Chemokine Regulation in Temporomandibular Joint Disease: A Comprehensive Review
Abstract
1. Introduction
2. Inflammation and TMJ
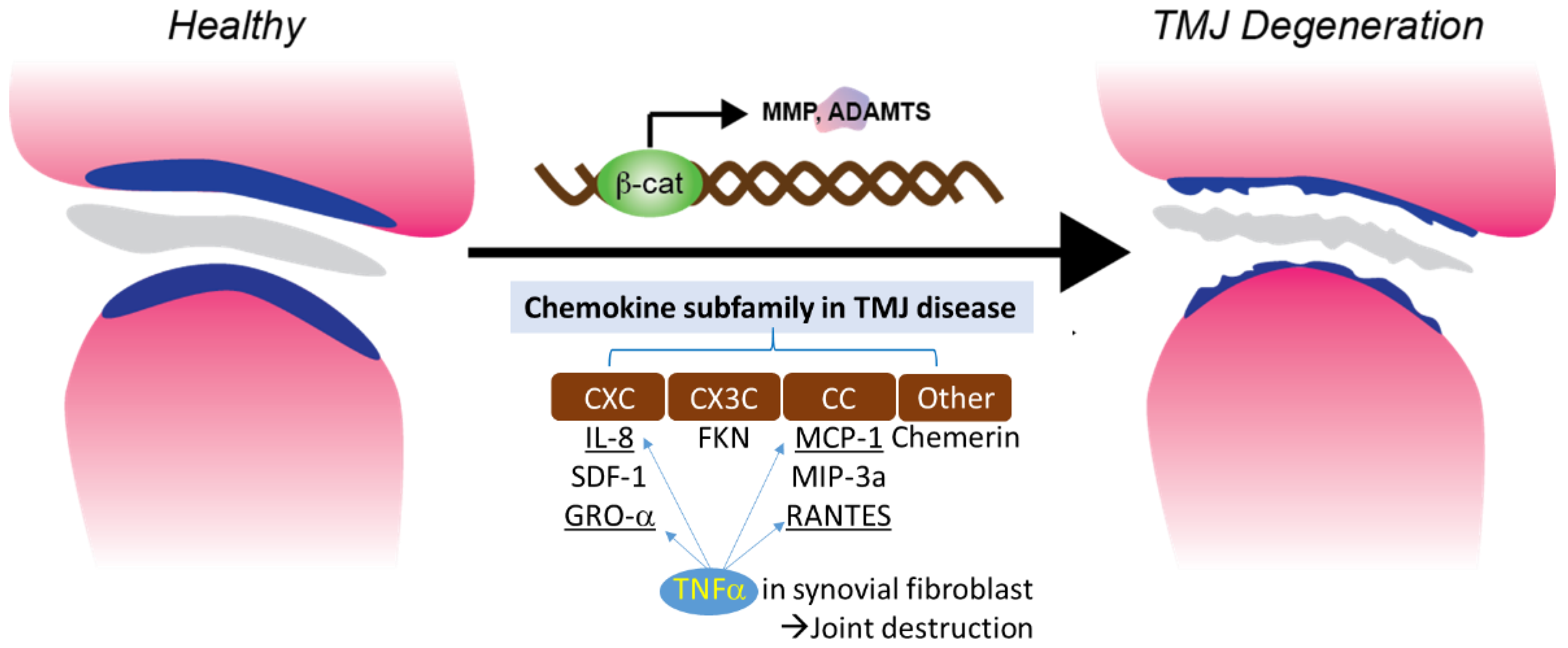
3. Chemokine and TMJ
3.1. CXC (α) and CX3C (δ) Subfamilies in TMJ Disease
3.1.1. IL-8
3.1.2. SDF-1/CXCR4
3.1.3. FKN (CXCL1)
3.2. CC(β) Subfamily in TMJ Disease
3.2.1. MCP-1 Chemokines
3.2.2. MIP3α-CCR6
3.2.3. RANTES-CCR1
3.3. Other Subfamilies in TMJ Disease
Chemerin-ChemR23
3.4. TNFα Induced Chemotaxis and Chemokine Regulation in Synovial Fibroblast
3.5. Chemokines and RA
4. Chemokine in Murine TMJ OA
5. Conclusions and Perspectives
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Gil-Martinez, A.; Paris-Alemany, A.; López-de-Uralde-Villanueva, I.; La Touche, R. Management of pain in patients with temporomandibular disorder (TMD): Challenges and solutions. J. Pain Res. 2018, 11, 571–587. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, M.; Schiffman, E.L. Temporomandibular Joint Disorders and Orofacial Pain. Dent. Clin. 2016, 60, 105–124. [Google Scholar] [CrossRef] [PubMed]
- Li, D.T.S.; Leung, Y.Y. Temporomandibular Disorders: Current Concepts and Controversies in Diagnosis and Management. Diagnostics 2021, 11, 459. [Google Scholar] [CrossRef] [PubMed]
- Valesan, L.F.; Da-Cas, C.D.; Réus, J.C.; Denardin, A.C.S.; Garanhani, R.R.; Bonotto, D.; Januzzi, E.; de Souza, B.D.M. Prevalence of temporomandibular joint disorders: A systematic review and meta-analysis. Clin. Oral Investig. 2021, 25, 441–453. [Google Scholar] [CrossRef]
- Schiffman, E.; Ohrbach, R.; Truelove, E.; Truelove, E.; Look, J.; Anderson, G.; Ceusters, W.; Smith, B. Diagnostic Criteria for Temporomandibular Disorders (DC/TMD) for Clinical and Research Applications: Recommendations of the International RDC/TMD Consortium Network and Orofacial Pain Special Interest Groupdagger. J. Oral Facial Pain Headache 2014, 28, 6–27. [Google Scholar] [CrossRef]
- John, M.T.; Dworkin, S.F.; Mancl, L.A. Reliability of clinical temporomandibular disorder diagnoses. Pain 2005, 118, 61–69. [Google Scholar] [CrossRef]
- Yakkaphan, P.; Smith, J.G.; Chana, P.; Renton, T.; Lambru, G. Temporomandibular disorder and headache prevalence: A systematic review and meta-analysis. Cephalalgia Rep. 2022, 5, 25158163221097352. [Google Scholar] [CrossRef]
- Cascone, P.; Gennaro, P.; Gabriele, G.; Chisci, G.; Mitro, V.; De Caris, F.; Iannetti, G. Temporomandibular synovial chondromatosis with numerous nodules. J. Craniofac. Surg. 2014, 25, 1114–1115. [Google Scholar] [CrossRef]
- Cascone, P.; Di Paolo, C.; Leonardi, R.; Pedullà, E. Temporomandibular disorders and orthognathic surgery. J. Craniofac. Surg. 2008, 19, 687–692. [Google Scholar] [CrossRef]
- Yin, Y.; He, S.; Xu, J.; You, W.; Li, Q.; Long, J.; Luo, L.; Kemp, G.J.; Sweeney, J.A.; Li, F.; et al. The neuro-pathophysiology of temporomandibular disorders-related pain: A systematic review of structural and functional MRI studies. J. Headache Pain 2020, 21, 78. [Google Scholar] [CrossRef]
- Jin, L.J.; Lamster, I.B.; Greenspan, J.S.; Pitts, N.B.; Scully, C.; Warnakulasuriya, S. Global burden of oral diseases: Emerging concepts, management and interplay with systemic health. Oral Dis. 2016, 22, 609–619. [Google Scholar] [CrossRef]
