Sex Determination and Differentiation in Decapod and Cladoceran Crustaceans: An Overview of Endocrine Regulation
Abstract
1. Introduction
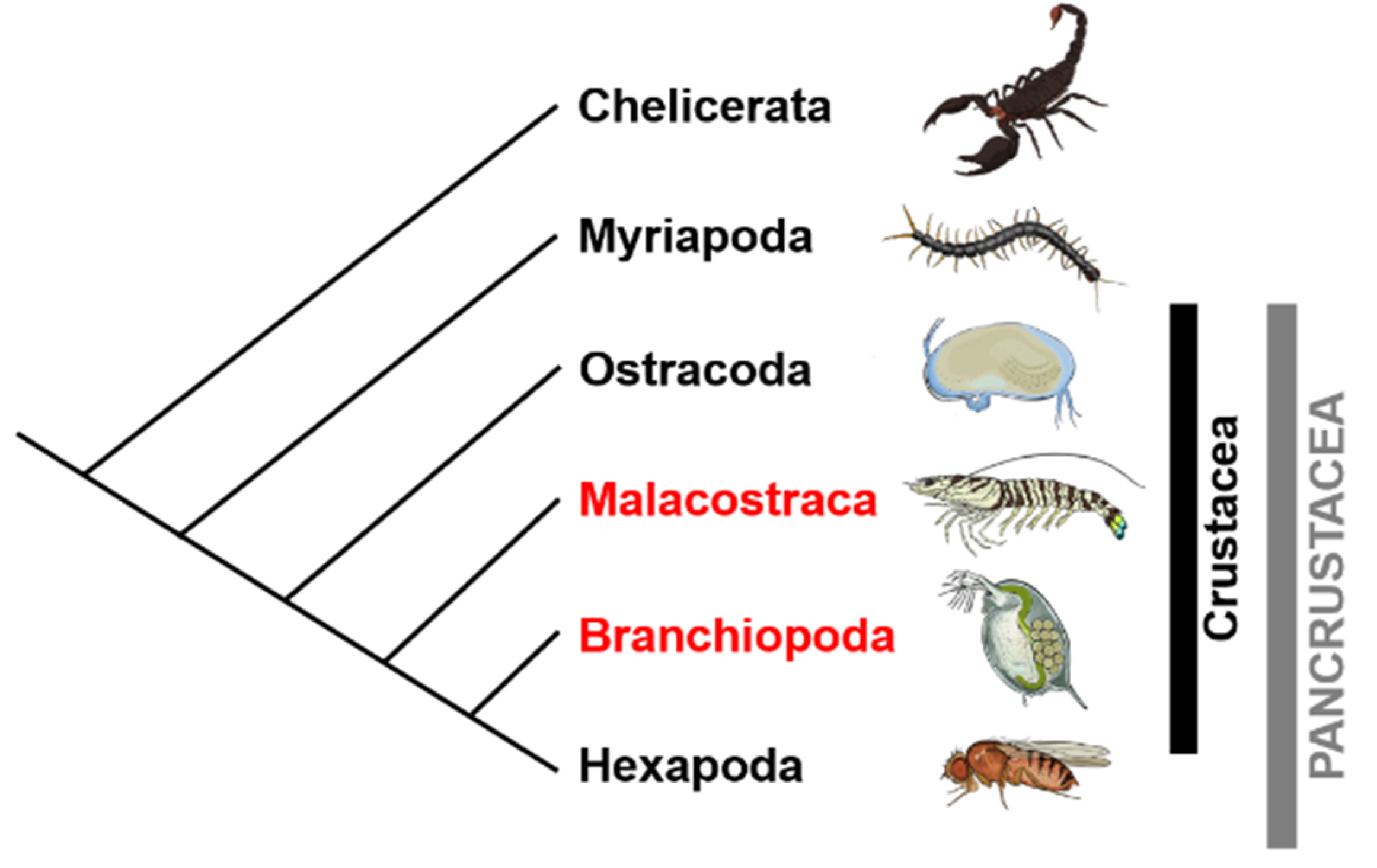
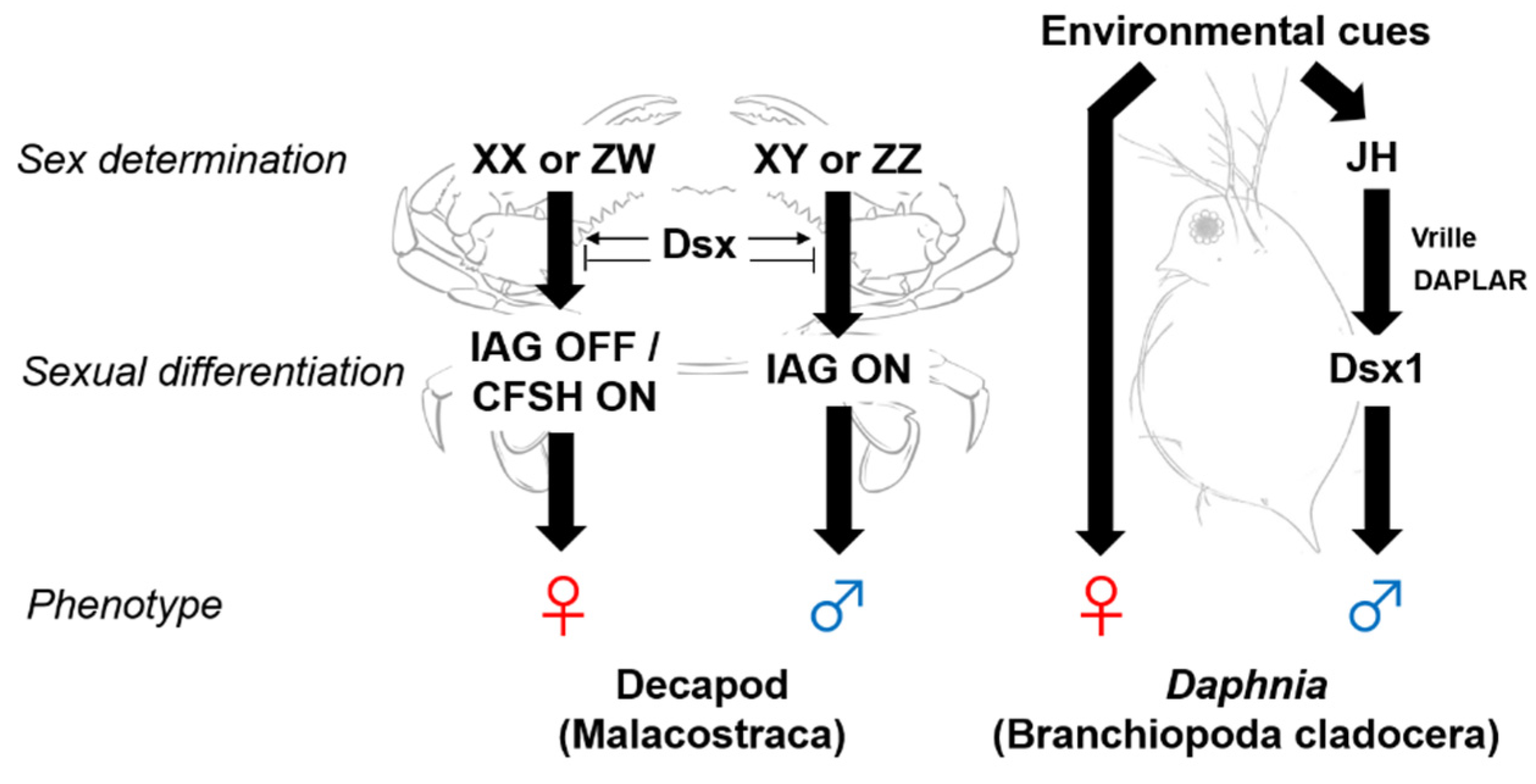
2. Sex Determination and Differentiation Mechanisms in Crustaceans
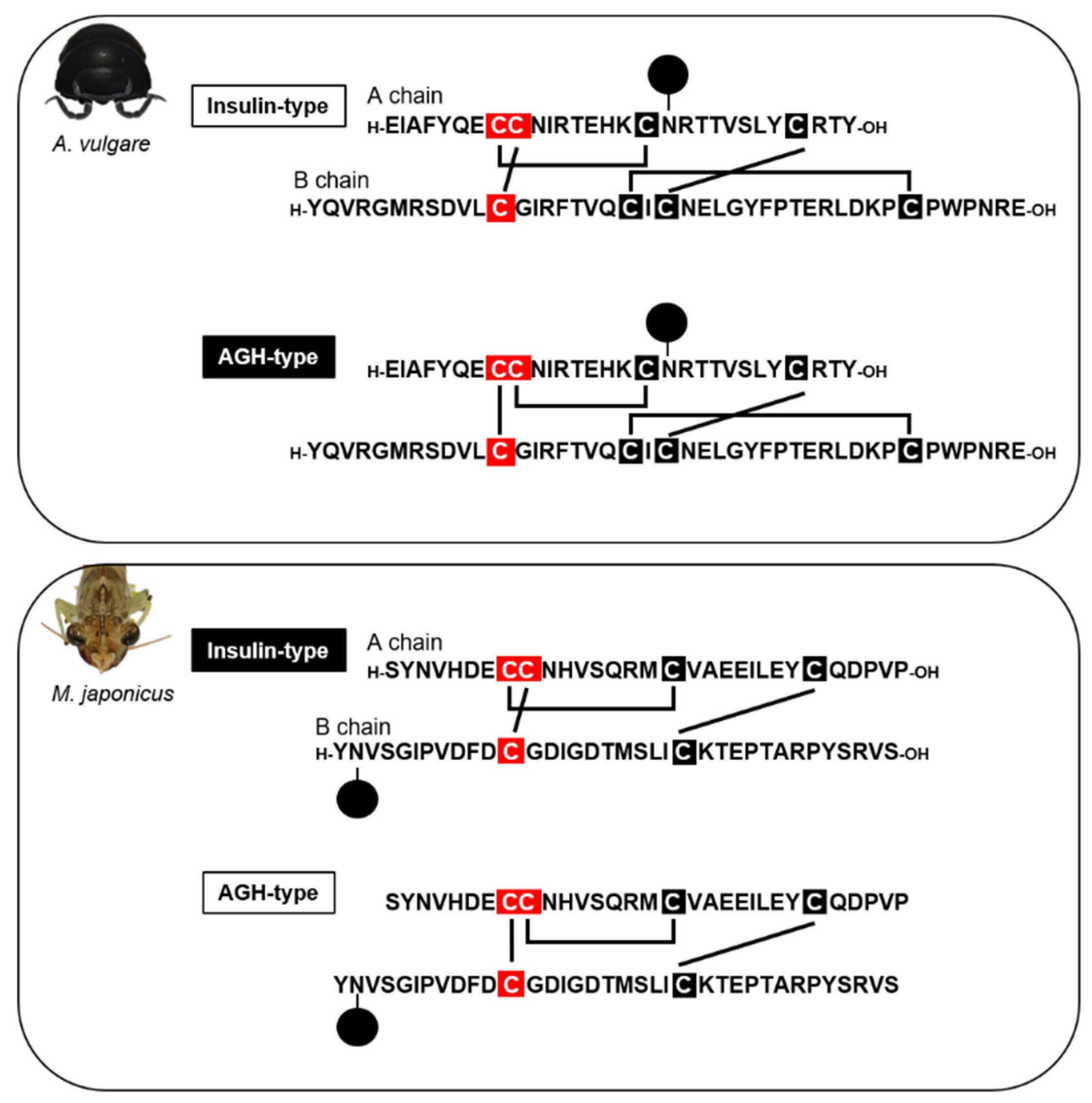
3. Androgenic Gland Factors
4. Crustacean Female Sex Hormone (CFSH)
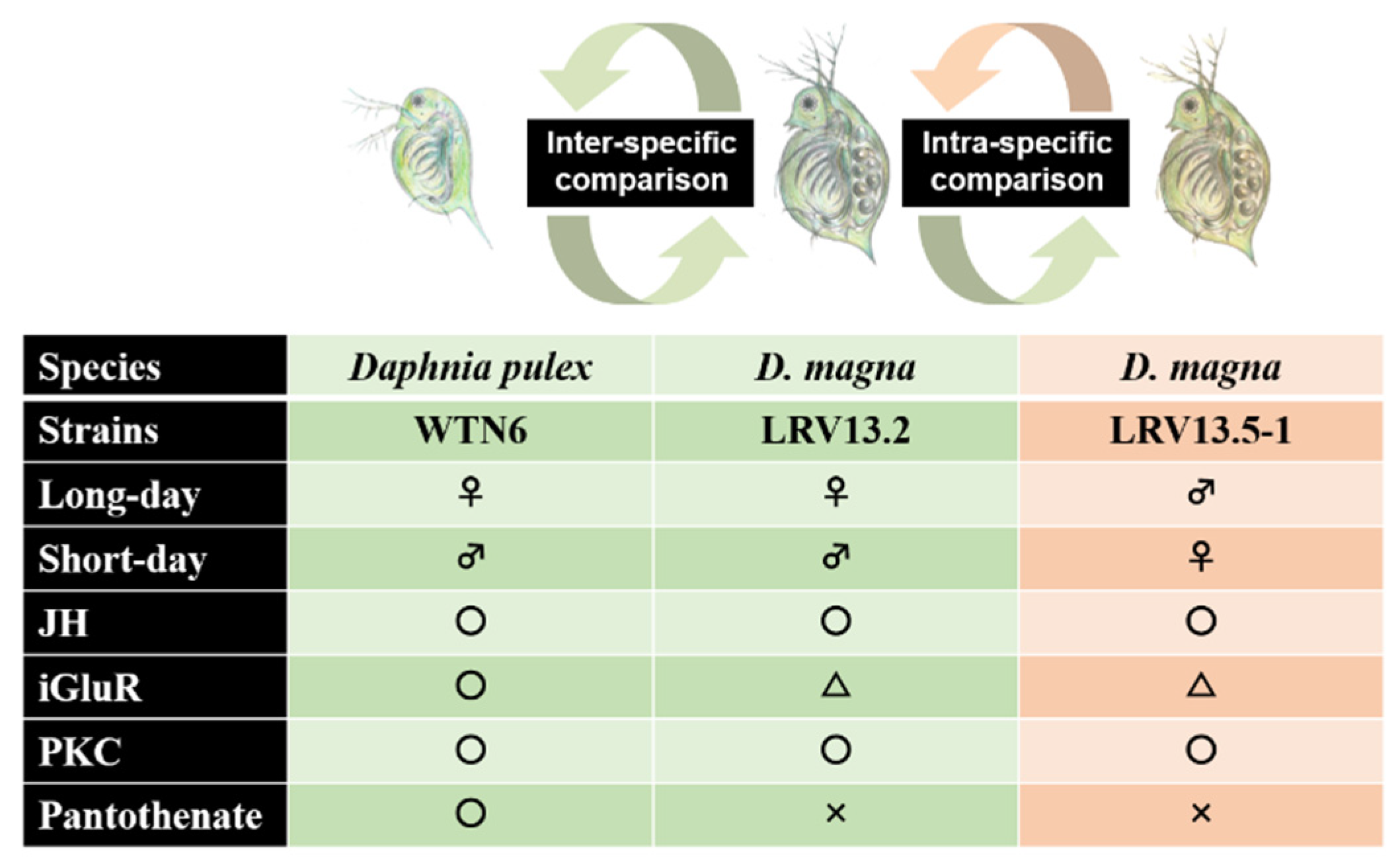
5. Juvenile Hormone as a Male Sex-Determinant in Cladocerans
6. Vertebrate-Type Steroid Hormones
7. Conclusions and Future Directions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Oakley, T.H.; Wolfe, J.M.; Lindgren, A.R.; Zaharoff, A.K. Phylotranscriptomics to bring the understudied into the fold: Monophyletic Ostracoda, fossil placement, and pancrustacean phylogeny. Mol. Biol. Evol. 2013, 30, 215–233. [Google Scholar] [CrossRef] [PubMed]
- Schwentner, M.; Combosch, D.J.; Nelson, J.P.; Giribet, G. A phylogenomic solution to the origin of insects by resolving crustacean-hexapod relationships. Curr. Biol. 2017, 27, 1818–1824. [Google Scholar] [CrossRef]
- Von Reumont, B.M.; Jenner, R.A.; Wills, M.A.; Dell’Ampio, E.; Pass, G.; Ebersberger, I.; Meyer, B.; Koenemann, S.; Iliffe, T.M.; Stamatakis, A.; et al. Pancrustacean phylogeny in the light of new phylogenomic data: Support for remipedia as the possible sister group of Hexapoda. Mol. Biol. Evol. 2012, 29, 1031–1045. [Google Scholar] [CrossRef]
- Gopal, C.; Gopikrishna, G.; Krishna, G.; Jahageerdar, S.S.; Rye, M.; Hayes, B.J.; Paulpandi, S.; Kiran, R.P.; Pillai, S.M.; Ravichandran, P. Weight and time of onset of female-superior sexual dimorphism in pond reared Penaeus monodon. Aquaculture 2010, 300, 237–239. [Google Scholar] [CrossRef]
- Mohanakumaran Nair, C.; Salin, K.R.; Raju, M.S.; Sebastian, M. Economic analysis of monosex culture of giant freshwater prawn (Macrobrachium rosenbergii De Man): A case study. Aquac. Res. 2006, 37, 949–954. [Google Scholar] [CrossRef]
- Chang, E.S.; Sagi, A. Male reproductive hormones. In Reproductive Biology of Crustaceans; Science Publishers: Enfield, UK, 2008. [Google Scholar]
- Zmora, N.; Chung, S. A novel hormone is required for the development of reproductive phenotypes in adult female crabs. Endocrinology 2014, 155, 230–239. [Google Scholar] [CrossRef]
- Manor, R.; Weil, S.; Oren, S.; Glazer, L.; Aflalo, E.D.; Ventura, T.; Chalifa-Caspi, V.; Lapidot, M.; Sagi, A. Insulin and gender: An insulin-like gene expressed exclusively in the androgenic gland of the male crayfish. Gen. Comp. Endocrinol. 2007, 150, 326–336. [Google Scholar] [CrossRef] [PubMed]
- Becking, T.; Giraud, I.; Raimond, M.; Moumen, B.; Chandler, C.; Cordaux, R.; Gilbert, C. Diversity and evolution of sex determination systems in terrestrial Isopods. Sci. Rep. 2017, 7, 1084. [Google Scholar] [CrossRef]
- Juchault, P.; Rigaud, T. Evidence for female heterogamety in two terrestrial crustaceans and the problem of sex chromosome evolution in Isopods. Heredity 1995, 75, 466–471. [Google Scholar] [CrossRef]
- Katakura, Y. Endocrine and genetic control of sex differentiation in the malacostracan Crustacea. Invertebr. Reprod. Dev. 1989, 16, 177–182. [Google Scholar] [CrossRef]
- Levy, T.; Rosen, O.; Eilam, B.; Azulay, D.; Aflalo, E.D.; Manor, R.; Shechter, A.; Sagi, A. A single injection of hypertrophied androgenic gland cells produces all-female aquaculture. Mar. Biotechnol. 2016, 18, 554–563. [Google Scholar] [CrossRef]
- Malecha, S.R.; Nevin, P.A.; Ha, P.; Barck, L.E.; Lamadridrose, Y.; Masuno, S.; Hedgecook, D. Sex-Ratios and sex-determination in progeny from crosses of surgically sex reversed freshwater prawns, Macrobrachium rosenbergii. Aquaculture 1992, 105, 201–218. [Google Scholar] [CrossRef]
- Parnes, S.; Khalaila, I.; Hulata, G.; Sagi, A. Sex determination in crayfish: Are intersex Cherax quadricarinatus (Decapoda, Parastacidae) genetically females? Genet. Res. 2003, 82, 107–116. [Google Scholar] [CrossRef]
- Yu, Y.; Zhang, X.J.; Yuan, J.B.; Wang, Q.C.; Li, S.H.; Huang, H.; Li, F.; Xiang, J. Identification of sex-determining loci in Pacific white shrimp Litopeneaus vannamei using linkage and association analysis. Mar. Biotechnol. 2017, 19, 277–286. [Google Scholar] [CrossRef]
- Chandler, J.C.; Fitzgibbon, Q.P.; Smith, G.; Elizur, A.; Ventura, T. Y-Linked iDmrt1 paralogue (iDMY) in the Eastern spiny lobster, Sagmariasus verreauxi: The first invertebrate sex-linked Dmrt. Dev. Biol. 2017, 430, 337–345. [Google Scholar] [CrossRef] [PubMed]
- Fang, S.; Zhang, Y.; Shi, X.; Zheng, H.; Li, S.; Zhang, Y.; Fazhan, H.; Waiho, K.; Tan, H.; Ikhwanuddin, M.; et al. Identification of male-specific SNP markers and development of PCR-based genetic sex identification technique in crucifix crab (Charybdis feriatus) with implication of an XX/XY sex determination system. Genomics 2019, 112, 404–411. [Google Scholar] [CrossRef] [PubMed]
- Lv, J.J.; Sun, D.F.; Huan, P.P.; Song, L.; Liu, P.; Li, J. QTL mapping and marker identification for sex-determining: Indicating XY sex determination system in the swimming crab (Portunus trituberculatus). Front. Genet. 2018, 9, 337. [Google Scholar] [CrossRef] [PubMed]
- Niiyama, H. The XY chromosomes of the shore-crab, Hemigrapsus sanguineus (de Haan). Jpn. J. Genet. 1938, 14, 34–38. [Google Scholar] [CrossRef]
- Chandler, J.C.; Elizur, A.; Ventura, T. The decapod researcher’s guide to the galaxy of sex determination. Hydrobiologia 2018, 825, 61–80. [Google Scholar] [CrossRef]
