The Sex Determination Cascade in the Silkworm
Abstract
1. Introduction
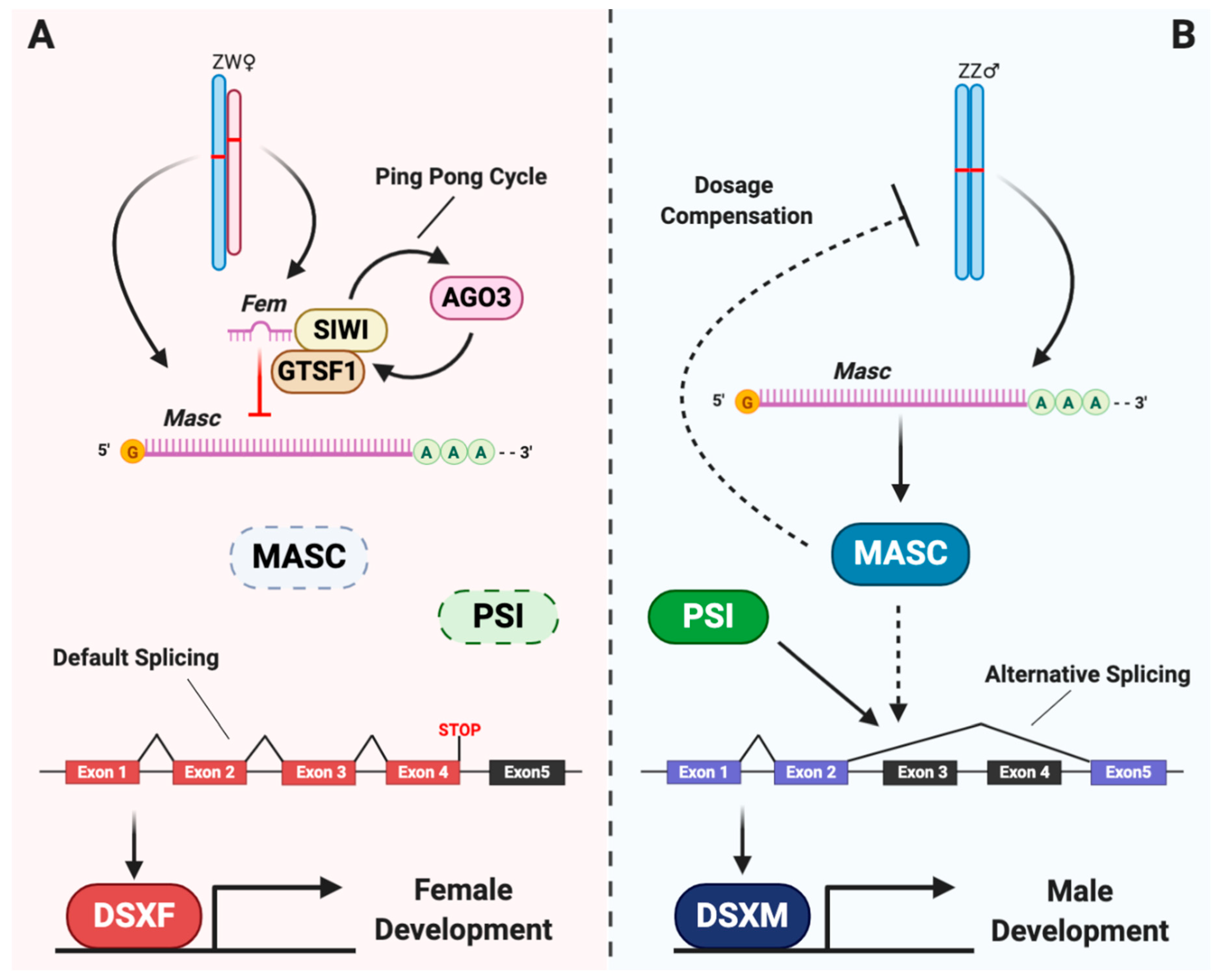
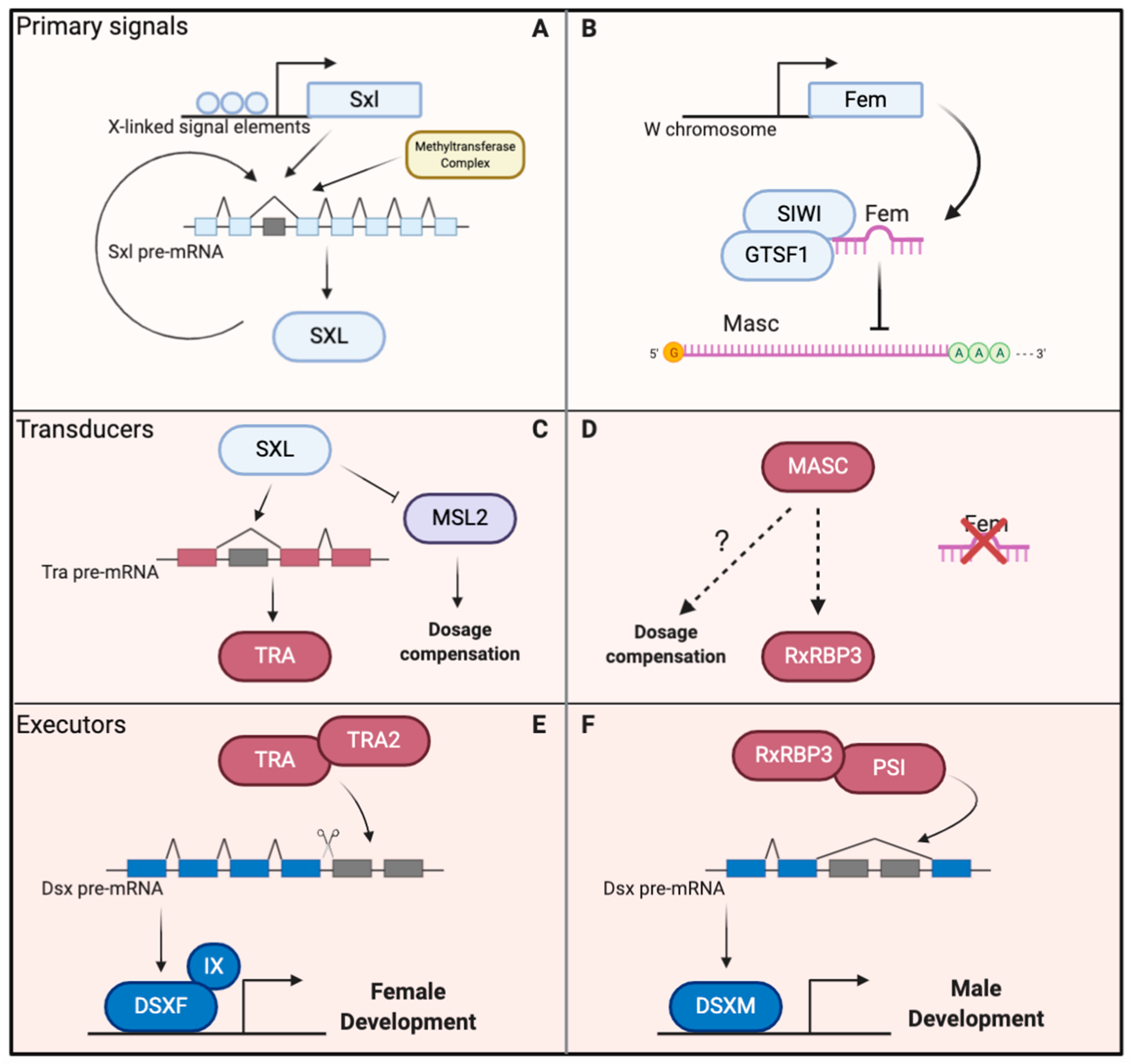
2. Overview of the Sex Determination Cascade in the Silkworm
3. Feminizers, the Primary Signals
4. Masculinizers, the Possible Transducers
5. Doublesex, the Executor
6. Conclusions and Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Otto, S.P. The Evolutionary Enigma of Sex. Am. Nat. 2009. [Google Scholar] [CrossRef] [PubMed]
- Bachtrog, D.; Mank, J.E.; Peichel, C.L.; Kirkpatrick, M.; Otto, S.P.; Ashman, T.L.; Hahn, M.W.; Kitano, J.; Mayrose, I.; Ming, R.; et al. Sex Determination: Why So Many Ways of Doing It? PLoS Biol. 2014, 12, e1001899. [Google Scholar] [CrossRef] [PubMed]
- Sahara, K.; Yoshido, A.; Traut, W. Sex chromosome evolution in moths and butterflies. Chromosom. Res. 2012, 20, 83–94. [Google Scholar] [CrossRef] [PubMed]
- Uller, T.; Pen, I.; Wapstra, E.; Beukeboom, L.W.; Komdeur, J. The evolution of sex ratios and sex-determining systems. Trends Ecol. Evol. 2007, 22, 292–297. [Google Scholar] [CrossRef]
- Salz, H.K.; Erickson, J.W. Sex determination in Drosophila: The view from the top. Fly (Austin) 2010, 4, 60–70. [Google Scholar] [CrossRef]
- Douglas, A. Harrison Sex Determination: Controlling the Master. Curr. Biol. 2007. [Google Scholar] [CrossRef]
- Billeter, J.C.; Rideout, E.J.; Dornan, A.J.; Goodwin, S.F. Control of Male Sexual Behavior in Drosophila by the Sex Determination Pathway. Curr. Biol. 2006, 16, 766–776. [Google Scholar] [CrossRef] [PubMed]
- Beye, M.; Hasselmann, M.; Fondrk, M.K.; Page, R.E.; Omholt, S.W. The gene csd is the primary signal for sexual development in the honeybee and encodes an SR-type protein. Cell 2003, 114, 419–429. [Google Scholar] [CrossRef]
- Gempe, T.; Hasselmann, M.; Schiøtt, M.; Hause, G.; Otte, M.; Beye, M. Sex determination in honeybees: Two separate mechanisms induce and maintain the female pathway. PLoS Biol. 2009, 7. [Google Scholar] [CrossRef]
- Shukla, J.N.; Palli, S.R. Sex determination in beetles: Production of all male progeny by Parental RNAi knockdown of transformer. Sci. Rep. 2012, 2. [Google Scholar] [CrossRef] [PubMed]
- Sawanth, S.K.; Gopinath, G.; Sambrani, N.; Arunkumar, K.P. The autoregulatory loop: A common mechanism of regulation of key sex determining genes in insects. J. Biosci. 2016, 41, 283–294. [Google Scholar] [CrossRef] [PubMed]
- Zou, Y.; Geuverink, E.; Beukeboom, L.W.; Verhulst, E.C.; van de Zande, L. A chimeric gene paternally instructs female sex determination in the haplodiploid wasp Nasonia. Science 2020, 370, 1115–1118. [Google Scholar] [CrossRef] [PubMed]
- Verhulst, E.C.; Beukeboom, L.W.; van de Zande, L. Maternal control of haplodiploid sex determination in the wasp Nasonia. Science 2010, 328, 620–623. [Google Scholar] [CrossRef]
- Hall, A.B.; Basu, S.; Jiang, X.; Qi, Y.; Timoshevskiy, V.A.; Biedler, J.K.; Sharakhova, M.V.; Elahi, R.; Anderson, M.A.E.; Chen, X.G.; et al. A male-determining factor in the mosquito Aedes aegypti. Science 2015, 348, 1268–1270. [Google Scholar] [CrossRef] [PubMed]
