Sex Chromosomes and Master Sex-Determining Genes in Turtles and Other Reptiles
Abstract
1. Introduction
2. Overview of GSD in Reptiles
2.1. Turtles
2.1.1. Pleurodiran Turtles
2.1.2. Cryptodiran Turtles
Trionychia
Durocryptodira
- Americhelydia
- Testudinoidea
2.1.3. General Considerations
2.2. Lizards
2.2.1. Dibamidae
2.2.2. Gekkota
2.2.3. Scinciformata
2.2.4. Laterata
2.2.5. Toxicofera
Anguimorpha
Iguana
- Anodonta
- Pleurodonta
2.3. Snakes
2.3.1. Caenophidia
2.3.2. Other Snakes
3. The Lessons from the Study of Sex Chromosomes in Reptiles
3.1. What We Learned
3.2. What Is Still to Be Discovered and How It Could Be Carried Out
4. Conclusions
Supplementary Materials
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Imarazene, B.; Du, K.; Beille, S.; Jouanno, E.; Feron, R.; Pan, Q.; Torres-Paz, J.; Lopez-Roques, C.; Castinel, A.; Gil, L.; et al. A supernumerary “B-sex” chromosome drives male sex determination in the Pachón cavefish, Astyanax mexicanus. Curr. Biol. 2021, 31, 4800–4809. [Google Scholar] [CrossRef] [PubMed]
- Ponnikas, S.; Sigeman, H.; Abbott, J.K.; Hansson, B. Why Do Sex Chromosomes Stop Recombining? Trends Genet. 2018, 34, 492–503. [Google Scholar] [CrossRef] [PubMed]
- Sinclair, A.H.; Berta, P.; Palmer, M.S.; Hawkins, J.R.; Griffiths, B.L.; Smith, M.J.; Foster, J.W.; Frischauf, A.M.; Lovell-Badge, R.; Goodfellow, P.N. A gene from the human sex-determining region encodes a protein with homology to a conserved DNA-binding motif. Nature 1990, 346, 240–244. [Google Scholar] [CrossRef] [PubMed]
- Koopman, P.; Gubbay, J.; Vivian, N.; Goodfellow, P.; Lovell-Badge, R. Male development of chromosomally female mice transgenic for Sry. Nature 1991, 351, 117–121. [Google Scholar] [CrossRef] [PubMed]
- Takehana, Y.; Matsuda, M.; Myosho, T.; Suster, M.L.; Kawakami, K.; Shin-I, T.; Kohara, Y.; Kuroki, Y.; Toyoda, A.; Fujiyama, A.; et al. Co-option of Sox3 as the male-determining factor on the Y chromosome in the fish Oryzias dancena. Nat. Commun. 2014, 5, 4157. [Google Scholar] [CrossRef]
- Smith, C.A.; Roeszler, K.N.; Ohnesorg, T.; Cummins, D.M.; Farlie, P.G.; Doran, T.J.; Sinclair, A.H. The avian Z-linked gene DMRT1 is required for male sex determination in the chicken. Nature 2009, 461, 267–271. [Google Scholar] [CrossRef]
- Ioannidis, J.; Taylor, G.; Zhao, D.; Liu, L.; Idoko-Akoh, A.; Gong, D.; Lovell-Badge, R.; Guioli, S.; McGrew, M.J.; Clinton, M. Primary sex determination in birds depends on DMRT1 dosage, but gonadal sex does not determine adult secondary sex characteristics. Proc. Natl. Acad. Sci. USA 2021, 118, e2020909118. [Google Scholar] [CrossRef]
- Chen, S.; Zhang, G.; Shao, C.; Huang, Q.; Liu, G.; Zhang, P.; Song, W.; An, N.; Chalopin, D.; Volff, J.-N.; et al. Whole-genome sequence of a flatfish provides insights into ZW sex chromosome evolution and adaptation to a benthic lifestyle. Nat. Genet. 2014, 46, 253–260. [Google Scholar] [CrossRef]
- Yoshimoto, S.; Okada, E.; Umemoto, H.; Tamura, K.; Uno, Y.; Nishida-Umehara, C.; Matsuda, Y.; Takamatsu, N.; Shiba, T.; Ito, M. A W-linked DM-domain gene, DM-W, participates in primary ovary development in Xenopus laevis. Proc. Natl. Acad. Sci. USA 2008, 105, 2469–2474. [Google Scholar] [CrossRef]
- Matsuda, M.; Nagahama, Y.; Shinomiya, A.; Sato, T.; Matsuda, C.; Kobayashi, T.; Morrey, C.E.; Shibata, N.; Asakawa, S.; Shimizu, N.; et al. DMY is a Y-specific DM-domain gene required for male development in the medaka fish. Nature 2002, 417, 559–563. [Google Scholar] [CrossRef]
- Nanda, I.; Kondo, M.; Hornung, U.; Asakawa, S.; Winkler, C.; Shimizu, A.; Shan, Z.; Haaf, T.; Shimizu, N.; Shima, A.; et al. A duplicated copy of DMRT1 in the sex-determining region of the Y chromosome of the medaka, Oryzias latipes. Proc. Natl. Acad. Sci. USA 2002, 99, 11778–11783. [Google Scholar] [CrossRef]
- Matsuda, M.; Sato, T.; Toyazaki, Y.; Nagahama, Y.; Hamaguchi, S.; Sakaizumi, M. Oryzias curvinotus Has DMY, a Gene That Is Required for Male Development in the Medaka, O. latipes. Zoolog. Sci. 2003, 20, 159–161. [Google Scholar] [CrossRef]
- Myosho, T.; Otake, H.; Masuyama, H.; Matsuda, M.; Kuroki, Y.; Fujiyama, A.; Naruse, K.; Hamaguchi, S.; Sakaizumi, M. Tracing the Emergence of a Novel Sex-Determining Gene in Medaka, Oryzias luzonensis. Genetics 2012, 191, 163–170. [Google Scholar] [CrossRef] [PubMed]
- Reichwald, K.; Petzold, A.; Koch, P.; Downie, B.R.; Hartmann, N.; Pietsch, S.; Baumgart, M.; Chalopin, D.; Felder, M.; Bens, M.; et al. Insights into Sex Chromosome Evolution and Aging from the Genome of a Short-Lived Fish. Cell 2015, 163, 1527–1538. [Google Scholar] [CrossRef] [PubMed]
- Hattori, R.S.; Murai, Y.; Oura, M.; Masuda, S.; Majhi, S.K.; Sakamoto, T.; Fernandino, J.I.; Somoza, G.M.; Yokota, M.; Strüssmann, C.A. A Y-linked anti-Müllerian hormone duplication takes over a critical role in sex determination. Proc. Natl. Acad. Sci. USA 2012, 109, 2955–2959. [Google Scholar] [CrossRef]
- Pan, Q.; Feron, R.; Yano, A.; Guyomard, R.; Jouanno, E.; Vigouroux, E.; Wen, M.; Busnel, J.-M.; Bobe, J.; Concordet, J.-P.; et al. Identification of the master sex determining gene in Northern pike (Esox lucius) reveals restricted sex chromosome differentiation. PLoS Genet. 2019, 15, e1008013. [Google Scholar] [CrossRef] [PubMed]
- Peichel, C.L.; McCann, S.R.; Ross, J.A.; Naftaly, A.F.S.; Urton, J.R.; Cech, J.N.; Grimwood, J.; Schmutz, J.; Myers, R.M.; Kingsley, D.M.; et al. Assembly of the threespine stickleback Y chromosome reveals convergent signatures of sex chromosome evolution. Genome Biol. 2020, 21, 177. [Google Scholar] [CrossRef]
- Kamiya, T.; Kai, W.; Tasumi, S.; Oka, A.; Matsunaga, T.; Mizuno, N.; Fujita, M.; Suetake, H.; Suzuki, S.; Hosoya, S.; et al. A Trans-Species Missense SNP in Amhr2 Is Associated with Sex Determination in the Tiger Pufferfish, Takifugu rubripes (Fugu). PLoS Genet. 2012, 8, e1002798. [Google Scholar] [CrossRef] [PubMed]
- Feron, R.; Zahm, M.; Cabau, C.; Klopp, C.; Roques, C.; Bouchez, O.; Eché, C.; Valière, S.; Donnadieu, C.; Haffray, P.; et al. Characterization of a Y-specific duplication/insertion of the anti-Mullerian Hormone type II receptor gene based on a chromosome-scale genome assembly of yellow perch, Perca flavescens. Mol. Ecol. Resour. 2020, 20, 531–543. [Google Scholar] [CrossRef]
- Nakamoto, M.; Uchino, T.; Koshimizu, E.; Kuchiishi, Y.; Sekiguchi, R.; Wang, L.; Sudo, R.; Endo, M.; Guiguen, Y.; Schartl, M.; et al. A Y-linked anti-Müllerian hormone type-II receptor is the sex-determining gene in ayu, Plecoglossus altivelis. PLoS Genet. 2021, 17, e1009705. [Google Scholar] [CrossRef]
- Cortez, D.; Marin, R.; Toledo-Flores, D.; Froidevaux, L.; Liechti, A.; Waters, P.D.; Grützner, F.; Kaessmann, H. Origins and Functional Evolution of Y Chromosomes across Mammals. Nature 2014, 508, 488–493. [Google Scholar] [CrossRef]
- Zhou, Y.; Shearwin-Whyatt, L.; Li, J.; Song, Z.; Hayakawa, T.; Stevens, D.; Fenelon, J.C.; Peel, E.; Cheng, Y.; Pajpach, F.; et al. Platypus and Echidna Genomes Reveal Mammalian Biology and Evolution. Nature 2021, 592, 756–762. [Google Scholar] [CrossRef]
- Graham, P.; Penn, J.K.M.; Schedl, P. Masters Change, Slaves Remain. BioEssays 2003, 25, 1–4. [Google Scholar] [CrossRef]
- Yano, A.; Guyomard, R.; Nicol, B.; Jouanno, E.; Quillet, E.; Klopp, C.; Cabau, C.; Bouchez, O.; Fostier, A.; Guiguen, Y. An Immune-Related Gene Evolved into the Master Sex-Determining Gene in Rainbow Trout, Oncorhynchus mykiss. Curr. Biol. 2012, 22, 1423–1428. [Google Scholar] [CrossRef]
- Bertho, S.; Herpin, A.; Branthonne, A.; Jouanno, E.; Yano, A.; Nicol, B.; Muller, T.; Pannetier, M.; Pailhoux, E.; Miwa, M.; et al. The Unusual Rainbow Trout Sex Determination Gene Hijacked the Canonical Vertebrate Gonadal Differentiation Pathway. Proc. Natl. Acad. Sci. USA 2018, 115, 12781–12786. [Google Scholar] [CrossRef] [PubMed]
- Bao, L.; Tian, C.; Liu, S.; Zhang, Y.; Elaswad, A.; Yuan, Z.; Khalil, K.; Sun, F.; Yang, Y.; Zhou, T.; et al. The Y Chromosome Sequence of the Channel Catfish Suggests Novel Sex Determination Mechanisms in Teleost Fish. BMC Biol. 2019, 17, 6. [Google Scholar] [CrossRef]
- Keinath, M.C.; Timoshevskaya, N.; Timoshevskiy, V.A.; Voss, S.R.; Smith, J.J. Miniscule Differences between Sex Chromosomes in the Giant Genome of a Salamander. Sci. Rep. 2018, 8, 17882. [Google Scholar] [CrossRef] [PubMed]
- Ion, A.; Telvi, L.; Chaussain, J.L.; Galacteros, F.; Valayer, J.; Fellous, M.; McElreavey, K. A Novel Mutation in the Putative DNA Helicase XH2 Is Responsible for Male-to-Female Sex Reversal Associated with an Atypical Form of the ATR-X Syndrome. Am. J. Hum. Genet. 1996, 58, 1185–1191. [Google Scholar] [PubMed]
- Charnier, M. Action of temperature on the sex ratio in the Agama agama (Agamidae, Lacertilia) embryo. Comptes Rendus Seances Soc. Biol. Fil. 1966, 160, 620–622. [Google Scholar]
- Pieau, C. Sex ratio in the embryos of 2 chelonians (Testudo graeca L. and Emys orbicularis L.) born of artificially incubated ova. Comptes Rendus Hebd. Seances Acad. Sci. Ser. Sci. Nat. 1971, 272, 3071–3074. [Google Scholar]
- Zardoya, R.; Meyer, A. The Evolutionary Position of Turtles Revised. Naturwissenschaften 2001, 88, 193–200. [Google Scholar] [CrossRef][Green Version]
- Crawford, N.G.; Faircloth, B.C.; McCormack, J.E.; Brumfield, R.T.; Winker, K.; Glenn, T.C. More than 1000 Ultraconserved Elements Provide Evidence That Turtles Are the Sister Group of Archosaurs. Biol. Lett. 2012, 8, 783–786. [Google Scholar] [CrossRef] [PubMed]
- Shen, X.-X.; Liang, D.; Wen, J.-Z.; Zhang, P. Multiple Genome Alignments Facilitate Development of NPCL Markers: A Case Study of Tetrapod Phylogeny Focusing on the Position of Turtles. Mol. Biol. Evol. 2011, 28, 3237–3252. [Google Scholar] [CrossRef]
