responses. Trends Genet. 2007, 23, 359–364. [Google Scholar] [CrossRef] [PubMed]
- Kampa, M.; Castanas, E. Human health effects of air pollution. Environ. Pollut. 2008, 151, 362–367. [Google Scholar] [CrossRef] [PubMed]
- Omidvarborna, H.; Kumar, A.; Kim, D.-S. Recent studies on soot modeling for diesel combustion. Renew. Sustain. Energy Rev. 2015, 48, 635–647. [Google Scholar] [CrossRef]
- Horwitz, C.; Buchanan, M. Pollution and Society. Available online: http://www.umich.edu/~gs265/index.html (accessed on 25 September 2017).
- Ritter, L.; International Program on Chemical Safety; Inter-Organization Programme for the Sound Management of Chemicals. A Review of Selected Persistent Organic Pollutants: DDT, Aldrin, Dieldrin, Endrin, Chlordane, Heptachlor, Hexachlorobenzene, Mirex, Toxaphene, Polycholorinated Biphenyls, Dioxins and Furans; 1995; publisher not identified. [Google Scholar]
- Ritter, L.; Solomon, K.; Forget, J.; Stemeroff, M.; O’Leary, C. Persistent Organic Pollutants; United Nations Environment Programme: Nairobi, Kenya, 1995. [Google Scholar]
- Kelly, B.C.; Ikonomou, M.G.; Blair, J.D.; Morin, A.E.; Gobas, F.A. Food web-specific biomagnification of persistent organic pollutants. Science 2007, 317, 236–239. [Google Scholar] [CrossRef] [PubMed]
- Beyer, A.; Mackay, D.; Matthies, M.; Wania, F.; Webster, E. Assessing Long-Range Transport Potential of Persistent Organic Pollutants. Environ. Sci. Technol. 2000, 34, 699–703. [Google Scholar] [CrossRef]
- Vallack, H.W.; Bakker, D.J.; Brandt, I.; Brostrom-Lunden, E.; Brouwer, A.; Bull, K.R.; Gough, C.; Guardans, R.; Holoubek, I.; Jansson, B.; et al. Controlling persistent organic pollutants-what next? Environ. Toxicol. Pharmacol. 1998, 6, 143–175. [Google Scholar] [CrossRef]
- Breivik, K.; Armitage, J.M.; Wania, F.; Sweetman, A.J.; Jones, K.C. Tracking the Global Distribution of Persistent Organic Pollutants Accounting for E-Waste Exports to Developing Regions. Environ. Sci. Technol. 2016, 50, 798–805. [Google Scholar] [CrossRef] [PubMed]
- Walker, C.H. Organic Pollutants: An Ecotoxicological Perspective, 2nd ed.; CRC Press: Boca Raton, FL, USA, 2008. [Google Scholar]
- Szabo, D.T.; Loccisano, A.E. POPs and Human Health Risk Assessment. In Dioxins and Health; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 4141; pp. 579–618. [Google Scholar] [CrossRef]
- Barringer, F. Scientists Find New Dangers in Tiny but Pervasive Particles in Air Pollution; New York Times: New York, NY, USA, 2012. [Google Scholar]
- Brown, J.S.; Gordon, T.; Price, O.; Asgharian, B. Thoracic and respirable particle definitions for human health risk assessment. Part. Fibre Toxicol. 2013, 10, 12. [Google Scholar] [CrossRef] [PubMed]
- Yamago, S.; Tokuyama, H.; Nakamura, E.; Kikuchi, K.; Kananishi, S.; Sueki, K.; Nakahara, H.; Enomoto, S.; Ambe, F. In vivo biological behavior of a water-miscible fullerene: 14C labeling, absorption, distribution, excretion and acute toxicity. Chem. Biol. 1995, 2, 385–389. [Google Scholar] [CrossRef]
- National PEP Weighing Laboratory, US-EPA, Region 4. PM2.5 Objectives and History. Available online: https://archive.epa.gov/pesticides/region4/sesd/pm25/web/html/p2.html (accessed on 25 September 2017).
