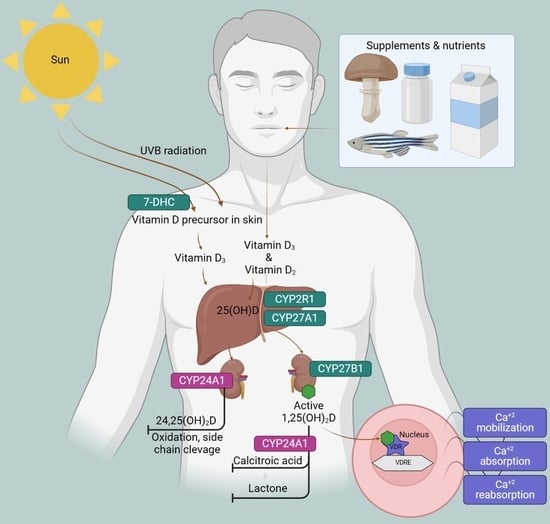
Research Trends of Vitamin D Metabolism Gene Polymorphisms Based on a Bibliometric Investigation
Abstract
1. Introduction
2. Materials and Methods
2.1. Database and Search Methodology
2.2. Data Extraction
2.3. Data Analysis
3. Results
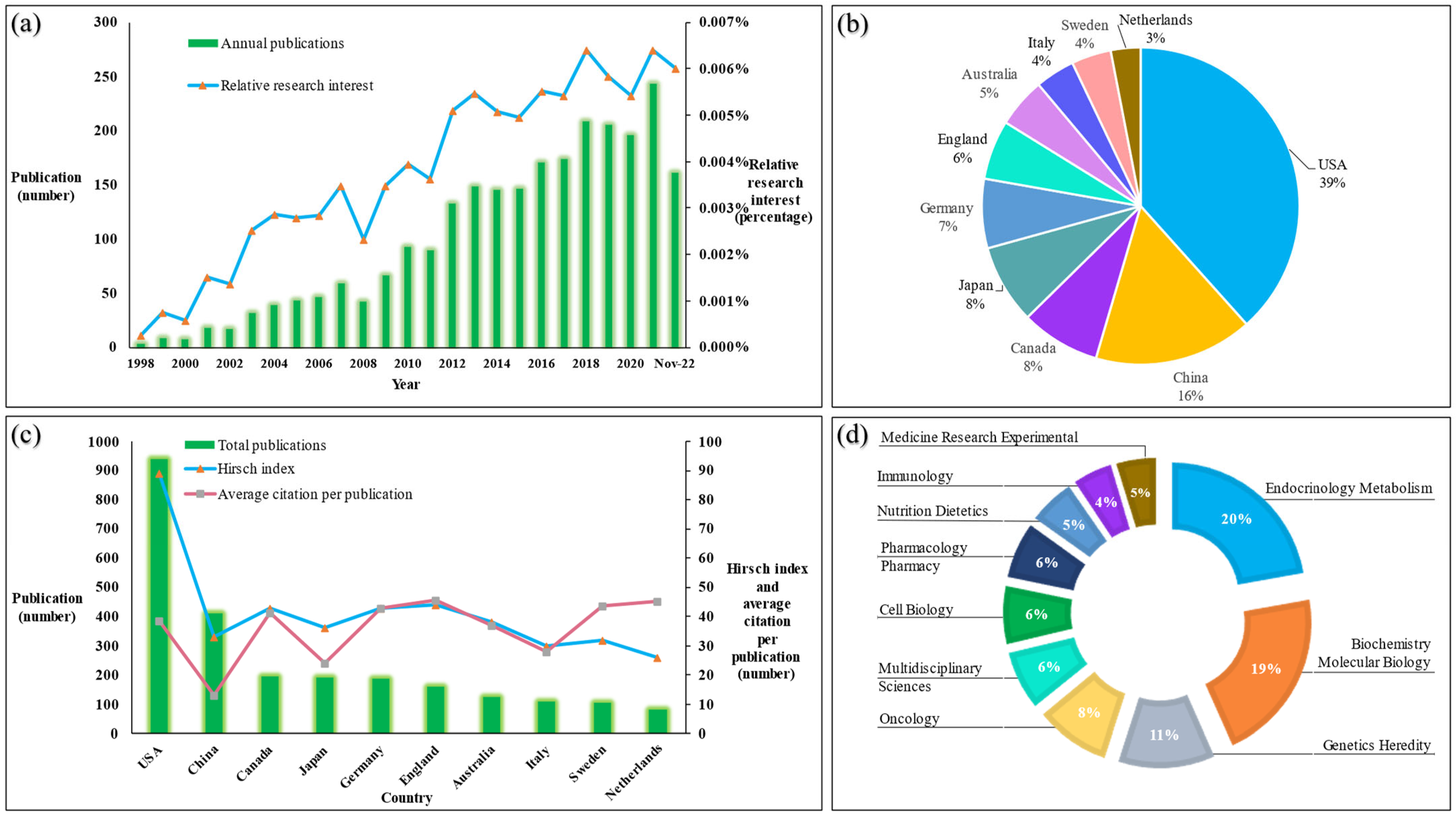
3.1. Publications about Vitamin D Metabolism Gene Polymorphisms (vitDMGPs)
3.2. Synopsis of Publications about Vitamin D Metabolism Gene Polymorphisms (vitDMGPs)
3.3. Countries of Publications and Scientific Categories
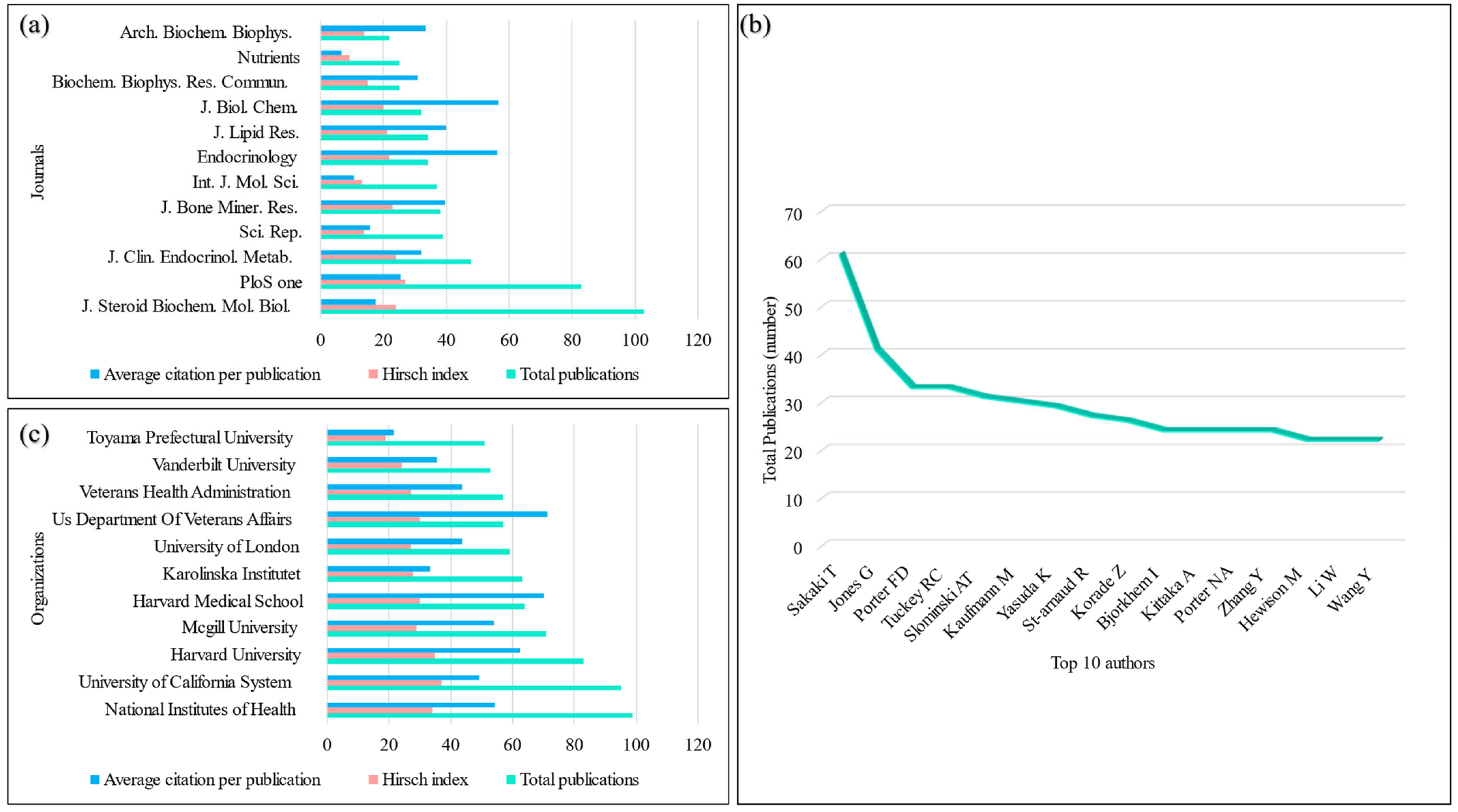
3.4. Journals of Publications and Scientific Disciplines
3.5. Authors of Publications
3.6. Affiliations of Publications
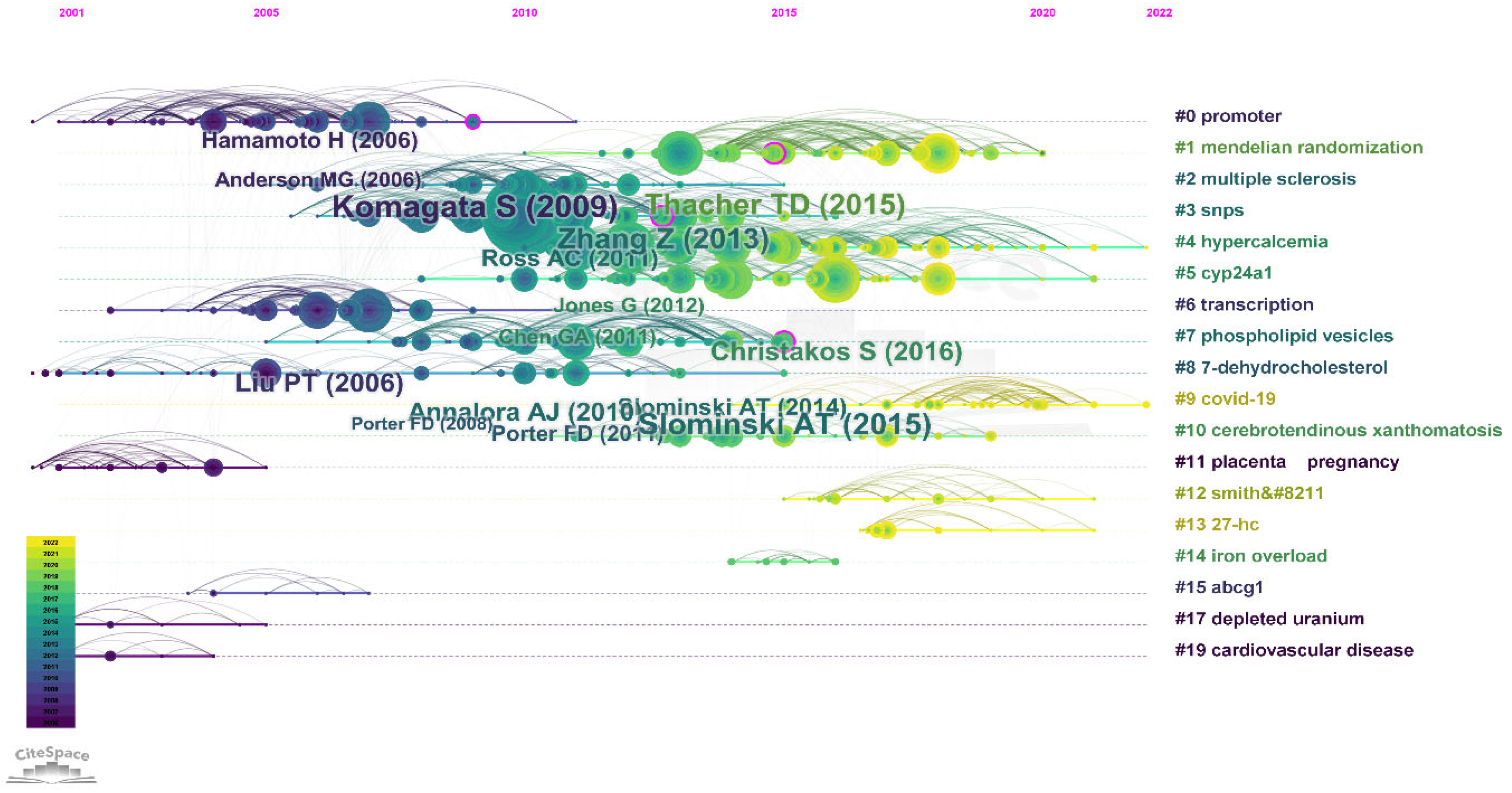
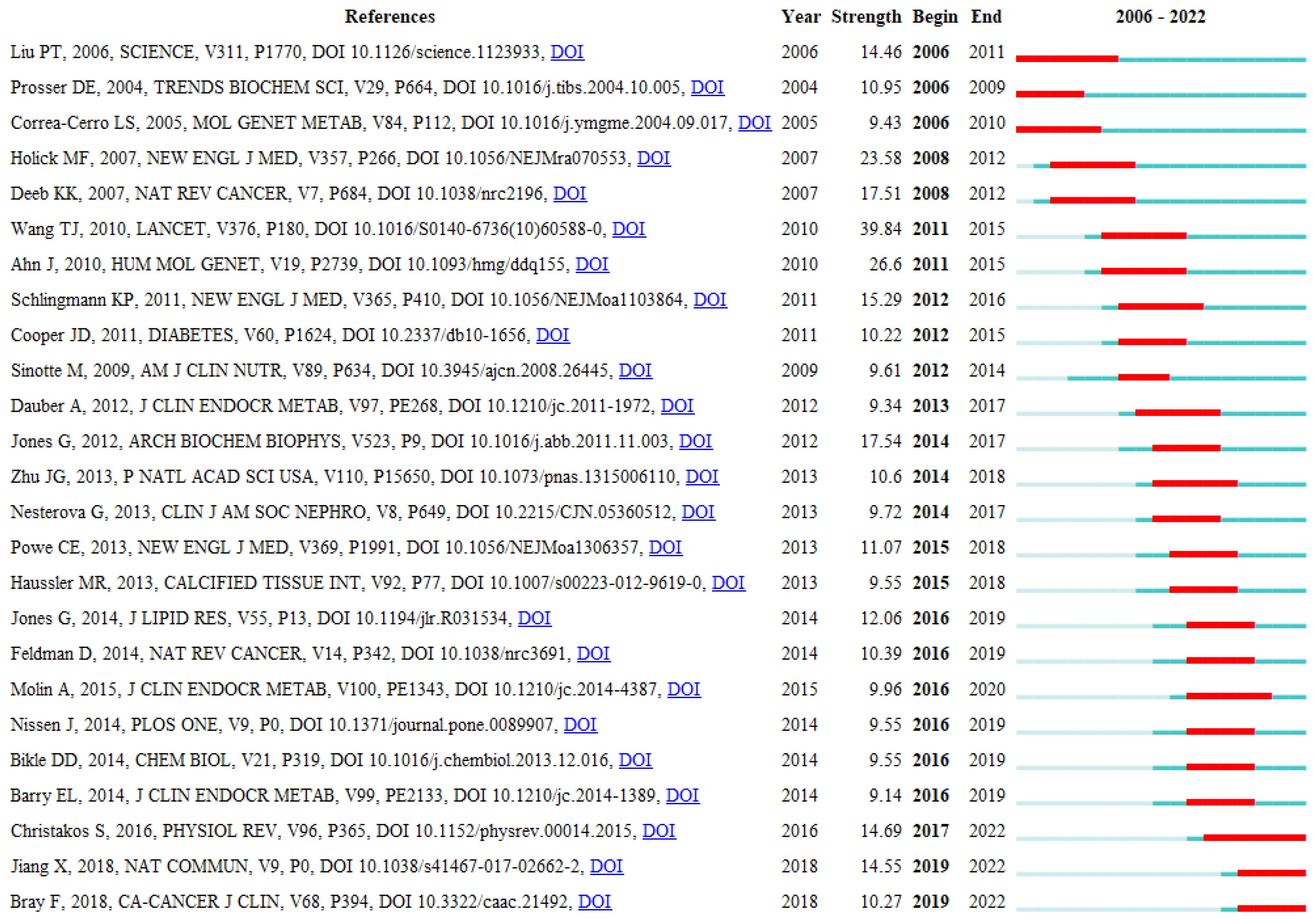
3.7. Analysis of Co-Cited References
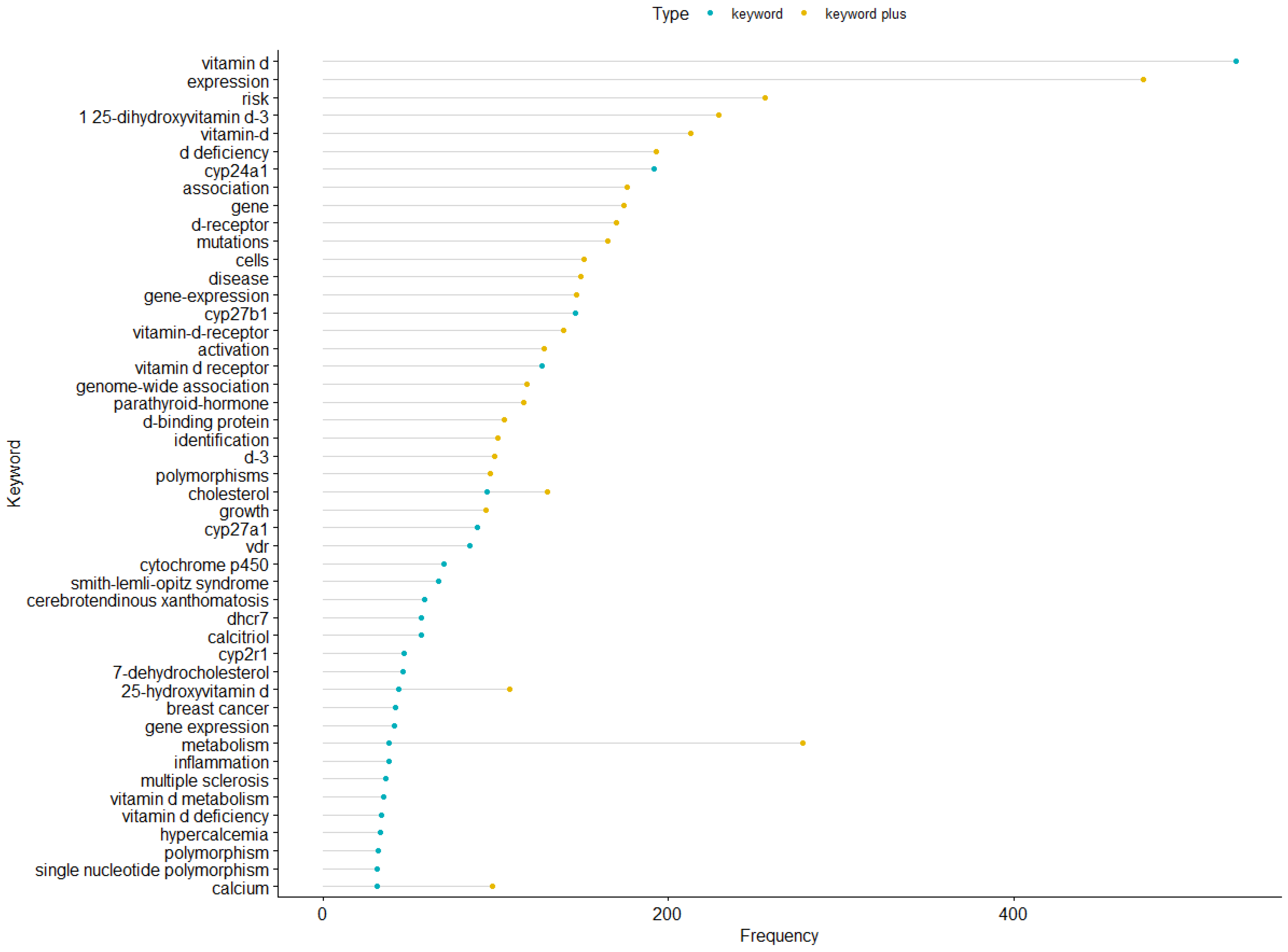
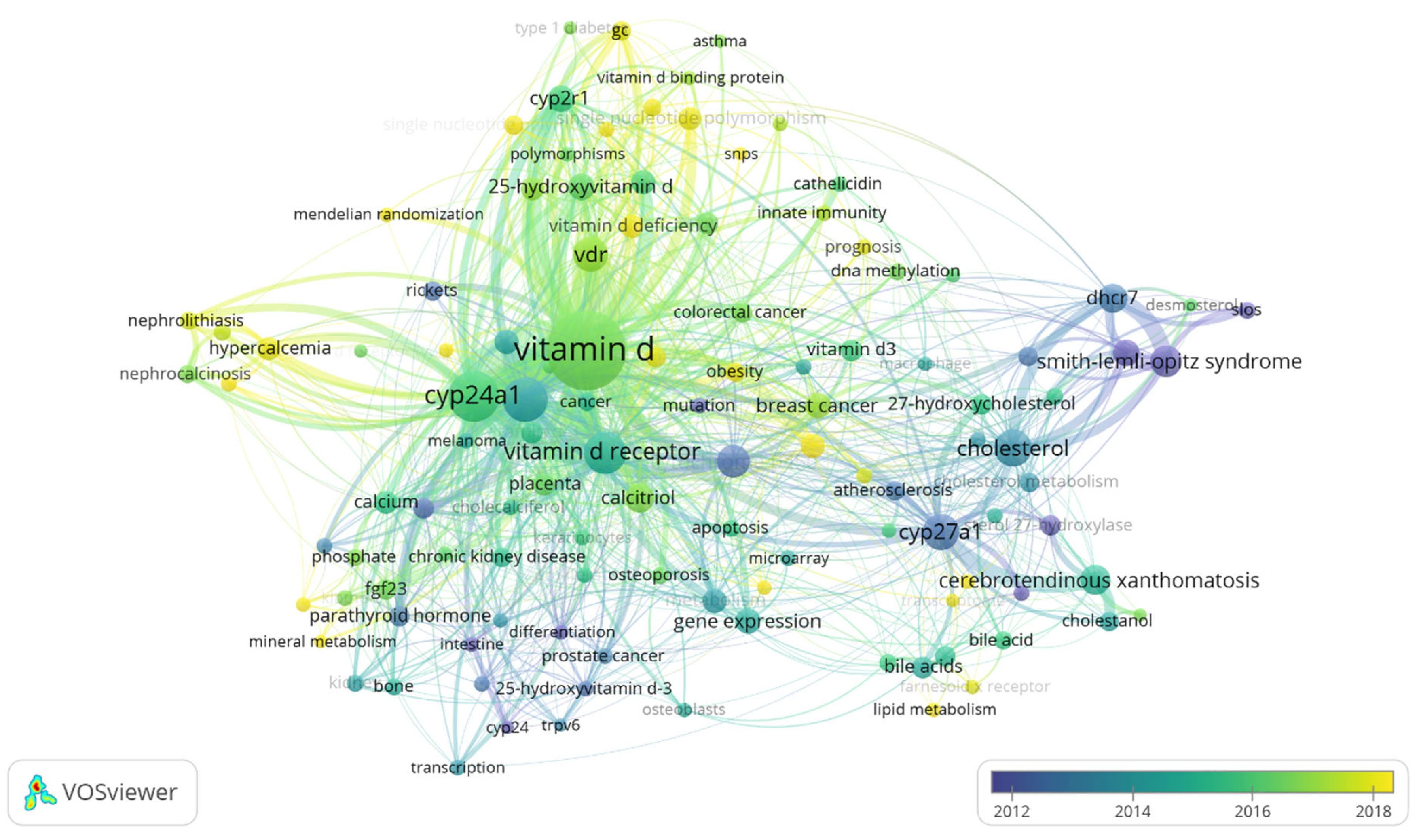
3.8. Hotspots of Papers Related to Vitamin D Metabolism Gene Polymorphisms (vitDMGPs)
4. Discussion
4.1. Trends of Publications Related Vitamin D Metabolism Gene Polymorphisms (vitDMGPs)
4.2. Studies Focused on Vitamin D Metabolism Gene Polymorphisms (vitDMGPs)
4.2.1. Global Most Cited Documents
4.2.2. Burst Documents That Co-Cited Vitamin D Metabolism Gene Polymorphisms (vitDMGPs)
4.3. Strengths and Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| Abbreviation | Explanation |
| 1,25(OH)2D3 | calcitriol |
| 25(OH)D | 25-hydroxyvitamin D |
| ABTB1 | ankyrin repeat and BTB domain containing 1 |
| COVID-19 | coronavirus disease 2019 |
| CYPs | cytochromes P450 |
| FAT1 | FAT atypical cadherin 1 |
| GC | group-specific component |
| GWAS | genome-wide association studies |
| H-index | Hirsch index |
| HIPK3 | homeodomain interacting protein kinase 3 |
| LLR | log-likelihood ratio |
| LPAR1 | lysophosphatidic acid receptor 1 |
| LSI | latent semantic indexing |
| MI | mutual information |
| PVT1 | plasmacytoma variant translocation 1 |
| RNA | ribonucleic acid |
| RRI | relative research interest |
| SNPs | single nucleotide polymorphisms |
| TC | total citation |
| VDR | vitamin D receptor |
| vitDMGPs | vitamin D metabolism gene polymorphisms |
| WOS | Web of Science |
References
- Bikle, D.D. Vitamin D Metabolism, Mechanism of Action, and Clinical Applications. Chem. Biol. 2014, 21, 319–329. [Google Scholar] [CrossRef] [PubMed]
- Neville, J.J.; Palmieri, T.; Young, A.R. Physical Determinants of Vitamin D Photosynthesis: A Review. JBMR Plus 2021, 5, e10460. [Google Scholar] [CrossRef] [PubMed]
- Touvier, M.; Deschasaux, M.; Montourcy, M.; Sutton, A.; Charnaux, N.; Kesse-Guyot, E.; Assmann, K.E.; Fezeu, L.; Latino-Martel, P.; Druesne-Pecollo, N.; et al. Determinants of Vitamin D Status in Caucasian Adults: Influence of Sun Exposure, Dietary Intake, Sociodemographic, Lifestyle, Anthropometric, and Genetic Factors. J. Investig. Dermatol. 2015, 135, 378–388. [Google Scholar] [CrossRef] [PubMed]
