Biological Complexities in Radiation Carcinogenesis and Cancer Radiotherapy: Impact of New Biological Paradigms
Abstract
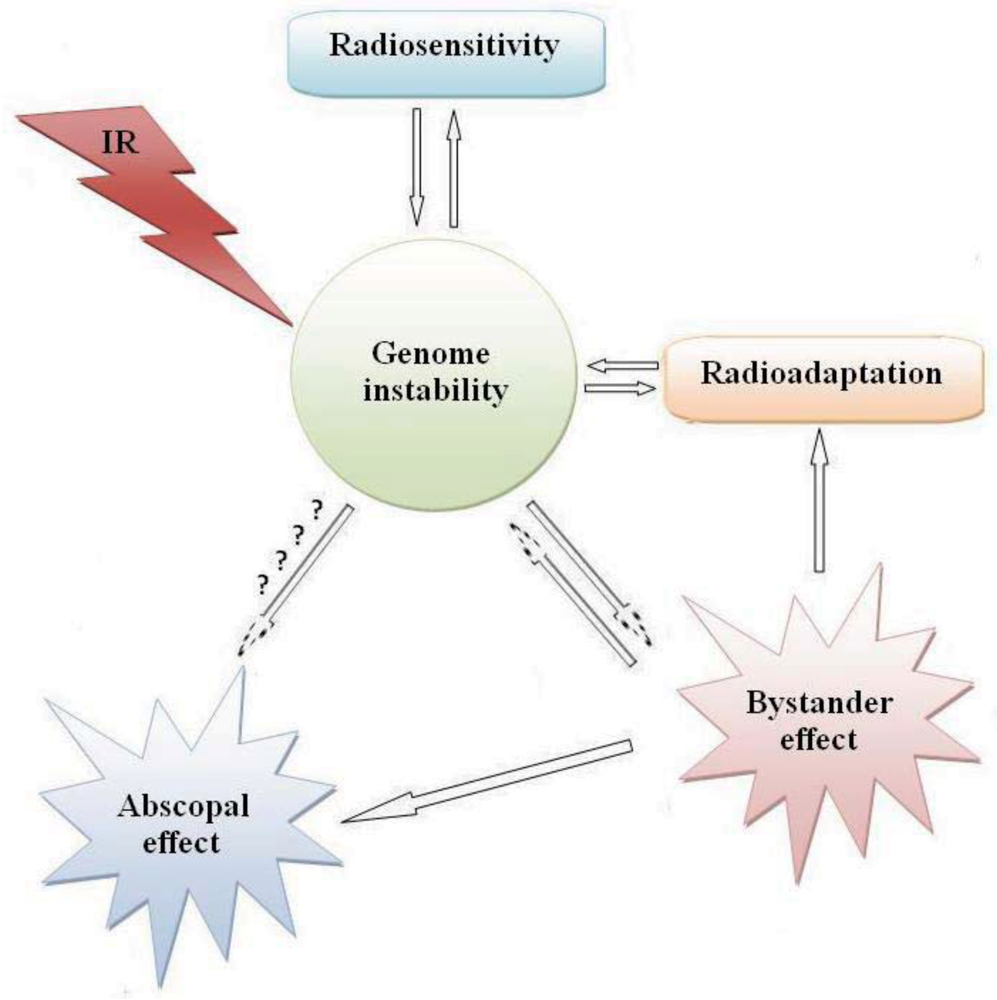
1. Introduction
2. Radiation Carcinogenesis
3. Impact of Radiosensitivity on Carcinogenesis and Cancer Therapy
4. Radioadaptation and Carcinogenesis
5. Bystander Effect and Carcinogenesis
6. Distant Bystander (Abscopal) Effect
7. Conclusions
References
- Upton, A.C. Historical perspective on radiation carcinogenesis. In Radiation Carcinogenesis; Upton, A.C., Alber, R.E., Burns, F.J., Shore, R.E., Eds.; Elsevier: New York, NY, USA, 1986; pp. 1–10. [Google Scholar]
- Jacob, P.; Bogdanova, T.I.; Buglova, E.; Chepurniy, M.; Demidchik, Y.; Gavrilin, Y.; Kenigsberg, J.; Meckbach, R.; Schotola, C.; Shinkarev, S.; et al. Thyroid cancer risk in areas of Ukraine and Belarus affected by the Chernobyl accident. Radiat. Res. 2006, 165, 1–8. [Google Scholar] [CrossRef]
- National Research Council, Health Effects of Exposure to Low Levels of Ionizing Radiation, BEIR V; National Academy Press: Washington, DC, USA, 1990.
- Shadley, J.D.; Wolff, S. Very low doses of X-rays can cause human lymphocytes to become less susceptible to ionizing radiation. Mutagenesis 1987, 2, 95–96. [Google Scholar]
- Wang, Z.; Saigusa, S.; Sasaki, M.S. Adaptive response to chromosome damage incultured human lymphocytes primed with low doses of X-rays. Mutat. Res. 1991, 246, 179–186. [Google Scholar]
- Mortazavi, S.M.J.; Mozdarani, H. The search for a possible optimum adapting dose under the optimum irradiation time scheme in cultured human lymphocytes. Int. J. Low Radiat. 2006, 3, 74–82. [Google Scholar]
- Morgan, W.F.; Sowa, M.B. Non-targeted bystander effects induced by ionizing radiation. Mutat. Res. 2007, 616, 159–164. [Google Scholar]
- Bowater, R.; Doherty, A.J. Making ends meet: Repairing breaks in bacterial DNA by non-homologous end-joining. PloS Genet. 2006, 2, e8. [Google Scholar]
- Iliakis, G.; Wu, W.; Wang, M.; Terzoudi, G.I.; Pantelias, G.E. Backup pathways of nonhomologous end joining may have a dominant role in the formation of chromosome aberrations. In Chromosmeal Alterations: Methods, Results and Importance in Human Health; Obe, G., Vijayalaxmi, Eds.; Springer-Verlag: Berlin, Germany, 2007; pp. 67–85. [Google Scholar]
- Obe, G.; Pfeiffer, P.; Savage, J.R.K.; Johannes, C.; Goedecke, W.; Jeppesen, P.; Natarajan, A.T.; Martinez-Lopez, W.; Folle, G.A.; Derts, M.E. Chromosomal aberrations: Formation, identification and distribution. Mutat. Res. 2002, 504, 17–36. [Google Scholar]
- Goedeccke, W. Double starnd break repair mechanisms in mammalian cells. In Chromoosmeal Alterations: Methods, Results and Importance in Human Health; Obe, G., Vijayalaxmi, Eds.; Springer-Verlag: Berlin, Germany, 2007; pp. 55–66. [Google Scholar]
- Heyer, W.D.; Li, X.; Rolfsmeier, M.; Zhang, X.P. Rad54: The Swiss Army Knife of homologous recombinations? Nucleic Acids Res. 2006, 34, 4115–4125. [Google Scholar]
- Ruis, B.L.; Fattah, K.R.; Hendrickson, E.A. The catalytic subunit of DNA-dependent protein kinase regulates proliferation, telomere length, and genomic stability in human somatic cells. Mol. Cell Biol. 2008, 28, 6182–6195. [Google Scholar]
- Fukasawa, K.; Wiener, F.; Vande Woude, G.F.; Mai, S. Genomic instability and apoptosis are frequent in p53 deficient young mice. Oncogene 1997, 15, 1295–1302. [Google Scholar]
- Brusa, G.; Benvenuti, M.; Mazzacurati, L.; Mancini, M.; Pattacini, L.; Martinelli, G.; Barbieri, E.; Greenberger, J.S.; Baccarani, M.; Santucci, M.A. p53 loss of function enhances genomic instability and accelerates clonal evolution of murine myeloid progenitors expressing the p(210)BCR-ABL tyrosine kinase. Haematologia 2003, 88, 622–630. [Google Scholar]
- Dimitrova, N.; de Lange, T. Cell cycle-dependent role of MRN at dysfunctional telomeres: ATM signaling-dependent induction of nonhomologous end joining (NHEJ) in G1 and resection-mediated inhibition of NHEJ in G2. Mol. Cell Biol. 2009, 29, 5552–5563. [Google Scholar]
- Greenwood, J.; Cooper, J.P. Trapping Rap1 at the telomere to prevent chromosome end fusions. EMBO J. 2009, 28, 3277–3278. [Google Scholar]
- Bailey, S.M.; Meyne, J.; Chen, D.J.; Kurimasa, A.; Li, G.C.; Lehnert, B.E.; Goodwin, E.H. DNA double-strand break repair proteins are required to cap the ends of mammalian chromosomes. Proc. Natl. Acad. Sci. USA 1999, 96, 14899–14904. [Google Scholar]
- Hsiao, S.J.; Smith, S. Sister telomeres rendered dysfunctional by persistent cohesion are fused by NHEJ. J. Cell Biol. 2009, 184, 515–526. [Google Scholar]
- Bender, M.A.; Griggs, H.G.; Bedford, J.S. Mechanisms of chromosomal aberration production: III. Chemical and ionizing radiation. Mutat. Res. 1974, 23, 197–212. [Google Scholar]
