TERRA and Telomere Maintenance in the Yeast Saccharomyces cerevisiae
Abstract
1. Introduction
2. TERRA Transcription and Primary Structure
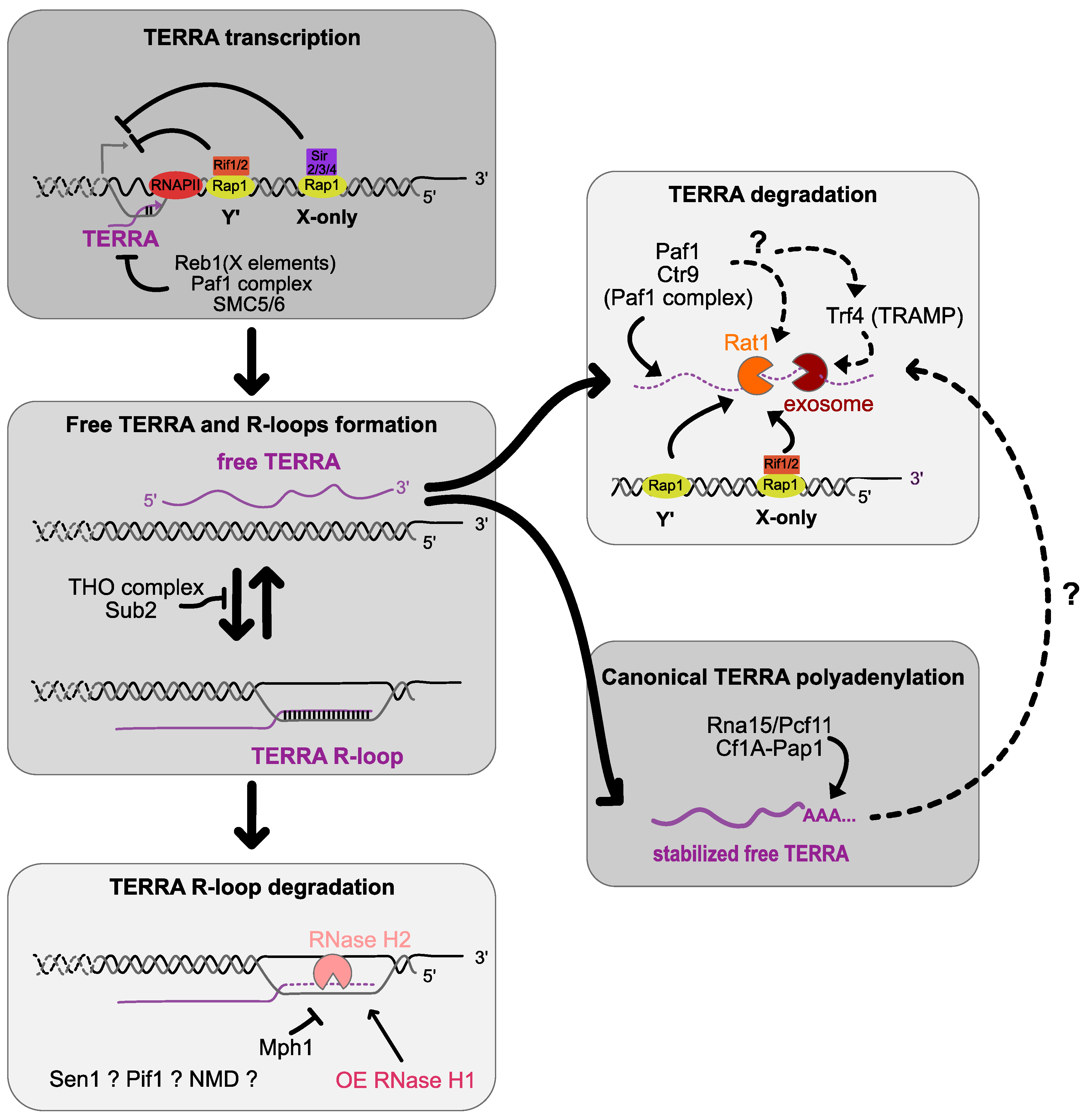
3. Transcriptional, Co-Transcriptional and Post-Transcriptional Regulations
4. TERRA RNA:DNA Hybrid Regulation
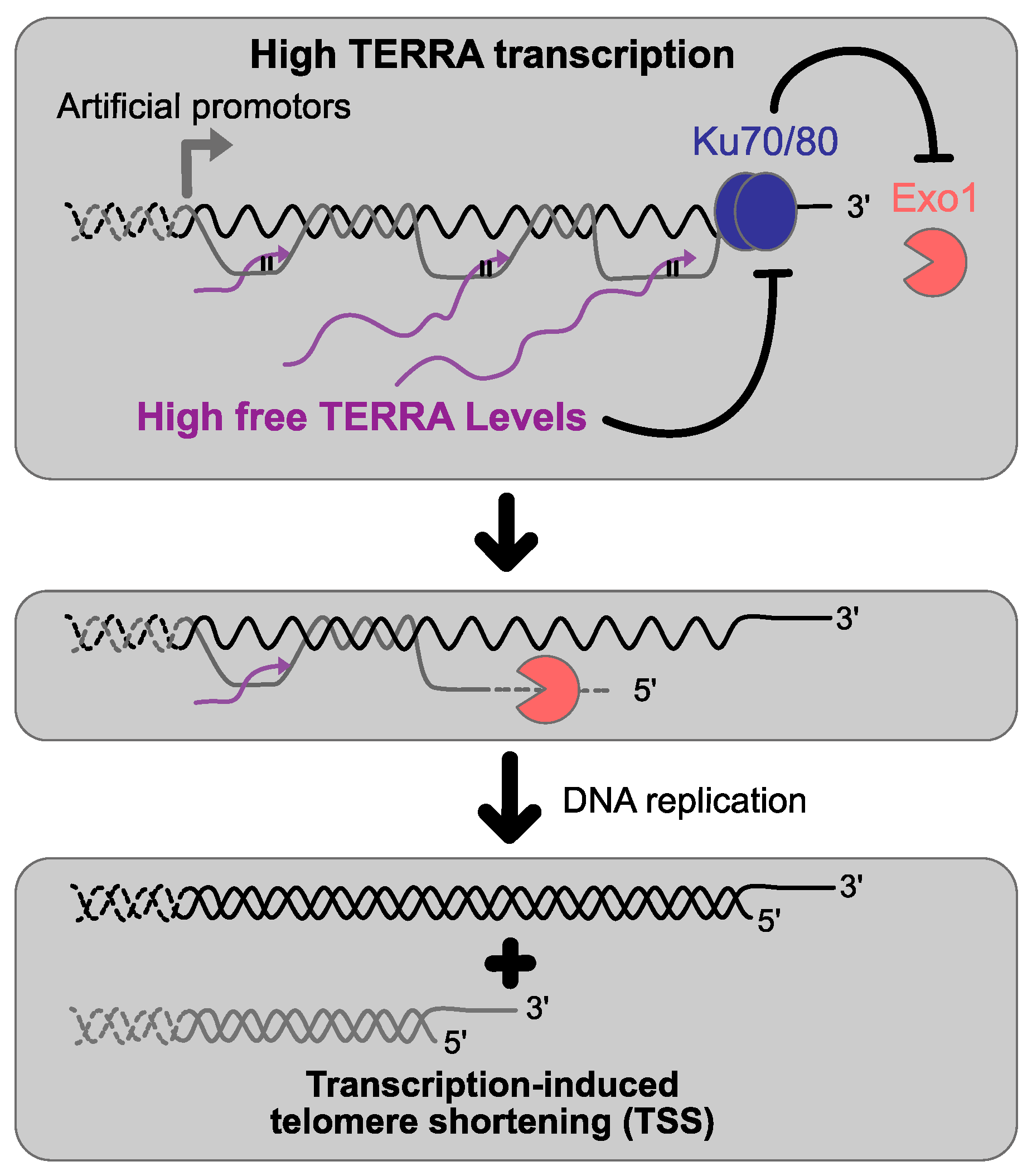
5. TERRA and Telomeres Length
6. TERRA Interacts with Telomerase and Regulates Its Function
7. TERRA and Telomere Replication
8. TERRA and R-Loop Functions during Senescence and Post-Senescence Survival
9. Extratelomeric Functions of TERRA
10. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- De Lange, T. Shelterin: The protein complex that shapes and safeguards human telomeres. Genes Dev. 2005, 19, 2100–2110. [Google Scholar] [CrossRef] [PubMed]
- Soudet, J.; Jolivet, P.; Teixeira, M.T. Elucidation of the DNA end-replication problem in Saccharomyces cerevisiae. Mol. Cell 2014, 53, 954–964. [Google Scholar] [CrossRef]
- Wang, S.S.; Zakian, V.A. Sequencing of Saccharomyces telomeres cloned using T4 DNA polymerase reveals two domains. Mol. Cell. Biol. 1990, 10, 4415–4419. [Google Scholar] [PubMed]
- McEachern, M.J.; Blackburn, E.H. A conserved sequence motif within the exceptionally diverse telomeric sequences of budding yeasts. Proc. Natl. Acad. Sci. USA 1994, 91, 3453–3457. [Google Scholar] [CrossRef]
- Wellinger, R.J.; Wolf, A.J.; Zakian, V.A. Saccharomyces telomeres acquire single-strand TG1-3 tails late in S phase. Cell 1993, 72, 51–60. [Google Scholar] [CrossRef] [PubMed]
- Dionne, I.; Wellinger, R.J. Processing of telomeric DNA ends requires the passage of a replication fork. Nucleic Acids Res. 1998, 26, 5365–5371. [Google Scholar] [CrossRef]
- Larrivee, M.; LeBel, C.; Wellinger, R.J. The generation of proper constitutive G-tails on yeast telomeres is dependent on the MRX complex. Genes Dev. 2004, 18, 1391–1396. [Google Scholar] [CrossRef]
- Gao, H.; Cervantes, R.B.; Mandell, E.K.; Otero, J.H.; Lundblad, V. RPA-like proteins mediate yeast telomere function. Nat. Struct. Mol. Biol. 2007, 14, 208. [Google Scholar] [CrossRef]
- Lundblad, V.; Szostak, J.W. A mutant with a defect in telomere elongation leads to senescence in yeast. Cell 1989, 57, 633–643. [Google Scholar] [CrossRef] [PubMed]
- Abdallah, P.; Luciano, P.; Runge, K.W.; Lisby, M.; Geli, V.; Gilson, E.; Teixeira, M.T. A two-step model for senescence triggered by a single critically short telomere. Nat. Cell Biol. 2009, 11, 988–993. [Google Scholar] [CrossRef] [PubMed]
- Khadaroo, B.; Teixeira, M.T.; Luciano, P.; Eckert-Boulet, N.; Germann, S.M.; Simon, M.N.; Gallina, I.; Abdallah, P.; Gilson, E.; Geli, V.; et al. The DNA damage response at eroded telomeres and tethering to the nuclear pore complex. Nat. Cell Biol. 2009, 11, 980–987. [Google Scholar] [CrossRef] [PubMed]
- Xu, Z.; Duc, K.D.; Holcman, D.; Teixeira, M.T. The length of the shortest telomere as the major determinant of the onset of replicative senescence. Genetics 2013, 194, 847–857. [Google Scholar] [CrossRef] [PubMed]
- Kaul, Z.; Cesare, A.J.; Huschtscha, L.I.; Neumann, A.A.; Reddel, R.R. Five dysfunctional telomeres predict onset of senescence in human cells. EMBO Rep. 2011, 13, 52–59. [Google Scholar] [CrossRef] [PubMed]