- Slade, G.; Ohrbach, R.; Greenspan, J.D.; Fillingim, R.B.; Bair, E.; Sanders, A.E.; Dubner, R.; Diatchenko, L.; Meloto, C.B.; Smith, S.; et al. Painful temporomandibular disorder: Decade of discovery from OPPERA studies. J. Dent. Res. 2016, 95, 1084–1092. [Google Scholar] [CrossRef]
- Stowell, A.W.; Gatchel, R.J.; Wildenstein, L. Cost-effectiveness of treatments for temporomandibular disorders: Biopsychosocial intervention versus treatment as usual. J. Am. Dent. Assoc. 2007, 138, 202–208. [Google Scholar] [CrossRef]
- Farina, D.; Bodin, C.; Gandolfi, S.; De Gasperi, W.; Borghesi, A.; Maroldi, R. TMJ disorders and pain: Assessment by contrast-enhanced MRI. Eur. J. Radiol. 2009, 70, 25–30. [Google Scholar] [CrossRef] [PubMed]
- Wiese, M.; Wenzel, A.; Hintze, H.; Petersson, A.; Knutsson, K.; Bakke, M.; List, T.; Svensson, P. Osseous changes and condyle position in TMJ tomograms: Impact of RDC/TMD clinical diagnoses on agreement between expected and actual findings. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endodontol. 2008, 106, e52–e63. [Google Scholar] [CrossRef] [PubMed]
- Miron, D.; Raz, R.; Luder, A. Congenital toxoplasmosis in Israel: To screen or not to screen. Isr. Med. Assoc. J. IMAJ 2002, 4, 119–122. [Google Scholar] [PubMed]
- Iwasaki, L.R.; Gonzalez, Y.M.; Liu, Y.; Liu, H.; Markova, M.; Gallo, L.M.; Nickel, J.C. TMJ energy densities in healthy men and women. Osteoarthr. Cartil. 2017, 25, 846–849. [Google Scholar] [CrossRef]
- Wang, X.D.; Zhang, J.N.; Gan, Y.H.; Zhou, Y.H. Current understanding of pathogenesis and treatment of TMJ osteoarthritis. J. Dent. Res. 2015, 94, 666–673. [Google Scholar] [CrossRef]
- Liu, Q.; Yang, H.; Zhang, M.; Zhang, J.; Lu, L.; Yu, S.; Wu, Y.; Wang, M. Initiation and progression of dental-stimulated temporomandibular joints osteoarthritis. Osteoarthr. Cartil. 2021, 29, 633–642. [Google Scholar] [CrossRef]
- Ferrillo, M.; Nucci, L.; Giudice, A.; Calafiore, D.; Marotta, N.; Minervini, G.; d’Apuzzo, F.; Ammendolia, A.; Perillo, L.; de Sire, A. Efficacy of conservative approaches on pain relief in patients with temporomandibular joint disorders: A systematic review with network meta-analysis. Cranio 2022, 1–17. [Google Scholar] [CrossRef]
- Raman, D.; Sobolik-Delmaire, T.; Richmond, A. Chemokines in health and disease. Exp. Cell Res. 2011, 317, 575–589. [Google Scholar] [CrossRef]
- Ogura, N.; Tobe, M.; Sakamaki, H.; Nagura, H.; Abiko, Y.; Kondoh, T. Tumor necrosis factor-α increases chemokine gene expression and production in synovial fibroblasts from human temporomandibular joint. J. Oral Pathol. Med. 2005, 34, 357–363. [Google Scholar] [CrossRef] [PubMed]
- Tipton, D.A.; Christian, J.; Blumer, A. Effects of cranberry components on IL-1beta-stimulated production of IL-6, IL-8 and VEGF by human TMJ synovial fibroblasts. Arch. Oral Biol. 2016, 68, 88–96. [Google Scholar] [CrossRef] [PubMed]
- Derwich, M.; Mitus-Kenig, M.; Pawlowska, E. Interdisciplinary Approach to the Temporomandibular Joint Osteoarthritis-Review of the Literature. Medicina 2020, 56, 225. [Google Scholar] [CrossRef]
- Yang, T.; Zhang, J.; Cao, Y.; Zhang, M.; Jing, L.; Jiao, K.; Yu, S.; Chang, W.; Chen, D.; Wang, M. Wnt5a/Ror2 mediates temporomandibular joint subchondral bone remodeling. J. Dent. Res. 2015, 94, 803–812. [Google Scholar] [CrossRef] [PubMed]
- Yuan, J.; Ding, W.; Wu, N.; Jiang, S.; Li, W. Protective Effect of Genistein on Condylar Cartilage through Downregulating NF-kappaB Expression in Experimentally Created Osteoarthritis Rats. BioMed Res. Int. 2019, 2019, 2629791. [Google Scholar] [CrossRef]