- Natarajan, A.T.; Obe, G.; van Zeeland, A.A.; Palitti, F.; Meijers, M.; Verdegaal-Immerzell, E.A.M. Molecular mechanisms involved in the production of chromosomal aberrations. II. Utilization of Neurospora endonuclease for the study of aberration production by X-rays in G1 and G2 stages of the cell cycle. Mutat. Res. 1980, 69, 293–305. [Google Scholar] [CrossRef]
- Mozdarani, H.; Bryant, P.E. The effects of 9-b-D arabinofuranosylanine on the formation of X-ray induced chromatid aberrations in X-irradiated G2 human cells. Mutagenesis 1987, 2, 371–374. [Google Scholar]
- Mozdarani, H.; Bryant, P.E. Induction and rejoining of chromatid breaks in X-irradiated A-T and normal human G2 fibroblasts. Int. J. Radiat. Biol. 1989, 56, 645–659. [Google Scholar]
- Kemp, L.M.; Jeggo, P.A. Radiation induced chromosome damage in X-ray sensitive mutants (xrs) of the Chinese hamster ovary cell line. Mutat. Res. DNA Repair Rep. 1986, 166, 255–263. [Google Scholar]
- Bryant, P.E.; Birch, D.A.; Jeggo, P.A. High chromosomal sensitivity of Chinese hamster xrs5 cells to restriction endonuclease induced DNA dsb. Int. J. Radiat. Biol. 1987, 52, 537–554. [Google Scholar]
- Bryant, P.E.; Finnegan, C.E.; Mozdarani, H. G2 chromatid breaks in murine scid cells. Mutagenesis 1998, 13, 481–485. [Google Scholar]
- Natarajan, A.T.; Obe, G. Molecular mechanisms involved in the production of chromosomal aberrations. III. Restriction endonucleases. Chromosoma 1984, 90, 120–127. [Google Scholar] [CrossRef]
- Bryant, P.E. Use of restriction endonucleases to study relationship between DNA double strand breaks, chromosomal aberrations and other end points in mammalian cells. Int. J. Radiat. Biol. 1988, 54, 869–890. [Google Scholar]
- Heim, S.; Mitelman, F. Cancer Cytogenetics, 3rd ed; Wiley-Blackwell: Hoboken, NJ, USA, 2009. [Google Scholar]
- Fusco, A.; Santoro, M. 20 years of RET/PTC in thyroid cancer: Clinico-pathological correlations. Arq. Bras. Endocrinol. Metabol. 2007, 51, 731–735. [Google Scholar]
- Viglietto, G.; Chiappetta, G.; Martinez-Tello, F.J.; Fukunaga, F.H.; Tallini, G.; Rigopoulou, D.; Visconti, R.; Mastro, A.; Santoro, M.; Fusco, A. RET/PTC oncogene activation is an early event in thyroid carcinogenesis. Oncogene 1995, 11, 1207–1210. [Google Scholar]
- Nikiforova, M.N.; Stringer, J.R.; Blough, R.; Medvedovic, M.; Fagin, J.A.; Nikiforov, Y.E. Proximity of chromoso-mal loci that participate in radiation-induced rearrangements in human cells. Science 2000, 290, 138–141. [Google Scholar]
- Nikiforov, Y.; Rowland, J.M.; Bove, K.E.; Monfore Munoz, H.; Fagin, J.A. Distinct patterns of ret rearrangements in morphological variants of radiation-induced and sporadic thyroid papillary carcinomas in children. Cancer Res. 1997, 57, 1690–1694. [Google Scholar]
- Santoro, M.; Melillo, R.M.; Fusco, A. RET/PTC activation in papillary thyroid carcinoma. Eur. J. Endocrinol. 2006, 155, 645–653. [Google Scholar]
- Elisei, R.; Romel, C.; Vorontsova, T.; Cosci, B.; Veremeychick, V.; Kuchinskaya, E.; Basolo, F.; Demidchik, E.P.; Miccoli, P.; Pinchera, A.; et al. RET/PTC rearrangments in tyroid nodules: Studies in irradiated and not irradiated, malignant and benign thyroid lesions in children and adults. J. Clin. Endocrinol. Metab. 2001, 86, 3211–3216. [Google Scholar]
- Hagmar, L.; Stromberg, U.; Bonassi, S.; Hansteen, I.L.; Knudsen, L.E.; Lindholm, C.; Norppa, H. Impact of types of lymphocyte chromosomal aberrations on human cancer risk: Results from Nordic and Italian cohorts. Cancer Res. 2004, 64, 2258–2263. [Google Scholar]
- Bonassi, S.; Abbondandolo, A.; Camurri, L.; Dal Prá, L.; de Ferrari, M.; Degrassi, F.; Forni, A.; Lamberti, L.; Lando, C.; Padovani, P.; et al. Are chromosome aberrations in circulating lymphocytes predictive of future cancer onset in humans? Preliminary results of an Italian cohort study. Cancer Genet. Cytogenet. 1995, 79, 133–135. [Google Scholar]
- Rabbitts, T.H. Chromosomal translocations in human cancer. Nature 1994, 372, 143–149. [Google Scholar]
- Auletta, A.; Ashby, J. Workshop on the relationship between short-term information and carcinogenicity. Environ. Mol. Mutagen. 1988, 11, 135–145. [Google Scholar]
- Shelby, M. The genetic toxicity of human carcinogens and its implications. Mutat. Res. 1988, 204, 3–15. [Google Scholar]
- Au, W.W.; Lane, R.G.; Legator, M.S.; Whorton, E.B.; Wilkinson, G.S.; Gaberhort, G.J. Biomarker monitoring of a population residing near uranium mining activities. Environ. Health Perspect. 1995, 103, 466–470. [Google Scholar]
- Mondello, C.; Smirnova, A.; Giulotto, E. Gene amplification, radiation sensitivity and DNA double-strand breaks. Mutat. Res. 2010, 704, 29–37. [Google Scholar]
- ICRP, Annual Report of the International Commission on Radiological Protection; Elsevier Science: New York, NY, USA, 1999.
