Endocrine Dysfunction in Female FMR1 Premutation Carriers: Characteristics and Association with Ill Health
Abstract
:1. Introduction
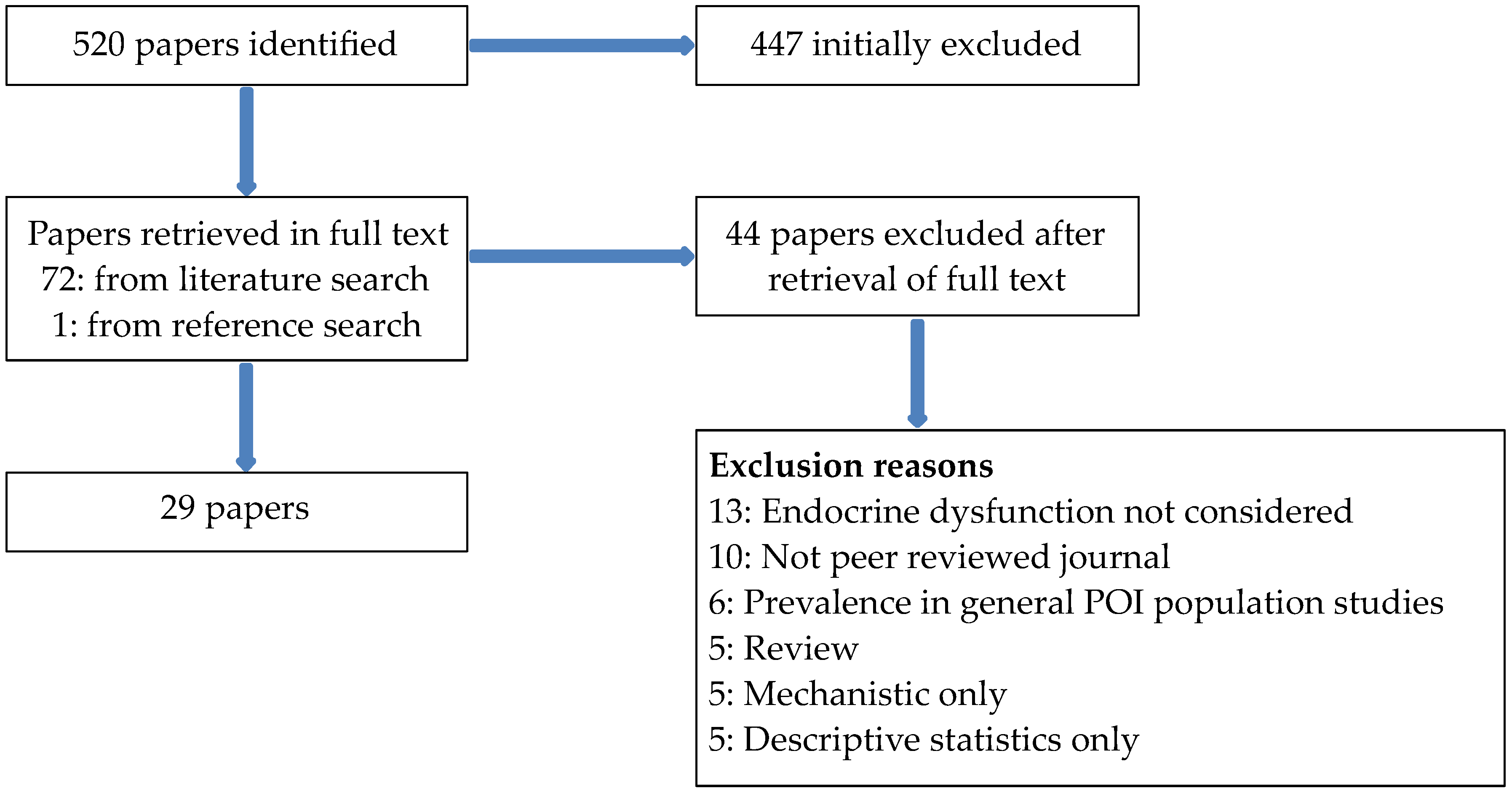
2. Materials and Methods
2.1. Literature Review
2.2. Pilot Study
3. Results
3.1. Literature Review
3.1.1. Reproductive System
Primary Ovarian Failure/Primary Ovarian Insufficiency
Premenopausal Ovarian Dysfunction
Relationship with Genetic Characteristics
3.1.2. Other Endocrine Disorders
3.1.3. Association between Endocrine Dysfunction and Other Health Conditions
3.2. Pilot Study
4. Discussion
4.1. Current Findings
4.2. Limitations of Existing Research
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Fu, Y.H.; Kuhl, D.P.; Pizzuti, A.; Pieretti, M.; Sutcliffe, J.S.; Richards, S.; Verkerk, A.J.; Holden, J.J.; Fenwick, R.G., Jr.; Warren, S.T.; et al. Variation of the CGG repeat at the fragile X site results in genetic instability: Resolution of the Sherman paradox. Cell 1991, 67, 1047–1058. [Google Scholar] [CrossRef]
- Lozano, R.; Rosero, C.A.; Hagerman, R.J. Fragile X spectrum disorders. Intractable Rare Dis. Res. 2014, 3, 134–146. [Google Scholar] [CrossRef] [PubMed]
- Wheeler, A.C.; Bailey, D.B., Jr.; Berry-Kravis, E.; Greenberg, J.; Losh, M.; Mailick, M.; Mila, M.; Olichney, J.M.; Rodriguez-Revenga, L.; Sherman, S.; et al. Associated features in females with an FMR1 premutation. J. Neurodev. Disord. 2014, 6, 30. [Google Scholar] [CrossRef] [PubMed]
- Hagerman, R.J.; Hall, D.A.; Coffey, S.; Leehey, M.; Bourgeois, J.; Gould, J.; Zhang, L.; Seritan, A.; Berry-Kravis, E.; Olichney, J.; et al. Treatment of fragile X-associated tremor ataxia syndrome (FXTAS) and related neurological problems. Clin. Interv. Aging 2008, 3, 251–262. [Google Scholar] [CrossRef]
- Jacquemont, S.; Hagerman, R.J.; Leehey, M.A.; Hall, D.A.; Levine, R.A.; Brunberg, J.A.; Zhang, L.; Jardini, T.; Gane, L.W.; Harris, S.W.; et al. Penetrance of the fragile X-associated tremor/ataxia syndrome in a premutation carrier population. JAMA 2004, 291, 460–469. [Google Scholar] [CrossRef] [PubMed]
- Hunter, J.E.; Rohr, J.K.; Sherman, S.L. Co-occurring diagnoses among FMR1 premutation allele carriers. Clin. Genet. 2010, 77, 374–381. [Google Scholar] [CrossRef] [PubMed]
- Coffey, S.M.; Cook, K.; Tartaglia, N.; Tassone, F.; Nguyen, D.V.; Pan, R.; Bronsky, H.E.; Yuhas, J.; Borodyanskaya, M.; Grigsby, J.; et al. Expanded clinical phenotype of women with the FMR1 premutation. Am. J. Med. Genet. A. 2008, 146A, 1009–1016. [Google Scholar] [CrossRef] [PubMed]
- Summers, S.M.; Cogswell, J.; Goodrich, J.E.; Mu, Y.; Nguyen, D.V.; Brass, S.D.; Hagerman, R.J. Prevalence of restless legs syndrome and sleep quality in carriers of the fragile X premutation. Clin. Genet. 2014, 86, 181–184. [Google Scholar] [CrossRef] [PubMed]
- Wittenberger, M.D.; Hagerman, R.J.; Sherman, S.L.; McConkie-Rosell, A.; Welt, C.K.; Rebar, R.W.; Corrigan, E.C.; Simpson, J.L.; Nelson, L.M. The FMR1 premutation and reproduction. Fertil. Steril. 2007, 87, 456–465. [Google Scholar] [CrossRef] [PubMed]
- Lokanga, R.A.; Entezam, A.; Kumari, D.; Yudkin, D.; Qin, M.; Smith, C.B.; Usdin, K. Somatic expansion in mouse and human carriers of fragile X premutation alleles. Hum. Mutat. 2013, 34, 157–166. [Google Scholar] [CrossRef] [PubMed]
- Hukema, R.K.; Buijsen, R.A.; Raske, C.; Severijnen, L.A.; Nieuwenhuizen-Bakker, I.; Minneboo, M.; Maas, A.; de Crom, R.; Kros, J.M.; Hagerman, P.J.; et al. Induced expression of expanded CGG RNA causes mitochondrial dysfunction in vivo. Cell Cycle 2014, 13, 2600–2608. [Google Scholar] [CrossRef] [PubMed]