- Zhang, S.; Teo, K.Y.W.; Chuah, S.J.; Lai, R.C.; Lim, S.K.; Toh, W.S. MSC exosomes alleviate temporomandibular joint osteoarthritis by attenuating inflammation and restoring matrix homeostasis. Biomaterials 2019, 200, 35–47. [Google Scholar] [CrossRef]
- Hughes, C.E.; Nibbs, R.J.B. A guide to chemokines and their receptors. FEBS J. 2018, 285, 2944–2971. [Google Scholar] [CrossRef]
- Gilchrist, A. Chemokines and Bone. In Bone Regulators and Osteoporosis Therapy; Handbook of Experimental Pharmacology; Stern, P.H., Ed.; Springer: Cham, Switzerland, 2020; Volume 262. [Google Scholar] [CrossRef]
- Ridiandries, A.; Tan, J.T.M.; Bursill, C.A. The Role of Chemokines in Wound Healing. Int. J. Mol. Sci. 2018, 19, 3217. [Google Scholar] [CrossRef]
- van der Vorst, E.P.; Doring, Y.; Weber, C. Chemokines. Arterioscler. Thromb. Vasc. Biol. 2015, 35, e52–e56. [Google Scholar] [CrossRef]
- Charo, I.F.; Ransohoff, R.M. The many roles of chemokines and chemokine receptors in inflammation. N. Engl. J. Med. 2006, 354, 610–621. [Google Scholar] [CrossRef]
- Legler, D.F.; Thelen, M. Chemokines: Chemistry, Biochemistry and Biological Function. Chimia 2016, 70, 856–859. [Google Scholar] [CrossRef] [PubMed]
- Baggiolini, M.; Dewald, B.; Moser, B. Interleukin-8 and related chemotactic cytokines—CXC and CC chemokines. Adv. Immunol. 1994, 55, 97–179. [Google Scholar] [PubMed]
- Baggiolini, M.; Loetscher, P.; Moser, B. Interleukin-8 and the chemokine family. Int. J. Immunopharmacol. 1995, 17, 103–108. [Google Scholar] [CrossRef]
- Larsen, C.G.; Anderson, A.O.; Appella, E.; Oppenheim, J.J.; Matsushima, K. The neutrophil-activating protein (NAP-1) is also chemotactic for T lymphocytes. Science 1989, 243, 1464–1466. [Google Scholar] [CrossRef]
- Kim, H.; Lee, B.K. Anti-Inflammatory Effect of Adipose-Derived Stromal Vascular Fraction on Osteoarthritic Temporomandibular Joint Synoviocytes. Tissue Eng. Regen. Med. 2020, 17, 351–362. [Google Scholar] [CrossRef] [PubMed]
- Borzi, R.M.; Mazzetti, I.; Marcu, K.B.; Facchini, A. Chemokines in cartilage degradation. Clin. Orthop. Relat. Res. 2004, 427, S53–S61. [Google Scholar] [CrossRef]
- Sukedai, M.; Tominaga, K.; Habu, M.; Matsukawa, A.; Nishihara, T.; Fukuda, J. Involvement of tumor necrosis factor-α and interleukin-8 in antigen-induced arthritis of the rabbit temporomandibular joint. J. Oral Pathol. Med. 2004, 33, 102–110. [Google Scholar] [CrossRef]
- Villalvilla, A.; Gomez, R.; Roman-Blas, J.A.; Largo, R.; Herrero-Beaumont, G. SDF-1 signaling: A promising target in rheumatic diseases. Expert Opin. Ther. Targets 2014, 18, 1077–1087. [Google Scholar] [CrossRef]
- Yang, J.; Li, Y.; Liu, Y.; Zhang, Q.; Zhang, Q.; Chen, J.; Yan, X.; Yuan, X. Role of the SDF-1/CXCR4 signaling pathway in cartilage and subchondral bone in temporomandibular joint osteoarthritis induced by overloaded functional orthopedics in rats. J. Orthop. Surg. Res. 2020, 15, 330. [Google Scholar] [CrossRef]
- Lu, L.; Zhang, X.; Zhang, M.; Zhang, H.; Liao, L.; Yang, T.; Zhang, J.; Xian, L.; Chen, D.; Wang, M. RANTES and SDF-1 Are Keys in Cell-based Therapy of TMJ Osteoarthritis. J. Dent. Res. 2015, 94, 1601–1609. [Google Scholar] [CrossRef] [PubMed]
