Abstract
Impulsivity has been proposed as an endophenotype for bipolar disorder (BD); moreover, impulsivity levels have been shown to carry prognostic significance and to be quality-of-life predictors. To date, reports about the genetic determinants of impulsivity in mood disorders are limited, with no studies on BD individuals. Individuals with BD and healthy controls (HC) were recruited in the context of an observational, multisite study (GECOBIP). Subjects were genotyped for three candidate single-nucleotide polymorphisms (SNPs) (5-HTTLPR, COMT rs4680, BDNF rs6265); impulsivity was measured through the Italian version of the Barratt Impulsiveness Scale (BIS-11). A mixed-effects regression model was built, with BIS scores as dependent variables, genotypes of the three polymorphisms as fixed effects, and centers of enrollment as random effect. Compared to HC, scores for all BIS factors were higher among subjects with euthymic BD (adjusted β for Total BIS score: 5.35, p < 0.001). No significant interaction effect was evident between disease status (HC vs. BD) and SNP status for any polymorphism. Considering the whole sample, BDNF Met/Met homozygosis was associated with lower BIS scores across all three factors (adjusted β for Total BIS score: −10.2, p < 0.001). A significant 5-HTTLPR x gender interaction was found for the SS genotype, associated with higher BIS scores in females only (adjusted β for Total BIS score: 12.0, p = 0.001). Finally, COMT polymorphism status was not significantly associated with BIS scores. In conclusion, BD diagnosis did not influence the effect on impulsivity scores for any of the three SNPs considered. Only one SNP—the BDNF rs6265 Met/Met homozygosis—was independently associated with lower impulsivity scores. The 5-HTTLPR SS genotype was associated with higher impulsivity scores in females only. Further studies adopting genome-wide screening in larger samples are needed to define the genetic basis of impulsivity in BD.
1. Introduction
Bipolar disorder (BD) is a severe, chronic, and common mental illness, characterized by episodes of elevated mood (hypomania or mania) alternating with depressive episodes [1]. BD is listed among the top causes of years lost due to disabilities [2] and is associated with a lifetime risk of suicide 20–30 times higher than the general population [3]. While mood changes represent the defining feature of the disease, individuals with BD display various mood-independent trait patterns encompassing temperament, character, and personality [4,5,6,7,8,9].
Impulsivity is one psychological dimension that has been shown to be significantly represented in individuals with BD [10,11,12], even during a phase of euthymia [13,14]; moreover, trait impulsivity is increased in unaffected relatives of individuals with BD [15] and in high-risk subjects [16]. As such, impulsivity has been proposed as an endophenotype for BD [17]. Impulsivity is defined as “a predisposition towards rapid, unplanned reactions to internal or external stimuli without regard to the negative consequences of these reactions to the impulsive individual or others” [18] and in BD individuals is associated with at-risk behaviors, such as suicidality, substance abuse and criminal actions [11,19]; it is also a predictor of quality of life [20], disease onset [21] and illness severity [13].
Importantly, impulsivity has a significant genetic background [22] and similar to other complex phenotypes, a great number of genetic variants seem to influence this trait, with small individual effects [23,24]. This may stem from impulsivity being a complex, multidimensional trait, which can be measured through a multitude of instruments (i.e., neuropsychological paradigms, other- or self-administered questionnaires), or through other surrogate measures (i.e., violent, aggressive, or suicidal behavior). Since impulsivity is considered a candidate endophenotype for BD, it may be hypothesized that individuals with BD carry an increased genetic load of impulsivity. However, to date, studies that explored this hypothesis are limited to single candidate genes [25,26,27].
The purpose of the present study is to elucidate the genetic basis of impulsivity in individuals with BD. In doing so, we focused on three single-nucleotide polymorphisms (SNPs)—COMT rs4680, 5-HTTLPR (SLC6A4) and BDNF rs6265. The choice of these three candidate genes was motivated by the following reasons: (1) all three SNPs have been associated with increased susceptibility for BD [28,29,30]; (2) they have been consistently shown to influence behavioral traits in both healthy subjects and individuals with psychiatric disorders (see below for more details). Indeed, the influence of these three SNPs on temperament and character dimensions in both HC and BD patients was investigated by our consortium (GECOBIP) and reported in a separate paper [31]. (3) Finally, all three SNPs have been shown to influence impulsivity measures in samples of HC and neuropsychiatric populations (see below for more details).
The COMT gene encodes for an enzyme involved in the regulation of dopamine levels, responsible for about 50% of dopamine clearance in the prefrontal cortex [32]. A common COMT polymorphism is a valine to methionine substitution at codon 158 (rs4680) [33] that is associated with markedly reduced enzyme activity [34]. COMT polymorphisms have been reported to influence temperament, personality, and cognition [35,36,37,38].
Several studies report an association between the Met substitution and increased impulsivity in healthy individuals [39] and among various neuropsychiatric disorders, including attention deficit hyperactivity disorder (ADHD) [40], binge-eating disorder [41] and borderline personality disorder [42]. To our knowledge, to date no studies on the effect of COMT polymorphism on impulsivity in BD are available.
The SLC6A4 gene encodes for the serotonin transporter (SERT or 5-HTT) protein [43] responsible for the uptake of serotonin from the synaptic cleft [44]. The 5-HTT gene-linked polymorphic region (5-HTTLPR) is a widely studied polymorphism that involves the transcriptional control region upstream of the SLC6A4 coding sequence [45]. The long and short variants of this region are associated with higher and lower transcriptional activity, respectively [45]. The polymorphism has been found to influence affective temperament [46], and to be associated with higher levels of negative emotionality [47], neuroticism [48], rumination [49], and hostility [50]. Of note, surrogate measures of impulsivity, such as response inhibition, gambling, or attentional tasks, have been shown to be heterogeneously influenced by the S allele in samples of healthy individuals [51,52]. Regarding the relationship between the 5-HTTLPR polymorphism and impulsivity in individuals with BD, increased ADHD-related impulsivity symptoms during childhood were found in a group of individuals with bipolar disorder type 2 [53]. Conversely, no association between HTTPLR status and impulsivity was found in a group of BD individuals with a history of suicide attempt [26].
The BDNF gene encodes for a member of the neurotrophin family, a class of proteins involved in the development, survival, and synaptic plasticity of neurons [54]. A valine to methionine substitution at codon 66 (rs6265) is the product of a common polymorphism in the 5′ pro-BDNF region. This substitution was shown to impair intracellular processing and secretion of the mature protein [55]. This SNP has been associated with several traits, such as sensation seeking [56] and harm avoidance [57]. The polymorphism has also been suggested to modulate various cognitive abilities in both healthy individuals and psychiatric populations, with Met substitution generally associated with worse cognitive performances [58,59,60]. The influence of the BDNF genotype on impulsivity-related domains appears not well defined, with conflicting results regarding risk of suicidal behavior [61,62,63] and substance abuse/dependence [64,65,66]. We found no reports on the effect of this SNP on impulsivity in samples of BD individuals.
In conclusion, the evidence on the genetic basis of impulsivity in individuals with BD is extremely limited. The present study aims to address this topic by investigating the effect of three functional genetic variants (5-HTTLPR, COMT rs4680, and BDNF rs6265) on impulsivity, as measured through the Barratt Impulsiveness Scale (BIS-11), in a sample of healthy controls (HC) and BD patients.
2. Materials and Methods
2.1. Study Sample
Subjects were recruited from 8 Italian clinical sites part of the GECOBIP consortium (Table 1). GECOPIB was built with the aim to explore the association between three SNPs and neuropsychological and clinical characteristics of individuals with BD.

Table 1.
Main characteristics of the subjects by case-control status variable.
Exclusion criteria for both HC and BD included: (1) age ≤18 years, (2) intelligence quotient ≤70, (3) medical or neurological comorbidities, (4) not being an Italian native speaker. Additional exclusion criteria for HC were a diagnosis of cognitive impairment or an history of alcohol and/or drug abuse. BD diagnosis had to be confirmed through the Structured Clinical Interview for DSM-IV TR axis I (SCID-I); individuals with BD diagnosed with any other axis I psychiatric comorbidity were excluded.
The study was approved by the local Ethics Committee and conducted according to the ethical principles stated in the Declaration of Helsinki.
2.2. Assessment Tools
Individuals were assessed for socioeconomical, neuropsychological and clinical characteristics, as reported in Porcelli et al. [31]
In the present study, we focused on the influence of selected genetic variants on the temperamental features of the sample, and in particular on the impulsiveness as measured by the Barratt Impulsivity Scale [67], Italian version [68]. BIS-11 is a self-report questionnaire composed of 30 items; every single item is answered on a 4-point scale (Rarely/Never, Occasionally, Often, Almost Always/Always). Each item contributes to define six first-order factors, which combined produce the following three second-order factors: “Attentional Impulsiveness”, “Motor Impulsiveness” and “Non-Planning Impulsiveness”.
2.3. Genotype Analysis
Three functional genetic variants within key candidate genes (COMT, BDNF, and SLC6A4) were genotyped in each individual recruited: the 5-HTTLPR variants within the SLC6A4 gene, the rs4680 within the COMT gene, and the rs6265 within the BDNF gene. Genomic DNA was collected from a saliva sample according to standard procedures (Oragene DNA Self-Collection Kit [69]). The COMT rs4680, the BDNF rs6265, and the 5-HTTLPR polymorphisms were analyzed by real-time polymerase chain reaction allelic discrimination assay, using predesigned TaqMan genotyping assays and standard assay conditions. Personnel performing the genetic analysis were blind to the socioeconomical, neuropsychological and clinical characteristics of the subjects.
2.4. Statistical Analysis
Data were summarized with frequency tables and figures. Between-group differences in the distribution of categorical and continuous variables were tested by means of Chi-squared test and Student’s T test, respectively. Since marginal BIS score showed a non-normal distribution (Figure S1—Supplementary Material), comparisons between HC and BD and among gene polymorphisms were performed by means of Mann–Whitney (MW) test, or Kruskal–Wallis (KW) test if the strata were more than two.