- Louis, E.J. The chromosome ends of Saccharomyces cerevisiae. Yeast 1995, 11, 1553–1573. [Google Scholar] [CrossRef] [PubMed]
- Brown, C.A.; Murray, A.W.; Verstrepen, K.J. Rapid expansion and functional divergence of subtelomeric gene families in yeasts. Curr. Biol. 2010, 20, 895–903. [Google Scholar] [CrossRef]
- Bergstrom, A.; Simpson, J.T.; Salinas, F.; Barre, B.; Parts, L.; Zia, A.; Nguyen Ba, A.N.; Moses, A.M.; Louis, E.J.; Mustonen, V.; et al. A high-definition view of functional genetic variation from natural yeast genomes. Mol. Biol. Evol. 2014, 31, 872–888. [Google Scholar] [CrossRef] [PubMed]
- Jolivet, P.; Serhal, K.; Graf, M.; Eberhard, S.; Xu, Z.; Luke, B.; Teixeira, M.T. A subtelomeric region affects telomerase-negative replicative senescence in Saccharomyces cerevisiae. Sci. Rep. 2019, 9, 1845. [Google Scholar] [CrossRef]
- Pasquier, E.; Wellinger, R.J. In vivo chromatin organization on native yeast telomeric regions is independent of a cis-telomere loopback conformation. Epigenetics Chromatin 2020, 13, 23. [Google Scholar] [CrossRef] [PubMed]
- Wright, J.H.; Gottschling, D.E.; Zakian, V.A. Saccharomyces telomeres assume a non-nucleosomal chromatin structure. Genes Dev. 1992, 6, 197–210. [Google Scholar] [CrossRef] [PubMed]
- Gilson, E.; Geli, V. How telomeres are replicated. Nat. Rev. Mol. Cell Biol. 2007, 8, 825–838. [Google Scholar] [CrossRef]
- Hocher, A.; Taddei, A. Subtelomeres as Specialized Chromatin Domains. Bioessays 2020, 42, e1900205. [Google Scholar] [CrossRef] [PubMed]
- Chan, C.S.; Tye, B.K. Organization of DNA sequences and replication origins at yeast telomeres. Cell 1983, 33, 563–573. [Google Scholar] [CrossRef] [PubMed]
- Chan, C.S.; Tye, B.K. A family of Saccharomyces cerevisiae repetitive autonomously replicating sequences that have very similar genomic environments. J. Mol. Biol. 1983, 168, 505–523. [Google Scholar] [CrossRef] [PubMed]
- Zakian, V.A.; Blanton, H.M. Distribution of telomere-associated sequences on natural chromosomes in Saccharomyces cerevisiae. Mol. Cell. Biol. 1988, 8, 2257–2260. [Google Scholar]
- Yue, J.X.; Li, J.; Aigrain, L.; Hallin, J.; Persson, K.; Oliver, K.; Bergstrom, A.; Coupland, P.; Warringer, J.; Lagomarsino, M.C.; et al. Contrasting evolutionary genome dynamics between domesticated and wild yeasts. Nat. Genet. 2017, 49, 913–924. [Google Scholar] [CrossRef] [PubMed]
- Azzalin, C.M.; Reichenback, P.; Khoriauli, L.; Giulotto, E.; Lingner, J. Telomeric repeat containing RNA and RNA surveillance factors at mammalian chromosome ends. Science 2007, 318, 798–801. [Google Scholar] [CrossRef]
- Kwapisz, M.; Morillon, A. Subtelomeric Transcription and its Regulation. J. Mol. Biol. 2020, 432, 4199–4219. [Google Scholar] [CrossRef]
- Bah, A.; Azzalin, C.M. The telomeric transcriptome: From fission yeast to mammals. Int. J. Biochem. Cell Biol. 2012, 44, 1055–1059. [Google Scholar] [CrossRef]
- Rudenko, G.; Van Der Ploeg, L.H. Transcription of telomere repeats in protozoa. EMBO J. 1989, 8, 2633–2638. [Google Scholar] [CrossRef]
- Morea, E.G.O.; Vasconcelos, E.J.R.; Alves, C.d.S.; Giorgio, S.; Myler, P.J.; Langoni, H.; Azzalin, C.M.; Cano, M.I.N. Exploring TERRA during Leishmania major developmental cycle and continuous in vitro passages. Int. J. Biol. Macromol. 2021, 174, 573–586. [Google Scholar] [CrossRef]
