Phenotypic and Genetic Stability of the Aldrovanda vesiculosa L. Plants Regenerated in Tissue Culture
Abstract
1. Introduction
2. Materials and Methods
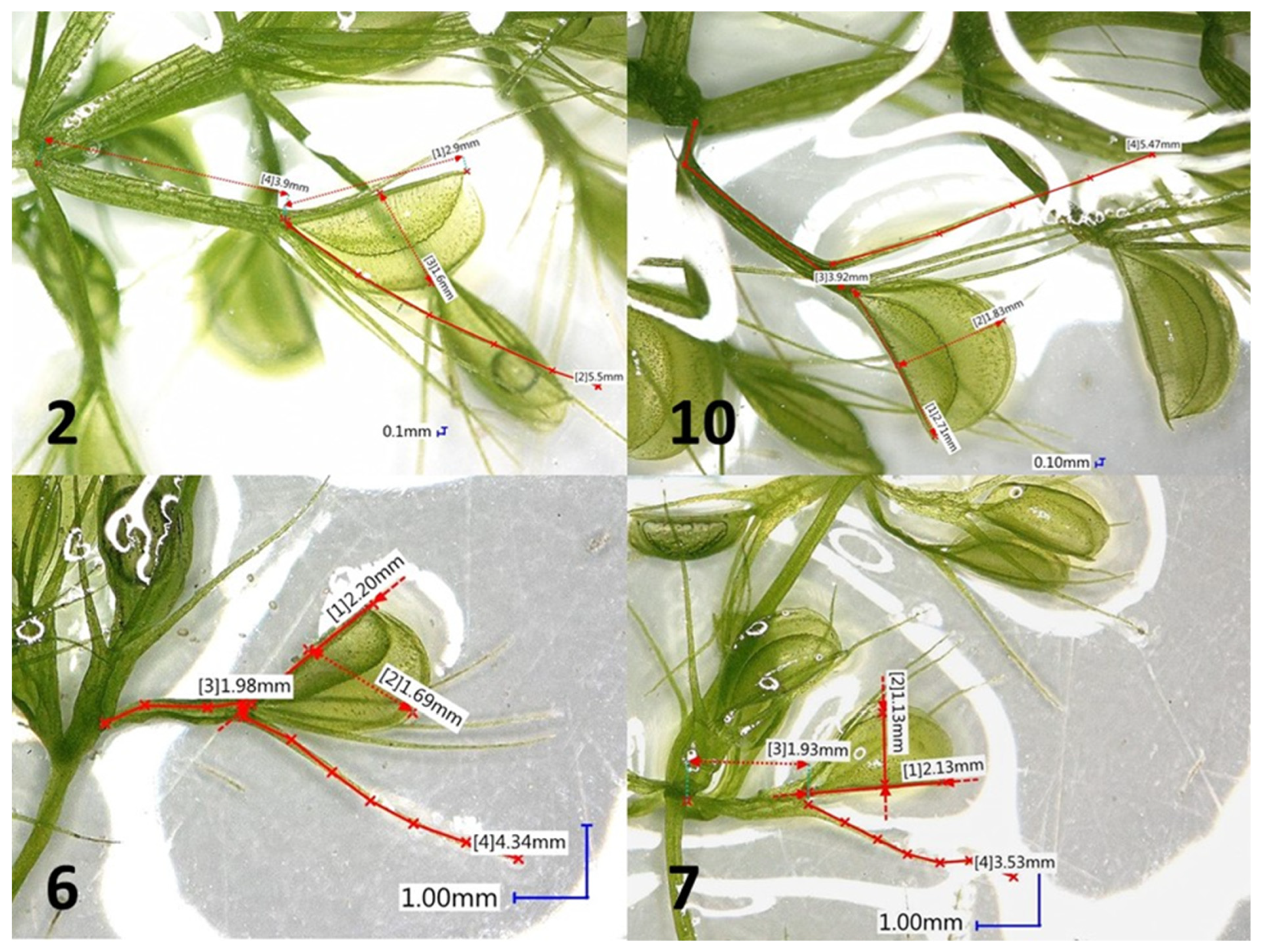
2.1. Tissue Culture Conditions and Morphological Observation
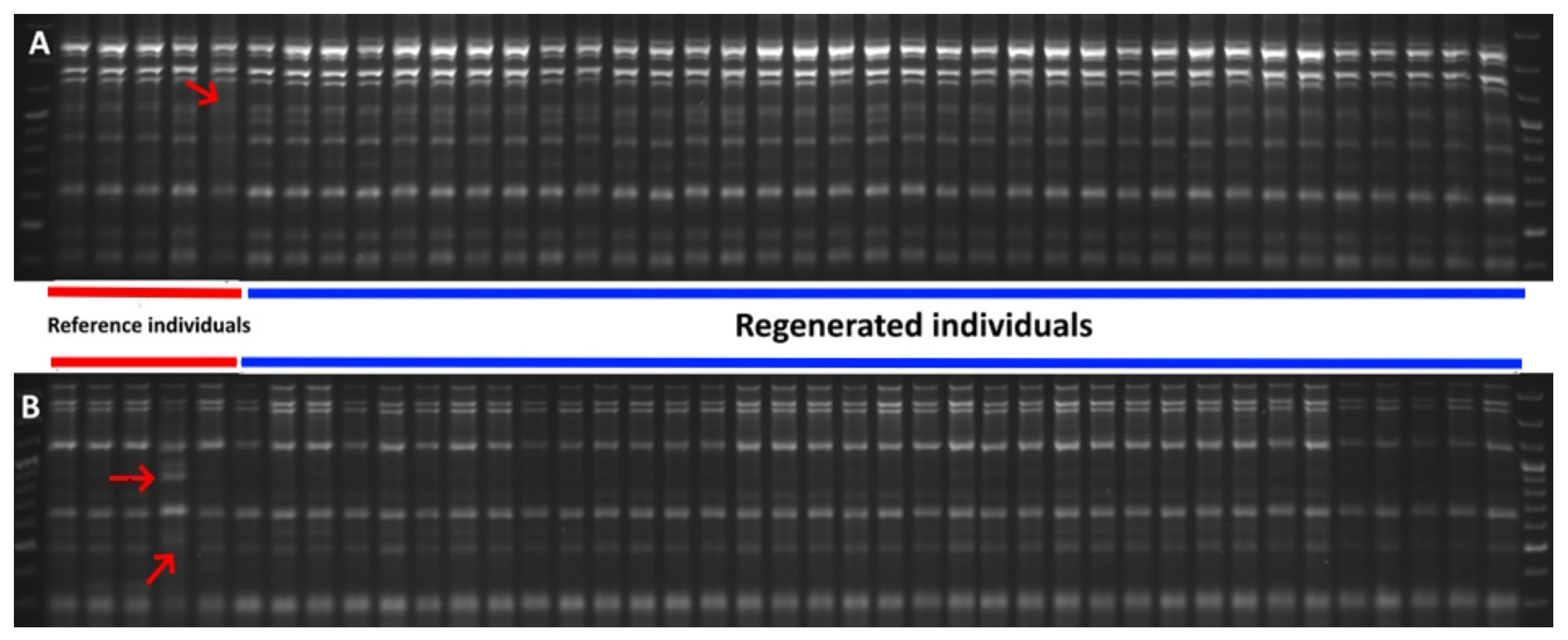
2.2. SCoT and SRAP Genetic Analysis
3. Results and Discussion
3.1. Phenotypic Variation
3.2. SCoT and SRAP Genetic Analysis
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| MS | Murashige and Skoog medium (1962) |
| PGRs | Plant growth regulators |
References
- Corlett, R.T. A bigger toolbox: Biotechnology in biodiversity conservation. Trends Biotechnol. 2017, 35, 55–65. [Google Scholar] [CrossRef]
- Fay, M.F. Conservation of rare and endangered plants using micropropagation. Vitr. Cell. Dev. Biol. Plant 1992, 28, 1–4. [Google Scholar] [CrossRef]
- Sarasan, V.; Cripps, R.; Ramsay, M.M.; Atherton, C.; McMichen, M.; Prendergast, G.; Rowntree, J.K. Conservation of rare and endangered plants using in vitro methods. Vitr. Cell. Dev. Biol. Plant 2006, 42, 206–214. [Google Scholar] [CrossRef]
- Chokheli, V.A.; Dmitriev, P.A.; Rajput, V.D.; Bakulin, S.D.; Azarov, A.S.; Varduni, T.V.; Stepanenko, V.V.; Tarigholizadeh, S.; Singh, R.K.; Verma, K.K.; et al. Recent development in micropropagation techniques for rare plant species. Plants 2020, 9, 1733. [Google Scholar] [CrossRef] [PubMed]
- Reed, B.M.; Sarasan, V.; Kane, M.; Bunn, E.; Pence, V.C. Biodiversity conservation and conservation biotechnology tools. Vitr. Cell. Dev. Biol. Plant 2011, 47, 1–4. [Google Scholar] [CrossRef]
- Pence, V.C. The possibilities and challenges of cryopreservation for conservation. Plant Cell Rep. 2011, 30, 241–250. [Google Scholar]