- Jacquemont, S.; Birnbaum, S.; Redler, S.; Steinbach, P.; Biancalana, V. Clinical utility gene card for: Fragile X mental retardation syndrome, fragile X-associated tremor/ataxia syndrome and fragile X-associated primary ovarian insufficiency. Eur. J. Hum. Genet. 2011, 19. [Google Scholar] [CrossRef] [PubMed]
- Hagerman, P.J.; Hagerman, R.J. The fragile-X premutation: A maturing perspective. Am. J. Hum. Genet. 2004, 74, 805–816. [Google Scholar] [CrossRef] [PubMed]
- Ross-Inta, C.; Omanska-Klusek, A.; Wong, S.; Barrow, C.; Garcia-Arocena, D.; Iwahashi, C.; Berry-Kravis, E.; Hagerman, R.J.; Hagerman, P.J.; Giulivi, C. Evidence of mitochondrial dysfunction in fragile X-associated tremor/ataxia syndrome. Biochem. J. 2010, 429, 545–552. [Google Scholar] [CrossRef] [PubMed]
- Napoli, E.; Ross-Inta, C.; Wong, S.; Omanska-Klusek, A.; Barrow, C.; Iwahashi, C.; Garcia-Arocena, D.; Sakaguchi, D.; Berry-Kravis, E.; Hagerman, R.; et al. Altered zinc transport disrupts mitochondrial protein processing/import in fragile X-associated tremor/ataxia syndrome. Hum. Mol. Genet. 2011, 20, 3079–3092. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Revenga, L.; Madrigal, I.; Pagonabarraga, J.; Xunclà, M.; Badenas, C.; Kulisevsky, J.; Gomez, B.; Milà, M. Penetrance of FMR1 premutation associated pathologies in fragile X syndrome families. Eur. J. Hum. Genet. 2009, 17, 1359–1362. [Google Scholar] [CrossRef] [PubMed]
- Allingham-Hawkins, D.J.; Babul-Hirji, R.; Chitayat, D.; Holden, J.J.; Yang, K.T.; Lee, C.; Hudson, R.; Gorwill, H.; Nolin, S.L.; Glicksman, A.; et al. Fragile X premutation is a significant risk factor for premature ovarian failure: The international collaborative POF in fragile X study—Preliminary data. Am. J. Med. Genet. 1999, 83, 322–325. [Google Scholar] [CrossRef]
- Au, J.; Akins, R.S.; Berkowitz-Sutherland, L.; Tang, H.T.; Chen, Y.; Boyd, A.; Tassone, F.; Nguyen, D.V.; Hagerman, R. Prevalence and risk of migraine headaches in adult fragile X premutation carriers. Clin. Genet. 2013, 84, 546–551. [Google Scholar] [CrossRef] [PubMed]
- Greco, C.M.; Soontrapornchai, K.; Wirojanan, J.; Gould, J.E.; Hagerman, P.J.; Hagerman, R.J. Testicular and pituitary inclusion formation in fragile X associated tremor/ataxia syndrome. J. Urol. 2007, 177, 1434–1437. [Google Scholar] [CrossRef] [PubMed]
- Buijsen, R.A.; Sellier, C.; Severijnen, L.A.; Oulad-Abdelghani, M.; Verhagen, R.F.; Berman, R.F.; Charlet-Berguerand, N.; Willemsen, R.; Hukema, R.K. FMRpolyg-positive inclusions in CNS and non-CNS organs of a fragile X premutation carrier with fragile X-associated tremor/ataxia syndrome. Acta Neuropathol. Commun. 2014, 2, 162. [Google Scholar] [CrossRef] [PubMed]
- Buijsen, R.A.M.; Visser, J.A.; Kramer, P.; Severijnen, E.A.W.F.M.; Gearing, M.; Charlet-Berguerand, N.; Sherman, S.L.; Berman, R.F.; Willemsen, R.; Hukema, R.K. Presence of inclusions positive for polyglycine containing protein, FMRpolyG, indicates that repeat-associated non-AUG translation plays a role in fragile X-associated primary ovarian insufficiency. Hum. Reprod. 2016, 31, 158–168. [Google Scholar] [CrossRef] [PubMed]
- Louis, E.; Moskowitz, C.; Friez, M.; Amaya, M.; Vonsattel, J.P. Parkinsonism, dysautonomia, and intranuclear inclusions in a fragile X carrier: A clinical-pathological study. Mov. Disord. 2006, 21, 420–425. [Google Scholar] [CrossRef] [PubMed]
