Canalized Morphogenesis Driven by Inherited Tissue Asymmetries in Hydra Regeneration
Abstract
:1. Introduction
2. Methods
2.1. Hydra Strains and Culture
2.2. Sample Preparation and Fluorescent Tissue Labeling
2.2.1. One-Color Labeling
2.2.2. Two-Color Labeling
2.2.3. Microscopy
2.2.4. Image Analysis
3. Results
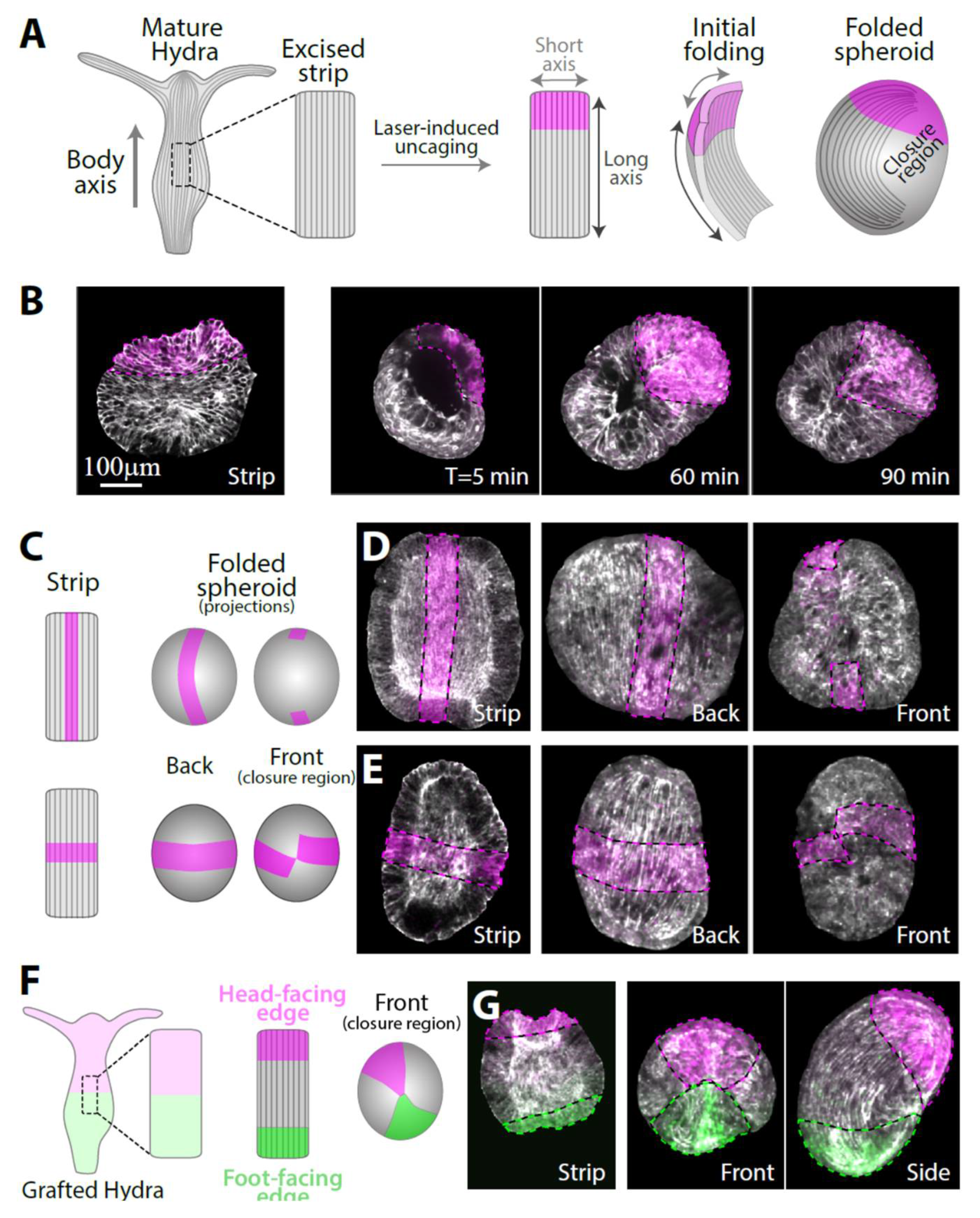
3.1. Folding of an Excised Tissue Strip into a Hollow Spheroid
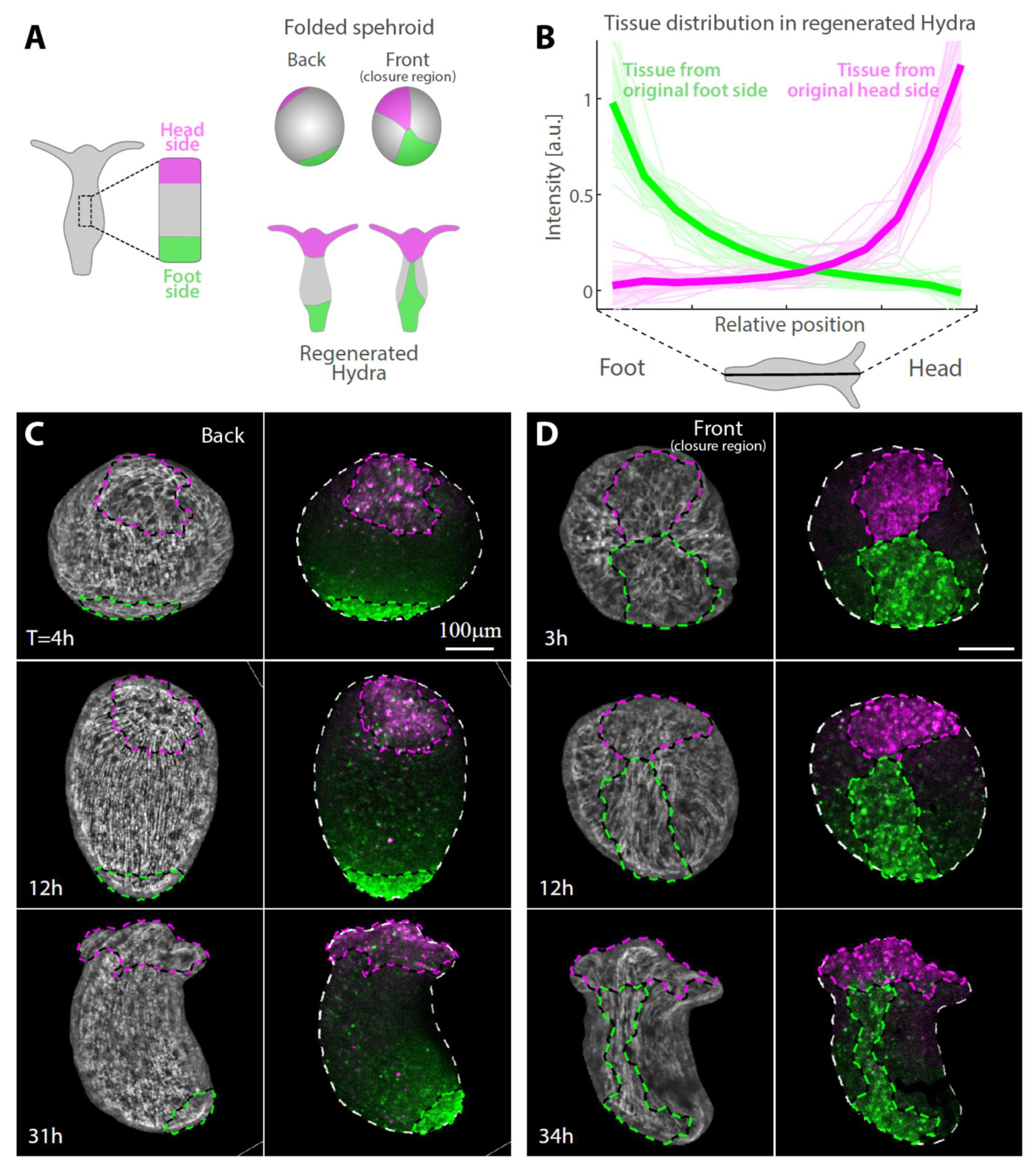
3.2. Tissue Reorganization during Regeneration
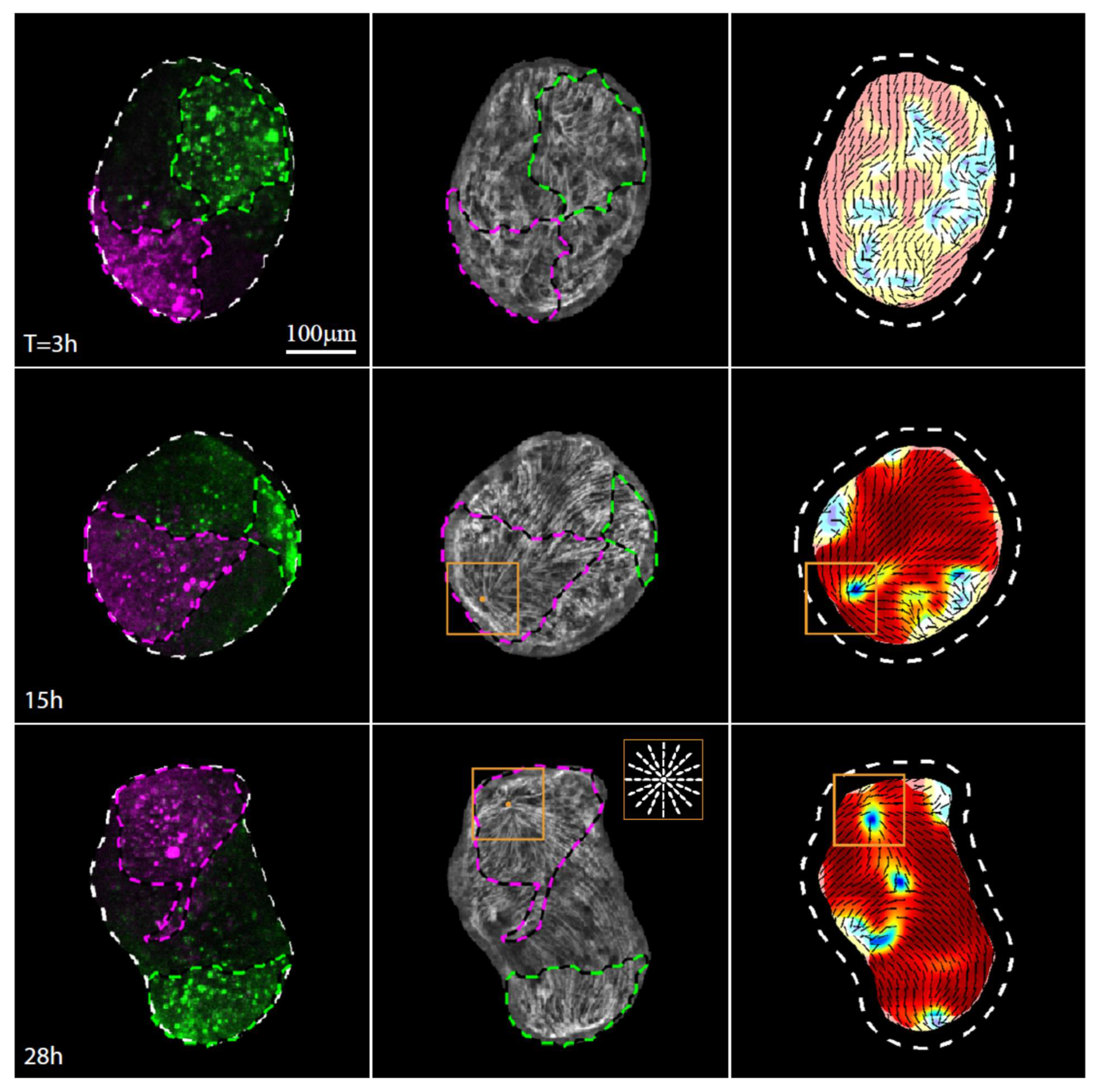
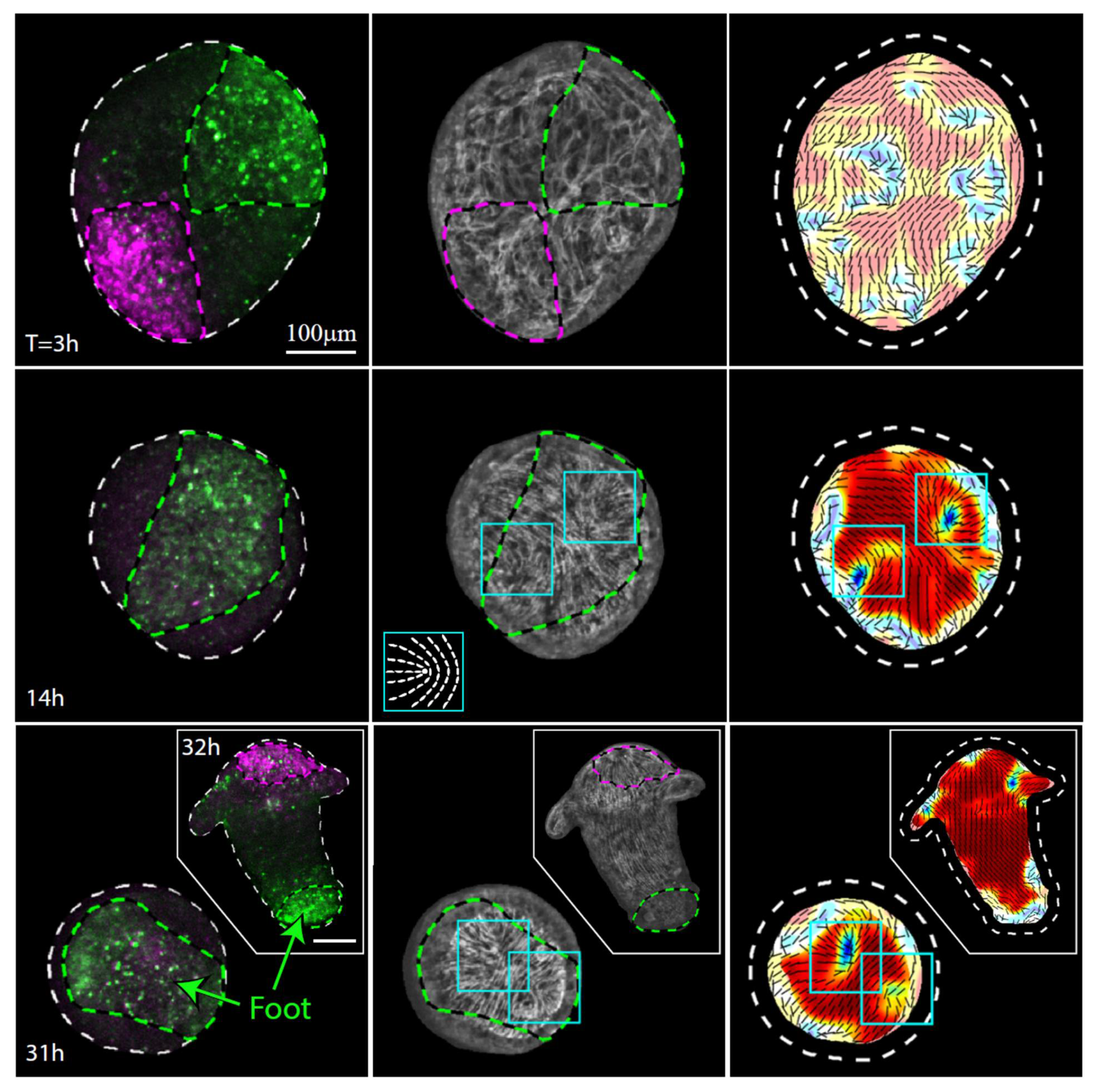
3.3. Establishment of a New Body Axis and Actin Fiber Reorganization
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Bode, H.R. Axial Patterning in Hydra. Cold Spring Harb. Perspect. Biol. 2009, 1, a000463. [Google Scholar] [CrossRef] [Green Version]
- Kass-Simon, G. Coordination of Juxtaposed Muscle Layers as Seen in Hydra. In Coelenterate Ecology and Behavior; Springer: Berlin/Heidelberg, Germany, 1976; pp. 705–714. [Google Scholar]
- Aufschnaiter, R.; Wedlich-Söldner, R.; Zhang, X.; Hobmayer, B. Apical and basal epitheliomuscular F-actin dynamics during Hydra bud evagination. Biol. Open 2017, 6, 1137–1148. [Google Scholar] [CrossRef] [Green Version]