- Yanthan, J.S.; Kehie, M.; Kumaria, S.; Tnadon, P. In vitro regeneration of Drosera burmannii Vahl.: A carnivorous plant of north-east India. 3 Biotech 2017, 7, 124. [Google Scholar] [CrossRef]
- Kondo, K.; Kokubugata, G.; Varghese, S.B.; Itoyama, M.; Breckpot, C.; Kromer, K.; Kamiński, R. Conservation of endangered Aldrovanda vesiculosa by tissue culture. Carniv. Plant Newsl. 1997, 26, 89–92. [Google Scholar] [CrossRef]
- Jala, A. Colchicine and duration time on survival rate and micropropagation of Dionaea muscipula Ellis. Afr. J. Plant Sci. 2014, 8, 291–297. [Google Scholar] [CrossRef]
- Tkalec, M.; Doboš, M.; Babić, M.; Jurak, E. The acclimation of carnivorous round-leaved sundew (Drosera rotundifolia L.) to solar radiation. Acta Physiol. Plant 2015, 37, 78. [Google Scholar] [CrossRef]
- Jadczak, P.; Kulpa, D.; Zbrojewska, A. In vitro micropropagation of Drosera rotundifolia. World Sci. News 2017, 66, 75–85. [Google Scholar]
- Devi, S.P.; Kumaria, S.; Rao, S.R.; Tandon, P. Genetic fidelity assessment in micropropagated plants using cytogenetical analysis and heterochromatin distribution: A case study with Nepenthes khasiana Hook f. Protoplasma 2015, 252, 1305–1312. [Google Scholar] [CrossRef]
- Caldeira, M.M.M.; de Jesus, J.V.M.; Magalhães, H.S.; de Carvalho, M.A.S.; Andrade, M.S.; Nunes, C.F. Tissue culture applied to carnivorous species. Rev. Sci. Agrár. Parana. 2020, 19, 312–320. [Google Scholar] [CrossRef]
- Barnhill‐Dilling, S.K.; Delborne, J.A. Whose intentions? What consequences? Interrogating “Intended Consequences” for conservation with environmental biotechnology. Conserv. Sci. Pract. 2021, 3, e406. [Google Scholar] [CrossRef]