A series of mixed-effects regression models were estimated, with BIS scores (Attentional, Motor, Non-Planning, Total Score) as dependent variables; disease status (HC or BD) and genotypes of the three polymorphisms (COMT rs4680, BDNF rs6265, 5-HTTLPR) were included as fixed effects. The estimated models considered the presence of a random effect (random intercept) represented by the center of enrollment. Results were presented by coefficients (β) adjusted for a series of confounders: age class (3 tertiles: (18–30.7), (30.7–47) and (47–73) years), gender, Raven IQ score (3 tertiles: (66–114), (114–127), and (127–128) points), educational level (3 tertiles: (5–13), (13–18), and (18–26) years). Interactions between gene polymorphisms and covariates were tested and included minimizing the Akaike’s Information Criterion (AIC) index.
The normality of the regression residuals was verified to evaluate the presence of outliers and deviations from the normal distribution. The observed power analysis and the replication attempt was assessed by means of a Retrospective Design Analysis (RDA) [70]. RDA was applied on the predicted BIS scores of the estimated mixed-effects regression models; this allowed to calculate, for each model and polymorphisms, the retrospective power of the highest observed effect size (Cohen d) among all possible group combinations, and the relative Type M and Type S errors.
The significance was set at 5%. All statistical analysis was performed using R Statistical Software; packages lme4 and lmerTest were used for the estimation of the mixed-effects regression model, while the PRDA package was adopted for the RDA [71].
3. Results
3.1. Characteristics of the Sample
Overall, 228 subjects were included in the analysis (132 HC, 96 BD). Three subjects with overall BIS score less than 35 were excluded; 225 subjects were considered for the subsequent analysis (131 HC, 94 BD). The sociodemographic characteristics and the results of the psychometric assessments are presented in Table 1. In general, individuals with BD were older (mean (standard deviation)—HC: 35.5 years (12.9); BD 45.7 (11.7); T-test p < 0.001), less educated (HC: 16.9 schooling years (3.8); BD: 12.7 (4.0); T-test p < 0.001), and had a lower socioeconomic status as measured by the Barratt Simplified Measure of Social Status (BSMSS) (HC: 44.5 (12.9); BD: 31.2 (15.7); T-test p < 0.001). The BD group performed significantly worse on the Raven IQ test (HC: 122.7 (7.9); BD: 112.5 (12.4); T-test p < 0.001).
3.2. Impulsivity Scores
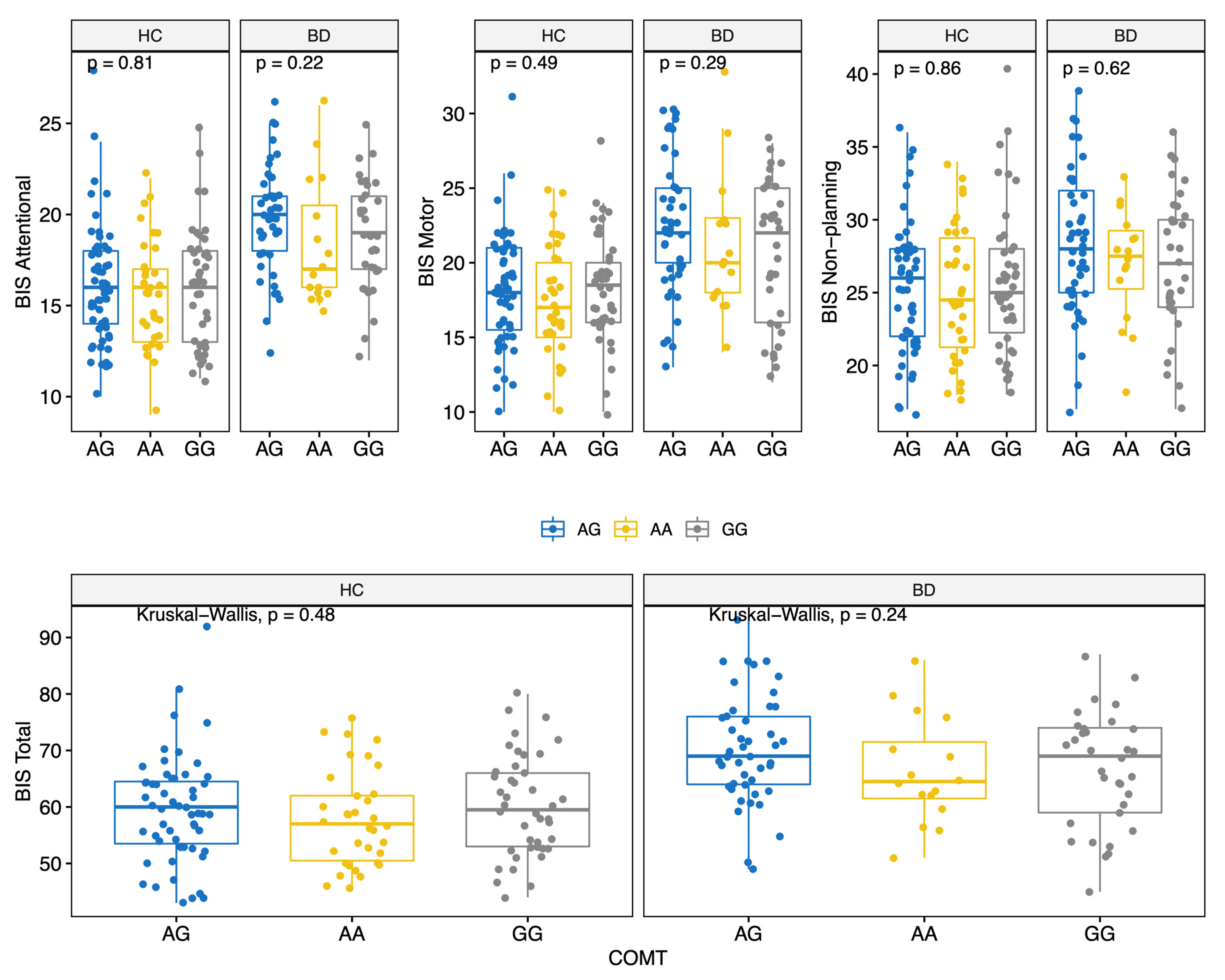
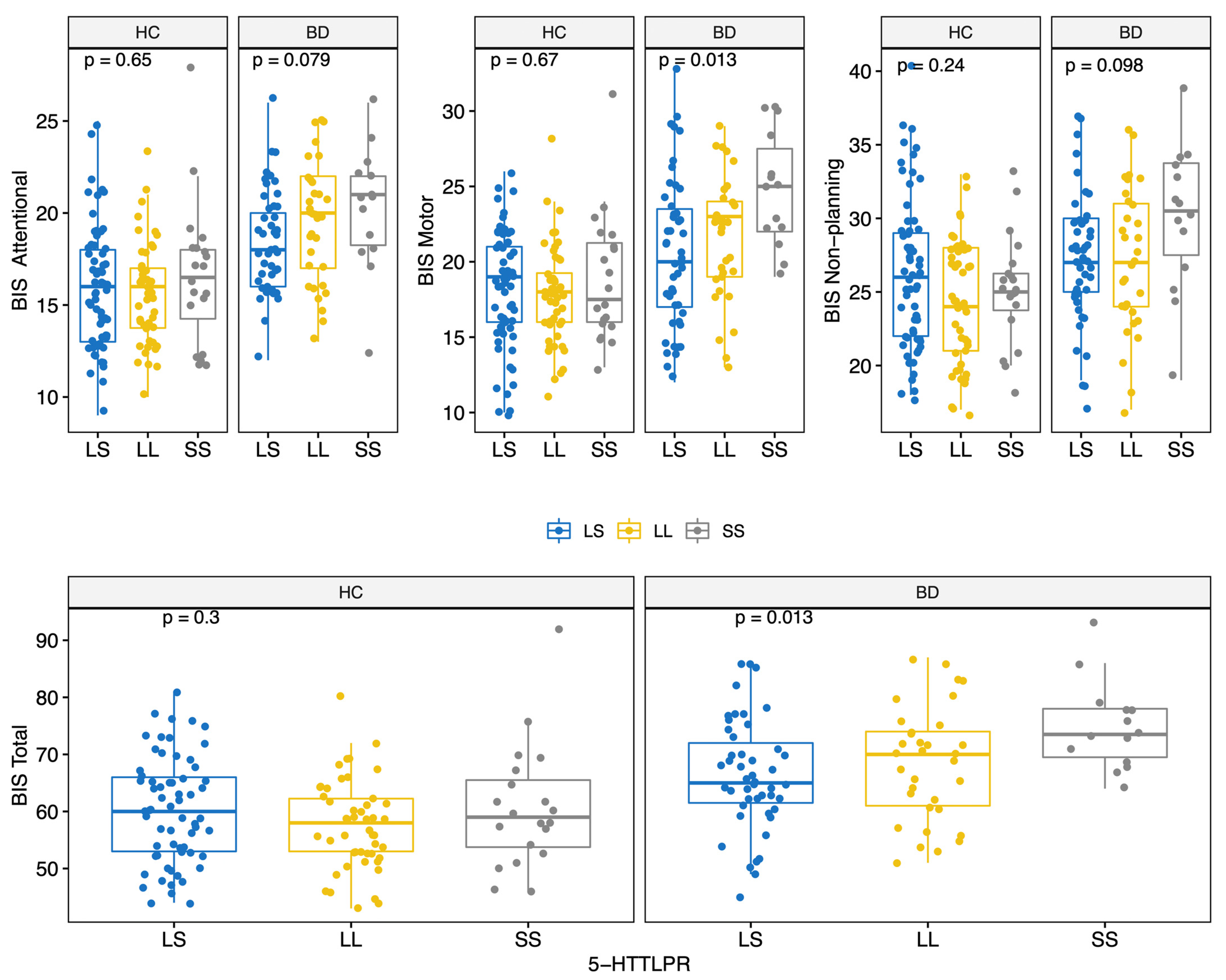
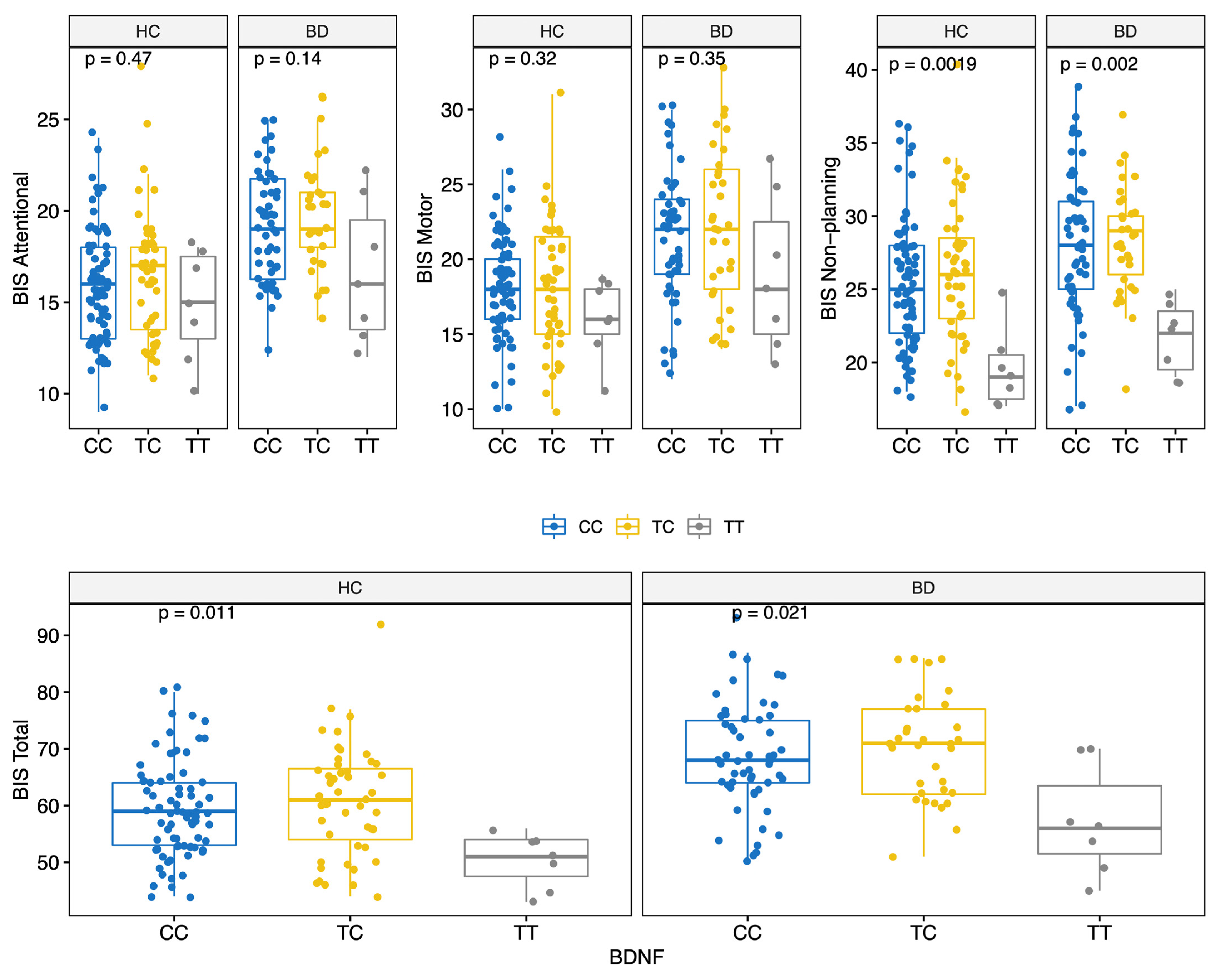