- Aryan, A.; Anderson, M.A.E.; Biedler, J.K.; Qi, Y.; Overcash, J.M.; Naumenko, A.N.; Sharakhova, M.V.; Mao, C.; Adelman, Z.N.; Tu, Z. Nix alone is sufficient to convert female Aedes aegypti into fertile males and myo-sex is needed for male flight. Proc. Natl. Acad. Sci. USA 2020, 117, 17702–17709. [Google Scholar] [CrossRef]
- Sharma, A.; Heinze, S.D.; Wu, Y.; Kohlbrenner, T.; Morilla, I.; Brunner, C.; Wimmer, E.A.; Van De Zande, L.; Robinson, M.D.; Beukeboom, L.W.; et al. Male sex in houseflies is determined by Mdmd, a paralog of the generic splice factor gene CWC22. Science 2017, 356, 642–645. [Google Scholar] [CrossRef] [PubMed]
- Meccariello, A.; Salvemini, M.; Primo, P.; Hall, B.; Koskinioti, P.; Dalíková, M.; Gravina, A.; Gucciardino, M.A.; Forlenza, F.; Gregoriou, M.E.; et al. Maleness-on-the-Y (MoY) orchestrates male sex determination in major agricultural fruit fly pests. Science 2019, 365, 1457–1460. [Google Scholar] [CrossRef]
- Krzywinska, E.; Dennison, N.J.; Lycett, G.J.; Krzywinski, J. A maleness gene in the malaria mosquito Anopheles gambiae. Science 2016, 353, 67–69. [Google Scholar] [CrossRef]
- Bopp, D.; Saccone, G.; Beye, M. Sex determination in insects: Variations on a common theme. Sex. Dev. 2014, 8, 20–28. [Google Scholar] [CrossRef] [PubMed]
- Geuverink, E.; Beukeboom, L.W. Phylogenetic distribution and evolutionary dynamics of the sex determination genes doublesex and transformer in insects. Sex. Dev. 2014, 8, 38–49. [Google Scholar] [CrossRef]
- Wang, Y.H.; Chen, X.E.; Yang, Y.; Xu, J.; Fang, G.Q.; Niu, C.Y.; Huang, Y.P.; Zhan, S. The Masc gene product controls masculinization in the black cutworm, Agrotis ipsilon. Insect Sci. 2019, 26, 1037–1044. [Google Scholar] [CrossRef]
- Suzuki, M.G. Sex determination: Insights from the silkworm. J. Genet. 2010, 89, 357–363. [Google Scholar] [CrossRef]
- Fujii, T.; Shimada, T. Sex determination in the silkworm, Bombyx mori: A female determinant on the W chromosome and the sex-determining gene cascade. Semin. Cell Dev. Biol. 2007, 18, 379–388. [Google Scholar] [CrossRef] [PubMed]
- Kiuchi, T.; Koga, H.; Kawamoto, M.; Shoji, K.; Sakai, H.; Arai, Y.; Ishihara, G.; Kawaoka, S.; Sugano, S.; Shimada, T.; et al. A single female-specific piRNA is the primary determiner of sex in the silkworm. Nature 2014, 509, 633–636. [Google Scholar] [CrossRef]
- Li, Z.; You, L.; Yan, D.; James, A.A.; Huang, Y.; Tan, A. Bombyx mori histone methyltransferase BmAsh2 is essential for silkworm piRNA-mediated sex determination. PLoS Genet. 2018, 14, e1007245. [Google Scholar] [CrossRef] [PubMed]
- Sakai, H.; Sumitani, M.; Chikami, Y.; Yahata, K.; Uchino, K.; Kiuchi, T.; Katsuma, S.; Aoki, F.; Sezutsu, H.; Suzuki, M.G. Transgenic Expression of the piRNA-Resistant Masculinizer Gene Induces Female-Specific Lethality and Partial Female-to-Male Sex Reversal in the Silkworm, Bombyx mori. PLoS Genet. 2016, 12, e1006203. [Google Scholar] [CrossRef]