- Zhang, L.; Yang, C.; Zhang, Y.; Li, L.; Zhang, X.; Zhang, Q.; Xiang, J. A genetic linkage map of Pacific white shrimp (Litopenaeus vannamei): Sex-linked microsatellite markers and high recombination rates. Genetica 2007, 131, 37–49. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Yuan, J.; Sun, Y.; Li, S.; Gao, Y.; Yu, Y.; Liu, C.; Wang, Q.; Lv, X.; Zhang, X.; et al. Penaeid shrimp genome provides insights into benthic adaptation and frequent molting. Nat. Commun. 2019, 10, 356. [Google Scholar] [CrossRef] [PubMed]
- Sagi, A.; Cohen, D.; Milner, Y. Effect of androgenic gland ablation on morphotypic differentiation and sexual characteristics of male freshwater prawns, Macrobrachium rosenbergii. Gen. Comp. Endocrinol. 1990, 77, 15–22. [Google Scholar] [CrossRef]
- Levy, T.; Rosen, O.; Manor, R.; Dotan, S.; Azulay, D.; Abramov, A.; Sklarz, M.Y.; Chalifa-Caspi, V.; Baruch, K.; Shechter, A.; et al. Production of WW males lacking the masculine Z chromosome and mining the Macrobrachium rosenbergii genome for sex-chromosomes. Sci. Rep. 2019, 9, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Kenny, N.J.; Sin, Y.W.; Shen, X.; Zhe, Q.; Wang, W.; Chan, T.F.; Tobe, S.S.; Shimeld, S.M.; Chu, K.H.; Hui, J.H. Genomic sequence and experimental tractability of a new decapod shrimp model, Neocaridina denticulata. Mar. Drugs 2014, 12, 1419–1437. [Google Scholar] [CrossRef]
- Gutekunst, J.; Andriantsoa, R.; Falckenhayn, C.; Hanna, K.; Stein, W.; Rasamy, J.; Lyko, F. Clonal genome evolution and rapid invasive spread of the marbled crayfish. Nat. Ecol. Evol. 2018, 2, 567. [Google Scholar] [CrossRef]
- Waiho, K.; Shi, X.; Fazhan, H.; Li, S.; Zhang, Y.; Zheng, H.; Liu, W.; Fang, S.; Ikhwanuddin, M.; Ma, H. High-density genetic linkage maps provide novel insights into ZW/ZZ sex determination system and growth performance in mud crab (Scylla paramamosain). Front. Genet. 2019, 5, 298. [Google Scholar] [CrossRef]
- Zhao, M.; Wang, W.; Zhang, F.; Ma, C.; Liu, Z.; Yang, M.H.; Chen, W.; Li, Q.; Cui, M.; Jiang, K.; et al. A chromosome-level genome of the mud crab (Scylla paramamosain Estampador) provides insights into the evolution of chemical and light perception in this crustacean. Mol. Ecol. Resour. 2021. [Google Scholar] [CrossRef] [PubMed]
- Chebbi, M.A.; Becking, T.; Moumen, B.; Giraud, I.; Gilbert, C.; Peccoud, J.; Cordaux, R. The genome of Armadillidium vulgare (Crustacea, Isopoda) provides insights into sex chromosome evolution in the context of cytoplasmic sex determination. Mol. Biol. Evol. 2019, 36, 727–741. [Google Scholar] [CrossRef]
- Kao, D.; Lai, A.G.; Stamataki, E.; Rosic, S.; Konstantinides, N.; Jarvis, E.; Donfrancesco, A.D.; Pouchkina-Stancheva, N.; Semon, M.; Grillo, M.; et al. The genome of the crustacean Parhyale hawaiensis, a model for animal development, regeneration, immunity and lignocellulose digestion. eLife 2016, 5, e20062. [Google Scholar] [CrossRef]
- Naylor, C.; Adams, J.; Greenwood, P. Population dynamics and adaptive sexual strategies in a brackish water crustacean, Gammarus duebeni. J. Anim. Ecol. 1988, 57, 493–507. [Google Scholar] [CrossRef]
- Innes, D.J. Sexual reproduction of Daphnia pulex in a temporary habitat. Oecologia 1997, 111, 53–60. [Google Scholar] [CrossRef]
- Toyota, K.; Miyakawa, H.; Hiruta, C.; Furuta, K.; Ogino, Y.; Shinoda, T.; Tatarazako, N.; Miyagawa, S.; Shaw, J.R.; Iguchi, T. Methyl farnesoate synthesis is necessary for the environmental sex determination in the water flea Daphnia pulex. J. Insect Physiol. 2015, 80, 22–30. [Google Scholar] [CrossRef]
- Colbourne, J.K.; Pfrender, M.E.; Gilbert, D.; Thomas, W.K.; Tucker, A.; Oakley, T.H.; Tokishita, S.; Aerts, A.; Arnold, G.J.; Basu, M.K.; et al. The ecoresponsive genome of Daphnia pulex. Science 2011, 331, 555–561. [Google Scholar] [CrossRef] [PubMed]
- Xu, S.; Ackerman, M.S.; Long, H.; Bright, L.; Spitze, K.; Ramsdell, J.S.; Thomas, W.K.; Lynch, M. A male-specific genetic map of the microcrustacean Daphnia pulex based on single-sperm whole-genome sequencing. Genetics 2015, 201, 31–38. [Google Scholar] [CrossRef]
- Hobæk, A.; Larsson, P. Sex determination in Daphnia magna. Ecology 1990, 71, 2255–2268. [Google Scholar] [CrossRef]
- Kleiven, O.T.; Larsson, P.; Hobæk, A. Sexual reproduction in Daphnia magna requires three stimuli. Oikos 1992, 65, 197–206. [Google Scholar] [CrossRef]
- Lee, B.Y.; Choi, B.S.; Kim, M.S.; Park, J.C.; Jeong, C.B.; Han, J.; Lee, J.S. The genome of the freshwater water flea Daphnia magna: A potential use for freshwater molecular ecotoxicology. Aquat. Toxicol. 2019, 210, 69–84. [Google Scholar] [CrossRef]
- Sassaman, C.; Weeks, S.C. The genetic mechanism of sex determination in the conchostracan shrimp Eulimnadia texana. Am. Nat. 1993, 141, 314–328. [Google Scholar] [CrossRef] [PubMed]
- Baldwin-Brown, J.G.; Weeks, S.C.; Long, A.D. A new standard for crustacean genomes: The highly contiguous, annotated genome assembly of the clam shrimp Eulimnadia texana reveals HOX gene order and identifies the sex chromosome. Genome Biol. Evol. 2018, 10, 143–156. [Google Scholar] [CrossRef]
- Kopp, A. Dmrt genes in the development and evolution of sexual dimorphism. Trends Genet. 2012, 28, 175–184. [Google Scholar] [CrossRef] [PubMed]
- Matson, C.K.; Zarkower, D. Sex and the singular DM domain: Insights into sexual regulation, evolution and plasticity. Nat. Rev. Genet. 2012, 13, 163–174. [Google Scholar] [CrossRef] [PubMed]