- Olsson, P.Q.; Benner, R.L. Atmospheric Chemistry and Physics: From Air Pollution to Climate Change. J. Am. Chem. Soc. 1999, 121, 1423. [Google Scholar] [CrossRef]
- Seinfeld, J.H.; Pandis, S.N. Atmospheric Chemistry and Physics: From Air Pollution to Climate Change; Wiley: Hoboken, NJ, USA, 1998. [Google Scholar]
- Hamra, G.B.; Guha, N.; Cohen, A.; Laden, F.; Raaschou-Nielsen, O.; Samet, J.M.; Vineis, P.; Forastiere, F.; Saldiva, P.; Yorifuji, T.; et al. Outdoor particulate matter exposure and lung cancer: A systematic review and meta-analysis. Environ. Health Perspect. 2014, 122, 906–911. [Google Scholar] [CrossRef] [PubMed]
- Raaschou-Nielsen, O.; Andersen, Z.J.; Beelen, R.; Samoli, E.; Stafoggia, M.; Weinmayr, G.; Hoffmann, B.; Fischer, P.; Nieuwenhuijsen, M.J.; Brunekreef, B.; et al. Air pollution and lung cancer incidence in 17 European cohorts: Prospective analyses from the European Study of Cohorts for Air Pollution Effects (ESCAPE). Lancet Oncol. 2013, 14, 813–822. [Google Scholar] [CrossRef]
- Raaschou-Nielsen, O.; Beelen, R.; Wang, M.; Hoek, G.; Andersen, Z.J.; Hoffmann, B.; Stafoggia, M.; Samoli, E.; Weinmayr, G.; Dimakopoulou, K.; et al. Particulate matter air pollution components and risk for lung cancer. Environ. Int. 2016, 87, 66–73. [Google Scholar] [CrossRef] [PubMed]
- Cesaroni, G.; Forastiere, F.; Stafoggia, M.; Andersen, Z.J.; Badaloni, C.; Beelen, R.; Caracciolo, B.; de Faire, U.; Erbel, R.; Eriksen, K.T.; et al. Long term exposure to ambient air pollution and incidence of acute coronary events: Prospective cohort study and meta-analysis in 11 European cohorts from the ESCAPE Project. BMJ 2014, 348, f7412. [Google Scholar] [CrossRef] [PubMed]
- Helmholtz Zentrum München-German Research Center for Environmental Health. Particulate Air Pollution Leads to Increased Heart Attack Risk; Science Daily: Rockvile, MD, USA, 2014. [Google Scholar]
- Doyle, K. Pollution Particles Damage Blood Vessels, May Lead to Heart Disease; Reuters: New York, NY, USA, 2016. [Google Scholar]
- Du, Y.; Xu, X.; Chu, M.; Guo, Y.; Wang, J. Air particulate matter and cardiovascular disease: The epidemiological, biomedical and clinical evidence. J. Thorac. Dis. 2015, 8, E8–E19. [Google Scholar]
- Silverman, D.T.; Samanic, C.M.; Lubin, J.H.; Blair, A.E.; Stewart, P.A.; Vermeulen, R.; Coble, J.B.; Rothman, N.; Schleiff, P.L.; Travis, W.D.; et al. The Diesel Exhaust in Miners study: A nested case-control study of lung cancer and diesel exhaust. J. Natl. Cancer Inst. 2012, 104, 855–868. [Google Scholar] [CrossRef] [PubMed]
- Attfield, M.D.; Schleiff, P.L.; Lubin, J.H.; Blair, A.; Stewart, P.A.; Vermeulen, R.; Coble, J.B.; Silverman, D.T. The Diesel Exhaust in Miners study: A cohort mortality study with emphasis on lung cancer. J. Natl. Cancer Inst. 2012, 104, 869–883. [Google Scholar] [CrossRef] [PubMed]
- Neslen, A. Diesel Cars May Be Worse Than Petrol for Carbon Emissions, Report Claims; Guardian: London, UK, 2016. [Google Scholar]
- Brugge, D.; Durant, J.L.; Rioux, C. Near-highway pollutants in motor vehicle exhaust: A review of epidemiologic evidence of cardiac and pulmonary health risks. Environ. Health 2007, 6, 23. [Google Scholar] [CrossRef] [PubMed]
- Garshick, E.; Laden, F.; Hart, J.E.; Rosner, B.; Davis, M.E.; Eisen, E.A.; Smith, T.J. Lung cancer and vehicle exhaust in trucking industry workers. Environ. Health Perspect. 2008, 116, 1327–1332. [Google Scholar] [CrossRef] [PubMed]