- Djuzenova, C.S.; Muhl, B.; Fehn, M.; Oppitz, U.; Muller, B.; Flentje, M. Radiosensitivity in breast cancer assessed by the Comet and micronucleus assays. Br. J. Cancer 2006, 94, 1194–1203. [Google Scholar]
- Shahidi, M.; Mozdarani, H.; Bryant, P.E. Radiation sensitivity of leukocytes from healthy individuals and breast cancer patients as measured by the alkaline and neutral comet assay. Cancer Lett. 2007, 257, 263–273. [Google Scholar]
- Shahidi, M.; Mozdarani, H.; Mueller, W.U. Radiosensitivity and repair kinetics of gamma-irradiated leukocytes from sporadic prostate cancer patients and healthy individuals assessed by alkaline comet assay. Iran. Biomed. J. 2010, 14, 67–75. [Google Scholar]
- Lengauer, C.; Kinzler, K.W.; Vogelstein, B. Genetic instabilities in human cancers. Nature 1998, 396, 643–649. [Google Scholar]
- Thompson, L.H.; Schild, D. Recombinational DNA repair and human disease. Mutat. Res. 2002, 509, 49–78. [Google Scholar]
- Rothfuss, A.; Schütz, P.; Bochum, S.; Volm, T.; Eberhardt, E.; Kreienberg, R.; Vogel, W.; Speit, G. Induced micronucleus frequencies in peripheral lymphocytes as a screening test for carriers of a BRCA1 mutation in breast cancer families. Cancer Res. 2000, 60, 390–394. [Google Scholar]
- Carney, J.P. Chromosomal breakage syndromes. Curr. Opin. Immunol. 1999, 11, 443–447. [Google Scholar]
- Pichierri, P.; Franchitto, A.; Palitti, F. Predisposition to cancer and radiosensitivity. Genet. Mol. Biol. 2000, 23, 1101–1105. [Google Scholar]
- Mohseni, A.; Mozdarani, H.; Mozdarani, S. DNA damage and repair of leukocytes from Fanconi anemia patients, carriers and healthy individuals as measured by the alkaline comet assay. Mutagenesis 2009, 24, 67–73. [Google Scholar]
- Taylor, A.M.R. Chromosome instability syndromes. Best Pract. Res. Clin. Haematol. 2001, 14, 3631–3644. [Google Scholar]
- Duker, N.J. Chromosome breakage syndromes and cancer. Am. J. Med. Genet. 2002, 115, 125–129. [Google Scholar]
- Higurashi, M.; Conen, P.E. In vitro chromosomal radiosensitivity in chromosomal breakage syndromes. Cancer 1973, 32, 380–383. [Google Scholar]
- Lavin, M.F.; Shiloh, Y. The genetic defect in ataxia-telangiectasia. Ann. Rev. Immunol. 1997, 15, 177–202. [Google Scholar]
- Carney, J.P.; Maser, R.S.; Olivares, H.; Davis, E.M.; Le Beau, M.; Yates, J.R., III; Hays, L.; Morgan, W.F.; Petrini, J.H.J. The hMre11/hRad50 protein complex and Nijmegen breakage syndrome: Linkage of double-strand break repair to the cellular DNA damage response. Cell 1998, 93, 477–486. [Google Scholar] [CrossRef]
- Varon, R.; Vissinga, C.; Platzer, M.; Cerosaletti, K.M.; Chrzanowska, K.H.; Saar, K.; Beckmann, G.; Seemanová, E.; Cooper, P.R.; Nowak, N.J.; et al. Nibrin, a novel DNA double-strand break repair protein, is mutated in Nijmegen breakage syndrome. Cell 1998, 93, 467–476. [Google Scholar]
- Stewart, G.S.; Maser, R.S.; Stankovic, T.; Bressan, D.A.; Kaplan, M.I.; Jaspers, N.G.J.; Raams, A.; Byrd1, P.J.; Petrini, J.H.J.; Taylor, A.M.R. The DNA double-strand break repair gene hmre11 is mutated in individuals with an ataxia-telangiectasia-like disorder. Cell 1999, 99, 577–587. [Google Scholar]
- Ellis, N.A.; Groden, J.; Ye, T.-Z.; Straughen, J.; Lennon, D.J.; Ciocci, S.; Proytcheva, M.; German, J. The Bloom’s syndrome gene product is homologous to RecQ helicases. Cell 1995, 83, 655–666. [Google Scholar] [CrossRef]
- Gray, M.D.; Shen, J.C.; Kamath-Loeb, A.S.; Blank, A.; Sopher, B.L.; Martin, G.M.; Oshima, J.; Loeb, L.A. The Werner syndrome protein is a DNA helicase. Nat. Genet. 1997, 17, 100–103. [Google Scholar]
- Howlett, N.G.; Taniguchi, T.; Olson, S.; Cox, B.; Waisfisz, Q.; de Die-Smulders, C.; Persky, N.; Grompe, M.; Joenje, H.; Pals, G.; et al. Biallelic inactivation of BRCA2 in Fanconi anemia. Science 2002, 297, 606–609. [Google Scholar]
- Patel, K.J.; Yu, V.P.; Lee, H.; Corcoran, A.; Thistlethwaite, F.C.; Evans, M.J.; Colledge, W.H.; Friedman, L.S.; Ponder, B.A.; Venkitaraman, A.R. Involvement of Brca2 in DNA repair. Mol. Cell. 1998, 1, 347–357. [Google Scholar]
- Stewart, G.; Elledge, S.J. The two faces of BRCA2, a FANCtastic discovery. Mol. Cell. 2002, 10, 2–4. [Google Scholar]
- Meybodi, M.A.; Mozdarani, H.; Vosogh, P. Cytogenetic sensitivity of G0 lymphocytes of Fanconi anemia patients and obligate carriers to mytomycin C and ionizing radiation. Cytogenet. Genome Res. 2007, 119, 191–195. [Google Scholar]
- Meybodi, M.A.; Mozdarani, H. DNA Damage in leukocytes from fanconi anemia (FA) patients and heterozygotes induced by mitomycin C and ionizing radiation as assessed by the comet and comet-FISH assay. Iran. Biomed. J. 2009, 13, 1–8. [Google Scholar]
- Sanford, K.K.; Parshad, R.; Price, F.M.; Gantt, R.; Jones, G.M.; Tarone, R.E. Deficient G2 phase repair of radiation-induced chromatin damage in the SENCAR mouse. Carcinogenesis 1989, 10, 1911–1916. [Google Scholar]
- Sanford, K.K.; Parshad, R.; Gantt, R.R.; Tarone, R.E. A deficiency in chromatin repair, genetic instability, and predisposition to cancer. Crit. Rev. Oncog. 1989, 1, 323–341. [Google Scholar]
- Parshad, R.; Price, F.M.; Bohr, V.A.; Cowans, K.H.; Zujewski, J.A.; Sanford, A.A. Deficient DNA repair capacity, a predisposing factor in breast cancer. Br. J. Cancer 1996, 74, 1–5. [Google Scholar]
- Baeyens, A.; Thierens, H.; Claes, K.; Poppe, B.; Messiaen, L.; de Ridder, L.; Vral, A. Chromosomal radiosensitivity in breast cancer patients with a known or putative genetic predisposition. Br. J. Cancer 2002, 87, 1379–1385. [Google Scholar]
