Transcriptome Analysis in Pyrus betulaefolia Roots in Response to Short-Term Boron Deficiency
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Materials
2.2. Analysis of Boron Forms
2.3. Analysis of JA and ABA
2.4. RNA-Seq Analisis
2.4.1. Library Preparation for RNA Sequencing
2.4.2. Library Clustering and Sequencing
2.4.3. RNA-Seq Data Analysis
2.5. cDNA Synthesis and Quantitative Real-Time (qRT)-PCR Analysis
2.6. Statistical Analysis
3. Results
3.1. Subsection
3.1.1. Boron Concentration in Various Plant Parts under the Short B Deficiency Treatment
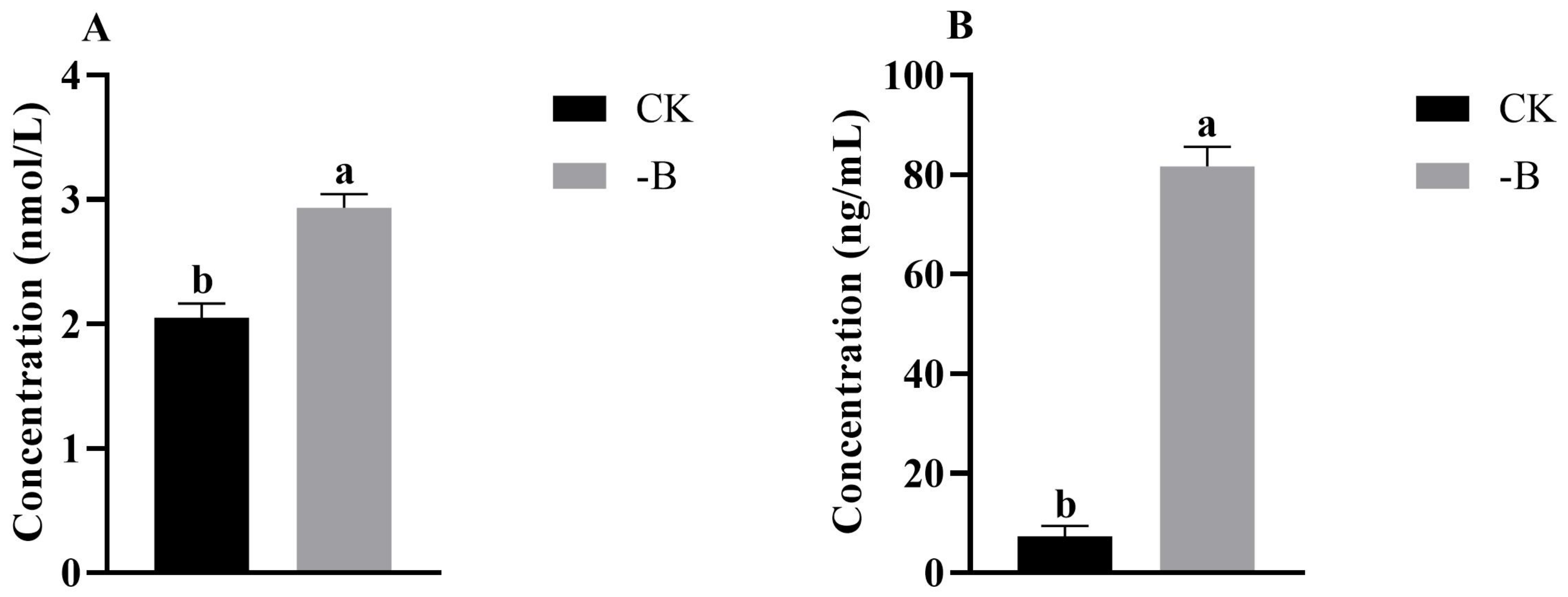
3.1.2. Analysis the Content of JA and ABA in Root Tips under the Short B Deficiency Treatment
3.1.3. RNA-Seq Transcriptome and Analysis of Differentially Expressed Genes
3.1.4. The Expression of Aquaporin Genes under B Deficiency
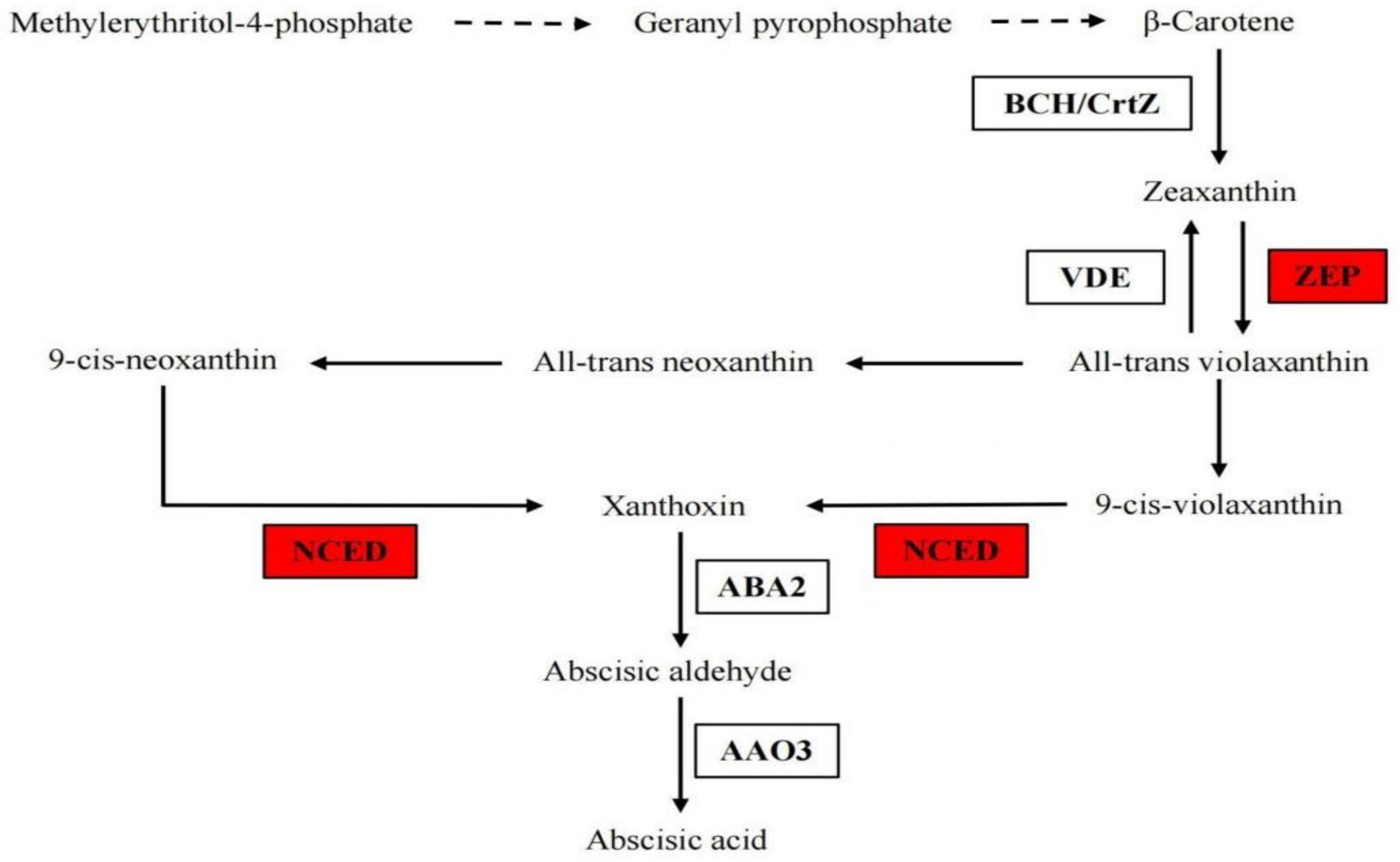
3.1.5. The Gene Expression in ABA Biosynthesis Pathway under B Deficiency
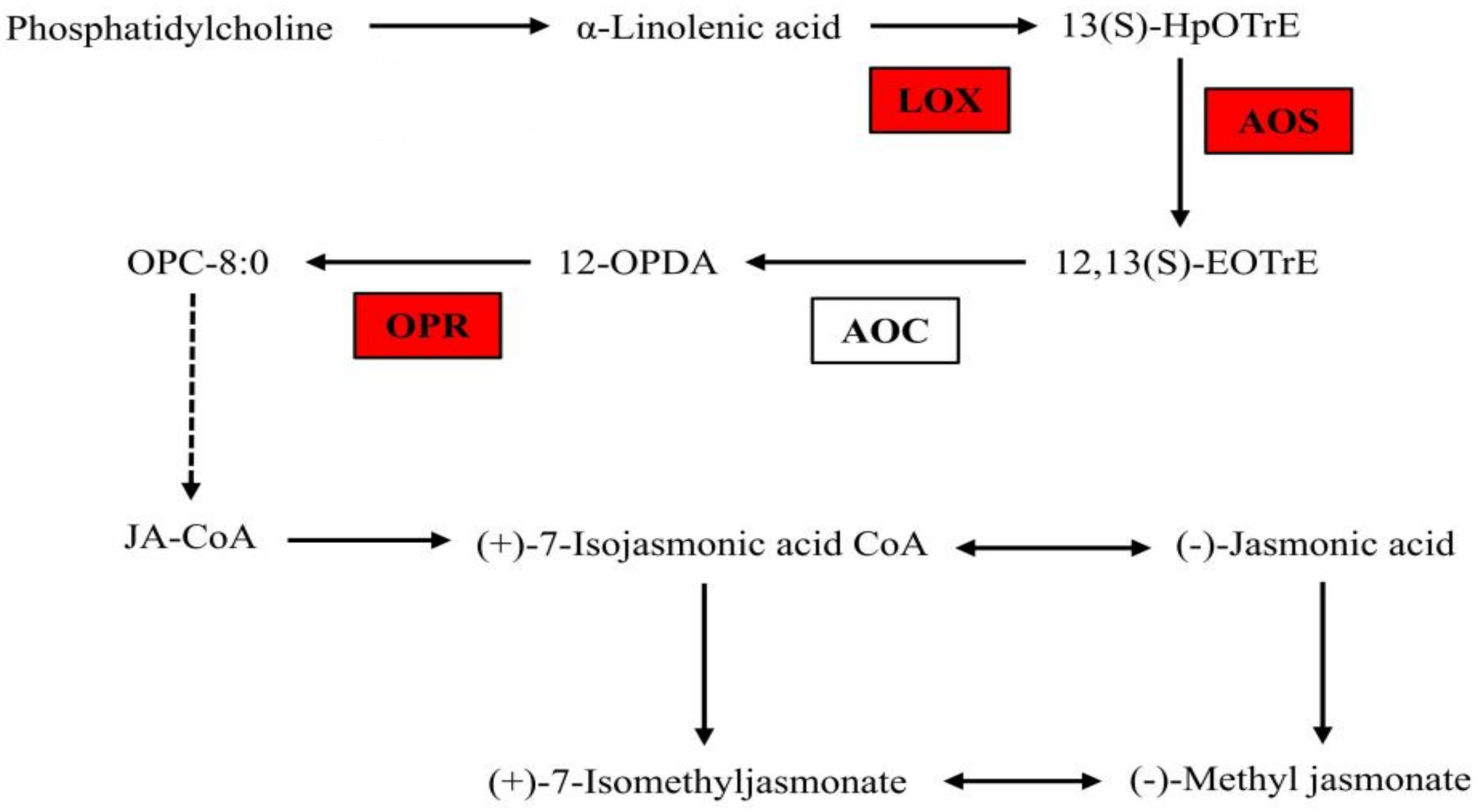
3.1.6. The Gene Expression in JA Biosynthesis Pathway under B Deficiency
3.1.7. The Differentially Expressed Transcription Factor Genes Response to Short Term B Deficiency
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Rerkasem, B.; Jamjod, S.; Pusadee, T. Productivity limiting impacts of boron deficiency, a review. Plant Soil 2020, 455, 23–40. [Google Scholar] [CrossRef]
- Reid, R. Understanding the boron transport network in plants. Plant Soil 2014, 385, 1–13. [Google Scholar] [CrossRef]
- Shireen, F.; Nawaz, M.A.; Chen, C.; Zhang, Q.K.; Zheng, Z.H.; Sohail, H.; Sun, J.Y.; Cao, H.S.; Huang, Y.; Bie, Z.L. Boron: Functions and Approaches to Enhance Its Availability in Plants for Sustainable Agriculture. Int. J. Mol. Sci. 2018, 19, 1856. [Google Scholar] [CrossRef] [PubMed]
