Hereditary Transthyretin Amyloidosis: Genetic Characterization of the TTR P.Val142Ile Variant in a Calabrian Kindred
Abstract
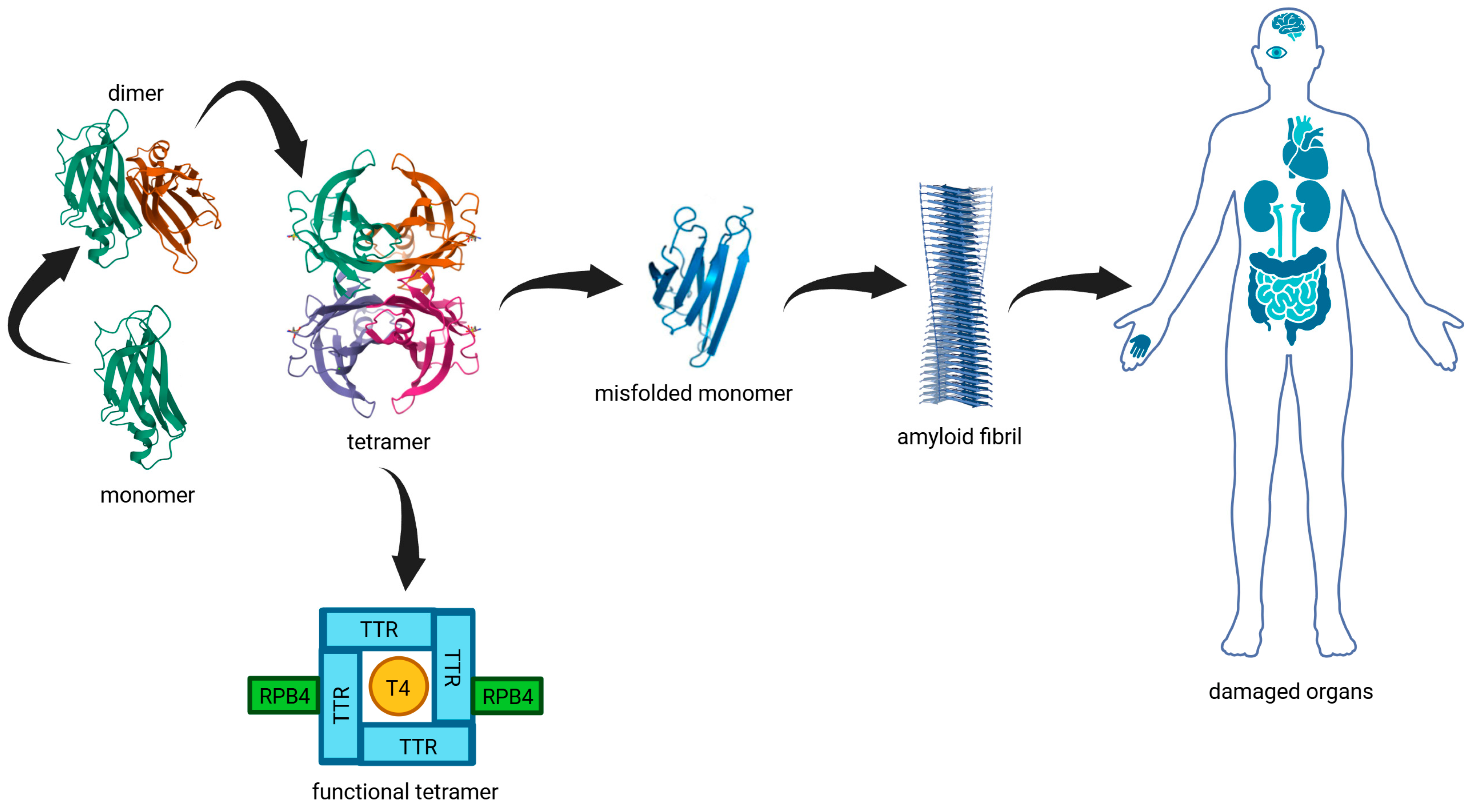
1. Introduction
2. Materials and Methods
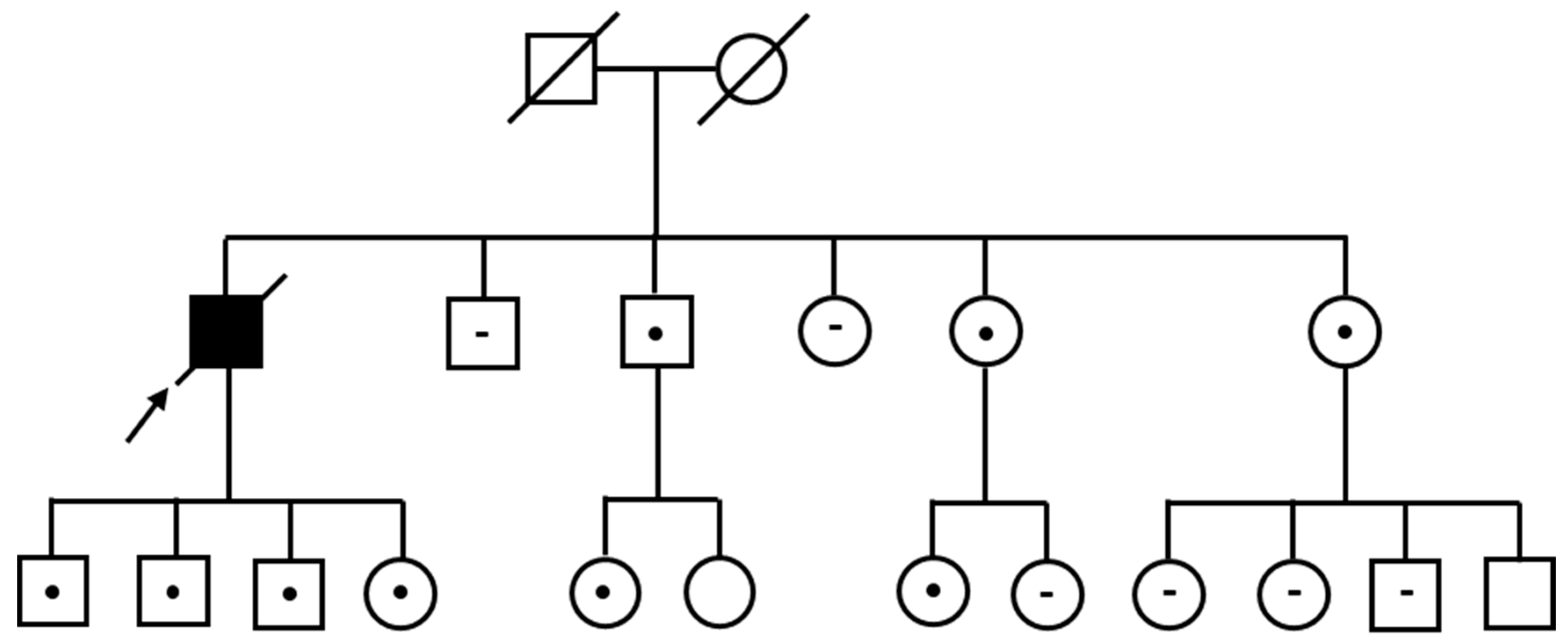
3. Results
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| ATTRwt | Wild-type transthyretin amyloidosis |
| ATTRv | Hereditary transthyretin amyloidosis |
| T4 | Thyroxine |
| RBP | Retinol-binding protein |
| PCR | Polymerase chain reaction |
| AF | Atrial flutter |
| ECG | Electrocardiogram |
| ATTR | Cardiac amyloidosis |
References
- Merlini, G.; Bellotti, V. Molecular mechanisms of amyloidosis. N. Engl. J. Med. 2003, 349, 583–596. [Google Scholar] [CrossRef]
- Chiti, F.; Dobson, C.M. Protein Misfolding, Amyloid Formation, and Human Disease: A Summary of Progress Over the Last Decade. Annu. Rev. Biochem. 2017, 86, 27–68. [Google Scholar] [CrossRef]
- Misra, P.; Blancas-Mejia, L.M.; Ramirez-Alvarado, M. Mechanistic Insights into the Early Events in the Aggregation of Immunoglobulin Light Chains. Biochemistry 2019, 58, 3155–3168. [Google Scholar] [CrossRef]
- Manolis, A.S.; Manolis, A.A.; Manolis, T.A.; Melita, H. Cardiac amyloidosis: An underdiagnosed/underappreciated disease. Eur. J. Intern. Med. 2019, 67, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Adam, R.D. Progress and challenges in the treatment of cardiac amyloidosis: A review of the literature. ESC Heart Fail. 2021, 8, 2380–2396. [Google Scholar] [CrossRef]
- Tomasoni, D. Treating amyloid transthyretin cardiomyopathy: Lessons learned from clinical trials. Front. Cardiovasc. Med. 2023, 10, 1154594. [Google Scholar] [CrossRef]
- Merlini, G.; Seldin, D.C.; Gertz, M.A. Amyloidosis: Pathogenesis and new therapeutic options. J. Clin. Oncol. 2011, 29, 1924–1933. [Google Scholar] [CrossRef]
- Ueda, M.; Horibata, Y.; Shono, M.; Misumi, Y.; Oshima, T.; Su, Y.; Tasaki, M.; Shinriki, S.; Kawahara, S.; Jono, H.; et al. Clinicopathological features of senile systemic amyloidosis: An ante- and postmortem study. Mod. Pathol. 2011, 24, 1533–1544. [Google Scholar] [CrossRef] [PubMed]