- Hildreth, P.E. Doublesex, a recessive gene that transforms both males and females of Drosophila into intersexes. Genetics 1965, 51, 659–678. [Google Scholar] [CrossRef] [PubMed]
- Matsuda, M.; Nagahama, Y.; Shinomiya, A.; Sato, T.; Matsuda, C.; Kobayashi, T.; Morrey, C.E.; Shibata, N.; Asakawa, S.; Shimizu, N.; et al. DMY is a Y-specific DM-domain gene required for male development in the medaka fish. Nature 2002, 417, 559–563. [Google Scholar] [CrossRef]
- Yoshimoto, S.; Okada, E.; Umemoto, H.; Tamura, K.; Uno, Y.; Nishida-Umehara, C.; Matsuda, Y.; Takamatsu, N.; Shiba, T.; Ito, M. A W-linked DM-domain gene, DM-W, participates in primary ovary development in Xenopus laevis. Proc. Natl. Acad. Sci. USA 2008, 105, 2469–2474. [Google Scholar] [CrossRef] [PubMed]
- Smith, C.A.; Roeszler, K.N.; Ohnesorg, T.; Cummins, D.M.; Farlie, P.G.; Doran, T.J.; Sinclair, A.H. The avian Z-linked gene DMRT1 is required for male sex determination in the chicken. Nature 2009, 461, 267–271. [Google Scholar] [CrossRef]
- Gruzin, M.; Mekheal, M.; Ruhlman, K.; Winkowski, M.; Petko, J. Developmental expression of doublesex-related transcripts in the common house spider, Parasteatoda tepidariorum. Gene Expr. Patterns 2020, 35, 119101. [Google Scholar] [CrossRef]
- Chandler, J.C.; Aizen, J.; Fitzgibbon, Q.P.; Elizur, A.; Ventura, T. Applying the power of transcriptomics: Understanding male sexual development in decapod Crustacea. Int. Comp. Biol. 2016, 56, 1144–1156. [Google Scholar] [CrossRef]
- Ventura, T.; Chandler, J.C.; Nguyen, T.V.; Hyde, C.J.; Elizur, A.; Fitzgibbon, Q.P.; Smith, G.G. Multi-Tissue transcriptome analysis identifies key sexual development-related genes of the ornate spiny lobster (Panulirus ornatus). Genes 2020, 11, 1150. [Google Scholar] [CrossRef]
- González-Castellano, I.; Manfin, C.; Pallavicini, A.; Martínez-Lage, A. De novo gonad transcriptome analysis of the common littoral shrimp Palaemon serratus: Novel insights into sex-related genes. BMC Genom. 2019, 20, 757. [Google Scholar] [CrossRef]
- Rotllant, G.; Nguyen, T.V.; Sbragaglia, V.; Rahi, L.; Dudley, K.J.; Hurwood, D.; Ventura, T.; Company, J.B.; Chand, V.; Aguzzi, J.; et al. Sex and tissue specific gene expression patterns identified following de novo transcriptomic analysis of the Norway lobster, Nephrops norvegicus. BMC Genom. 2017, 18, 622. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Miao, G.; Fazhan, H.; Waiho, K.; Zheng, H.; Li, S.; Ikhwaniddin, M.; Ma, H. Transcriptome-seq provides insights into sex-preference pattern of gene expression between testis and ovary of the crucifix crab (Charybdis feriatus). Physiol. Genom. 2018, 50, 393–405. [Google Scholar] [CrossRef]
- Wang, Y.; Yu, Y.; Li, S.; Zhang, X.; Xiang, J.; Li, F. Sex-Specific transcriptome sequencing of zoea I larvae and identification of sex-linked genes using bulked segregant analysis in Pacific white shrimp Litopenaeus vannamei. Mar. Biotechnol. 2020, 22, 423–432. [Google Scholar] [CrossRef]
- Rodríguez, E.M.; Medesani, D.A.; Fingerman, M. Endocrine disruption in crustaceans due to pollutants: A review. Comp. Biochem. Physiol. A Mol. Integr. Physiol. 2007, 146, 661–671. [Google Scholar] [CrossRef]
- Charniaux-Cotton, H. Discovery in an amphipod crustacean (Orchestia gammarella) of an endocrine gland responsible for the differentiation of primary and secondary male sex characteristics. C. R. Acad. Sci. Paris 1954, 239, 780–782. [Google Scholar] [PubMed]
- Olmstead, A.W.; LeBlanc, G.A. Effects of endocrine-active chemicals on the development of sex characteristics of Daphnia magna. Environ. Toxicol. Chem. 2000, 19, 2107–2113. [Google Scholar] [CrossRef]
- Charniaux-Cotton, H. Étude du déterminisme des caractères sexuels secondaires par castration chirurgicale et implantation d’ovaire chez un Crustacé Amphipode (Orchestia gammarella). C. R. Acad. Sci. Paris 1953, 236, 141–143. [Google Scholar]
- Katakura, Y. Transformation of ovary into testis following implantation of androgenic glands in Armadillidium vulgare, an isopod crustacean. Annot. Zool. Jpn. 1960, 33, 241–244. [Google Scholar]
- Suzuki, S. Androgenic gland hormone is a sex-reversing factor but cannot be a sex-determining factor in the female crustacean isopods Armadillidium vulgare. Gen. Comp. Endocrinol. 1999, 115, 370–378. [Google Scholar] [CrossRef] [PubMed]
- Katakura, Y.; Hasegawa, Y. Masculinization of females of the isopod crustacean, Armadillidium vulgare, following injections of an active extract of the androgenic gland. Gen. Comp. Endocrinol. 1983, 49, 57–62. [Google Scholar] [CrossRef]
- Hasegawa, Y.; Haino-Fukushima, K.; Katakura, Y. Isolation and properties of androgenic gland hormone from the terrestrial isopod, Armadillidium vulgare. Gen. Comp. Endocrinol. 1987, 67, 101–110. [Google Scholar] [CrossRef]
- Okuno, A.; Hasegawa, Y.; Nagasawa, H. Purification and properties of androgenic gland hormone from the terrestrial isopod Armadillidium vulgare. Zool. Sci. 1997, 14, 837–842. [Google Scholar] [CrossRef]
- Kato, M.; Hiruta, C.; Tochinai, S. Androgenic gland implantation induces partial masculinization in marmorkrebs Procambarus fallax f. virginalis. Zool. Sci. 2015, 32, 459–464. [Google Scholar] [CrossRef][Green Version]
- Tropea, C.; Hermida, G.N.; Greco, L.S.L. Effects of androgenic gland ablation on growth and reproductive parameters of Cherax quadricarinatus males (Parastacidae, Decapoda). Gen. Comp. Endocrinol. 2011, 174, 211–218. [Google Scholar] [CrossRef] [PubMed]