- Livshits, A.; Shani-Zerbib, L.; Maroudas-Sacks, Y.; Braun, E.; Keren, K. Structural inheritance of the actin cytoskeletal organization determines the body axis in regenerating Hydra. Cell Rep. 2017, 18, 1410–1421. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hobmayer, B.; Rentzsch, F.; Kuhn, K.; Happel, C.M.; Von Laue, C.C.; Snyder, P.; Rothbächer, U.; Holstein, T.W. WNT signalling molecules act in axis formation in the diploblastic metazoan Hydra. Nature 2000, 407, 186–189. [Google Scholar] [CrossRef] [PubMed]
- Lengfeld, T.; Watanabe, H.; Simakov, O.; Lindgens, D.; Gee, L.; Law, L.; Schmidt, H.A.; Özbek, S.; Bode, H.; Holstein, T.W. Multiple Wnts are involved in Hydra organizer formation and regeneration. Dev. Biol. 2009, 330, 186–199. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nakamura, Y.; Tsiairis, C.D.; Özbek, S.; Holstein, T.W. Autoregulatory and repressive inputs localize Hydra Wnt3 to the head organizer. Proc. Natl. Acad. Sci. USA 2011, 108, 9137–9142. [Google Scholar] [CrossRef] [Green Version]
- Bode, H.R. The head organizer in Hydra. Int. J. Dev. Biol. 2012, 56, 473–478. [Google Scholar] [CrossRef] [Green Version]