- Scott, D. Chromosomal radiosensitivity and low penetrance predisposition to cancer. Cytogenet. Genome Res. 2004, 104, 365–370. [Google Scholar]
- Scott, D.; Barber, J.B.; Spreadborough, A.R.; Burrill, W.; Roberts, S.A. Increased chromosomal radiosensitivity in breast cancer patients: A comparison of two assays. Int. J. Radiat. Biol. 1999, 75, 1–10. [Google Scholar]
- Sánchez, P.; Peñarroja, R.; Gallegos, F.; Bravo, J.L.; Rojas, E.; Benítez-Bribiesca, L. DNA damage in peripheral lymphocytes of untreated breast cancer patients. Arch. Med. Res. 2004, 35, 480–483. [Google Scholar]
- Mozdarani, H.; Mansouri, Z.; Haeri, S.A. Cytogenetic radiosensitivity of G0-lymphocytes of breast and esophageal cancer patients using the micronucleous assay. J. Radiat. Res. 2005, 46, 111–116. [Google Scholar]
- Helzlsouer, K.J.; Harris, E.L.; Parshad, R.; Perry, H.R.; Price, F.M.; Sanford, K.K. DNA repair proficiency: Potential susceptiblity factor for breast cancer. J. Natl. Cancer Inst. 1996, 88, 754–755. [Google Scholar]
- Patel, R.K.; Trivedi, A.H.; Arora, D.C.; Bhatavdekar, J.M.; Patel, D.D. DNA repair proficiency in breast cancer patients and their first-degree relatives. Int. J. Cancer 1997, 73, 20–24. [Google Scholar]
- Cybulski, C.; Huzarski, T.; Górski, B.; Masojć, B.; Mierzejewski, M.; Debniak, T.; Gliniewicz, B.; Matyjasik, J.; Złowocka, E.; Kurzawski, G.; et al. A novel founder CHEK2 mutation is associated with increased prostate cancer risk. Cancer Res. 2004, 64, 2677–2679. [Google Scholar]
- Angèle, S.; Jones, C.; Reis Filho, J.S.; Fulford, L.G.; Treilleux, I.; Lakhani, S.R.; Hall, J. Expression of ATM, p53, and the MRE11-Rad50-NBS1 complex in myoepithelial cells from benign and malignant proliferations of the breast. J. Clin. Pathol. 2004, 57, 1179–1184. [Google Scholar]
- Edwards, S.M.; Kote-Jarai, Z.; Meitz, J.; Hamoudi, R.; Hope, Q.; Osin, P.; Jackson, R.; Southgate, C.; Singh, R.; Falconer, A.; Dearnaley, D.P.; et al. Two percent of men with early-onset prostate cancer harbor germline mutations in the BRCA2 gene. Am. J. Hum. Genet. 2003, 72, 1–12. [Google Scholar] [CrossRef]
- Parshad, R.; Sanford, K.K.; Jones, G.M. Chromatid damage after G2 phase x-irradiation of cells from cancer-prone individuals implicates deficiency in DNA repair. Proc. Natl. Acad. Sci. USA 1983, 80, 5612–5616. [Google Scholar]
- Sanford, K.K.; Parshad, R.; Stanbridge, E.J.; Frost, J.K.; Jones, G.M.; Wilkinson, J.E.; Tarone, R.E. Chromosomal radiosensitivity during the G2 cell cycle period and cytopathology of human normal × tumor cell hybrids. Cancer Res. 1986, 46, 2045–2049. [Google Scholar]
- Mozdarani, H.; Liu, N.; Jones, N.J.; Bryant, P.E. The XRCC2 human repair gene influences recombinational rearrangements leading to chromatid breaks. Int. J. Radiat. Biol. 2001, 77, 859–865. [Google Scholar]
- Terzoudi, G.I.; Hatzi, V.I.; Donta-Bakoyianni, C.; Pantelias, G.E. Chromatin dynamics during cell cycle mediate conversion of DNA damage into chromatid breaks and affect formation of chromosomal aberrations: Biological and clinical significance. Mutat. Res. 2011, 711, 174–186. [Google Scholar]
- Belyaev, I.Y. Radiation-induced DNA repair foci: Spatio-temporal aspects of formation, application for assessment of radiosensitivity and biological dosimetry. Mutat. Res. 2010, 704, 132–141. [Google Scholar]
- Alsbeih, G.; Al-Harbi, N.; Al-Buhairi, M.; Al-Hadyan, K.; Al-Hamed, M. Association between TP53 codon 72 single-nucleotide polymorphism and radiation sensitivity of human fibroblasts. Radiat. Res. 2007, 167, 535–540. [Google Scholar]
- Andreassen, C.N.; Alsner, J.; Overgaard, J. Does variability in normal tissue reactions after radiotherapy have a genetic basis—Where and how to look for it? Radiother. Oncol. 2002, 64, 131–140. [Google Scholar] [CrossRef]
- Andreassen, C.N.; Alsner, J. Genetic variants and normal tissue toxicity after radiotherapy: A systematic review. Radiother. Oncol. 2009, 92, 299–309. [Google Scholar]
- Rosenstein, B.S. Identification of SNPs associated with susceptibility for development of adverse reactions to radiotherapy. Pharmacogenomics 2011, 12, 267–275. [Google Scholar]
- Azria, D.; Ozsahin, M.; Kramar, A.; Peters, S.; Atencio, D.P.; Crompton, N.E.A.; Mornex, F.; Pèlegrin, A.; Dubois, J.B.; Mirimanoff, R.O.; et al. Single nucleotide polymorphisms, apoptosis and the development of severe late adverse effects after radiotherapy. Clin. Cancer Res. 2008, 14, 6284–6288. [Google Scholar]
- Michikawa, Y.; Suga, T.; Ishikawa, A.; Hayashi, H.; Oka, A.; Inoko, H.; Iwakawa, M.; Imai, T. Genome wide screen identifies microsatellite markers associated with acute adverse effects following radiotherapy in cancer patients. BMC Med. Genet. 2010, 11, 123. [Google Scholar]
- Luckey, T.D. Radiation Hormesis; CRC Press: Boca Raton, FL, USA, 1991. [Google Scholar]
- Jolly, D.; Meyer, J. A brief review of radiationhormesis. Australas. Phys. Eng. Sci. Med. 2009, 32, 180–187. [Google Scholar]
- Muckerheide, J. Low-Level Radiation Health Effects: A Compilation of Data and Programs; RSH Inc.: Needham, MA, USA, 2001. [Google Scholar]
- McClintock, B. The significance of responses of the genome to challenge. Science 1984, 226, 792–801. [Google Scholar]