BD scored higher on all BIS-11 factors (Attentional, Motor, and Non-Planning) and overall score (HC: 59.0 (12.0); BD: 68.5 (12.8); MW p < 0.001) (Table 2 and Figure S1). The prevalence of the genotypes for the three polymorphisms studied did not differ significantly between the two groups (Table 2). Moreover, allelic and genotypic frequencies were consistent with those reported for the Caucasian ethnicity [72,73,74]. Of note, only 14 subjects (6.2%) carried the BDNF TT genotype. There were no significant differences in BIS scores between different COMT genotypes (Figure 1); conversely, significant differences in BIS scores were found for the 5-HTTLPR and BDNF polymorphisms. Specifically, BIS Motor factor and overall scores were lower for BD individuals carrying the 5-HTTLPR L allele (genotypes LL or LS) (KS test < 0.05) (Figure 2). Among both HC and BD subjects, BDNF TT homozygosis was associated with lower Non-Planning and overall BIS scores (KS test p < 0.05; Figure 3).

Table 2.
BIS-11 scores and polymorphism distribution by case-control status variable.
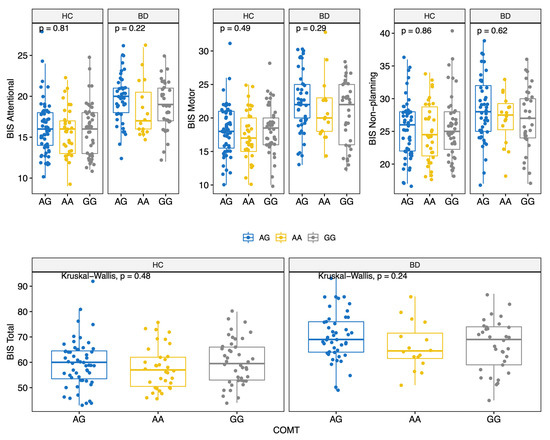
Figure 1.
BIS-11 score (Attentional, Motor, Non-Planning and Total) by COMT polymorphisms for HC and BD.
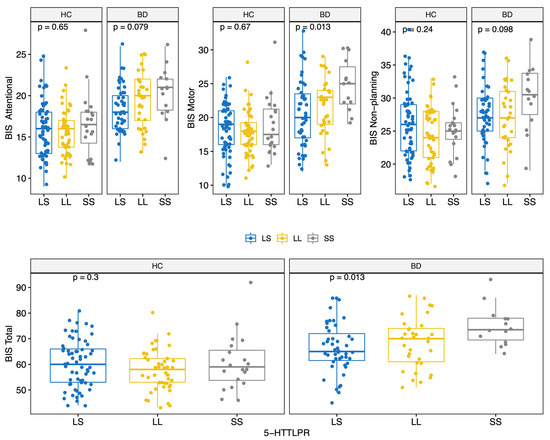
Figure 2.
BIS-11 score (Attentional, Motor, Non-Planning and Total) by 5-HTTLPR polymorphisms for HC and BD.
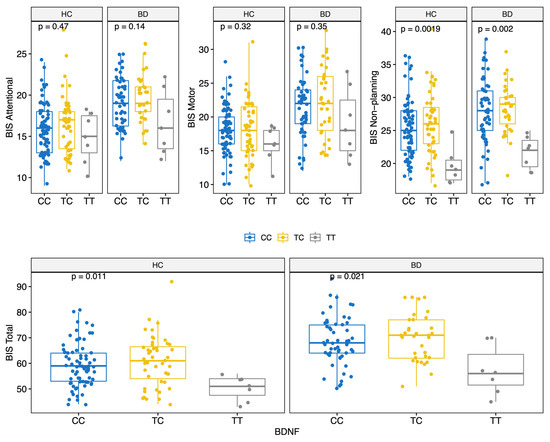
Figure 3.
BIS score (Attentional, Motor, Non-Planning and Total) by BDNF polymorphisms for HC and BD.
3.3. Mixed-Effects Regression Model
A total of four regression models were estimated. The β estimates for the BIS Total score are shown in Table 3; the results of the mixed-effect regression models for each BIS factor are reported in Table S1 (Supplementary Material). The fitted models explained only a discrete quote of variance (marginal R2 comprised between 23% and 33.1%). The quantile-quantile diagram reported a good adaption of the regression residuals to the normal distribution, with some deviation only for the Attentional BIS dimension (Figure S2—Supplementary Material).

Table 3.
Coefficients (β) estimated by the mixed-effects regression model considering the Total BIS score as dependent variable.
3.3.1. Main Effects
Total BIS scores were significantly influenced by gender, with female sex associated with lower BIS scores; there was no association between BIS scores and age. Higher educational level, but not higher Raven IQ, was associated with lower Total BIS scores. BD diagnosis was associated with higher impulsivity scores across all BIS factors.
The BDNF rs6265 Met/Met homozygosis was the genotype most consistently associated with lower impulsivity levels, being associated with both lower Total BIS scores and lower scores across each BIS factor when compared to the Val/Val genotype. BIS scores did not differ between the Val/Val homozygosis and the Val/Met heterozygosis. Regarding the 5-HTTLPR polymorphism, LL homozygosis was associated with lower BIS scores in the Non-Planning factor if compared to the LS heterozygosis, but not with lower Total BIS scores. The SS genotype had no effect on impulsivity scores. Finally, COMT rs4680 genotype status did not influence any BIS scores.