- Hosogane, N.; Huang, Z.; Rawlins, B.A.; Liu, X.; Boachie-Adjei, O.; Boskey, A.L.; Zhu, W. Stromal derived factor-1 regulates bone morphogenetic protein 2-induced osteogenic differentiation of primary mesenchymal stem cells. Int. J. Biochem. Cell Biol. 2010, 42, 1132–1141. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Lin, S.; Sun, Y.; Guo, J.; Lu, Y.; Suen, C.W.; Zhang, J.; Zha, Z.; Ho, K.W.; Pan, X.; et al. Attenuation of subchondral bone abnormal changes in osteoarthritis by inhibition of SDF-1 signaling. Osteoarthr. Cartil. 2017, 25, 986–994. [Google Scholar] [CrossRef]
- Wei, F.; Moore, D.C.; Li, Y.; Zhang, G.; Wei, X.; Lee, J.K.; Wei, L. Attenuation of osteoarthritis via blockade of the SDF-1/CXCR4 signaling pathway. Arthritis Res. Ther. 2012, 14, R177. [Google Scholar] [CrossRef] [PubMed]
- Nanki, T.; Imai, T.; Kawai, S. Fractalkine/CX3CL1 in rheumatoid arthritis. Mod. Rheumatol. 2017, 27, 392–397. [Google Scholar] [CrossRef]
- Fan, W.; Zhu, X.; He, Y.; Zhu, M.; Wu, Z.; Huang, F.; He, H. The role of satellite glial cells in orofacial pain. J. Neurosci. Res. 2019, 97, 393–401. [Google Scholar] [CrossRef]
- Brennan, F.M.; McInnes, I.B. Evidence that cytokines play a role in rheumatoid arthritis. J. Clin. Investig. 2008, 118, 3537–3545. [Google Scholar] [CrossRef]
- Conte, F.D.P.; Barja-Fidalgo, C.; Verri, W.A., Jr.; Cunha, F.Q.; Rae, G.A.; Penido, C.; Henriques, M.D.G.M. Endothelins modulate inflammatory reaction in zymosan-induced arthritis: Participation of LTB4, TNF-α, and CXCL-1. J. Leukoc. Biol. 2008, 84, 652–660. [Google Scholar] [CrossRef]
- Clark, A.K.; Yip, P.K.; Grist, J.; Gentry, C.; Staniland, A.A.; Marchand, F.; Dehvari, M.; Wotherspoon, G.; Winter, J.; Ullah, J.; et al. Inhibition of spinal microglial cathepsin S for the reversal of neuropathic pain. Proc. Natl. Acad. Sci. USA 2007, 104, 10655–10660. [Google Scholar] [CrossRef]
- Clark, A.K.; Yip, P.K.; Malcangio, M. The liberation of fractalkine in the dorsal horn requires microglial cathepsin S. J. Neurosci. 2009, 29, 6945–6954. [Google Scholar] [CrossRef]
- Nieto, F.R.; Clark, A.K.; Grist, J.; Hathway, G.J.; Chapman, V.; Malcangio, M. Neuron-immune mechanisms contribute to pain in early stages of arthritis. J. Neuroinflamm. 2016, 13, 96. [Google Scholar] [CrossRef]
- Munoz-Lora, V.R.M.; Abdalla, H.B.; Cury, A.A.D.B.; Clemente-Napimoga, J.T. Modulatory effect of botulinum toxin type A on the microglial P2X7/CatS/FKN activated-pathway in antigen-induced arthritis of the temporomandibular joint of rats. Toxicon 2020, 187, 116–121. [Google Scholar] [CrossRef] [PubMed]
- Bonfante, R.; Napimoga, M.H.; Macedo, C.G.; Abdalla, H.B.; Pieroni, V.; Clemente-Napimoga, J.T. The P2X7 Receptor, Cathepsin S and Fractalkine in the Trigeminal Subnucleus Caudalis Signal Persistent Hypernociception in Temporomandibular Rat Joints. Neuroscience 2018, 391, 120–130. [Google Scholar] [CrossRef] [PubMed]
- Ellingsen, T.; Hornung, N.; Møller, B.K.; Poulsen, J.H.; Stengaard-Pedersen, K. Differential effect of methotrexate on the increased CCR2 density on circulating CD4 T lymphocytes and monocytes in active chronic rheumatoid arthritis, with a down regulation only on monocytes in responders. Ann. Rheum. Dis. 2007, 66, 151–157. [Google Scholar] [CrossRef] [PubMed]
- Yao, T.C.; Kuo, M.L.; See, L.C.; Ou, L.S.; Lee, W.I.; Chan, C.K.; Huang, J.L. RANTES and monocyte chemoattractant protein 1 as sensitive markers of disease activity in patients with juvenile rheumatoid arthritis: A six-year longitudinal study. Arthritis Rheum. Off. J. Am. Coll. Rheumatol. 2006, 54, 2585–2593. [Google Scholar] [CrossRef] [PubMed]