- Park, J.; Zhu, Q.; Mirek, E.; Na, L.; Raduwan, H.; Anthony, T.G.; Belden, W.J. BMAL1 associates with chromosome ends to control rhythms in TERRA and telomeric heterochromatin. PLoS ONE 2019, 14, e0223803. [Google Scholar] [CrossRef] [PubMed]
- Majerová, E.; Mandáková, T.; Vu, G.T.; Fajkus, J.; Lysak, M.A.; Fojtová, M. Chromatin features of plant telomeric sequences at terminal vs. internal positions. Front. Plant Sci. 2014, 5, 593. [Google Scholar] [CrossRef] [PubMed]
- Majerová, E.; Fojtová, M.; Mozgová, I.; Bittová, M.; Fajkus, J. Hypomethylating drugs efficiently decrease cytosine methylation in telomeric DNA and activate telomerase without affecting telomere lengths in tobacco cells. Plant Mol. Biol. 2011, 77, 371–380. [Google Scholar] [CrossRef]
- Solovei, I.; Gaginskaya, E.R.; Macgregor, H.C. The arrangement and transcription of telomere DNA sequences at the ends of lampbrush chromosomes of birds. Chromosome Res. 1994, 2, 460–470. [Google Scholar] [CrossRef] [PubMed]
- Vrbsky, J.; Akimcheva, S.; Watson, J.M.; Turner, T.L.; Daxinger, L.; Vyskot, B.; Aufsatz, W.; Riha, K. siRNA-mediated methylation of Arabidopsis telomeres. PLoS Genet. 2010, 6, e1000986. [Google Scholar] [CrossRef]
- Luke, B.; Panza, A.; Redon, S.; Iglesias, N.; Li, Z.; Lingner, J. The Rat1p 5’ to 3’ Exonuclease Degrades Telomeric Repeat-Containing RNA and Promotes Telomere Elongation in Saccharomyces cerevisiae. Mol. Cell 2008, 32, 465–477. [Google Scholar] [CrossRef] [PubMed]
- Mak, H.C.; Pillus, L.; Ideker, T. Dynamic reprogramming of transcription factors to and from the subtelomere. Genome Res. 2009, 19, 1014–1025. [Google Scholar] [CrossRef]
- Feuerhahn, S.; Iglesias, N.; Panza, A.; Porro, A.; Lingner, J. TERRA biogenesis, turnover and implications for function. FEBS Lett. 2010, 584, 3812–3818. [Google Scholar] [CrossRef] [PubMed]
- Lalonde, M.; Chartrand, P. TERRA, a multifaceted regulator of telomerase activity at telomeres. J. Mol. Biol. 2020, 432, 4232–4243. [Google Scholar] [CrossRef]
- Schoeftner, S.; Blasco, M.A. Developmentally regulated transcription of mammalian telomeres by DNA-dependent RNA polymerase II. Nat. Cell Biol. 2008, 10, 228–236. [Google Scholar] [CrossRef]
- Iglesias, N.; Redon, S.; Pfeiffer, V.; Dees, M.; Lingner, J.; Luke, B. Subtelomeric repetitive elements determine TERRA regulation by Rap1/Rif and Rap1/Sir complexes in yeast. EMBO Rep. 2011, 12, 587–593. [Google Scholar] [CrossRef] [PubMed]
- Pfeiffer, V.; Lingner, J. TERRA promotes telomere shortening through exonuclease 1-mediated resection of chromosome ends. PLoS Genet. 2012, 8, e1002747. [Google Scholar] [CrossRef] [PubMed]
- Bauer, S.L.; Grochalski, T.N.T.; Smialowska, A.; Åström, S.U. Sir2 and Reb1 antagonistically regulate nucleosome occupancy in subtelomeric X-elements and repress TERRAs by distinct mechanisms. PLoS Genet. 2022, 18, e1010419. [Google Scholar] [CrossRef] [PubMed]
- Guintini, L.; Paillé, A.; Graf, M.; Luke, B.; Wellinger, R.J.; Conconi, A. Transcription of ncRNAs promotes repair of UV induced DNA lesions in Saccharomyces cerevisiae subtelomeres. PLoS Genet. 2022, 18, e1010167. [Google Scholar] [CrossRef] [PubMed]
