Nipple Sparing Mastectomy as a Risk-Reducing Procedure for BRCA-Mutated Patients
Abstract
1. Introduction
2. Methods
2.1. Eligibility Criteria
2.2. Study Selection
2.3. Data Collection Process
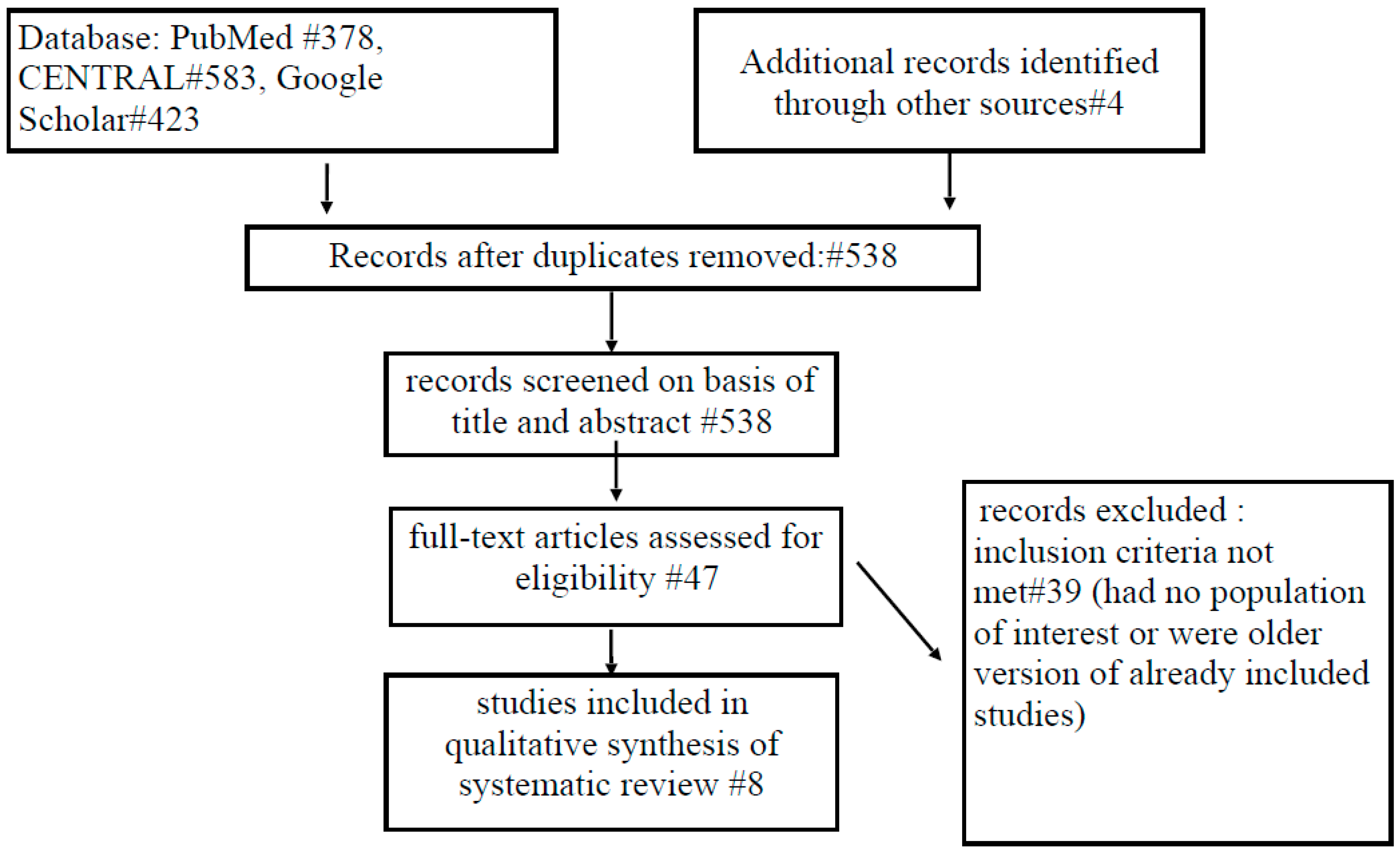
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Kuchenbaecker, K.B.; Hopper, J.L.; Barnes, D.R.; Phillips, K.-A.; Mooij, T.M.; Roos-Blom, M.-J.; Jervis, S.; Van Leeuwen, F.E.; Milne, R.L.; Andrieu, N.; et al. Risks of Breast, Ovarian, and Contralateral Breast Cancer for BRCA1 and BRCA2 Mutation Carriers. JAMA 2017, 317, 2402–2416. [Google Scholar] [CrossRef]
- Chen, S.; Parmigiani, G. Meta-Analysis ofBRCA1andBRCA2Penetrance. J. Clin. Oncol. 2007, 25, 1329–1333. [Google Scholar] [CrossRef] [PubMed]
- Ingham, S.L.; Sperrin, M.; Baildam, A.; Ross, G.L.; Clayton, R.; Lallo, F.; Buchan, I.; Howell, A.; Evans, D.G.R. Risk-reducing surgery increases survival in 1/2 mutation carriers unaffected at time of family referral. Breast Cancer Res. Treat. 2013, 142, 611–618. [Google Scholar] [CrossRef]
- Liede, A.; Cai, M.; Crouter, T.F.; Niepel, D.; Callaghan, F.; Evans, D.G. Risk-reducing mastectomy rates in the US: A closer examination of the Angelina Jolie effect. Breast Cancer Res. Treat. 2018, 171, 435–442. [Google Scholar] [CrossRef]
- Co, M.; Chiu, R.; Chiu, T.M.; Chong, Y.C.; Lau, S.; Lee, Y.H.; To, H.M.; Kwong, A. Nipple-Sparing Mastectomy and Its Application on Gene Mutation Carrier. Clin. Breast Cancer 2017, 17, 581–584. [Google Scholar] [CrossRef]