- Kaeppler, S.M.; Kaeppler, H.F.; Rhee, Y. Epigenetic aspects of somaclonal variation in plants. Plant Mol. Biol. 2000, 43, 179–188. [Google Scholar] [CrossRef]
- Bairu, M.W.; Aremu, A.O.; Van Staden, J. Somaclonal variation in plants: Causes and detection methods. Plant Growth Regul. 2011, 63, 147–173. [Google Scholar] [CrossRef]
- Adamec, L. Ecological requirements and the European distribution of the aquatic carnivorous plant Aldrovanda vesiculosa L.—A review. Folia Geobot. 1995, 30, 53–61. [Google Scholar] [CrossRef]
- Adamec, L. Biological flora of Central Europe: Aldrovanda vesiculosa L. Perspect. Plant Evol. Syst. 2018, 35, 8–21. [Google Scholar] [CrossRef]
- Cameron, K.M.; Wurdack, K.J.; Jobson, R.W. Molecular evidence for the common origin of snap-traps among carnivorous plants. Am. J. Bot. 2002, 89, 1503–1509. [Google Scholar] [CrossRef]
- Poppinga, S.; Bauer, U.; Speck, T.; Volkov, A.G. Motile traps. In Carnivorous Plants: Physiology, Ecology and Evolution; Ellison, A.M., Adamec, L., Eds.; Oxford University Press: Oxford, UK, 2018; p. 193. [Google Scholar]
- Cross, A.T.; Adamec, L.; Turner, S.R.; Dixon, K.W.; Merritt, D.J. Seed reproductive biology of the rare aquatic carnivorous plant Aldrovanda vesiculosa (Droseraceae). Bot. J. Linn. Soc. 2016, 180, 515–529. [Google Scholar] [CrossRef]
- Kamiński, R. Studies on the ecology of Aldrovanda vesiculosa L. I. Ecological differentiation of A. vesiculosa population under the influence of chemical factors in the habitat. Ekol. Pol. 1987, 35, 559–590. [Google Scholar]
- Adamec, L. The biology and cultivation of red Australian Aldrovanda vesiculosa. Carniv. Plant Newsl. 1999, 28, 128–132. [Google Scholar] [CrossRef]
