A COL5A2 In-Frame Deletion in a Chihuahua with Ehlers-Danlos Syndrome
Abstract
1. Introduction
2. Materials and Methods
2.1. Animal Selection
2.2. Clinical and Histopathological Examinations
2.3. DNA Extraction
2.4. Whole-Genome Sequencing and Variant Calling
2.5. Gene Analysis
2.6. PCR and Sanger Sequencing
3. Results
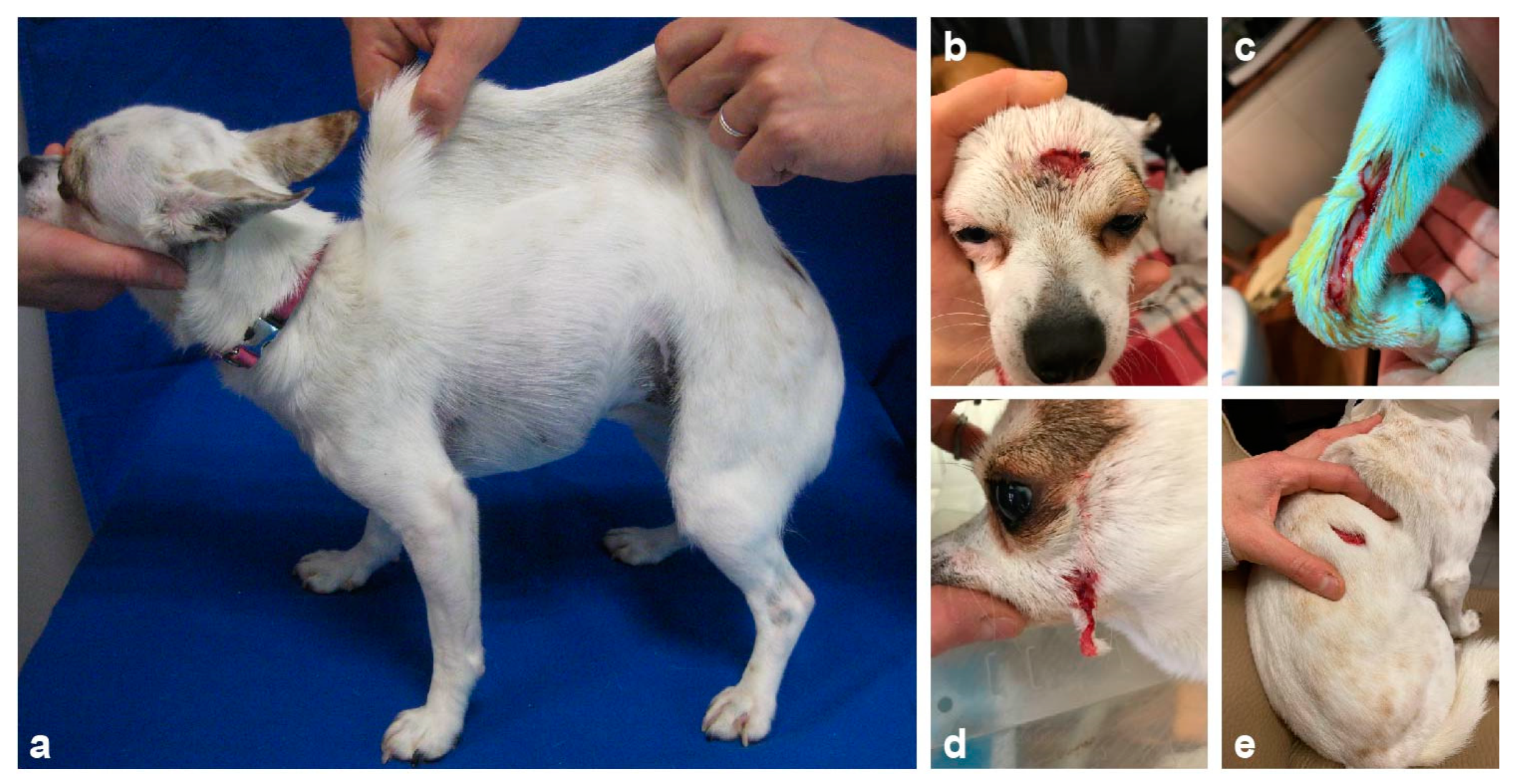
3.1. Clinical History and Examination
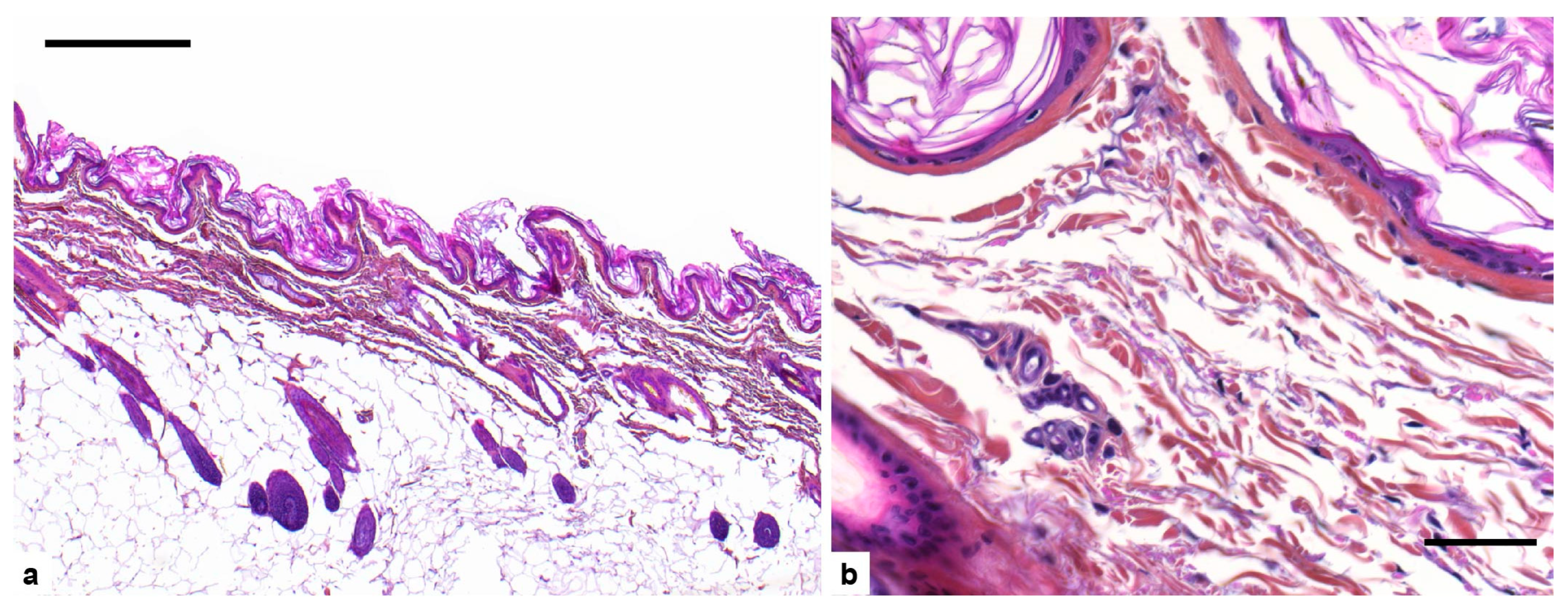
3.2. Histopathological Examination
3.3. Genetic Analysis
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Malfait, F.; Francomano, C.; Byers, P.; Belmont, J.; Berglund, B.; Black, J.; Bloom, L.; Bowen, J.M.; Brady, A.F.; Burrows, N.P.; et al. The 2017 international classification of the Ehlers-Danlos syndromes. Am. J. Med. Genet. Part. C Semin. Med. Genet. 2017, 175, 8–26. [Google Scholar] [CrossRef] [PubMed]
- Malfait, F.; Castori, M.; Francomano, C.A.; Giunta, C.; Kosho, T.; Byers, P.H. The Ehlers–Danlos syndromes. Nat. Rev. Dis. Prim. 2020, 6, 64. [Google Scholar] [CrossRef] [PubMed]
- Arlein, M.S. Generalized acute cutaneous asthenia in a dog. J. Am. Vet. Med. Assoc. 1947, 111, 52. [Google Scholar] [PubMed]
- Hegreberg, G.A.; Padgett, G.A.; Gorham, J.R.; Henson, J.B. A Connective Tissue Disease of Dogs and Mink Resembling the Ehlers-Danlos Syndrome of Man. J. Hered. 1969, 60, 249–254. [Google Scholar] [CrossRef]
- Hegreberg, G.A.; Padgett, G.A.; Ott, R.L.; Henson, J.B. A Heritable Connective Tissue Disease of Dogs and Mink Resembling Ehlers-Danlos Syndrome of Man I. Skin Tensile Strength Properties. J. Investig. Dermatol. 1970, 54, 377–380. [Google Scholar] [CrossRef]
- Bellini, M.H.; Caldini, E.T.E.G.; Scapinelli, M.P.; Simões, M.J.; Machado, D.B.; Nürmberg, R. Increased elastic microfibrils and thickening of fibroblastic nuclear lamina in canine cutaneous asthenia. Vet. Dermatol. 2009, 20, 139–143. [Google Scholar] [CrossRef]
- Uri, M.; Verin, R.; Ressel, L.; Buckley, L.; McEwan, N. Ehlers–Danlos Syndrome Associated with Fatal Spontaneous Vascular Rupture in a Dog. J. Comp. Pathol. 2015, 152, 211–216. [Google Scholar] [CrossRef]
- Rasch, S.N. Surgical and medical treatment of ocular disease in a dog with Ehlers-Danlos syndrome. Clin. Case Rep. 2017, 5, 880–886. [Google Scholar] [CrossRef]