- Wang, N.N.; Yang, C.Q.; Pan, Z.Y.; Liu, Y.Z.; Peng, S.A. Boron deficiency in woody plants: Various responses and tolerance mechanisms. Front. Plant Sci. 2015, 6, 916. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.Q.; Liu, Y.Z.; An, J.C.; Li, S.; Jin, L.F.; Zhou, G.F.; Wei, Q.J.; Yan, H.Q.; Wang, N.N.; Fu, L.N.; et al. Digital Gene Expression Analysis of Corky Split Vein Caused by Boron Deficiency in ‘Newhall’ Navel Orange (Citrus sinensis Osbeck) for Selecting Differentially Expressed Genes Related to Vascular Hypertrophy. PLoS ONE 2013, 8, e65737. [Google Scholar] [CrossRef]
- Sutinen, S.; Vuorinen, M.; Rikala, R. Developmental disorders in buds and needles of mature Norway spruce, Picea abies (L.) Karst., in relation to needle boron concentrations. Trees-Struct. Funct. 2006, 20, 559–570. [Google Scholar] [CrossRef]
- Mei, L.; Li, Q.H.; Wang, H.; Sheng, O.; Peng, S.A. Boron deficiency affects root vessel anatomy and mineral nutrient allocation of Poncirus trifoliata (L.) Raf. Acta Physiol. Plant. 2016, 38, 86. [Google Scholar] [CrossRef]
- Wang, N.N.; Yan, T.S.; Fu, L.N.; Zhou, G.F.; Liu, Y.Z.; Peng, S.A. Differences in boron distribution and forms in four citrus scion-rootstock combinations with contrasting boron efficiency under boron-deficient conditions. Trees-Struct. Funct. 2014, 28, 1589–1598. [Google Scholar] [CrossRef]
- Liu, G.D.; Jiang, C.C.; Wang, Y.H. Distribution of boron and its forms in young “Newhall” navel orange (Citrus sinensis Osb.) plants grafted on two rootstocks in response to deficient and excessive boron. Soil Sci. Plant Nutr. 2011, 57, 93–104. [Google Scholar]