- Yang, F.; Zhang, Z.; Hu, B.; Yu, Y.; Tan, A. The role of Bmznf-2 in the silkworm. Insect Sci. 2020, 1–44. [Google Scholar] [CrossRef]
- Chen, K.; Yu, Y.; Yang, D.; Yang, X.; Tang, L.; Liu, Y.; Luo, X.; Walter, J.R.; Liu, Z.; Xu, J.; et al. Gtsf1 is essential for proper female sex determination and transposon silencing in the silkworm, Bombyx mori. PLoS Genet. 2020. [Google Scholar] [CrossRef] [PubMed]
- Verhulst, E.C.; van de Zande, L.; Beukeboom, L.W. Insect sex determination: It all evolves around transformer. Curr. Opin. Genet. Dev. 2010, 20, 376–383. [Google Scholar] [CrossRef]
- Sánchez, L. Sex-determining mechanisms in insects. Int. J. Dev. Biol. 2008, 52, 837–856. [Google Scholar] [CrossRef]
- Matson, C.K.; Zarkower, D. Sex and the singular DM domain: Insights into sexual regulation, evolution and plasticity. Nat. Rev. Genet. 2012, 13, 163–174. [Google Scholar] [CrossRef]
- Traut, W.; Sahara, K.; Marec, F. Sex chromosomes and sex determination in Lepidoptera. Sex. Dev. 2007, 1, 332–346. [Google Scholar] [CrossRef]
- Xu, J.; Chen, S.; Zeng, B.; James, A.A.; Tan, A.; Huang, Y. Bombyx mori P-element Somatic Inhibitor (BmPSI) Is a Key Auxiliary Factor for Silkworm Male Sex Determination. PLoS Genet. 2017, 13, e1006576. [Google Scholar] [CrossRef]
- Lee, J.; Kiuchi, T.; Kawamoto, M.; Shimada, T.; Katsuma, S. Identification and functional analysis of a Masculinizer orthologue in Trilocha varians (Lepidoptera: Bombycidae). Insect Mol. Biol. 2015, 24, 561–569. [Google Scholar] [CrossRef] [PubMed]
- Fukui, T.; Kiuchi, T.; Shoji, K.; Kawamoto, M.; Shimada, T.; Katsuma, S. In vivo masculinizing function of the Ostrinia furnacalis Masculinizer gene. Biochem. Biophys. Res. Commun. 2018, 503, 1768–1772. [Google Scholar] [CrossRef]
- Suzuki, M.G.; Imanishi, S.; Dohmae, N.; Nishimura, T.; Shimada, T.; Matsumoto, S. Establishment of a Novel In Vivo Sex-Specific Splicing Assay System To Identify a trans-Acting Factor That Negatively Regulates Splicing of Bombyx mori dsx Female Exons. Mol. Cell. Biol. 2008, 28, 333–343. [Google Scholar] [CrossRef]
- Suzuki, M.G.; Funaguma, S.; Kanda, T.; Tamura, T.; Shimada, T. Role of the male BmDSX protein in the sexual differentiation of Bombyx mori. Evol. Dev. 2005, 7, 58–68. [Google Scholar] [CrossRef]
- Xu, J.; Zhan, S.; Chen, S.; Zeng, B.; Li, Z.; James, A.A.; Tan, A.; Huang, Y. Sexually dimorphic traits in the silkworm, Bombyx mori, are regulated by doublesex. Insect Biochem. Mol. Biol. 2017, 80, 42–51. [Google Scholar] [CrossRef]
- Hasimoto, H. The role of the W-chromosome in the sex determination of Bombyx mori. Jpn. J. Genet. 1933, 8, 245–247. [Google Scholar] [CrossRef]
- Abe, H.; Fujii, T.; Tanaka, N.; Yokoyama, T.; Kakehashi, H.; Ajimura, M.; Mita, K.; Banno, Y.; Yasukochi, Y.; Oshiki, T.; et al. Identification of the female-determining region of the W chromosome in Bombyx mori. Genetica 2008, 133, 269–282. [Google Scholar] [CrossRef]