- Ueda, K.; Kawai, T.; Senoo, H.; Shimizu, A.; Ishiko, A.; Nagata, M. Histopathological and electron microscopic study in dogs with patellar luxation and skin hyperextensibility. J. Vet. Med. Sci. 2018, 80, 1309–1316. [Google Scholar] [CrossRef]
- Hegreberg, G.A.; Padgett, G.A.; Henson, J.B. Connective tissue disease of dogs and mink resembling Ehlers-Danlos syndrome of man. 3. Histopathologic changes of the skin. Arch. Pathol. 1970, 90, 159–166. [Google Scholar]
- Freeman, L.J.; Hegreberg, G.A.; Robinette, J.D. Ehlers-Danlos syndrome in dogs and cats. Semin. Vet. Med. Surg. (Small Anim). 1987, 2, 221–227. [Google Scholar] [PubMed]
- Barnett, K.C.; Cottrell, B.D. Ehlers-Danlos syndrome in a dog: Ocular, cutaneous and articular abnormalities. J. Small Anim. Pract. 1987, 28, 941–946. [Google Scholar] [CrossRef]
- Matthews, B.R.; Lewis, G.T. Ehlers-Danlos syndrome in a dog. Can. Vet. J. = La Rev. Vet. Can. 1990, 31, 389–390. [Google Scholar]
- Rodríguez, F.; Herráez, P.; Monteros, A.E.; de los Calabuig, P.; Rodríguez, J.L. Collagen Dysplasia in a Litter of Garaffiano Shepherd Dogs. J. Vet. Med. Ser. A 1996, 43, 509–512. [Google Scholar] [CrossRef]
- Jelínek, F.; Karban, J. Cutaneous Asthenia in One Dog. Acta Vet. Brno 1998, 67, 109–113. [Google Scholar] [CrossRef][Green Version]
- Paciello, O.; Lamagna, F.; Lamagna, B.; Papparella, S. Ehlers-Danlos-like syndrome in 2 dogs: Clinical, histologic, and ultrastructural findings. Vet. Clin. Pathol. 2003, 32, 13–18. [Google Scholar] [CrossRef]
- Barrera, R.; Mañe, C.; Duran, E.; Vives, M.A.; Zaragoza, C. Ehlers-Danlos syndrome in a dog. Can. Vet. J. = La Rev. Vet. Can. 2004, 45, 355–356. [Google Scholar]
- Jaffey, J.A.; Bullock, G.; Teplin, E.; Guo, J.; Villani, N.A.; Mhlanga-Mutangadura, T.; Schnabel, R.D.; Cohn, L.A.; Johnson, G.S. A homozygous ADAMTS 2 nonsense mutation in a Doberman Pinscher dog with Ehlers Danlos syndrome and extreme skin fragility. Anim. Genet. 2019, 50, 543–545. [Google Scholar] [CrossRef]
- Colige, A.; Sieron, A.L.; Li, S.-W.; Schwarze, U.; Petty, E.; Wertelecki, W.; Wilcox, W.; Krakow, D.; Cohn, D.H.; Reardon, W.; et al. Human Ehlers-Danlos Syndrome Type VII C and Bovine Dermatosparaxis Are Caused by Mutations in the Procollagen I N-Proteinase Gene. Am. J. Hum. Genet. 1999, 65, 308–317. [Google Scholar] [CrossRef]