- Thacher, T.D.; Clarke, B.L. Vitamin D Insufficiency. Mayo Clin. Proc. 2011, 86, 50–60. [Google Scholar] [CrossRef] [PubMed]
- Christakos, S.; Dhawan, P.; Verstuyf, A.; Verlinden, L.; Carmeliet, G. Vitamin D: Metabolism, Molecular Mechanism of Action, and Pleiotropic Effects. Physiol. Rev. 2015, 96, 365–408. [Google Scholar] [CrossRef]
- Jenkinson, C.; Taylor, A.E.; Hassan-Smith, Z.K.; Adams, J.S.; Stewart, P.M.; Hewison, M.; Keevil, B.G. High Throughput LC–MS/MS Method for the Simultaneous Analysis of Multiple Vitamin D Analytes in Serum. J. Chromatogr. B 2016, 1014, 56–63. [Google Scholar] [CrossRef]
- Sugimoto, H.; Shiro, Y. Diversity and Substrate Specificity in the Structures of Steroidogenic Cytochrome P450 Enzymes. Biol. Pharm. Bull. 2012, 35, 818–823. [Google Scholar] [CrossRef]
- Haussler, M.R.; Whitfield, G.K.; Kaneko, I.; Haussler, C.A.; Hsieh, D.; Hsieh, J.-C.; Jurutka, P.W. Molecular Mechanisms of Vitamin D Action. Calcif. Tissue Int. 2013, 92, 77–98. [Google Scholar] [CrossRef]
- Pike, J.W.; Meyer, M.B.; Bishop, K.A. Regulation of Target Gene Expression by the Vitamin D Receptor—an Update on Mechanisms. Rev. Endocr. Metab. Disord. 2012, 13, 45–55. [Google Scholar] [CrossRef]
- Bouillon, R.; Schuit, F.; Antonio, L.; Rastinejad, F. Vitamin D Binding Protein: A Historic Overview. Front. Endocrinol. 2020, 10, 910. [Google Scholar] [CrossRef]
- Abouzid, M.; Kruszyna, M.; Burchardt, P.; Kruszyna, Ł.; Główka, F.K.; Karaźniewicz-Łada, M. Vitamin D Receptor Gene Polymorphism and Vitamin D Status in Population of Patients with Cardiovascular Disease—A Preliminary Study. Nutrients 2021, 13, 3117. [Google Scholar] [CrossRef]
- Abouzid, M.; Główka, F.; Karaźniewicz-Łada, M. Seasonal Pattern of Vitamin D Hydroxyl Metabolite Concentrations and Their Association with Cardiac Medications—An Observational Study. J. King Saud Univ.-Sci. 2022, 34, 102187. [Google Scholar] [CrossRef]
- Giovannucci, E.; Liu, Y.; Hollis, B.W.; Rimm, E.B. 25-Hydroxyvitamin D and Risk of Myocardial Infarction in Men: A Prospective Study. Arch. Intern. Med. 2008, 168, 1174–1180. [Google Scholar] [CrossRef]
- Pekkanen, M.P.; Ukkola, O.; Hedberg, P.; Piira, O.P.; Lepojärvi, S.; Lumme, J.; Tulppo, M.P.; Huikuri, H.V. Serum 25-Hydroxyvitamin D Is Associated with Major Cardiovascular Risk Factors and Cardiac Structure and Function in Patients with Coronary Artery Disease. Nutr. Metab. Cardiovasc. Dis. 2015, 25, 471–478. [Google Scholar] [CrossRef]
- Shoben, A.B.; Kestenbaum, B.; Levin, G.; Hoofnagle, A.N.; Psaty, B.M.; Siscovick, D.S.; de Boer, I.H. Seasonal Variation in 25-Hydroxyvitamin D Concentrations in the Cardiovascular Health Study. Am. J. Epidemiol. 2011, 174, 1363–1372. [Google Scholar] [CrossRef]
- De la Guía-Galipienso, F.; Martínez-Ferran, M.; Vallecillo, N.; Lavie, C.J.; Sanchis-Gomar, F.; Pareja-Galeano, H. Vitamin D and Cardiovascular Health. Clin. Nutr. Edinb. Scotl. 2021, 40, 2946–2957. [Google Scholar] [CrossRef]