- Adamec, L. Ecophysiological characteristics of turions of aquatic plants. A review. Aquat. Bot. 2018, 148, 64–77. [Google Scholar] [CrossRef]
- Adamec, L. Seasonal growth dynamics and overwintering of the aquatic carnivorous plant Aldrovanda vesiculosa at experimental field sites. Folia Geobot. 1999, 34, 287–297. [Google Scholar] [CrossRef]
- Adamec, L. Ecophysiological characterization of dormancy states in turions of the aquatic carnivorous plant Aldrovanda vesiculosa. Biol. Plant. 2003, 47, 395–402. [Google Scholar] [CrossRef]
- Cross, A.T. Turion development is an ecological trait in all populations of the aquatic carnivorous plant Aldrovanda vesiculosa. Carniv. Plant Newsl. 2013, 42, 57–61. [Google Scholar] [CrossRef]
- Adamec, L.; Lev, J. The introduction of the aquatic carnivorous plant Aldrovanda vesiculosa L. to new potential sites in the Czech Republic: A five-year investigation. Folia Geobot. 1999, 34, 299–305. [Google Scholar] [CrossRef]
- Adamec, L.; Tichý, M. Flowering of Aldrovanda vesiculosa in outdoor culture in the Czech Republic and isozyme variability of its European populations. Carniv. Plant Newsl. 1997, 26, 99–103. [Google Scholar] [CrossRef]
- Cross, A.; Adamec, L. Red List Assessment—Aldrovanda vesiculosa. In The IUCN Red List of Threatened Species; IUCN 2020: T162346A275442310; IUCN Red List: Cambridge, UK, 2020. [Google Scholar] [CrossRef]
- Bilz, M.; Kell, S.P.; Maxted, N.; Lansdown, R.V. European Red List of Vascular Plants; Publications Office of the European Union: Luxembourg, 2011; Available online: https://iucnredlist.org/resources/bilz2011 (accessed on 30 July 2025).
- Council of Europe. European Treaty Series—No. 104. Available online: https://rm.coe.int/1680078aff (accessed on 30 July 2025).