- Nergadze, S.G.; Farnung, B.O.; Wischnewski, H.; Khoriauli, L.; Vitelli, V.; Chawla, R.; Giulotto, E.; Azzalin, C.M. CpG-island promoters drive transcription of human telomeres. RNA 2009, 15, 2186–2194. [Google Scholar] [CrossRef] [PubMed]
- Farnung, B.O.; Brun, C.M.; Arora, R.; Lorenzi, L.E.; Azzalin, C.M. Telomerase efficiently elongates highly transcribing telomeres in human cancer cells. PLoS ONE 2012, 7, e35714. [Google Scholar] [CrossRef] [PubMed]
- Graf, M.; Bonetti, D.; Lockhart, A.; Serhal, K.; Kellner, V.; Maicher, A.; Jolivet, P.; Teixeira, M.T.; Luke, B. Telomere Length Determines TERRA and R-Loop Regulation through the Cell Cycle. Cell 2017, 170, 72–85.e14. [Google Scholar] [CrossRef] [PubMed]
- Arnoult, N.; Van Beneden, A.; Decottignies, A. Telomere length regulates TERRA levels through increased trimethylation of telomeric H3K9 and HP1alpha. Nat. Struct. Mol. Biol. 2012, 19, 948–956. [Google Scholar] [CrossRef] [PubMed]
- Porro, A.; Feuerhahn, S.; Reichenbach, P.; Lingner, J. Molecular dissection of telomeric repeat-containing RNA biogenesis unveils the presence of distinct and multiple regulatory pathways. Mol. Cell. Biol. 2010, 30, 4808–4817. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, J.; Lydall, D. Paf1 and Ctr9, core components of the PAF1 complex, maintain low levels of telomeric repeat containing RNA. Nucleic Acids Res. 2018, 46, 621–634. [Google Scholar] [CrossRef] [PubMed]
- Bah, A.; Wischnewski, H.; Shchepachev, V.; Azzalin, C.M. The telomeric transcriptome of Schizosaccharomyces pombe. Nucleic Acids Res. 2012, 40, 2995–3005. [Google Scholar] [CrossRef] [PubMed]
- Sandell, L.L.; Gottschling, D.E.; Zakian, V.A. Transcription of a yeast telomere alleviates telomere position effect without affecting chromosome stability. Proc. Natl. Acad. Sci. USA 1994, 91, 12061–12065. [Google Scholar] [CrossRef]
- Balk, B.; Dees, M.; Bender, K.; Luke, B. The differential processing of telomeres in response to increased telomeric transcription and RNA-DNA hybrid accumulation. RNA Biol. 2014, 11, 95–100. [Google Scholar] [CrossRef] [PubMed]
- Cusanelli, E.; Romero, C.A.; Chartrand, P. Telomeric Noncoding RNA TERRA Is Induced by Telomere Shortening to Nucleate Telomerase Molecules at Short Telomeres. Mol. Cell 2013, 51, 780–791. [Google Scholar] [CrossRef] [PubMed]
- Moradi-Fard, S.; Sarthi, J.; Tittel-Elmer, M.; Lalonde, M.; Cusanelli, E.; Chartrand, P.; Cobb, J.A. Smc5/6 Is a Telomere-Associated Complex that Regulates Sir4 Binding and TPE. PLoS Genet. 2016, 12, e1006268. [Google Scholar] [CrossRef]
- Balk, B.; Maicher, A.; Dees, M.; Klermund, J.; Luke-Glaser, S.; Bender, K.; Luke, B. Telomeric RNA-DNA hybrids affect telomere-length dynamics and senescence. Nat. Struct. Mol. Biol. 2013, 20, 1199–1205. [Google Scholar] [CrossRef] [PubMed]
- Pfeiffer, V.; Crittin, J.; Grolimund, L.; Lingner, J. The THO complex component Thp2 counteracts telomeric R-loops and telomere shortening. EMBO J. 2013, 32, 2861–2871. [Google Scholar] [CrossRef]
- Lafuente-Barquero, J.; Luke-Glaser, S.; Graf, M.; Silva, S.; Gomez-Gonzalez, B.; Lockhart, A.; Lisby, M.; Aguilera, A.; Luke, B. The Smc5/6 complex regulates the yeast Mph1 helicase at RNA-DNA hybrid-mediated DNA damage. PLoS Genet. 2017, 13, e1007136. [Google Scholar] [CrossRef]