3.3.2. Interaction Effects
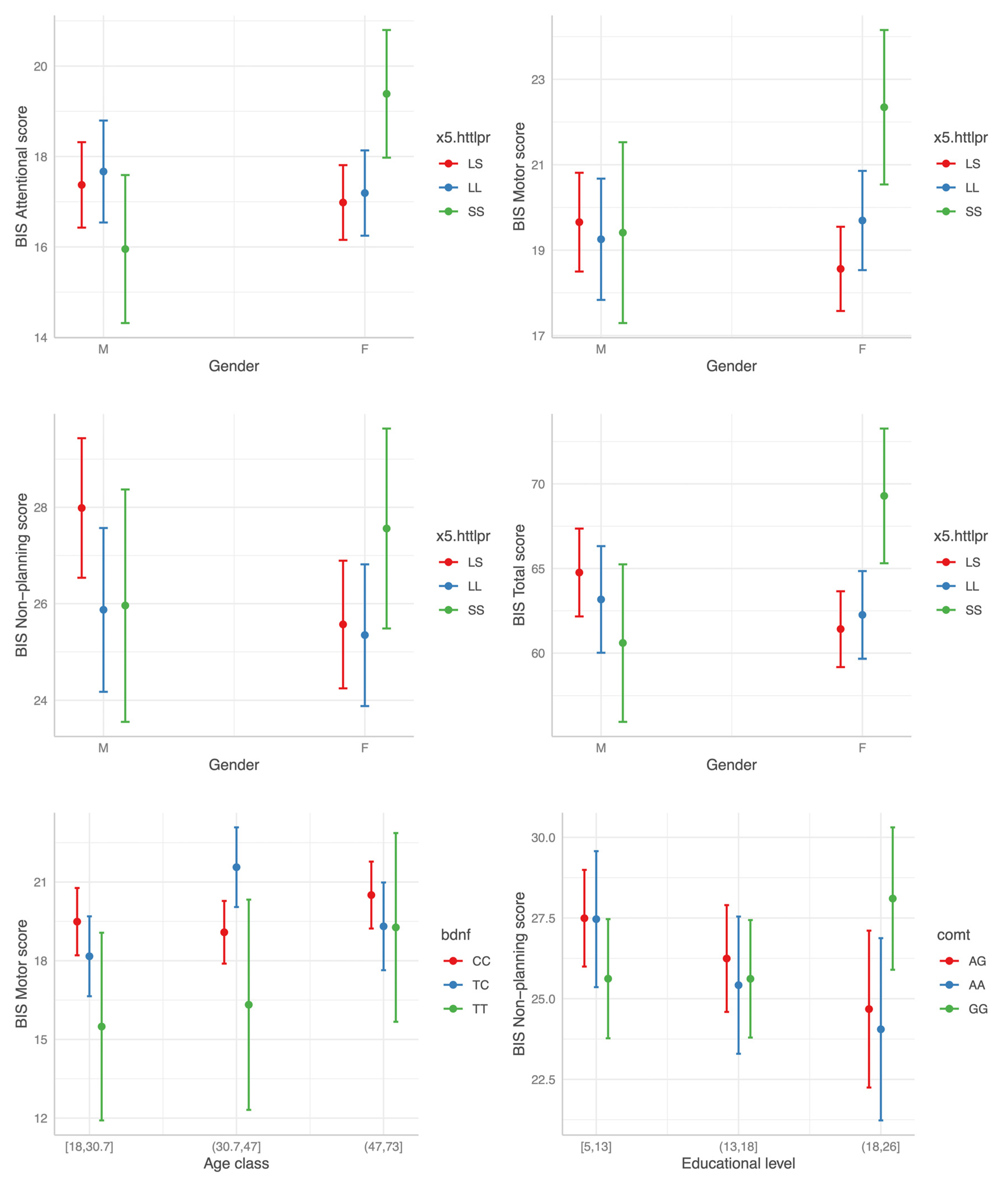
Of note, no significant interaction effect on impulsivity levels was evident between disease status (HC vs. BD) and any polymorphism status, apparently implying no differences in the modulatory effect of the three SNPs on impulsivity between HC and BD individuals. The most consistent interaction found was that between the 5-HTTLPR SS genotype status and gender. In females the SS homozygosis was associated with higher BIS scores across all the three BIS factors when compared to the LS heterozygosis (Figure 4). Of note, no main effect of the SS genotype was evident. Two other significant interactions were found: higher Motor BIS score for the combination BDNF TC and age class (31.7–47) and elevated BIS Non-Planning scores for high education levels with GG genotype in COMT. However, in both cases the effect was limited to only one BIS factor and did not influence Total BIS scores (Figure 4; Table 3).
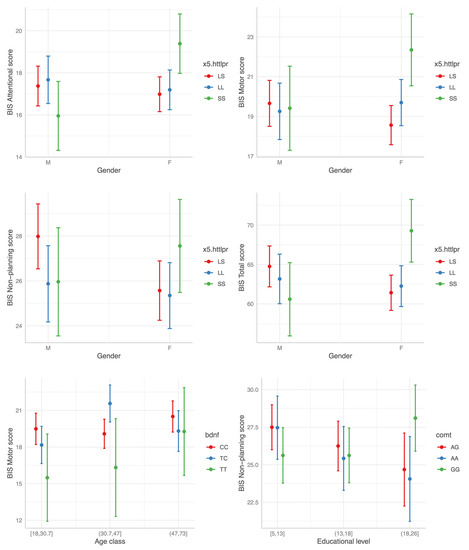
Figure 4.
The interaction effect between polymorphism status and gender, age and educational level (from top left to bottom right) on BIS scores.
No other significant interaction effect was found.
3.4. Retrospective Design Analysis
The results of the RDA on the maximum observed effects size between pairwise comparisons are reported in the (Supplementary Materials section Table S2). Overall, Type S error was small, indicating a high probability of a correct direction of the measured differences. Despite being significant, Type M error was generally small for BIS factor and overall scores for the interaction between the 5-HTTLPR polymorphism and gender. Type M error values were higher for the three BIS factor scores for both BDNF and COMT polymorphisms. In summary, the RDA confirmed that all the observed results were supported by an adequate power and a low Type M error.
4. Discussion
The main findings of the study in our sample of euthymic BD individuals and HC can be summarized as follows:
- (1)
- In our sample of euthymic BD individuals, BD diagnosis was associated with higher impulsivity scores across all BIS factors when compared to HC; this finding confirms earlier reports [75], ultimately suggesting that impulsivity may represent a state-independent trait of the disease.
- (2)
- The effect of the interaction between BD diagnosis and polymorphism status on impulsivity levels was not significant for any of the three polymorphisms considered.
- (3)
- In the whole sample (HC+BD), BDNF Met/Met homozygosis (but not Val/Met heterozygosis) was associated with lower BIS scores when compared to the wild-type genotype (Val/Val).
- (4)
- A genotype × gender interaction was evident for the 5-HTTLPR polymorphism in females, with the SS genotype (but not the LS heterozygosis) being associated with higher BIS scores. Conversely, no main effect was evident for the SS homozygosis.
- (5)
- No significant association was found between COMT genotype status and impulsivity levels.
4.1. No Differences in the Effect of SNPs on Impulsivity between HC and BD
One of the primary goals of our study was to determine whether the three polymorphisms differed in the two samples in terms of allele frequencies or effect on impulsivity levels. This is a relevant hypothesis to be tested since, if confirmed, it supports the role of impulsivity as an endophenotype for BD [76,77]. However, neither two were confirmed in our sample. Specifically, allele frequencies were comparable between HC and BD individuals. Moreover, the polymorphism status effect on impulsivity scores was not dependent on diagnosis, implying similar modulatory effects on impulsivity in HC and BD individuals. Previous evidence on the role of these three SNPs in modulating impulsivity in BD subjects is limited to two studies on 5-HTTLPR. One found a differential effect of SNP between BD and HC samples; however, the study was focused on ADHD impulsivity features retrospectively measured [53]. The other study, in which cognitive impulsivity was measured through neuropsychological tools, found no differences in the effect of the SNP genotype status on impulsivity between two small samples of HC and BD individuals (non-significant effect in both) [52]. Therefore, since other genes might influence the complex trait of impulsivity and might interact with the diagnosis of BD, genome-wide screening studies on larger samples, with homogeneous impulsivity measures, are needed.
4.2. The Effect of BDNF on BIS Scores
In our sample the BDNF rs6265 genotype associated with low BDNF transcriptional efficiency (Met/Met) was associated with reduced impulsivity levels. While the Met substitution is traditionally associated with low transcriptional efficiency, studies in both healthy subjects [78] and individuals with BD [79] suggest a lack of association between genotype status and circulating BDNF plasma levels. Nonetheless, several studies point towards increased impulsivity levels in individuals carrying the Met allele. Specifically, in healthy individuals Met substitution is associated with worse performances in Go/No Go and Stop Signal Tasks [80]. Regarding substance use disorders, the Met allele is generally associated with an increased risk of abuse [65,80,81,82], with only one study indicating a protective effect for this allele [66]. Of note, none of these studies adopted BIS-11 to assess impulsivity. This could limit the comparability between studies, since different measures of impulsivity (e.g., self-report questionnaires and behavioral tasks) have been often shown to be poorly convergent [83]. Moreover, none of the studies adjusted the effect of the genotype for educational level or Raven IQ estimates, that in our model were independently associated with impulsivity scores.
4.3. HTTLPR x Gender Interaction Effect on Impulsivity Measures
We found that the effect of 5-HTTLPR polymorphism on impulsivity was dependent on gender, with the SS genotype associated with higher impulsivity levels across all the BIS-1 factors in females, but not in males.
The relationship between 5-HTTLPR status and serotoninergic brain activity is unclear. While the S allele has been traditionally associated with higher serotoninergic activity, a more complex picture emerges from recent studies, in which the SNP effect varies when different serotonin receptors are considered [84,85,86]. Regarding the relationship between 5-HTTLPR gender and serotoninergic activity, the SS genotype was found to be associated with lower 5-Hydroxyindoleacetic acid (5-HIAA) levels in men, but higher 5-HIAA levels in females [87]. If this assumption holds true also for our sample, it may suggest that, in females, higher central serotoninergic tone is associated with higher impulsivity. In support of this, it was previously shown how acute tryptophan depletion, a model simulating a low serotoninergic tone, increases impulsivity in males but decreases it in females [88].
Although a study measuring impulsivity through BIS-11 found no interactions between the 5-HTTLPR status and impulsivity scores [89], several other reports are in line with our result of higher impulsivity in females carrying the S allele. In healthy females, the SS genotype was found to be associated with deficits in working memory [90], which is closely dependent on Attentional processes [91]. Another model of Attentional and Motor impulsivity is represented by dysregulated eating behavior [92,93]. In women samples, the 5-HTTLPR S allele increased the risk of eating disturbances [94] and was associated with higher impulsivity in those prone to binge eating [95]. Furthermore, the SS genotype has been shown to be associated with higher disinhibition in adolescent girls [96], as well as hostility and aggressive behaviors [97], features that may be interpreted as surrogates of Motor impulsivity [98]. However, when suicidal behavior is considered as a surrogate measure of impulsivity, opposite results seem to emerge. Indeed, previous studies found an increased risk of suicide risk [99] and higher impulsivity in suicide attempts [100] among men carrying the S allele, as well as higher suicidal behavior in women with the LL genotype [101]. This could be the result of various factors. First, we cannot control for the effect of diagnosis of suicidal behavior, since in our sample data on suicidal behavior history were not available. Second, suicide risk may not necessarily represent a reliable surrogate of impulsivity, since only some types of suicidal behavior seem to be associated with impulsivity [102]; however, previous evidence found BIS-11 scores to be higher among suicide attempters [103,104].