- Chiari, Y.; Cahais, V.; Galtier, N.; Delsuc, F. Phylogenomic Analyses Support the Position of Turtles as the Sister Group of Birds and Crocodiles (Archosauria). BMC Biol. 2012, 10, 65. [Google Scholar] [CrossRef]
- Shaffer, H.B.; Minx, P.; Warren, D.E.; Shedlock, A.M.; Thomson, R.C.; Valenzuela, N.; Abramyan, J.; Amemiya, C.T.; Badenhorst, D.; Biggar, K.K.; et al. The Western Painted Turtle Genome, a Model for the Evolution of Extreme Physiological Adaptations in a Slowly Evolving Lineage. Genome Biol. 2013, 14, 1–23. [Google Scholar] [CrossRef]
- Wang, Z.; Pascual-Anaya, J.; Zadissa, A.; Li, W.; Niimura, Y.; Huang, Z.; Li, C.; White, S.; Xiong, Z.; Fang, D.; et al. The Draft Genomes of Soft-Shell Turtle and Green Sea Turtle Yield Insights into the Development and Evolution of the Turtle-Specific Body Plan. Nat. Genet. 2013, 45, 701–706. [Google Scholar] [CrossRef] [PubMed]
- Crawford, N.G.; Parham, J.F.; Sellas, A.B.; Faircloth, B.C.; Glenn, T.C.; Papenfuss, T.J.; Henderson, J.B.; Hansen, M.H.; Simison, W.B. A Phylogenomic Analysis of Turtles. Mol. Phylogenet. Evol. 2015, 83, 250–257. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Y.; Wiens, J.J. Combining Phylogenomic and Supermatrix Approaches, and a Time-Calibrated Phylogeny for Squamate Reptiles (Lizards and Snakes) Based on 52 Genes and 4162 Species. Mol. Phylogenet. Evol. 2016, 94, 537–547. [Google Scholar] [CrossRef] [PubMed]
- Pereira, A.G.; Sterli, J.; Moreira, F.R.R.; Schrago, C.G. Multilocus Phylogeny and Statistical Biogeography Clarify the Evolutionary History of Major Lineages of Turtles. Mol. Phylogenet. Evol. 2017, 113, 59–66. [Google Scholar] [CrossRef]
- Shaffer, H.B.; McCartney-Melstad, E.; Near, T.J.; Mount, G.G.; Spinks, P.Q. Phylogenomic Analyses of 539 Highly Informative Loci Dates a Fully Resolved Time Tree for the Major Clades of Living Turtles (Testudines). Mol. Phylogenet. Evol. 2017, 115, 7–15. [Google Scholar] [CrossRef]
- Thomson, R.C.; Spinks, P.Q.; Shaffer, H.B. A Global Phylogeny of Turtles Reveals a Burst of Climate-Associated Diversification on Continental Margins. Proc. Natl. Acad. Sci. USA 2021, 118, e2012215118. [Google Scholar] [CrossRef] [PubMed]
- Deeming, D.C. Prevalence of TSD in Crocodilians. In Temperature Dependent Sex Determination in Vertebrates; Valenzuela, N., Lance, V.A., Eds.; Smithsonian Books: Whashington, DC, USA, 2004; pp. 33–41. [Google Scholar]
- Zhou, Q.; Zhang, J.; Bachtrog, D.; An, N.; Huang, Q.; Jarvis, E.D.; Gilbert, M.T.P.; Zhang, G. Complex Evolutionary Trajectories of Sex Chromosomes across Bird Taxa. Science 2014, 346, 1246338. [Google Scholar] [CrossRef] [PubMed]
- Gunski, R.J.; Cañedo, A.D.; Garnero, A.D.V.; Ledesma, M.A.; Coria, N.; Montalti, D.; Degrandi, T.M. Multiple Sex Chromosome System in Penguins (Pygoscelis, Spheniscidae). Comp. Cytogenet. 2017, 11, 541–552. [Google Scholar] [CrossRef] [PubMed]
- Sigeman, H.; Ponnikas, S.; Hansson, B. Whole-Genome Analysis across 10 Songbird Families within Sylvioidea Reveals a Novel Autosome-Sex Chromosome Fusion. Biol. Lett. 2020, 16, 20200082. [Google Scholar] [CrossRef]
- Küpper, C.; Augustin, J.; Edwards, S.; Székely, T.; Kosztolányi, A.; Burke, T.; Janes, D.E. Triploid Plover Female Provides Support for a Role of the W Chromosome in Avian Sex Determination. Biol. Lett. 2012, 8, 787–789. [Google Scholar] [CrossRef]
- Kuroiwa, A. Sex-Determining Mechanism in Avians. Adv. Exp. Med. Biol. 2017, 1001, 19–31. [Google Scholar] [CrossRef]
- Ewert, M.A.; Nelson, C.E. Sex Determination in Turtles: Diverse Patterns and Some Possible Adaptive Values. Copeia 1991, 1991, 50–69. [Google Scholar] [CrossRef]
- Janzen, F.J.; Phillips, P.C. Exploring the Evolution of Environmental Sex Determination, Especially in Reptiles. J. Evol. Biol. 2006, 19, 1775–1784. [Google Scholar] [CrossRef]
- Valenzuela, N.; Adams, D.C. Chromosome Number and Sex Determination Coevolve in Turtles. Evol. Int. J. Org. Evol. 2011, 65, 1808–1813. [Google Scholar] [CrossRef] [PubMed]
- Petzold, A.; Vargas-Ramírez, M.; Kehlmaier, C.; Vamberger, M.; Branch, W.R.; Preez, L.D.; Hofmeyr, M.D.; Meyer, L.; Schleicher, A.; Siroký, P.; et al. A Revision of African Helmeted Terrapins (Testudines: Pelomedusidae: Pelomedusa), with Descriptions of Six New Species. Zootaxa 2014, 3795, 523–548. [Google Scholar] [CrossRef]
- Fritz, U.; Branch, W.R.; Hofmeyr, M.D.; Maran, J.; Prokop, H.; Schleicher, A.; Širokỳ, P.; Stuckas, H.; Vargas-Ramírez, M.; Vences, M. Molecular Phylogeny of African Hinged and Helmeted Terrapins (Testudines: Pelomedusidae: Pelusios and Pelomedusa). Zool. Scr. 2011, 40, 115–125. [Google Scholar] [CrossRef]
- Noonan, B.P. Does the Phylogeny of Pelomedusoid Turtles Reflect Vicariance Due to Continental Drift? J. Biogeogr. 2000, 27, 1245–1249. [Google Scholar] [CrossRef]
- Vargas-Ramírez, M.; Castaño-Mora, O.V.; Fritz, U. Molecular Phylogeny and Divergence Times of Ancient South American and Malagasy River Turtles (Testudines: Pleurodira: Podocnemididae). Org. Divers. Evol. 2008, 8, 388–398. [Google Scholar] [CrossRef]
- Ayres, M.; Sampaio, M.M.; Barros, R.M.; Dias, L.B.; Cunha, O.R. A Karyological Study of Turtles from the Brazilian Amazon Region. Cytogenetics 1969, 8, 401–409. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.C.; Clark, H.F. Chromosome Studies of the Cultured Cells of Two Species of Side-Necked Turtles (Podocnemis unifilis and P. Expansa). Chromosoma 1969, 26, 245–253. [Google Scholar] [CrossRef] [PubMed]
- Killebrew, F.C. Mitotic Chromosomes of Turtles: I. the Pelomedusidae. J. Herpetol. 1975, 281–285. [Google Scholar] [CrossRef]
- Bull, J.J. Sex Determination in Reptiles. Q. Rev. Biol. 1980, 55, 3–21. [Google Scholar] [CrossRef]
- Fantin, C.; Monjeló, L.A.D.S. Cytogenetic Studies in Podocnemis Expansa and Podocnemis Sextuberculata (Testudines, Podocnemididae), Turtles of the Brazilian Amazon. Caryologia 2011, 64, 154–157. [Google Scholar] [CrossRef][Green Version]
- Gunski, R.J.; Cunha, I.S.; Degrandi, T.M.; Ledesma, M.; Garnero, A.D.V. Cytogenetic Comparison of Podocnemis expansa and Podocnemis unifilis: A Case of Inversion and Duplication Involving Constitutive Heterochromatin. Genet. Mol. Biol. 2013, 36, 353–356. [Google Scholar] [CrossRef]
- Ventura, K.; Moreira, C.N.; Moretti, R.; Yonenaga-Yassuda, Y.; Rodrigues, M.T. The Lowest Diploid Number in Testudines: Banding Patterns, Telomeric and 45S RDNA FISH in Peltocephalus dumerilianus, 2n = 26 and FN = 52 (Pleurodira, Podocnemididae). Genet. Mol. Biol. 2014, 37, 61–63. [Google Scholar] [CrossRef]
- Noronha, R.C.R.; Barros, L.M.R.; Araújo, R.E.F.; Marques, D.F.; Nagamachi, C.Y.; Martins, C.; Pieczarka, J.C. New Insights of Karyoevolution in the Amazonian Turtles Podocnemis expansa and Podocnemis unifilis (Testudines, Podocnemidae). Mol. Cytogenet. 2016, 9, 73. [Google Scholar] [CrossRef]
- Rhodin, A.G.J.; Mittermeier, R.A.; Gardner, A.L.; Medem, F. Karyotypic Analysis of the Podocnemis Turtles. Copeia 1978, 1978, 723–728. [Google Scholar] [CrossRef]
- Alho, C.J.R.; Danni, T.M.S.; Padua, L.F.M. Temperature-Dependent Sex Determination in Podocnemis expansa (Testudinata: Pelomedusidae). Biotropica 1985, 17, 75–78. [Google Scholar] [CrossRef]
- Valenzuela, N. Constant, Shift, and Natural Temperature Effects on Sex Determination in Podocnemis expansa Turtles. Ecology 2001, 82, 3010–3024. [Google Scholar] [CrossRef]
- De Souza, R.R.; Vogt, R.C. Incubation Temperature Influences Sex and Hatchling Size in the Neotropical Turtle Podocnemis unifilis. J. Herpetol. 1994, 28, 453–464. [Google Scholar] [CrossRef]
- Vogt, R.C. Amazon Turtles; Wust Editions, Grafica Biblos: Lima, Peru, 2008. [Google Scholar]
- Páez, V.P.; Correa, J.C.; Cano, A.M.; Bock, B.C. A Comparison of Maternal and Temperature Effects on Sex, Size, and Growth of Hatchlings of the Magdalena River Turtle (Podocnemis lewyana) Incubated under Field and Controlled Laboratory Conditions. Copeia 2009, 2009, 698–704. [Google Scholar] [CrossRef]
- Gómez-Saldarriaga, C.; Valenzuela, N.; Ceballos, C.P. Effects of Incubation Temperature on Sex Determination in the Endangered Magdalena River Turtle, Podocnemis lewyana. Chelonian Conserv. Biol. 2016, 15, 43–53. [Google Scholar] [CrossRef]
- Vogt, R.C. Reproduction of The Cabeçudo, Peltocephalus dumerilianus, in the Biological Reserve of Rio Trombetas, Brazil. Chelonian Conserv. Biol. 1994, 1, 145–148. [Google Scholar]
- De La Ossa, J.; Vogt, R.C.; De La Ossa-Lacayo, A. Sexo Termo Dependencia y Su Relación Con La Idoneidad En Neonatos de Peltocephalus dumerilianus (Testudines, Podocnemididae). Rev. Asoc. Colomb. Cienc. Biol. 2014, 1, 145–151. [Google Scholar]
- Garcia, G. Ecology, Human Impact and Conservation for the Madagascan Side-Necked Turtle (Eretmochelys madagascariensis Grandidier, 1876) at Ankarafantsika National Park, Madagascar. Ph.D. Thesis, The Durrell Institut of Conservation and Ecology, The University of Kent, Canterbury, UK, 2005. [Google Scholar]
- Ewert, M.A.; Etchberger, C.R.; Nelson, C.E. Turtle Sex-Determining Modes and TSD Patterns, and Some TSD Pattern Correlates. In Temperature Dependent Sex Determination in Vertebrates; Valenzuela, N., Lance, V.A., Eds.; Smithsonian Books: Whashington, DC, USA, 2004; pp. 21–32. [Google Scholar]
- Bull, J.J.; Legler, J.M.; Vogt, R.C. Non-Temperature Dependent Sex Determination in Two Suborders of Turtles. Copeia 1985, 1985, 784–786. [Google Scholar] [CrossRef]
- Thompson, M.B. Influence of Incubation Temperature and Water Potential on Sex Determination in Emydura macquarii (Testudines: Pleurodira). Herpetologica 1988, 44, 86–90. [Google Scholar]
- Georges, A. Sex Determination Is Independent of Incubation Temperature in Another Chelid Turtle, Chelodina longicollis. Copeia 1988, 1988, 248–254. [Google Scholar] [CrossRef]
- Georges, A.; McInnes, S. Temperature Fails to Influence Hatchling Sex in Another Genus and Species of Chelid Turtle, Elusor macrurus. J. Herpetol. 1998, 32, 596–598. [Google Scholar] [CrossRef]