- Adams, D.; Koike, H.; Slama, M.; Coelho, T. Hereditary transthyretin amyloidosis: A model of medical progress for a fatal disease. Nat. Rev. Neurol. 2019, 15, 387–404. [Google Scholar] [CrossRef] [PubMed]
- Perfetto, F.; Zampleri, M.; Fumagalli, C.; Allinovi, M.; Cappelli, F. Circulating biomarkers in diagnosis and management of cardiac amyloidosis: A review for internist. Intern. Emerg. Med. 2022, 17, 957–969. [Google Scholar] [CrossRef]
- Hamilton, J.A.; Benson, M.D. Transthyretin: A review from a structural perspective. Cell Mol. Life Sci. 2001, 58, 1491–1521. [Google Scholar] [CrossRef]
- Purkey, H.E.; Dorrell, M.I.; Kelly, J.W. Evaluating the binding selectivity of transthyretin amyloid fibril inhibitors in blood plasma. Proc. Natl. Acad. Sci. USA 2001, 98, 5566–5571. [Google Scholar] [CrossRef]
- Sanguinetti, C.; Minniti, M.; Susini, V.; Caponi, L.; Panichella, G.; Castiglione, V.; Aimo, A.; Emdin, M.; Vergaro, G.; Franzini, M. The journey of Human transthyretin: Synthesis, structure stability, and catabolism. Biomedicines 2022, 10, 1906. [Google Scholar] [CrossRef]
- Lai, Z.; Col’on, W.; Kelly, J.W. The acid-mediated denaturation pathway of transthyretin yields a conformational intermediate that can self-assemble into amyloid. Biochemistry 1996, 35, 6470–6482. [Google Scholar] [CrossRef]
- Lahuerta, C.; Menao, S.; Gracia-Gutierrez, A.; Bueno-Juana, E.; Guillén, N.; Sorribas, V.; Gracia, A.A.; Aibar, M.A. Diagnosis of genetic amyloidosis through the analysis of transthyretin gene mutation using high-resolution melting. Int. J. Cardiol. 2020, 301, 220–225. [Google Scholar] [CrossRef]
- Zaros, C.; Genin, E.; Hellman, U.; Saporta, M.A.; Languille, L.; Wadington-Cruz, M.; Suhr, O.; Misrahi, M.; Plant’e-Bordeneuve, V. On the origin of the transthyretin Val30Met familial amyloid polyneuropathy. Ann. Hum. Genet. 2008, 72 Pt 4, 478–484. [Google Scholar] [CrossRef] [PubMed]
- Waddington-Cruz, M.; Wixner, J.; Amass, L.; Kiszko, J.; Chapman, D.; Ando, Y. THAOS investigators. Characteristics of patients with late- vs. Early-onset Val30Met transthyretin amyloidosis from the transthyretin amyloidosis outcomes survey (THAOS). Neurol. Ther. 2021, 10, 753–766. [Google Scholar] [CrossRef] [PubMed]