Our results should be interpreted considering some limitations. First, we considered the effect of only three SNPs on impulsivity levels. This surely represents the main limitation of our study, considering the well-known pitfalls of candidate gene studies, such as being unpowered to detect effects of specific variants on genetically complex traits such as impulsivity [105]. Moreover, candidate gene-by-environment interaction findings often suffer from the “replication crisis” phenomenon [106]. Still, these three polymorphisms have been shown to significantly impact various neuropsychological and psychopathological dimensions among various disorders. Second, it is possible that a modulating effect of the three SNPs studied may have been hidden by the relatively small sample size employed in our study; however, the numerosity of our sample was comparable to the majority of other studies with similar design [26,52,107]. Third, the two groups had significant differences in terms of sociodemographic variables; however, although the mixed regression models accounted for these differences, an effect on our findings cannot be completely ruled out. Since impulsivity is a multidimensional and likely multifactorial trait, it is likely to be influenced by many clinical and non-clinical variables; in this context, a significant number of predictors of impulsivity changes in our sample could have been missed due to practical concerns. Finally, BIS is a self-administered questionnaire, with the intrinsic limitations of a subjective assessment. Other neuropsychological tests can be used to objectively assess the impulsivity trait, such as the Go/No Go, the Continuous Performance Test, the Stop Signal Task and the Iowa Gambling Task. However, the BIS is a validated instrument, and it is among the most widely adopted instruments for the assessment of impulsivity among clinical and non-clinical populations.
5. Conclusions
In our sample of BD individuals and HC, we found impulsivity levels, as measured by the BIS instrument, to be independently influenced by the BDNF rs6265 polymorphism. In addition, an interaction between the 5-HTTLPR status and gender was found, with the SS genotype associated with higher impulsivity levels among females. Of note, BD diagnosis did not exert any significant influence on the relationship between SNP status and impulsivity scores, suggesting a comparable effect of the polymorphisms among HC and individuals with BD.
Impulsivity represents a trait-like feature of BD and has been demonstrated to have prognostic implications in the disease; however, only few studies so far have explored the contribution of the genetic component in modulating impulsivity levels in these patients. Since impulsivity is a complex and multidimensional trait, studies employing new generation sequencing techniques, together with big data analysis methods, are needed to better define the genetic basis of trait impulsivity in BD.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/genes13030482/s1, Figure S1: Distribution of BIS-11 score (Attentional, Motor, Non-Planning and Total) among HC and BD; Figure S2: Quantile-quantile diagram for each mixed-effects regression model and p-value of Shapiro test for assessing the normal distribution of the residuals. Table S1: Coefficients (β) and relative 95% Confidence Interval estimated by the mixed-effects regression model considering as dependent variable the Attentional, Motor, Non-Planning, and Total BIS score with respect to the reference category; Table S2: Retrospective Design Analysis on the highest observed effect size (Cohen d), the ratio between the observed standard deviation (SDratio) and the sample size of each subgroup (n1 and n2) reporting the observed power (P) and the Type M (M) and Type S (S) errors. In bold is the observed power greater than 60%.
Author Contributions
Conceptualization of the GECOBIP project: P.B.; Design of this particular study: P.B., G.D. (Giuseppe Delvecchio) and A.S.; genetic analyses: G.D. (Giuseppe Damante); statistical analysis: P.G. and L.F.; investigation: M.L., G.M.M., A.F., M.S., V.A., M.C., M.B. (Marcella Bellani), C.P., M.G.R., M.B. (Matteo Balestrieri), G.D. (Giuseppe Damante), C.B. and R.R.; data curation: A.B. and P.G.; writing—original draft preparation: A.B., A.P. and G.D. (Giuseppe Delvecchio); writing—review and editing: all authors. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the Italian Ministry of Health, grant number GR-2016-02362413.
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study.
Data Availability Statement
Authors agree to make data and materials supporting the results or analyses presented in this paper available upon reasonable request.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Grande, I.; Berk, M.; Birmaher, B.; Vieta, E. Bipolar disorder. Lancet 2016, 387, 1561–1572. [Google Scholar] [CrossRef]
- WHO. Global Health Estimates: Life Expectancy and Leading Causes of Death and Disability; WHO: Geneva, Switzerland, 2019. [Google Scholar]
- Dong, M.; Lu, L.; Zhang, L.; Zhang, Q.; Ungvari, G.S.; Ng, C.H.; Yuan, Z.; Xiang, Y.; Wang, G.; Xiang, Y.-T. Prevalence of suicide attempts in bipolar disorder: A systematic review and meta-analysis of observational studies. Epidemiol. Psychiatr. Sci. 2020, 29, e63. [Google Scholar] [CrossRef] [PubMed]
- Fountoulakis, K.N.; Gonda, X.; Koufaki, I.; Hyphantis, T.; Cloninger, C.R. The Role of Temperament in the Etiopathogenesis of Bipolar Spectrum Illness. Harv. Rev. Psychiatry 2016, 24, 36–52. [Google Scholar] [CrossRef] [PubMed]
- Fletcher, K.; Parker, G.; Barrett, M.; Synnott, H.; McCraw, S. Temperament and personality in bipolar II disorder. J. Affect. Disord. 2012, 136, 304–309. [Google Scholar] [CrossRef] [PubMed]
- Engström, C.; Brändström, S.; Sigvardsson, S.; Cloninger, R.; Nylander, P.-O. Bipolar disorder: I. Temperament and character. J. Affect. Disord. 2004, 82, 131–134. [Google Scholar] [CrossRef]
- Zaninotto, L.; Souery, D.; Calati, R.; Di Nicola, M.; Montgomery, S.; Kasper, S.; Zohar, J.; Mendlewicz, J.; Robert Cloninger, C.; Serretti, A.; et al. Temperament and character profiles in bipolar I, bipolar II and major depressive disorder: Impact over illness course, comorbidity pattern and psychopathological features of depression. J. Affect. Disord. 2015, 184, 51–59. [Google Scholar] [CrossRef]
- Sparding, T.; Pålsson, E.; Joas, E.; Hansen, S.; Landén, M. Personality traits in bipolar disorder and influence on outcome. BMC Psychiatry 2017, 17, 159. [Google Scholar] [CrossRef]
- Meyer, T.D.; Hautzinger, M. Screening for bipolar disorders using the Hypomanic Personality Scale. J. Affect. Disord. 2003, 75, 149–154. [Google Scholar] [CrossRef]
- Johnson, S.L.; Carver, C.S.; Tharp, J.A. Suicidality in Bipolar Disorder: The Role of Emotion-Triggered Impulsivity. Suicide Life Threat. Behav. 2017, 47, 177–192. [Google Scholar] [CrossRef]
- Richard-Lepouriel, H.; Kung, A.-L.; Hasler, R.; Bellivier, F.; Prada, P.; Gard, S.; Ardu, S.; Kahn, J.-P.; Dayer, A.; Henry, C.; et al. Impulsivity and its association with childhood trauma experiences across bipolar disorder, attention deficit hyperactivity disorder and borderline personality disorder. J. Affect. Disord. 2019, 244, 33–41. [Google Scholar] [CrossRef]
- Ozten, M.; Erol, A. Impulsivity differences between bipolar and unipolar depression. Indian J. Psychiatry 2019, 61, 156–160. [Google Scholar] [CrossRef] [PubMed]
- Rote, J.; Dingelstadt, A.-M.-L.; Aigner, A.; Bauer, M.; Fiebig, J.; König, B.; Kunze, J.; Pfeiffer, S.; Pfennig, A.; Quinlivan, E.; et al. Impulsivity predicts illness severity in long-term course of bipolar disorder: A prospective approach. Aust. N. Z. J. Psychiatry 2018, 52, 876–886. [Google Scholar] [CrossRef]
- Henna, E.; Hatch, J.P.; Nicoletti, M.; Swann, A.C.; Zunta-Soares, G.; Soares, J.C. Is impulsivity a common trait in bipolar and unipolar disorders? Bipolar Disord. 2013, 15, 223–227. [Google Scholar] [CrossRef] [PubMed]
- Sanches, M.; Scott-Gurnell, K.; Patel, A.; Caetano, S.C.; Zunta-Soares, G.B.; Hatch, J.P.; Olvera, R.; Swann, A.C.; Soares, J.C. Impulsivity in children and adolescents with mood disorders and unaffected offspring of bipolar parents. Compr. Psychiatry 2014, 55, 1337–1341. [Google Scholar] [CrossRef] [PubMed]