- Rusby, J.E.; Smith, B.L.; Gui, G.P.H. Nipple-sparing mastectomy. Br. J. Surg. 2010, 97, 305–316. [Google Scholar] [CrossRef] [PubMed]
- Grinstein, O.; Krug, B.; Hellmic, M.; Siedek, F.; Malter, W.; Burke, C.; Schmutzler, R.; Maintz, D.; Rhiem, K. Residual glandular tissue (RGT) in 1/2 germline mutation carriers with unilateral and bilateral prophylactic mastectomies. Surg. Oncol. 2019, 29, 126–133. [Google Scholar] [CrossRef] [PubMed]
- Kaidar-Person, O.; Boersma, L.J.; Poortmans, P.; Sklair-Levy, M.; Offersen, B.V.; Cardoso, M.-J.; De Ruysscher, D. Residual Glandular Breast Tissue After Mastectomy: A Systematic Review. Ann. Surg. Oncol. 2020, 27, 2288–2296. [Google Scholar] [CrossRef]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G.; The PRISMA Group. Preferred Reporting Items for Systematic Reviews and Meta-Analyses: The PRISMA Statement. PLoS Med. 2009, 6, e1000097. [Google Scholar] [CrossRef]
- Oxford Centre for Evidence Based Medicine—Levels of Evidence. Available online: https://www.cebm.net/2009/06/oxford-centre-evidence-based-medicine-levels-evidence-march-2009/ (accessed on 18 January 2021).
- Wells, G.A.; Shea, B.; O’Connell, D.; Peterson, J.; Welch, V.; Losos, M.; Tugwell, P. The Newcastle-Ottawa Scale (NOS) for Assessing the Quality of Nonrandomised Studies in Meta-Analyses. 2019. Available online: http://www.ohri.ca/programs/clinical_epidemiology/oxford.asp (accessed on 18 January 2021).
