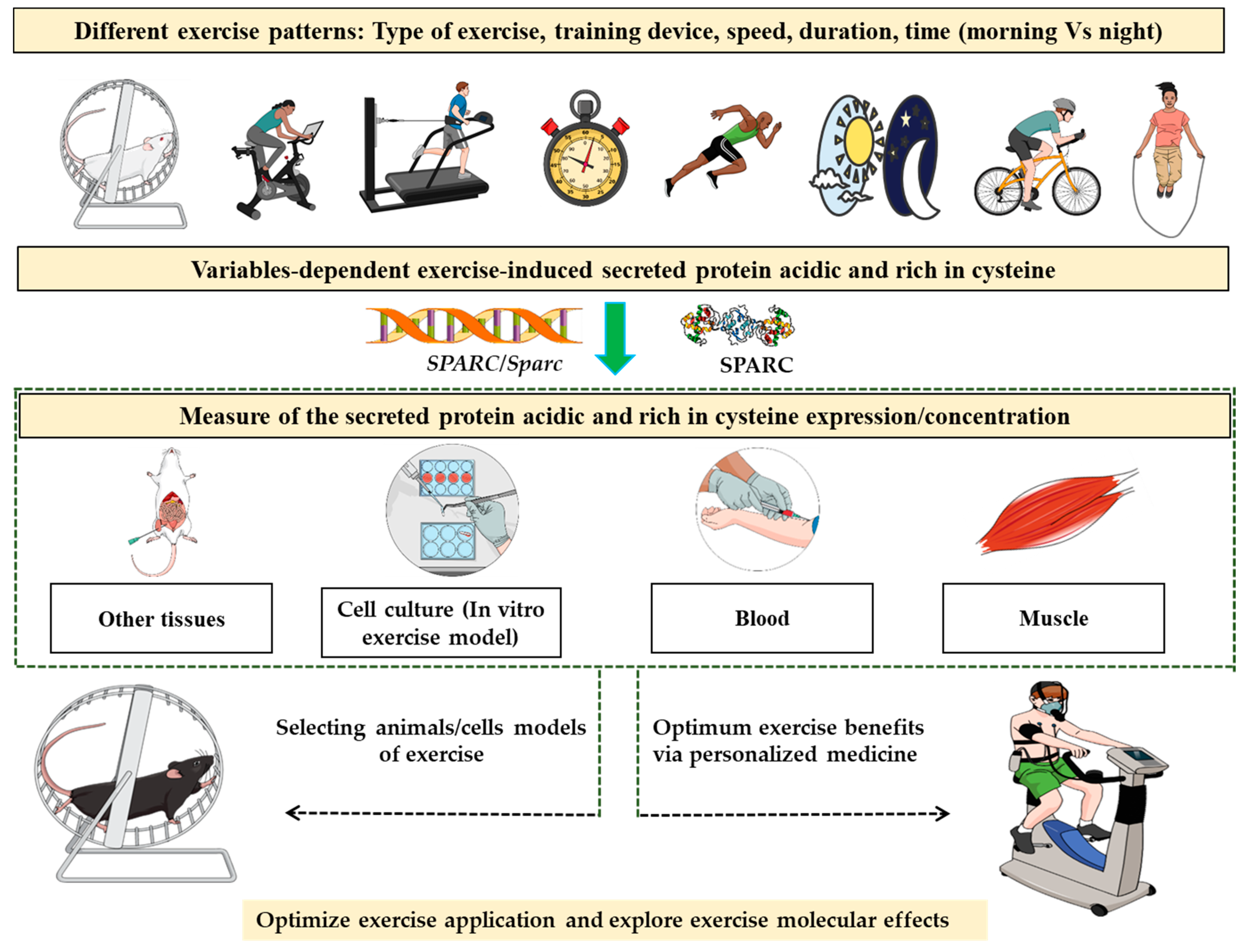
Measuring Exercise-Induced Secreted Protein Acidic and Rich in Cysteine Expression as a Molecular Tool to Optimize Personalized Medicine
Abstract
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Hosker, D.K.; Elkins, R.M.; Potter, M.P. Promoting Mental Health and Wellness in Youth Through Physical Activity, Nutrition, and Sleep. Child. Adolesc. Psychiatr. Clin. N. Am. 2019, 28, 171–193. [Google Scholar] [CrossRef] [PubMed]
- Peluso, M.A.; de Andrade, L.H.G. Physical activity and mental health: The association between exercise and mood. Clinics 2005, 60, 61–70. [Google Scholar] [CrossRef]
- Myers, J.; McAuley, P.; Lavie, C.J.; Despres, J.P.; Arena, R.; Kokkinos, P. Physical activity and cardiorespiratory fitness as major markers of cardiovascular risk: Their independent and interwoven importance to health status. Prog. Cardiovasc. Dis. 2015, 57, 306–314. [Google Scholar] [CrossRef]
- Katzmarzyk, P.T.; Ross, R.; Blair, S.N.; Després, J.P. Should we target increased physical activity or less sedentary behavior in the battle against cardiovascular disease risk development? Atherosclerosis 2020, 311, 107–115. [Google Scholar] [CrossRef]
- Ghanemi, A.; Yoshioka, M.; St-Amand, J. Obese Animals as Models for Numerous Diseases: Advantages and Applications. Medicina 2021, 57, 399. [Google Scholar] [CrossRef]
- Ghanemi, A.; St-Amand, J. Redefining obesity toward classifying as a disease. Eur. J. Intern. Med. 2018, 55, 20–22. [Google Scholar] [CrossRef]
- Ghanemi, A.; Yoshioka, M.; St-Amand, J. Broken Energy Homeostasis and Obesity Pathogenesis: The Surrounding Concepts. J. Clin. Med. 2018, 7, 453. [Google Scholar] [CrossRef]
- Ghanemi, A.; Yoshioka, M.; St-Amand, J. Obesity as a Neuroendocrine Reprogramming. Medicina 2021, 57, 66. [Google Scholar] [CrossRef] [PubMed]
- Owen, P.J.; Miller, C.T.; Mundell, N.L.; Verswijveren, S.; Tagliaferri, S.D.; Brisby, H.; Bowe, S.J.; Belavy, D.L. Which specific modes of exercise training are most effective for treating low back pain? Network meta-analysis. Br. J. Sports Med. 2020, 54, 1279–1287. [Google Scholar] [CrossRef]
- Ghanemi, A.; Melouane, A.; Yoshioka, M.; St-Amand, J. Exercise and High-Fat Diet in Obesity: Functional Genomics Perspectives of Two Energy Homeostasis Pillars. Genes 2020, 11, 875. [Google Scholar] [CrossRef] [PubMed]
- Wilkinson, T.J.; Shur, N.F.; Smith, A.C. “Exercise as medicine” in chronic kidney disease. Scand. J. Med. Sci. Sports 2016, 26, 985–988. [Google Scholar] [CrossRef]
- Ghanemi, A.; Yoshioka, M.; St-Amand, J. Regeneration during Obesity: An Impaired Homeostasis. Animals 2020, 10, 2344. [Google Scholar] [CrossRef]
- Idorn, M.; Straten, P.T. Exercise and cancer: From “healthy” to “therapeutic”? Cancer Immunol. Immunother. 2017, 66, 667–671. [Google Scholar] [CrossRef]