- Allen, E.G.; Sullivan, A.K.; Marcus, M.; Small, C.; Dominguez, C.; Epstein, M.P.; Charen, K.; He, W.; Taylor, K.C.; Sherman, S.L. Examination of reproductive aging milestones among women who carry the FMR1 premutation. Hum. Reprod. 2007, 22, 2142–2152. [Google Scholar] [CrossRef] [PubMed]
- Sullivan, A.K.; Marcus, M.; Epstein, M.P.; Allen, E.G.; Anido, A.E.; Paquin, J.J.; Yadav-Shah, M.; Sherman, S.L. Association of FMR1 repeat size with ovarian dysfunction. Hum. Reprod. 2005, 20, 402–412. [Google Scholar] [CrossRef] [PubMed]
- Vianna-Morgante, A.M.; Costa, S.S.; De Pavanello, R.C.M.; Otto, P.A.; Mingroni-Netto, R.C. Premature ovarian failure (POF) in Brazilian fragile X carriers. Genet. Mol. Biol. 1999, 22, 471–474. [Google Scholar] [CrossRef]
- Vianna-Morgante, A.M. Twinning and premature ovarian failure in premutation fragile X carriers. Am. J. Med. Genet. 1999, 83, 326. [Google Scholar] [CrossRef]
- Schwartz, C.E.; Dean, J.; Howard-Peebles, P.N.; Bugge, M.; Mikkelsen, M.; Tommerup, N.; Hull, C.; Hagerman, R.; Holden, J.J.; Stevenson, R.E. Obstetrical and gynecological complications in fragile X carriers: A multicenter study. Am. J. Med. Genet. 1994, 51, 400–402. [Google Scholar] [CrossRef] [PubMed]
- Murray, A.; Ennis, S.; MacSwiney, F.; Webb, J.; Morton, N.E. Reproductive and menstrual history of females with fragile X expansions. Eur. J. Hum. Genet. 2000, 8, 247–252. [Google Scholar] [CrossRef] [PubMed]
- Hunter, J.E.; Epstein, M.P.; Tinker, S.W.; Charen, K.H.; Sherman, S.L. Fragile X-associated primary ovarian insufficiency: Evidence for additional genetic contributions to severity. Genet. Epidemiol. 2008, 32, 553–559. [Google Scholar] [CrossRef] [PubMed]
- Hundscheid, R.D.; Smits, A.P.; Thomas, C.M.; Kiemeney, L.A.; Braat, D.D. Female carriers of fragile X premutations have no increased risk for additional diseases other than premature ovarian failure. Am. J. Med. Genet. Part A 2003, 117A, 6–9. [Google Scholar] [CrossRef] [PubMed]
- Chonchaiya, W.; Nguyen, D.V.; Au, J.; Campos, L.; Berry-Kravis, E.M.; Lohse, K.; Mu, Y.; Utari, A.; Hervey, C.; Wang, L.; et al. Clinical involvement in daughters of men with fragile X-associated tremor ataxia syndrome. Clin. Genet. 2010, 78, 38–46. [Google Scholar] [CrossRef] [PubMed]
- Elizur, S.E.; Lebovitz, O.; Derech-Haim, S.; Dratviman-Storobinsky, O.; Feldman, B.; Dor, J.; Orvieto, R.; Cohen, Y. Elevated levels of FMR1 mRNA in granulosa cells are associated with low ovarian reserve in FMR1 premutation carriers. PLoS ONE 2014, 9, e105121. [Google Scholar] [CrossRef] [PubMed]
- Ennis, S.; Ward, D.; Murray, A. Nonlinear association between CGG repeat number and age of menopause in FMR1 premutation carriers. Eur. J. Hum. Genet. 2006, 14, 253–255. [Google Scholar] [CrossRef] [PubMed]
- Hall, D.; Todorova-Koteva, K.; Pandya, S.; Bernard, B.; Ouyang, B.; Walsh, M.; Pounardjian, T.; Deburghraeve, C.; Zhou, L.; Losh, M.; et al. Neurological and endocrine phenotypes of fragile X carrier women. Clin. Genet. 2016, 89, 60–67. [Google Scholar] [CrossRef] [PubMed]
- Hundscheid, R.D.; Sistermans, E.A.; Thomas, C.M.; Braat, D.D.; Straatman, H.; Kiemeney, L.A.; Oostra, B.A.; Smits, A.P. Imprinting effect in premature ovarian failure confined to paternally inherited fragile X premutations. Am. J. Hum. Genet. 2000, 66, 413–418. [Google Scholar] [CrossRef] [PubMed]