- Kaźmierczakowa, R.; Zarzycki, K.; Mirek, Z. Polska Czerwona Księga Roślin: Paprotniki i Rośliny Kwiatowe (Polish Red Data Book of Plants: Pteridophytes and Flowering Plants); Instytut Ochrony Przyrody PAN: Kraków, Poland, 2014; p. 895. [Google Scholar]
- Fleischmann, A.; Cross, A.T.; Gibson, R.; Gonella, P.M.; Dixon, K.W. Systematics and evolution of Droseraceae. In Carnivorous Plants: Physiology, Ecology and Evolution; Ellison, A., Adamec, L., Eds.; Oxford Academic: Oxford, UK, 2017; pp. 45–57. [Google Scholar]
- Kamiński, R. Restitution of Waterwheel plant (Aldrovanda vesiculosa L.) in Poland and recognition of factors deciding on its survival in temperate climate. Prace Ogrodu Bot. Univ. Wrocławskiego 2006, 8, 105. (In Polish) [Google Scholar]
- Kamiński, R. Aldrovanda vesiculosa L. In Polska Czerwona Księga Roślin: Paprotniki i Rośliny Kwiatowe, 3rd ed.; Polska Akademia Nauk: Kraków, Poland, 2014. [Google Scholar]
- Murashige, T.; Skoog, F. A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol. Plant. 1962, 15, 473–497. [Google Scholar] [CrossRef]
- Parzymies, M.; Pogorzelec, M.; Świstowska, A. Optimization of propagation of the Polish strain of Aldrovanda vesiculosa in tissue culture. Biology 2022, 11, 1389. [Google Scholar] [CrossRef]
- Palfalvi, G.; Hackl, T.; Terhoeven, N.; Shibata, T.F.; Nishiyama, T.; Ankenbrand, M.; Becker, D.; Förster, F.; Freund, M.; Iosip, A.; et al. Genomes of the Venus flytrap and close relatives unveil the roots of plant carnivory. Curr. Biol. 2020, 30, 2312–2320. [Google Scholar] [CrossRef]
- Collard, B.C.Y.; Mackill, D.J. Start codon targeted (SCoT) polymorphism: A simple, novel DNA marker technique for generating gene-targeted markers in plants. Plant Mol. Biol. Rep. 2009, 27, 86–93. [Google Scholar] [CrossRef]
- Li, G.; Quiros, C.F. Sequence-related amplified polymorphism (SRAP), a new marker system based on a simple PCR reaction: Its application to mapping and gene tagging in Brassica. Theor. Appl. Genet. 2001, 103, 455–461. [Google Scholar] [CrossRef]
- Karp, A. Origins, causes and uses of variation in plant tissue cultures. In Plant Cell and Tissue Culture; Vasil, I.K., Thorpe, T.A., Eds.; Kluwer Academic Publishers: Dordrecht, The Netherlands, 1994; pp. 139–151. [Google Scholar]
- Skirvin, R.M.; McPheeters, K.D.; Norton, M. Sources and frequency of somaclonal variation. HortScience 1994, 29, 1232–1237. [Google Scholar] [CrossRef]
- Harding, K. Genetic integrity of cryopreserved plant cells: A review. CryoLetters 2004, 25, 3–22. [Google Scholar]
- Hortsmann, M.; Heier, L.; Kruppert, S.; Weiss, L.C.; Tollrian, R.; Adamec, L.; Westermeier, A.; Speck, T.; Poppinga, S. Comparative prey spectra analyses on the endangered aquatic carnivorous waterwheel plant (Aldrovanda vesiculosa, Droseraceae) at several naturalized microsites in the Czech Republic and Germany. Integr. Organismal Biol. 2019, 1, oby012. [Google Scholar] [CrossRef]
- Israeli, Y.; Reuveni, O.; Lahav, E. Qualitative aspects of somaclonal variations in banana propagated by in vitro techniques. Sci. Hortic. 1991, 48, 71–88. [Google Scholar] [CrossRef]
- Sharma, S.; Bryan, G.; Winfield, M.; Milliam, S. Stability of potato (Solanum tuberosum L.) plants regenerated via somatic embryos, axillary bud proliferated shoots, microtubers and true potato seeds: A comparative phenotypic, cytogenetic and molecular assessment. Planta 2007, 226, 1449–1458. [Google Scholar] [CrossRef]