- Santos-Pereira, J.M.; Aguilera, A. R loops: New modulators of genome dynamics and function. Nat. Rev. Genet. 2015, 16, 583–597. [Google Scholar] [CrossRef] [PubMed]
- Symington, L.S.; Gautier, J. Double-strand break end resection and repair pathway choice. Annu. Rev. Genet. 2011, 45, 247–271. [Google Scholar] [CrossRef] [PubMed]
- Wagner, C.B.; Luke, B. DNA-RNA Hybrids at Telomeres in Budding Yeast. Methods Mol. Biol. 2022, 2528, 145–157. [Google Scholar] [CrossRef] [PubMed]
- Lockhart, A.; Pires, V.B.; Bento, F.; Kellner, V.; Luke-Glaser, S.; Yakoub, G.; Ulrich, H.D.; Luke, B. RNase H1 and H2 Are Differentially Regulated to Process RNA-DNA Hybrids. Cell Rep. 2019, 29, 2890–2900.e2895. [Google Scholar] [CrossRef] [PubMed]
- Hyjek, M.; Figiel, M.; Nowotny, M. RNases H: Structure and mechanism. DNA Repair (Amst.) 2019, 84, 102672. [Google Scholar] [CrossRef]
- Chávez, S.; Beilharz, T.; Rondón, A.G.; Erdjument-Bromage, H.; Tempst, P.; Svejstrup, J.Q.; Lithgow, T.; Aguilera, A. A protein complex containing Tho2, Hpr1, Mft1 and a novel protein, Thp2, connects transcription elongation with mitotic recombination in Saccharomyces cerevisiae. EMBO J. 2000, 19, 5824–5834. [Google Scholar] [CrossRef]
- Yu, T.Y.; Kao, Y.W.; Lin, J.J. Telomeric transcripts stimulate telomere recombination to suppress senescence in cells lacking telomerase. Proc. Natl. Acad. Sci. USA 2014, 111, 3377–3382. [Google Scholar] [CrossRef] [PubMed]
- Dahlseid, J.N.; Lew-Smith, J.; Lelivelt, M.J.; Enomoto, S.; Ford, A.; Desruisseaux, M.; McClellan, M.; Lue, N.; Culbertson, M.R.; Berman, J. mRNAs encoding telomerase components and regulators are controlled by UPF genes in Saccharomyces cerevisiae. Eukaryot. Cell 2003, 2, 134–142. [Google Scholar] [CrossRef]
- Enomoto, S.; Glowczewski, L.; Lew-Smith, J.; Berman, J.G. Telomere cap components influence the rate of senescence in telomerase-deficient yeast cells. Mol. Cell. Biol. 2004, 24, 837–845. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Sayani, S.; Janis, M.; Lee, C.Y.; Toesca, I.; Chanfreau, G.F. Widespread impact of nonsense-mediated mRNA decay on the yeast intronome. Mol. Cell 2008, 31, 360–370. [Google Scholar] [CrossRef]
- Misino, S.; Busch, A.; Wagner, C.B.; Bento, F.; Luke, B. TERRA increases at short telomeres in yeast survivors and regulates survivor associated senescence (SAS). Nucleic Acids Res. 2022, 50, 12829–12843. [Google Scholar] [CrossRef] [PubMed]
- Teixeira, M.T.; Arneric, M.; Sperisen, P.; Lingner, J. Telomere length homeostasis is achieved via a switch between telomerase- extendible and -nonextendible states. Cell 2004, 117, 323–335. [Google Scholar] [CrossRef]
- Marcand, S.; Gilson, E.; Shore, D. A protein-counting mechanism for telomere length regulation in yeast. Science 1997, 275, 986–990. [Google Scholar] [CrossRef]
- Laprade, H.; Lalonde, M.; Guérit, D.; Chartrand, P. Live-cell imaging of budding yeast telomerase RNA and TERRA. Methods 2017, 114, 46–53. [Google Scholar] [CrossRef] [PubMed]
- Gallardo, F.; Laterreur, N.; Cusanelli, E.; Ouenzar, F.; Querido, E.; Wellinger, R.J.; Chartrand, P. Live cell imaging of telomerase RNA dynamics reveals cell cycle-dependent clustering of telomerase at elongating telomeres. Mol. Cell 2011, 44, 819–827. [Google Scholar] [CrossRef] [PubMed]