- Hundscheid, R.D.; Braat, D.D.; Kiemeney, L.A.; Smits, A.P.; Thomas, C.M. Increased serum FSH in female fragile X premutation carriers with either regular menstrual cycles or on oral contraceptives. Hum. Reprod. 2001, 16, 457–462. [Google Scholar] [CrossRef] [PubMed]
- Mailick, M.R.; Hong, J.; Greenberg, J.; Smith, L.; Sherman, S. Curvilinear association of CGG repeats and age at menopause in women with FMR1 premutation expansions. Am. J. Med. Genet. Part B: Neuropsychiatr. Genet. 2014, 165, 705–711. [Google Scholar] [CrossRef] [PubMed]
- Mallolas, J.; Duran, M.; Sanchez, A.; Jimenez, D.; Castellvi-Bel, S.; Rife, M.; Mila, M. Implications of the FMR1 gene in menopause: Study of 147 Spanish women. Menopause 2001, 8, 106–110. [Google Scholar] [CrossRef] [PubMed]
- Murray, A.; Webb, J.; MacSwiney, F.; Shipley, E.L.; Morton, N.E.; Conway, G.S. Serum concentrations of follicle stimulating hormone may predict premature ovarian failure in FRAXA premutation women. Hum. Reprod. 1999, 14, 1217–1218. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Revenga, L.P.; Madrigal, I.P.; Badenas, C.P.; Xuncla, M.B.S.; Jimenez, L.P.; Mila, M.P. Premature ovarian failure and fragile X female premutation carriers: No evidence for a skewed X-chromosome inactivation pattern. Menopause Sept./Oct. 2009, 16, 944–949. [Google Scholar] [CrossRef] [PubMed]
- Rohr, J.; Allen, E.G.; Charen, K.; Giles, J.; He, W.; Dominguez, C.; Sherman, S.L. Anti-müllerian hormone indicates early ovarian decline in fragile X mental retardation (FMR1) premutation carriers: A preliminary study. Hum. Reprod. 2008, 23, 1220–1225. [Google Scholar] [CrossRef] [PubMed]
- Spath, M.A.; Nillesen, W.N.; Smits, A.P.; Feuth, T.B.; Braat, D.D.; van Kessel, A.G.; Yntema, H.G. X chromosome inactivation does not define the development of premature ovarian failure in fragile X premutation carriers. Am. J. Med. Genet. Part A 2010, 152A, 387–393. [Google Scholar] [CrossRef] [PubMed]
- Spath, M.A.; Feuth, T.B.; Smits, A.P.; Yntema, H.G.; Braat, D.D.; Thomas, C.M.; van Kessel, A.G.; Sherman, S.L.; Allen, E.G. Predictors and risk model development for menopausal age in fragile X premutation carriers. Genet. Med. 2011, 13, 643–650. [Google Scholar] [CrossRef] [PubMed]
- Tejada, M.I.; Garcia-Alegria, E.B.S.; Bilbao, A.; Martinez-Bouzas, C.; Beristain, E.B.S.; Poch, M.; Ramos-Arroyo, M.A.; Lopez, B.; Fernandez Carvajal, I.; Ribate, M.-P.; et al. Analysis of the molecular parameters that could predict the risk of manifesting premature ovarian failure in female premutation carriers of fragile X syndrome. Menopause 2008, 15, 945–949. [Google Scholar] [CrossRef] [PubMed]
- Welt, C.K.; Smith, P.C.; Taylor, A.E. Evidence of early ovarian aging in fragile X premutation carriers. J. Clin. Endocrinol. Metab. 2004, 89, 4569–4574. [Google Scholar] [CrossRef] [PubMed]
- Wheeler, A.C.; Raspa, M.; Green, A.; Bishop, E.; Bann, C.; Edwards, A.; Bailey, D.B., Jr. Health and reproductive experiences of women with an FMR1 premutation with and without fragile X premature ovarian insufficiency. Front. Genet. 2014, 5, 300. [Google Scholar] [CrossRef] [PubMed]
- Winarni, T.I.; Chonchaiya, W.; Sumekar, T.A.; Ashwood, P.; Morales, G.M.; Tassone, F.; Nguyen, D.V.; Faradz, S.M.; Van de Water, J.; Cook, K.; et al. Immune-mediated disorders among women carriers of fragile X premutation alleles. Am. J. Med. Genet. Part A 2012, 158A, 2473–2481. [Google Scholar] [CrossRef] [PubMed]