- Kheiri, B.; Abdalla, A.; Osman, M.; Ahmed, S.; Hassan, M.; Bachuwa, G. Vitamin D Deficiency and Risk of Cardiovascular Diseases: A Narrative Review. Clin. Hypertens. 2018, 24, 9. [Google Scholar] [CrossRef]
- Kim, D.H.; Sabour, S.; Sagar, U.N.; Adams, S.; Whellan, D.J. Prevalence of Hypovitaminosis D in Cardiovascular Diseases (from the National Health and Nutrition Examination Survey 2001 to 2004). Am. J. Cardiol. 2008, 102, 1540–1544. [Google Scholar] [CrossRef]
- Barbarawi, M.; Kheiri, B.; Zayed, Y.; Barbarawi, O.; Dhillon, H.; Swaid, B.; Yelangi, A.; Sundus, S.; Bachuwa, G.; Alkotob, M.L.; et al. Vitamin D Supplementation and Cardiovascular Disease Risks in More Than 83,000 Individuals in 21 Randomized Clinical Trials: A Meta-Analysis. JAMA Cardiol. 2019, 4, 765–776. [Google Scholar] [CrossRef]
- Kowalówka, M.; Główka, A.K.; Karaźniewicz-łada, M.; Kosewski, G. Clinical Significance of Analysis of Vitamin d Status in Various Diseases. Nutrients 2020, 12, 2788. [Google Scholar] [CrossRef]
- Abouzid, M.; Karaźniewicz-Łada, M.; Pawlak, K.; Burchardt, P.; Kruszyna, Ł.; Główka, F. Measurement of Plasma 25-Hydroxyvitamin D2, 25-Hydroxyvitamin D3 and 3-Epi-25-Hydroxyvitamin D3 in Population of Patients with Cardiovascular Disease by UPLC-MS/MS Method. J. Chromatogr. B 2020, 1159, 122350. [Google Scholar] [CrossRef] [PubMed]
- Law, J.; Bauin, S.; Courtial, J.P.; Whittaker, J. Policy and the Mapping of Scientific Change: A Co-Word Analysis of Research into Environmental Acidification. Scientometrics 1988, 14, 251–264. [Google Scholar] [CrossRef]
- Thompson, D.F.; Walker, C.K. A Descriptive and Historical Review of Bibliometrics with Applications to Medical Sciences. Pharmacotherapy 2015, 35, 551–559. [Google Scholar] [CrossRef] [PubMed]
- Moral-Muñoz, J.A.; Herrera-Viedma, E.; Santisteban-Espejo, A.; Cobo, M.J. Software Tools for Conducting Bibliometric Analysis in Science: An up-to-Date Review. Prof. Inf. 2020, 29, 1699–2407. [Google Scholar] [CrossRef]
- Abouzid, M.; Główka, A.K.; Karaźniewicz-Łada, M. Trend Research of Vitamin D Receptor: Bibliometric Analysis. Health Inform. J. 2021, 27, 14604582211043158. [Google Scholar] [CrossRef]
- Bektan Kanat, B.; Yavuzer, H. Top 100 Articles on Vitamin D: Bibliometric versus Altmetric Analysis. Bratisl. Lek. Listy 2022, 123, 160–171. [Google Scholar] [CrossRef]
- Yang, A.; Lv, Q.; Chen, F.; Wang, D.; Liu, Y.; Shi, W. Identification of Recent Trends in Research on Vitamin D: A Quantitative and Co-Word Analysis. Med. Sci. Monit. 2019, 25, 643–655. [Google Scholar] [CrossRef]
- Lu, Y.; Zhang, X.; Wu, S.; Zhang, S.; Tan, J. A Bibliometric Analysis of Global Research on Vitamin D and Reproductive Health between 2012 and 2021: Learning from the Past, Planning for the Future. Front. Nutr. 2022, 9, 973332. [Google Scholar] [CrossRef]