- Gentile, L.; Coelho, T.; Dispenzieri, A.; Conceição, I.; Waddington-Cruz, M.; Kristen, A.; Wixner, J.; Diemberger, I.; Gonzalez-Moreno, J.; Cariou, E.; et al. A 15-year consolidated overview of data in over 6000 patients from the Transthyretin Amyloidosis Outcomes Survey (THAOS). Orphanet J. Rare Dis. 2023, 18, 350. [Google Scholar] [CrossRef] [PubMed]
- Ando, Y.; Coelho, T.; Berk, J.L.; Cruz, M.W.; Ericzon, B.-G.; Ikeda, S.-i.; Lewis, W.D.; Obici, L.; Planté-Bordeneuve, V.; Rapezzi, C.; et al. Guideline of transthyretin-related hereditary amyloidosis for clinicians. Orphanet J. Rare Dis. 2013, 8, 31. [Google Scholar] [CrossRef]
- Almeida, M.R.; Alves, I.L.; Terazaki, H.; Ando, Y.; Saraiva, M.J. Comparative studies of two transthyretin variants with protective effects on familial amyloidotic polyneuropathy: TTR R104H and TTR T119M. Biochem. Biophys. Res. Commun. 2000, 270, 1024–1028. [Google Scholar] [CrossRef]
- Maurer, M.S.; Schwartz, J.H.; Gundapaneni, B.; Elliott, P.M.; Merlini, G.; Waddington-Cruz, M.; Kristen, A.V.; Grogan, M.; Witteles, R.; Damy, T.; et al. ATTR-ACT Study Investigators. Tafamidis Treatment for Patients with Transthyretin Amyloid Cardiomyopathy. N. Engl. J. Med. 2018, 379, 1007–1016. [Google Scholar] [CrossRef]
- Obi, C.A.; Mostertz, W.C.; Griffin, J.M.; Judge, D.P. ATTR Epidemiology, Genetics, and Prognostic Factors. Methodist Debakey Cardiovasc. J. 2022, 18, 17–26. [Google Scholar] [CrossRef] [PubMed]
- Chandrashekar, P.; Alhuneafat, L.; Mannello, M.; Al-Rashdan, L.; Kim, M.M.; Dungu, J.; Alexander, K.; Masri, A. Prevalence and Outcomes of p.Val142Ile TTR Amyloidosis Cardiomyopathy: A Systematic Review. Circ. Genom. Precis. Med. 2021, 14, e003356. [Google Scholar] [CrossRef]
- Gentile, L.; Di Bella, G.; Minutoli, F.; Cucinotta, F.; Obici, L.; Mussinelli, R.; Arimatea, I.; Russo, M.; Toscano, A.; Vita, G.; et al. Description of a large cohort of Caucasian patients with V122I ATTRv amyloidosis: Neurological and cardiological features. J. Peripher. Nerv. Syst. 2020, 25, 273–278. [Google Scholar] [CrossRef]