- Khalaila, I.; Manor, R.; Weil, S.; Granot, Y.; Keller, R.; Sagi, A. The eyestalk-androgenic gland-testis endocrine axis in the crayfish Cherax quadricarinatus. Gen. Comp. Endocrinol. 2002, 127, 147–156. [Google Scholar] [CrossRef]
- Sroyraya, M.; Chotwiwatthanakun, C.; Stewart, M.J.; Soonklang, N.; Kornthong, N.; Phoungpetchara, I.; Hanna, P.J.; Sobhon, P. Bilateral eyestalk ablation of the blue swimmer crab, Portunus pelagicus, produces hypertrophy of the androgenic gland and an increase of cells producing insulin-like androgenic gland hormone. Tissue Cell 2010, 42, 293–300. [Google Scholar] [CrossRef] [PubMed]
- Chung, J.S.; Manor, R.; Sagi, A. Cloning of an insulin-like androgenic gland factor (IAG) from the blue crab, Callinectes sapidus: Implications for eyestalk regulation of IAG expression. Gen. Comp. Endocrinol. 2011, 173, 4–10. [Google Scholar] [CrossRef]
- Li, F.; Bai, H.; Zhang, W.; Fu, H.; Jiang, F.; Liang, G.; Jin, S.; Sun, S.; Qiao, H. Cloning of genomic sequences of three crustacean hyperglycemic hormone superfamily genes and elucidation of their roles of regulating insulin-like androgenic gland hormone gene. Gene 2015, 561, 68–75. [Google Scholar] [CrossRef]
- Rosen, O.; Weil, S.; Manor, R.; Roth, Z.; Khalaila, I.; Sagi, A. A crayfish insulin-like-binding protein: Another piece in the androgenic gland insulin-like hormone puzzle is revealed. J. Biol. Chem. 2013, 288, 22289–22298. [Google Scholar] [CrossRef]
- Chandler, J.C.; Aizen, J.; Elizur, A.; Hollander-Cohen, L.; Battaglene, S.C.; Ventura, T. Discovery of a novel insulin-like peptide and insulin binding proteins in the Eastern rock lobster Sagmariasus verreauxi. Gen. Comp. Endocrinol. 2015, 215, 76–87. [Google Scholar] [CrossRef]
- Aizen, J.; Chandler, J.C.; Fitzgibbon, Q.P.; Sagi, A.; Battaglene, S.C.; Elizur, A.; Ventura, T. Production of recombinant insulin-like androgenic gland hormones from three decapod species: In vitro testicular phosphorylation and activation of a newly identified tyrosine kinase receptor from the Eastern spiny lobster, Sagmariasus verreauxi. Gen. Comp. Endocrinol. 2016, 229, 8–18. [Google Scholar] [CrossRef]
- Li, S.; Li, F.; Yu, K.; Xiang, J. Identification and characterization of a doublesex gene which regulates the expression of insulin-like androgenic gland hormone in Fenneropenaeus chinensis. Gene 2018, 649, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Zheng, J.; Cai, L.; Jia, Y.; Chi, M.; Cheng, S.; Liu, S.; Gu, Z. Identification and functional analysis of the doublesex gene in the redclaw crayfish, Cherax quadricarinatus. BMC Dev. Biol. 2020. [Google Scholar] [CrossRef]
- Levy, T.; Sagi, A. The “IAG-switch”—A key controlling element in decapod crustacean sex differentiation. Front. Endocrinol. 2020, 11, 651. [Google Scholar] [CrossRef] [PubMed]
- Ge, H.-L.; Tan, K.; Shi, L.-L.; Sun, R.; Wang, W.-M.; Li, Y.-H. Comparison of effects of dsRNA and siRNA RNA interference on insulin-like androgenic gland gene (IAG) in red swamp crayfish Procambarus clarkii. Gene 2020, 752, 144783. [Google Scholar] [CrossRef]
- Rosen, O.; Manor, R.; Weil, S.; Gafni, O.; Afalo, E.D.; Ventura, T.; Sagi, A. A sexual shift induced by silencing of a single insulin-like gene in crayfish: Ovarian upregulation and testicular degeneration. PLoS ONE 2010, 5, e15281. [Google Scholar] [CrossRef]
- Ventura, T.; Manor, R.; Afalo, E.D.; Weil, S.; Raviv, S.; Glazer, L.; Sagi, A. Temporal silencing of an androgenic gland-specific insulin-like gene affecting phenotypical gender differences and spermatogenesis. Endocrinology 2009, 150, 1278–1286. [Google Scholar] [CrossRef]
- Ventura, T.; Manor, R.; Afalo, E.D.; Weil, A.; Rosen, O.; Sagi, A. Timing sexual differentiation: Full functional sex reversal achieved through silencing of a single insulin-like gene in the prawn, Macrobrachium rosenbergii. Biol. Reprod. 2012, 86, 90. [Google Scholar] [CrossRef]
- Katayama, H. Structure-Activity relationship of crustacean peptide hormones. Biosci. Biotechnol. Biochem. 2016, 80, 633–641. [Google Scholar] [CrossRef]
- Katayama, H.; Hojo, H.; Ohira, T.; Ishii, A.; Nozaki, T.; Goto, K.; Nakahara, Y.; Takahashi, T.; Hasegawa, Y.; Nagasawa, H.; et al. Correct disulfide pairing is required for the biological activity of crustacean androgenic gland hormone (AGH): Synthetic studies of AGH. Biochemistry 2010, 49, 1798–1807. [Google Scholar] [CrossRef]
- Katayama, H.; Kubota, N.; Hojo, H.; Okada, A.; Kotaka, S.; Tsutsui, N.; Ohira, T. Direct evidence for the function of crustacean insulin-like androgenic gland factor (IAG): Total chemical synthesis of IAG. Bioorg. Med. Chem. 2014, 22, 5783–5789. [Google Scholar] [CrossRef] [PubMed]
- Banzai, K.; Izumi, S.; Ohira, T. Molecular cloning and expression analysis of cDNAs encoding an insulin-like androgenic gland factor from three palaemonid species, Macrobrachum lar, Palaemon paucidens and P. pacificus. Jpn. Agric. Res. Q. 2012, 46, 105–114. [Google Scholar] [CrossRef][Green Version]
- Katayama, H.; Mukainakano, T.; Kogure, J.; Ohira, T. Chemical synthesis of the crustacean insulin-like peptide with four disulfide bonds. J. Pept. Sci. 2018, 24, e3132. [Google Scholar] [CrossRef]
- Katayama, H.; Hiromichi, N. Chemical synthesis of N-glycosylated insulin-like androgenic gland factor from the freshwater prawn Macrobrachium rosenbergii. J. Pept. Sci. 2019, 25, e3215. [Google Scholar] [CrossRef] [PubMed]
- Veenstra, J.A. The power of next-generation sequencing as illustrated by the neuropeptidome of the crayfish Procambarus clarkii. Gen. Comp. Endocrinol. 2015, 224, 84–95. [Google Scholar] [CrossRef]