- Xia, Q.; Zhou, Z.; Lu, C.; Cheng, D.; Dai, F.; Li, B.; Zhao, P.; Zha, X.; Cheng, T.; Chai, C.; et al. A draft sequence for the genome of the domesticated silkworm (Bombyx mori). Science 2004, 306, 1937–1940. [Google Scholar] [CrossRef]
- Abe, H.; Mita, K.; Yasukochi, Y.; Oshiki, T.; Shimada, T. Retrotransposable elements on the W chromosome of the silkworm, Bombyx mori. Cytogenet. Genome Res. 2005, 110, 144–151. [Google Scholar] [CrossRef] [PubMed]
- Kawaoka, S.; Hayashi, N.; Suzuki, Y.; Abe, H.; Sugano, S.; Tomari, Y.; Shimada, T.; Katsuma, S. The Bombyx ovary-derived cell line endogenously expresses PIWI/PIWI-interacting RNA complexes. RNA 2009, 15, 1258–1264. [Google Scholar] [CrossRef] [PubMed]
- Kawaoka, S.; Kadota, K.; Arai, Y.; Suzuki, Y.; Fujii, T.; Abe, H.; Yasukochi, Y.; Mita, K.; Sugano, S.; Shimizu, K.; et al. The silkworm W chromosome is a source of female-enriched piRNAs. Rna 2011, 17, 2144–2151. [Google Scholar] [CrossRef] [PubMed]
- Kawaoka, S.; Mitsutake, H.; Kiuchi, T.; Kobayashi, M.; Yoshikawa, M.; Suzuki, Y.; Sugano, S.; Shimada, T.; Kobayashi, J.; Tomari, Y.; et al. A role for transcription from a piRNA cluster in de novo piRNA production. RNA 2012, 18, 265–273. [Google Scholar] [CrossRef] [PubMed]
- Hara, K.; Fujii, T.; Suzuki, Y.; Sugano, S.; Shimada, T.; Katsuma, S.; Kawaoka, S. Altered expression of testis-specific genes, piRNAs, and transposons in the silkworm ovary masculinized by a W chromosome mutation. BMC Genom. 2012, 13. [Google Scholar] [CrossRef] [PubMed]
- Kawaoka, S.; Arai, Y.; Kadota, K.; Suzuki, Y.; Hara, K.; Sugano, S.; Shimizu, K.; Tomari, Y.; Shimada, T.; Katsuma, S. Zygotic amplification of secondary piRNAs during silkworm embryogenesis. RNA 2011, 17, 1401–1407. [Google Scholar] [CrossRef]
- Ishizu, H.; Siomi, H.; Siomi, M.C. Biology of Piwi-interacting RNAs: New insights into biogenesis and function inside and outside of germlines. Genes Dev. 2012, 26, 2361–2373. [Google Scholar] [CrossRef]
- Sato, K.; Siomi, M.C. The piRNA pathway in Drosophila ovarian germ and somatic cells. Proc. Jpn. Acad. Ser. B Phys. Biol. Sci. 2020, 96, 32–42. [Google Scholar] [CrossRef]
- Sakakibara, K.; Siomi, M.C. The PIWI-Interacting RNA Molecular Pathway: Insights from Cultured Silkworm Germline Cells. BioEssays 2018, 40. [Google Scholar] [CrossRef]
- Czech, B.; Munafò, M.; Ciabrelli, F.; Eastwood, E.L.; Fabry, M.H.; Kneuss, E.; Hannon, G.J. PiRNA-guided genome defense: From biogenesis to silencing. Annu. Rev. Genet. 2018, 52, 131–157. [Google Scholar] [CrossRef] [PubMed]
- Ozata, D.M.; Gainetdinov, I.; Zoch, A.; O’Carroll, D.; Zamore, P.D. PIWI-interacting RNAs: Small RNAs with big functions. Nat. Rev. Genet. 2019, 20, 89–108. [Google Scholar] [CrossRef]
- Ohtani, H.; Iwasaki, Y.W.; Shibuya, A.; Siomi, H.; Siomi, M.C.; Saito, K. DmGTSF1 is necessary for Piwi-piRISC-mediated transcriptional transposon silencing in the Drosophila ovary. Genes Dev. 2013, 27, 1656–1661. [Google Scholar] [CrossRef]