- Barros, R.M.; Sampaio, M.M.; Assis, M.F.; Ayres, M.; Cunha, O.R. General Considerations on the Karyotypic Evolution of Chelonia from the Amazon Region of Brazil. Cytologia 1976, 41, 559–565. [Google Scholar] [CrossRef]
- Noleto, R.B.; Kantek, D.L.Z.; Swarça, A.C.; Dias, A.L.; Fenocchio, A.S.; Cestari, M.M. Karyotypic Characterization of Hydromedusa tectifera (Testudines, Pleurodira) from the Upper Iguaçu River in the Brazilian State of Paraná. Genet. Mol. Biol. 2006, 29, 263–266. [Google Scholar] [CrossRef]
- Reed, K.M.; Greenbaum, I.F.; Hanks, B.G.; Bickham, J.W.; Rhodin, A.G.J.; Mittermeier, R.A.; Fedullo, L.P. Cytogenetic Analysis Of The Pleurodine Turtle Phrynops hogei And Its Taxonomic Implications. Amphib.-Reptil. 1991, 12, 203–212. [Google Scholar] [CrossRef]
- Killebrew, F.C. Mitotic Chromosomes of Turtles: II. The Chelidae. Texas J. Sci. 1976, 27, 149–154. [Google Scholar]
- McBee, K.; Bickham, J.W.; Rhodin, A.G.; Mittermeier, R.A. Karyotypic Variation in the Genus Platemys (Testudines: Pleurodira). Copeia 1985, 1985, 445–449. [Google Scholar] [CrossRef]
- Ezaz, T.; Valenzuela, N.; Grützner, F.; Miura, I.; Georges, A.; Burke, R.L.; Graves, J.A.M. An XX/XY Sex Microchromosome System in a Freshwater Turtle, Chelodina longicollis (Testudines: Chelidae) with Genetic Sex Determination. Chromosome Res. Int. J. Mol. Supramol. Evol. Asp. Chromosome Biol. 2006, 14, 139–150. [Google Scholar] [CrossRef] [PubMed]
- Martinez, P.A.; Ezaz, T.; Valenzuela, N.; Georges, A.; Marshall Graves, J.A. An XX/XY Heteromorphic Sex Chromosome System in the Australian Chelid Turtle Emydura macquarii: A New Piece in the Puzzle of Sex Chromosome Evolution in Turtles. Chromosome Res. Int. J. Mol. Supramol. Evol. Asp. Chromosome Biol. 2008, 16, 815–825. [Google Scholar] [CrossRef] [PubMed]
- Lee, L.; Montiel, E.E.; Valenzuela, N. Discovery of Putative XX/XY Male Heterogamety in Emydura subglobosa Turtles Exposes a Novel Trajectory of Sex Chromosome Evolution in Emydura. Cytogenet. Genome Res. 2019, 158, 160–169. [Google Scholar] [CrossRef]
- Mazzoleni, S.; Augstenová, B.; Clemente, L.; Auer, M.; Fritz, U.; Praschag, P.; Protiva, T.; Velenský, P.; Kratochvíl, L.; Rovatsos, M. Sex Is Determined by XX/XY Sex Chromosomes in Australasian Side-Necked Turtles (Testudines: Chelidae). Sci. Rep. 2020, 10, 4276. [Google Scholar] [CrossRef] [PubMed]
- Matsubara, K.; O’Meally, D.; Azad, B.; Georges, A.; Sarre, S.D.; Graves, J.A.M.; Matsuda, Y.; Ezaz, T. Amplification of Microsatellite Repeat Motifs Is Associated with the Evolutionary Differentiation and Heterochromatinization of Sex Chromosomes in Sauropsida. Chromosoma 2016, 125, 111–123. [Google Scholar] [CrossRef] [PubMed]
- Cavalcante, M.G.; Bastos, C.E.M.C.; Nagamachi, C.Y.; Pieczarka, J.C.; Vicari, M.R.; Noronha, R.C.R. Physical Mapping of Repetitive DNA Suggests 2n Reduction in Amazon Turtles Podocnemis (Testudines: Podocnemididae). PLoS ONE 2018, 13, e0197536. [Google Scholar] [CrossRef]
- Clemente, L.; Mazzoleni, S.; Bellavia, E.P.; Augstenová, B.; Auer, M.; Praschag, P.; Protiva, T.; Velenský, P.; Wagner, P.; Fritz, U.; et al. Interstitial Telomeric Repeats Are Rare in Turtles. Genes 2020, 11, 657. [Google Scholar] [CrossRef] [PubMed]
- Viana, P.F.; Feldberg, E.; Cioffi, M.B.; de Carvalho, V.T.; Menezes, S.; Vogt, R.C.; Liehr, T.; Ezaz, T. The Amazonian Red Side-Necked Turtle Rhinemys rufipes (Spix, 1824) (Testudines, Chelidae) Has a GSD Sex-Determining Mechanism with an Ancient XY Sex Microchromosome System. Cells 2020, 9, 2088. [Google Scholar] [CrossRef]
- Lee, L.; Montiel, E.E.; Navarro-Domínguez, B.M.; Valenzuela, N. Chromosomal Rearrangements during Turtle Evolution Altered the Synteny of Genes Involved in Vertebrate Sex Determination. Cytogenet. Genome Res. 2019, 157, 77–88. [Google Scholar] [CrossRef]
- Webb, G.J.; Choquenot, D.; Whitehead, P.J. Nests, Eggs, and Embryonic Development of Carettochelys insculpta (Chelonia: Carettochelidae) from Northern Australia. J. Zool. 1986, 1, 521–550. [Google Scholar] [CrossRef]
- Bickham, J.W.; Bull, J.J.; Legler, J.M. Karyotypes and Evolutionary Relationships of Trionychoid Turtles. Cytologia 1983, 48, 177–183. [Google Scholar] [CrossRef]
- Sato, H.; Ota, H. Karyotype of the Chinese Soft-Shelled Turtle, Pelodiscus sinensis, from Japan and Taiwan, with Chromosomal Data for Dogania subplana. Curr. Herpetol. 2001, 20, 19–25. [Google Scholar] [CrossRef][Green Version]
- Stock, A.D. Karyological Relationships in Turtles (Reptilia: Chelonia). Can. J. Genet. Cytol. 1972, 14, 859–868. [Google Scholar] [CrossRef] [PubMed]
- Vogt, R.C.; Bull, J.J. Genetic Sex Determination in the Spiny Softshell Trionyx spiniferus (Testudines: Trionychidae)? Copeia 1982, 1982, 699–700. [Google Scholar] [CrossRef]
- Choo, B.L.; Chou, L.M. Does Incubation Temperature Influence the Sex of Embryos in Trionyx sinensis? J. Herpetol. 1992, 26, 341–342. [Google Scholar] [CrossRef]
- Janzen, F.J. The Influence of Incubation Temperature and Family on Eggs, Embryos, and Hatchlings of the Smooth Softshell Turtle (Apalone mutica). Physiol. Zool. 1993, 66, 349–373. [Google Scholar] [CrossRef]
- Kawai, A.; Nishida-Umehara, C.; Ishijima, J.; Tsuda, Y.; Ota, H.; Matsuda, Y. Different Origins of Bird and Reptile Sex Chromosomes Inferred from Comparative Mapping of Chicken Z-Linked Genes. Cytogenet. Genome Res. 2007, 117, 92–102. [Google Scholar] [CrossRef] [PubMed]
- Mu, Y.; Zhao, B.; Tang, W.-Q.; Sun, B.-J.; Zeng, Z.-G.; Valenzuela, N.; Du, W.-G. Temperature-Dependent Sex Determination Ruled out in the Chinese Soft-Shelled Turtle (Pelodiscus sinensis) via Molecular Cytogenetics and Incubation Experiments across Populations. Sex. Dev. Genet. Mol. Biol. Evol. Endocrinol. Embryol. Pathol. Sex Determ. Differ. 2015, 9, 111–117. [Google Scholar] [CrossRef] [PubMed]
- Kawagoshi, T.; Uno, Y.; Matsubara, K.; Matsuda, Y.; Nishida, C. The ZW Micro-Sex Chromosomes of the Chinese Soft-Shelled Turtle (Pelodiscus sinensis, Trionychidae, Testudines) Have the Same Origin as Chicken Chromosome 15. Cytogenet. Genome Res. 2009, 125, 125–131. [Google Scholar] [CrossRef] [PubMed]
- Badenhorst, D.; Stanyon, R.; Engstrom, T.; Valenzuela, N. A ZZ/ZW Microchromosome System in the Spiny Softshell Turtle, Apalone spinifera, Reveals an Intriguing Sex Chromosome Conservation in Trionychidae. Chromosome Res. Int. J. Mol. Supramol. Evol. Asp. Chromosome Biol. 2013, 21, 137–147. [Google Scholar] [CrossRef]
- Rovatsos, M.; Praschag, P.; Fritz, U.; Kratochvšl, L. Stable Cretaceous Sex Chromosomes Enable Molecular Sexing in Softshell Turtles (Testudines: Trionychidae). Sci. Rep. 2017, 7, 42150. [Google Scholar] [CrossRef]
- Rovatsos, M.; Kratochvíl, L. Evolution of Dosage Compensation Does Not Depend on Genomic Background. Mol. Ecol. 2021, 30, 1836–1845. [Google Scholar] [CrossRef]
- Bista, B.; Wu, Z.; Literman, R.; Valenzuela, N. Thermosensitive Sex Chromosome Dosage Compensation in ZZ/ZW Softshell Turtles, Apalone spinifera. Philos. Trans. R. Soc. Lond. B. Biol. Sci. 2021, 376, 20200101. [Google Scholar] [CrossRef] [PubMed]
- Mrosovsky, N.; Fretey, J.; Lescure, J.; Pieau, C.; Rimblot, F. Sexual Differentiation as a Function of the Incubation Temperature of Eggs in the Sea-Turtle Dermochelys coriacea (Vandelli, 1761). Amphib.-Reptil. 1985, 6, 83–92. [Google Scholar] [CrossRef]
- Mrosovsky, N.; Dutton, P.H.; Whitmore, C.P. Sex Ratios of Two Species of Sea Turtle Nesting in Suriname. Can. J. Zool. 1984, 62, 2227–2239. [Google Scholar] [CrossRef]
- Mrosovsky, N.; Bass, A.; Corliss, L.A.; Richardson, J.I.; Richardson, T.H. Pivotal and Beach Temperatures for Hawksbill Turtles Nesting in Antigua. Can. J. Zool. 1992, 70, 1920–1925. [Google Scholar] [CrossRef]
- Hewavisenthi, S.; Parmenter, C.J. Hydric Environment and Sex Determination in the Flatback Turtle (Natator depressus Garman) (Chelonia: Cheloniidae). Aust. J. Zool. 2000, 48, 653–659. [Google Scholar] [CrossRef]
- Yntema, C.L.; Mrosovsky, N. Incubation Temperature and Sex Ratio in Hatchling Loggerhead Turtles: A Preliminary Report. Mar. Turt. Newsl. 1979, 11, 9–10. [Google Scholar]
- McCoy, C.J.; Vogt, R.C.; Censky, E.J. Temperature-Controlled Sex Determination in the Sea Turtle Lepidochelys olivacea. J. Herpetol. 1983, 17, 404–406. [Google Scholar] [CrossRef]
- Carrasco, A.; Marquez, M.; Benitez, V.; Diaz, F.; Jimenez, Q. The Effect of Temperature Change on the Sex Ratio of Kemp’s Ridley Nests in the Hatchery Center at Rancho Nuevo, Tamaulipas, Mexico. In Proceedings of the Nineteenth Annual Symposium on Sea Turtle Conservation and Biology, South Padre Island, TX, USA, 2–6 March 1999; Kalb, H., Wibbels, T., Eds.; NOAA Tech Memo, NMFS-SEFSC-443. Southeast Fisheries Science Center (U.S.): Miami, FL, USA, 2000; pp. 128–129. [Google Scholar]
- Yntema, C.L. Effects of Incubation Temperatures on Sexual Differentiation in the Turtle, Chelydra serpentina. J. Morphol. 1976, 150, 453–461. [Google Scholar] [CrossRef] [PubMed]
- Vogt, R.C.; Flores-Villela, O. Effects of Incubation Temperature on Sex Determination in a Community of Neotropical Freshwater Turtles in Southern Mexico. Herpetologica 1992, 48, 265–270. [Google Scholar]
- Janzen, F.J.; Paukstis, G.L. Environmental Sex Determination in Reptiles: Ecology, Evolution, and Experimental Design. Q. Rev. Biol. 1991, 66, 149–179. [Google Scholar] [CrossRef]
- Bull, J.J.; Moon, R.G.; Legler, J.M. Male Heterogamety in Kinosternid Turtles (Genus Staurotypus). Cytogenet. Cell Genet. 1974, 13, 419–425. [Google Scholar] [CrossRef]
- Kawagoshi, T.; Uno, Y.; Nishida, C.; Matsuda, Y. The Staurotypus Turtles and Aves Share the Same Origin of Sex Chromosomes but Evolved Different Types of Heterogametic Sex Determination. PLoS ONE 2014, 9, e105315. [Google Scholar] [CrossRef] [PubMed]