- Serebrianyĭ, A.M. Radiation adaptive response as a stress reaction of a cell (in Russian). Radiats. Biol. Radioecol. 2011, 51, 399–404. [Google Scholar]
- Samson, L.; Cairns, J. A new pathway for DNA repair in Escherichia coli. Nature 1977, 267, 281–283. [Google Scholar]
- Laval, F.; Laval, J. Adaptive response in mammalian cells: Crossreactivity of different pretreatments on cytotoxicity as contrasted to mutagenicity. Proc. Natl. Acad. Sci. USA 1984, 81, 1062–1066. [Google Scholar]
- Olivieri, G.; Bodycote, J.; Wolff, S. Adaptive response of human lymphocytes to low concentrations of radioactive thymidine. Science 1984, 223, 594–597. [Google Scholar]
- Wolff , S. Failla Memorial Lecture. Is radiation all bad? The search for adaptation. Radiat. Res. 1992, 131, 117–123. [Google Scholar] [CrossRef]
- Wolff, S. The adaptive response in radiobiology: Evolving insights and implications. Environ. Health Perspect. 1998, 106, S277–S283. [Google Scholar]
- Rigaud, O.; Moustacchi, E. Radioadaptation for gene mutation and the possible molecular mechanisms of the adaptive response. Mutat. Res. 1996, 358, 127–134. [Google Scholar]
- Stoilov, L.M.; Mullenders, L.H.; Darroudi, F.; Natarajan, A.T. Adaptive response to DNA and chromosomal damage induced by X-rays in human blood lymphocytes. Mutagenesis 2007, 22, 117–122. [Google Scholar]
- Bryant, P.E. Origin of chromosome anerrations: Mechanisms. In Chromoosmeal Alterations: Methods, Results and Importance in Human Health; Obe, G., Vijayalaxmi, Eds.; Springer-Verlag: Berlin, Germany, 2007; pp. 177–199. [Google Scholar]
- Bryant, P.E.; Mozdarani, H. Mechanisms underlying the conversion of DNA double strand breaks into chromatid breaks. Int. J. Low Radiat. 2004, 1, 223–230. [Google Scholar]
- Tedeschi, B.; Caporossi, D.; Vernole, P.; Padovani, L.; Appolloni, M.; Anzidei, P.; Mauro, F. Do human lymphocytes exposed to the fallout of the Chernobyl accident exhibit an adaptive response? II. Challenge with bleomycin. Mutat. Res. 1995, 332, 39–44. [Google Scholar] [CrossRef]
- Mozdarani, H.; Saberi, A.H. Induction of cytogenetic adaptive response of bone marrow cells to radiation by therapeutic doses of bleomycin sulfate and actionmycin D as assayed by the micronucleus test. Cancer Lett. 1994, 78, 141–150. [Google Scholar]
- Barquinero, J.F.; Barrios, L.; Caballin, M.R.; Miro, R.; Ribas, M. Occupational exposure to radiation induces an adaptive response in human lymphocytes. Int. J. Radiat. Biol. 1995, 67, 187–191. [Google Scholar]
- Barquinero, J.F.; Barrios, L.; Caballin, M.R.; Miro, R.; Ribas, M.; Subias, A.; Egozcue, J. Decreased sensitivity to the cytogenetic effects of bleomycin in individuals occupationally exposed to ionizing radiation. Mutat. Res. 1996, 354, 81–86. [Google Scholar]
- Gourabi, H.; Mozdarani, H. A cytokinesis blocked micronucleus study of the radiadaptive response of lymphocytes of individuals ocupationally exposed to chronic doses ofradiation. Mutagenesis 1998, 13, 475–480. [Google Scholar]
- Mozdarani, H.; Gourabi, H. Chromosomal aberrations and micronuclei in lymphocytes of medical radiation workers exposed to ionizing radiation below dose limit. Yakhteh Med. J. 2001, 3, 153–158. [Google Scholar]
- Tedeschi, B. Do human lymphocytes exposed to fall out of Chernobyl accident exhibit an adaptive response. Mutat. Res. 1996, 354, 77–80. [Google Scholar]
- Mitchel, R.E.J.; Jackson, J.S.; McCann, R.A.; Boreham, D.R. Adaptive response modification of latency for radiation-induced myeloid leukemia in CBA/H mice. Radiat. Res. 1999, 152, 273–279. [Google Scholar]
- Mitchel, R.E.J.; Jackson, J.S.; Morrison, D.P.; Carlisle, S.M. Low doses of radiation increase the latency of spontaneous lymphomas and spinal osteosarcomas in cancer-prone, radiation-sensitive Trp53 heterozygous mice. Radiat. Res. 2003, 159, 320–327. [Google Scholar]
- Cohen, B.L. Test of the linear no-threshold theory of radiation carcinogenesis for inhaled radon decay products. Health Phys. 1995, 68, 157–174. [Google Scholar]
- Deetjen, P. Biological and therapeutical properties of radon. In Radon and Thoron in the Human Environment; Katase, A., Shimo, M., Eds.; WorldScientific: Singapore, 1998; pp. 515–522. [Google Scholar]
- Becker, K. Health effects of high radon environments in central Europe: Another test for the LNT hypothesis. Nonlinearity Biol. Toxicol. Med. 2005, 1, 3–35. [Google Scholar]
- Chen, W.L.; Luan, Y.C.; Shici, M.C.; Chen, S.T.; Kung, H.T.; Soong, K.L.; Yeh, Y.C.; Chou, S.H.; Mong, S.H.; Wu, J.T.; et al. Is chronic radiation an effective prophylaxis against cancer? J. Am. Phys. Surg. 2004, 9, 6–10. [Google Scholar]
- Sakamoto, K.; Myonin, M.; Hosor, Y.; Ogawa, Y.; Nemoto, K.; Takai, Y.; Kakuto, Y.; Yamada, S.; Watabe, N. Fundamental and clinical studies on cancer control with total or upper half body irradiation. J. Jpn. Soc. Ther. Radiol. Oncol. 1997, 9, 161–175. [Google Scholar]
- Aurengo, A.; Averbeck, D.; Bonnin, A.; LeGuen, B.; Masse, R.; Monier, R.; Tubiana, M.; Valleron, A.J.; de Vathaire, F. Dose-Effect Relationships and Estimation of the Carcinogenic Effectsof Low Doses of Ionizing Radiation; Academies of Sciences and Medicine: Paris, France, 2005. [Google Scholar]
- UNSCEAR (United Nations Scientific Committee on the Effects of Atomic Radiation), Health Effects Due to Radiation from the Chernobyl Accident; Report to the General Assembly with Scientific Annexes, Volume II, Annex D; United Nations: New York, NY, USA, 2011.