- Dönertas, D.; Sienski, G.; Brennecke, J. Drosophila Gtsf1 is an essential component of the Piwi-mediated transcriptional silencing complex. Genes Dev. 2013, 27, 1693–1705. [Google Scholar] [CrossRef] [PubMed]
- Sienski, G.; Dönertas, D.; Brennecke, J. Transcriptional silencing of transposons by Piwi and maelstrom and its impact on chromatin state and gene expression. Cell 2012, 151, 964–980. [Google Scholar] [CrossRef] [PubMed]
- Chen, K.; Chen, S.; Xu, J.; Yu, Y.; Liu, Z.; Tan, A.; Huang, Y. Maelstrom regulates spermatogenesis of the silkworm, Bombyx mori. Insect Biochem. Mol. Biol. 2019, 109, 43–51. [Google Scholar] [CrossRef]
- Izumi, N.; Shoji, K.; Sakaguchi, Y.; Honda, S.; Kirino, Y.; Suzuki, T.; Katsuma, S.; Tomari, Y. Identification and Functional Analysis of the Pre-piRNA 3′ Trimmer in Silkworms. Cell 2016, 164, 962–973. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Liu, Y.; Yang, X.; Liu, Z.; Luo, X.; Xu, J.; Huang, Y. Dysfunction of dimorphic sperm impairs male fertility in the silkworm. Cell Discov. 2020, 6. [Google Scholar] [CrossRef] [PubMed]
- Katsuma, S.; Sugano, Y.; Kiuchi, T.; Shimada, T. Two conserved cysteine residues are required for the masculinizing activity of the silkworm Masc protein. J. Biol. Chem. 2015, 290, 26114–26124. [Google Scholar] [CrossRef] [PubMed]
- Fukui, T.; Kawamoto, M.; Shoji, K.; Kiuchi, T.; Sugano, S.; Shimada, T.; Suzuki, Y.; Katsuma, S. The Endosymbiotic Bacterium Wolbachia Selectively Kills Male Hosts by Targeting the Masculinizing Gene. PLoS Pathog. 2015, 11, e1005048. [Google Scholar] [CrossRef]
- Zhao, Q.; Li, J.; Wen, M.Y.; Wang, H.; Wang, Y.; Wang, K.X.; Wan, Q.X.; Zha, X.F. A novel splice variant of the masculinizing gene masc with piRNA-cleavage-site defect functions in female external genital development in the silkworm, bombyx mori. Biomolecules 2019, 9, 318. [Google Scholar] [CrossRef] [PubMed]
- Zhuo, J.C.; Zhang, H.H.; Xie, Y.C.; Li, H.J.; Hu, Q.L.; Zhang, C.X. Identification of a female determinant gene for the sexual determination of a hemipteran insect, the brown planthopper. bioRxiv 2019. [Google Scholar] [CrossRef]
- Krzywinska, E.; Ferretti, L.; Li, J.; Li, J.-C.; Chen, C.-H.; Krzywinski, J. femaleless controls sex determination and dosage compensation pathways in females of the Anopheles mosquitoes. Curr. Biol. 2021, 1–8. [Google Scholar] [CrossRef]
- Siebel, C.W.; Kanaar, R.; Rio, D.C. Regulation of tissue-specific P-element pre-mRNA splicing requires the RNA-binding protein PSI. Genes Dev. 1994, 8, 1713–1725. [Google Scholar] [CrossRef] [PubMed]
- Labourier, E.; Adams, M.D.; Rio, D.C. Modulation of P-element pre-mRNA splicing by a direct interaction between PSI and U1 snRNP 70K protein. Mol. Cell 2001, 8, 363–373. [Google Scholar] [CrossRef]
- Adams, M.D.; Tarng, R.S.; Rio, D.C. The alternative splicing factor PSI regulates P-element third intron splicing in vivo. Genes Dev. 1997, 11, 129–138. [Google Scholar] [CrossRef] [PubMed]