- Malik, A.A.; Baig, M.; Butt, N.S.; Imran, M.; Alzahrani, S.H.; Gazzaz, Z.J. Bibliometric Analysis of Global Research Productivity on Vitamin D and Bone Metabolism (2001–2020): Learn from the Past to Plan Future. Nutrients 2022, 14, 542. [Google Scholar] [CrossRef]
- Caballero-Villarraso, J.; Jiménez-Jiménez, M.J.; Escribano, B.M.; Agüera, E.; Santamaría, A.; Túnez, I. Implications of Vitamin D in Multiple Sclerosis and Other Neurodegenerative Processes: Bibliometric Analysis and Systematic Review. CNS Neurol. Disord.-Drug Targets 2019, 18, 478–490. [Google Scholar] [CrossRef]
- Shah, M.W.; Ahmad, T.; Khan, M.; Muhammad, S.; Sun, G. Global Research on Vitamin D and Coronavirus Disease 2019: A Bibliometric and Visualized Study. Medicine 2022, 101, e29768. [Google Scholar] [CrossRef]
- Abouzid, M.; Główka, F.; Kagan, L.; Karaźniewicz-Łada, M. Vitamin D Metabolism Gene Polymorphisms and Their Associated Disorders: A Literature Review. Curr. Drug Metab. 2022, 23, 630–651. [Google Scholar] [CrossRef]
- Alathari, B.E.; Sabta, A.A.; Kalpana, C.A.; Vimaleswaran, K.S. Vitamin D Pathway-Related Gene Polymorphisms and Their Association with Metabolic Diseases: A Literature Review. J. Diabetes Metab. Disord. 2020, 19, 1701–1729. [Google Scholar] [CrossRef]
- Chen, C. CiteSpace II: Detecting and Visualizing Emerging Trends and Transient Patterns in Scientific Literature. J. Am. Soc. Inf. Sci. Technol. 2006, 57, 359–377. [Google Scholar] [CrossRef]
- Van Eck, N.J.; Waltman, L. Software Survey: VOSviewer, a Computer Program for Bibliometric Mapping. Scientometrics 2010, 84, 523–538. [Google Scholar] [CrossRef]
- Aria, M.; Cuccurullo, C. Bibliometrix: An R-Tool for Comprehensive Science Mapping Analysis. J. Informetr. 2017, 11, 959–975. [Google Scholar] [CrossRef]
- Xing, D.; Zhao, Y.; Dong, S.; Lin, J. Global Research Trends in Stem Cells for Osteoarthritis: A Bibliometric and Visualized Study. Int. J. Rheum. Dis. 2018, 21, 1372–1384. [Google Scholar] [CrossRef]
- Colledge, L. Field-Weighted Citation Impact. In Snowball Metrics Recipe Book; Snowball Metrics Steering Group, Elsevier: Amsterdam, The Netherlands, 2017; pp. 101–112. [Google Scholar]
- Salzman, J.; Chen, R.E.; Olsen, M.N.; Wang, P.L.; Brown, P.O. Cell-Type Specific Features of Circular RNA Expression. PLOS Genet. 2013, 9, e1003777. [Google Scholar] [CrossRef]
- Wang, T.J.; Zhang, F.; Richards, J.B.; Kestenbaum, B.; van Meurs, J.B.; Berry, D.; Kiel, D.P.; Streeten, E.A.; Ohlsson, C.; Koller, D.L.; et al. Common Genetic Determinants of Vitamin D Insufficiency: A Genome-Wide Association Study. Lancet 2010, 376, 180–188. [Google Scholar] [CrossRef]
- Ahn, J.; Yu, K.; Stolzenberg-Solomon, R.; Simon, K.C.; McCullough, M.L.; Gallicchio, L.; Jacobs, E.J.; Ascherio, A.; Helzlsouer, K.; Jacobs, K.B.; et al. Genome-Wide Association Study of Circulating Vitamin D Levels. Hum. Mol. Genet. 2010, 19, 2739–2745. [Google Scholar] [CrossRef]