- Veenstra, J.A. Similarities between decapod and insect neuropeptidomes. PeerJ 2016, 4, e2043. [Google Scholar] [CrossRef]
- Oliphant, A.; Alexander, J.L.; Swain, M.T.; Webster, S.G.; Wilcockson, D.C. Transcriptomic analysis of crustacean neuropeptide signaling during the moult cycle in the green shore crab, Carcinus maenas. BMC Genom. 2018, 19, 711. [Google Scholar] [CrossRef] [PubMed]
- Liu, A.; Liu, J.; Liu, F.; Huang, Y.; Wang, G.; Ye, H. Crustacean female sex hormone from the mud crab Scylla paramamosain is highly expressed in prepubertal males and inhibits the development of androgenic gland. Front. Physiol. 2018, 9, 924. [Google Scholar] [CrossRef]
- Jiang, Q.; Lu, B.; Lin, D.; Huang, H.; Chen, X.; Ye, H. Role of crustacean female sex hormone (CFSH) in sex differentiation in early juvenile mud crabs, Scylla paramamosain. Gen. Comp. Endocrinol. 2020, 289, 113383. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Q.; Lu, B.; Wang, G.; Ye, H. Transcriptional inhibition of Sp-IAG by crustacean female sex hormone in the mud crab, Scylla paramamosain. Int. J. Mol. Sci. 2020, 21, 5300. [Google Scholar] [CrossRef]
- Kotaka, S.; Ohira, T. cDNA cloning and in situ localization of a crustacean female sex hormone‑like molecule in the kuruma prawn Marsupenaeus japonicus. Fish. Sci. 2018, 84, 53–60. [Google Scholar] [CrossRef]
- Powell, D.; Knibb, W.; Remilton, C.; Elizur, A. De-Novo transcriptome analysis of the banana shrimp (Fenneropenaeus merguiensis) and identification of genes associated with reproduction and development. Mar. Genom. 2015, 22, 71–78. [Google Scholar] [CrossRef] [PubMed]
- Toullec, J.; Corre, E.; Mandon, P.; Gonzalez-Aravena, M.; Ollivaux, C. Characterization of the neuropeptidome of a Southern Ocean decapod, the Antarctic shrimp Chorismus antarcticus: Focusing on a new decapod ITP-like peptide belonging to the CHH peptide family. Gen. Comp. Endocrinol. 2017, 252, 60–78. [Google Scholar] [CrossRef] [PubMed]
- Ventura, T.; Cummins, S.F.; Fitzgibbon, Q.; Battaglene, S.; Elizur, A. Analysis of the central nervous system transcriptome of the Eastern rock lobster Sagmariasus verreauxi reveals its putative neuropeptidome. PLoS ONE 2014, 9, e97323. [Google Scholar] [CrossRef]
- Suwansaard, S.; Thongbuakaew, T.; Wang, T.; Zhao, M.; Elizur, A.; Hanna, P.J.; Sretarugsa, P.; Commins, S.F.; Sobhon, P. In silico neuropeptidome of female Macrobrachium rosenbergii based on transcriptome and peptide mining of eyestalk, central nervous system and ovary. PLoS ONE 2015, 10, e0123848. [Google Scholar] [CrossRef]
- Thongbuakaew, T.; Suwansa-ard, S.; Sretarugsa, P.; Sobhon, P.; Cummins, S.F. Identification and characterization of a crustacean female sex hormone in the giant freshwater prawn, Macrobrachium rosenbergii. Aquaculture 2019, 507, 56–68. [Google Scholar] [CrossRef]
- Nguyen, T.V.; Cummins, S.F.; Elizur, A.; Ventura, T. Transcriptomic characterization and curation of candidate neuropeptides regulating reproduction in the eyestalk ganglia of the Australian crayfish, Cherax quadricarinatus. Sci. Rep. 2016, 6, 38658. [Google Scholar] [CrossRef]
- Tsutsui, N.; Kotaka, S.; Ohira, T.; Sakamoto, T. Characterization of distinct ovarian isoform of crustacean female sex hormone in the kuruma prawn Marsupenaeus japonicus. Comp. Biochem. Physiol. Part A 2018, 217, 7–16. [Google Scholar] [CrossRef] [PubMed]
- Nijhout, H.F. Insect Hormones; Princeton University Press: Princeton, NJ, USA, 1994. [Google Scholar]
- Sugime, Y.; Oguchi, K.; Gotoh, H.; Hayashi, Y.; Matsunami, M.; Shigenobu, S.; Koshikawa, S.; Miura, T. Termite soldier mandibles are elongated by dachshund under hormonal and Hox gene controls. Development 2019, 146, dev171942. [Google Scholar] [CrossRef]
- Gotoh, H.; Cornette, R.; Koshikawa, S.; Okada, Y.; Lavine, L.C.; Emlen, D.J.; Miura, T. Juvenile hormone regulates extreme mandible growth in male stag beetles. PLoS ONE 2011, 6, e21139. [Google Scholar] [CrossRef]
- Ishikawa, A.; Ogawa, K.; Gotoh, H.; Walsh, T.K.; Tagu, D.; Brisson, J.A.; Rispe, C.; Jaubert-Possamai, S.; Kanbe, T.; Tsubota, T.; et al. Juvenile hormone titre and related gene expression during the change of reproductive modes in the pea aphid. Insect Mol. Biol. 2012, 27, 49–60. [Google Scholar] [CrossRef] [PubMed]
- Miyakawa, H.; Toyota, K.; Sumiya, E.; Iguchi, T. Comparison of JH signaling in insects and crustaceans. Curr. Opin. Insect Sci. 2014, 1, 81–87. [Google Scholar] [CrossRef]
- Miyakawa, H.; Sato, T.; Song, Y.; Tollefsen, K.E.; Iguchi, T. Ecdysteroid and juvenile hormone biosynthesis, receptors and their signaling in the freshwater microcrustacean Daphnia. J. Steroid Biochem. Mol. Biol. 2018, 184, 62–68. [Google Scholar] [CrossRef] [PubMed]
- Laufer, H.; Borst, D.; Baker, F.C.; Reuter, C.C.; Tsai, L.W.; Schooley, D.A.; Carrasco, C.; Sinkus, M. Identification of a juvenile hormone-like compound in a crustacean. Science 1987, 235, 202–205. [Google Scholar] [CrossRef]
- Tobe, S.S.; Young, D.A.; Khoo, H.W. Production of methyl farnesoate by the mandibular organs of the mud crab, Scylla serrate: Validation of a radiochemical assay. Gen. Comp. Endocrinol. 1989, 73, 342–353. [Google Scholar] [CrossRef]
- Laufer, H.; Biggers, W.J. Unifying concepts learned from methyl farnesoate for invertebrate reproduction and post-embryonic development. Am. Zool. 2001, 41, 442–457. [Google Scholar] [CrossRef]
- Nagaraju, G.P.C. Is methyl farnesoate a crustacean hormone? Aquaculture 2007, 272, 39–54. [Google Scholar] [CrossRef]