- Balducci, S.; Sacchetti, M.; Haxhi, J.; Orlando, G.; D’Errico, V.; Fallucca, S.; Menini, S.; Pugliese, G. Physical exercise as therapy for type 2 diabetes mellitus. Diabetes Metab. Res. Rev. 2014, 30, 13–23. [Google Scholar] [CrossRef] [PubMed]
- Simpson, R.J.; Kunz, H.; Agha, N.; Graff, R. Exercise and the Regulation of Immune Functions. Prog. Mol. Biol. Transl. Sci. 2015, 135, 355–380. [Google Scholar] [PubMed]
- Ghanemi, A.; Yoshioka, M.; St-Amand, J. Coronavirus Disease 2019 (COVID-19) Crisis: Losing Our Immunity When We Need It the Most. Biology 2021, 10, 545. [Google Scholar] [CrossRef] [PubMed]
- Ghanemi, A.; Yoshioka, M.; St-Amand, J. Will an obesity pandemic replace the coronavirus disease-2019 (COVID-19) pandemic? Med. Hypotheses 2020, 144, 110042. [Google Scholar] [CrossRef]
- Sharman, J.E.; La Gerche, A.; Coombes, J.S. Exercise and cardiovascular risk in patients with hypertension. Am. J. Hypertens. 2015, 28, 147–158. [Google Scholar] [CrossRef] [PubMed]
- Benedetti, M.G.; Furlini, G.; Zati, A.; Mauro, G.L. The Effectiveness of Physical Exercise on Bone Density in Osteoporotic Patients. Biomed. Res. Int. 2018, 2018, 4840531. [Google Scholar] [CrossRef]
- Metsios, G.S.; Moe, R.H.; Kitas, G.D. Exercise and inflammation. Best Pract. Res. Clin. Rheumatol. 2020, 34, 101504. [Google Scholar] [CrossRef] [PubMed]
- Ströhle, A. Sports psychiatry: Mental health and mental disorders in athletes and exercise treatment of mental disorders. Eur. Arch. Psychiatry Clin. Neurosci. 2019, 269, 485–498. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhang, H.; Ma, X.; Di, Q. Mental Health Problems during the COVID-19 Pandemics and the Mitigation Effects of Exercise: A Longitudinal Study of College Students in China. Int. J. Environ. Res. Public Health 2020, 17, 3722. [Google Scholar]
- Mikkelsen, K.; Stojanovska, L.; Polenakovic, M.; Bosevski, M.; Apostolopoulos, V. Exercise and mental health. Maturitas 2017, 106, 48–56. [Google Scholar] [CrossRef]
- Jurado-Fasoli, L.; De-la, O.A.; Molina-Hidalgo, C.; Migueles, J.H.; Castillo, M.J.; Amaro-Gahete, F.J. Exercise training improves sleep quality: A randomized controlled trial. Eur. J. Clin. Investig. 2020, 50, e13202. [Google Scholar] [CrossRef]
- Yang, P.Y.; Ho, K.H.; Chen, H.C.; Chien, M.Y. Exercise training improves sleep quality in middle-aged and older adults with sleep problems: A systematic review. J. Physiother. 2012, 58, 157–163. [Google Scholar] [CrossRef]
- Kwon, J.H.; Moon, K.M.; Min, K.-W. Exercise-Induced Myokines can Explain the Importance of Physical Activity in the Elderly: An Overview. Healthcare 2020, 8, 378. [Google Scholar] [CrossRef] [PubMed]
- Aoi, W.; Sakuma, K. Skeletal muscle: Novel and intriguing characteristics as a secretory organ. BioDiscovery 2013, 7. [Google Scholar] [CrossRef]