- Nelson, E.R.; Wardell, S.E.; Jasper, J.S.; Park, S.; Suchindran, S.; Howe, M.K.; Carver, N.J.; Pillai, R.V.; Sullivan, P.M.; Sondhi, V.; et al. 27-Hydroxycholesterol Links Hypercholesterolemia and Breast Cancer Pathophysiology. Science 2013, 342, 1094–1098. [Google Scholar] [CrossRef] [PubMed]
- Schauber, J.; Dorschner, R.A.; Coda, A.B.; Büchau, A.S.; Liu, P.T.; Kiken, D.; Helfrich, Y.R.; Kang, S.; Elalieh, H.Z.; Steinmeyer, A.; et al. Injury Enhances TLR2 Function and Antimicrobial Peptide Expression through a Vitamin D–Dependent Mechanism. J. Clin. Investig. 2007, 117, 803–811. [Google Scholar] [CrossRef] [PubMed]
- Cheng, J.B.; Levine, M.A.; Bell, N.H.; Mangelsdorf, D.J.; Russell, D.W. Genetic Evidence That the Human CYP2R1 Enzyme Is a Key Vitamin D 25-Hydroxylase. Proc. Natl. Acad. Sci. USA 2004, 101, 7711–7715. [Google Scholar] [CrossRef] [PubMed]
- Schlingmann, K.P.; Kaufmann, M.; Weber, S.; Irwin, A.; Goos, C.; John, U.; Misselwitz, J.; Klaus, G.; Kuwertz-Bröking, E.; Fehrenbach, H.; et al. Mutations in CYP24A1 and Idiopathic Infantile Hypercalcemia. N. Engl. J. Med. 2011, 365, 410–421. [Google Scholar] [CrossRef] [PubMed]
- Hewison, M. Vitamin D and the Immune System: New Perspectives on an Old Theme. Endocrinol. Metab. Clin. N. Am. 2010, 39, 365–379. [Google Scholar] [CrossRef]
- Motola, D.L.; Cummins, C.L.; Rottiers, V.; Sharma, K.K.; Li, T.; Li, Y.; Suino-Powell, K.; Xu, H.E.; Auchus, R.J.; Antebi, A.; et al. Identification of Ligands for DAF-12 That Govern Dauer Formation and Reproduction in C. Elegans. Cell 2006, 124, 1209–1223. [Google Scholar] [CrossRef]
- Petta, S.; Cammà, C.; Scazzone, C.; Tripodo, C.; Di Marco, V.; Bono, A.; Cabibi, D.; Licata, G.; Porcasi, R.; Marchesini, G.; et al. Low Vitamin D Serum Level Is Related to Severe Fibrosis and Low Responsiveness to Interferon-Based Therapy in Genotype 1 Chronic Hepatitis C. Hepatology 2010, 51, 1158–1167. [Google Scholar] [CrossRef]
- Chen, C. The CiteSpace Manual; Drexel University College of Computing & Informatics: Philadelphia, PA, USA, 2014. [Google Scholar]
- Komagata, S.; Nakajima, M.; Takagi, S.; Mohri, T.; Taniya, T.; Yokoi, T. Human CYP24 Catalyzing the Inactivation of Calcitriol Is Post-Transcriptionally Regulated by MiR-125b. Mol. Pharmacol. 2009, 76, 702–709. [Google Scholar] [CrossRef]
- Slominski, A.T.; Li, W.; Kim, T.-K.; Semak, I.; Wang, J.; Zjawiony, J.K.; Tuckey, R.C. Novel Activities of CYP11A1 and Their Potential Physiological Significance. J. Steroid Biochem. Mol. Biol. 2015, 151, 25–37. [Google Scholar] [CrossRef]
- Liu, P.T.; Stenger, S.; Li, H.; Wenzel, L.; Tan, B.H.; Krutzik, S.R.; Ochoa, M.T.; Schauber, J.; Wu, K.; Meinken, C.; et al. Toll-Like Receptor Triggering of a Vitamin D-Mediated Human Antimicrobial Response. Science 2006, 311, 1770–1773. [Google Scholar] [CrossRef]