- Bauer, A.; de Lucia, M.; Leuthard, F.; Jagannathan, V.; Leeb, T. Compound heterozygosity for TNXB genetic variants in a mixed-breed dog with Ehlers-Danlos syndrome. Anim. Genet. 2019, 50, 546–549. [Google Scholar] [CrossRef]
- Bristow, J.; Tee, M.; Gitelman, S.; Mellon, S.; Miller, W. Tenascin-X: A novel extracellular matrix protein encoded by the human XB gene overlapping P450c21B. J. Cell Biol. 1993, 122, 265–278. [Google Scholar] [CrossRef] [PubMed]
- Burch, G.H.; Gong, Y.; Liu, W.; Dettman, R.W.; Curry, C.J.; Smith, L.; Miller, W.L.; Bristow, J. Tenascin–X deficiency is associated with Ehlers–Danlos syndrome. Nat. Genet. 1997, 17, 104–108. [Google Scholar] [CrossRef] [PubMed]
- Mao, J.R.; Taylor, G.; Dean, W.B.; Wagner, D.R.; Afzal, V.; Lotz, J.C.; Rubin, E.M.; Bristow, J. Tenascin-X deficiency mimics Ehlers-Danlos syndrome in mice through alteration of collagen deposition. Nat. Genet. 2002, 30, 421–425. [Google Scholar] [CrossRef] [PubMed]
- Bauer, A.; Bateman, J.F.; Lamandé, S.R.; Hanssen, E.; Kirejczyk, S.G.M.; Yee, M.; Ramiche, A.; Jagannathan, V.; Welle, M.; Leeb, T.; et al. Identification of Two Independent COL5A1 Variants in Dogs with Ehlers–Danlos Syndrome. Genes 2019, 10, 731. [Google Scholar] [CrossRef]
- Birk, D.E. Type V collagen: Heterotypic type I/V collagen interactions in the regulation of fibril assembly. Micron 2001, 32, 223–237. [Google Scholar] [CrossRef]
- Jagannathan, V.; Drögemüller, C.; Leeb, T.; Aguirre, G.; André, C.; Bannasch, D.; Becker, D.; Davis, B.; Ekenstedt, K.; Faller, K.; et al. A comprehensive biomedical variant catalogue based on whole genome sequences of 582 dogs and eight wolves. Anim. Genet. 2019, 50, 695–704. [Google Scholar] [CrossRef]
- Kiener, S.; Wiener, D.J.; Hopke, K.; Diesel, A.B.; Jagannathan, V.; Mauldin, E.A.; Casal, M.L.; Leeb, T. ABHD5 frameshift deletion in Golden Retrievers with ichthyosis. G3 (Bethesda) 2022, 12, jkab397. [Google Scholar] [CrossRef]
- Jacinto, J.G.P.; Häfliger, I.M.; Veiga, I.M.B.; Letko, A.; Benazzi, C.; Bolcato, M.; Drögemüller, C. A Heterozygous Missense Variant in the COL5A2 in Holstein Cattle Resembling the Classical Ehlers–Danlos Syndrome. Animals 2020, 10, 2002. [Google Scholar] [CrossRef]