- Janes, D.E.; Organ, C.L.; Stiglec, R.; O’Meally, D.; Sarre, S.D.; Georges, A.; Graves, J.A.M.; Valenzuela, N.; Literman, R.A.; Rutherford, K.; et al. Molecular Evolution of Dmrt1 Accompanies Change of Sex-Determining Mechanisms in Reptilia. Biol. Lett. 2014, 10, 20140809. [Google Scholar] [CrossRef] [PubMed]
- Radhakrishnan, S.; Valenzuela, N. Chromosomal Context Affects the Molecular Evolution of Sex-Linked Genes and Their Autosomal Counterparts in Turtles and Other Vertebrates. J. Hered. 2017, 108, 720–730. [Google Scholar] [CrossRef]
- Sites, J.W.; Bickham, J.W.; Haiduk, M.W.; Derived, X. Chromosome in the Turtle Genus Staurotypus. Science 1979, 206, 1410–1412. [Google Scholar] [CrossRef]
- Kuchling, G.; Goode, E.V.; Praschag, P. Endoscopic imaging of gonads, sex ratio, and temperature-dependent sex determination in juvenile captive-bred radiated tortoises, Astrochelys radiata. Chelonian Conserv. Biol. 2013, 6, 171–177. [Google Scholar]
- Ligon, D.B.; Bidwell, J.R.; Lovern, M.B. Incubation Temperature Effects on Hatchling Growth and Metabolic Rate in the African Spurred Tortoise, Geochelone sulcata. Can. J. Zool. 2009, 87, 64–72. [Google Scholar] [CrossRef]
- Sancho, A.; Gutzke, W.H.; Snell, H.L.; Rea, S.; Wilson, M.; Burke, R.L. Temperature Sex Determination, Incubation Duration, and Hatchling Sexual Dimorphism in the Española Giant Tortoise (Chelonoidis hoodensis) of the Galápagos Islands. Amphib. Reptile Conserv. 2017, 11, 44–50. [Google Scholar]
- Spotila, J.R.; Zimmerman, L.C.; Binckley, C.A.; Grumbles, J.S.; Rostal, D.C.; List, A.; Beyer, E.C.; Phillips, K.M.; Kemp, S.J. Effects of Incubation Conditions on Sex Determination, Hatching Success, and Growth of Hatchling Desert Tortoises, Gopherus agassizii. Herpetol. Monogr. 1994, 8, 103–116. [Google Scholar] [CrossRef]
- Burke, R.; Ewert, M.; McLemore, J.; Jackson, D. Temperature-Dependent Sex Determination and Hatching Success in the Gopher Tortoise (Gopherus polyphemus). Chelonian Conserv. Biol. 1996, 86–88. [Google Scholar]
- Demuth, J.P. The Effects of Constant and Fluctuating Incubation Temperatures on Sex Determination, Growth, and Performance in the Tortoise Gopherus polyphemus. Can. J. Zool. 2001, 79, 1609–1620. [Google Scholar] [CrossRef]
- Emer, S.A. Temperature-Dependent Sex Determination in Manouria emys Emys, The Asian Forest Tortoise. Master’s Thesis, Georgia State University, Atlanta, GA, USA, 5 April 2007. Available online: https://scholarworks.gsu.edu/biology_theses/11 (accessed on 17 November 2021).
- Eendebak, B. Incubation Period and Sex Ratio of Hermann’s Tortoise, Testudo hermanni boettgeri. Chelonian Conserv. Biol. 1995, 1, 227–231. [Google Scholar]
- Kuchling, G.; Goode, E.V.; Praschag, P. Endoscopic Imaging of Gonads, Sex Ratio and Temperature Dependent Sex Determination in Captive Bred Juvenile Burmese Star Tortoises Geochelone platynota. Asian Herpetol. Res. 2011, 2, 240–244. [Google Scholar] [CrossRef]
- Pearson, D.W. Ecological Husbandry and Reproduction of Madagascar Spider (Pyxis arachnoides) and Flat-Tailed (Pyxis planicauda) Tortoises. Chelonian Conserv. Biol. 2013, 6, 146–152. [Google Scholar]
- Zhu, X.P.; Chen, Y.L.; Wei, C.Q.; Liu, Y.H.; Gui, J.F. Temperature Effects on Sex Determination in Yellow Pond Turtle (Mauremys mutica Cantor). Acta Ecol. Sin. 2006, 26, 620–625. [Google Scholar]
- Du, W.G.; Wang, L.; Shen, J.W. Optimal Temperatures for Egg Incubation in Two Geoemydid Turtles: Ocadia sinensis and Mauremys mutica. Aquaculture 2010, 305, 138–142. [Google Scholar] [CrossRef]
- Wu, M.; Zhao, B.; Zhang, W.; Lu, H. Effects of Incubation Temperature on Embryonic Development and Hatchling Traits in the Asian Yellow Pond Turtle, Mauremys mutica. Acta Ecol. Sin. 2014, 34, 5398–5404. [Google Scholar] [CrossRef]
- Okada, Y.; Yabe, T.; Oda, S.-I. Temperature-Dependent Sex Determination in the Japanese Pond Turtle, Mauremys japonica (Reptilia: Geoemydidae). Curr. Herpetol. 2010, 29, 1–10. [Google Scholar] [CrossRef]
- Hou, L. Sex Determination by Temperature for Incubation in Chinemys reevesii. Acta Herpetol. Sin. 1985, 4, 130. [Google Scholar]
- Farrell, L. Update: Temperature Sex Determination (TSD) Project for the Yellow-Margined Box Turtle, Cuora flavomarginata. Turt. Surviv. Alliance Newsl. 2007, 2007, 18. [Google Scholar]
- Ewert, M.A.; Jackson, D.R.; Nelson, C.E. Patterns of Temperature-Dependent Sex Determination in Turtles. J. Exp. Zool. 1994, 270, 3–15. [Google Scholar] [CrossRef]
- Pewphong, R.; Kitana, N.; Kitana, J. Effect of temperature on development: The case of the Malayan snail-eating turtle Malayemys macrocephala. In Naturalists, Explorers and Field Scientists in South-East Asia and Australasia; Das, I., Tuen, A.A., Eds.; Springer: Berlin/Heidelberg, Germany, 2016; pp. 157–169. [Google Scholar]
- WHO De Smet. The Chromosomes of 11 Species of Chelonia (Reptilia). Acta Zool. Pathol. Antverp. 1978, 70, 15–34. [Google Scholar]
- Bickham, J.W. Two-Hundred-Million-Year-Old Chromosomes: Deceleration of the Rate of Karyotypic Evolution in Turtles. Science 1981, 212, 1291–1293. [Google Scholar] [CrossRef] [PubMed]
- Haiduk, M.W.; Bickham, J.W. Chromosomal Homologies and Evolution of Testudinoid Turtles with Emphasis on the Systematic Placement of Platysternon. Copeia 1982, 1982, 60–66. [Google Scholar] [CrossRef]
- Carr, J.L.; Bickham, J.W. Phylogenetic Implications of Karyotypic Variation in the Batagurinae (Testudines: Emydidae). Genetica 1986, 70, 89–106. [Google Scholar] [CrossRef]
- Bickham, J.W.; Carr, J.L. Taxonomy and Phylogeny of the Higher Categories of Cryptodiran Turtles Based on a Cladistic Analysis of Chromosomal Data. Copeia 1983, 1983, 918–932. [Google Scholar] [CrossRef]
- Siripiyasing, P.; Tanomtong, A.; Jumrusthanasan, S.; Patawang, I.; Phimphan, S.; Sanoamuang, L. First Cytogenetic Study of Malayan Snail-Eating Turtle, Malayemys Macrocephala (Testudines, Geoemydidae) in Thailand. Cytologia 2013, 78, 125–132. [Google Scholar] [CrossRef]
- Sharma, G.P.; Kaur, P.; Nakhasi, U. Female Heterogamety in the Indian Cryptodiran Chelonian, Kachuga smithi Gray. In Dr. B.S. Chauhan Commemoration; Tiwari, K.K., Srivistava, C.B., Eds.; Zoological Society of India: Orissa, India, 1975. [Google Scholar]
- Mazzoleni, S.; Augstenová, B.; Clemente, L.; Auer, M.; Fritz, U.; Praschag, P.; Protiva, T.; Velenský, P.; Kratochvíl, L.; Rovatsos, M. Turtles of the Genera Geoemyda and Pangshura (Testudines: Geoemydidae) Lack Differentiated Sex Chromosomes: The End of a 40-Year Error Cascade for Pangshura. PeerJ 2019, 7, e6241. [Google Scholar] [CrossRef]
- Carr, J.L.; Bickham, J.W. Sex Chromosomes of the Asian Black Pond Turtle, Siebenrockiella crassicollis (Testudines: Emydidae). Cytogenet. Cell Genet. 1981, 31, 178–183. [Google Scholar] [CrossRef] [PubMed]
- Kawagoshi, T.; Nishida, C.; Matsuda, Y. The Origin and Differentiation Process of X and Y Chromosomes of the Black Marsh Turtle (Siebenrockiella crassicollis, Geoemydidae, Testudines). Chromosome Res. 2012, 20, 95–110. [Google Scholar] [CrossRef][Green Version]
- Matsuda, Y.; Nishida-Umehara, C.; Tarui, H.; Kuroiwa, A.; Yamada, K.; Isobe, T.; Ando, J.; Fujiwara, A.; Hirao, Y.; Nishimura, O.; et al. Highly Conserved Linkage Homology between Birds and Turtles: Bird and Turtle Chromosomes Are Precise Counterparts of Each Other. Chromosome Res. 2005, 13, 601–615. [Google Scholar] [CrossRef]
- Hammes, A.; Guo, J.K.; Lutsch, G.; Leheste, J.R.; Landrock, D.; Ziegler, U.; Gubler, M.C.; Schedl, A. Two Splice Variants of the Wilms’ Tumor 1 Gene Have Distinct Functions during Sex Determination and Nephron Formation. Cell 2001, 106, 319–329. [Google Scholar] [CrossRef]
- Eozenou, C.; Gonen, N.; Touzon, M.S.; Jorgensen, A.; Yatsenko, S.A.; Fusee, L.; Kamel, A.K.; Gellen, B.; Guercio, G.; Singh, P.; et al. Testis Formation in XX Individuals Resulting from Novel Pathogenic Variants in Wilms’ Tumor 1 (WT1) Gene. Proc. Natl. Acad. Sci. USA 2020, 117, 13680–13688. [Google Scholar] [CrossRef]
- Rhen, T.; Fagerlie, R.; Schroeder, A.; Crossley, D.A.; Lang, J.W. Molecular and Morphological Differentiation of Testes and Ovaries in Relation to the Thermosensitive Period of Gonad Development in the Snapping Turtle, Chelydra serpentina. Differ. Res. Biol. Divers. 2015, 89, 31–41. [Google Scholar] [CrossRef] [PubMed]
- Taylor, E.H. Philippine turtles. Philipp. J. Sci. 1920, 16, 111–144. [Google Scholar] [CrossRef]
- Diesmos, A.C.; Gee, G.V.; Diesmos, M.L.; Brown, R.M.; Widmann, P.J.; Dimalibot, J.C. Rediscovery of the Philippine Forest Turtle, Heosemys leytensis (Chelonia; Bataguridae), from Palawan Island, Philippines. Asiat. Herpetol. Res. 2004, 10, 22–27. [Google Scholar]
- Diesmos, A.C.; Parham, J.F.; Stuart, B.L.; Brown, R.M. The Phylogenetic Position of the Recently Rediscovered Philippine Forest Turtle (Bataguridae: Heosemys leytensis). Proc. Calif. Acad. Sci. 2005, 56, 31. [Google Scholar]
- Bickham, J.W. A Cytosystematic Study of Turtles in the Genera Clemmys, Mauremys and Sacalia. Herpetologica 1975, 31, 198–204. [Google Scholar]
- Montiel, E.E.; Badenhorst, D.; Tamplin, J.; Burke, R.L.; Valenzuela, N. Discovery of the Youngest Sex Chromosomes Reveals First Case of Convergent Co-Option of Ancestral Autosomes in Turtles. Chromosoma 2017, 126, 105–113. [Google Scholar] [CrossRef]
- Badenhorst, D.; Hillier, L.W.; Literman, R.; Montiel, E.E.; Radhakrishnan, S.; Shen, Y.; Minx, P.; Janes, D.E.; Warren, W.C.; Edwards, S.V.; et al. Physical Mapping and Refinement of the Painted Turtle Genome (Chrysemys picta) Inform Amniote Genome Evolution and Challenge Turtle-Bird Chromosomal Conservation. Genome Biol. Evol. 2015, 7, 2038–2050. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Literman, R.; Radhakrishnan, S.; Tamplin, J.; Burke, R.; Dresser, C.; Valenzuela, N. Development of Sexing Primers in Glyptemys insculpta and Apalone spinifera Turtles Uncovers an XX/XY Sex-Determining System in the Critically-Endangered Bog Turtle Glyptemys muhlenbergii. Conserv. Genet. Resour. 2017, 9, 651–658. [Google Scholar] [CrossRef]