- Russo, M.; Obici, L.; Bartolomei, I.; Cappelli, F.; Luigetti, M.; Fenu, S.; Cavallaro, T.; Chiappini, M.G.; Gemelli, C.; Pradotto, L.G.; et al. ATTRv amyloidosis Italian Registry: Clinical and epidemiological data. Amyloid 2020, 27, 259–265. [Google Scholar] [CrossRef]
- Micaglio, E.; Santangelo, G.; Moscardelli, S.; Rusconi, D.; Musca, F.; Verde, A.; Campiglio, L.; Bursi, F.; Guazzi, M. Case Report: A rare homozygous patient affected by TTR systemic amyloidosis with a prominent heart involvement. Front. Cardiovasc. Med. 2023, 10, 1164916. [Google Scholar] [CrossRef] [PubMed]
- Jacobson, D.R.; Gorevic, P.D.; Buxbaum, J.N. A homozygous transthyretin variant associated with senile systemic amyloidosis: Evidence for a late-onset disease of genetic etiology. Am. J. Hum. Genet. 1990, 47, 127–136. [Google Scholar] [PubMed]
- Carvalho, E.; Dias, A.; Coelho, T.; Sousa, A.; Alves-Ferreira, M.; Santos, M.; Lemos, C. Hereditary transthyretin amyloidosis: A myriad of factors that influence phenotypic variability. J. Neurol. 2024, 271, 5746–5761. [Google Scholar] [CrossRef]
- Cappelli, F.; Del Franco, A.; Vergaro, G.; Mazzoni, C.; Argirò, A.; Pieroni, M.; Giacomin, E.; Poli, S.; Allinovi, M.; Olivotto, I.; et al. Prevalence of transthyretin-related amyloidosis in Tuscany: Data from the regional population-based registry. Int. J. Cardiol. 2023, 382, 87–90. [Google Scholar] [CrossRef]
- Luigetti, M.; Guglielmino, V.; Antonini, G.; Casali, C.; Ceccanti, M.; Chiappini, M.G.; De Giglio, L.; Di Lazzaro, V.; Di Muzio, A.; Goglia, M.; et al. ATTRv in Lazio-Italy: A High-Prevalence Region in a Non-Endemic Country. Genes 2021, 12, 829. [Google Scholar] [CrossRef]
- Bruno, F.; Laganà, V.; Di Lorenzo, R.; Bruni, A.C.; Maletta, R. Calabria as a Genetic Isolate: A Model for the Study of Neurodegenerative Diseases. Biomedicines 2022, 10, 2288. [Google Scholar] [CrossRef] [PubMed]
- Mazzarotto, F.; Argirò, A.; Zampieri, M.; Magri, C.; Giotti, I.; Boschi, B.; Frusconi, S.; Gennarelli, M.; Buxbaum, J.; Polimanti, R.; et al. Investigation on the high recurrence of the ATTRv-causing transthyretin variant Val142Ile in central Italy. Eur. J. Hum. Genet. 2023, 31, 541–547. [Google Scholar] [CrossRef] [PubMed]