- Aoi, W.; Hirano, N.; Lassiter, D.G.; Björnholm, M.; Chibalin, A.V.; Sakuma, K.; Tanimura, Y.; Mizushima, K.; Takagi, T.; Naito, Y.; et al. Secreted protein acidic and rich in cysteine (SPARC) improves glucose tolerance via AMP-activated protein kinase activation. Faseb J. 2019, 33, 10551–10562. [Google Scholar] [CrossRef]
- Aoi, W.; Naito, Y.; Takagi, T.; Tanimura, Y.; Takanami, Y.; Kawai, Y.; Sakuma, K.; Hang, L.P.; Mizushima, K.; Hirai, Y.; et al. A novel myokine, secreted protein acidic and rich in cysteine (SPARC), suppresses colon tumorigenesis via regular exercise. Gut 2013, 62, 882–889. [Google Scholar] [CrossRef]
- Melouane, A.; Ghanemi, A.; Aubé, S.; Yoshioka, M.; St-Amand, J. Differential gene expression analysis in ageing muscle and drug discovery perspectives. Ageing Res. Rev. 2018, 41, 53–63. [Google Scholar] [CrossRef]
- Melouane, A.; Ghanemi, A.; Yoshioka, M.; St-Amand, J. Functional genomics applications and therapeutic implications in sarcopenia. Mutat. Res. Rev. Mutat. Res. 2019, 781, 175–185. [Google Scholar] [CrossRef] [PubMed]
- Riedl, I.; Yoshioka, M.; Nishida, Y.; Tobina, T.; Paradis, R.; Shono, N.; Tanaka, H.; St-Amand, J. Regulation of skeletal muscle transcriptome in elderly men after 6 weeks of endurance training at lactate threshold intensity. Exp. Gerontol. 2010, 45, 896–903. [Google Scholar] [CrossRef] [PubMed]
- Scheler, M.; Irmler, M.; Lehr, S.; Hartwig, S.; Staiger, H.; Al-Hasani, H.; Beckers, J.; de Angelis, M.H.; Häring, H.U.; Weigert, C. Cytokine response of primary human myotubes in an in vitro exercise model. Am. J. Physiol. Cell Physiol. 2013, 305, C877–C886. [Google Scholar] [CrossRef] [PubMed]
- Nikolić, N.; Bakke, S.S.; Kase, E.T.; Rudberg, I.; Halle, I.F.; Rustan, A.C.; Thoresen, G.H.; Aas, V. Correction: Electrical Pulse Stimulation of Cultured Human Skeletal Muscle Cells as an In Vitro Model of Exercise. PLoS ONE 2013, 8, e33203. [Google Scholar] [CrossRef] [PubMed]
- Tarum, J.; Folkesson, M.; Atherton, P.J.; Kadi, F. Electrical pulse stimulation: An in vitro exercise model for the induction of human skeletal muscle cell hypertrophy. A proof-of-concept study. Exp. Physiol. 2017, 102, 1405–1413. [Google Scholar] [CrossRef] [PubMed]
- Feng, Y.Z.; Nikolić, N.; Bakke, S.S.; Kase, E.T.; Guderud, K.; Hjelmesæth, J.; Aas, V.; Rustan, A.C.; Thoresen, G.H. Myotubes from lean and severely obese subjects with and without type 2 diabetes respond differently to an in vitro model of exercise. Am. J. Physiol. Cell. Physiol. 2015, 308, C548–C556. [Google Scholar] [CrossRef]
- Melouane, A.; Yoshioka, M.; Kanzaki, M.; St-Amand, J. Sparc, an EPS-induced gene, modulates the extracellular matrix and mitochondrial function via ILK/AMPK pathways in C2C12 cells. Life Sci. 2019, 229, 277–287. [Google Scholar] [CrossRef]
- Melouane, A.; Carbonell, A.; Yoshioka, M.; Puymirat, J.; St-Amand, J. Implication of SPARC in the modulation of the extracellular matrix and mitochondrial function in muscle cells. PLoS ONE 2018, 13, e0192714. [Google Scholar] [CrossRef] [PubMed]