- Kenna, H.A.; Tartter, M.; Hall, S.S.; Lightbody, A.A.; Nguyen, Q.; de los Angeles, C.P.; Reiss, A.L.; Rasgon, N.L. High rates of comorbid depressive and anxiety disorders among women with premutation of the FMR1 gene. Am. J. Med. Genet. Part B Neuropsychiatr. Genet. 2013, 162B, 872–878. [Google Scholar] [CrossRef] [PubMed]
- Roberts, J.E.; Tonnsen, B.L.; McCary, L.M.; Ford, A.L.; Golden, R.N.; Bailey, D.B. Trajectory and predictors of depression and anxiety disorders in mothers with the FMR1 premutation. Biol. Psychiatry 2016, 79, 850–857. [Google Scholar] [CrossRef] [PubMed]
- Leehey, M.; Lee, W.; Tassone, F.; Hagerman, R. Fibromyalgia in fragile X mental retardation 1 gene premutation carriers. Rheumatology 2011, 50, 2233–2236. [Google Scholar] [CrossRef] [PubMed]
- Seritan, A.L.; Bourgeois, J.A.; Schneider, A.; Mu, Y.; Hagerman, R.J.; Nguyen, D.V. Ages of onset of mood and anxiety disorders in fragile X premutation carriers. Curr. Psychiatry Rev. 2013, 9, 65–71. [Google Scholar] [CrossRef] [PubMed]
- Schneider, A.; Johnston, C.; Tassone, F.; Sansone, S.; Hagerman, R.J.; Ferrer, E.; Rivera, S.M.; Hessl, D. Broad autism spectrum and obsessive-compulsive symptoms in adults with the fragile X premutation. Clin. Neuropsychol. 2016, 30, 929–943. [Google Scholar] [CrossRef] [PubMed]
- Hessl, D.; Tassone, F.; Loesch, D.Z.; Berry-Kravis, E.; Leehey, M.A.; Gane, L.W.; Barbato, I.; Rice, C.; Gould, E.; Hall, D.A.; et al. Abnormal elevation of FMR1 mRNA is associated with psychological symptoms in individuals with the fragile X premutation. Am. J. Med. Genet. B Neuropsychiatr. Genet. 2005, 139B, 115–121. [Google Scholar] [CrossRef] [PubMed]
| Paper | Groups | n | Mean Age | Features Studied | Findings |
|---|---|---|---|---|---|
| Allen et al., 2007 [23] | PMC—low repeats PMC—mid repeats PMC—high repeats NC | 127 237 70 521 | 42.7 (14.3) 45.8 (12.0) 35.8 (13.6) 56.1 (9.4) | POI Menstrual cycle Fertility Obstetric features | All PMC repeat lengths significantly associated with earlier premature ovarian failure (mid > high > low) Low and middle sized repeats show shorter and more skipped menstrual cycles. Mid-sized repeats showed irregular menstrual cycles. Low repeat lengths show early menarche. Mid-sized repeats associated with reduced fertility. Mid-sized repeats associated with increased DZ twinning. NSD in spontaneous abortion rates. |
| Allingham Hawkins et al., 1999 [17] | PMC FXS NC | 395 128 237 | - | Early menopause | Increased in PMC |
| Chonchaiya et al., 2010 [31] | PMC w parent w FXTAS NC | 110 43 | 44.8 (8.2) 43.8 (8.1) | POI Menopausal symptoms Infertility | NSD Increased in PMC NSD |
| Coffey et al., 2008 [7] | PMC w FXTAS PMC w/o FXTAS NC | 15 63 36 | - - - | POI | NSD regardless of FXTAS status |
| Elizur et al., 2014 [32] | PMC NC | 21 15 | 31.5 (3.4) 30.8 (4.3) | Response to controlled ovarian hyperstimulation | PMC had higher FSH:LH and gonadotrophin dosage and fewer retrieved oocytes. Number of retrieved oocytes showed non-linear association with repeat number and negative correlation with granulosa cell FMR1 mRNA in PMCs. |
| Ennis et al., 2006 [33] | PMC | 45 | - | Early menopause | Curvilinear association with CGG number |
| Hall et al., 2016 [34] | PMC NC | 33 13 | 54.2 (16.8) 47.0 (10.1) | Ovarian dysfunction | NSD |
| Hundscheid et al., 2000 [35] | PMC (paternal origin) PMC (maternal origin) | 82 27 | - - | Early menopause/POI | Earlier/increased in PMC with paternally inherited mutation |