- Amarasinghe, A.K.; Macdiarmid, R.; Adams, M.D.; Rio, D.C. An in vitro-selected RNA-binding site for the KH domain protein PSI acts as a splicing inhibitor element. RNA 2001, 7, 1239–1253. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Chmiel, N.H.; Rio, D.C.; Doudna, J.A. Distinct contributions of KH domains to substrate binding affinity of Drosophila P-element somatic inhibitor protein. RNA 2006, 12, 283–291. [Google Scholar] [CrossRef] [PubMed]
- Ignjatovic, T.; Yang, J.C.; Butler, J.; Neuhaus, D.; Nagai, K. Structural basis of the interaction between P-element somatic inhibitor and U1-70k essential for the alternative splicing of P-element transposase. J. Mol. Biol. 2005, 351, 52–65. [Google Scholar] [CrossRef]
- Wang, Y.; Zhao, Q.; Wan, Q.X.; Wang, K.X.; Zha, X.F. P-element somatic inhibitor protein binding a target sequence in dsx pre-mrna conserved in Bombyx mori and Spodoptera litura. Int. J. Mol. Sci. 2019, 20, 2361. [Google Scholar] [CrossRef]
- Gopinath, G.; Arunkumar, K.P.; Mita, K.; Nagaraju, J. Role of Bmznf-2, a Bombyx mori CCCH zinc finger gene, in masculinisation and differential splicing of Bmtra-2. Insect Biochem. Mol. Biol. 2016, 75, 32–44. [Google Scholar] [CrossRef]
- Zheng, Z.Z.; Sun, X.; Zhang, B.; Pu, J.; Jiang, Z.Y.; Li, M.; Fan, Y.J.; Xu, Y.Z. Alternative splicing regulation of doublesex gene by RNA-binding proteins in the silkworm Bombyx mori. RNA Biol. 2019, 16, 809–820. [Google Scholar] [CrossRef]
- Pane, A.; Salvemini, M.; Delli Bovi, P.; Polito, C.; Saccone, G. The transformer gene in Ceratitis capitata provides a genetic basis for selecting and remembering the sexual fate. Development 2002, 129, 3715–3725. [Google Scholar]
- Concha, C.; Scott, M.J. Sexual development in Lucilia cuprina (Diptera, Calliphoridae) is controlled by the transformer gene. Genetics 2009, 182, 785–798. [Google Scholar] [CrossRef]
- Liu, G.; Wu, Q.; Li, J.; Zhang, G.; Wan, F. RNAi-mediated knock-down of transformer and transformer 2 to generate male-only progeny in the oriental fruit fly, Bactrocera dorsalis (Hendel). PLoS ONE 2015, 10, e0128892. [Google Scholar] [CrossRef]
- Li, F.; Vensko, S.P.; Belikoff, E.J.; Scott, M.J. Conservation and Sex-Specific Splicing of the transformer Gene in the Calliphorids Cochliomyia hominivorax, Cochliomyia macellaria and Lucilia sericata. PLoS ONE 2013, 8, e56303. [Google Scholar] [CrossRef] [PubMed]
- Kyrou, K.; Hammond, A.M.; Galizi, R.; Kranjc, N.; Burt, A.; Beaghton, A.K.; Nolan, T.; Crisanti, A. A CRISPR–Cas9 gene drive targeting doublesex causes complete population suppression in caged Anopheles gambiae mosquitoes. Nat. Biotechnol. 2018, 36, 1062–1071. [Google Scholar] [CrossRef] [PubMed]
- Simoni, A.; Hammond, A.M.; Beaghton, A.K.; Galizi, R.; Taxiarchi, C.; Kyrou, K.; Meacci, D.; Gribble, M.; Morselli, G.; Burt, A.; et al. A male-biased sex-distorter gene drive for the human malaria vector Anopheles gambiae. Nat. Biotechnol. 2020, 38, 1054–1060. [Google Scholar] [CrossRef] [PubMed]