- Ghanemi, A.; Melouane, A.; Yoshioka, M.; St-Amand, J. Exercise Training of Secreted Protein Acidic and Rich in Cysteine (Sparc) KO Mice Suggests That Exercise-Induced Muscle Phenotype Changes Are SPARC-Dependent. Appl. Sci. 2020, 10, 9108. [Google Scholar] [CrossRef]
- Phan, E.; Ahluwalia, A.; Tarnawski, A.S. Role of SPARC—Matricellular protein in pathophysiology and tissue injury healing. Implications for gastritis and gastric ulcers. Med. Sci. Monit. 2007, 13, RA25–RA30. [Google Scholar]
- Harries, L.W.; McCulloch, L.J.; Holley, J.E.; Rawling, T.J.; Welters, H.J.; Kos, K. A role for SPARC in the moderation of human insulin secretion. PLoS ONE 2013, 8, e68253. [Google Scholar] [CrossRef] [PubMed]
- Hu, L.; He, F.; Huang, M.; Zhao, Q.; Cheng, L.; Said, N.; Zhou, Z.; Liu, F.; Dai, Y.S. SPARC promotes insulin secretion through down-regulation of RGS4 protein in pancreatic β cells. Sci. Rep. 2020, 10, 17581. [Google Scholar] [CrossRef] [PubMed]
- Ghanemi, A.; Melouane, A.; Yoshioka, M.; St-Amand, J. Secreted protein acidic and rich in cysteine and bioenergetics: Extracellular matrix, adipocytes remodeling and skeletal muscle metabolism. Int. J. Biochem. Cell Biol. 2019, 117, 105627. [Google Scholar] [CrossRef]
- Ghanemi, A.; Yoshioka, M.; St-Amand, J. Secreted Protein Acidic and Rich in Cysteine: Metabolic and Homeostatic Properties beyond the Extracellular Matrix Structure. Appl. Sci. 2020, 10, 2388. [Google Scholar] [CrossRef]
- Rosset, E.M.; Bradshaw, A.D. SPARC/osteonectin in mineralized tissue. Matrix Biol. 2016, 52–54, 78–87. [Google Scholar] [CrossRef]
- Ghanemi, A.; Yoshioka, M.; St-Amand, J. Secreted Protein Acidic and Rich in Cysteine as A Regeneration Factor: Beyond the Tissue Repair. Life 2021, 11, 38. [Google Scholar] [CrossRef]
- Petersson, S.J.; Jørgensen, L.H.; Andersen, D.C.; Nørgaard, R.C.; Jensen, C.H.; Schrøder, H.D. SPARC is up-regulated during skeletal muscle regeneration and inhibits myoblast differentiation. Histol. Histopathol. 2013, 28, 1451–1460. [Google Scholar]
- Jørgensen, L.H.; Petersson, S.J.; Sellathurai, J.; Andersen, D.C.; Thayssen, S.; Sant, D.J.; Jensen, C.H.; Schrøder, H.D. Secreted protein acidic and rich in cysteine (SPARC) in human skeletal muscle. J. Histochem. Cytochem. 2009, 57, 29–39. [Google Scholar] [CrossRef]
- Ghanemi, A.; Yoshioka, M.; St-Amand, J. Secreted protein acidic and rich in cysteine and cancer: A homeostatic hormone? Cytokine 2020, 127, 154996. [Google Scholar] [CrossRef] [PubMed]
- Ghanemi, A.; Yoshioka, M.; St-Amand, J. Secreted protein acidic and rich in cysteine and inflammation: Another homeostatic property? Cytokine 2020, 133, 155179. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.H.; Jun, H.S. Role of Myokines in Regulating Skeletal Muscle Mass and Function. Front. Physiol. 2019, 10, 42. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.S.; Galvão, D.A.; Newton, R.U.; Gray, E.; Taaffe, D.R. Exercise-induced myokines and their effect on prostate cancer. Nat. Rev. Urol. 2021, 18, 519–542. [Google Scholar] [CrossRef]