- Homola, E.; Chang, E.S. Methyl farnesoate: Crustacean juvenile hormone in search of functions. Comp. Biochem. Physiol. 1997, 117, 347–356. [Google Scholar] [CrossRef]
- Toyota, K.; Yamane, F.; Ohira, T. Impacts of methyl farnesoate and 20-hydroxyecdysone on larval mortality and metamorphosis in the kuruma prawn Marsupenaeus japonicus. Front. Endocrinol. 2020, 11, 475. [Google Scholar] [CrossRef] [PubMed]
- Hebert, P.D.N. Niche overlap among species in the Daphnia carinata complex. J. Anim. Ecol. 1977, 46, 399–409. [Google Scholar] [CrossRef]
- Banta, A.M.; Brown, L.A. Control of sex in Cladocera. II. The unstable nature of the excretory products involved in male production. Physiol. Zool. 1929, 2, 93–98. [Google Scholar] [CrossRef]
- Smith, G. The life-cycle of Cladocera, with remarks on the physiology of growth and reproduction in crustacea. Proc. R. Soc. Lond. B Biol. Sci. 1915, 88, 418–435. [Google Scholar] [CrossRef]
- Barton, N.H.; Charlesworth, B. Why sex and recombination? Science 1998, 281, 1986–1990. [Google Scholar] [CrossRef]
- Abe, R.; Watanabe, H.; Yamamuro, M.; Iguchi, T.; Tatarazako, N. Establishment of a short-term, in vivo screening method for detecting chemicals with juvenile hormone activity using adult Daphnia magna. J. Appl. Toxicol. 2015, 35, 75–82. [Google Scholar] [CrossRef]
- Abe, R.; Toyota, K.; Miyakawa, H.; Watanabe, H.; Oka, T.; Miyagawa, S.; Nishide, H.; Uchiyama, I.; Tollefsen, K.E.; Iguchi, T.; et al. Diofenolan induces male offspring production through binding to the juvenile hormone receptor in Daphnia magna. Aquat. Toxicol. 2015, 159, 44–51. [Google Scholar] [CrossRef]
- Oda, S.; Tatarazako, N.; Watanabe, H.; Morita, M.; Iguchi, T. Production of male neonates in four cladoceran species exposed to a juvenile hormone analog, fenoxycarb. Chemosphere 2005, 60, 74–78. [Google Scholar] [CrossRef]
- Oda, S.; Tatarazako, N.; Watanabe, H.; Morita, M.; Iguchi, T. Production of male neonates in Daphnia magna (Cladocera, Crustacea) exposed to juvenile hormones and their analogs. Chemosphere 2005, 61, 1168–1174. [Google Scholar] [CrossRef] [PubMed]
- Oda, S.; Tatarazako, N.; Watanabe, H.; Morita, M.; Iguchi, T. Genetic differences in the production of male neonates in Daphnia magna exposed to juvenile hormone analogs. Chemosphere 2006, 63, 1477–1484. [Google Scholar] [CrossRef]
- Olmstead, A.W.; LeBlanc, G.A. Juvenoid hormone methyl farnesoate is a sex determinant in the crustacean Daphnia magna. J. Exp. Zool. 2002, 293, 736–739. [Google Scholar] [CrossRef] [PubMed]
- Tatarazako, N.; Oda, S.; Watanabe, H.; Morita, M.; Iguchi, T. Juvenile hormone agonists affect the occurrence of male Daphnia. Chemosphere 2003, 53, 827–833. [Google Scholar] [CrossRef]
- Kim, K.; Kotov, A.A.; Taylor, D.J. Hormonal induction of undescribed males resolves cryptic species of cladocerans. Proc. R. Soc. B 2006, 273, 141–147. [Google Scholar] [CrossRef] [PubMed]
- Sinev, A.Y.; Sanoamuang, L. Hormonal induction of males as a method for studying tropical cladocerans: Description of males of four chydorid species (Cladocera: Anomopoda: Chydoridae). Zootaxa 2011, 2826, 45–56. [Google Scholar] [CrossRef]
- Toyota, K.; Kato, Y.; Sato, M.; Sugiura, N.; Miyagawa, S.; Miyakawa, H.; Watanabe, H.; Oda, S.; Ogino, Y.; Hiruta, C.; et al. Molecular cloning of doublesex genes of four cladocera (water flea) species. BMC Genom. 2013, 14, 239. [Google Scholar] [CrossRef] [PubMed]
- Miyakawa, H.; Toyota, K.; Hirakawa, I.; Ogino, Y.; Miyagawa, S.; Oda, S.; Tatarazako, N.; Miura, T.; Colbourne, J.K.; Iguchi, T. A mutation in the receptor Methoprene-tolerant alters juvenile hormone response in insects and crustaceans. Nat. Commun. 2013, 4, 1856. [Google Scholar] [CrossRef] [PubMed]
- Miyakawa, H.; Iguchi, T. Comparative luciferase assay for establishing reliable in vitro screening system of juvenile hormone agonists. J. Appl. Toxicol. 2017, 37, 1082–1090. [Google Scholar] [CrossRef]
- Tanaka, T.; Iguchi, T.; Miyakawa, H. Establishment of a high-sensitivity reporter system in mammalian cells for detecting juvenoids using juvenile hormone receptors of Daphnia pulex. J. Appl. Toxicol. 2019, 39, 241–246. [Google Scholar] [CrossRef] [PubMed]
- Hirano, M.; Toyota, K.; Ishibashi, H.; Tominaga, N.; Sato, T.; Tatarazako, N.; Iguchi, T. Molecular insights into structural and ligand binding features of methoprene-tolerant in daphnids. Chem. Res. Toxicol. 2020, 33, 2785–2792. [Google Scholar] [CrossRef] [PubMed]
- Eads, B.D.; Colbourne, J.K.; Bohuski, E.; Andrews, J. Profiling sex-biased gene expression during parthenogenetic reproduction in Daphnia pulex. BMC Genom. 2007, 8, 464. [Google Scholar] [CrossRef] [PubMed]
- Kato, Y.; Kobayashi, K.; Watanabe, H.; Iguchi, T. Environmental sex determination in the branchiopod crustacean Daphnia magna: Deep conservation of a doublesex gene in the sex-determining pathway. PLoS Genet. 2011, 7, e1001345. [Google Scholar] [CrossRef] [PubMed]
- Ishak, N.S.M.; Nong, Q.D.; Matsuura, Y.; Kato, Y.; Watanabe, H. Co-Option of the bZIP transcription factor Vrille as the activator of Doublesex1 in environmental sex determination of the crustacean Daphnia magna. PLoS Genet. 2017, 13, e1006953. [Google Scholar] [CrossRef]
- Kato, Y.; Perez, C.A.G.; Ishak, N.S.M.; Nong, Q.D.; Sudo, Y.; Matsuura, T.; Wada, T.; Watanabe, H. A 5′UTR-overlapping lncRNA activates the male-determining gene doublesex1 in the crustacean Daphnia magna. Curr. Biol. 2018, 28, 1811–1817. [Google Scholar] [CrossRef]