- Tan, A.; Fu, G.; Jin, L.; Guo, Q.; Li, Z.; Niu, B.; Meng, Z.; Morrison, N.I.; Alphey, L.; Huang, Y. Transgene-based, female-specific lethality system for genetic sexing of the silkworm, Bombyx mori. Proc. Natl. Acad. Sci. USA 2013, 110, 6766–6770. [Google Scholar] [CrossRef]
- Xu, J.; Wang, Y.; Li, Z.; Ling, L.; Zeng, B.; James, A.A.; Tan, A.; Huang, Y. Transcription activator-like effector nuclease (TALEN)-mediated female-specific sterility in the silkworm, Bombyx mori. Insect Mol. Biol. 2014, 23, 800–807. [Google Scholar] [CrossRef]
- Suzuki, M.G.; Ohbayashi, F.; Mita, K.; Shimada, T. The mechanism of sex-specific splicing at the doublesex gene is different between Drosophila melanogaster and Bombyx mori. Insect Biochem. Mol. Biol. 2001, 31, 1201–1211. [Google Scholar] [CrossRef]
- Suzuki, M.G.; Funaguma, S.; Kanda, T.; Tamura, T.; Shimada, T. Analysis of the biological functions of a doublesex homologue in Bombyx mori. Dev. Genes Evol. 2003, 213, 345–354. [Google Scholar] [CrossRef]
- Chen, X.; Cao, Y.; Zhan, S.; Tan, A.; Palli, S.R.; Huang, Y. Disruption of sex-specific doublesex exons results in male- and female-specific defects in the black cutworm, Agrotis ipsilon. Pest Manag. Sci. 2019, 75, 1697–1706. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Liu, Q.; Liu, H.; Bi, H.; Wang, Y.; Chen, X.; Wu, N.; Xu, J.; Zhang, Z.; Huang, Y.; et al. Mutation of doublesex in Hyphantria cunea results in sex-specific sterility. Pest Manag. Sci. 2020, 76, 1673–1682. [Google Scholar] [CrossRef]
- Wang, Y.; Chen, X.; Liu, Z.; Xu, J.; Li, X.; Bi, H.; Andongma, A.A.; Niu, C.; Huang, Y. Mutation of doublesex induces sex-specific sterility of the diamondback moth Plutella xylostella. Insect Biochem. Mol. Biol. 2019, 112, 103180. [Google Scholar] [CrossRef]
- Garrett-Engele, C.M.; Siegal, M.L.; Manoli, D.S.; Williams, B.C.; Li, H.; Baker, B.S. intersex, a gene required for female sexual development in Drosophila, is expressed in both sexes and functions together with doublesex to regulate terminal differentiation. Development 2002, 129, 4661–4675. [Google Scholar]
- Xu, J.; Yu, Y.; Chen, K.; Huang, Y. Intersex regulates female external genital and imaginal disc development in the silkworm. Insect Biochem. Mol. Biol. 2019, 108, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Liu, W.; Yang, D.; Chen, S.; Chen, K.; Liu, Z.; Yang, X.; Meng, J.; Zhu, G.; Dong, S.; et al. Regulation of olfactory-based sex behaviors in the silkworm by genes in the sexdetermination cascade. PLoS Genet. 2020, 16, e1008622. [Google Scholar] [CrossRef]
- Luo, S.D.; Shi, G.W.; Baker, B.S. Direct targets of the D. melanogaster DSXF protein and the evolution of sexual development. Development 2011, 138, 2761–2771. [Google Scholar] [CrossRef]