- Joyce, N.C.; Oskarsson, B.; Jin, L.W. Muscle biopsy evaluation in neuromuscular disorders. Phys. Med. Rehabil. Clin. N. Am. 2012, 23, 609–631. [Google Scholar] [CrossRef]
- Pearl, G.S.; Ghatak, N.R. Muscle biopsy. Arch. Pathol. Lab. Med. 1995, 119, 303–306. [Google Scholar]
- Garneau, L.; Parsons, S.A.; Smith, S.R.; Mulvihill, E.E.; Sparks, L.M.; Aguer, C. Plasma Myokine Concentrations After Acute Exercise in Non-obese and Obese Sedentary Women. Front. Physiol. 2020, 11, 18. [Google Scholar] [CrossRef] [PubMed]
- Brown, J.C.; Damjanov, N.; Courneya, K.S.; Troxel, A.B.; Zemel, B.S.; Rickels, M.R.; Ky, B.; Rhim, A.D.; Rustgi, A.K.; Schmitz, K.H. A randomized dose-response trial of aerobic exercise and health-related quality of life in colon cancer survivors. Psychooncology 2018, 27, 1221–1228. [Google Scholar] [CrossRef] [PubMed]
- Dunn, A.L.; Trivedi, M.H.; Kampert, J.B.; Clark, C.G. Chambliss HO: Exercise treatment for depression: Efficacy and dose response. Am. J. Prev. Med. 2005, 28, 1–8. [Google Scholar] [CrossRef]
- Malliaras, P.; Johnston, R.; Street, G.; Littlewood, C.; Bennell, K.; Haines, T.; Buchbinder, R. The Efficacy of Higher Versus Lower Dose Exercise in Rotator Cuff Tendinopathy: A Systematic Review of Randomized Controlled Trials. Arch. Phys. Med. Rehabil. 2020, 101, 1822–1834. [Google Scholar] [CrossRef] [PubMed]
- Miyamoto, T.; Shimizu, Y.; Matsuo, Y.; Otaru, T.; Kanzawa, Y.; Miyamae, N.; Yamada, E.; Katsuno, T. Effects of exercise intensity and duration on a myokine, secreted protein acidic and rich in cysteine. Eur. J. Sport Sci. 2021, 8, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Songsorn, P.; Ruffino, J.; Vollaard, N.B. No effect of acute and chronic supramaximal exercise on circulating levels of the myokine SPARC. Eur. J. Sport Sci. 2017, 17, 447–452. [Google Scholar] [CrossRef] [PubMed]
- Galloza, J.; Castillo, B.; Micheo, W. Benefits of Exercise in the Older Population. Phys. Med. Rehabil. Clin. N. Am. 2017, 28, 659–669. [Google Scholar] [CrossRef]
- Lee, P.G.; Jackson, E.A.; Richardson, C.R. Exercise Prescriptions in Older Adults. Am. Fam. Physician. 2017, 95, 425–432. [Google Scholar] [PubMed]
- Baker, M.K.; Atlantis, E.; Singh, M.A.F. Multi-modal exercise programs for older adults. Age Ageing 2007, 36, 375–381. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, K.; Yamanouchi, K.; Nishihara, M. Transdisciplinary Approach for Sarcopenia. Molecular mechanism of sarcopenia: The role of skeletal muscle niche component SPARC in the regulation of myogenesis and adipogenesis and its alteration with age. Clin Calcium 2014, 24, 1471–1478. [Google Scholar]