- Gross, T.L.; Ihrke, J.P.; Walder, E.J.; Affolter, V.K. Degenerative, Dysplastic and Depositional Diseases of Dermal Connective Tissue. In Skin Diseases of the Dog and Cat: Clinical and Histopathologic Diagnosis; Blackwell Science Ltd.: Oxford, UK, 2005; pp. 386–389. [Google Scholar]
- Burgeson, R.E.; El Adli, F.A.; Kaitila, I.I.; Hollister, D.W. Fetal membrane collagens: Identification of two new collagen alpha chains. Proc. Natl. Acad. Sci. USA 1976, 73, 2579–2583. [Google Scholar] [CrossRef]
- Gay, S.; Kent Rhodes, R.; Gay, R.E.; Miller, E.J. Collagen molecules comprised of alpha 1(V)-chains (B-chains): An apparent localization in the exocytoskeleton. Coll. Relat. Res. 1981, 1, 53–58. [Google Scholar] [CrossRef]
- Imamura, Y.; Scott, I.C.; Greenspan, D.S. The pro-alpha3(V) collagen chain. Complete primary structure, expression domains in adult and developing tissues, and comparison to the structures and expression domains of the other types V and XI procollagen chains. J. Biol. Chem. 2000, 275, 8749–8759. [Google Scholar] [CrossRef] [PubMed]
- Wenstrup, R.J.; Florer, J.B.; Davidson, J.M.; Phillips, C.L.; Pfeiffer, B.J.; Menezes, D.W.; Chervoneva, I.; Birk, D.E. Murine model of the Ehlers-Danlos syndrome. col5a1 haploinsufficiency disrupts collagen fibril assembly at multiple stages. J. Biol. Chem. 2006, 281, 12888–12895. [Google Scholar] [CrossRef] [PubMed]
- Chanut-Delalande, H.; Bonod-Bidaud, C.; Cogne, S.; Malbouyres, M.; Ramirez, F.; Fichard, A.; Ruggiero, F. Development of a Functional Skin Matrix Requires Deposition of Collagen V Heterotrimers. Mol. Cell. Biol. 2004, 24, 6049. [Google Scholar] [CrossRef] [PubMed]
- Symoens, S.; Syx, D.; Malfait, F.; Callewaert, B.; De Backer, J.; Vanakker, O.; Coucke, P.; De Paepe, A. Comprehensive molecular analysis demonstrates type V collagen mutations in over 90% of patients with classic EDS and allows to refine diagnostic criteria. Hum. Mutat. 2012, 33, 1485–1493. [Google Scholar] [CrossRef]
| Gene | EDS Type | Inheritance |
|---|---|---|
| ADAMTS2 | Dermatosparaxis | AR |
| AEBP1 | Classical-like type 2 | AR |
| B3GALT6 | Spondylo-dysplastic | AR |
| B4GALT7 | Spondylo-dysplastic | AD |
| CHST14 | Musculocontractural | AR |
| COL1A1 | Classical, Vascular, Artrochalasia | AD |
| COL1A2 | Artrochalasia, Cardiac valvular | AD |
| COL3A1 | Vascular | AD |