- Thacher, T.D.; Fischer, P.R.; Singh, R.J.; Roizen, J.; Levine, M.A. CYP2R1 Mutations Impair Generation of 25-Hydroxyvitamin D and Cause an Atypical Form of Vitamin D Deficiency. J. Clin. Endocrinol. Metab. 2015, 100, E1005–E1013. [Google Scholar] [CrossRef]
- Zhang, Z.; He, J.-W.; Fu, W.-Z.; Zhang, C.-Q.; Zhang, Z.-L. An Analysis of the Association between the Vitamin D Pathway and Serum 25-Hydroxyvitamin D Levels in a Healthy Chinese Population. J. Bone Miner. Res. 2013, 28, 1784–1792. [Google Scholar] [CrossRef]
- Prosser, D.E.; Jones, G. Enzymes Involved in the Activation and Inactivation of Vitamin D. Trends Biochem. Sci. 2004, 29, 664–673. [Google Scholar] [CrossRef]
- Correa-Cerro, L.S.; Porter, F.D. 3β-Hydroxysterol Δ7-Reductase and the Smith–Lemli–Opitz Syndrome. Mol. Genet. Metab. 2005, 84, 112–126. [Google Scholar] [CrossRef]
- Holick, M.F. Vitamin D Deficiency. N. Engl. J. Med. 2007, 357, 266–281. [Google Scholar] [CrossRef]
- Deeb, K.K.; Trump, D.L.; Johnson, C.S. Vitamin D Signalling Pathways in Cancer: Potential for Anticancer Therapeutics. Nat. Rev. Cancer 2007, 7, 684–700. [Google Scholar] [CrossRef]
- Cooper, J.D.; Smyth, D.J.; Walker, N.M.; Stevens, H.; Burren, O.S.; Wallace, C.; Greissl, C.; Ramos-Lopez, E.; Hyppönen, E.; Dunger, D.B.; et al. Inherited Variation in Vitamin D Genes Is Associated With Predisposition to Autoimmune Disease Type 1 Diabetes. Diabetes 2011, 60, 1624–1631. [Google Scholar] [CrossRef]
- Sinotte, M.; Diorio, C.; Bérubé, S.; Pollak, M.; Brisson, J. Genetic Polymorphisms of the Vitamin D Binding Protein and Plasma Concentrations of 25-Hydroxyvitamin D in Premenopausal Women. Am. J. Clin. Nutr. 2009, 89, 634–640. [Google Scholar] [CrossRef]
- Dauber, A.; Nguyen, T.T.; Sochett, E.; Cole, D.E.C.; Horst, R.; Abrams, S.A.; Carpenter, T.O.; Hirschhorn, J.N. Genetic Defect in CYP24A1, the Vitamin D 24-Hydroxylase Gene, in a Patient with Severe Infantile Hypercalcemia. J. Clin. Endocrinol. Metab. 2012, 97, E268–E274. [Google Scholar] [CrossRef]
- Jones, G.; Prosser, D.E.; Kaufmann, M. 25-Hydroxyvitamin D-24-Hydroxylase (CYP24A1): Its Important Role in the Degradation of Vitamin D. Arch. Biochem. Biophys. 2012, 523, 9–18. [Google Scholar] [CrossRef]
- Zhu, J.G.; Ochalek, J.T.; Kaufmann, M.; Jones, G.; DeLuca, H.F. CYP2R1 Is a Major, but Not Exclusive, Contributor to 25-Hydroxyvitamin D Production in Vivo. Proc. Natl. Acad. Sci. USA 2013, 110, 15650–15655. [Google Scholar] [CrossRef] [PubMed]
- Nesterova, G.; Malicdan, M.C.; Yasuda, K.; Sakaki, T.; Vilboux, T.; Ciccone, C.; Horst, R.; Huang, Y.; Golas, G.; Introne, W.; et al. 1,25-(OH)2D-24 Hydroxylase (CYP24A1) Deficiency as a Cause of Nephrolithiasis. Clin. J. Am. Soc. Nephrol. 2013, 8, 649–657. [Google Scholar] [CrossRef] [PubMed]
- Powe, C.E.; Evans, M.K.; Wenger, J.; Zonderman, A.B.; Berg, A.H.; Nalls, M.; Tamez, H.; Zhang, D.; Bhan, I.; Karumanchi, S.A.; et al. Vitamin D–Binding Protein and Vitamin D Status of Black Americans and White Americans. N. Engl. J. Med. 2013, 369, 1991–2000. [Google Scholar] [CrossRef] [PubMed]