- Clough, E.; Jimenez, E.; Kim, Y.A.; Whitworth, C.; Neville, M.C.; Hempel, L.U.; Pavlou, H.J.; Chen, Z.X.; Sturgill, D.; Dale, R.K.; et al. Sex- and tissue-specific functions of drosophila doublesex transcription factor target genes. Dev. Cell 2014, 31, 761–773. [Google Scholar] [CrossRef]
- Baral, S.; Arumugam, G.; Deshmukh, R.; Kunte, K. Genetic architecture and sex-specific selection govern modular, male-biased evolution of doublesex. Sci. Adv. 2019, 5, 1–9. [Google Scholar] [CrossRef]
- Guo, J.; Tang, H.W.; Li, J.; Perrimon, N.; Yan, D. Xio is a component of the Drosophila sex determination pathway and RNA N6-methyladenosine methyltransferase complex. Proc. Natl. Acad. Sci. USA 2018, 115, 3674–3679. [Google Scholar] [CrossRef] [PubMed]
- Lence, T.; Akhtar, J.; Bayer, M.; Schmid, K.; Spindler, L.; Ho, C.H.; Kreim, N.; Andrade-Navarro, M.A.; Poeck, B.; Helm, M.; et al. M6A modulates neuronal functions and sex determination in Drosophila. Nature 2016, 540, 242–247. [Google Scholar] [CrossRef] [PubMed]
- Kan, L.; Grozhik, A.V.; Vedanayagam, J.; Patil, D.P.; Pang, N.; Lim, K.S.; Huang, Y.C.; Joseph, B.; Lin, C.J.; Despic, V.; et al. The m6A pathway facilitates sex determination in Drosophila. Nat. Commun. 2017, 8, 1–16. [Google Scholar] [CrossRef]
- Haussmann, I.U.; Bodi, Z.; Sanchez-Moran, E.; Mongan, N.P.; Archer, N.; Fray, R.G.; Soller, M. M6 A potentiates Sxl alternative pre-mRNA splicing for robust Drosophila sex determination. Nature 2016, 540, 301–304. [Google Scholar] [CrossRef] [PubMed]
- Yuzawa, T.; Matsuoka, M.; Sumitani, M.; Aoki, F.; Sezutsu, H.; Suzuki, M.G. Transgenic and knockout analyses of Masculinizer and doublesex illuminated the unique functions of doublesex in germ cell sexual development of the silkworm, Bombyx mori. BMC Dev. Biol. 2020, 20, 1–15. [Google Scholar] [CrossRef]
- Xu, X.; Wang, K.; Zha, X. An antisense lncRNA functions in alternative splicing of Bmdsx in the silkworm, Bombyx mori. Biochem. Biophys. Res. Commun. 2019, 516, 639–644. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Li, J.; Wan, Q.X.; Zhao, Q.; Wang, K.X.; Zha, X.F. Spliceosomal protein gene BmSPX regulates reproductive organ development in bombyx mori. Int. J. Mol. Sci. 2020, 21, 2579. [Google Scholar] [CrossRef]
- Geffroy, B.; Douhard, M. The Adaptive Sex in Stressful Environments. Trends Ecol. Evol. 2019, 34, 628–640. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yang, X.; Chen, K.; Wang, Y.; Yang, D.; Huang, Y. The Sex Determination Cascade in the Silkworm. Genes 2021, 12, 315. https://doi.org/10.3390/genes12020315
Yang X, Chen K, Wang Y, Yang D, Huang Y. The Sex Determination Cascade in the Silkworm. Genes. 2021; 12(2):315. https://doi.org/10.3390/genes12020315
Chicago/Turabian StyleYang, Xu, Kai Chen, Yaohui Wang, Dehong Yang, and Yongping Huang. 2021. "The Sex Determination Cascade in the Silkworm" Genes 12, no. 2: 315. https://doi.org/10.3390/genes12020315
APA StyleYang, X., Chen, K., Wang, Y., Yang, D., & Huang, Y. (2021). The Sex Determination Cascade in the Silkworm. Genes, 12(2), 315. https://doi.org/10.3390/genes12020315