- Garshick, E.; Laden, F.; Hart, J.E.; Davis, M.E.; Eisen, E.A.; Smith, T.J. Lung cancer and elemental carbon exposure in trucking industry workers. Environ. Health Perspect. 2012, 120, 1301–1306. [Google Scholar] [CrossRef] [PubMed]
- Lee, P.C.; Talbott, E.O.; Roberts, J.M.; Catov, J.M.; Bilonick, R.A.; Stone, R.A.; Sharma, R.K.; Ritz, B. Ambient air pollution exposure and blood pressure changes during pregnancy. Environ. Res. 2012, 117, 46–53. [Google Scholar] [CrossRef] [PubMed]
- Fleischer, N.L.; Merialdi, M.; van Donkelaar, A.; Vadillo-Ortega, F.; Martin, R.V.; Betran, A.P.; Souza, J.P. Outdoor air pollution, preterm birth, and low birth weight: Analysis of the world health organization global survey on maternal and perinatal health. Environ. Health Perspect. 2014, 122, 425–430. [Google Scholar] [CrossRef] [PubMed]
- Erickson, A.C.; Arbour, L. The shared pathoetiological effects of particulate air pollution and the social environment on fetal-placental development. J. Environ. Public Health 2014, 2014, 901017. [Google Scholar] [CrossRef] [PubMed]
- Byrne, C.D.; Phillips, D.I. Fetal origins of adult disease: Epidemiology and mechanisms. J. Clin. Pathol. 2000, 53, 822–828. [Google Scholar] [CrossRef] [PubMed]
- Woodruff, T.J.; Parker, J.D.; Darrow, L.A.; Slama, R.; Bell, M.L.; Choi, H.; Glinianaia, S.; Hoggatt, K.J.; Karr, C.J.; Lobdell, D.T.; et al. Methodological issues in studies of air pollution and reproductive health. Environ. Res. 2009, 109, 311–320. [Google Scholar] [CrossRef] [PubMed]
- Cohen, A.J.; Ross Anderson, H.; Ostro, B.; Pandey, K.D.; Krzyzanowski, M.; Kunzli, N.; Gutschmidt, K.; Pope, A.; Romieu, I.; Samet, J.M.; et al. The global burden of disease due to outdoor air pollution. J. Toxicol. Environ. Health A 2005, 68, 1301–1307. [Google Scholar] [CrossRef] [PubMed]
- McGrath, M. Four Major Cities Move to Ban Diesel Vehicles by 2025; BBC News: London, UK, 2016. [Google Scholar]
- Harte, J.; Holdren, C.; Schneider, R.; Shirley, C. Toxics A to Z: A Guide to Everyday Pollution Hazards; University of California Press: Oakland, CA, USA, 1991. [Google Scholar]
- Gordon, B.; Mackay, R.; Rehfuess, E. Polluted Cities: The Air Children Breathe. In Inheriting the World: The Atlas of Children’s Health & the Environment; WHO: Washington, DC, USA, 2004; p. 28. [Google Scholar]
- Delorme, N.; Le Blanc, C.; Dezzani, A.; Bely, M.; Ferret, A.; Laminette, S.; Roudier, J.; Colinet, E. A NEMS-Array Control IC for Subattogram Mass Sensing Applications in 28 nm CMOS Technology. IEEE J. Solid-State Circuits 2016, 51, 249–258. [Google Scholar] [CrossRef]
- Lussac, E.; Barattin, R.; Cardinael, P.; Agasse, V. Review on Micro-Gas Analyzer Systems: Feasibility, Separations and Applications. Crit. Rev. Anal. Chem. 2016, 46, 455–468. [Google Scholar] [CrossRef] [PubMed]
- Wetchakun, K.; Samerjai, T.; Tamaekong, N.; Liewhiran, C.; Siriwong, C.; Kruefu, V.; Wisitsoraat, A.; Tuantranont, A.; Phanichphant, S. Semiconducting metal oxides as sensors for environmentally hazardous gases. Sens. Actuators B Chem. 2011, 160, 580–591. [Google Scholar] [CrossRef]