- Hanaoka, H.; Uraguchi, S.; Takano, J.; Tanaka, M.; Fujiwara, T. OsNIP3;1, a rice boric acid channel, regulates boron distribution and is essential for growth under boron-deficient conditions. Plant J. 2014, 78, 890–902. [Google Scholar] [CrossRef]
- Miwa, K.; Takano, J.; Fujiwara, T. Improvement of seed yields under boron-limiting conditions through overexpression of BOR1, a boron transporter for xylem loading, in Arabidopsis thaliana. Plant J. 2006, 46, 1084–1091. [Google Scholar] [CrossRef]
- Peng, L.S.; Zeng, C.Y.; Shi, L.; Cai, H.M.; Xu, F.S. Transcriptional Profiling Reveals Adaptive Responses to Boron Deficiency Stress in Arabidopsis. Z. Fur Nat. Sect. C-A J. Biosci. 2012, 67, 510–524. [Google Scholar]
- Zhou, T.; Hua, Y.P.; Huang, Y.P.; Ding, G.D.; Shi, L.; Xu, F.S. Physiological and Transcriptional Analyses Reveal Differential Phytohormone Responses to Boron Deficiency in Brassica napus Genotypes. Front. Plant Sci. 2016, 7, 221. [Google Scholar] [CrossRef]
- Eggert, K.; von Wiren, N. Response of the plant hormone network to boron deficiency. New Phytol. 2017, 216, 868–881. [Google Scholar] [CrossRef]
- Gonzalez-Fontes, A.; Rexach, J.; Quiles-Pando, C.; Begona Herrera-Rodriguez, M.; Camacho-Cristobal, J.J.; Teresa Navarro-Gochicoa, M. Transcription factors as potential participants in the signal transduction pathway of boron deficiency. Plant Signal. Behav. 2013, 8, e26114. [Google Scholar] [CrossRef]
- Song, X.; Wang, X.; Song, B.; Wu, Z.; Zhao, X.; Huang, W.; Riaz, M. Transcriptome analysis reveals the molecular mechanism of boron deficiency tolerance in leaves of boron-efficient Beta vulgaris seedlings. Plant Physiol. Biochem. 2021, 168, 294–304. [Google Scholar] [CrossRef]
- Kasajima, I.; Ide, Y.; Hirai, M.Y.; Fujiwara, T. WRKY6 is involved in the response to boron deficiency in Arabidopsis thaliana. Physiol. Plant. 2010, 139, 80–92. [Google Scholar] [CrossRef]