- Javois, L.C.; Bode, P.M.; Bode, H.R. Patterning of the head in hydra as visualized by a monoclonal antibody: II. The initiation and localization of head structures in regenerating pieces of tissue. Dev. Biol. 1988, 129, 390–399. [Google Scholar] [CrossRef]
- Wang, R.; Steele, R.E.; Collins, E.-M.S. Wnt signaling determines body axis polarity in regenerating Hydra tissue fragments. Dev. Biol. 2020, 467, 88–94. [Google Scholar] [CrossRef]
- Livshits, A.; Garion, L.; Maroudas-Sacks, Y.; Shani-Zerbib, L.; Keren, K.; Braun, E. Plasticity of body axis polarity in Hydra regeneration under constraints. bioRxiv 2021. [CrossRef]
- Bode, P.M.; Bode, H.R. Formation of pattern in regenerating tissue pieces of Hydra attenuata: III. The shaping of the body column. Dev. Biol. 1984, 106, 315–325. [Google Scholar] [CrossRef]
- Trembley, A. Mémoires Pour Servir à L’histoire D’un Genre de Polypes D’eau Douce, à Bras en Forme de Cornes; Jean & Herman Verbeek: Leiden, The Netherlands, 1744. [Google Scholar]
- MacWilliams, H.K. Hydra transplantation phenomena and the mechanism of Hydra head regeneration: II. Properties of the head activation. Dev. Biol. 1983, 96, 239–257. [Google Scholar] [CrossRef]