- Fanget, S.; Hentz, S.; Puget, P.; Arcamone, J.; Matheron, M.; Colinet, E.; Andreucci, P.; Duraffourg, L.; Myers, E.; Roukes, M.L. Gas sensors based on gravimetric detection—A review. Sens. Actuators B Chem. 2011, 160, 804–821. [Google Scholar] [CrossRef]
- Lee, H.J.; Park, K.K.; Kupnik, M.; Khuri-Yakub, B.T. Functionalization layers for CO2 sensing using capacitive micromachined ultrasonic transducers. Sens. Actuators B Chem. 2012, 174, 87–93. [Google Scholar] [CrossRef]
- Barauskas, D.; Pelenis, D.; Virzonis, D.; Baltrus, J.P.; Baltrusaitis, J. Greenhouse Gas Molecule CO2 Detection Using a Capacitive Micromachined Ultrasound Transducer. Anal. Chem. 2016, 88, 6662–6665. [Google Scholar] [CrossRef] [PubMed]
- Kanan, S.M.; El-Kadri, O.M.; Abu-Yousef, I.A.; Kanan, M.C. Semiconducting metal oxide based sensors for selective gas pollutant detection. Sensors 2009, 9, 8158–8196. [Google Scholar] [CrossRef] [PubMed]
- Mirzaei, A.; Janghorban, K.; Hashemi, B.; Neri, G. Metal-core@metal oxide-shell nanomaterials for gas-sensing applications: A review. J. Nanopart. Res. 2015, 17. [Google Scholar] [CrossRef]
- Ding, B.; Kim, J.; Miyazaki, Y.; Shiratori, S. Electrospun nanofibrous membranes coated quartz crystal microbalance as gas sensor for NH3 detection. Sens. Actuators B Chem. 2004, 101, 373–380. [Google Scholar] [CrossRef]
- Xianfeng, W.; Bin, D.; Jianyong, Y.; Moran, W.; Fukui, P. A highly sensitive humidity sensor based on a nanofibrous membrane coated quartz crystal microbalance. Nanotechnology 2010, 21, 055502. [Google Scholar]
- Venstra, W.J.; Capener, M.J.; Elliott, S.R. Nanomechanical gas sensing with nonlinear resonant cantilevers. Nanotechnology 2014, 25, 425501. [Google Scholar] [CrossRef] [PubMed]
- Fraiwan, A.; Lee, H.; Choi, S. A paper-based cantilever array sensor: Monitoring volatile organic compounds with naked eye. Talanta 2016, 158, 57–62. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.J.; Park, K.K.; Kupnik, M.; Oralkan, O.; Khuri-Yakub, B.T. Highly sensitive detection of DMMP using a CMUT-based chemical sensor. In Proceedings of the Sensors, Kona, HI, USA, 1–4 November 2010. [Google Scholar] [CrossRef]
- Kwon, O.S.; Park, C.S.; Park, S.J.; Noh, S.; Kim, S.; Kong, H.J.; Bae, J.; Lee, C.S.; Yoon, H. Carboxylic Acid-Functionalized Conducting-Polymer Nanotubes as Highly Sensitive Nerve-Agent Chemiresistors. Sci. Rep. 2016, 6, 33724. [Google Scholar] [CrossRef] [PubMed]
- Cavallo, P.; Frontera, E.; Acevedo, D.F.; Olejnik, R.; Slobodian, P.; Saha, P.; Barbero, C.A. Functionalized polyanilines made by nucleophilic addition reaction, applied in gas sensors field. Synth. Met. 2016, 215, 127–133. [Google Scholar] [CrossRef]
- Krichevsky, D.M.; Zasedatelev, A.V.; Tolbin, A.Y.; Yu, M.Z.; Krasovskii, V.I.; Karpo, A.B.; Tomilova, L.G. A low-symmetrical zinc phthalocyanine-based Langmuir-Blodgett thin films for NO2 gas sensor applications. J. Phys. Conf. Ser. 2016, 737, 012030. [Google Scholar] [CrossRef]
- Akiyama, N. A sensor array based on trigonal-selenium nanowires for the detection of gas mixtures. Sens. Actuators B Chem. 2016, 223, 131–137. [Google Scholar] [CrossRef]
- Ma, H.; Qin, S.; Wang, L.; Wang, G.; Zhao, X.; Ding, E. The study on methane sensing with high-temperature low-power CMOS compatible silicon microheater. Sens. Actuators B Chem. 2017, 244, 17–23. [Google Scholar] [CrossRef]