- Wessa, M.; Kollmann, B.; Linke, J.; Schönfelder, S.; Kanske, P. Increased impulsivity as a vulnerability marker for bipolar disorder: Evidence from self-report and experimental measures in two high-risk populations. J. Affect. Disord. 2015, 178, 18–24. [Google Scholar] [CrossRef]
- Lombardo, L.E.; Bearden, C.E.; Barrett, J.; Brumbaugh, M.S.; Pittman, B.; Frangou, S.; Glahn, D.C. Trait impulsivity as an endophenotype for bipolar I disorder: Trait impulsivity as an endophenotype for BD-I. Bipolar Disord. 2012, 14, 565–570. [Google Scholar] [CrossRef] [PubMed]
- Moeller, F.G.; Barratt, E.S.; Dougherty, D.M.; Schmitz, J.M.; Swann, A.C. Psychiatric Aspects of Impulsivity. Am. J. Psychiatry 2001, 158, 1783–1793. [Google Scholar] [CrossRef] [PubMed]
- Pettinati, H.M.; O’Brien, C.P.; Dundon, W.D. Current Status of Co-Occurring Mood and Substance Use Disorders: A New Therapeutic Target. Am. J. Psychiatry 2013, 170, 23–30. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.-S.; Cha, B.; Lee, D.; Kim, S.-M.; Moon, E.; Park, C.-S.; Kim, B.-J.; Lee, C.-S.; Lee, S. The Relationship between Impulsivity and Quality of Life in Euthymic Patients with Bipolar Disorder. Psychiatry Investig. 2013, 10, 246. [Google Scholar] [CrossRef]
- Ng, T.H.; Stange, J.P.; Black, C.L.; Titone, M.K.; Weiss, R.B.; Abramson, L.Y.; Alloy, L.B. Impulsivity predicts the onset of DSM-IV-TR or RDC hypomanic and manic episodes in adolescents and young adults with high or moderate reward sensitivity. J. Affect. Disord. 2016, 198, 88–95. [Google Scholar] [CrossRef]
- Bezdjian, S.; Baker, L.A.; Tuvblad, C. Genetic and environmental influences on impulsivity: A meta-analysis of twin, family and adoption studies. Clin. Psychol. Rev. 2011, 31, 1209–1223. [Google Scholar] [CrossRef] [PubMed]
- Khadka, S.; Narayanan, B.; Meda, S.A.; Gelernter, J.; Han, S.; Sawyer, B.; Aslanzadeh, F.; Stevens, M.C.; Hawkins, K.A.; Anticevic, A.; et al. Genetic association of impulsivity in young adults: A multivariate study. Transl. Psychiatry 2014, 4, e451. [Google Scholar] [CrossRef] [PubMed]
- Sanchez-Roige, S.; Fontanillas, P.; Elson, S.L.; Gray, J.C.; de Wit, H.; MacKillop, J.; Palmer, A.A. Genome-wide association studies of impulsive personality traits (BIS-11 and UPPSP) and drug experimentation in up to 22,861 adult research participants identify loci in the CACNA1I and CADM2 genes. J. Neurosci. 2019, 39, 2562–2572. [Google Scholar] [CrossRef] [PubMed]
- Jiménez, E.; Arias, B.; Mitjans, M.; Goikolea, J.M.; Roda, E.; Ruíz, V.; Pérez, A.; Sáiz, P.A.; Paz García-Portilla, M.; Burón, P.; et al. Association between GSK3β gene and increased impulsivity in bipolar disorder. Eur. Neuropsychopharmacol. 2014, 24, 510–518. [Google Scholar] [CrossRef] [PubMed]
- Malloy-Diniz, L.F.; Neves, F.S.; de Moraes, P.H.P.; De Marco, L.A.; Romano-Silva, M.A.; Krebs, M.-O.; Corrêa, H. The 5-HTTLPR polymorphism, impulsivity and suicide behavior in euthymic bipolar patients. J. Affect. Disord. 2011, 133, 221–226. [Google Scholar] [CrossRef] [PubMed]
- Greenwood, T.A.; Joo, E.-J.; Shekhtman, T.; Sadovnick, A.D.; Remick, R.A.; Keck, P.E.; McElroy, S.L.; Kelsoe, J.R. Association of dopamine transporter gene variants with childhood ADHD features in bipolar disorder. Am. J. Med. Genet. B Neuropsychiatr. Genet. 2013, 162, 137–145. [Google Scholar] [CrossRef] [PubMed]
- Rao, S.; Han, X.; Shi, M.; Siu, C.O.; Waye, M.M.Y.; Liu, G.; Wing, Y.K. Associations of the serotonin transporter promoter polymorphism (5-HTTLPR) with bipolar disorder and treatment response: A systematic review and meta-analysis. Prog. Neuropsychopharmacol. Biol. Psychiatry 2019, 89, 214–226. [Google Scholar] [CrossRef]
- Chiou, Y.-J.; Huang, T.-L. Brain-derived neurotrophic factor (BDNF) and bipolar disorder. Psychiatry Res. 2019, 274, 395–399. [Google Scholar] [CrossRef]
- Ahmadi, L.; Kazemi Nezhad, S.R.; Behbahani, P.; Khajeddin, N.; Pourmehdi-Boroujeni, M. Genetic Variations of DAOA (rs947267 and rs3918342) and COMT Genes (rs165599 and rs4680) in Schizophrenia and Bipolar I Disorder. Basic Clin. Neurosci. J. 2018, 9, 429–438. [Google Scholar] [CrossRef]
- Porcelli, S.; Marsano, A.; Caletti, E.; Sala, M.; Abbiati, V.; Bellani, M.; Perlini, C.; Rossetti, M.G.; Mandolini, G.M.; Pigoni, A.; et al. Temperament and Character Inventory in Bipolar Disorder versus Healthy Controls and Modulatory Effects of 3 Key Functional Gene Variants. Neuropsychobiology 2017, 76, 209–221. [Google Scholar] [CrossRef]
- Käenmäki, M.; Tammimäki, A.; Myöhänen, T.; Pakarinen, K.; Amberg, C.; Karayiorgou, M.; Gogos, J.A.; Männistö, P.T. Quantitative role of COMT in dopamine clearance in the prefrontal cortex of freely moving mice: Quantitative role of COMT in the prefrontal cortex. J. Neurochem. 2010, 114, 1745–1755. [Google Scholar] [CrossRef] [PubMed]
- Olsson, C.A.; Byrnes, G.B.; Anney, R.J.L.; Collins, V.; Hemphill, S.A.; Williamson, R.; Patton, G.C. COMT Val158 Met and 5HTTLPR functional loci interact to predict persistence of anxiety across adolescence: Results from the Victorian Adolescent Health Cohort Study. Genes Brain Behav. 2007, 6, 647–652. [Google Scholar] [CrossRef]
- Lachman, H.M.; Papolos, D.F.; Saito, T.; Yu, Y.-M.; Szumlanski, C.L.; Weinshilboum, R.M. Human catechol-O-methyltransferase pharmacogenetics: Description of a functional polymorphism and its potential application to neuropsychiatric disorders. Pharmacogenetics 1996, 6, 243–250. [Google Scholar] [CrossRef] [PubMed]
- Pigoni, A.; Lazzaretti, M.; Mandolini, G.M.; Delvecchio, G.; Altamura, A.C.; Soares, J.C.; Brambilla, P. The impact of COMT polymorphisms on cognition in Bipolar Disorder: A review. J. Affect. Disord. 2019, 243, 545–551. [Google Scholar] [CrossRef] [PubMed]
- Calati, R.; Porcelli, S.; Giegling, I.; Hartmann, A.M.; Möller, H.-J.; De Ronchi, D.; Serretti, A.; Rujescu, D. Catechol-o-methyltransferase gene modulation on suicidal behavior and personality traits: Review, meta-analysis and association study. J. Psychiatr. Res. 2011, 45, 309–321. [Google Scholar] [CrossRef] [PubMed]
- Glavina Jelas, I.; Devic, I.; Karlovic, D. Cloninger’s temperament and character dimensions and dopaminergic genes: DAT1 VNTR and COMT Val158Met polymorphisms. Psychiatr. Danub. 2018, 30, 47–56. [Google Scholar] [CrossRef] [PubMed]
- Kotyuk, E.; Duchek, J.; Head, D.; Szekely, A.; Goate, A.M.; Balota, D.A. A genetic variant (COMT) coding dopaminergic activity predicts personality traits in healthy elderly. Personal. Individ. Differ. 2015, 82, 61–66. [Google Scholar] [CrossRef] [PubMed]
- Enge, S.; Sach, M.; Reif, A.; Lesch, K.-P.; Miller, R.; Fleischhauer, M. Cumulative Dopamine Genetic Score predicts behavioral and electrophysiological correlates of response inhibition via interactions with task demand. Cogn. Affect. Behav. Neurosci. 2020, 20, 59–75. [Google Scholar] [CrossRef]
- Paloyelis, Y.; Asherson, P.; Mehta, M.A.; Faraone, S.V.; Kuntsi, J. DAT1 and COMT Effects on Delay Discounting and Trait Impulsivity in Male Adolescents with Attention Deficit/Hyperactivity Disorder and Healthy Controls. Neuropsychopharmacology 2010, 35, 2414–2426. [Google Scholar] [CrossRef]
- Leehr, E.J.; Schag, K.; Brückmann, C.; Plewnia, C.; Zipfel, S.; Nieratschker, V.; Giel, K.E. A Putative Association of COMT Val(108/158)Met with Impulsivity in Binge Eating Disorder: COMT and BED. Eur. Eat. Disord. Rev. 2016, 24, 169–173. [Google Scholar] [CrossRef]
- Lazzaretti, M.; Fabbro, D.; Sala, M.; Del Toso, K.; de Vidovich, G.; Marraffini, E.; Morandotti, N.; Gambini, F.; Barale, F.; Balestrieri, M.; et al. Association between Low-Activity Allele of Cathecolamine-O-Methyl-Transferase (COMT) and Borderline Personality Disorder in an Italian Population. Behav. Med. 2013, 39, 25–28. [Google Scholar] [CrossRef] [PubMed]
- Coleman, J.A.; Green, E.M.; Gouaux, E. X-ray structures and mechanism of the human serotonin transporter. Nature 2016, 532, 334–339. [Google Scholar] [CrossRef] [PubMed]
- Möller, I.R.; Slivacka, M.; Nielsen, A.K.; Rasmussen, S.G.F.; Gether, U.; Loland, C.J.; Rand, K.D. Conformational dynamics of the human serotonin transporter during substrate and drug binding. Nat. Commun. 2019, 10, 1687. [Google Scholar] [CrossRef] [PubMed]