- Brooun, M.; Gee, L.; Reinhardt, B.; Bode, H.R. Formation of the head organizer in hydra involves the canonical Wnt pathway. Dev. 2005, 132, 2907–2916. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gufler, S.; Artes, B.; Bielen, H.; Krainer, I.; Eder, M.K.; Falschlunger, J.; Bollmann, A.; Ostermann, T.; Valovka, T.; Hartl, M.; et al. Beta-Catenin acts in a position-independent regeneration response in the simple eumetazoan Hydra. Dev. Biol. 2018, 433, 310–323. [Google Scholar] [CrossRef] [PubMed]
- Tursch, A.; Bartsch, N.; Holstein, T.W. MAPK signaling links the injury response to Wnt-regulated patterning in Hydra regeneration. bioRxiv 2020. [CrossRef]
- Cazet, J.F.; Cho, A.; Juliano, C.E. Generic injuries are sufficient to induce ectopic Wnt organizers in Hydra. eLife 2021, 10, e60562. [Google Scholar] [CrossRef]
- Maroudas-Sacks, Y.; Garion, L.; Shani-Zerbib, L.; Livshits, A.; Braun, E.; Keren, K. Topological defects in the ne-matic order of actin fibers as organization centers of Hydra morphogenesis. Nat. Phys. 2021, 17, 251–259. [Google Scholar] [CrossRef]