- Omi, S.; Yamanouchi, K.; Nakamura, K.; Matsuwaki, T.; Nishihara, M. Reduced fibrillar collagen accumulation in skeletal muscle of secreted protein acidic and rich in cysteine (SPARC)-null mice. J. Vet. Med. Sci. 2019, 81, 1649–1654. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, K.; Yamanouchi, K.; Nishihara, M. Secreted protein acidic and rich in cysteine internalization and its age-related alterations in skeletal muscle progenitor cells. Aging Cell 2014, 13, 175–184. [Google Scholar] [CrossRef]
- Nakamura, K.; Nakano, S.; Miyoshi, T.; Yamanouchi, K.; Matsuwaki, T.; Nishihara, M. Age-related resistance of skeletal muscle-derived progenitor cells to SPARC may explain a shift from myogenesis to adipogenesis. Aging (Albany NY) 2012, 4, 40–48. [Google Scholar] [CrossRef]
- Song, H.; Ding, L.; Zhang, S.; Wang, W. MiR-29 family members interact with SPARC to regulate glucose metabolism. Biochem. Biophys. Res. Commun. 2018, 497, 667–674. [Google Scholar] [CrossRef]
- Kos, K.; Wilding, J.P. SPARC: A key player in the pathologies associated with obesity and diabetes. Nat. Rev. Endocrinol. 2010, 6, 225–235. [Google Scholar] [CrossRef]
- Pierrakos, C.; Vincent, J.L. Sepsis biomarkers: A review. Crit Care 2010, 14, R15. [Google Scholar] [CrossRef]
- Capecchi, R.; Puxeddu, I.; Pratesi, F.; Migliorini, P. New biomarkers in SLE: From bench to bedside. Rheumatology (Oxford) 2020, 59, v12–v18. [Google Scholar] [CrossRef]
- Kunc, P.; Fabry, J.; Lucanska, M.; Pecova, R. Biomarkers of Bronchial Asthma. Physiol. Res. 2020, 69, S29–S34. [Google Scholar] [CrossRef]
- Aronson, J.K.; Ferner, R.E. Biomarkers-A General Review. Curr. Protoc. Pharmacol. 2017, 76, 9–23. [Google Scholar] [CrossRef] [PubMed]
- Sandokji, I.; Greenberg, J.H. Novel biomarkers of acute kidney injury in children: An update on recent findings. Curr. Opin. Pediatr. 2020, 32, 354–359. [Google Scholar] [CrossRef]
- Kawata, K.; Tierney, R.; Langford, D. Blood and cerebrospinal fluid biomarkers. Handb. Clin. Neurol. 2018, 158, 217–233. [Google Scholar] [PubMed]
- Avramouli, A.; Vlamos, P. Buccal Mucosa Biomarkers in Alzheimer’s Disease. Adv. Exp. Med. Biol. 2020, 1195, 49–56. [Google Scholar]
- Zhou, L.T.; Lv, L.L.; Liu, B.C. Urinary Biomarkers of Renal Fibrosis. Adv. Exp. Med. Biol. 2019, 1165, 607–623. [Google Scholar]
- Shen, L.; Liu, X.; Zhang, H.; Lin, J.; Feng, C.; Iqbal, J. Biomarkers in autism spectrum disorders: Current progress. Clin. Chim. Acta. 2020, 502, 41–54. [Google Scholar] [CrossRef] [PubMed]
- Steckl, A.J.; Ray, P. Stress Biomarkers in Biological Fluids and Their Point-of-Use Detection. ACS Sens. 2018, 3, 2025–2044. [Google Scholar] [CrossRef]
- Gururajan, A.; Clarke, G.; Dinan, T.G.; Cryan, J.F. Molecular biomarkers of depression. Neurosci. Biobehav. Rev. 2016, 64, 101–133. [Google Scholar] [CrossRef]