- Lesch, K.-P.; Bengel, D.; Heils, A.; Sabol, S.Z.; Greenberg, B.D.; Petri, S.; Benjamin, J.; Muller, C.R.; Hamer, D.H.; Murphy, D.L. Association of Anxiety-Related Traits with a Polymorphism in the Serotonin Transporter Gene Regulatory Region. Science 1996, 274, 1527–1531. [Google Scholar] [CrossRef]
- Gonda, X.; Fountoulakis, K.N.; Rihmer, Z.; Lazary, J.; Laszik, A.; Akiskal, K.K.; Akiskal, H.S.; Bagdy, G. Towards a genetically validated new affective temperament scale: A delineation of the temperament ’phenotype’ of 5-HTTLPR using the TEMPS-A. J. Affect. Disord. 2009, 112, 19–29. [Google Scholar] [CrossRef]
- Plieger, T.; Montag, C.; Felten, A.; Reuter, M. The serotonin transporter polymorphism (5-HTTLPR) and personality: Response style as a new endophenotype for anxiety. Int. J. Neuropsychopharmacol. 2014, 17, 851–858. [Google Scholar] [CrossRef]
- Luo, Y.L.L.; Welker, K.M.; Way, B.; DeWall, N.; Bushman, B.J.; Wildschut, T.; Sedikides, C. 5-HTTLPR polymorphism is associated with nostalgia proneness: The role of neuroticism. Soc. Neurosci. 2019, 14, 183–190. [Google Scholar] [CrossRef]
- Schepers, R.; Markus, C.R. The interaction between 5-HTTLPR genotype and ruminative thinking on BMI. Br. J. Nutr. 2017, 118, 629–637. [Google Scholar] [CrossRef]
- Talati, A.; Odgerel, Z.; Wickramaratne, P.J.; Norcini-Pala, A.; Skipper, J.L.; Gingrich, J.A.; Weissman, M.M. Associations between serotonin transporter and behavioral traits and diagnoses related to anxiety. Psychiatry Res. 2017, 253, 211–219. [Google Scholar] [CrossRef]
- Nomura, M.; Kaneko, M.; Okuma, Y.; Nomura, J.; Kusumi, I.; Koyama, T.; Nomura, Y. Involvement of Serotonin Transporter Gene Polymorphisms (5-HTT) in Impulsive Behavior in the Japanese Population. PLoS ONE 2015, 10, e0119743. [Google Scholar] [CrossRef]
- Lage, G.M.; Malloy-Diniz, L.F.; Matos, L.O.; Bastos, M.A.R.; Abrantes, S.S.C.; Corrêa, H. Impulsivity and the 5-HTTLPR Polymorphism in a Non-Clinical Sample. PLoS ONE 2011, 6, e16927. [Google Scholar] [CrossRef] [PubMed]
- Eun, T.K.; Jeong, S.H.; Lee, K.Y.; Kim, S.H.; Ahn, Y.M.; Bang, Y.W.; Joo, E.-J. Association between the 5-HTTLPR Genotype and Childhood Characteristics in Mood Disorders. Clin. Psychopharmacol. Neurosci. 2016, 14, 88–95. [Google Scholar] [CrossRef] [PubMed]
- Notaras, M.; van den Buuse, M. Brain-Derived Neurotrophic Factor (BDNF): Novel Insights into Regulation and Genetic Variation. Neuroscientist 2019, 25, 434–454. [Google Scholar] [CrossRef] [PubMed]
- Egan, M.F.; Kojima, M.; Callicott, J.H.; Goldberg, T.E.; Kolachana, B.S.; Bertolino, A.; Zaitsev, E.; Gold, B.; Goldman, D.; Dean, M.; et al. The BDNF val66met Polymorphism Affects Activity-Dependent Secretion of BDNF and Human Memory and Hippocampal Function. Cell 2003, 112, 257–269. [Google Scholar] [CrossRef]
- In Kang, J.; Song, D.-H.; Namkoong, K.; Kim, S.J. Interaction effects between COMT and BDNF polymorphisms on boredom susceptibility of sensation seeking traits. Psychiatry Res. 2010, 178, 132–136. [Google Scholar] [CrossRef]
- Kim, S.J.; Cho, S.-J.; Jang, H.M.; Shin, J.; Park, P.-W.; Lee, Y.J.; Cho, I.H.; Choi, J.-E.; Lee, H.-J. Interaction between Brain-Derived Neurotrophic Factor Val66Met Polymorphism and Recent Negative Stressor in Harm Avoidance. Neuropsychobiology 2010, 61, 19–26. [Google Scholar] [CrossRef]
- Toh, Y.L.; Ng, T.; Tan, M.; Tan, A.; Chan, A. Impact of brain-derived neurotrophic factor genetic polymorphism on cognition: A systematic review. Brain Behav. 2018, 8, e01009. [Google Scholar] [CrossRef]
- Mandolini, G.M.; Lazzaretti, M.; Pigoni, A.; Delvecchio, G.; Soares, J.C.; Brambilla, P. The impact of BDNF Val66Met polymorphism on cognition in Bipolar Disorder: A review. J. Affect. Disord. 2019, 243, 552–558. [Google Scholar] [CrossRef]
- Kambeitz, J.P.; Bhattacharyya, S.; Kambeitz-Ilankovic, L.M.; Valli, I.; Collier, D.A.; McGuire, P. Effect of BDNF val66met polymorphism on declarative memory and its neural substrate: A meta-analysis. Neurosci. Biobehav. Rev. 2012, 36, 2165–2177. [Google Scholar] [CrossRef]
- Karg, K.; Burmeister, M.; Shedden, K.; Sen, S. The Serotonin Transporter Promoter Variant (5-HTTLPR), Stress, and Depression Meta-analysis Revisited: Evidence of Genetic Moderation. Arch. Gen. Psychiatry 2011, 68, 444. [Google Scholar] [CrossRef]
- González-Castro, T.B.; Salas-Magaña, M.; Juárez-Rojop, I.E.; López-Narváez, M.L.; Tovilla-Zárate, C.A.; Hernández-Díaz, Y. Exploring the association between BDNF Val66Met polymorphism and suicidal behavior: Meta-analysis and systematic review. J. Psychiatr. Res. 2017, 94, 208–217. [Google Scholar] [CrossRef] [PubMed]
- Youssef, M.M.; Underwood, M.D.; Huang, Y.-Y.; Hsiung, S.; Liu, Y.; Simpson, N.R.; Bakalian, M.J.; Rosoklija, G.B.; Dwork, A.J.; Arango, V.; et al. Association of BDNF Val66Met Polymorphism and Brain BDNF Levels with Major Depression and Suicide. Int. J. Neuropsychopharmacol. 2018, 21, 528–538. [Google Scholar] [CrossRef] [PubMed]
- He, L.; Liao, Y.; Wu, Q.; Liu, T. Association Between Brain-Derived Neurotrophic Factor Val66Met Polymorphism and Methamphetamine Use Disorder: A Meta-Analysis. Front. Psychiatry 2020, 11, 585852. [Google Scholar] [CrossRef]
- Su, H.; Tao, J.; Zhang, J.; Xie, Y.; Sun, Y.; Li, L.; Xu, K.; Han, B.; Lu, Y.; Sun, H.; et al. An association between BDNF Val66Met polymorphism and impulsivity in methamphetamine abusers. Neurosci. Lett. 2014, 582, 16–20. [Google Scholar] [CrossRef] [PubMed]
- Wojnar, M.; Brower, K.J.; Strobbe, S.; Ilgen, M.; Matsumoto, H.; Nowosad, I.; Sliwerska, E.; Burmeister, M. Association between Val66Met brain-derived neurotrophic factor (BDNF) gene polymorphism and post-treatment relapse in alcohol dependence. Alcohol. Clin. Exp. Res. 2009, 33, 693–702. [Google Scholar] [CrossRef] [PubMed]
- Patton, J.H.; Stanford, M.S.; Barratt, E.S. Factor structure of the Barratt impulsiveness scale. J. Clin. Psychol. 1995, 51, 768–774. [Google Scholar] [CrossRef]
- Fossati, A.; Di Ceglie, A.; Acquarini, E.; Barratt, E.S. Psychometric properties of an Italian version of the Barratt Impulsiveness Scale-11 (BIS-11) in nonclinical subjects. J. Clin. Psychol. 2001, 57, 815–828. [Google Scholar] [CrossRef]
- Rylander-Rudqvist, T.; Håkansson, N.; Tybring, G.; Wolk, A. Quality and Quantity of Saliva DNA Obtained from the Self-administrated Oragene Method—A Pilot Study on the Cohort of Swedish Men. Cancer Epidemiol. Biomark. Prev. 2006, 15, 1742–1745. [Google Scholar] [CrossRef]
- Altoè, G.; Bertoldo, G.; Zandonella Callegher, C.; Toffalini, E.; Calcagnì, A.; Finos, L.; Pastore, M. Enhancing Statistical Inference in Psychological Research via Prospective and Retrospective Design Analysis. Front. Psychol. 2020, 10, 2893. [Google Scholar] [CrossRef]
- Callegher, C.; Bertoldo, G.; Toffalini, E.; Vesely, A.; Andreella, A.; Pastore, M.; Altoè, G. PRDA: An R package for Prospective and Retrospective Design Analysis. J. Open Source Softw. 2021, 6, 2810. [Google Scholar] [CrossRef]
- Shen, T.; You, Y.; Joseph, C.; Mirzaei, M.; Klistorner, A.; Graham, S.L.; Gupta, V. BDNF Polymorphism: A Review of Its Diagnostic and Clinical Relevance in Neurodegenerative Disorders. Aging Dis. 2018, 9, 523–536. [Google Scholar] [CrossRef] [PubMed]
- Culverhouse, R.C.; Saccone, N.L.; Horton, A.C.; Ma, Y.; Anstey, K.J.; Banaschewski, T.; Burmeister, M.; Cohen-Woods, S.; Etain, B.; Fisher, H.L.; et al. Collaborative meta-analysis finds no evidence of a strong interaction between stress and 5-HTTLPR genotype contributing to the development of depression. Mol. Psychiatry 2018, 23, 133–142. [Google Scholar] [CrossRef] [PubMed]
- Liao, Y.-J.; Jiang, J.-R.; Jin, S.-Q. The association between COMT Val158Met polymorphism and migraine risk: A meta-analysis. Cephalalgia 2017, 37, 592–598. [Google Scholar] [CrossRef] [PubMed]
- Saddichha, S.; Schuetz, C. Is impulsivity in remitted bipolar disorder a stable trait? A meta-analytic review. Compr. Psychiatry 2014, 55, 1479–1484. [Google Scholar] [CrossRef]
- Gottesman, I.I.; Gould, T.D. The Endophenotype Concept in Psychiatry: Etymology and Strategic Intentions. Am. J. Psychiatry 2003, 160, 636–645. [Google Scholar] [CrossRef]
- Hasler, G.; Drevets, W.C.; Gould, T.D.; Gottesman, I.I.; Manji, H.K. Toward Constructing an Endophenotype Strategy for Bipolar Disorders. Biol. Psychiatry 2006, 60, 93–105. [Google Scholar] [CrossRef]