- Murad, M.H.; Sultan, S.; Haffar, S.; Bazerbachi, F. Methodological quality and synthesis of case series and case reports. BMJ Evidence-Based Med. 2018, 23, 60–63. [Google Scholar] [CrossRef] [PubMed]
- Hartmann, L.C.; Sellers, T.A.; Schaid, D.J.; Frank, T.S.; Soderberg, C.L.; Sitta, D.L.; Frost, M.H.; Grant, C.S.; Donohue, J.H.; Woods, J.E.; et al. Efficacy of Bilateral Prophylactic Mastectomy in 1 and 2 Gene Mutation Carriers. J. Natl. Cancer Inst. 2001, 93, 1633–1637. [Google Scholar] [CrossRef]
- Domchek, S.M.; Friebel, T.M.; Singer, C.F.; Evans, D.G.; Lynch, H.T.; Isaacs, C.; Rebbeck, T.R. Association of risk-reducing surgery in 1 or 2 mutation carriers with cancer risk and mortality. JAMA 2010, 304, 967–975. [Google Scholar] [CrossRef]
- Harness, J.K.; Vetter, T.S.; Salibian, A.H. Areola and Nipple-Areola-Sparing Mastectomy for Breast Cancer Treatment and Risk Reduction: Report of an Initial Experience in a Community Hospital Setting. Ann. Surg. Oncol. 2010, 18, 917–922. [Google Scholar] [CrossRef] [PubMed]
- Peled, A.W.; Irwin, C.S.; Hwang, E.S.; Ewing, C.A.; Alvarado, M.; Esserman, L.J. Total skin-sparing mastectomy in mutation carriers. Ann. Surg. Oncol. 2014, 21, 37–41. [Google Scholar] [CrossRef] [PubMed]
- Yao, K.; Liederbach, E.; Tang, R.; Lei, L.; Czechura, T.; Sisco, M.; Howard, M.; Hulick, P.J.; Weissman, S.; Winchester, D.J.; et al. Nipple-sparing mastectomy in 1/2 mutation carriers: An interim analysis and review of the literature. Ann Surg Oncol. 2015, 22, 370–376. [Google Scholar] [CrossRef] [PubMed]
- Manning, A.; Wood, C.; Eaton, A.; Stempel, M.; Capko, D.; Pusic, A.; Morrow, M.; Sacchini, V. Nipple-sparing mastectomy in patients with 1/2 mutations and variants of uncertain significance. Br. J. Surg. 2015, 102, 1354–1359. [Google Scholar] [CrossRef]
- Jakub, J.W.; Peled, A.W.; Gray, R.J.; Greenup, R.A.; Kiluk, J.V.; Sacchini, V.; McLaughlin, S.A.; Tchou, J.; Vierkant, R.A.; Degnim, A.C.; et al. Oncological safety of prophylactic nipple-sparing mastectomy in a population with mutations. A multi-institutional study. JAMA Surg. 2018, 153, 123–129. [Google Scholar]
- De Al Cantara Filho, P.; Capko, D.; Barry, J.M. Nipple-sparing mastectomy for breast cancer and risk reducing surgery: The Memorial Sloan-Kettering Cancer Center experience. Ann. Surg. Oncol. 2011, 18, 3117–3122. [Google Scholar] [CrossRef] [PubMed]
- Valero, M.G.; Moo, T.-A.; Muhsen, S.; Zabor, E.C.; Stempel, M.; Pusic, A.; Gemignani, M.L.; Morrow, M.; Sacchini, V.S. Use of bilateral prophylactic nipple-sparing mastectomy in patients with high risk of breast cancer. Br. J. Surg. 2020, 107, 1307–1312. [Google Scholar] [CrossRef]
- Papassotiropoulos, B.; Güth, U.; Chiesa, F.; Rageth, C.; Amann, E.; Baege, A.; Elfgen, C.; Varga, Z.; Moskovszky, L.; Endhardt, K.; et al. Prospective Evaluation of Residual Breast Tissue After Skin- or Nipple-Sparing Mastectomy: Results of the SKINI-Trial. Ann. Surg. Oncol. 2019, 26, 1254–1262. [Google Scholar] [CrossRef]
- Reynolds, C.; Davidson, J.A.; Lindor, N.M.; Glazebrook, K.N.; Jakub, J.W.; Degnim, A.C.; Sandhu, N.P.; Walsh, M.F.; Hartmann, L.C.; Boughey, J.C. Prophylactic and Therapeutic Mastectomy in Mutation Carriers: Can the Nipple Be Preserved? Ann. Surg. Oncol. 2011, 18, 3102–3109. [Google Scholar] [CrossRef]
- Gradishar, W.J.; Anderson, B.O.; Abraham, J.; Aft, R.; Agnese, D.; Allison, K.H.; Blair, S.L.; Burstein, H.J.; Dang, C.; Elias, A.D.; et al. Breast Cancer, Version 3.2020, NCCN Clinical Practice Guidelines in Oncology. J. Natl. Compr. Cancer Netw. 2020, 18, 452–478. [Google Scholar] [CrossRef] [PubMed]
- ESMO Guidelines. Available online: https://www.esmo.org/guidelines/breast-cancer (accessed on 31 December 2020).