- Etienne, H.; Bertrand, B. Somaclonal variation in Coffea arabica: Effects of genotype and embryogenic cell suspension age on frequency and phenotype of variants. Tree Physiol. 2003, 21, 1031–1038. [Google Scholar] [CrossRef]
- Podwyszyńska, M. Somaclonal variation in micropropagated tulips based on phenotype observation. J. Fruit Ornam. Plant Res. 2005, 13, 109–122. [Google Scholar]
- Rewers, M.; Kisiala, A.B.; Drouin, J.; Sliwinska, E.; Cholewa, E. In vitro-regenerated wetland sedge Eriophorum vaginatum L. is genetically stable. Acta Physiol. Plant. 2012, 34, 2197–2206. [Google Scholar] [CrossRef]
- Liu, F.; Huang, L.; Li, Y.; Reinhoud, P.; Jongsma, M.A.; Wang, C.-Y. Shoot organogenesis in leaf explants of Hydrangea macrophylla ‘Hyd1’ and assessing genetic stability of regenerants using ISSR markers. Plant Cell Tissue Organ Cult. 2011, 104, 111–117. [Google Scholar] [CrossRef]
- Babu, G.A.; Vinoth, A.; Ravindhran, R. Direct shoot regeneration and genetic fidelity analysis in finger millet using ISSR markers. Plant Cell Tissue Organ Cult. 2017, 132, 157–164. [Google Scholar] [CrossRef]
- Elansary, H.O.M.; Adamec, L.; Storchova, H. Uniformity of organellar DNA in Aldrovanda vesiculosa, an endangered aquatic carnivorous species, distributed across four continents. Aquat. Bot. 2010, 92, 214–220. [Google Scholar] [CrossRef]
- Maldonado San Martin, A.P.; Adamec, L.; Suda, J.; Mes, T.H.M.; Storchova, H. Genetic variation within the endangered species Aldrovanda vesiculosa (Droseraceae) as revealed by RAPD analysis. Aquat. Bot. 2003, 75, 159–172. [Google Scholar] [CrossRef]
- Nishihara, S.; Shiga, T.; Nishihiro, J. The discovery of a new locality for Aldrovanda vesiculosa (Droseraceae), a critically endangered free-floating plant in Japan. J. Asia-Pac. Biodivers. 2023, 16, 227–233. [Google Scholar] [CrossRef]
- Kawiak, A.; Łojkowska, E. Application of RAPD in the determination of genetic fidelity in micropropagated Drosera plantlets. Vitr. Cell. Dev. Biol. Plant 2004, 40, 592–595. [Google Scholar] [CrossRef]
- Chuang, S.J.; Chen, C.L.; Chen, J.J.; Chou, W.Y.; Sung, J.M. Detection of somaclonal variation in micro-propagated Echinacea purpurea using AFLP marker. Sci. Hortic. 2009, 120, 121–126. [Google Scholar] [CrossRef]
- Hirano, T.; Godo, T.; Mii, M. Conservation of endangered endemic plants of the Bonin Islands using micropropagation techniques. Vitr. Cell. Dev. Biol. Plant 2005, 41, 242–246. [Google Scholar] [CrossRef]
- Kułak, V.; Longboat, S.; Brunet, N.D.; Shukla, M.; Saxena, P. In vitro technology in plant conservation: Relevance to biocultural diversity. Plants 2022, 11, 503. [Google Scholar] [CrossRef] [PubMed]
- Ochatt, S.J.; Patat-Ochat, E.M.; Moessner, A. Ploidy level determination within the context of in vitro breeding. Plant Cell Tissue Organ Cult. (PCTOC) 2011, 104, 329–341. [Google Scholar] [CrossRef]
- Thiem, B.; Kikowska, M.; Krawczyk, A.; Więckowska, B.; Sliwinska, E. Phenolic acid and DNA contents of micropropagated Eryngium planum L. Plant Cell Tissue Organ Cult. (PCTOC) 2013, 114, 197–206. [Google Scholar] [CrossRef]