- Luckey, T.D. Low dose irradiation reduces cancer death rates. Radiat. Protect. Manag. 1997, 14, 58–64. [Google Scholar]
- Luckey, T.D. Radiation hormesis in cancer mortality. Int. J. Occup. Med. Tox. 1994, 3, 175–191. [Google Scholar]
- Vaiserman, A.M. Radiationhormesis: Historical perspective and implications for low-dose cancer risk assessment. Dose Response 2010, 8, 172–191. [Google Scholar]
- Luckey, T.D. Nuclear triage and the dirty bomb. Radiat. Protect. Manag. 2004, 20, 11–18. [Google Scholar]
- NEA Committee, Chernobyl: Ten Years on Radiological and Health Impact; Nuclear Energy Agency, OCED (Organization for Economic Co-Operation and Development): Paris, France, 1995.
- National Research Council, Health Effects of Exposure to Radon, BEIR VI; National Academy Press: Washington, DC, USA, 1999.
- National Research Council, Health Risks from Exposure to Low Levels of Ionizing Radiation, BEIR VII, Phase 2; National Academy Press: Washington, DC, USA, 2005.
- Jaworowski, Z. Radiationhormesis—A remedy for fear. Hum. Exp. Toxicol. 2010, 29, 263–270. [Google Scholar]
- Azzam, E.I.; Raaphorst, G.P.; Mitchel, R.E.J. Radiation-induced adaptive response for protection against micronucleus formation and neoplastic transformation in C3H 10T1/2 mouse embryo cells. Radiat. Res. 1994, 138, S28–S31. [Google Scholar]
- Broome, E.J.; Brown, D.L.; Mitchel, R.E. Dose responses for adaption to low doses of (60)Co gamma rays and (3)H beta particles in normal human fibroblasts. Radiat. Res. 2002, 158, 181–186. [Google Scholar]
- Hamada, N.; Maeda, M.; Otsuka, K.; Tomita, M. Signaling pathways underpinning the manifestations of ionizing radiation-induced bystander effects. Curr. Mol. Pharmacol. 2011, 4, 79–95. [Google Scholar]
- Feinendegen, L.E.; Bond, V.P.; Booz, J.; Mühlensiepen, H. Biochemical and cellular mechanisms of low-dose effects. Int. J. Radiat. Biol. Relat. Stud. Phys. Chem. Med. 1988, 53, 23–37. [Google Scholar]
- Wolff, S. Are radiation-induced effects hormetic? Science 1989, 245, 575–621. [Google Scholar]
- Sankaranarayanan, K.; von Duyn, A.; Loos, M.J.; Natarajan, A.T. Adaptive response of human lymphocytes to low-level radiation from radioisotopes or X-rays. Mutat. Res. 1989, 211, 7–12. [Google Scholar]
- Bosi, A.; Olivieri, G. Variability of the adaptive response to ionizing radiations in humans. Mutat. Res. 1989, 211, 13–17. [Google Scholar]
- Davalos, A.; Yannone, S.; Kawahara, M.; Campisi, J. Molecular Mediators of Radio-Adaptation in Human Cells. DOE Low Dose Radiation Research Investigators’ Workshop VI, Washington, DC, USA, 30 July–2 August 2006.
- UNSCEAR (United Nations Scientific Committee on the Effects of Atomic Radiation), Effects of Ionizing Radiation; Report to the General Assembly with Scientific Annexes, Volume I; United Nations, New York, NY, USA, 2006.
- Liu, S.Z. Biological effects of low level exposures to ionizing radiation: Theory and practice. Hum. Exp. Toxicol. 2010, 29, 275–281. [Google Scholar]
- Baverstock, K. Radiation-induced genomic instability: A paradigm-breaking phenomenon and its relevance to environmentally induced cancer. Mutat. Res. Fund. Mol. Mech. Mutagen. 2000, 454, 89–109. [Google Scholar]
- Mothersill, C.; Seymour, C. Radiation-induced bystander effects: Past history and future directions. Radiat. Res. 2001, 155, 757–765. [Google Scholar]
- Goldberg, Z.; Lehnert, B.E. Radiation-induced effects in unirradiated cells: A review and implications in cancer. Int. J. Oncol. 2002, 21, 337–349. [Google Scholar]
- Lorimore, S.A.; Coates, P.J.; Wright, E.G. Radiation-induced genomic instability and bystander effects: Inter-related nontargeted effects of exposure to ionizing radiation. Oncogene 2003, 22, 7058–7069. [Google Scholar]
- Mothersill, C.; Seymour, C. Radiation-induced bystander effects, carcinogenesis and models. Oncogene 2003, 22, 7028–7033. [Google Scholar]
- Wright, E.G. Manifestations and mechanisms of non-targeted effects of ionizing radiation. Mutat. Res. 2010, 687, 28–33. [Google Scholar]
- Mothersill, C.; Seymour, C. Medium from irradiated human epithelial cells but not human fibroblasts reduces the clonogenic survival of unirradiated cells. Int. J. Radiat. Biol. 1997, 71, 421–427. [Google Scholar]
- Lehnert, B.E.; Goodwin, E.H. A new mechanism for DNA alterations induced by alpha particles such as those emitted by radon and radon progeny. Environ. Health Perspect. 1997, 105, 1095–1101. [Google Scholar] [Green Version]
- Prise, K.M.; Belyakov, O.V.; Folkard, M.; Michael, B.D. Studies of bystander effects in human fibroblasts using a charged particle microbeam. Int. J. Radiat. Biol. 1998, 74, 793–798. [Google Scholar]
- Shao, C.; Stewart, V.; Folkard, M.; Michael, B.D.; Prise, K.M. Nitric oxide-mediated signaling in the bystandard response of individually targeted glioma cells. Cancer Res. 2003, 63, 8437–8442. [Google Scholar]
- Lorimore, S.A.; Kadhim, M.A.; Pocock, D.A.; Papworth, D.; Stevens, D.L.; Goodhead, D.T.; Wright, E.G. Chromosomal instability in the descendants of unirradiated surviving cells after alpha-particle irradiation. Proc. Natl. Acad. Sci. USA 1998, 95, 5730–5733. [Google Scholar]
- Azzam, E.I.; de Toledo, S.M.; Gooding, T.; Little, J.B. Intercellular communication is involved in the bystander regulation of gene expression in human cells exposed to very low fluences of alpha particles. Radiat. Res. 1998, 150, 497–504. [Google Scholar]