- Watt, F.E. Osteoarthritis biomarkers: Year in review. Osteoarthritis Cartilage 2018, 26, 312–318. [Google Scholar] [CrossRef]
- Chen, X.H.; Huang, S.; Kerr, D. Biomarkers in clinical medicine. IARC Sci. Publ. 2011, 303–322. [Google Scholar]
- Jing, J.; Gao, Y. Urine biomarkers in the early stages of diseases: Current status and perspective. Discov. Med. 2018, 25, 57–65. [Google Scholar]
- Agah, E.; Saleh, F.; Sanjari, M.H.; Saghazadeh, A.; Tafakhori, A.; Rezaei, N. CSF and blood biomarkers in amyotrophic lateral sclerosis: Protocol for a systematic review and meta-analysis. Syst. Rev. 2018, 7, 237. [Google Scholar] [CrossRef]
- Bhide, A.A.; Cartwright, R.; Khullar, V.; Digesu, G.A. Biomarkers in overactive bladder. Int. Urogynecol. J. 2013, 24, 1065–1072. [Google Scholar] [CrossRef]
- Ahn, M.H.; Kim, H.A. Biomarkers for adult-onset still’s disease: Up-to-date. Expert. Rev. Mol. Diagn. 2019, 19, 655–657. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Modur, V.; Carayannopoulos, L.N.; Laterza, O.F. Biomarkers in Pharmaceutical Research. Clin. Chem. 2015, 61, 1343–1353. [Google Scholar] [CrossRef] [PubMed]
- Ghanemi, A.; Yoshioka, M.; St-Amand, J. Secreted Protein Acidic and Rich in Cysteine as a Molecular Physiological and Pathological Biomarker. Biomolecules 2021, 11, 1689. [Google Scholar] [CrossRef]
- Curigliano, G. Personalizing medicine through personalized communication: Individuality of the patient across borders and cultures. J. Natl. Compr. Canc. Netw. 2010, 8, 277–278. [Google Scholar] [CrossRef]
- Ziegelstein, R.C. Perspectives in Primary Care: Knowing the Patient as a Person in the Precision Medicine Era. Ann. Fam. Med. 2018, 16, 4–5. [Google Scholar] [CrossRef]
- Erden, A. Personalized Medicine: How Studying Individual Differences Can Shed Light on Treating Thousands of Patients: An Interview with Richard Lifton, PhD. Yale. J. Biol. Med. 2015, 88, 407–411. [Google Scholar]
- Abettan, C.; Welie, J.V.M. The impact of twenty-first century personalized medicine versus twenty-first century medicine’s impact on personalization. Philos. Ethics Humanit Med. 2020, 15, 11. [Google Scholar] [CrossRef] [PubMed]
- Boersma, G.J.; Benthem, L.; van Beek, A.P.; van Dijk, G.; Scheurink, A.J. Personality, a key factor in personalized medicine? Eur. J. Pharmacol. 2011, 667, 23–25. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Khatri, V.P.; Petrelli, N.J. Precision Medicine. Surg. Oncol. Clin. N. Am. 2020, 29, xv–xvi. [Google Scholar] [CrossRef] [PubMed]
- Ramaswami, R.; Bayer, R.; Galea, S. Precision Medicine from a Public Health Perspective. Annu. Rev. Public Health 2018, 39, 153–168. [Google Scholar] [CrossRef]
- Beckmann, J.S.; Lew, D. Reconciling evidence-based medicine and precision medicine in the era of big data: Challenges and opportunities. Genome Med. 2016, 8, 134. [Google Scholar] [CrossRef]
- Antman, E.M.; Loscalzo, J. Precision medicine in cardiology. Nat. Rev. Cardiol. 2016, 13, 591–602. [Google Scholar] [CrossRef] [PubMed]