| Hundscheid et al., 2001 [36] | PMC not on OCP NC/FXS not on OCP PMC on OCP NC/FXS on OCP | 17 28 13 21 | 36 (2.2) 34 (4.7) 34 (5.3) 31 (4.5) | FSH Inhibin B 17β oestradiol | Higher in PMC regardless of contraceptive use NSD NSD |
| Hundscheid et al., 2003 [30] | PMC NC | 152 112 | 52.7 (13.5) 41.1 (12.5) | Early menopause/POI Obstetric features | Increased in PMC NSD in number of pregnancies, children, spont. abortion, fetal loss or twinning |
| Hunter et al., 2008 [29] | PMC—low repeats PMC—mid repeats PMC—high repeats NC | 134 248 78 541 | 49.1 (15.9) 43.3 (12.6) 38.8 (11.7) 42.4 (15.9) | Early menopause | Compared to NC, rates of early menopause highest in with mid-sized repeats (4x) then high and low sized repeats (2x). Significant genetic component to age of menopause even after controlling for PMC status. |
| Mailick et al., 2014 [37] | Postmenopausal PMC | 88 | - | Age at menopause | Curvilinear association between age at menopause and repeat length. NS relationship between menopause age and X inactivation ratio. |
| Mallolas et al., 2001 [38] | PMC w POI/early menopause PMC w/o POI/early menopause | 21 206 | - - | Parental origin of mutation | NSD |
| Murray et al. 1999 [39] | PMC NC/FXS | 19 32 | FSH | Increased FSH levels in PMC group | |
| Murray et al., 2000 [28] | PMC NC/FXS | 116 236 | - | Early menopause Obstetric features Inhibin | Menopause occurs at significantly younger age in PMC. Repeat size and skewed X inactivation not related to age of menopause. Subset showed NSD in twinning or unfavourable pregnancy outcome. No relationship between repeat size and inhibin concentrations in PMC. |
| Rodriguez-Revenga et al., 2009 [40] | PMC w POI PMC w/o POI NC | 40 220 220 | - | POI | No relationship between skewed X-inactivation and POI. |
| Rohr et al., 2008 [41] | PMC < 70 repeats PMC > 70 repeats | - - | 18–50 18–50 | FSH AMH | PMC with >70 repeats had higher FSH levels than those with <70 repeats in 31–40 year old age group PMC with >70 repeats had higher AMH levels than those with <70 repeats in all age groups tested |
| Schwartz et al., 1994 [27] | PMC FXS NC (relatives) NC (non-relatives) | 92 39 109 50 | 46 33 37 33 | Menstrual cycle Early menopause Gynaecological complications Spontaneous abortions | More irregular in PMC Increased in PMC Increased in PMC compared to related NCs NSD |
| Spath et al., 2010 [42] | PMC w POI PMC w/o POI NC w POI | 37 64 25 | 45.9 (13.2) 47.1 (12.7) 31.7 (19.5) | POI | No relationship between skewed X-inactivation and POI |
| Spath et al., 2011 [43] | PMC NC | 517 551 | 51.5 53.5 | Age at menopause | Within PMC, age at menopause significantly predicted using multivariate analysis by CGG repeat length, mean menopausal age of first degree relatives and smoking. CGG repeat length showed non-linear relationship with risk increased between 62 and 120 repeats. |
| Sullivan et al., 2005 [24] * | PMC NC | 183 324 | 44.3 (13.5) 42.3 (14.6) | POI/early menopause FSH | PMC at increased risk of POI and early menopause compared to NC; highest rates with mid-sized repeat lengths; no association between POI/early menopause and parental origin. Subgroup showed no increase in FSH in PMC but further sub-analysis showed increased levels in PMC aged 30–39 years old. No relationship between FSH and repeat length, X inactivation ratio or parental origin. |