- Singh, S.R.; Dalal, S.; Singh, R.; Dhawan, A.K.; Kalia, R.K. Evaluation of genetic fidelity of in vitro raised plants of Dendrocalamus asper (Schult. & Schult. F.) Backer ex K. Heyne using DNA-based markers. Acta Physiol. Plant. 2012, 35, 419–430. [Google Scholar] [CrossRef]
- Nybom, H.; Weising, K.; Rotter, B. DNA fingerprinting in botany: Past, present, future. Investig. Genet. 2014, 5, 1. [Google Scholar] [CrossRef]

| Primer Name | Sequence (5′ 3′) |
|---|---|
| SCoT18 | ACCATGGCTACCACCGCC |
| SCoT22 | AACCATGGCTACCACCAC |
| SCoT28 | CCATGGCTACCACCGCCA |
| Forward Primer | Sequence | Reverse Primer | Sequence | Combinations Used |
|---|---|---|---|---|
| me2 | TGAGTCCAAACCGGAGC | em2 | GACTGCGTACGAATTTGC | me2-m2 |
| me5 | TGAGTCCAAACCGGAAG | em5 | GACTGCGTACGAATTAAC | me5-em5 |
| me10 | TGAGTCCAAACCGGAAA | em11 | GACTGCGTACGAATTCTA | me10-em12 |
| me11 | TGAGTCCAAACCGGAAC | em12 | GACTGCGTACGAATTCTC | me11-em11 |
| me12 | TGAGTCCAAACCGGAGA | em13 | GACTGCGTACGAATTCTG | me12-em13 |
| Step | SCoT | SRAP | ||||
|---|---|---|---|---|---|---|
| Temperature (°C) | Time (s) | Number of Cycles | Temperature (°C) | Time (s) | Number of Cycles | |
| Initial denaturation | 94 | 180 | 94 | 240 | ||
| Denaturation | 94 | 60 | 35 | 94 | 60 | 5 |
| Annealing | 56 | 60 | 35 | 60 | ||
| Elongation | 72 | 120 | 72 | 60 | ||
| Denaturation | 94 | 60 | 36 | |||
| Annealing | 50 | 60 | ||||
| Elongation | 72 | 60 | ||||
| Final elongation | 72 | 300 | 72 | 420 | ||
| Clone Mark | Main Shoot Length (mm) | Number of Lateral Shoots/ Explant | Length of Lateral Shoots (mm) | Fresh Weight of Plants (mg) |
|---|---|---|---|---|
| 2 | 10.63 A * | 1.60 B | 4.15 A | 31.67 A |
| 10 | 10.28 A | 2.30 B | 4.34 A | 31.53 A |
| 6 | 5.07 B | 7.00 A | 2.20 B | 17.0 AB |
| 7 | 4.76 B | 4.70 AB | 2.54 B | 7.89 B |
| Clone Mark | No. of Whorls per 1 cm of Shoot | Length of Traps (mm) | Width of Traps (mm) | Length of Bristles (mm) | Length of Petioles (mm) |
|---|---|---|---|---|---|
| 2 | 3.0 A * | 2.77 A | 1.71 A | 5.67 A | 3.88 A |
| 10 | 3.0 A | 2.98 A | 1.75 A | 5.64 A | 3.82 A |
| 6 | 2.3 B | 1.93 B | 1.36 B | 4.09 B | 1.86 B |
| 7 | 2.10 B | 2.11 B | 1.17 B | 3.61 B | 1.95 B |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Parzymies, M.; Głębocka, K.; Pogorzelec, M.; Banach-Albińska, B.; Świstowska, A.; Arciszewski, M. Phenotypic and Genetic Stability of the Aldrovanda vesiculosa L. Plants Regenerated in Tissue Culture. Genes 2025, 16, 1003. https://doi.org/10.3390/genes16091003
Parzymies M, Głębocka K, Pogorzelec M, Banach-Albińska B, Świstowska A, Arciszewski M. Phenotypic and Genetic Stability of the Aldrovanda vesiculosa L. Plants Regenerated in Tissue Culture. Genes. 2025; 16(9):1003. https://doi.org/10.3390/genes16091003
Chicago/Turabian StyleParzymies, Marzena, Katarzyna Głębocka, Magdalena Pogorzelec, Barbara Banach-Albińska, Alicja Świstowska, and Michał Arciszewski. 2025. "Phenotypic and Genetic Stability of the Aldrovanda vesiculosa L. Plants Regenerated in Tissue Culture" Genes 16, no. 9: 1003. https://doi.org/10.3390/genes16091003
APA StyleParzymies, M., Głębocka, K., Pogorzelec, M., Banach-Albińska, B., Świstowska, A., & Arciszewski, M. (2025). Phenotypic and Genetic Stability of the Aldrovanda vesiculosa L. Plants Regenerated in Tissue Culture. Genes, 16(9), 1003. https://doi.org/10.3390/genes16091003