- Puigcorb, J.; Vogel, D.; Michel, B.; Vil, A.; Grcia, I.; Can, C.; Morante, J.R. High temperature degradation of Pt/Ti electrodes in micro-hotplate gas sensors. J. Micromech. Microeng. 2003, 13, S119–S124. [Google Scholar] [CrossRef]
- Karpov, E.E.; Karpov, E.F.; Suchkov, A.; Mironov, S.; Baranov, A.; Sleptsov, V.; Calliari, L. Energy efficient planar catalytic sensor for methane measurement. Sens. Actuators A Phys. 2013, 194, 176–180. [Google Scholar] [CrossRef]
- Asgharian, B.; Price, O.T.; Oldham, M.; Chen, L.C.; Saunders, E.L.; Gordon, T.; Mikheev, V.B.; Minard, K.R.; Teeguarden, J.G. Computational modeling of nanoscale and microscale particle deposition, retention and dosimetry in the mouse respiratory tract. Inhal. Toxicol. 2014, 26, 829–842. [Google Scholar] [CrossRef] [PubMed]
- Nichols, J.E.; Niles, J.A.; Vega, S.P.; Argueta, L.B.; Eastaway, A.; Cortiella, J. Modeling the lung: Design and development of tissue engineered macro- and micro-physiologic lung models for research use. Exp. Biol. Med. (Maywood) 2014, 239, 1135–1169. [Google Scholar] [CrossRef] [PubMed]
- Geiser, M.; Kreyling, W.G. Deposition and biokinetics of inhaled nanoparticles. Part. Fibre Toxicol. 2010, 7, 2. [Google Scholar] [CrossRef] [PubMed]
- Nalayanda, D.D.; Wang, Q.; Fulton, W.B.; Wang, T.H.; Abdullah, F. Engineering an artificial alveolar-capillary membrane: A novel continuously perfused model within microchannels. J. Pediatr. Surg. 2010, 45, 45–51. [Google Scholar] [CrossRef] [PubMed]
- Benam, K.H.; Villenave, R.; Lucchesi, C.; Varone, A.; Hubeau, C.; Lee, H.H.; Alves, S.E.; Salmon, M.; Ferrante, T.C.; Weaver, J.C.; et al. Small airway-on-a-chip enables analysis of human lung inflammation and drug responses in vitro. Nat. Methods 2016, 13, 151–157. [Google Scholar] [CrossRef] [PubMed]
- Punde, T.H.; Wu, W.H.; Lien, P.C.; Chang, Y.L.; Kuo, P.H.; Chang, M.D.; Lee, K.Y.; Huang, C.D.; Kuo, H.P.; Chan, Y.F.; et al. A biologically inspired lung-on-a-chip device for the study of protein-induced lung inflammation. Integr. Biol. 2015, 7, 162–169. [Google Scholar] [CrossRef] [PubMed]
- Sellgren, K.L.; Butala, E.J.; Gilmour, B.P.; Randell, S.H.; Grego, S. A biomimetic multicellular model of the airways using primary human cells. Lab Chip 2014, 14, 3349–3358. [Google Scholar] [CrossRef] [PubMed]
- Huh, D.; Matthews, B.D.; Mammoto, A.; Montoya-Zavala, M.; Hsin, H.Y.; Ingber, D.E. Reconstituting Organ-Level Lung Functions on a Chip. Science 2010, 328, 1662–1668. [Google Scholar] [CrossRef] [PubMed]
- Waters, C.M.; Roan, E.; Navajas, D. Mechanobiology in lung epithelial cells: Measurements, perturbations, and responses. Compr. Phys. 2012, 2, 1–29. [Google Scholar] [CrossRef]
- Stucki, A.O.; Stucki, J.D.; Hall, S.R.; Felder, M.; Mermoud, Y.; Schmid, R.A.; Geiser, T.; Guenat, O.T. A lung-on-a-chip array with an integrated bio-inspired respiration mechanism. Lab Chip 2015, 15, 1302–1310. [Google Scholar] [CrossRef] [PubMed]
- Douville, N.J.; Zamankhan, P.; Tung, Y.C.; Li, R.; Vaughan, B.L.; Tai, C.F.; White, J.; Christensen, P.J.; Grotberg, J.B.; Takayama, S. Combination of fluid and solid mechanical stresses contribute to cell death and detachment in a microfluidic alveolar model. Lab Chip 2011, 11, 609–619. [Google Scholar] [CrossRef] [PubMed]
- Nesmith, A.P.; Agarwal, A.; McCain, M.L.; Parker, K.K. Human airway musculature on a chip: An in vitro model of allergic asthmatic bronchoconstriction and bronchodilation. Lab Chip 2014, 14, 3925–3936. [Google Scholar] [CrossRef] [PubMed]