- Wilby, O.K.; Webster, G. Experimental studies on axial polarity in hydra. J. Embryol. Exp. Morphol. 1970, 24, 595–613. [Google Scholar] [CrossRef]
- Marcum, B.A.; Campbell, R.D.; Romero, J. Polarity Reversal in Nerve-Free Hydra. Science 1977, 197, 771–773. [Google Scholar] [CrossRef]
- Petersen, H.O.; Höger, S.K.; Looso, M.; Lengfeld, T.; Kuhn, A.; Warnken, U.; Nishimiya-Fujisawa, C.; Schnölzer, M.; Krüger, M.; Özbek, S.; et al. A Comprehensive Transcriptomic and Proteomic Analysis of Hydra Head Regeneration. Mol. Biol. Evol. 2015, 32, 1928–1947. [Google Scholar] [CrossRef]
- Pincus, Z.; Theriot, J.A. Comparison of quantitative methods for cell-shape analysis. J. Microsc. 2007, 227, 140–156. [Google Scholar] [CrossRef]
- Sinigaglia, C.; Peron, S.; Eichelbrenner, J.; Chevalier, S.; Steger, J.; Barreau, C.; Houliston, E.; Leclère, L. Pattern regulation in a regenerating jellyfish. eLife 2020, 9, e54868. [Google Scholar] [CrossRef]
- Waddington, C.H. Canalization of development and the inheritance of acquired characters. Nature 1942, 150, 563–565. [Google Scholar] [CrossRef]
- Braun, E.; Keren, K. Hydra Regeneration: Closing the Loop with Mechanical Processes in Morphogenesis. BioEssays 2018, 40, e1700204. [Google Scholar] [CrossRef] [PubMed]