| COL5A1 | Classical | AD |
| COL5A2 | Classical | AD |
| COL12A1 | Myopathic | AD |
| C1R | Periodontal | AD |
| C1S | Periodontal | AD |
| DSE | Musculocontractural | AR |
| FKBP14 | Kyphoscoliotic | AR |
| PRDM5 | Brittle cornea syndrome | AR |
| PLOD1 | Kyphoscoliotic | AR |
| SLC39A13 | Spondylo-dysplastic | AR |
| TNXB | Classical-like | AR |
| ZNF469 | Brittle cornea syndrome | AR |
| Filtering Step | Heterozygous Variants | Homozygous Variants |
|---|---|---|
| All variants in the affected dog | 4,191,411 | 2,457,482 |
| Private variants | 18,956 | 1258 |
| Protein-changing private variants | 104 | 2 |
| Protein-changing private variants in 20 candidate genes | 1 | 0 |
| Major criteria | 1. Skin hyperextensibility and atrophic scarring |
| 2. Generalized joint hypermobility | |
| Minor criteria | 1. Easy bruising |
| 2. Soft, doughy skin | |
| 3. Skin fragility (or traumatic splitting) | |
| 4. Molluscoid pseudotumors | |
| 5. Subcutaneous spheroids | |
| 6. Hernia (or history thereof) | |
| 7. Epicanthal folds | |
| 8. Complication of joint hypermobility (e.g., sprains, luxation/subluxation, pain, flexible flatfoot) | |
| 9. Family history of a first degree relative who meets clinical criteria |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kiener, S.; Chevallier, L.; Jagannathan, V.; Briand, A.; Cochet-Faivre, N.; Reyes-Gomez, E.; Leeb, T. A COL5A2 In-Frame Deletion in a Chihuahua with Ehlers-Danlos Syndrome. Genes 2022, 13, 934. https://doi.org/10.3390/genes13050934
Kiener S, Chevallier L, Jagannathan V, Briand A, Cochet-Faivre N, Reyes-Gomez E, Leeb T. A COL5A2 In-Frame Deletion in a Chihuahua with Ehlers-Danlos Syndrome. Genes. 2022; 13(5):934. https://doi.org/10.3390/genes13050934
Chicago/Turabian StyleKiener, Sarah, Lucie Chevallier, Vidhya Jagannathan, Amaury Briand, Noëlle Cochet-Faivre, Edouard Reyes-Gomez, and Tosso Leeb. 2022. "A COL5A2 In-Frame Deletion in a Chihuahua with Ehlers-Danlos Syndrome" Genes 13, no. 5: 934. https://doi.org/10.3390/genes13050934
APA StyleKiener, S., Chevallier, L., Jagannathan, V., Briand, A., Cochet-Faivre, N., Reyes-Gomez, E., & Leeb, T. (2022). A COL5A2 In-Frame Deletion in a Chihuahua with Ehlers-Danlos Syndrome. Genes, 13(5), 934. https://doi.org/10.3390/genes13050934