- Huang, Y.P.; Wang, S.L.; Wang, C.; Ding, G.D.; Cai, H.M.; Shi, L.; Xu, F.S. Induction of jasmonic acid biosynthetic genes inhibits Arabidopsis growth in response to low boron. J. Integr. Plant Biol. 2021, 63, 937–948. [Google Scholar] [CrossRef]
- Du, C.W.; Wang, Y.H.; Xu, F.S.; Yang, Y.H.; Wang, H.Y. Study on the physiological mechanism of boron utilization efficiency in rape cultivars. J. Plant Nutr. 2002, 25, 231–244. [Google Scholar] [CrossRef]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2(T)(-Delta Delta C) method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef]
- Tanaka, M.; Fujiwara, T. Physiological roles and transport mechanisms of boron: Perspectives from plants. Pflug. Arch.-Eur. J. Physiol. 2008, 456, 671–677. [Google Scholar] [CrossRef] [PubMed]
- Li, G.W.; Santoni, V.; Maurel, C. Plant aquaporins: Roles in plant physiology. Biochim. Biophys. Acta-Gen. Subj. 2014, 1840, 1574–1582. [Google Scholar] [CrossRef] [PubMed]
- Takano, J.; Wada, M.; Ludewig, U.; Schaaf, G.; von Wiren, N.; Fujiwara, T. The Arabidopsis major intrinsic protein NIP5;1 is essential for efficient boron uptake and plant development under boron limitation. Plant Cell 2006, 18, 1498–1509. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.L.; Yoshinari, A.; Shimada, T.; Hara-Nishimura, I.; Mitani-Ueno, N.; Ma, J.F.; Naito, S.; Takano, J. Polar Localization of the NIP5;1 Boric Acid Channel Is Maintained by Endocytosis and Facilitates Boron Transport in Arabidopsis Roots. Plant Cell 2017, 29, 824–842. [Google Scholar] [CrossRef]