- Collinet, C.; Lecuit, T. Programmed and self-organized flow of information during morphogenesis. Nat. Rev. Mol. Cell Biol. 2021, 22, 245–265. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shani-Zerbib, L.; Garion, L.; Maroudas-Sacks, Y.; Braun, E.; Keren, K. Canalized Morphogenesis Driven by Inherited Tissue Asymmetries in Hydra Regeneration. Genes 2022, 13, 360. https://doi.org/10.3390/genes13020360
Shani-Zerbib L, Garion L, Maroudas-Sacks Y, Braun E, Keren K. Canalized Morphogenesis Driven by Inherited Tissue Asymmetries in Hydra Regeneration. Genes. 2022; 13(2):360. https://doi.org/10.3390/genes13020360
Chicago/Turabian StyleShani-Zerbib, Lital, Liora Garion, Yonit Maroudas-Sacks, Erez Braun, and Kinneret Keren. 2022. "Canalized Morphogenesis Driven by Inherited Tissue Asymmetries in Hydra Regeneration" Genes 13, no. 2: 360. https://doi.org/10.3390/genes13020360
APA StyleShani-Zerbib, L., Garion, L., Maroudas-Sacks, Y., Braun, E., & Keren, K. (2022). Canalized Morphogenesis Driven by Inherited Tissue Asymmetries in Hydra Regeneration. Genes, 13(2), 360. https://doi.org/10.3390/genes13020360