- Gorman, G. The Chromosomes of the Reptilian, a Cytotaxonomy Interpretation. In Cytotaxonomy and Vertebrate Evolution; Chiarelli, A.B., Capanna, E., Eds.; Academic Press: Cambridge, MA, USA, 1973. [Google Scholar]
- Cao, D.; Wang, M.; Ge, Y.; Gong, S. Draft Genome of the Big-Headed Turtle Platysternon megacephalum. Sci. Data 2019, 6, 60. [Google Scholar] [CrossRef]
- Rohilla, M.S.; Rao, R.J.; Tiwari, P.K. Use of Peripheral Blood Lymphocyte Culture in the Karyological Analysis of Indian Freshwater Turtles, Lissemys punctata and Geoclemys hamiltoni. Curr. Sci. 2006, 90, 1130–1134. [Google Scholar]
- Literman, R.; Burrett, A.; Bista, B.; Valenzuela, N. Putative Independent Evolutionary Reversals from Genotypic to Temperature-Dependent Sex Determination Are Associated with Accelerated Evolution of Sex-Determining Genes in Turtles. J. Mol. Evol. 2018, 86, 11–26. [Google Scholar] [CrossRef]
- Bista, B.; Valenzuela, N. Turtle Insights into the Evolution of the Reptilian Karyotype and the Genomic Architecture of Sex Determination. Genes 2020, 11, 416. [Google Scholar] [CrossRef] [PubMed]
- Deveson, I.W.; Holleley, C.E.; Blackburn, J.; Marshall Graves, J.A.; Mattick, J.S.; Waters, P.D.; Georges, A. Differential Intron Retention in Jumonji Chromatin Modifier Genes Is Implicated in Reptile Temperature-Dependent Sex Determination. Sci. Adv. 2017, 3, e1700731. [Google Scholar] [CrossRef]
- Ge, C.; Ye, J.; Weber, C.; Sun, W.; Zhang, H.; Zhou, Y.; Cai, C.; Qian, G.; Capel, B. The Histone Demethylase KDM6B Regulates Temperature-Dependent Sex Determination in a Turtle Species. Science 2018, 360, 645–648. [Google Scholar] [CrossRef]
- Weber, C.; Zhou, Y.; Lee, J.G.; Looger, L.L.; Qian, G.; Ge, C.; Capel, B. Temperature-Dependent Sex Determination Is Mediated by PSTAT3 Repression of Kdm6b. Science 2020, 368, 303–306. [Google Scholar] [CrossRef] [PubMed]
- Wilson Sayres, M.A.; Makova, K.D. Genome Analyses Substantiate Male Mutation Bias in Many Species. BioEssays 2011, 33, 938–945. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, N.J.; Nelson, N.J.; Cree, A.; Pledger, S.; Keall, S.N.; Daugherty, C.H. Support for a Rare Pattern of Temperature-Dependent Sex Determination in Archaic Reptiles: Evidence from Two Species of Tuatara (Sphenodon). Front. Zool. 2006, 3, 9. [Google Scholar] [CrossRef] [PubMed]
- Vidal, N.; Hedges, S.B. The molecular evolutionary tree of lizards, snakes, and amphisbaenians. Comptes Rendus Biol. 2009, 332, 129–139. [Google Scholar] [CrossRef]
- Burbrink, F.T.; Grazziotin, F.G.; Pyron, R.A.; Cundall, D.; Donnellan, S.; Irish, F.; Keogh, J.S.; Kraus, F.; Murphy, R.W.; Noonan, B.; et al. Interrogating Genomic-Scale Data for Squamata (Lizards, Snakes, and Amphisbaenians) Shows No Support for Key Traditional Morphological Relationships. Syst. Biol. 2020, 69, 502–520. [Google Scholar] [CrossRef]
- Cole, C.J.; Gans, C. The Karyotype of Dibamus novaeguineae (Squamata: Dibamidae). Herpetologica 1997, 229–232. [Google Scholar]
- Wagner, E. Temperature-Dependent Sex Determination in a Gekko Lizard. Q. Rev. Biol. 1980, 55, 21. [Google Scholar]
- King, M.; Rofe, R. Karyotypic Variation in the Australian Gekko Phyllodactylus marmoratus (Gray) (Gekkonidae: Reptilia). Chromosoma 1976, 54, 75–87. [Google Scholar] [CrossRef] [PubMed]
- Gorman, G.C.; Gress, F. Sex Chromosomes of a Pygopodid Lizard, Lialis burtonis. Experientia 1970, 26, 206–207. [Google Scholar] [CrossRef]
- Solleder, E.; Schmid, M. XX/XY-Sex Chromosomes in Gekko Gecko (Sauria, Reptilia). Amphib.-Reptil. 1984, 5, 339–345. [Google Scholar] [CrossRef]
- Kawai, A.; Ishijima, J.; Nishida, C.; Kosaka, A.; Ota, H.; Kohno, S.; Matsuda, Y. The ZW Sex Chromosomes of Gekko hokouensis (Gekkonidae, Squamata) Represent Highly Conserved Homology with Those of Avian Species. Chromosoma 2009, 118, 43–51. [Google Scholar] [CrossRef]
- Gamble, T. A Review of Sex Determining Mechanisms in Geckos (Gekkota: Squamata). Sex. Dev. 2010, 4, 88–103. [Google Scholar] [CrossRef] [PubMed]
- Pokorná, M.; Rens, W.; Rovatsos, M.; Kratochvíl, L. A ZZ/ZW Sex Chromosome System in the Thick-Tailed Gecko (Underwoodisaurus milii; Squamata: Gekkota: Carphodactylidae), a Member of the Ancient Gecko Lineage. Cytogenet. Genome Res. 2014, 142, 190–196. [Google Scholar] [CrossRef]
- Koubová, M.; Johnson Pokorná, M.; Rovatsos, M.; Farkačová, K.; Altmanová, M.; Kratochvíl, L. Sex Determination in Madagascar Geckos of the Genus Paroedura (Squamata: Gekkonidae): Are Differentiated Sex Chromosomes Indeed so Evolutionary Stable? Chromosome Res. 2014, 22, 441–452. [Google Scholar] [CrossRef]
- Rovatsos, M.; Johnson Pokorná, M.; Altmanová, M.; Kratochvíl, L. Mixed-Up Sex Chromosomes: Identification of Sex Chromosomes in the X1X1X2X2/X1X2Y System of the Legless Lizards of the Genus Lialis (Squamata: Gekkota: Pygopodidae). Cytogenet. Genome Res. 2016, 149, 282–289. [Google Scholar] [CrossRef] [PubMed]
- Gamble, T.; Coryell, J.; Ezaz, T.; Lynch, J.; Scantlebury, D.P.; Zarkower, D. Restriction Site-Associated DNA Sequencing (RAD-Seq) Reveals an Extraordinary Number of Transitions among Gecko Sex-Determining Systems. Mol. Biol. Evol. 2015, 32, 1296–1309. [Google Scholar] [CrossRef]
- Gamble, T.; McKenna, E.; Meyer, W.; Nielsen, S.V.; Pinto, B.J.; Scantlebury, D.P.; Higham, T.E. XX/XY Sex Chromosomes in the South American Dwarf Gecko (Gonatodes humeralis). J. Hered. 2017, 109, 462–468. [Google Scholar] [CrossRef]
- Nielsen, S.V.; Daza, J.D.; Pinto, B.J.; Gamble, T. ZZ/ZW Sex Chromosomes in the Endemic Puerto Rican Leaf-Toed Gecko (Phyllodactylus wirshingi). Cytogenet. Genome Res. 2019, 157, 89–97. [Google Scholar] [CrossRef]
- Keating, S.E.; Griffing, A.H.; Nielsen, S.V.; Scantlebury, D.P.; Gamble, T. Conserved ZZ/ZW Sex Chromosomes in Caribbean Croaking Geckos (Aristelliger: Sphaerodactylidae). J. Evol. Biol. 2020, 33, 1316–1326. [Google Scholar] [CrossRef]
- Jeffries, D.L.; Lavanchy, G.; Sermier, R.; Sredl, M.J.; Miura, I.; Borzée, A.; Barrow, L.N.; Canestrelli, D.; Crochet, P.-A.; Dufresnes, C.; et al. A Rapid Rate of Sex-Chromosome Turnover and Non-Random Transitions in True Frogs. Nat. Commun. 2018, 9, 4088. [Google Scholar] [CrossRef]
- Gammerdinger, W.J.; Kocher, T.D. Unusual Diversity of Sex Chromosomes in African Cichlid Fishes. Genes 2018, 9, 480. [Google Scholar] [CrossRef] [PubMed]
- Matsubara, K.; Gamble, T.; Matsuda, Y.; Zarkower, D.; Sarre, S.D.; Georges, A.; Marshall Graves, J.A.; Ezaz, T. Non-homologous sex chromosomes in two geckos (Gekkonidae: Gekkota) with female heterogamy. Cytogenet. Genome Res. 2014, 143, 251–258. [Google Scholar] [CrossRef] [PubMed]
- Srikulnath, K.; Uno, Y.; Nishida, C.; Ota, H.; Matsuda, Y. Karyotype Reorganization in the Hokou Gecko (Gekko Hokouensis, Gekkonidae): The Process of Microchromosome Disappearance in Gekkota. PLoS ONE 2015, 10, e0134829. [Google Scholar] [CrossRef] [PubMed]
- Rovatsos, M.; Farkačová, K.; Altmanová, M.; Johnson Pokorná, M.; Kratochvíl, L. The Rise and Fall of Differentiated Sex Chromosomes in Geckos. Mol. Ecol. 2019, 28, 3042–3052. [Google Scholar] [CrossRef] [PubMed]
- Pyron, R.A.; Burbrink, F.T.; Wiens, J.J. A Phylogeny and Revised Classification of Squamata, Including 4161 Species of Lizards and Snakes. BMC Evol. Biol. 2013, 13, 93. [Google Scholar] [CrossRef]
- Keating, S.E.; Blumer, M.; Grismer, L.L.; Lin, A.; Nielsen, S.V.; Thura, M.K.; Wood, P.L.; Quah, E.S.H.; Gamble, T. Sex Chromosome Turnover in Bent-Toed Geckos (Cyrtodactylus). Genes 2021, 12, 116. [Google Scholar] [CrossRef] [PubMed]
- Ota, H.; Hikida, T.; Matsui, M.; Mori, A. Karyotypes of Two Species of the Genus Cyrtodactylus (Squamata: Gekkonidae) from Sarawak, Malaysia. Caryologia 1992, 45, 43–49. [Google Scholar] [CrossRef][Green Version]
- Pensabene, E.; Kratochvíl, L.; Rovatsos, M. Independent Evolution of Sex Chromosomes in Eublepharid Geckos, A Lineage with Environmental and Genotypic Sex Determination. Life 2020, 10, 342. [Google Scholar] [CrossRef] [PubMed]
- Kluge, A.G. Phylogenetic Relationships and Evolutionary Trends in the Eublepharine Lizard Genus Coleonyx. Copeia 1975, 1975, 24–35. [Google Scholar] [CrossRef]
- Dial, B.E.; Grismer, L.L. A Phylogenetic Analysis of Physiological-Ecological Character Evolution in the Lizard Genus Coleonyx and Its Implications for Historical Biogeographic Reconstruction. Syst. Biol. 1992, 41, 178–195. [Google Scholar] [CrossRef]
- Viets, B.E.; Ewert, M.A.; Talent, L.G.; Nelson, C.E. Sex-Determining Mechanisms in Squamate Reptiles. J. Exp. Zool. 1994, 270, 45–56. [Google Scholar] [CrossRef]
- Pokorná, M.; Rábová, M.; Ráb, P.; Ferguson-Smith, M.A.; Rens, W.; Kratochvíl, L. Differentiation of Sex Chromosomes and Karyotypic Evolution in the Eye-Lid Geckos (Squamata: Gekkota: Eublepharidae), a Group with Different Modes of Sex Determination. Chromosome Res. 2010, 18, 809–820. [Google Scholar] [CrossRef]
- Augstenová, B.; Pensabene, E.; Veselý, M.; Kratochvíl, L.; Rovatsos, M. Are Geckos Special in Sex Determination? Independently Evolved Differentiated ZZ/ZW Sex Chromosomes in Carphodactylid Geckos. Genome Biol. Evol. 2021, 13, evab119. [Google Scholar] [CrossRef] [PubMed]
- Rovatsos, M.; Gamble, T.; Nielsen, S.V.; Georges, A.; Ezaz, T.; Kratochvíl, L. Do Male and Female Heterogamety Really Differ in Expression Regulation? Lack of Global Dosage Balance in Pygopodid Geckos. Philos. Trans. R. Soc. Lond. B. Biol. Sci. 2021, 376, 20200102. [Google Scholar] [CrossRef] [PubMed]
- Matsubara, K.; Knopp, T.; Sarre, S.D.; Georges, A.; Ezaz, T. Karyotypic Analysis and FISH Mapping of Microsatellite Motifs Reveal Highly Differentiated XX/XY Sex Chromosomes in the Pink-Tailed Worm-Lizard (Aprasia parapulchella, Pygopodidae, Squamata). Mol. Cytogenet. 2013, 6, 60. [Google Scholar] [CrossRef] [PubMed]