- Belloni, P.; Latini, P.; Palitti, F. Radiation-induced bystander effect in healthy G0 human lymphocytes: Biological and clinical significance. Mutat. Res. 2011, 713, 32–38. [Google Scholar] [CrossRef]
- Little, J.B. Cellular radiation effects and the bystander response. Mutat. Res. 2006, 597, 113–118. [Google Scholar]
- Kanasugi, Y.; Hamada, N.; Wada, S.; Funayama, T.; Sakashita, T.; Kakizaki, T.; Kobayashi, Y.; Takakura, K. Role of DNA-PKcs in the bystander effect after low- or high-LET irradiation. Int. J. Radiat. Biol. 2007, 83, 73–80. [Google Scholar]
- Konopacka, M.; Rzeszowska-Wolny, R.J. The bystander effect-induced formation of micronucleated cells is inhibited by antioxidants, but the parallel induction of apoptosis and loss of viability are not affected. Mutat. Res. 2006, 593, 32–38. [Google Scholar]
- Rzeszowska-Wolny, J.; Przybyszewski, W.M.; Widel, M. Ionizing radiation-induced bystander effects, potential targets for modulation of radiotherapy. Eur. J. Pharmacol. 2009, 625, 156–164. [Google Scholar]
- Kashino, G.; Suzuki, K.; Matsuda, N.; Kodama, S.; Ono, K.; Watanabe, M.; Prise, K.M. Radiation induced bystander signals are independent of DNA damage and DNA repair capacity of the irradiated cells. Mutat. Res. 2007, 619, 134–138. [Google Scholar] [CrossRef]
- Sokolov, M.V.; Smilenov, L.B.; Hall, E.J.; Panyutin, I.G.; Bonner, W.M.; Sedelnikova, O.A. Ionizing radiation induces DNA double-strand breaks in bystander primary human fibroblasts. Oncogene 2005, 24, 7257–7265. [Google Scholar]
- Harada, T.; Kashino, G.; Suzuki, K.; Matsuda, N.; Kodama, S.; Watanabe, M. Different involvement of radical species in irradiated and bystander cells. Int. J. Radiat. Biol. 2008, 84, 809–814. [Google Scholar]
- Sokolov, M.V.; Dickey, J.S.; Bonner, W.M.; Sedelnikova, O.A. Gamma-H2AX in bystander cells: Not just a radiation-triggered event, a cellular response to stress mediated by intercellular communication. Cell Cycle 2007, 6, 2210–2212. [Google Scholar]
- Sedelnikova, O.A.; Nakamura, A.; Kovalchuk, O.; Koturbash, I.; Mitchell, S.A.; Marino, S.A.; Brenner, D.J.; Bonner, W.M. DNA double-strand breaks form in bystander cells after microbeam irradiation of three-dimensional human tissue models. Cancer Res. 2007, 67, 4295–4302. [Google Scholar]
- Belyakov, O.; Malcolmson, A.; Folkard, M.; Prise, K.; Michael, B. Direct evidence for a bystander effect of ionizing radiation in primary human fibroblasts. Br. J. Cancer 2001, 84, 674–679. [Google Scholar]
- Ballarini, F.; Biaggi, M.; Ottolenghi, A.; Sapora, O. Cellular communication and bystander effects: A critical review for modelling low-dose radiation action. Mutat. Res. 2002, 501, 1–12. [Google Scholar]
- Prise, K.M.; O'Sullivan, J.M. Radiation-induced bystander signalling in cancer therapy. Nat. Rev. Cancer 2009, 9, 351–360. [Google Scholar]
- Lorimore, S.A.; Coates, P.J.; Scobie, G.E.; Milne, G.; Wright, E.G. Inflammatory-type responses after exposure to ionizing radiation in vivo: A mechanism for radiation-induced bystander effects? Oncogene 2001, 20, 7085–7095. [Google Scholar] [CrossRef]
- Marples, B.; Collis, S.J. Low-dose hyper-radiosensitivity: Past, present, and future. Int. J. Radiat. Oncol. Biol. Phys. 2008, 70, 1310–1318. [Google Scholar]
- Iyer, R.; Lehnert, B.E. Low dose, low-LET ionizing radiation-induced radioadaptation and associated early responses in unirradiated cells. Mutat.Res. 2002, 503, 1–9. [Google Scholar]
- Rothkamm, K.; Löbrich, M. Evidence for a lack of DNA double-strand break repair in human cells exposed to very low x-ray doses. Proc. Natl. Acad. Sci. USA 2003, 100, 5057–5062. [Google Scholar]
- Ojima, M.; Ban, N.; Kai, M. DNA double-strand breaks induced by very low X-ray doses are largely due to bystander effects. Radiat. Res. 2008, 170, 365–371. [Google Scholar] [CrossRef]
- Little, M.P. Do non-targeted effects increase or decrease low dose risk in relation to the linear-non-threshold (LNT) model? Mutat. Res. 2010, 687, 17–27. [Google Scholar] [CrossRef]
- Suit, H.; Goldberg, S.; Niemierko, A.; Ancukiewicz, M.; Hall, E.; Goitein, M.; Wong, W.; Paganetti, H. Secondary carcinogenesis in patients treated with radiation: A review of data on radiation-induced cancers in human, non-human primate, canine and rodent subjects. Radiat. Res. 2007, 167, 12–42. [Google Scholar]
- Tubiana, M. Can we reduce the incidence of second primary malignancies occurring after radiotherapy? A critical review. Radiother. Oncol. 2009, 91, 4–15. [Google Scholar]
- Wideł, M.; Przybyszewski, W.; Rzeszowska-Wolny, J. Radiation-induced bystander effect: The important part of ionizing radiation response (in Polish). Potential clinical implications. Postepy Hig. Med. Dosw. (Online) 2009, 63, 377–388. [Google Scholar]
- Sjostedt, S.; Bezak, E. Non-targeted effects of ionising radiation and radiotherapy. Australas. Phys. Eng. Sci. Med. 2010, 33, 219–231. [Google Scholar]
- Blyth, B.J.; Sykes, P.J. Radiation-induced bystander effects: What are they, and how relevant are they to human radiation exposures? Radiat. Res. 2011, 176, 139–157. [Google Scholar] [CrossRef]
- Mackonis, E.C.; Suchowerska, N.; Zhang, M.; Ebert, M.; McKenzie, D.R.; Jackson, M. Cellular response to modulated radiation fields. Phys. Med. Biol. 2007, 52, 5469–5482. [Google Scholar]
- Iyer, R.; Lehnert, B.E. Effects of ionizing radiation in targeted and nontargeted cells. Arch. Biochem. Biophys. 2000, 376, 14–25. [Google Scholar]