- Pierer, M.; Rethage, J.; Seibl, R.; Lauener, R.; Brentano, F.; Wagner, U.; Hantzschel, H.; Michel, B.A.; Gay, R.E.; Gay, S.; et al. Chemokine secretion of rheumatoid arthritis synovial fibroblasts stimulated by Toll-like receptor 2 ligands. J. Immunol. 2004, 172, 1256–1265. [Google Scholar] [CrossRef] [PubMed]
- Daisuke, I.; Toshio, H.; Naomi, O. Effect of IL-17 for monocyte chemotactic protein production by human temporomandibular joint synovial fibroblasts. Int. J. Oral Med. Sci. 2019, 18, 1–9. [Google Scholar] [CrossRef]
- Ogura, N.; Satoh, K.; Akutsu, M.; Tobe, M.; Kuyama, K.; Kuboyama, N.; Sakamaki, H.; Kujiraoka, H.; Kondoh, T. MCP-1 production in temporomandibular joint inflammation. J. Dent. Res. 2010, 89, 1117–1122. [Google Scholar] [CrossRef] [PubMed]
- Rossi, D.L.; Vicari, A.P.; Franz-Bacon, K.; McClanahan, T.K.; Zlotnik, A. Identification through bioinformatics of two new macrophage proinflammatory human chemokines: MIP-3alpha and MIP-3beta. J. Immunol. 1997, 158, 1033–1036. [Google Scholar] [CrossRef]
- Schutyser, E.; Struyf, S.; Van Damme, J. The CC chemokine CCL20 and its receptor CCR6. Cytokine Growth Factor Rev. 2003, 14, 409–426. [Google Scholar] [CrossRef]
- Cavanagh, L.L.; Boyce, A.; Smith, L.; Padmanabha, J.; Filgueira, L.; Pietschmann, P.; Thomas, R. Rheumatoid arthritis synovium contains plasmacytoid dendritic cells. Arthritis Res. Ther. 2005, 7, R230–R240. [Google Scholar] [CrossRef] [PubMed]
- Tran, C.N.; Lundy, S.K.; Fox, D.A. Synovial biology and T cells in rheumatoid arthritis. Pathophysiology 2005, 12, 183–189. [Google Scholar] [CrossRef] [PubMed]
- Matsui, T.; Akahoshi, T.; Namai, R.; Hashimoto, A.; Kurihara, Y.; Rana, M.; Nishimura, A.; Endo, H.; Kitasato, H.; Kawai, S.; et al. Selective recruitment of CCR6-expressing cells by increased production of MIP-3 α in rheumatoid arthritis. Clin. Exp. Immunol. 2001, 125, 155–161. [Google Scholar] [CrossRef]
- Kwon, J.H.; Keates, S.; Simeonidis, S.; Grall, F.; Libermann, T.A.; Keates, A.C. ESE-1, an enterocyte-specific Ets transcription factor, regulates MIP-3alpha gene expression in Caco-2 human colonic epithelial cells. J. Biol. Chem. 2003, 278, 875–884. [Google Scholar] [CrossRef]
- Monasterio, G.; Castillo, F.; Rojas, L.; Cafferata, E.A.; Alvarez, C.; Carvajal, P.; Núñez, C.; Flores, G.; Díaz, W.; Vernal, R. Th1/Th17/Th22 immune response and their association with joint pain, imagenological bone loss, RANKL expression and osteoclast activity in temporomandibular joint osteoarthritis: A preliminary report. J. Oral Rehabil. 2018, 45, 589–597. [Google Scholar] [CrossRef]
- Crincoli, V.; Anelli, M.G.; Quercia, E.; Piancino, M.G.; Di Comite, M. Temporomandibular Disorders and Oral Features in Early Rheumatoid Arthritis Patients: An Observational Study. Int. J. Med. Sci. 2019, 16, 253–263. [Google Scholar] [CrossRef] [PubMed]
- Goldring, M.B.; Goldring, S.R. Osteoarthritis. J. Cell. Physiol. 2007, 213, 626–634. [Google Scholar] [CrossRef]
- Liao, F.; Shirakawa, A.K.; Foley, J.F.; Rabin, R.L.; Farber, J.M. Human B cells become highly responsive to macrophage-inflammatory protein-3 α/CC chemokine ligand-20 after cellular activation without changes in CCR6 expression or ligand binding. J. Immunol. 2002, 168, 4871–4880. [Google Scholar] [CrossRef]