- Moravec, M.; Wischnewski, H.; Bah, A.; Hu, Y.; Liu, N.; Lafranchi, L.; King, M.C.; Azzalin, C.M. TERRA promotes telomerase-mediated telomere elongation in Schizosaccharomyces pombe. EMBO Rep. 2016, 17, 999–1012. [Google Scholar] [CrossRef]
- Redon, S.; Reichenbach, P.; Lingner, J. The non-coding RNA TERRA is a natural ligand and direct inhibitor of human telomerase. Nucleic Acids Res. 2010, 38, 5797–5806. [Google Scholar] [CrossRef] [PubMed]
- Azhibek, D.; Skvortsov, D.; Andreeva, A.; Zatsepin, T.; Arutyunyan, A.; Zvereva, M.; Dontsova, O. TERRA mimicking ssRNAs prevail over the DNA substrate for telomerase in vitro due to interactions with the alternative binding site. J. Mol. Recognit. 2016, 29, 242–247. [Google Scholar] [CrossRef] [PubMed]
- Redon, S.; Zemp, I.; Lingner, J. A three-state model for the regulation of telomerase by TERRA and hnRNPA1. Nucleic Acids Res. 2013, 41, 9117–9128. [Google Scholar] [CrossRef] [PubMed]
- Miller, K.M.; Rog, O.; Cooper, J.P. Semi-conservative DNA replication through telomeres requires Taz1. Nature 2006, 440, 824–828. [Google Scholar] [CrossRef] [PubMed]
- Sfeir, A.; Kosiyatrakul, S.T.; Hockemeyer, D.; MacRae, S.L.; Karlseder, J.; Schildkraut, C.L.; de Lange, T. Mammalian telomeres resemble fragile sites and require TRF1 for efficient replication. Cell 2009, 138, 90–103. [Google Scholar] [CrossRef]
- Makovets, S.; Herskowitz, I.; Blackburn, E.H. Anatomy and dynamics of DNA replication fork movement in yeast telomeric regions. Mol. Cell. Biol. 2004, 24, 4019–4031. [Google Scholar] [CrossRef]
- Ivessa, A.S.; Lenzmeier, B.A.; Bessler, J.B.; Goudsouzian, L.K.; Schnakenberg, S.L.; Zakian, V.A. The Saccharomyces cerevisiae helicase Rrm3p facilitates replication past nonhistone protein-DNA complexes. Mol. Cell 2003, 12, 1525–1536. [Google Scholar] [CrossRef] [PubMed]
- Ivessa, A.S.; Zhou, J.Q.; Schulz, V.P.; Monson, E.K.; Zakian, V.A. Saccharomyces Rrm3p, a 5’ to 3’ DNA helicase that promotes replication fork progression through telomeric and subtelomeric DNA. Genes Dev. 2002, 16, 1383–1396. [Google Scholar] [CrossRef] [PubMed]
- Bonetti, D.; Martina, M.; Clerici, M.; Lucchini, G.; Longhese, M.P. Multiple pathways regulate 3’ overhang generation at S. cerevisiae telomeres. Mol. Cell 2009, 35, 70–81. [Google Scholar] [CrossRef]
- Oh, J.; Symington, L.S. Role of the Mre11 Complex in Preserving Genome Integrity. Genes (Basel) 2018, 9, 589. [Google Scholar] [CrossRef] [PubMed]
- Booth, C.; Griffith, E.; Brady, G.; Lydall, D. Quantitative amplification of single-stranded DNA (QAOS) demonstrates that cdc13-1 mutants generate ssDNA in a telomere to centromere direction. Nucleic Acids Res. 2001, 29, 4414–4422. [Google Scholar] [CrossRef] [PubMed]
- Gravel, S.; Larrivee, M.; Labrecque, P.; Wellinger, R.J. Yeast Ku as a regulator of chromosomal DNA end structure. Science 1998, 280, 741–744. [Google Scholar] [CrossRef] [PubMed]
- Polotnianka, R.M.; Li, J.; Lustig, A.J. The yeast Ku heterodimer is essential for protection of the telomere against nucleolytic and recombinational activities. Curr. Biol. 1998, 8, 831–834. [Google Scholar] [CrossRef]
- Maringele, L.; Lydall, D. EXO1-dependent single-stranded DNA at telomeres activates subsets of DNA damage and spindle checkpoint pathways in budding yeast yku70Delta mutants. Genes Dev. 2002, 16, 1919–1933. [Google Scholar] [CrossRef] [PubMed]