- Kostmann, A.; Kratochvíl, L.; Rovatsos, M. First Report of Sex Chromosomes in Plated Lizards (Squamata: Gerrhosauridae). Sex. Dev. 2020, 14, 60–65. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, S.V.; Pinto, B.J.; Guzmán-Méndez, I.A.; Gamble, T. First Report of Sex Chromosomes in Night Lizards (Scincoidea: Xantusiidae). J. Hered. 2020, 111, 307–317. [Google Scholar] [CrossRef]
- Kratochvíl, L.; Vukić, J.; Červenka, J.; Kubička, L.; Pokorná, M.J.; Kukačková, D.; Rovatsos, M.; Piálek, L. Mixed-Sex Offspring Produced via Cryptic Parthenogenesis in a Lizard. Mol. Ecol. 2020, 29, 4118–4127. [Google Scholar] [CrossRef]
- Katoh-Fukui, Y.; Tsuchiya, R.; Shiroishi, T.; Nakahara, Y.; Hashimoto, N.; Noguchi, K.; Higashinakagawa, T. Male-to-Female Sex Reversal in M33 Mutant Mice. Nature 1998, 393, 688–692. [Google Scholar] [CrossRef]
- Biason-Lauber, A.; Konrad, D.; Meyer, M.; DeBeaufort, C.; Schoenle, E.J. Ovaries and Female Phenotype in a Girl with 46,XY Karyotype and Mutations in the CBX2 Gene. Am. J. Hum. Genet. 2009, 84, 658–663. [Google Scholar] [CrossRef]
- Kuroiwa, A.; Handa, S.; Nishiyama, C.; Chiba, E.; Yamada, F.; Abe, S.; Matsuda, Y. Additional Copies of CBX2 in the Genomes of Males of Mammals Lacking SRY, the Amami Spiny Rat (Tokudaia osimensis) and the Tokunoshima Spiny Rat (Tokudaia tokunoshimensis). Chromosome Res. 2011, 19, 635–644. [Google Scholar] [CrossRef] [PubMed]
- Robert, K.A.; Thompson, M.B. Sex Determination. Viviparous Lizard Selects Sex of Embryos. Nature 2001, 412, 698–699. [Google Scholar] [CrossRef]
- Wapstra, E.; Olsson, M.; Shine, R.; Edwards, A.; Swain, R.; Joss, J.M.P. Maternal Basking Behaviour Determines Offspring Sex in a Viviparous Reptile. Proc. Biol. Sci. 2004, 271 (Suppl. S4), S230–S232. [Google Scholar] [CrossRef]
- Shine, R.; Elphick, M.J.; Donnellan, S. Co-Occurrence of Multiple, Supposedly Incompatible Modes of Sex Determination in a Lizard Population. Ecol. Lett. 2002, 5, 486–489. [Google Scholar] [CrossRef]
- Pen, I.; Uller, T.; Feldmeyer, B.; Harts, A.; While, G.M.; Wapstra, E. Climate-Driven Population Divergence in Sex-Determining Systems. Nature 2010, 468, 436–438. [Google Scholar] [CrossRef]
- Cunningham, G.D.; While, G.M.; Wapstra, E. Climate and Sex Ratio Variation in a Viviparous Lizard. Biol. Lett. 2017, 13, 20170218. [Google Scholar] [CrossRef] [PubMed]
- Radder, R.S.; Quinn, A.E.; Georges, A.; Sarre, S.D.; Shine, R. Genetic Evidence for Co-Occurrence of Chromosomal and Thermal Sex-Determining Systems in a Lizard. Biol. Lett. 2008, 4, 176–178. [Google Scholar] [CrossRef] [PubMed]
- Quinn, A.E.; Radder, R.S.; Sarre, S.D.; Georges, A.; Ezaz, T.; Shine, R. Isolation and Development of a Molecular Sex Marker for Bassiana duperreyi, a Lizard with XX/XY Sex Chromosomes and Temperature-Induced Sex Reversal. Mol. Genet. Genom. MGG 2009, 281, 665–672. [Google Scholar] [CrossRef]
- Hill, P.L.; Burridge, C.P.; Ezaz, T.; Wapstra, E. Conservation of Sex-Linked Markers among Conspecific Populations of a Viviparous Skink, Niveoscincus ocellatus, Exhibiting Genetic and Temperature-Dependent Sex Determination. Genome Biol. Evol. 2018, 10, 1079–1087. [Google Scholar] [CrossRef]
- Cornejo-Páramo, P.; Dissanayake, D.S.B.; Lira-Noriega, A.; Martínez-Pacheco, M.L.; Acosta, A.; Ramírez-Suástegui, C.; Méndez-de-la-Cruz, F.R.; Székely, T.; Urrutia, A.O.; Georges, A.; et al. Viviparous Reptile Regarded to Have Temperature-Dependent Sex Determination Has Old XY Chromosomes. Genome Biol. Evol. 2020, 12, 924–930. [Google Scholar] [CrossRef] [PubMed]
- Kostmann, A.; Kratochvíl, L.; Rovatsos, M. Poorly Differentiated XX/XY Sex Chromosomes Are Widely Shared across Skink Radiation. Proc. Biol. Sci. 2021, 288, 20202139. [Google Scholar] [CrossRef]
- Dissanayake, D.S.B.; Holleley, C.E.; Hill, L.K.; O’Meally, D.; Deakin, J.E.; Georges, A. Identification of Y Chromosome Markers in the Eastern Three-Lined Skink (Bassiana duperreyi) Using in Silico Whole Genome Subtraction. BMC Genom. 2020, 21, 667. [Google Scholar] [CrossRef]
- Hughes, J.J.; Alkhunaizi, E.; Kruszka, P.; Pyle, L.C.; Grange, D.K.; Berger, S.I.; Payne, K.K.; Masser-Frye, D.; Hu, T.; Christie, M.R.; et al. Loss-of-Function Variants in PPP1R12A: From Isolated Sex Reversal to Holoprosencephaly Spectrum and Urogenital Malformations. Am. J. Hum. Genet. 2020, 106, 121–128. [Google Scholar] [CrossRef]
- Varmuza, S.; Jurisicova, A.; Okano, K.; Hudson, J.; Boekelheide, K.; Shipp, E.B. Spermiogenesis Is Impaired in Mice Bearing a Targeted Mutation in the Protein Phosphatase 1cgamma Gene. Dev. Biol. 1999, 205, 98–110. [Google Scholar] [CrossRef]
- Patawang, I.; Chuaykern, Y.; Supanuam, P.; Maneechot, N.; Pinthong, K.; Tanomtong, A. Cytogenetics of the skinks (Reptilia, Scincidae) from Thailand; IV: Newly investigated karyotypic features of Lygosoma quadrupes and Scincella melanostica. Caryologia 2018, 71, 29–34. [Google Scholar] [CrossRef]
- Wright, J.W. Evolution of the X1X2Y Sex Chromosome Mechanism in the Scincid Lizard Scincella laterale (Say). Chromosoma 1973, 43, 101–108. [Google Scholar] [PubMed]
- Castiglia, R.; Bezerra, A.; Flores-Villela, O.; Annesi, F.; Muñoz, A.; Gornung, E. Comparative Cytogenetics of Two Species of Ground Skinks: Scincella assata and S. cherriei (Squamata: Scincidae: Lygosominae) from Chiapas, Mexico. Acta Herpetol. 2013, 8, 69–73. [Google Scholar] [CrossRef]
- Bull, J. Sex Chromosome Differentiation: An Intermediate Stage in a Lizard. Can. J. Genet. Cytol. 1978, 20, 205–209. [Google Scholar] [CrossRef]
- Peccinini-Seale, D.; Rocha, C.F.D.; Almeida, T.M.B.; Araújo, A.F.B.; De Sena, M.A. Cytogenetics of the Brazilian Whiptail Lizard Cnemidophorus littoralis (Teiidae) from a Restinga Area (Barra de Maricá) in Southeastern Brazil. Braz. J. Biol. 2004, 64, 661–667. [Google Scholar] [CrossRef] [PubMed]
- Cole, C.J.; Dessauer, H.C.; Townsend, C.R.; Arnold, M.G. Unisexual Lizards of the Genus Gymnophthalmus (Reptilia, Teiidae) in the Neotropics: Genetics, Origin, and Systematics. Am. Mus. Novit. 1990, 2994, 1–29. [Google Scholar]
- Pellegrino, K.C.; Rodrigues, M.T.; Yonenaga-Yassuda, Y. Chromosomal Polymorphisms Due to Supernumerary Chromosomes and Pericentric Inversions in the Eyelidless Microteiid Lizard Nothobachia ablephara (Squamata, Gymnophthalmidae). Chromosome Res. 1999, 7, 247–254. [Google Scholar] [CrossRef]
- Yonenaga-Yassudal, Y.; Rodrigues, M.T. Supernumerary Chromosome Variation, Heteromorphic Sex Chromosomes and Banding Patterns in Microteiid Lizards of the Genus Micrablepharus (Squamata, Gymnophthalmidae). Chromosome Res. 1999, 7, 21–29. [Google Scholar] [CrossRef] [PubMed]
- Yonenaga-Yassuda, Y.; Rodrigues, M.T.; Pellegrino, K.C.M. Chromosomal Banding Patterns in the Eyelid-Less Microteiid Lizard Radiation: The X1X1X2X2: X1X2Y Sex Chromosome System in Calyptommatus and the Karyotypes of Psilophthalmus and Tretioscincus (Squamata, Gymnophthalmidae). Genet. Mol. Biol. 2005, 28, 700–709. [Google Scholar] [CrossRef]
- Cole, C.J.; Gans, C. Chromosomes of Bipes, Mesobaena, and Other Amphisbaenians (Reptilia), with Comments on Their Evolution. Am. Mus. Novit. 1987, 2689, 1–9. [Google Scholar]
- Rovatsos, M.; Vukić, J.; Mrugała, A.; Suwala, G.; Lymberakis, P.; Kratochvíl, L. Little Evidence for Switches to Environmental Sex Determination and Turnover of Sex Chromosomes in Lacertid Lizards. Sci. Rep. 2019, 9, 7832. [Google Scholar] [CrossRef] [PubMed]
- Rovatsos, M.; Vukić, J.; Kratochvíl, L. Mammalian X Homolog Acts as Sex Chromosome in Lacertid Lizards. Heredity 2016, 117, 8–13. [Google Scholar] [CrossRef] [PubMed]
- Achermann, J.C.; Ito, M.; Hindmarsh, P.C.; Jameson, J.L. A Mutation in the Gene Encoding Steroidogenic Factor-1 Causes XY Sex Reversal and Adrenal Failure in Humans. Nat. Genet. 1999, 22, 125–126. [Google Scholar] [CrossRef]
- Ikeda, Y.; Tagami, A.; Maekawa, M.; Nagai, A. The Conditional Deletion of Steroidogenic Factor 1 (Nr5a1) in Sox9-Cre Mice Compromises Testis Differentiation. Sci. Rep. 2021, 11, 4486. [Google Scholar] [CrossRef]
- Oike, A.; Kodama, M.; Yasumasu, S.; Yamamoto, T.; Nakamura, Y.; Ito, E.; Nakamura, M. Participation of Androgen and Its Receptor in Sex Determination of an Amphibian Species. PLoS ONE 2017, 12, e0178067. [Google Scholar] [CrossRef]
- Parma, P.; Veyrunes, F.; Pailhoux, E. Sex Reversal in Non-Human Placental Mammals. Sex. Dev. 2016, 10, 326–344. [Google Scholar] [CrossRef]
- Danielyan, F.; Arakelyan, M.; Stepanyan, I. Hybrids of Darevskia Valentini, D. Armeniaca and D. Unisexualis from a Sympatric Population in Armenia. Amphib.-Reptil. 2008, 29, 487–504. [Google Scholar] [CrossRef]
- King, M.; King, D. Chromosomal Evolution in the Lizard Genus Varanus (Reptilia). Aust. J. Biol. Sci. 1975, 28, 89–108. [Google Scholar] [CrossRef] [PubMed]
- King, M.; Mengden, G.A.; King, D. A Pericentric-Inversion Polymorphism and s ZZ/ZW Sex-Chromosome System in Varanus acanthurus Boulenger Analyzed by G-and C-Banding and Ag Staining. Genetica 1982, 58, 39–45. [Google Scholar] [CrossRef]
- Matsubara, K.; Sarre, S.D.; Georges, A.; Matsuda, Y.; Marshall Graves, J.A.; Ezaz, T. Highly Differentiated ZW Sex Microchromosomes in the Australian Varanus Species Evolved through Rapid Amplification of Repetitive Sequences. PLoS ONE 2014, 9, e95226. [Google Scholar] [CrossRef]
- Patawang, I.; Tanomtong, A.; Getlekha, N.; Phimphan, S.; Pinthong, K.; Neeratanaphan, L. Standardized Karyotype and Idiogram of Bengal Monitor Lizard, Varanus bengalensis (Squamata, Varanidae). Cytologia 2017, 82, 75–82. [Google Scholar] [CrossRef][Green Version]
- Johnson Pokorná, M.; Altmanová, M.; Rovatsos, M.; Velenský, P.; Vodička, R.; Rehák, I.; Kratochvíl, L. First Description of the Karyotype and Sex Chromosomes in the Komodo Dragon (Varanus Komodoensis). Cytogenet. Genome Res. 2016, 148, 284–291. [Google Scholar] [CrossRef]