- Schall, T.J.; Bacon, K.; Toy, K.J.; Goeddel, D.V. Selective attraction of monocytes and T lymphocytes of the memory phenotype by cytokine RANTES. Nature 1990, 347, 669–671. [Google Scholar] [CrossRef]
- Rot, A.; Krieger, M.; Brunner, T.; Bischoff, S.C.; Schall, T.J.; Dahinden, C.A. RANTES and macrophage inflammatory protein 1 α induce the migration and activation of normal human eosinophil granulocytes. J. Exp. Med. 1992, 176, 1489–1495. [Google Scholar] [CrossRef]
- von Luettichau, I.; Nelson, P.J.; Pattison, J.M.; Van De Rijn, M.; Huie, P.; Warnke, R.; Wiedermann, C.J.; Stahl, R.A.; Sibley, R.K.; Krensky, A.M. RANTES chemokine expression in diseased and normal human tissues. Cytokine 1996, 8, 89–98. [Google Scholar] [CrossRef]
- Agere, S.A.; Akhtar, N.; Watson, J.M.; Ahmed, S. RANTES/CCL5 Induces Collagen Degradation by Activating MMP-1 and MMP-13 Expression in Human Rheumatoid Arthritis Synovial Fibroblasts. Front. Immunol. 2017, 8, 1341. [Google Scholar] [CrossRef]
- Feng, S.Y.; Lei, J.; Chen, H.M.; Wang, Y.X.; Yap, A.U.J.; Fu, K.Y. Increased chemokine RANTES in synovial fluid and its role in early-stage degenerative temporomandibular joint disease. J. Oral Rehabil. 2020, 47, 1150–1160. [Google Scholar] [CrossRef]
- Ogura, N.; Tobe, M.; Sakamaki, H.; Nagura, H.; Hosaka, H.; Akiba, M.; Abiko, Y.; Kondoh, T. Interleukin-1beta increases RANTES gene expression and production in synovial fibroblasts from human temporomandibular joint. J. Oral Pathol. Med. 2004, 33, 629–633. [Google Scholar] [CrossRef]
- Bondue, B.; Wittamer, V.; Parmentier, M. Chemerin and its receptors in leukocyte trafficking, inflammation and metabolism. Cytokine Growth Factor Rev. 2011, 22, 331–338. [Google Scholar] [CrossRef] [PubMed]
- Meder, W.; Wendland, M.; Busmann, A.; Kutzleb, C.; Spodsberg, N.; John, H.; Richter, R.; Schleuder, D.; Meyer, M.; Forssmann, W.G. Characterization of human circulating TIG2 as a ligand for the orphan receptor ChemR23. FEBS Lett. 2003, 555, 495–499. [Google Scholar] [CrossRef]
- Jin, C.H.; Yi, K.W.; Ha, Y.R.; Shin, J.H.; Park, H.T.; Kim, T.; Hur, J.Y. Chemerin Expression in the Peritoneal Fluid, Serum, and Ovarian Endometrioma of Women with Endometriosis. Am. J. Reprod. Immunol. 2015, 74, 379–386. [Google Scholar] [CrossRef] [PubMed]
- Ernst, M.C.; Issa, M.; Goralski, K.B.; Sinal, C.J. Chemerin exacerbates glucose intolerance in mouse models of obesity and diabetes. Endocrinology 2010, 151, 1998–2007. [Google Scholar] [CrossRef] [PubMed]
- Kaneko, K.; Miyabe, Y.; Takayasu, A.; Fukuda, S.; Miyabe, C.; Ebisawa, M.; Yokoyama, W.; Watanabe, K.; Imai, T.; Muramoto, K.; et al. Chemerin activates fibroblast-like synoviocytes in patients with rheumatoid arthritis. Arthritis Res. Ther. 2011, 13, R158. [Google Scholar] [CrossRef] [PubMed]
- Wittamer, V.; Franssen, J.D.; Vulcano, M.; Mirjolet, J.F.; Le Poul, E.; Migeotte, I.; Brézillon, S.; Tyldesley, R.; Blanpain, C.; Detheux, M.; et al. Specific recruitment of antigen-presenting cells by chemerin, a novel processed ligand from human inflammatory fluids. J. Exp. Med. 2003, 198, 977–985. [Google Scholar] [CrossRef]
- Ha, Y.J.; Kang, E.J.; Song, J.S.; Park, Y.B.; Lee, S.K.; Choi, S.T. Plasma chemerin levels in rheumatoid arthritis are correlated with disease activity rather than obesity. Jt. Bone Spine 2013, 81, 189–190. [Google Scholar] [CrossRef] [PubMed]