- Luykx, J.J.; Boks, M.P.M.; Breetvelt, E.J.; Aukes, M.F.; Strengman, E.; da Pozzo, E.; Dell’osso, L.; Marazziti, D.; van Leeuwen, A.; Vreeker, A.; et al. BDNF Val66Met homozygosity does not influence plasma BDNF levels in healthy human subjects. Prog. Neuropsychopharmacol. Biol. Psychiatry 2013, 43, 185–187. [Google Scholar] [CrossRef]
- Chang, Y.-H.; Wang, T.-Y.; Lee, S.-Y.; Chen, S.-L.; Huang, C.-C.; Chen, P.S.; Yang, Y.K.; Hong, J.-S.; Lu, R.-B. Memory Impairment and Plasma BDNF Correlates of the BDNF Val66Met Polymorphism in Patients With Bipolar II Disorder. Front. Genet. 2018, 9, 583. [Google Scholar] [CrossRef]
- Enge, S.; Fleischhauer, M.; Gärtner, A.; Reif, A.; Lesch, K.-P.; Kliegel, M.; Strobel, A. Brain-Derived Neurotrophic Factor (Val66Met) and Serotonin Transporter (5-HTTLPR) Polymorphisms Modulate Plasticity in Inhibitory Control Performance Over Time but Independent of Inhibitory Control Training. Front. Hum. Neurosci. 2016, 10, 370. [Google Scholar] [CrossRef]
- Cheng, C.Y.; Hong, C.J.; Yu, Y.W.Y.; Chen, T.J.; Wu, H.C.; Tsai, S.J. Brain-derived neurotrophic factor (Val66Met) genetic polymorphism is associated with substance abuse in males. Mol. Brain Res. 2005, 140, 86–90. [Google Scholar] [CrossRef]
- Greenwald, M.K.; Steinmiller, C.L.; Śliwerska, E.; Lundahl, L.; Burmeister, M. BDNF Val/Met genotype is associated with drug-seeking phenotypes in heroin-dependent individuals: A pilot study: BDNF and drug-seeking behavior. Addict. Biol. 2013, 18, 836–845. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Gomide Vasconcelos, A.; Sergeant, J.; Corrêa, H.; Mattos, P.; Malloy-Diniz, L. When self-report diverges from performance: The usage of BIS-11 along with neuropsychological tests. Psychiatry Res. 2014, 218, 236–243. [Google Scholar] [CrossRef] [PubMed]
- Fisher, P.M.; Holst, K.K.; Adamsen, D.; Klein, A.B.; Frokjaer, V.G.; Jensen, P.S.; Svarer, C.; Gillings, N.; Baare, W.F.C.; Mikkelsen, J.D.; et al. BDNF Val66met and 5-HTTLPR polymorphisms predict a human in vivo marker for brain serotonin levels: Genetic Predictors of Human Brain Serotonin Marker. Hum. Brain Mapp. 2015, 36, 313–323. [Google Scholar] [CrossRef] [PubMed]
- Baldinger, P.; Kraus, C.; Rami-Mark, C.; Gryglewski, G.; Kranz, G.S.; Haeusler, D.; Hahn, A.; Spies, M.; Wadsak, W.; Mitterhauser, M.; et al. Interaction between 5-HTTLPR and 5-HT1B genotype status enhances cerebral 5-HT1A receptor binding. NeuroImage 2015, 111, 505–512. [Google Scholar] [CrossRef]
- Spies, M.; Nasser, A.; Ozenne, B.; Jensen, P.S.; Knudsen, G.M.; Fisher, P.M. Common HTR2A variants and 5-HTTLPR are not associated with human in vivo serotonin 2A receptor levels. Hum. Brain Mapp. 2020, 41, 4518–4528. [Google Scholar] [CrossRef]
- Williams, R.B.; Marchuk, D.A.; Gadde, K.M.; Barefoot, J.C.; Grichnik, K.; Helms, M.J.; Kuhn, C.M.; Lewis, J.G.; Schanberg, S.M.; Stafford-Smith, M.; et al. Central Nervous System Serotonin Function and Cardiovascular Responses to Stress. Psychosom. Med. 2001, 63, 300–305. [Google Scholar] [CrossRef]
- Walderhaug, E.; Herman, A.I.; Magnusson, A.; Morgan, M.J.; Landrø, N.I. The short (S) allele of the serotonin transporter polymorphism and acute tryptophan depletion both increase impulsivity in men. Neurosci. Lett. 2010, 473, 208–211. [Google Scholar] [CrossRef][Green Version]
- Stoltenberg, S.F.; Christ, C.C.; Highland, K.B. Serotonin system gene polymorphisms are associated with impulsivity in a context dependent manner. Prog. Neuropsychopharmacol. Biol. Psychiatry 2012, 39, 182–191. [Google Scholar] [CrossRef]
- Weiss, E.M.; Schulter, G.; Fink, A.; Reiser, E.M.; Mittenecker, E.; Niederstätter, H.; Nagl, S.; Parson, W.; Papousek, I. Influences of COMT and 5-HTTLPR Polymorphisms on Cognitive Flexibility in Healthy Women: Inhibition of Prepotent Responses and Memory Updating. PLoS ONE 2014, 9, e85506. [Google Scholar] [CrossRef]
- Awh, E.; Vogel, E.K.; Oh, S.-H. Interactions between attention and working memory. Neuroscience 2006, 139, 201–208. [Google Scholar] [CrossRef]
- Meule, A.; de Zwaan, M.; Müller, A. Attentional and motor impulsivity interactively predict ‘food addiction’ in obese individuals. Compr. Psychiatry 2017, 72, 83–87. [Google Scholar] [CrossRef] [PubMed]
- Ebneter, D.; Latner, J.; Rosewall, J.; Chisholm, A. Impulsivity in restrained eaters: Emotional and external eating are associated with attentional and motor impulsivity. Eat. Weight Disord. Stud. Anorex. Bulim. Obes. 2012, 17, e62–e65. [Google Scholar] [CrossRef] [PubMed]
- Stoltenberg, S.F.; Anderson, C.; Nag, P.; Anagnopoulos, C. Association between the serotonin transporter triallelic genotype and eating problems is moderated by the experience of childhood trauma in women. Int. J. Eat. Disord. 2012, 45, 492–500. [Google Scholar] [CrossRef] [PubMed]
- Akkermann, K.; Nordquist, N.; Oreland, L.; Harro, J. Serotonin transporter gene promoter polymorphism affects the severity of binge eating in general population. Prog. Neuropsychopharmacol. Biol. Psychiatry 2010, 34, 111–114. [Google Scholar] [CrossRef] [PubMed]
- Paaver, M.; Kurrikoff, T.; Nordquist, N.; Oreland, L.; Harro, J. The effect of 5-HTT gene promoter polymorphism on impulsivity depends on family relations in girls. Prog. Neuropsychopharmacol. Biol. Psychiatry 2008, 32, 1263–1268. [Google Scholar] [CrossRef]
- Gonda, X.; Fountoulakis, K.N.; Csukly, G.; Bagdy, G.; Pap, D.; Molnár, E.; Laszik, A.; Lazary, J.; Sarosi, A.; Faludi, G.; et al. Interaction of 5-HTTLPR genotype and unipolar major depression in the emergence of aggressive/hostile traits. J. Affect. Disord. 2011, 132, 432–437. [Google Scholar] [CrossRef]
- Pawliczek, C.M.; Derntl, B.; Kellermann, T.; Kohn, N.; Gur, R.C.; Habel, U. Inhibitory control and trait aggression: Neural and behavioral insights using the emotional stop signal task. NeuroImage 2013, 79, 264–274. [Google Scholar] [CrossRef]
- Limosin, F.; Loze, J.-Y.; Boni, C.; Hamon, M.; Adès, J.; Rouillon, F.; Gorwood, P. Male-specific association between the 5-HTTLPR S allele and suicide attempts in alcohol-dependent subjects. J. Psychiatr. Res. 2005, 39, 179–182. [Google Scholar] [CrossRef]
- Jiménez-Treviño, L.; Saiz, P.A.; García-Portilla, M.P.; Blasco-Fontecilla, H.; Carli, V.; Iosue, M.; Jaussent, I.; López-Castroman, J.; Vaquero-Lorenzo, C.; Sarchiapone, M.; et al. 5-HTTLPR–brain-derived neurotrophic factor (BDNF) gene interactions and early adverse life events effect on impulsivity in suicide attempters. World J. Biol. Psychiatry 2019, 20, 137–149. [Google Scholar] [CrossRef]
- Gaysina, D.; Zainullina, A.; Gabdulhakov, R.; Khusnutdinova, E. The Serotonin Transporter Gene: Polymorphism and Haplotype Analysis in Russian Suicide Attempters. Neuropsychobiology 2006, 54, 70–74. [Google Scholar] [CrossRef]
- Gvion, Y.; Levi-Belz, Y.; Hadlaczky, G.; Apter, A. On the role of impulsivity and decision-making in suicidal behavior. World J. Psychiatry 2015, 5, 255. [Google Scholar] [CrossRef] [PubMed]
- Doihara, C.; Kawanishi, C.; Ohyama, N.; Yamada, T.; Nakagawa, M.; Iwamoto, Y.; Odawara, T.; Hirayasu, Y. Trait impulsivity in suicide attempters: Preliminary study: Impulsivity in suicide attempters. Psychiatry Clin. Neurosci. 2012, 66, 529–532. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; He, C.Z.; Yu, Y.M.; Qiu, X.H.; Yang, X.X.; Qiao, Z.X.; Sui, H.; Zhu, X.Z.; Yang, Y.J. Associations between impulsivity, aggression, and suicide in Chinese college students. BMC Public Health 2014, 14, 551. [Google Scholar] [CrossRef]
- Border, R.; Johnson, E.C.; Evans, L.M.; Smolen, A.; Berley, N.; Sullivan, P.F.; Keller, M.C. No Support for Historical Candidate Gene or Candidate Gene-by-Interaction Hypotheses for Major Depression Across Multiple Large Samples. Am. J. Psychiatry 2019, 176, 376–387. [Google Scholar] [CrossRef]
- Duncan, L.E.; Keller, M.C. A Critical Review of the First 10 Years of Candidate Gene-by-Environment Interaction Research in Psychiatry. Am. J. Psychiatry 2011, 168, 1041–1049. [Google Scholar] [CrossRef] [PubMed]
- Pasyk, S.; Sanger, N.; Kapczinski, F.; Samaan, Z. Evaluation of BDNF as a biomarker for impulsivity in a psychiatric population. Diagnostics 2020, 10, 419. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).