- Hei, T.K.; Zhou, H.; Chai, Y.; Ponnaiya, B.; Ivanov, V.N. Radiation induced non-targeted response: Mechanism and potential clinical implications. Curr. Mol. Pharmacol. 2011, 4, 96–105. [Google Scholar]
- Mole, R.H. Whole body irradiation-radiobiology or medicine? Br. J. Radiol. 1953, 26, 234–241. [Google Scholar] [CrossRef]
- Nobler, M.P. The abscopal effect in malignant lymphoma and its relationship to lymphocyte circulation. Radiology 1969, 93, 410–412. [Google Scholar]
- Antoniades, J.; Brady, L.W.; Lightfoot, D.A. Lymphangiographic demonstration of the abscopal effect in patients with malignant lymphomas. Int. J. Radiat. Oncol. Biol. Phys. 1977, 2, 141–147. [Google Scholar]
- Rees, G.J. Abscopal regression in lymphoma: A mechanism in common with total body irradiation? Clin. Radiol. 1981, 32, 475–480. [Google Scholar] [CrossRef]
- Ehlers, G.; Fridman, M. Abscopal effect of radiation in papillary adenocarcinoma. Br. J. Radiol. 1973, 46, 220–222. [Google Scholar]
- Kingsley, D.P. An interesting case of possible abscopal effect in malignant melanoma. Br. J. Radiol. 1975, 48, 863–866. [Google Scholar]
- Rees, G.J.; Ross, C.M. Abscopal regression following radiotherapy for adenocarcinoma. Br. J. Radiol. 1983, 56, 63–66. [Google Scholar]
- Sham, R.L. The abscopal effect and chronic lymphocytic leukemia. Am. J. Med. 1995, 98, 307–308. [Google Scholar]
- Ohba, K.; Omagari, K.; Nakamura, T.; Ikuno, N.; Saeki, S.; Matsuo, I.; Kinoshita, H.; Masuda, J.; Hazama, H.; Sakamoto, I.; et al. Abscopal regression of hepatocellular carcinoma after radiotherapy for bone metastasis. Gut 1998, 43, 575–577. [Google Scholar] [CrossRef]
- Takaya, M.; Niibe, Y.; Tsunoda, S.; Jobo, T.; Imai, M.; Kotani, S.; Unno, N.; Hayakawa, K. Abscopal effect of radiation on toruliform para-aortic lymph node metastases of advanced uterine cervical carcinoma—A case report. Anticancer Res. 2007, 27, 499–503. [Google Scholar]
- Okuma, K.; Yamashita, H.; Niibe, Y.; Hayakawa, K.; Nakagawa, K. Abscopal effect of radiation on lung metastases of hepatocellular carcinoma: A case report. J. Med. Case Reports 2011, 5, 111. [Google Scholar]
- Perego, D.; Faravelli, A. Unexpected consequence of splenectomy in composite lymphoma. The abscopal effect. Haematologica 2000, 85, 211. [Google Scholar]
- Vartak, S.; George, K.C.; Singh, B.B. Antitumor effects of local hyperthermia on a mouse fibrosarcoma. Anticancer Res. 1993, 13, 727–729. [Google Scholar]
- Formenti, S.C.; Demaria, S. Systemic effects of local radiotherapy. Lancet Oncol. 2009, 10, 718–726. [Google Scholar]
- Dewan, M.Z.; Galloway, A.E.; Kawashima, N.; Dewyngaert, J.K.; Babb, J.S.; Formenti, S.C.; Demaria, S. Fractionated but not single-dose radiotherapy induces an immune-mediated abscopal effect when combined with anti-CTLA-4 antibody. Clin. Cancer Res. 2009, 15, 5379–5388. [Google Scholar]
- Demaria, S.; Ng, B.; Devitt, M.L.; Babb, J.S.; Kawashima, N.; Liebes, L.; Formenti, S.C. Ionizing radiation inhibition of distant untreated tumors (abscopal effect) is immune mediated. Int. J. Radiat. Oncol. Biol. Phys. 2004, 58, 862–870. [Google Scholar]
- Camphausen, K.; Moses, M.A.; Ménard, C.; Sproull, M.; Beecken, W.D.; Folkman, J.; O'Reilly, M.S. Radiation abscopal antitumor effect is mediated through p53. Cancer Res. 2003, 63, 1990–1993. [Google Scholar]
- Kinashi, Y.; Tanaka, H.; Masunaga, S.; Suzuki, M.; Kashino, G.; Yong, L.; Takahashi, S.; Ono, K. Ascorbic acid 2-glucocide reduces micronucleus induction in distant splenic T lymphocytes following head irradiation. Mutat. Res. 2010, 695, 69–74. [Google Scholar]
- Ilnytskyy, Y.; Kovalchuk, O. Non-targeted radiation effects-an epigenetic connection. Mutat. Res. 2011, 714, 113–125. [Google Scholar]
- Kaminski, J.M.; Shinohara, E.; Summers, J.B.; Niermann, K.J.; Morimoto, A.; Brousal, J. The controversial abscopal effect. Cancer Treat. Rev. 2005, 31, 159–172. [Google Scholar]
- Joiner, M.C.; Marples, B.; Lambin, P.; Short, S.C.; Turesson, I. Low-dose hypersensitivity: Current status and possible mechanisms. Int. J. Radiat. Oncol. Biol. Phys. 2001, 49, 379–389. [Google Scholar]
- Mortazavi, S.M.J.; Ikushima, T.; Mozdarani, H. Variablity of chromosomal radioadaptive response in human lymphocytes. Iran. J. Radiat. Res. 2003, 1, 55–61. [Google Scholar]
- Kadhim, M.A.; Moore, S.R.; Goodwin, E.H. Interrelationships amongst radiation-induced genomic instability, bystander effects, and the adaptive response. Mutat. Res. 2004, 568, 21–32. [Google Scholar]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Mozdarani, H. Biological Complexities in Radiation Carcinogenesis and Cancer Radiotherapy: Impact of New Biological Paradigms. Genes 2012, 3, 90-114. https://doi.org/10.3390/genes3010090
Mozdarani H. Biological Complexities in Radiation Carcinogenesis and Cancer Radiotherapy: Impact of New Biological Paradigms. Genes. 2012; 3(1):90-114. https://doi.org/10.3390/genes3010090
Chicago/Turabian StyleMozdarani, Hossein. 2012. "Biological Complexities in Radiation Carcinogenesis and Cancer Radiotherapy: Impact of New Biological Paradigms" Genes 3, no. 1: 90-114. https://doi.org/10.3390/genes3010090
APA StyleMozdarani, H. (2012). Biological Complexities in Radiation Carcinogenesis and Cancer Radiotherapy: Impact of New Biological Paradigms. Genes, 3(1), 90-114. https://doi.org/10.3390/genes3010090