- Feng, Y.N.; Cui, R.; Wang, S.L.; He, M.L.; Hua, Y.P.; Shi, L.; Ye, X.S.; Xu, F.S. Transcription factor BnaA9.WRKY47 contributes to the adaptation of Brassica napus to low boron stress by up-regulating the boric acid channel gene BnaA3.NIP5;1. Plant Biotechnol. J. 2020, 18, 1241–1254. [Google Scholar] [CrossRef]
- Feng, Y.N.; Cui, R.; Huang, Y.P.; Shi, L.; Wang, S.L.; Xu, F.S. Repression of transcription factor AtWRKY47 confers tolerance to boron toxicity in Arabidopsis thaliana. Ecotoxicol. Environ. Saf. 2021, 220, 112406. [Google Scholar] [CrossRef]
- Noriega, G.; Cruz, D.S.; Batlle, A.; Tomaro, M.; Balestrasse, K. Heme Oxygenase is Involved in the Protection Exerted by Jasmonic Acid Against Cadmium Stress in Soybean Roots. J. Plant Growth Regul. 2012, 31, 79–89. [Google Scholar] [CrossRef]
- Chen, X.; Humphreys, J.L.; Ru, Y.Q.; He, Y.T.; Wu, F.H.; Mai, J.W.; Li, M.; Li, Y.L.; Shabala, S.; Yu, M.; et al. Jasmonate signaling and remodeling of cell wall metabolism induced by boron deficiency in pea shoots. Environ. Exp. Bot. 2022, 201, 104947. [Google Scholar] [CrossRef]
- Gomez-Soto, D.; Galvan, S.; Rosales, E.; Bienert, P.; Abreu, I.; Bonilla, I.; Bolanos, L.; Reguera, M. Insights into the role of phytohormones regulating pAtNIP5;1 activity and boron transport in Arabidopsis thaliana. Plant Sci. 2019, 287, 110198. [Google Scholar] [CrossRef]


| Samples | Raw Reads | Clean Reads | Q20 (%) | Q30 (%) | GC Content (%) | Total Map |
|---|---|---|---|---|---|---|
| B1 | 36,056,316 | 35,817,088 | 97.48 | 92.83 | 46.70 | 19,854,248 (91.74%) |
| B2 | 36,047,720 | 35,884,386 | 97.93 | 93.82 | 46.90 | 19,901,778 (91.90%) |
| B3 | 40,023,668 | 39,819,658 | 97.98 | 93.99 | 46.70 | 19,905,043 (91.79%) |
| CK1 | 39,896,460 | 39,708,496 | 97.92 | 93.81 | 46.65 | 17,908,544 (91.39%) |
| CK2 | 40,025,276 | 39,803,556 | 97.67 | 93.25 | 46.67 | 17,942,193 (91.83%) |