| Tejada et al., 2008 [44] | PMC w POI PMC w/o POI | 25 17 | - - | POI | NSD in mRNA, X inactivation ratio and CGG repeat lengths. POI associated most with >100 repeats. |
| Vianna-Morgante et al., 1999 [25] | PMC FXS NC | 101 37 55 | 43.3 (11.8) 36.6 (9.4) 40.4 (12.7) | Early menopause/POI | PMC at increased risk of early menopause compared to FXS and NC. No relationship between POI and parental origin. |
| Welt et al., 2004 [45] | PMC NC | 11 22 | 34.5 (5.7) 34.6 (5.8) | Menstrual cycle FSH LH Inhibin A Inhibin B Oestradiol Progesterone | Reduced total cycle length in PMC driven by reduced follicular phase Increased in PMC throughout follicular and luteal phases NSD Decreased in PMC in early and mid-luteal phases Decreased in PMC in early and mid-follicular and early luteal phases NSD Decreased in PMC in follicular phase |
| Wheeler et al., 2014 [46] | PMC w POI PMC w/o POI | 73 365 | 48.6 (11.7) 48.9 (12.2) | Menstrual cycle Fertility Obstetric features | POI associated with absent or irregular menstrual periods POI associated with greater use reproductive assistance/fertility drugs POI associated with lower rates of precipitous labour (under 2 h) |
| Paper | Groups | n | Mean Age | Features Studied | Findings |
|---|---|---|---|---|---|
| Chonchaiya et al., 2010 [31] | PMC w parent w FXTAS NC | 110 43 | 44.8 (8.2) 43.8 (8.1) | Thyroid disease Diabetes | NSD NSD |
| Coffey et al., 2008 [7] | PMC w FXTAS PMC w/o FXTAS NC | 18 127 69 | 59.2 (10.3) 42.3 (11.5) 45.8 (14.9) | Thyroid disease Type II diabetes | Increased only in PMC w FXTAS when compared to subset of age matched controls (57.1 years old) NSD |
| Hall et al., 2016 [34] | PMC NC | 33 13 | 54.2 (16.8) 47.0 (10.1) | Thyroid disease Pituitary/adrenal dysfunction Thyroid antibodies Low 8 a.m. cortisol Prediabetes | NSD None detected in either group NSD NSD NSD |
| Hundscheid et al., 2003 [30] | PMC NC | 152 112 | 52.7 (13.5) 41.1 (12.5) | Thyroid disease Type II diabetes | NSD NSD |
| Hunter et al., 2010 [6] | PMC NC | 203 334 | 37.1 (8.4) 32.5 (10.1) | Thyroid disease Type II diabetes | NSD. Presence of irregular menstruation significantly associated with presence of thyroid problems NSD |
| Winarni et al. (2012) [47] | PMC w FXTAS PMC w/o FXTAS NC PMC w FXTAS PMC w/o FXTAS NC PMC w POI PMC w/o POI NC | 56 288 72 45 158 57 41 147 50 | - - - >40 >40 >40 >40 >40 >40 | Autoimmune thyroid disease | PMC w FXTAS > PMC w/o FXTAS = NC PMC w FXTAS > PMC w/o FXTAS = NC NSD |
| Paper | Groups | n | Mean Age | Features studied | Findings |
|---|---|---|---|---|---|
| Hunter et al., 2010 [6] | PMC NC | 203 334 | 37.1 (8.4) 32.5 (10.1) | Anxiety Depression | Ovarian reserve did not predict either depression or anxiety in PMCs |
| Kenna et al., 2013 [48] | Premenopausal PMC Perimenopausal PMC Postmenopausal PMC | 17 7 22 | 46.2 (6.2) 43.7 (5.7) 50.1 (4.1) | Anxiety Depression | NSD between groups; no significant relationship between prevalence of anxiety or depression and age of menopause or postmenopausal use of HRT |
| Roberts et al., 2016 [49] | PMC w/POI PMC w/o POI | 34 49 | - - | Anxiety Depression | POI did not significantly predict anxiety POI showed trend towards significantly predicting depression |
| Wheeler et al., 2014 [46] | PMC w POI PMC w/o POI | 73 365 | 48.9 (12.2) 48.6 (11.7) | Anxiety Depression ADHD Learning disability Speech/language dis. Hypertension Heart disease Diabetes Autoimmune disorder Thyroid disease GI problems Seizures Specific physical symptoms | NSD NSD NSD NSD NSD NSD NSD NSD NSD NSD NSD NSD PMC with POI reported significantly more muscle weakness, dizziness and nausea |