- Weil, A.R. Precision Medicine. Health Aff. (Millwood) 2018, 37, 687. [Google Scholar] [CrossRef]
- Xie, F.; Chan, J.C.; Ma, R.C. Precision medicine in diabetes prevention, classification and management. J. Diabetes Investig. 2018, 9, 998–1015. [Google Scholar] [CrossRef]
- Forrest, S.J.; Geoerger, B.; Janeway, K.A. Precision medicine in pediatric oncology. Curr. Opin. Pediatr. 2018, 30, 17–24. [Google Scholar] [CrossRef] [PubMed]
- König, I.R.; Fuchs, O.; Hansen, G.; von Mutius, E.; Kopp, M.V. What is precision medicine? Eur. Respir. J. 2017, 50. [Google Scholar] [CrossRef]
- Yardley, J.E.; Campbell, M.D. Moving Toward Precision Medicine with Diabetes, Exercise and Physical Activity. Can. J. Diabetes 2020, 44, 679. [Google Scholar] [CrossRef] [PubMed]
- Bonafiglia, J.T.; Ramic, L.; Islam, H. Challenges to personalized exercise medicine: Lack of repeatability and influence of genetics. J. Physiol. 2020, 598, 5021–5023. [Google Scholar] [CrossRef] [PubMed]
- Buford TW, Roberts MD, Church TS: Toward exercise as personalized medicine. Sports Med. 2013, 43, 157–165. [CrossRef] [PubMed]
- Garner, S.; Fenton, T.; Martin, L.; Creaser, C.; Johns, C.; Barnabe, C. Personalized diet and exercise recommendations in early rheumatoid arthritis: A feasibility trial. Musculoskeletal Care 2018, 16, 167–172. [Google Scholar]
- Kostek, M. Precision Medicine and Exercise Therapy in Duchenne Muscular Dystrophy. Sports (Basel) 2019, 7, 64. [Google Scholar] [CrossRef]
- Ross, R.; Goodpaster, B.H.; Koch, L.G.; Sarzynski, M.A.; Kohrt, W.M.; Johannsen, N.M.; Skinner, J.S.; Castro, A.; Irving, B.A.; Noland, R.C.; et al. Precision exercise medicine: Understanding exercise response variability. Br. J. Sports Med. 2019, 53, 1141–1153. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ghanemi, A.; Yoshioka, M.; St-Amand, J. Measuring Exercise-Induced Secreted Protein Acidic and Rich in Cysteine Expression as a Molecular Tool to Optimize Personalized Medicine. Genes 2021, 12, 1832. https://doi.org/10.3390/genes12111832
Ghanemi A, Yoshioka M, St-Amand J. Measuring Exercise-Induced Secreted Protein Acidic and Rich in Cysteine Expression as a Molecular Tool to Optimize Personalized Medicine. Genes. 2021; 12(11):1832. https://doi.org/10.3390/genes12111832
Chicago/Turabian StyleGhanemi, Abdelaziz, Mayumi Yoshioka, and Jonny St-Amand. 2021. "Measuring Exercise-Induced Secreted Protein Acidic and Rich in Cysteine Expression as a Molecular Tool to Optimize Personalized Medicine" Genes 12, no. 11: 1832. https://doi.org/10.3390/genes12111832
APA StyleGhanemi, A., Yoshioka, M., & St-Amand, J. (2021). Measuring Exercise-Induced Secreted Protein Acidic and Rich in Cysteine Expression as a Molecular Tool to Optimize Personalized Medicine. Genes, 12(11), 1832. https://doi.org/10.3390/genes12111832