| CK3 | 40,046,248 | 39,810,086 | 97.75 | 93.42 | 46.67 | 19,909,829 (91.57%) |
| Gene Description | Log2Fold Change | ID |
|---|---|---|
| TIP | 2.18 | LOC103937833 |
| 0.11 | LOC103930013 | |
| −0.48 | LOC103930703 | |
| −0.24 | LOC103938012 | |
| 0.10 | LOC103941273 | |
| 0.34 | LOC103944840 | |
| −0.14 | LOC103948949 | |
| 0.26 | LOC103957101 | |
| 0.60 | LOC103966432 | |
| NIP | 2.60 | LOC103962656 |
| −0.72 | LOC103944046 | |
| 0.67 | LOC103950753 | |
| −0.34 | LOC125477097 |
| Gene Description | Log2Fold Change | ID |
|---|---|---|
| ZEP | 1.44 | LOC103937745 |
| 1.17 | LOC103945929 | |
| −0.02 | LOC103956812 | |
| NE | LOC125470765 | |
| NCED | 1.55 | LOC103957334 |
| −0.16 | LOC125476311 | |
| −0.15 | LOC103935979 | |
| NE | LOC103955326 | |
| BCH | −0.14 | LOC103958212 |
| 0.34 | LOC103964324 | |
| NE | LOC103953392 | |
| VDE | 0.11 | LOC103968091 |
| ABA2 | −0.17 | LOC103957656 |
| −0.60 | LOC125478256 | |
| NE | LOC103945571 | |
| NE | LOC125478257 | |
| AAO | −0.75 | LOC103937039 |
| ND | LOC103929500 |
| Gene Description | Log2Fold Change | ID |
|---|---|---|
| LOX | 1.23 | LOC103941411 |
| 1.24 | LOC125471779 | |
| NE | LOC103931981 | |
| NE | LOC103944208 | |
| OPR | 2.69 | LOC103952517 |
| −0.64 | LOC103951930 | |
| 0.23 | LOC103952512 | |
| −0.74 | LOC103952514 | |
| 0.06 | LOC103952519 | |
| −0.14 | LOC103952523 | |
| −0.21 | LOC125478814 | |
| AOS | −0.19 | LOC103954131 |
| 0.10 | LOC103955456 | |
| 2.44 | LOC103942464 | |
| NE | LOC103930300 | |
| AOC | 0.07 | LOC103929185 |
| −0.17 | LOC103947146 | |
| −0.93 | LOC103947801 |
| Function | Log2Fold Change | ID |
|---|---|---|
| MYB | 3.53 | LOC103953105 |
| 2.58 | LOC103941953 | |
| 1.65 | LOC103946050 | |
| 1.61 | LOC103937539 | |
| 1.49 | LOC103959532 | |