- Bertuch, A.A.; Lundblad, V. EXO1 contributes to telomere maintenance in both telomerase-proficient and telomerase-deficient Saccharomyces cerevisiae. Genetics 2004, 166, 1651–1659. [Google Scholar] [CrossRef] [PubMed]
- Maicher, A.; Kastner, L.; Dees, M.; Luke, B. Deregulated telomere transcription causes replication-dependent telomere shortening and promotes cellular senescence. Nucleic Acids Res. 2012, 40, 6649–6659. [Google Scholar] [CrossRef] [PubMed]
- Lundblad, V.; Blackburn, E.H. An alternative pathway for yeast telomere maintenance rescues est1- senescence. Cell 1993, 73, 347–360. [Google Scholar] [CrossRef]
- Kockler, Z.W.; Comeron, J.M.; Malkova, A. A unified alternative telomere-lengthening pathway in yeast survivor cells. Mol. Cell 2021, 81, 1816–1829.e5. [Google Scholar] [CrossRef] [PubMed]
- Bryan, T.M.; Englezou, A.; Dalla-Pozza, L.; Dunham, M.A.; Reddel, R.R. Evidence for an alternative mechanism for maintaining telomere length in human tumors and tumor-derived cell lines. Nat. Med. 1997, 3, 1271–1274. [Google Scholar] [CrossRef] [PubMed]
- Reddel, R.R.; Bryan, T.M.; Colgin, L.M.; Perrem, K.T.; Yeager, T.R. Alternative lengthening of telomeres in human cells. Radiat. Res. 2001, 155, 194–200. [Google Scholar] [CrossRef] [PubMed]
- Cesare, A.J.; Reddel, R.R. Alternative lengthening of telomeres: Models, mechanisms and implications. Nat. Rev. Genet. 2010, 11, 319–330. [Google Scholar] [CrossRef]
- Aguilera, P.; Dubarry, M.; Hardy, J.; Lisby, M.; Simon, M.N.; Géli, V. Telomeric C-circles localize at nuclear pore complexes in Saccharomyces cerevisiae. EMBO J. 2022, 41, e108736. [Google Scholar] [CrossRef]
- Lydeard, J.R.; Jain, S.; Yamaguchi, M.; Haber, J.E. Break-induced replication and telomerase-independent telomere maintenance require Pol32. Nature 2007, 448, 820–823. [Google Scholar] [CrossRef] [PubMed]
- Sobinoff, A.P.; Pickett, H.A. Mechanisms that drive telomere maintenance and recombination in human cancers. Curr. Opin. Genet. Dev. 2020, 60, 25–30. [Google Scholar] [CrossRef] [PubMed]
- Hu, J.; Hwang, S.S.; Liesa, M.; Gan, B.; Sahin, E.; Jaskelioff, M.; Ding, Z.; Ying, H.; Boutin, A.T.; Zhang, H.; et al. Antitelomerase Therapy Provokes ALT and Mitochondrial Adaptive Mechanisms in Cancer. Cell 2012, 148, 651–663. [Google Scholar] [CrossRef] [PubMed]
- Wanat, J.J.; Logsdon, G.A.; Driskill, J.H.; Deng, Z.; Lieberman, P.M.; Johnson, F.B. TERRA and the histone methyltransferase Dot1 cooperate to regulate senescence in budding yeast. PLoS ONE 2018, 13, e0195698. [Google Scholar] [CrossRef]
- Pérez-Martínez, L.; Öztürk, M.; Butter, F.; Luke, B. Npl3 stabilizes R-loops at telomeres to prevent accelerated replicative senescence. EMBO Rep. 2020, 21, e49087. [Google Scholar] [CrossRef] [PubMed]
- Arora, R.; Lee, Y.; Wischnewski, H.; Brun, C.M.; Schwarz, T.; Azzalin, C.M. RNaseH1 regulates TERRA-telomeric DNA hybrids and telomere maintenance in ALT tumour cells. Nat. Commun. 2014, 5, 5220. [Google Scholar] [CrossRef] [PubMed]