- Toyota, K.; Miyakawa, H.; Yamaguchi, K.; Shigenobu, S.; Ogino, Y.; Tatarazako, N.; Miyagawa, S.; Iguchi, T. NMDA receptor activation upstream of methyl farnesoate signaling for short day-induced male offspring production in the water flea, Daphnia pulex. BMC Genom. 2015, 16, 186. [Google Scholar] [CrossRef]
- Toyota, K.; Sato, T.; Tatarazako, N.; Iguchi, T. Protein kinase C is involved with upstream signaling of methyl farnesoate for photoperiod-dependent sex determination in the water flea Daphnia pulex. Biol. Open 2017, 6, 161–164. [Google Scholar] [CrossRef]
- Lan, J.; Skeberdis, V.A.; Jover, T.; Grooms, S.Y.; Lin, Y.; Araneda, R.C.; Zheng, X.; Bennett, M.V.L.; Zukin, R.S. Protein kinase C modulates NMDA receptor trafficking and gating. Nat. Neurosci. 2001, 4, 382–390. [Google Scholar] [CrossRef]
- Toyota, K.; Gavin, A.; Miyagawa, S.; Viant, M.R.; Iguchi, T. Metabolomics reveals an involvement of pantothenate for male production responding to the short-day stimulus in the water flea, Daphnia pulex. Sci. Rep. 2016, 6, 25125. [Google Scholar] [CrossRef] [PubMed]
- Toyota, K.; Cuenca, M.C.; Dhandapani, V.; Suppa, A.; Rossi, V.; Colbourne, J.K.; Orsini, L. Transgenerational response to early spring warming in Daphnia. Sci. Rep. 2019, 9, 4449. [Google Scholar] [CrossRef] [PubMed]
- Toyota, K.; Sato, T.; Iguchi, T.; Ohira, T. Methyl farnesoate regulatory mechanisms underlying photoperiod-dependent sex determination in the freshwater crustacean Daphnia magna. J. Appl. Toxicol. 2021, 41, 216–223. [Google Scholar] [CrossRef]
- Song, Y.; Villeneuve, D.L.; Toyota, K.; Iguchi, T.; Tollefsen, K.E. Ecdysone receptor agonism leading to lethal molting disruption in arthropods: Review and adverse outcome pathway development. Environ. Sci. Technol. 2017, 51, 4142–4157. [Google Scholar] [CrossRef]
- Gersch, M.; Eibisch, H.; Bohm, G.A.; Koolman, J. Ecdysteroid production by the cephalic gland of the crayfish Orconectes limosus. Gen. Comp. Endocrinol. 1979, 39, 505–511. [Google Scholar] [CrossRef]
- Passano, L.M.; Jyssum, S. The role of the Y-organ in crab proecdysis and limb regeneration. Comp. Biochem. Physiol. 1963, 9, 195–213. [Google Scholar] [CrossRef]
- Shyamal, S.; Das, S.; Guruacharya, A.; Mykles, D.L.; Durica, D.S. Transcriptomic analysis of crustacean molting gland (Y-organ) regulation via the mTOR signaling pathway. Sci. Rep. 2018, 8, 7307. [Google Scholar] [CrossRef] [PubMed]
- Yano, I. Induced ovarian maturation and spawning in greasyback shrimp, Metapenaeus ensis, by progesterone. Aquaculture 1985, 47, 223–229. [Google Scholar] [CrossRef]
- Yano, I. Effect of 17α-hydroxy-progesterone on vitellogenin secretion in kuruma prawn, Penaeus japonicus. Aquaculture 1987, 61, 49–57. [Google Scholar] [CrossRef]
- Okumura, T.; Sakiyama, K. Hemolymph levels of vertebrate-type steroid hormones in female kuruma prawn Marsupenaeus japonicus (Crustacea: Decapoda: Penaeidae) during natural reproductive cycle and induced ovarian development by eyestalk ablation. Fish. Sci. 2004, 70, 372–380. [Google Scholar] [CrossRef]
- Sarojini, S. Comparison of the effects of androgenic hormone and testosterone propionate on the female ocypod crab. Curr. Sci. 1963, 32, 411–412. [Google Scholar]
- Watts, M.M.; Pascoe, D.; Carroll, K. Population responses of the freshwater amphipod Gammarus pulex (L.) to an environmental estrogen, 17α-ethinylestradiol. Environ. Toxicol. Chem. 2002, 21, 445–450. [Google Scholar] [CrossRef] [PubMed]
- Sugestya, I.N.G.; Widodo, M.S.; Soeprijanto, A. Effect of 17β-estradiol on feminization, growth rate and survival rate of pacific white shrimp (Litopenaeus vannamei, Boone 1931) postlarvae. J. Exp. Life Sci. 2018, 8, 37–42. [Google Scholar] [CrossRef]
- Lin, J.; Shi, X.; Fang, S.; Zhang, Y.; You, C.; Ma, H.; Lin, F. Comparative transcriptome analysis combining SMRT and NGS sequencing provides novel insights into sex differentiation and development in mud crab (Scylla paramamosain). Aquaculture 2019, 513, 734447. [Google Scholar] [CrossRef]
- Sumiya, E.; Ogino, Y.; Miyakawa, H.; Hiruta, C.; Toyota, K.; Miyagawa, S.; Iguchi, T. Roles of ecdysteroids for progression of reproductive cycle in the fresh water crustacean Daphnia magna. Front. Zool. 2014, 11, 60. [Google Scholar] [CrossRef]
- Sumiya, E.; Ogino, Y.; Toyota, K.; Miyakawa, H.; Miyagawa, S.; Iguchi, T. Neverland regulates embryonic moltings through the regulation of ecdysteroid synthesis in the water flea Daphnia magna, and may thus act as a target for chemical disruption of molting. J. Appl. Toxicol. 2016, 36, 1476–1485. [Google Scholar] [CrossRef]
- Baldwin, W.S.; Milam, D.L.; LeBlanc, G.A. Physiological and biochemical perturbations in Daphnia magna following exposure to the model environmental estrogen diethylstilbestrol. Environ. Toxicol. Chem. 1995, 14, 945–952. [Google Scholar] [CrossRef]
- Clubbs, R.L.; Brooks, B.W. Daphnia magna responses to a vertebrate estrogen receptor agonist and an antagonist: A multigenerational study. Ecotoxicol. Environ. Saf. 2007, 67, 385–398. [Google Scholar] [CrossRef] [PubMed]
- Kashian, D.R.; Dodson, S.I. Effects of vertebrate hormones on development and sex determination in Daphnia magna. Environ. Toxicol. Chem. 2004, 23, 1282–1288. [Google Scholar] [CrossRef]
- LeBlanc, G.A.; McLachlan, J.B. Molt-Independent growth inhibition of Daphnia magna by a vertebrate antiandrogen. Environ. Toxicol. 1999, 18, 1450–1455. [Google Scholar] [CrossRef]