- Jones, G.; Prosser, D.E.; Kaufmann, M. Cytochrome P450-Mediated Metabolism of Vitamin D. J. Lipid Res. 2014, 55, 13–31. [Google Scholar] [CrossRef] [PubMed]
- Feldman, D.; Krishnan, A.V.; Swami, S.; Giovannucci, E.; Feldman, B.J. The Role of Vitamin D in Reducing Cancer Risk and Progression. Nat. Rev. Cancer 2014, 14, 342–357. [Google Scholar] [CrossRef] [PubMed]
- Molin, A.; Baudoin, R.; Kaufmann, M.; Souberbielle, J.C.; Ryckewaert, A.; Vantyghem, M.C.; Eckart, P.; Bacchetta, J.; Deschenes, G.; Kesler-Roussey, G.; et al. CYP24A1 Mutations in a Cohort of Hypercalcemic Patients: Evidence for a Recessive Trait. J. Clin. Endocrinol. Metab. 2015, 100, E1343–E1352. [Google Scholar] [CrossRef]
- Nissen, J.; Rasmussen, L.B.; Ravn-Haren, G.; Andersen, E.W.; Hansen, B.; Andersen, R.; Mejborn, H.; Madsen, K.H.; Vogel, U. Common Variants in CYP2R1 and GC Genes Predict Vitamin D Concentrations in Healthy Danish Children and Adults. PloS ONE 2014, 9, e89907. [Google Scholar] [CrossRef]
- Barry, E.L.; Rees, J.R.; Peacock, J.L.; Mott, L.A.; Amos, C.I.; Bostick, R.M.; Figueiredo, J.C.; Ahnen, D.J.; Bresalier, R.S.; Burke, C.A.; et al. Genetic Variants in CYP2R1, CYP24A1, and VDR Modify the Efficacy of Vitamin D3 Supplementation for Increasing Serum 25-Hydroxyvitamin D Levels in a Randomized Controlled Trial. J. Clin. Endocrinol. Metab. 2014, 99, E2133–E2137. [Google Scholar] [CrossRef]
- Jiang, X.; O’Reilly, P.F.; Aschard, H.; Hsu, Y.-H.; Richards, J.B.; Dupuis, J.; Ingelsson, E.; Karasik, D.; Pilz, S.; Berry, D.; et al. Genome-Wide Association Study in 79,366 European-Ancestry Individuals Informs the Genetic Architecture of 25-Hydroxyvitamin D Levels. Nat. Commun. 2018, 9, 260. [Google Scholar] [CrossRef]
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global Cancer Statistics 2018: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA. Cancer J. Clin. 2018, 68, 394–424. [Google Scholar] [CrossRef]
- Deerwester, S.; Dumais, S.T.; Furnas, G.W.; Landauer, T.K.; Harshman, R. Indexing by Latent Semantic Analysis. J. Am. Soc. Inf. Sci. 1990, 41, 391–407. [Google Scholar] [CrossRef]
- Chen, C.; Ibekwe-SanJuan, F.; Hou, J. The Structure and Dynamics of Cocitation Clusters: A Multiple-Perspective Cocitation Analysis. J. Am. Soc. Inf. Sci. Technol. 2010, 61, 1386–1409. [Google Scholar] [CrossRef]
- Zheng, L. Using Mutual Information as a Cocitation Similarity Measure. Scientometrics 2019, 119, 1695–1713. [Google Scholar] [CrossRef]
- Abouzid, M.; Karaźniewicz-Łada, M.; Abdelazeem, B.; Brašić, J.R. Replication Data for: Research Trends of Vitamin D Metabolism Gene Polymorphisms Based on a Bibliometric Investigation. Harvard Dataverse, V1. 2022. Available online: https://dataverse.harvard.edu/dataset.xhtml?persistentId=doi:10.7910/DVN/BQGSYI (accessed on 6 January 2023).