- Iannucci, A.; Altmanová, M.; Ciofi, C.; Ferguson-Smith, M.; Milan, M.; Pereira, J.C.; Pether, J.; Rehák, I.; Rovatsos, M.; Stanyon, R.; et al. Conserved Sex Chromosomes and Karyotype Evolution in Monitor Lizards (Varanidae). Heredity 2019, 123, 215–227. [Google Scholar] [CrossRef] [PubMed]
- Johnson Pokorná, M.; Rovatsos, M.; Kratochvíl, L. Sex Chromosomes and Karyotype of the (Nearly) Mythical Creature, the Gila Monster, Heloderma suspectum (Squamata: Helodermatidae). PLoS ONE 2014, 9, e104716. [Google Scholar] [CrossRef]
- Rovatsos, M.; Rehák, I.; Velenský, P.; Kratochvíl, L. Shared Ancient Sex Chromosomes in Varanids, Beaded Lizards, and Alligator Lizards. Mol. Biol. Evol. 2019, 36, 1113–1120. [Google Scholar] [CrossRef]
- Augstenová, B.; Pensabene, E.; Kratochvíl, L.; Rovatsos, M. Cytogenetic Evidence for Sex Chromosomes and Karyotype Evolution in Anguimorphan Lizards. Cells 2021, 10, 1612. [Google Scholar] [CrossRef] [PubMed]
- Hattori, R.S.; Somoza, G.M.; Fernandino, J.I.; Colautti, D.C.; Miyoshi, K.; Gong, Z.; Yamamoto, Y.; Strüssmann, C.A. The Duplicated Y-Specific amhy Gene Is Conserved and Linked to Maleness in Silversides of the Genus Odontesthes. Genes 2019, 10, 679. [Google Scholar] [CrossRef]
- Bej, D.K.; Miyoshi, K.; Hattori, R.S.; Strüssmann, C.A.; Yamamoto, Y. A Duplicated, Truncated amh Gene Is Involved in Male Sex Determination in an Old World Silverside. G3 Genes Genomes Genet. 2017, 7, 2489–2495. [Google Scholar] [CrossRef]
- Curzon, A.Y.; Shirak, A.; Dor, L.; Zak, T.; Perelberg, A.; Seroussi, E.; Ron, M. A Duplication of the Anti-Müllerian Hormone Gene Is Associated with Genetic Sex Determination of Different Oreochromis niloticus Strains. Heredity 2020, 125, 317–327. [Google Scholar] [CrossRef]
- Harlow, P. Temperature-Dependent Sex Determination in Lizards. In Temperature Dependent Sex Determination in Vertebrates; Valenzuela, N., Lance, V.A., Eds.; Smithsonian Books: Whashington, DC, USA, 2004; pp. 42–52. [Google Scholar]
- Sidhom, M.; Said, K.; Chatti, N.; Guarino, F.M.; Odierna, G.; Petraccioli, A.; Picariello, O.; Mezzasalma, M. Karyological Characterization of the Common Chameleon (Chamaeleo chamaeleon) Provides Insights on the Evolution and Diversification of Sex Chromosomes in Chamaeleonidae. Zool. Jena Ger. 2020, 141, 125738. [Google Scholar] [CrossRef]
- Andrews, R.M. Incubation Temperature and Sex Ratio of the Veiled Chameleon (Chamaeleo calyptratus). J. Herpetol. 2005, 39, 515–518. [Google Scholar] [CrossRef]
- Rovatsos, M.; Johnson Pokorná, M.; Altmanová, M.; Kratochvíl, L. Female Heterogamety in Madagascar Chameleons (Squamata: Chamaeleonidae: Furcifer): Differentiation of Sex and Neo-Sex Chromosomes. Sci. Rep. 2015, 5, 13196. [Google Scholar] [CrossRef]
- Rovatsos, M.; Altmanová, M.; Augstenová, B.; Mazzoleni, S.; Velenský, P.; Kratochvíl, L. ZZ/ZW Sex Determination with Multiple Neo-Sex Chromosomes Is Common in Madagascan Chameleons of the Genus Furcifer (Reptilia: Chamaeleonidae). Genes 2019, 10, 1020. [Google Scholar] [CrossRef] [PubMed]
- Rovatsos, M.; Altmanová, M.; Johnson Pokorná, M.; Velenský, P.; Sánchez Baca, A.; Kratochvíl, L. Evolution of Karyotypes in Chameleons. Genes 2017, 8, 382. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, S.V.; Banks, J.L.; Diaz, R.E.; Trainor, P.A.; Gamble, T. Dynamic Sex Chromosomes in Old World Chameleons (Squamata: Chamaeleonidae). J. Evol. Biol. 2018, 31, 484–490. [Google Scholar] [CrossRef] [PubMed]
- Pokorná, M.; Giovannotti, M.; Kratochvíl, L.; Kasai, F.; Trifonov, V.A.; O’Brien, P.C.M.; Caputo, V.; Olmo, E.; Ferguson-Smith, M.A.; Rens, W. Strong Conservation of the Bird Z Chromosome in Reptilian Genomes Is Revealed by Comparative Painting despite 275 Million Years Divergence. Chromosoma 2011, 120, 455–468. [Google Scholar] [CrossRef] [PubMed]
- Kratochvíl, L.; Gamble, T.; Rovatsos, M. Sex Chromosome Evolution among Amniotes: Is the Origin of Sex Chromosomes Non-Random? Philos. Trans. R. Soc. Lond. B. Biol. Sci. 2021, 376, 20200108. [Google Scholar] [CrossRef]
- Ganesh, S.; Raman, R. Sex Reversal by Testosterone and Not by Estradiol or Temperature in Calotes versicolor, the Lizard Lacking Sex Chromosomes. J. Exp. Zool. 1995, 271, 139–144. [Google Scholar] [CrossRef]
- Doddamani, L.S.I.; Vani, V.; Seshagiri, P.B. A Tropical Oviparous Lizard, Calotes versicolor, Exhibiting a Potentially Novel FMFM Pattern of Temperature-Dependent Sex Determination. J. Exp. Zool. Part Ecol. Genet. Physiol. 2012, 317, 32–46. [Google Scholar] [CrossRef]
- Wilson, C.A.; Priyanka; Titus, T.; Batzel, P.; Postlethwait, J.H.; Raman, R. A Search for Sex-Linked Loci in the Agamid Lizard, Calotes versicolor. Sex. Dev. 2019, 13, 143–150. [Google Scholar] [CrossRef] [PubMed]
- Alam, S.M.I.; Altmanová, M.; Prasongmaneerut, T.; Georges, A.; Sarre, S.D.; Nielsen, S.V.; Gamble, T.; Srikulnath, K.; Rovatsos, M.; Kratochvíl, L.; et al. Cross-Species BAC Mapping Highlights Conservation of Chromosome Synteny across Dragon Lizards (Squamata: Agamidae). Genes 2020, 11, 698. [Google Scholar] [CrossRef] [PubMed]
- Zeng, X.M.; Wang, Y.Z.; Liu, Z.J.; Fang, Z.L.; Wu, G.F.; Papenfuss, T.J.; Macey, R.J. Karyotypes of Nine Species in the Genus Phrynocephalus, with Discussion of Karyotypic Evolution of Chinese Phrynocephalus. Acta Zool. Sin. 1997, 43, 399–410. [Google Scholar]
- Yang, W.; Qi, Y.; Fu, J. Exploring the Genetic Basis of Adaptation to High Elevations in Reptiles: A Comparative Transcriptome Analysis of Two Toad-Headed Agamas (Genus Phrynocephalus). PLoS ONE 2014, 9, e112218. [Google Scholar] [CrossRef] [PubMed]
- Gao, W.; Sun, Y.-B.; Zhou, W.-W.; Xiong, Z.-J.; Chen, L.; Li, H.; Fu, T.-T.; Xu, K.; Xu, W.; Ma, L.; et al. Genomic and Transcriptomic Investigations of the Evolutionary Transition from Oviparity to Viviparity. Proc. Natl. Acad. Sci. USA 2019, 116, 3646–3655. [Google Scholar] [CrossRef]
- Uller, T.; Mott, B.; Odierna, G.; Olsson, M. Consistent Sex Ratio Bias of Individual Female Dragon Lizards. Biol. Lett. 2006, 2, 569–572. [Google Scholar] [CrossRef][Green Version]
- Ezaz, T.; Quinn, A.E.; Miura, I.; Sarre, S.D.; Georges, A.; Marshall Graves, J.A. The Dragon Lizard Pogona vitticeps Has ZZ/ZW Micro-Sex Chromosomes. Chromosome Res. 2005, 13, 763–776. [Google Scholar] [CrossRef]
- Ezaz, T.; Quinn, A.E.; Sarre, S.D.; O’Meally, D.; Georges, A.; Graves, J.A.M. Molecular Marker Suggests Rapid Changes of Sex-Determining Mechanisms in Australian Dragon Lizards. Chromosome Res. 2009, 17, 91–98. [Google Scholar] [CrossRef]
- Quinn, A.E.; Ezaz, T.; Sarre, S.D.; Graves, J.M.; Georges, A. Extension, Single-Locus Conversion and Physical Mapping of Sex Chromosome Sequences Identify the Z Microchromosome and Pseudo-Autosomal Region in a Dragon Lizard, Pogona vitticeps. Heredity 2010, 104, 410–417. [Google Scholar] [CrossRef]
- Young, M.J.; O’Meally, D.; Sarre, S.D.; Georges, A.; Ezaz, T. Molecular Cytogenetic Map of the Central Bearded Dragon, Pogona vitticeps (Squamata: Agamidae). Chromosome Res. 2013, 21, 361–374. [Google Scholar] [CrossRef]
- Georges, A.; Li, Q.; Lian, J.; O’Meally, D.; Deakin, J.; Wang, Z.; Zhang, P.; Fujita, M.; Patel, H.R.; Holleley, C.E.; et al. High-Coverage Sequencing and Annotated Assembly of the Genome of the Australian Dragon Lizard Pogona vitticeps. GigaScience 2015, 4, 45. [Google Scholar] [CrossRef] [PubMed]
- Deakin, J.E.; Edwards, M.J.; Patel, H.; O’Meally, D.; Lian, J.; Stenhouse, R.; Ryan, S.; Livernois, A.M.; Azad, B.; Holleley, C.E.; et al. Anchoring Genome Sequence to Chromosomes of the Central Bearded Dragon (Pogona vitticeps) Enables Reconstruction of Ancestral Squamate Macrochromosomes and Identifies Sequence Content of the Z Chromosome. BMC Genom. 2016, 17, 447. [Google Scholar] [CrossRef] [PubMed]
- Ezaz, T.; Azad, B.; O’Meally, D.; Young, M.J.; Matsubara, K.; Edwards, M.J.; Zhang, X.; Holleley, C.E.; Deakin, J.E.; Marshall Graves, J.A.; et al. Sequence and Gene Content of a Large Fragment of a Lizard Sex Chromosome and Evaluation of Candidate Sex Differentiating Gene R-Spondin 1. BMC Genom. 2013, 14, 899. [Google Scholar] [CrossRef]
- Luo, X.; Ikeda, Y.; Lala, D.; Rice, D.; Wong, M.; Parker, K.L. Steroidogenic Factor 1 (SF-1) Is Essential for Endocrine Development and Function. J. Steroid Biochem. Mol. Biol. 1999, 69, 13–18. [Google Scholar] [CrossRef]
- Quinn, A.E.; Georges, A.; Sarre, S.D.; Guarino, F.; Ezaz, T.; Graves, J.A.M. Temperature Sex Reversal Implies Sex Gene Dosage in a Reptile. Science 2007, 316, 411. [Google Scholar] [CrossRef] [PubMed]
- Holleley, C.E.; O’Meally, D.; Sarre, S.D.; Marshall Graves, J.A.; Ezaz, T.; Matsubara, K.; Azad, B.; Zhang, X.; Georges, A. Sex Reversal Triggers the Rapid Transition from Genetic to Temperature-Dependent Sex. Nature 2015, 523, 79–82. [Google Scholar] [CrossRef] [PubMed]
- Pokorná, M.; Kratochvíl, L. Phylogeny of Sex-Determining Mechanisms in Squamate Reptiles: Are Sex Chromosomes an Evolutionary Trap? Zool. J. Linn. Soc. 2009, 156, 168–183. [Google Scholar] [CrossRef]
- Gorman, G.C.; Atkins, L. Chromosomal Heteromorphism in Some Male Lizards of the Genus Anolis. Am. Nat. 1966, 100, 579–583. [Google Scholar] [CrossRef]
- Cole, C.J.; Lowe, C.H.; Wright, J.W. Sex Chromosomes in Lizards. Science 1967, 155, 1028–1029. [Google Scholar] [CrossRef]
- Alföldi, J.; Di Palma, F.; Grabherr, M.; Williams, C.; Kong, L.; Mauceli, E.; Russell, P.; Lowe, C.B.; Glor, R.E.; Jaffe, J.D.; et al. The Genome of the Green Anole Lizard and a Comparative Analysis with Birds and Mammals. Nature 2011, 477, 587–591. [Google Scholar] [CrossRef]
- Rovatsos, M.; Altmanová, M.; Pokorná, M.J.; Kratochvíl, L. Novel X-Linked Genes Revealed by Quantitative Polymerase Chain Reaction in the Green Anole, Anolis carolinensis. G3 Genes Genomes Genet. 2014, 4, 2107–2113. [Google Scholar] [CrossRef]