- Tatarazako, N.; Takao, Y.; Kishi, K.; Onikura, N.; Arizono, K.; Iguchi, T. Styrene dimers and trimers affect reproduction of daphnid (Ceriodaphnia dubia). Chemosphere 2002, 48, 597–601. [Google Scholar] [CrossRef]
- Zou, E.; Fingerman, M. Synthetic estrogenic agents do not interfere with sex differentiation but do inhibit molting of the cladoceran Daphnia magna. Bull. Environ. Contam. Toxicol. 1997, 58, 596–602. [Google Scholar] [CrossRef] [PubMed]
- OECD. Guidelines for testing of chemicals. In Daphnia magna Reproduction Test; OECD: Paris, France, 2012. [Google Scholar]
- Kato, Y.; Kobayashi, K.; Oda, S.; Tatarazako, N.; Watanabe, H.; Iguchi, T. Cloning and characterization of the ecdysone receptor and ultraspiracle protein from the water flea Daphnia magna. J. Endocrinol. 2007, 193, 183–194. [Google Scholar] [CrossRef]
- Evenseth, L.M.; Kristiansen, K.; Song, Y.; Tollefsen, K.E.; Sylte, I. In silico site-directed mutagenesis of the Daphnia magna ecdysone receptor identifies critical amino acids for species-specific and inter-species differences in agonist binding. Comput. Toxicol. 2019, 12, 100091. [Google Scholar] [CrossRef]
- Abayed, F.A.A.; Manor, R.; Aflalo, E.D.; Sagi, A. Screening for Dmrt genes from embryo to mature Macrobrachium rosenbergii prawns. Gen. Comp. Endocrinol. 2019, 282, 113205. [Google Scholar] [CrossRef]
- Zhong, P.; Zhou, T.; Zhang, Y.; Chen, Y.; Yi, J.; Lin, W.; Guo, Z.; Xu, A.; Yang, S.; Chan, S.; et al. Potential involvement of a DMRT family member (Mr-Dsx) in the regulation of sexual differentiation and moulting in the giant river prawn Macrobrachium rosenbergii. Aquac. Res. 2019, 50, 3037–3049. [Google Scholar] [CrossRef]
- Sun, D.A.; Patel, N.H. The amphipod crustacean Parhyale hawaiensis: An emerging comparative model of arthropod development, evolution, and regeneration. WIREs Dev. Biol. 2019, 8, e355. [Google Scholar] [CrossRef]
- Toyota, K.; Hiruta, C.; Ogino, Y.; Miyagawa, S.; Okamura, T.; Onishi, Y.; Tatarazako, N.; Ighuchi, T. Comparative developmental staging of female and male water fleas Daphnia pulex and Daphnia magna during embryogenesis. Zool. Sci. 2016, 33, 31–37. [Google Scholar] [CrossRef] [PubMed]
- Bruce, H.S.; Patel, N.H. Knockout of crustacean leg patterning genes suggests that insect wings and body walls evolved from ancient leg segments. Nat. Ecol. Evol. 2020, 4, 1703–1712. [Google Scholar] [CrossRef]
- Clark-Hachtel, C.M.; Tomoyasu, Y. Two sets of candidate crustacean wing homologues and their implication for the origin of insect wings. Nat. Ecol. Evol. 2020, 4, 1694–1702. [Google Scholar] [CrossRef] [PubMed]
- Toyota, K.; Miyagawa, S.; Ogino, Y.; Iguchi, T. Microinjection-based RNA interference method in the water flea, Daphnia pulex and Daphnia magna. In RNA Interference; InTech: London, UK, 2016. [Google Scholar] [CrossRef]
- Mykles, D.L.; Hui, J.H. Neocaridina denticulata: A decapod crustacean model for functional genomics. Int. Comp. Biol. 2015, 55, 891–897. [Google Scholar] [CrossRef] [PubMed]
- Sin, Y.W.; Kenny, N.J.; Qu, Z.; Chan, K.W.; Chan, K.W.S.; Cheong, S.P.S.; Leung, R.W.T.; Chan, T.F.; Bendena, W.G.; Chu, K.H.; et al. Identification of putative ecdysteroid and juvenile hormone pathway genes in the shrimp Neocaridina denticulata. Gen. Comp. Endocrinol. 2015, 214, 167–176. [Google Scholar] [CrossRef]
| Species | Taxonomy | Sex Determination Manner | Draft or Complete Genome |
|---|---|---|---|
| Pacific white shrimp Litopenaeus vannamei | Malacostraca Decapoda | GSD with ZZ/ZW [21] | [22] |
| Giant freshwater prawn Macrobrachium rosenbergii | Malacostraca Decapoda | GSD with ZZ/ZW [23] | [24] |
| Cherry shrimp Neocaridina denticulate | Malacostraca Decapoda | Not available | [25] |
| Marbled crayfish Procambarus fallax f. virginalis | Malacostraca Decapoda | GSD (no male has reported) | [26] |
| Mud crab Scylla paramamosain | Malacostraca Decapoda | GSD with ZZ/ZW [27] | [28] |
| Wood louse Armadillidium vulgare | Malacostraca Isopoda | GSD with ZZ/ZW [9,10] | [29] |
| Parhyale hawaiensis | Malacostraca Amphipoda | Not available | [30] |
| Gammarus duebeni | Malacostraca Amphipoda | ESD [31] | Not available |
| Water flea Daphnia pulex | Branchiopoda Cladocera | ESD [32,33] | [34,35] |
| Water flea Daphnia magna | Branchiopoda Cladocera | ESD [36,37] | [38] |
| Clam shrimp Eulimnadia texana | Branchiopoda Spinicaudata | GSD with androdioecious (male and hermaphrodite) [39] | [40] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Toyota, K.; Miyakawa, H.; Hiruta, C.; Sato, T.; Katayama, H.; Ohira, T.; Iguchi, T. Sex Determination and Differentiation in Decapod and Cladoceran Crustaceans: An Overview of Endocrine Regulation. Genes 2021, 12, 305. https://doi.org/10.3390/genes12020305
Toyota K, Miyakawa H, Hiruta C, Sato T, Katayama H, Ohira T, Iguchi T. Sex Determination and Differentiation in Decapod and Cladoceran Crustaceans: An Overview of Endocrine Regulation. Genes. 2021; 12(2):305. https://doi.org/10.3390/genes12020305
Chicago/Turabian StyleToyota, Kenji, Hitoshi Miyakawa, Chizue Hiruta, Tomomi Sato, Hidekazu Katayama, Tsuyoshi Ohira, and Taisen Iguchi. 2021. "Sex Determination and Differentiation in Decapod and Cladoceran Crustaceans: An Overview of Endocrine Regulation" Genes 12, no. 2: 305. https://doi.org/10.3390/genes12020305
APA StyleToyota, K., Miyakawa, H., Hiruta, C., Sato, T., Katayama, H., Ohira, T., & Iguchi, T. (2021). Sex Determination and Differentiation in Decapod and Cladoceran Crustaceans: An Overview of Endocrine Regulation. Genes, 12(2), 305. https://doi.org/10.3390/genes12020305