| Paper | DOI | TC | TC Per Year | Normalized TC |
|---|---|---|---|---|
| [39] Salzman, J. et al., 2013, PLOS Genet. | 10.1371/journal.pgen.1003777 | 1299 | 129.90 | 27.38 |
| [40] Wang, T.J. et al., 2010, Lancet Lond. Engl. | 10.1016/S0140-6736(10)60588-0 | 1158 | 89.08 | 14.87 |
| [41] Ahn, J. et al., 2010, Hum. Mol. Genet. | 10.1093/hmg/ddq155 | 579 | 44.54 | 7.43 |
| [42] Nelson, E.R. et al., 2013, Science | 10.1126/science.1241908 | 499 | 49.90 | 10.52 |
| [43] Schauber, J. et al., 2007, J. Clin. Invest. | 10.1172/JCI30142 | 498 | 31.13 | 8.67 |
| [44] Cheng, J.B. et al. 2004, Proc. Natl. Acad. Sci. U.S.A. | 10.1073/pnas.0402490101 | 477 | 25.11 | 8.54 |
| [45] Schlingmann, K.P. et al., 2011, New Engl. J. Med. | 10.1056/NEJMoa1103864 | 382 | 31.83 | 8.02 |
| [46] Hewison, M., 2010, Endocrin. Metab. Clin. North Am. | 10.1016/j.ecl.2010.02.010 | 365 | 28.08 | 4.69 |
| [47] Motola, D.L. et al., 2006, Cell | 10.1016/j.cell.2006.01.037 | 352 | 20.71 | 6.66 |
| [48] Petta, S. et al., 2010, Hepatology | 10.1002/hep.23489 | 333 | 25.62 | 4.28 |
| ClusterID | Size | Silhouette | Top Terms (LSI) α | Top Terms (LLR, p-Level) β | Top Terms (MI) γ | Average Year |
|---|---|---|---|---|---|---|
| 4 | 63 | 0.922 | idiopathic infantile hypercalcemia | idiopathic infantile hypercalcemia (1176.6, 10−4) | α-oh derivative (1.2) | 2015 |
| 3 | 62 | 0.894 | genetic variant | african american (576.35, 10−4) | antenatal cholecalciferol supplementation (1.52) | 2010 |
| 1 | 53 | 0.862 | controlling vitamin | mendelian randomization study (538.14, 10−4) | α-oh derivative (1.24) | 2015 |
| 5 | 51 | 0.796 | colorectal cancer | gene polymorphism (426.57, 10−4) | upregulated cyp24a1 (1.94) | 2014 |
| 0 | 45 | 0.816 | 25-dihydroxyvitamin d-3 | mutational analysis (339.68, 10−4) | α-oh derivative (0.35) | 2004 |
| 2 | 45 | 0.836 | multiple sclerosis | multiple sclerosis (1174.9, 10−4) | altered vitamin (0.73) | 2009 |
| 7 | 42 | 0.956 | 20-hydroxyvitamin d3 | 20-hydroxyvitamin d3 (562.32, 10−4) | α-oh derivative (0.19) | 2011 |
| 8 | 33 | 0.982 | lemli-opitz syndrome | lemli-opitz syndrome (895.15, 10−4) | dhcr7-het mouse model (0.08) | 2006 |
| 6 | 30 | 0.894 | old theme | old theme (339.83, 10−4) | α-oh derivative (0.27) | 2005 |
| 10 | 24 | 0.992 | cerebrotendinous xanthomatosis | cerebrotendinous xanthomatosis (1407.51, 10−4) | early identification (0.1) | 2014 |
| 9 | 20 | 0.952 | cyp11a1-derived vitamin | cyp11a1-derived vitamin (119.56, 10−4) | vdr-associated lncrna (0.03) | 2018 |
| 12 | 13 | 0.988 | glial cholesterol synthesis | prescription medication (97.39, 10−4) | targeting 7-dehydrocholesterol reductase (0.01) | 2017 |
| 11 | 12 | 0.999 | intestinal caco-2 cell | thp-1 macrophage (67.29, 10−4) | multiple sclerosis (0.01) | 2002 |
| 13 | 8 | 0.998 | cancer development | emerging role (78.89, 10−4) | 2017 | |
| 14 | 6 | 0.997 | deferasirox pharmacokinetics | deferasirox pharmacokinetics (77.92, 10−4) | 2015 | |
| 15 | 6 | 1 | amyloid-β peptide production | amyloid-β peptide production (49.27, 10−4) | 2005 | |
| 17 | 5 | 0.998 | uranium | rat (47.47, 10−4) | 2002 | |
| 19 | 4 | 0.999 | cardiovascular disease | role (32.35, 10−4) | 2002 |
| Cluster No. | Keywords’ Counts | Focus |
|---|---|---|
| 1 | 31 | CYP27A1 and bile acid and lipid metabolism |
| 2 | 29 | CYP27B1 and vitamin D receptor and vitamin D metabolism |
| 3 | 24 | Vitamin D genetics and deficiency |
| 4 | 13 | Cytochrome P450, DHCR7, cholesterol, and Smith–Lemli–Opitz Syndrome |
| 5 | 11 | CYP24A1, hypercalcemia, vitamin D metabolism, and nephrocalcinosis |
| 6 | 6 | 25(OH)D3 and pregnancy |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abouzid, M.; Karaźniewicz-Łada, M.; Abdelazeem, B.; Brašić, J.R. Research Trends of Vitamin D Metabolism Gene Polymorphisms Based on a Bibliometric Investigation. Genes 2023, 14, 215. https://doi.org/10.3390/genes14010215
Abouzid M, Karaźniewicz-Łada M, Abdelazeem B, Brašić JR. Research Trends of Vitamin D Metabolism Gene Polymorphisms Based on a Bibliometric Investigation. Genes. 2023; 14(1):215. https://doi.org/10.3390/genes14010215
Chicago/Turabian StyleAbouzid, Mohamed, Marta Karaźniewicz-Łada, Basel Abdelazeem, and James Robert Brašić. 2023. "Research Trends of Vitamin D Metabolism Gene Polymorphisms Based on a Bibliometric Investigation" Genes 14, no. 1: 215. https://doi.org/10.3390/genes14010215
APA StyleAbouzid, M., Karaźniewicz-Łada, M., Abdelazeem, B., & Brašić, J. R. (2023). Research Trends of Vitamin D Metabolism Gene Polymorphisms Based on a Bibliometric Investigation. Genes, 14(1), 215. https://doi.org/10.3390/genes14010215