- Misino, S.; Bonetti, D.; Luke-Glaser, S.; Luke, B. Increased TERRA levels and RNase H sensitivity are conserved hallmarks of post-senescent survivors in budding yeast. Differentiation 2018, 100, 37–45. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Hong, X.; Wang, L.; Liang, C.Y.; Liu, J.P. Sir4 Deficiency Reverses Cell Senescence by Sub-Telomere Recombination. Cells 2021, 10, 778. [Google Scholar] [CrossRef]
- Yadav, T.; Zhang, J.-M.; Ouyang, J.; Leung, W.; Simoneau, A.; Zou, L. TERRA and RAD51AP1 promote alternative lengthening of telomeres through an R- to D-loop switch. Mol. Cell 2022, 82, 3985–4000.e3984. [Google Scholar] [CrossRef] [PubMed]
- Pires, V.B.; Lohner, N.; Wagner, T.; Wagner, C.B.; Wilkens, M.; Hajikazemi, M.; Paeschke, K.; Butter, F.; Luke, B. RNA-DNA hybrids prevent resection at dysfunctional telomeres. Cell Rep. 2023, 42, 112077. [Google Scholar] [CrossRef] [PubMed]
- Fallet, E.; Jolivet, P.; Soudet, J.; Lisby, M.; Gilson, E.; Teixeira, M.T. Length-dependent processing of telomeres in the absence of telomerase. Nucleic Acids Res. 2014, 42, 3648–3665. [Google Scholar] [CrossRef] [PubMed]
- Silva, B.; Arora, R.; Azzalin, C.M. The alternative lengthening of telomeres mechanism jeopardizes telomere integrity if not properly restricted. Proc. Natl. Acad. Sci. USA 2022, 119, e2208669119. [Google Scholar] [CrossRef] [PubMed]
- Sole, C.; Nadal-Ribelles, M.; de Nadal, E.; Posas, F. A novel role for lncRNAs in cell cycle control during stress adaptation. Curr. Genet. 2015, 61, 299–308. [Google Scholar] [CrossRef]
- Wang, Z.; Deng, Z.; Dahmane, N.; Tsai, K.; Wang, P.; Williams, D.R.; Kossenkov, A.V.; Showe, L.C.; Zhang, R.; Huang, Q.; et al. Telomeric repeat-containing RNA (TERRA) constitutes a nucleoprotein component of extracellular inflammatory exosomes. Proc. Natl. Acad. Sci. USA 2015, 112, E6293–E6300. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Lieberman, P.M. The crosstalk of telomere dysfunction and inflammation through cell-free TERRA containing exosomes. RNA Biol. 2016, 13, 690–695. [Google Scholar] [CrossRef] [PubMed]
- Galigniana, N.M.; Charó, N.L.; Uranga, R.; Cabanillas, A.M.; Piwien-Pilipuk, G. Oxidative stress induces transcription of telomeric repeat-containing RNA (TERRA) by engaging PKA signaling and cytoskeleton dynamics. Biochim. Biophys. Acta Mol. Cell Res. 2020, 1867, 118643. [Google Scholar] [CrossRef] [PubMed]
- Perez-Romero, C.A.; Lalonde, M.; Chartrand, P.; Cusanelli, E. Induction and relocalization of telomeric repeat-containing RNAs during diauxic shift in budding yeast. Curr. Genet. 2018, 64, 1117–1127. [Google Scholar] [CrossRef] [PubMed]


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zeinoun, B.; Teixeira, M.T.; Barascu, A. TERRA and Telomere Maintenance in the Yeast Saccharomyces cerevisiae. Genes 2023, 14, 618. https://doi.org/10.3390/genes14030618
Zeinoun B, Teixeira MT, Barascu A. TERRA and Telomere Maintenance in the Yeast Saccharomyces cerevisiae. Genes. 2023; 14(3):618. https://doi.org/10.3390/genes14030618
Chicago/Turabian StyleZeinoun, Bechara, Maria Teresa Teixeira, and Aurélia Barascu. 2023. "TERRA and Telomere Maintenance in the Yeast Saccharomyces cerevisiae" Genes 14, no. 3: 618. https://doi.org/10.3390/genes14030618
APA StyleZeinoun, B., Teixeira, M. T., & Barascu, A. (2023). TERRA and Telomere Maintenance in the Yeast Saccharomyces cerevisiae. Genes, 14(3), 618. https://doi.org/10.3390/genes14030618