- Metcalfe, K.A.; Cil, T.D.; Semple, J.L.; Li, L.D.X.; Bagher, S.; Zhong, T.; Virani, S.; Narod, S.; Pal, T. Long-Term Psychosocial Functioning in Women with Bilateral Prophylactic Mastectomy: Does Preservation of the Nipple-Areolar Complex Make a Difference? Ann. Surg. Oncol. 2015, 22, 3324–3330. [Google Scholar] [CrossRef] [PubMed]
- Keller, K.; Meisel, C.; Grübling, N.; Petzold, A.; Wimberger, P.; Kast, K. Patient-Reported Satisfaction after Prophylactic Operations of the Breast. Breast Care 2019, 14, 217–223. [Google Scholar] [CrossRef]
- Salibian, A.A.; Bekisz, J.M.; Frey, J.D.; Miller, B.; Choi, M.; Karp, N.S. Prophylactic nipple-sparing mastectomy in young previvors: Examining decision-making, reconstructive outcomes, and patient satisfaction in + patients under 30. Breast J. 2019, 26, 971–975. [Google Scholar] [CrossRef] [PubMed]
- Carbine, N.E.; Lostumbo, L.; Wallace, J.; Ko, H. Risk-reducing mastectomy for the prevention of primary breast cancer. Cochrane Database Syst. Rev. 2018, 4, CD002748. [Google Scholar] [CrossRef]
- Meijers-Heijboer, H.; van Geel, B.; van Putten, W.L.; Henzen-Logmans, S.C.; Seynaeve, C.; Menke-Pluymers, M.B.; Bartles, C.C.; Verhoog, L.C.; van den Ouweland, A.M.; Neirmeijer, M.F.; et al. Breast cancer after prophylactic bilateral mastectomy in women with a 1 or 2 mutation. N. Engl. J. Med. 2001, 345, 159–164. [Google Scholar] [CrossRef] [PubMed]
| #BRCA Mutation Carriers Undergoing Risk-Reducing NAC-Sparing Mastectomy | Breast Cancer Diagnosis Following Risk-Reducing NAC-Sparing Mastectomy | Median Follow-up (Years) | Median Age at Time of Surgery (Years) | 1 | 2 | Level of Evidence | Quality Score | |
|---|---|---|---|---|---|---|---|---|
| Hartmann 2001 [13] | 26 | 0 | 13.4 | 36.5 (§) 44 (§§) | NR | NR | 3 | 7 |
| Domchek 2010 [14] | 247 | 0 | 2.5–3.7 (in different groups) | 40.7 | 159 | 88 | 3 | 7 |
| Harness 2010 [15] | 6 | 0 | 1.5 | NR | NR | NR | 4 | 5 |
| Peled 2014 [16] | 26 | 0 | 4.3 | 41.2 | 14 | 12 | 4 | 6 |
| Yao 2015 [17] | 150 | 1 | 2.7 | 41 (mean) | NR # | NR # | 4 | 7 |
| Manning 2015 [18] | 63 | 0 | 2.2 | 39 | NR * | NR * | 4 | 6 |
| Jakub 2018 [19] | 202 | 0 | 2.8 and 4.7 (in different groups) | 41 | NR ** | NR ** | 4 | 7 |
| Valero 2020 [21] | 117 | 0 | 3 | 41.5 | 72 | 45 | 4 | 7 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rocco, N.; Montagna, G.; Criscitiello, C.; Nava, M.B.; Privitera, F.; Taher, W.; Gloria, A.; Catanuto, G. Nipple Sparing Mastectomy as a Risk-Reducing Procedure for BRCA-Mutated Patients. Genes 2021, 12, 253. https://doi.org/10.3390/genes12020253
Rocco N, Montagna G, Criscitiello C, Nava MB, Privitera F, Taher W, Gloria A, Catanuto G. Nipple Sparing Mastectomy as a Risk-Reducing Procedure for BRCA-Mutated Patients. Genes. 2021; 12(2):253. https://doi.org/10.3390/genes12020253
Chicago/Turabian StyleRocco, Nicola, Giacomo Montagna, Carmen Criscitiello, Maurizio Bruno Nava, Francesca Privitera, Wafa Taher, Antonio Gloria, and Giuseppe Catanuto. 2021. "Nipple Sparing Mastectomy as a Risk-Reducing Procedure for BRCA-Mutated Patients" Genes 12, no. 2: 253. https://doi.org/10.3390/genes12020253
APA StyleRocco, N., Montagna, G., Criscitiello, C., Nava, M. B., Privitera, F., Taher, W., Gloria, A., & Catanuto, G. (2021). Nipple Sparing Mastectomy as a Risk-Reducing Procedure for BRCA-Mutated Patients. Genes, 12(2), 253. https://doi.org/10.3390/genes12020253