- Jacobson, D.R.; Alexander, A.A.; Tagoe, C.; Garvey, W.T.; Williams, S.M.; Tishkoff, S.; Modiano, D.; Sirima, S.B.; Kalidi, I.; Toure, A.; et al. The prevalence and distribution of the amyloidogenic transthyretin (TTR) V122I allele in Africa. Mol. Genet. Genomic. Med. 2016, 4, 548–556. [Google Scholar] [CrossRef] [PubMed]
- De Lillo, A.; Pathak, G.A.; Low, A.; De Angelis, F.; Alaiwi, S.A.; Miller, E.J.; Fuciarelli, M.; Polimanti, R. Clinical spectrum of Transthyretin amyloidogenic mutations among diverse population origins. Hum. Genomics. 2024, 18, 31. [Google Scholar] [CrossRef]
- Fumagalli, C.; Longhi, S.; Aimo, A.; Argirò, A.; Barilaro, A.; Biagini, E.; Biagioni, G.; Ceccanti, M.; Cipriani, A.; Chimenti, C.; et al. Change in prevalence of ATTR variants in Italy-Results from a National Survey. Eur. Heart J. Qual. Care Clin. Outcomes. 2025, qcaf024. [Google Scholar] [CrossRef]
- Giambona, A.; Damiani, G.; Vinciguerra, M.; Jakil, C.; Cannata, M.; Cassarà, F.; Picciotto, F.; Schillaci, G.; Cigna, V.; Renda, D.; et al. Incidence of haemoglobinopathies in Sicily: The impact of screening and prenatal diagnosis. Int. J. Clin. Pract. 2015, 69, 1129–1138. [Google Scholar] [CrossRef]
- Ben-Chetrit, E.; Touitou, I. Familial mediterranean Fever in the world. Arthritis Rheum. 2009, 61, 1447–1453. [Google Scholar] [CrossRef]
- Estivill, X.; Bancells, C.; Ramos, C. Geographic distribution and regional origin of 272 cystic fibrosis mutations in European populations. The Biomed CF Mutation Analysis Consortium. Hum. Mutat. 1997, 10, 135–154. [Google Scholar] [CrossRef]
| Geographic Region | Most Common TTR Mutation |
|---|---|
| Portugal, Spain, France | Val50Met |
| Sweden | Val50Met |
| Japan | Val50Met |
| South America (e.g., Brazil) | Val50Met |
| West Africa | Val142Ile |
| United States | Val142Ile, Thr80Ala, Val50Met |
| United Kingdom, Ireland | Thr80Ala |
| Denmark | Leu131Met |
| Italy | Ile88Leu |
| London (UK) | Val142Ile |
| Caribbean | Val142Ile |
| Degree of Kinship | Result | Symptoms |
|---|---|---|
| Brother 1 | WT | apparently good health |
| Brother 2 | HZ | arterial hypertension, hypercholesterolemia, cerebral ischemia, type 2 diabetes mellitus, elevated microalbuminuria levels |