- Skolimowski, M.; Nielsen, M.W.; Abeille, F.; Skafte-Pedersen, P.; Sabourin, D.; Fercher, A.; Papkovsky, D.; Molin, S.; Taboryski, R.; Sternberg, C.; et al. Modular microfluidic system as a model of cystic fibrosis airways. Biomicrofluidics 2012, 6, 34109. [Google Scholar] [CrossRef] [PubMed]
- Tavana, H.; Zamankhan, P.; Christensen, P.J.; Grotberg, J.B.; Takayama, S. Epithelium damage and protection during reopening of occluded airways in a physiologic microfluidic pulmonary airway model. Biomed. Microdevices 2011, 13, 731–742. [Google Scholar] [CrossRef] [PubMed]
- Huh, D.; Leslie, D.C.; Matthews, B.D.; Fraser, J.P.; Jurek, S.; Hamilton, G.A.; Thorneloe, K.S.; McAlexander, M.A.; Ingber, D.E. A Human Disease Model of Drug Toxicity-Induced Pulmonary Edema in a Lung-on-a-Chip Microdevice. Sci. Transl. Med. 2012, 4, ARTN–159ra147. [Google Scholar] [CrossRef] [PubMed]
- Morrow, P.E. Possible mechanisms to explain dust overloading of the lungs. Fundam. Appl. Toxicol. 1988, 10, 369–384. [Google Scholar] [CrossRef]
- Pope, C.A., 3rd; Burnett, R.T.; Thun, M.J.; Calle, E.E.; Krewski, D.; Ito, K.; Thurston, G.D. Lung cancer, cardiopulmonary mortality, and long-term exposure to fine particulate air pollution. JAMA 2002, 287, 1132–1141. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.; Gao, B.; Zhou, Z.; Chang, Y. The movement and deposition of PM2.5 in the upper respiratory tract for the patients with heart failure: An elementary CFD study. Biomed. Eng. Online 2016, 15, 138. [Google Scholar] [CrossRef] [PubMed]
- Schins, R.P.; Lightbody, J.H.; Borm, P.J.; Shi, T.; Donaldson, K.; Stone, V. Inflammatory effects of coarse and fine particulate matter in relation to chemical and biological constituents. Toxicol. Appl. Pharmacol. 2004, 195, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Lewtas, J. Air pollution combustion emissions: Characterization of causative agents and mechanisms associated with cancer, reproductive, and cardiovascular effects. Mutat. Res. 2007, 636, 95–133. [Google Scholar] [CrossRef] [PubMed]
- Dagher, Z.; Garcon, G.; Billet, S.; Gosset, P.; Ledoux, F.; Courcot, D.; Aboukais, A.; Shirali, P. Activation of different pathways of apoptosis by air pollution particulate matter (PM2.5) in human epithelial lung cells (L132) in culture. Toxicology 2006, 225, 12–24. [Google Scholar] [CrossRef] [PubMed]
- Deng, X.; Zhang, F.; Rui, W.; Long, F.; Wang, L.; Feng, Z.; Chen, D.; Ding, W. PM2.5-induced oxidative stress triggers autophagy in human lung epithelial A549 cells. Toxicol In Vitro 2013, 27, 1762–1770. [Google Scholar] [CrossRef] [PubMed]
- Mahalingaiah, S.; Hart, J.E.; Laden, F.; Terry, K.L.; Boynton-Jarrett, R.; Aschengrau, A.; Missmer, S.A. Air pollution and risk of uterine leiomyomata. Epidemiology 2014, 25, 682–688. [Google Scholar] [CrossRef] [PubMed]
- Nursan, C.; Muge, A.T.; Cemile, D.; Pinar, T.; Sevin, A. Parent’s knowledge and perceptions of the health effects of environmental hazards in Sakarya, Turkey. J. Pak. Med. Assoc. 2014, 64, 38–41. [Google Scholar] [PubMed]
- Puett, R.C.; Hart, J.E.; Yanosky, J.D.; Spiegelman, D.; Wang, M.; Fisher, J.A.; Hong, B.; Laden, F. Particulate matter air pollution exposure, distance to road, and incident lung cancer in the nurses’ health study cohort. Environ. Health Perspect. 2014, 122, 926–932. [Google Scholar] [CrossRef] [PubMed]