- Berg, V.; Sveinbjörnsson, B.; Bendiksen, S.; Brox, J.; Meknas, K.; Figenschau, Y. Human articular chondrocytes express ChemR23 and chemerin; ChemR23 promotes inflammatory signalling upon binding the ligand chemerin21-157. Arthritis Res. Ther. 2010, 12, R228. [Google Scholar] [CrossRef] [PubMed]
- Conde, J.; Gomez, R.; Bianco, G.; Scotece, M.; Lear, P.; Dieguez, C.; Gomez-Reino, J.; Lago, F.; Gualillo, O. Expanding the adipokine network in cartilage: Identification and regulation of novel factors in human and murine chondrocytes. Ann. Rheum. Dis. 2011, 70, 551–559. [Google Scholar] [CrossRef] [PubMed]
- Kaya, G.S.; Yavuz, G.Y.; Kiziltunc, A. Expression of chemerin in the synovial fluid of patients with temporomandibular joint disorders. J. Oral Rehabil. 2018, 45, 289–294. [Google Scholar] [CrossRef]
- Takahashi, T.; Kondoh, T.; Fukuda, M.; Yamazaki, Y.; Toyosaki, T.; Suzuki, R. Proinflammatory cytokines detectable in synovial fluids from patients with temporomandibular disorders. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endodontol. 1998, 85, 135–141. [Google Scholar] [CrossRef]
- Emshoff, R.; Puffer, P.; Rudisch, A.; Gaßner, R. Temporomandibular joint pain: Relationship to internal derangement type, osteoarthrosis, and synovial fluid mediator level of tumor necrosis factor-α. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endodontol. 2000, 90, 442–449. [Google Scholar] [CrossRef]
- Baggiolini, M.; Walz, A.; Kunkel, S.L. Neutrophil-activating peptide-1/interleukin 8, a novel cytokine that activates neutrophils. J. Clin. Investig. 1989, 84, 1045–1049. [Google Scholar] [CrossRef]
- Harris, E.D., Jr. Rheumatoid arthritis. Pathophysiology and implications for therapy. N. Engl. J. Med. 1990, 322, 1277–1289. [Google Scholar]
- Horiuchi, T.; Yoshida, T.; Koshihara, Y.; Sakamoto, H.; Kanai, H.; Yamamoto, S.; Ito, H. The increase of parathyroid hormone-related peptide and cytokine levels in synovial fluid of elderly rheumatoid arthritis and osteoarthritis. Endocr. J. 1999, 46, 643–649. [Google Scholar] [CrossRef]
- Chin, J.E.; Winterrowd, G.E.; Krzesicki, R.F.; Sanders, M.E. Role of cytokines in inflammatory synovitis. The coordinate regulation of intercellular adhesion molecule 1 and HLA class I and class II antigens in rheumatoid synovial fibroblasts. Arthritis Rheum. Off. J. Am. Coll. Rheumatol. 1990, 33, 1776–1786. [Google Scholar] [CrossRef]
- Tobe, M.; Ogura, N.; Abiko, Y.; Nagura, H. Interleukin-1beta stimulates interleukin-8 production and gene expression in synovial cells from human temporomandibular joint. J. Oral Maxillofac. Surg. 2002, 60, 741–747. [Google Scholar] [CrossRef]
- Patterson, A.M.; Schmutz, C.; Davis, S.; Gardner, L.; Ashton, B.A.; Middleton, J. Differential binding of chemokines to macrophages and neutrophils in the human inflamed synovium. Arthritis Res. 2002, 4, 209–214. [Google Scholar] [CrossRef]
- Ibi, M.; Horie, S.; Kyakumoto, S.; Chosa, N.; Yoshida, M.; Kamo, M.; Ohtsuka, M.; Ishisaki, A. Cell-cell interactions between monocytes/macrophages and synoviocyte-like cells promote inflammatory cell infiltration mediated by augmentation of MCP-1 production in temporomandibular joint. Biosci. Rep. 2018, 38, BSR20171217. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Ma, K.; Yi, D.; Oh, C.D.; Chen, D. Nociceptive behavioural assessments in mouse models of temporomandibular joint disorders. Int. J. Oral Sci. 2020, 12, 26. [Google Scholar] [CrossRef] [PubMed]