| Winarni et al. (2012) [47] | PMC w POI PMC w/o POI NC | 41 147 50 | >40 >40 >40 | Immune mediated disorders | PMC w POI > PMC w/o POI > NC |
| Group | Endocrine | Non-endocrine | ||||||
|---|---|---|---|---|---|---|---|---|
| Analysis | PMC | PMC | NC | p | ||||
| % | n = 18 | % | n = 14 | % | n = 15 | |||
| Medication | ||||||||
| Takes medication | 61.1 | 18 | 50 | 14 | 28.6 | 14 | 0.20 | |
| Number of medications taken | ||||||||
| 0–3 | 72.2 | 18 | 92.9 | 14 | 85.7 | 14 | ||
| 3+ | 27.8 | 7.1 | 14.3 | 0.30 | ||||
| Type of medication used | ||||||||
| Cardiovascular | 0 | 17 | 7.2 | 14 | 7.1 | 14 | 0.52 | |
| Digestive | 38.9 | 18 | 21.4 | 14 | 13.3 | 15 | 0.26 | |
| Metabolic | 5.9 | 17 | 7.1 | 14 | 7.1 | 14 | 1.00 | |
| Endocrine | 17.6 | 18 | 0 | 14 | 0 | 15 | 0.10 | |
| Neurological | 11.8 | 18 | 14.3 | 14 | 0 | 15 | 0.44 | |
| Mental health | 29.4 | 18 | 21.4 | 14 | 14.3 | 14 | 0.61 | |
| Analgesic | 29.4 | 18 | 7.1 | 14 | 0 | 15 | 0.04 * | |
| Other | 23.5 | 18 | 21.4 | 14 | 7.1 | 14 | 0.50 | |
| Health difficulties | ||||||||
| Number of health difficulties | 18 | 15 | ||||||
| 0–3 | 55.6 | 92.9 | 14 | 86.7 | ||||
| 4+ | 44.4 | 7.1 | 14 | 13.3 | 0.04 * | |||
| Cardiovascular | 27.8 | 18 | 7.1 | 14 | 13.3 | 15 | 0.36 | |
| Digestive | 38.9 | 18 | 21.4 | 14 | 13.3 | 15 | 0.26 | |
| Immune | 22.2 | 18 | 21.4 | 14 | 26.7 | 15 | 1.00 | |
| Muscular/skeletal | 27.8 | 18 | 7.1 | 14 | 20.0 | 15 | 0.38 | |
| Respiratory | 5.6 | 18 | 14.3 | 14 | 0 | 15 | 0.38 | |
| Sensory | 27.8 | 18 | 14.3 | 14 | 33.3 | 15 | 0.54 | |
| Neurological | 72.2 | 18 | 28.6 | 14 | 46.7 | 15 | 0.049 * | |
| Tremor | 22.2 | 18 | 0 | 14 | 0 | 15 | 0.03 * | |
| Headache | 55.6 | 18 | 7.1 | 14 | 46.7 | 15 | 0.01 * | |
| Paraesthesia | 22.2 | 18 | 0 | 14 | 6.7 | 15 | 0.17 | |
| Seizures | 5.6 | 18 | 23.1 | 13 | 0 | 15 | 0.09 | |
| Mental health | 77.8 | 18 | 35.7 | 14 | 66.7 | 15 | 0.06 | |
| Anxiety/depression | 58.8 | 17 | 35.7 | 14 | 71.4 | 14 | 0.19 | |
| OCD | 44.4 | 18 | 0 | 14 | 23.1 | 13 | 0.009 * | |
| Pain-associated conditions | 55.6 | 18 | 14.3 | 14 | 26.7 | 15 | 0.045 * | |
| Arthritis | 16.7 | 18 | 0 | 14 | 20.3 | 15 | 0.29 | |
| Fibromyalgia | 22.2 | 18 | 0 | 14 | 0 | 15 | 0.03* | |
| Endometriosis | 0 | 18 | 0 | 14 | 0 | 15 | N/A | |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Campbell, S.; Eley, S.E.A.; McKechanie, A.G.; Stanfield, A.C. Endocrine Dysfunction in Female FMR1 Premutation Carriers: Characteristics and Association with Ill Health. Genes 2016, 7, 101. https://doi.org/10.3390/genes7110101
Campbell S, Eley SEA, McKechanie AG, Stanfield AC. Endocrine Dysfunction in Female FMR1 Premutation Carriers: Characteristics and Association with Ill Health. Genes. 2016; 7(11):101. https://doi.org/10.3390/genes7110101
Chicago/Turabian StyleCampbell, Sonya, Sarah E. A. Eley, Andrew G. McKechanie, and Andrew C. Stanfield. 2016. "Endocrine Dysfunction in Female FMR1 Premutation Carriers: Characteristics and Association with Ill Health" Genes 7, no. 11: 101. https://doi.org/10.3390/genes7110101
APA StyleCampbell, S., Eley, S. E. A., McKechanie, A. G., & Stanfield, A. C. (2016). Endocrine Dysfunction in Female FMR1 Premutation Carriers: Characteristics and Association with Ill Health. Genes, 7(11), 101. https://doi.org/10.3390/genes7110101