| 1.47 | LOC125472789 | |
| 1.24 | LOC125477110 | |
| 1.19 | LOC103944477 | |
| 1.02 | LOC103931986 | |
| WRKY | 3.52 | LOC103958164 |
| 3.37 | LOC103952502 | |
| 2.59 | LOC103964384 | |
| 2.56 | LOC103943771 | |
| 2.08 | LOC103940628 | |
| 1.93 | LOC125473081 | |
| 1.58 | LOC125469162 | |
| 1.55 | LOC103953687 | |
| 1.53 | LOC103956875 | |
| 1.35 | LOC103951248 | |
| 1.33 | LOC103945359 | |
| 1.31 | LOC103953663 | |
| 1.15 | LOC125468857 | |
| 1.06 | LOC103941856 | |
| bHLH | 2.20 | LOC103929974 |
| 1.28 | LOC103941211 | |
| 1.55 | LOC103945773 | |
| 2.69 | LOC103958789 | |
| 1.29 | LOC125475930 | |
| ERF | 3.90 | LOC103947947 |
| 3.54 | LOC103950248 | |
| 3.26 | LOC103955515 | |
| 2.84 | LOC103932979 | |
| 2.83 | LOC103929680 | |
| 2.35 | LOC103928186 | |
| 2.22 | LOC103941900 | |
| 2.13 | LOC103935996 | |
| 1.63 | LOC103959790 | |
| 1.28 | LOC103953878 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, J.; Chen, T.; Wang, C.-L.; Liu, X. Transcriptome Analysis in Pyrus betulaefolia Roots in Response to Short-Term Boron Deficiency. Genes 2023, 14, 817. https://doi.org/10.3390/genes14040817
Liu J, Chen T, Wang C-L, Liu X. Transcriptome Analysis in Pyrus betulaefolia Roots in Response to Short-Term Boron Deficiency. Genes. 2023; 14(4):817. https://doi.org/10.3390/genes14040817
Chicago/Turabian StyleLiu, Jing, Tao Chen, Chun-Lei Wang, and Xiao Liu. 2023. "Transcriptome Analysis in Pyrus betulaefolia Roots in Response to Short-Term Boron Deficiency" Genes 14, no. 4: 817. https://doi.org/10.3390/genes14040817
APA StyleLiu, J., Chen, T., Wang, C.-L., & Liu, X. (2023). Transcriptome Analysis in Pyrus betulaefolia Roots in Response to Short-Term Boron Deficiency. Genes, 14(4), 817. https://doi.org/10.3390/genes14040817