- Qiao, Y.; Yi, D.; Reed, D.A.; Mercuri, L.G.; Chen, D. A novel approach to establishing a temporomandibular joint fibrocartilage cell line. J. Dent. Sci. 2022, 17, 1378–1386. [Google Scholar] [CrossRef] [PubMed]

| Chemokines | Cells | Main Findings | |
|---|---|---|---|
| CXC(α) | IL-8 | macrophages, epithelial cells, airway smooth muscle cells, and endothelial cells | IL-8 is a powerful neutrophil attraction and activation cytokine in TMJ RA and OA. Upregulation of IL-8 in SMSCs caused by IL-1β also occurs by activating the NF-κB pathway |
| SDF-1 | hematopoietic stem cells | Activation of the SDF-1/CXCR4 signaling pathway regulates the expression of various inflammatory factors, including IL-1β, IL-6, TNF-α, and MMPs involved in TMJ pathology. | |
| GRO-α | neutrophils | Growth of new, small blood vessels in the TMJ synovium | |
| CC(β) | MCP-1 | monocytes, lymphocytes | IL-1β-stimulated temporomandibular joint synovial cells produce and release MCP-1, which is associated with the early stages of temporomandibular joint inflammation. MCP-1 may be a major factor in the onset, subsequent progression, and chronicity of TMJ synovial inflammation |
| MIP-1α, 1β | monocytes, T lymphocytes | Recruiting inflammatory cells, wound healing, inhibition of stem cells, and maintaining effector immune response | |
| MIP-3α | lymphocytes and dendritic cells | Increase in MIP-3a may trigger the migration of dendritic cells, T cells, and B cells into the synovial tissue and body fluids of patients with TMJ-ID, and may lead to the onset and progression of inflammatory alterations in TMJ. | |
| RANTES | monocytes, T lymphocytes | RANTES/CCR1 signals are key signals that may play a synergistic role in GFP BMSCs for recruiting OA cartilage from the temporomandibular joint. | |
| CX3C(δ) | Fkn (fractalkine, CX3CL1) | monocytes, natural killer cells, T cells, and smooth muscle cells | In the trigeminal nervous system, the persistent albumin-induced model of arthritis hyperphagia in TMJ activates the trigeminal tail subnuclear signal through the P2X7/CatS/FKN pathway |
| Other | Chemerin-ChemR23 | dendritic cells, macrophages, adipocytes | The interaction of inflammatory factors and Chemerin increases the inflammatory effect. Chemerin levels were positively correlated with TMJ pain. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Qiao, Y.; Li, J.; Yuh, C.; Ko, F.; Mercuri, L.G.; Alkhudari, J.; Pourzal, R.; Oh, C.-d. Chemokine Regulation in Temporomandibular Joint Disease: A Comprehensive Review. Genes 2023, 14, 408. https://doi.org/10.3390/genes14020408
Qiao Y, Li J, Yuh C, Ko F, Mercuri LG, Alkhudari J, Pourzal R, Oh C-d. Chemokine Regulation in Temporomandibular Joint Disease: A Comprehensive Review. Genes. 2023; 14(2):408. https://doi.org/10.3390/genes14020408
Chicago/Turabian StyleQiao, Yusen, Jun Li, Catherine Yuh, Frank Ko, Louis G. Mercuri, Jad Alkhudari, Robin Pourzal, and Chun-do Oh. 2023. "Chemokine Regulation in Temporomandibular Joint Disease: A Comprehensive Review" Genes 14, no. 2: 408. https://doi.org/10.3390/genes14020408
APA StyleQiao, Y., Li, J., Yuh, C., Ko, F., Mercuri, L. G., Alkhudari, J., Pourzal, R., & Oh, C.-d. (2023). Chemokine Regulation in Temporomandibular Joint Disease: A Comprehensive Review. Genes, 14(2), 408. https://doi.org/10.3390/genes14020408