- Billet, S.; Abbas, I.; Le Goff, J.; Verdin, A.; Andre, V.; Lafargue, P.E.; Hachimi, A.; Cazier, F.; Sichel, F.; Shirali, P.; et al. Genotoxic potential of Polycyclic Aromatic Hydrocarbons-coated onto airborne Particulate Matter (PM 2.5) in human lung epithelial A549 cells. Cancer Lett. 2008, 270, 144–155. [Google Scholar] [CrossRef] [PubMed]
- Travis, W.D.; Brambilla, E.; Burke, A.P.; Marx, A.; Nicholson, A.G. WHO Classification of Tumours of the Lung, Pleura, Thymus and Heart, 4th ed.; International Agency for Research on Cancer: Washington, DC, USA, 2015. [Google Scholar]
- Socinski, M.A. Clinical issues in the management of non-small-cell lung cancer and the role of platinum-based therapy. Clin. Lung Cancer 2004, 5, 274–289. [Google Scholar] [CrossRef] [PubMed]
- Hensing, T.A.; Peterman, A.H.; Schell, M.J.; Lee, J.H.; Socinski, M.A. The impact of age on toxicity, response rate, quality of life, and survival in patients with advanced, Stage IIIB or IV nonsmall cell lung carcinoma treated with carboplatin and paclitaxel. Cancer 2003, 98, 779–788. [Google Scholar] [CrossRef] [PubMed]
- Culy, C.R.; Faulds, D. Gefitinib. Drugs 2002, 62, 2237–2248. [Google Scholar] [CrossRef] [PubMed]
- Su, S.; Wang, T.; Duan, J.A.; Zhou, W.; Hua, Y.Q.; Tang, Y.P.; Yu, L.; Qian, D.W. Anti-inflammatory and analgesic activity of different extracts of Commiphora myrrha. J. Ethnopharmacol. 2011, 134, 251–258. [Google Scholar] [CrossRef] [PubMed]
- Anderson, M. As the Wind Blows: The Effects of Long-Term Exposure to Air Pollution on Mortality. NBEI Working Pap. Ser. 2015, 21578. [Google Scholar] [CrossRef]

| Company | Country | Developing the OoC Models | Providing Services or Chips in the Field of OoC |
|---|---|---|---|
| AxoSim Technologies | US | x | |
| CNBio Innovations | UK | x | |
| Emulate | US | x | |
| Mimetas | The Netherlands | x | |
| TissUse | Germany | x | |
| Hesperosinc | US | x | |
| Hurel Corporation | US | x | |
| InSphero | US | x | |
| Microfluidic Chipshop | Germany | x | |
| Micronit | The Netherlands | x | |
| Molecular Devices | US | x | |
| Neofluidics | US | x | |
| Nortis | US | x | |
| TARA Biosystems | US | x |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Schulze, F.; Gao, X.; Virzonis, D.; Damiati, S.; Schneider, M.R.; Kodzius, R. Air Quality Effects on Human Health and Approaches for Its Assessment through Microfluidic Chips. Genes 2017, 8, 244. https://doi.org/10.3390/genes8100244
Schulze F, Gao X, Virzonis D, Damiati S, Schneider MR, Kodzius R. Air Quality Effects on Human Health and Approaches for Its Assessment through Microfluidic Chips. Genes. 2017; 8(10):244. https://doi.org/10.3390/genes8100244
Chicago/Turabian StyleSchulze, Frank, Xinghua Gao, Darius Virzonis, Samar Damiati, Marlon R. Schneider, and Rimantas Kodzius. 2017. "Air Quality Effects on Human Health and Approaches for Its Assessment through Microfluidic Chips" Genes 8, no. 10: 244. https://doi.org/10.3390/genes8100244
APA StyleSchulze, F., Gao, X., Virzonis, D., Damiati, S., Schneider, M. R., & Kodzius, R. (2017). Air Quality Effects on Human Health and Approaches for Its Assessment through Microfluidic Chips. Genes, 8(10), 244. https://doi.org/10.3390/genes8100244