| Sister 1 | WT | hypercholesterolemia |
| Sister 2 | HZ | Hashimoto’s thyroiditis + hypercholesterolemia |
| Sister 3 | HZ | hypotension |
| Son | HZ | apparently good health |
| Son | HZ | apparently good health |
| Son | HZ | apparently good health |
| Daughter | HZ | extrasystoles |
| Brother’s 2 daughter 1 | HZ | hypercholesterolemia |
| Brother’s 2 daughter 2 | Not tested | - |
| Sister 2’s son 1 | WT | apparently good health |
| Sister 2’s son 2 | Not tested | - |
| Sister 3’s daughter 1 | WT | apparently good health |
| Sister 3’s daughter 2 | WT | apparently good health |
| Sister 3’s son 1 | WT | apparently good health |
| Sister 3’s son 2 | Not tested | - |
| Cousin | HZ | hypercholesterolemia |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dinatolo, F.; Procopio, R.; Rocca, V.; Lo Feudo, E.; Dattola, A.; D’Antona, L.; Fabiani, F.; Colao, E.; Amato, R.; Trapasso, F.; et al. Hereditary Transthyretin Amyloidosis: Genetic Characterization of the TTR P.Val142Ile Variant in a Calabrian Kindred. Genes 2025, 16, 960. https://doi.org/10.3390/genes16080960
Dinatolo F, Procopio R, Rocca V, Lo Feudo E, Dattola A, D’Antona L, Fabiani F, Colao E, Amato R, Trapasso F, et al. Hereditary Transthyretin Amyloidosis: Genetic Characterization of the TTR P.Val142Ile Variant in a Calabrian Kindred. Genes. 2025; 16(8):960. https://doi.org/10.3390/genes16080960
Chicago/Turabian StyleDinatolo, Francesca, Radha Procopio, Valentina Rocca, Elisa Lo Feudo, Adele Dattola, Lucia D’Antona, Fernanda Fabiani, Emma Colao, Rosario Amato, Francesco Trapasso, and et al. 2025. "Hereditary Transthyretin Amyloidosis: Genetic Characterization of the TTR P.Val142Ile Variant in a Calabrian Kindred" Genes 16, no. 8: 960. https://doi.org/10.3390/genes16080960
APA StyleDinatolo, F., Procopio, R., Rocca, V., Lo Feudo, E., Dattola, A., D’Antona, L., Fabiani, F., Colao, E., Amato, R., Trapasso, F., Viglietto, G., & Iuliano, R. (2025). Hereditary Transthyretin Amyloidosis: Genetic Characterization of the TTR P.Val142Ile Variant in a Calabrian Kindred. Genes, 16(8), 960. https://doi.org/10.3390/genes16080960