- Rupp, S.M.; Webster, T.H.; Olney, K.C.; Hutchins, E.D.; Kusumi, K.; Sayres, M.A.W. Evolution of Dosage Compensation in Anolis carolinensis, a Reptile with XX/XY Chromosomal Sex Determination. Genome Biol. Evol. 2017, 9, 231–240. [Google Scholar] [CrossRef]
- Marin, R.; Cortez, D.; Lamanna, F.; Pradeepa, M.M.; Leushkin, E.; Julien, P.; Liechti, A.; Halbert, J.; Brüning, T.; Mössinger, K.; et al. Convergent Origination of a Drosophila-like Dosage Compensation Mechanism in a Reptile Lineage. Genome Res. 2017, 27, 1974–1987. [Google Scholar] [CrossRef] [PubMed]
- Gamble, T.; Geneva, A.J.; Glor, R.E.; Zarkower, D. Anolis Sex Chromosomes Are Derived from a Single Ancestral Pair. Evol. Int. J. Org. Evol. 2014, 68, 1027–1041. [Google Scholar] [CrossRef] [PubMed]
- Rovatsos, M.; Altmanová, M.; Pokorná, M.; Kratochvíl, L. Conserved Sex Chromosomes across Adaptively Radiated Anolis Lizards. Evol. Int. J. Org. Evol. 2014, 68, 2079–2085. [Google Scholar] [CrossRef] [PubMed]
- Kichigin, I.G.; Giovannotti, M.; Makunin, A.I.; Ng, B.L.; Kabilov, M.R.; Tupikin, A.E.; Barucchi, V.C.; Splendiani, A.; Ruggeri, P.; Rens, W.; et al. Evolutionary Dynamics of Anolis Sex Chromosomes Revealed by Sequencing of Flow Sorting-Derived Microchromosome-Specific DNA. Mol. Genet. Genom. MGG 2016, 291, 1955–1966. [Google Scholar] [CrossRef]
- Giovannotti, M.; Trifonov, V.A.; Paoletti, A.; Kichigin, I.G.; O’Brien, P.C.M.; Kasai, F.; Giovagnoli, G.; Ng, B.L.; Ruggeri, P.; Cerioni, P.N.; et al. New Insights into Sex Chromosome Evolution in Anole Lizards (Reptilia, Dactyloidae). Chromosoma 2017, 126, 245–260. [Google Scholar] [CrossRef]
- Rovatos, M.; Pokorná, M.; Altmanová, M.; Kratochvíl, L. Cretaceous Park of Sex Determination: Sex Chromosomes Are Conserved across Iguanas. Biol. Lett. 2014, 10, 20131093. [Google Scholar] [CrossRef]
- Altmanová, M.; Rovatsos, M.; Johnson Pokorná, M.; Veselý, M.; Wagner, F.; Kratochvíl, L. All Iguana Families with the Exception of Basilisks Share Sex Chromosomes. Zool. Jena Ger. 2018, 126, 98–102. [Google Scholar] [CrossRef] [PubMed]
- Gamble, T.; Zarkower, D. Identification of Sex-Specific Molecular Markers Using Restriction Site-Associated DNA Sequencing. Mol. Ecol. Resour. 2014, 14, 902–913. [Google Scholar] [CrossRef] [PubMed]
- Acosta, A.; Martínez-Pacheco, M.L.; Díaz-Barba, K.; Porras, N.; Gutiérrez-Mariscal, M.; Cortez, D. Deciphering Ancestral Sex Chromosome Turnovers Based on Analysis of Male Mutation Bias. Genome Biol. Evol. 2019, 11, 3054–3067. [Google Scholar] [CrossRef]
- Conrad, T.; Akhtar, A. Dosage Compensation in Drosophila Melanogaster: Epigenetic Fine-Tuning of Chromosome-Wide Transcription. Nat. Rev. Genet. 2012, 13, 123–134. [Google Scholar] [CrossRef]
- Nielsen, S.V.; Guzmán-Méndez, I.A.; Gamble, T.; Blumer, M.; Pinto, B.J.; Kratochvíl, L.; Rovatsos, M. Escaping the evolutionary trap? Sex chromosome turnover in basilisks and related lizards (Corytophanidae: Squamata). Biol. Lett. 2019, 15, 20190498. [Google Scholar] [CrossRef]
- Acosta, A.; Suárez-Varón, G.; Rodríguez-Miranda, L.A.; Lira-Noriega, A.; Aguilar-Gómez, D.; Gutiérrez-Mariscal, M.; Hernández-Gallegos, O.; Méndez-de-la-Cruz, F.; Cortez, D. Corytophanids Replaced the Pleurodont XY System with a New Pair of XY Chromosomes. Genome Biol. Evol. 2019, 11, 2666–2677. [Google Scholar] [CrossRef] [PubMed]
- Mezzasalma, M.; Guarino, F.M.; Odierna, G. Lizards as Model Organisms of Sex Chromosome Evolution: What We Really Know from a Systematic Distribution of Available Data? Genes 2021, 12, 1341. [Google Scholar] [CrossRef] [PubMed]
- Becak, W.; Becak, M.L.; Nazareth, H.R.; Ohno, S. Close karyological kinship between the reptilian suborder Serpentes and the class Aves. Chromosoma 1964, 15, 606–617. [Google Scholar] [CrossRef]
- Ohno, S. Sex Chromosomes and Sex-Linked Genes; Springer Science & Business Media: Berlin/Heidelberg, Germany, 1967; Volume 1. [Google Scholar]
- Rovatsos, M.; Altmanová, M.; Pokorná, M.J.; Augstenová, B.; Kratochvíl, L. Cytogenetics of the Javan File Snake (Acrochordus javanicus) and the Evolution of Snake Sex Chromosomes. J. Zool. Syst. Evol. Res. 2018, 56, 117–125. [Google Scholar] [CrossRef]
- Rovatsos, M.; Pokorná, M.J.; Kratochvíl, L. Differentiation of Sex Chromosomes and Karyotype Characterisation in the Dragonsnake Xenodermus javanicus (Squamata: Xenodermatidae). Cytogenet. Genome Res. 2015, 147, 48–54. [Google Scholar] [CrossRef] [PubMed]
- Matsubara, K.; Tarui, H.; Toriba, M.; Yamada, K.; Nishida-Umehara, C.; Agata, K.; Matsuda, Y. Evidence for Different Origin of Sex Chromosomes in Snakes, Birds, and Mammals and Step-Wise Differentiation of Snake Sex Chromosomes. Proc. Natl. Acad. Sci. USA 2006, 103, 18190–18195. [Google Scholar] [CrossRef]
- Vicoso, B.; Emerson, J.J.; Zektser, Y.; Mahajan, S.; Bachtrog, D. Comparative Sex Chromosome Genomics in Snakes: Differentiation, Evolutionary Strata, and Lack of Global Dosage Compensation. PLoS Biol. 2013, 11, e1001643. [Google Scholar] [CrossRef]
- Rovatsos, M.; Vukić, J.; Lymberakis, P.; Kratochvíl, L. Evolutionary Stability of Sex Chromosomes in Snakes. Proc. Biol. Sci. 2015, 282, 20151992. [Google Scholar] [CrossRef] [PubMed]
- Rovatsos, M.; Augstenová, B.; Altmanová, M.; Sloboda, M.; Kodym, P.; Kratochvíl, L. Triploid Colubrid Snake Provides Insight into the Mechanism of Sex Determination in Advanced Snakes. Sex. Dev. Genet. Mol. Biol. Evol. Endocrinol. Embryol. Pathol. Sex Determ. Differ. 2018, 12, 251–255. [Google Scholar] [CrossRef] [PubMed]
- Booth, W.; Schuett, G.W. The Emerging Phylogenetic Pattern of Parthenogenesis in Snakes. Biol. J. Linn. Soc. 2016, 118, 172–186. [Google Scholar] [CrossRef]
- Mallery Jr, C.S.; Carrillo, M.M. A Case Study of Sex-Linkage in Python Regius (Serpentes: Boidae), with New Insights into Sex Determination in Henophidia. Phyllomedusa 2016, 15, 29–42. [Google Scholar] [CrossRef]
- Gamble, T.; Castoe, T.A.; Nielsen, S.V.; Banks, J.L.; Card, D.C.; Schield, D.R.; Schuett, G.W.; Booth, W. The Discovery of XY Sex Chromosomes in a Boa and Python. Curr. Biol. 2017, 27, 2148–2153.e4. [Google Scholar] [CrossRef] [PubMed]
- Augstenová, B.; Johnson Pokorná, M.; Altmanová, M.; Frynta, D.; Rovatsos, M.; Kratochvíl, L. ZW, XY, and yet ZW: Sex Chromosome Evolution in Snakes Even More Complicated. Evol. Int. J. Org. Evol. 2018. [Google Scholar] [CrossRef]
- Augstenová, B.; Mazzoleni, S.; Kostmann, A.; Altmanová, M.; Frynta, D.; Kratochvíl, L.; Rovatsos, M. Cytogenetic Analysis Did Not Reveal Differentiated Sex Chromosomes in Ten Species of Boas and Pythons (Reptilia: Serpentes). Genes 2019, 10, 934. [Google Scholar] [CrossRef]
- Matsubara, K.; Kumazawa, Y.; Ota, H.; Nishida, C.; Matsuda, Y. Karyotype Analysis of Four Blind Snake Species (Reptilia: Squamata: Scolecophidia) and Karyotypic Changes in Serpentes. Cytogenet. Genome Res. 2019, 157, 98–106. [Google Scholar] [CrossRef]
- Palmer, D.H.; Rogers, T.F.; Dean, R.; Wright, A.E. How to identify sex chromosomes and their turnover. Mol. Ecol. 2019, 28, 4709–4724. [Google Scholar] [CrossRef]
- Vicoso, B. Molecular and Evolutionary Dynamics of Animal Sex-Chromosome Turnover. Nat. Ecol. Evol. 2019, 3, 1632–1641. [Google Scholar] [CrossRef]
- Foster, J.W.; Dominguez-Steglich, M.A.; Guioli, S.; Kwok, C.; Weller, P.A.; Stevanović, M.; Weissenbach, J.; Mansour, S.; Young, I.D.; Goodfellow, P.N. Campomelic Dysplasia and Autosomal Sex Reversal Caused by Mutations in an SRY-Related Gene. Nature 1994, 372, 525–530. [Google Scholar] [CrossRef] [PubMed]
- Wagner, T.; Wirth, J.; Meyer, J.; Zabel, B.; Held, M.; Zimmer, J.; Pasantes, J.; Bricarelli, F.D.; Keutel, J.; Hustert, E.; et al. Autosomal Sex Reversal and Campomelic Dysplasia Are Caused by Mutations in and around the SRY-Related Gene SOX9. Cell 1994, 79, 1111–1120. [Google Scholar] [CrossRef]
- Bi, W.; Huang, W.; Whitworth, D.J.; Deng, J.M.; Zhang, Z.; Behringer, R.R.; de Crombrugghe, B. Haploinsufficiency of Sox9 Results in Defective Cartilage Primordia and Premature Skeletal Mineralization. Proc. Natl. Acad. Sci. USA 2001, 98, 6698–6703. [Google Scholar] [CrossRef] [PubMed]
- Gao, F.; Maiti, S.; Alam, N.; Zhang, Z.; Deng, J.M.; Behringer, R.R.; Lécureuil, C.; Guillou, F.; Huff, V. The Wilms Tumor Gene, Wt1, Is Required for Sox9 Expression and Maintenance of Tubular Architecture in the Developing Testis. Proc. Natl. Acad. Sci. USA 2006, 103, 11987–11992. [Google Scholar] [CrossRef]
- Sun, W.; Cai, H.; Zhang, G.; Zhang, H.; Bao, H.; Wang, L.; Ye, J.; Qian, G.; Ge, C. Dmrt1 Is Required for Primary Male Sexual Differentiation in Chinese Soft-Shelled Turtle Pelodiscus Sinensis. Sci. Rep. 2017, 7, 4433. [Google Scholar] [CrossRef]
- Rasys, A.M.; Park, S.; Ball, R.E.; Alcala, A.J.; Lauderdale, J.D.; Menke, D.B. CRISPR-Cas9 Gene Editing in Lizards through Microinjection of Unfertilized Oocytes. Cell Rep. 2019, 28, 2288–2292.e3. [Google Scholar] [CrossRef]
- Zhou, Y.; Sun, W.; Cai, H.; Bao, H.; Zhang, Y.; Qian, G.; Ge, C. The Role of Anti-Müllerian Hormone in Testis Differentiation Reveals the Significance of the TGF-β Pathway in Reptilian Sex Determination. Genetics 2019, 213, 1317–1327. [Google Scholar] [CrossRef]
- Stöck, M.; Kratochvíl, L.; Kuhl, H.; Rovatsos, M.; Evans, B.J.; Suh, A.; Valenzuela, N.; Veyrunes, F.; Zhou, Q.; Gamble, T.; et al. A brief review of vertebrate sex evolution with a pledge for integrative research: Towards “sexomics”. Philos. Trans. R. Soc. Lond. B. Biol. Sci. 2021, 376, 20200426. [Google Scholar] [CrossRef]
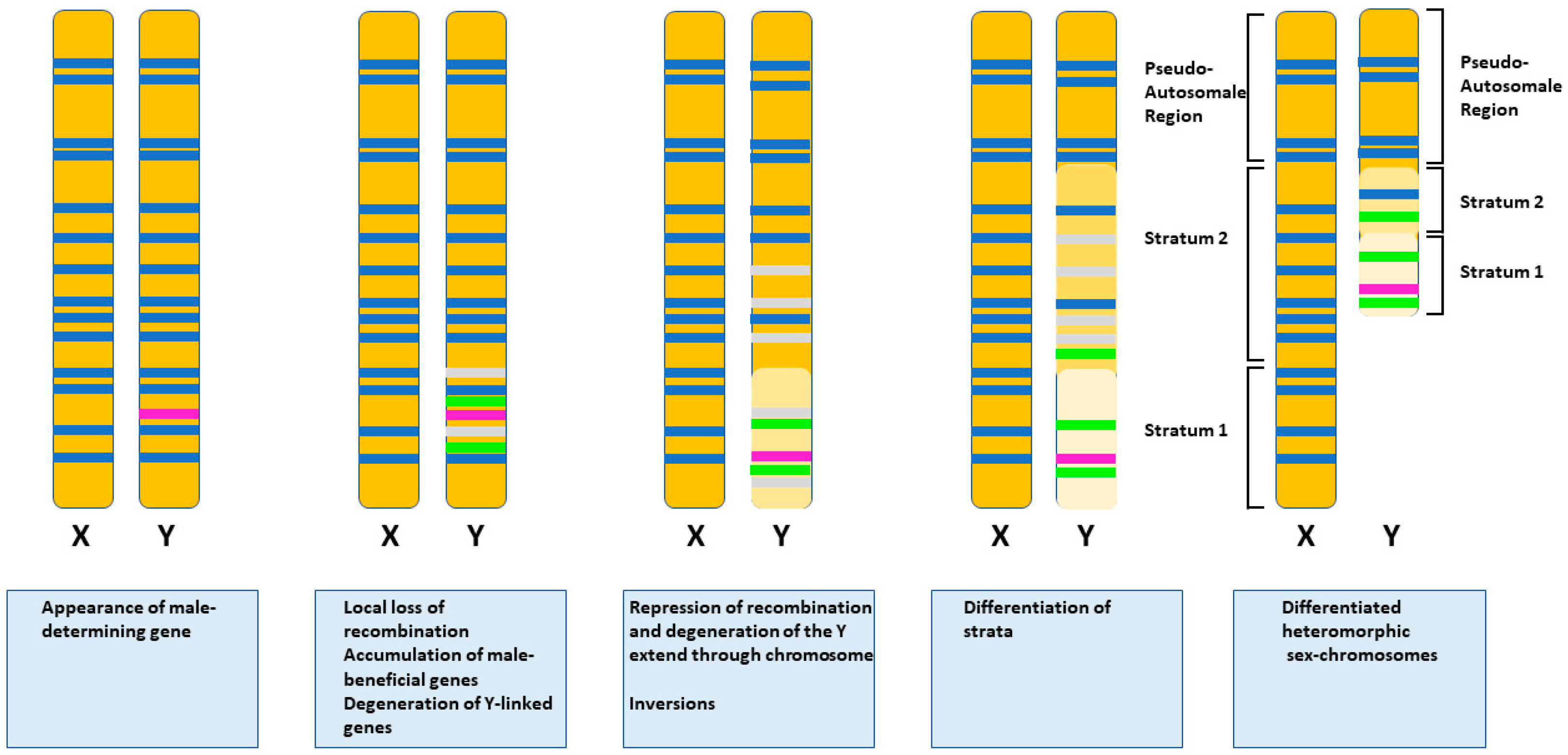
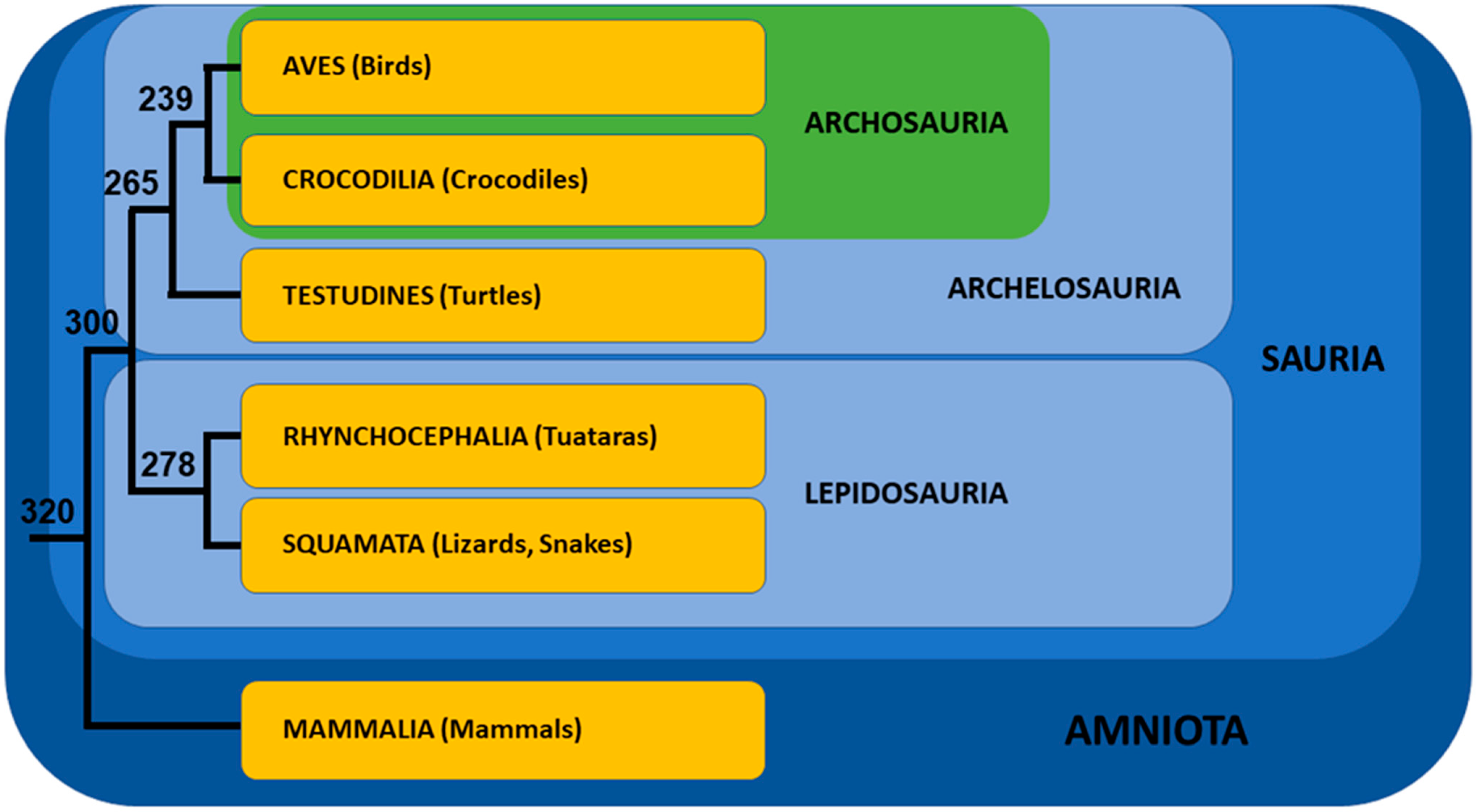
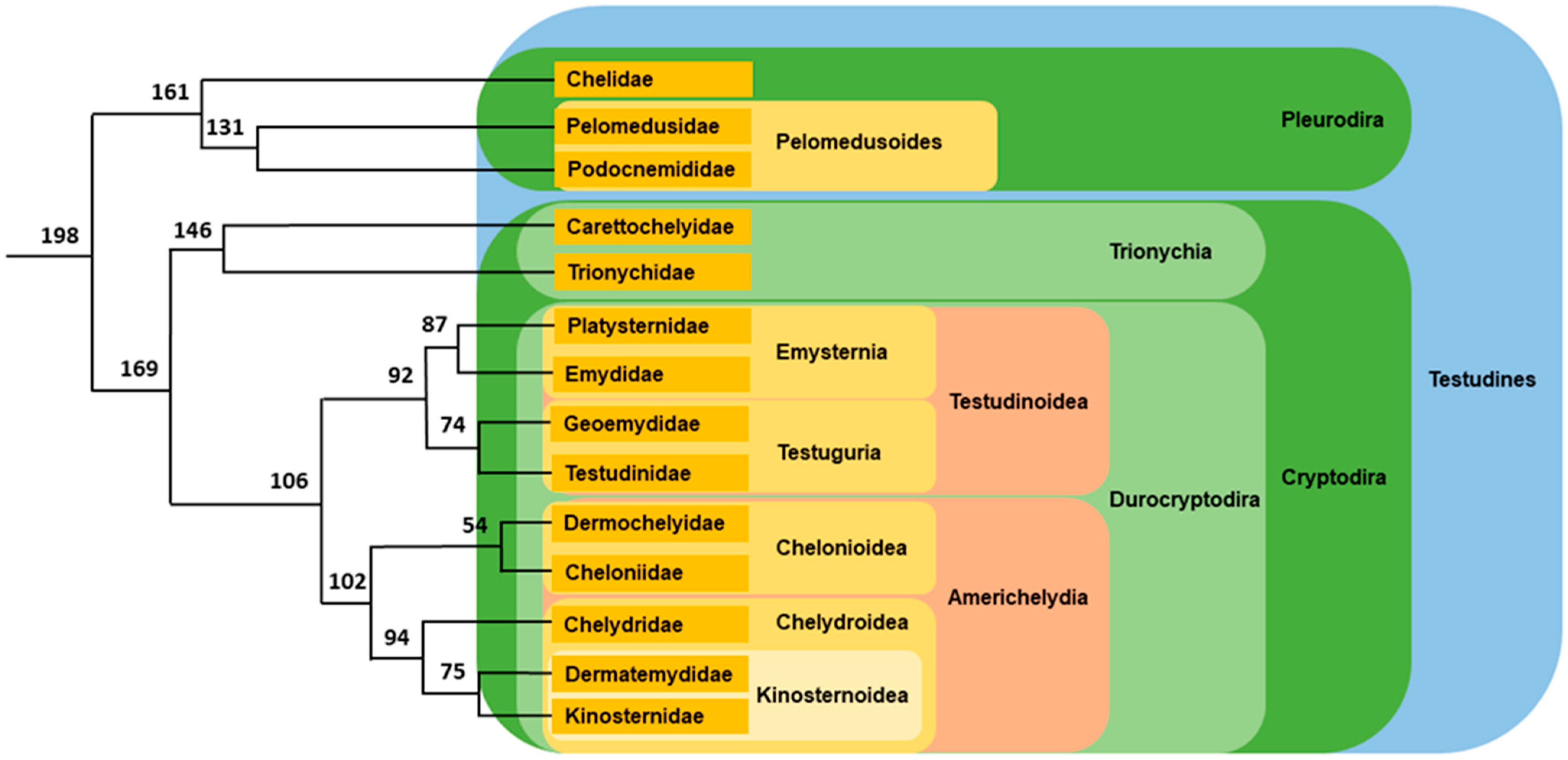
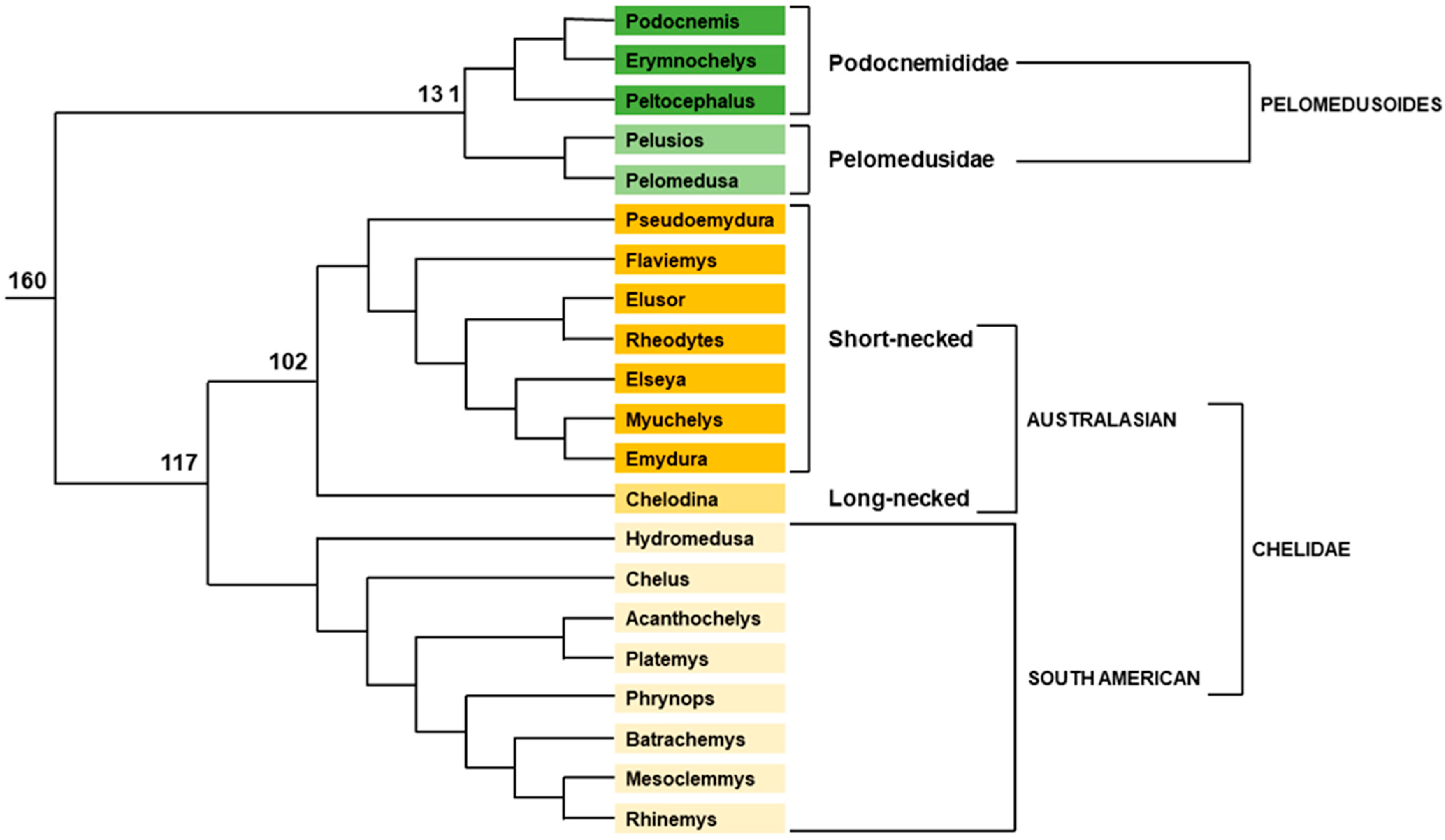

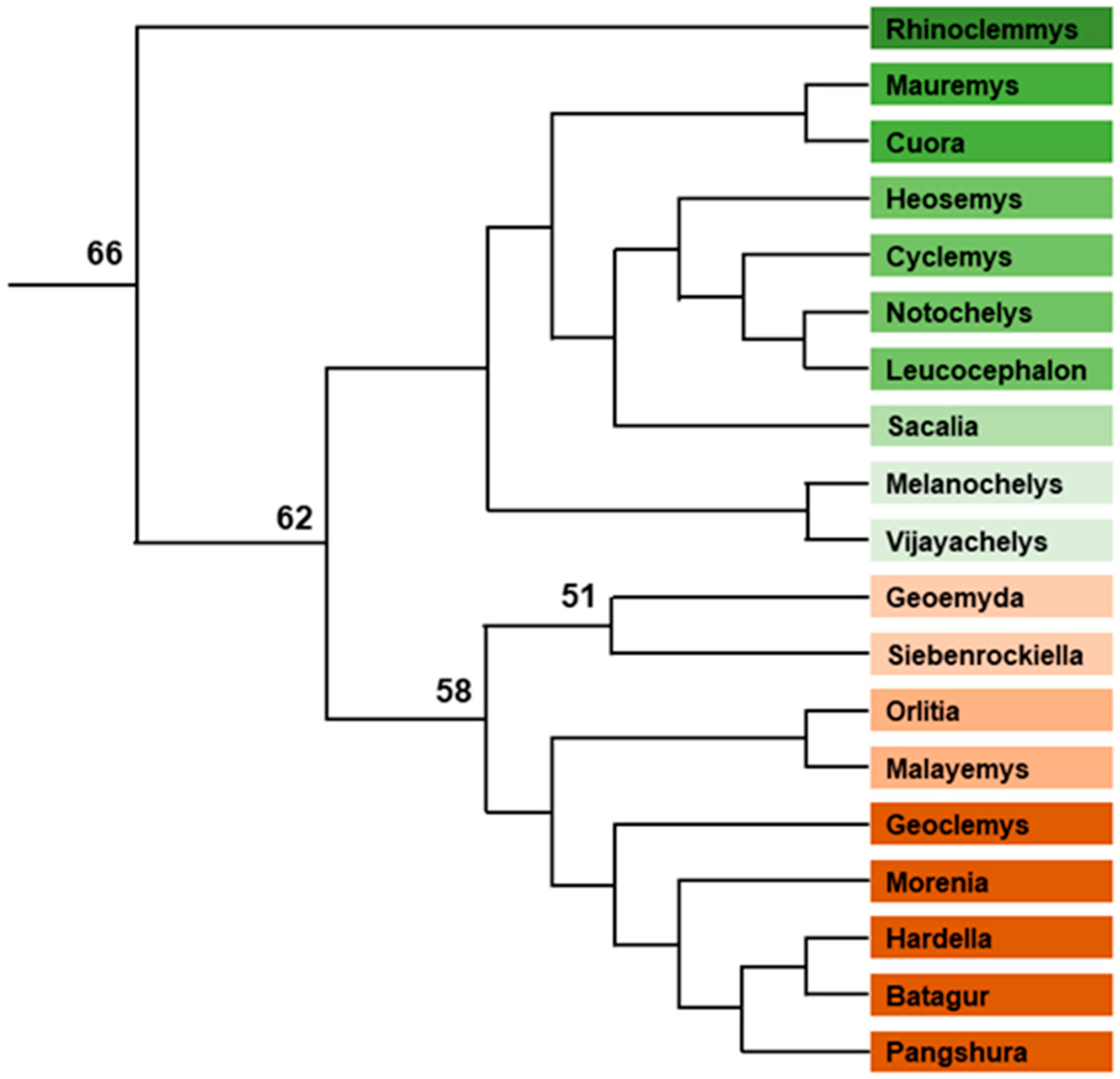

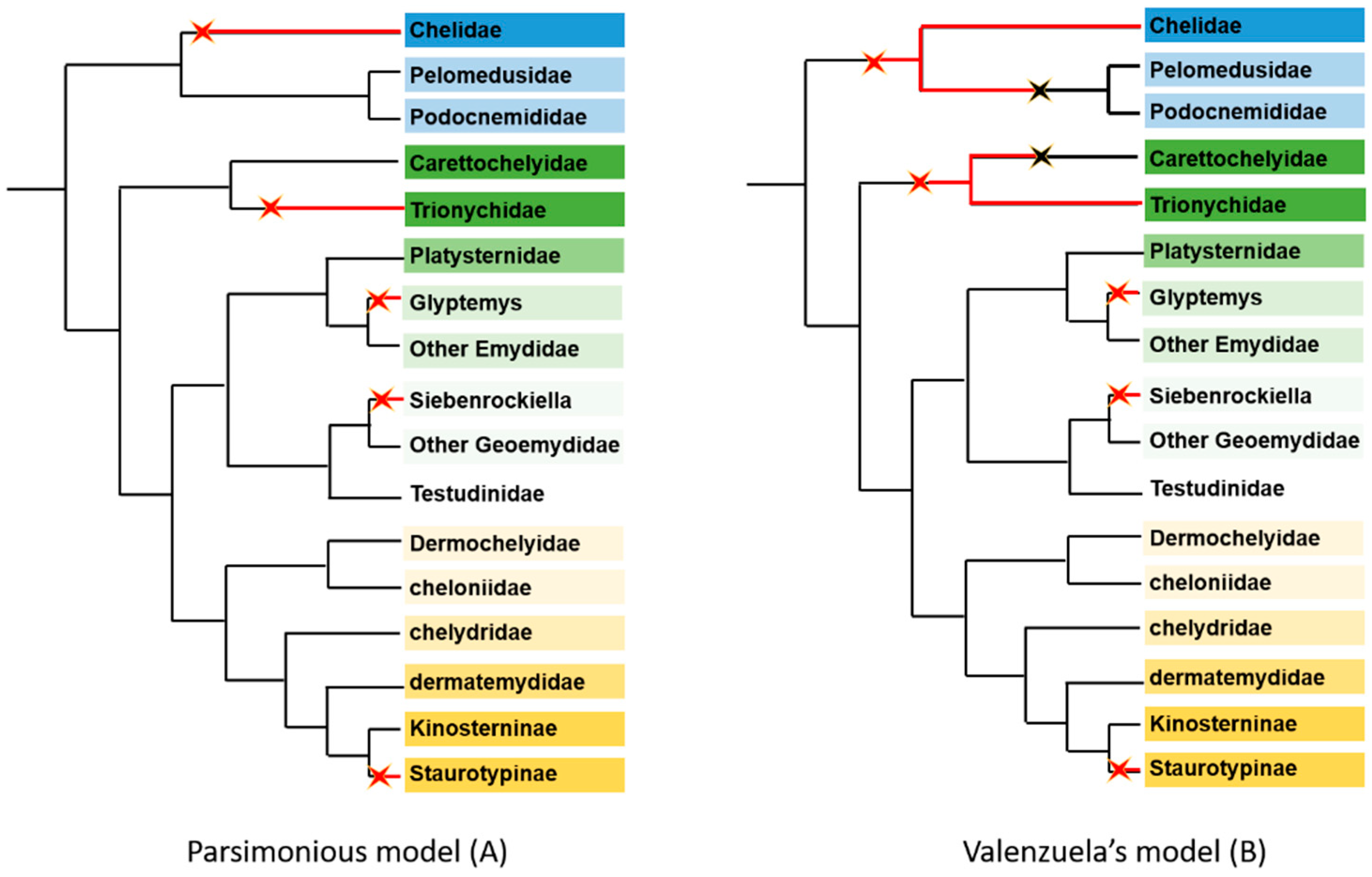
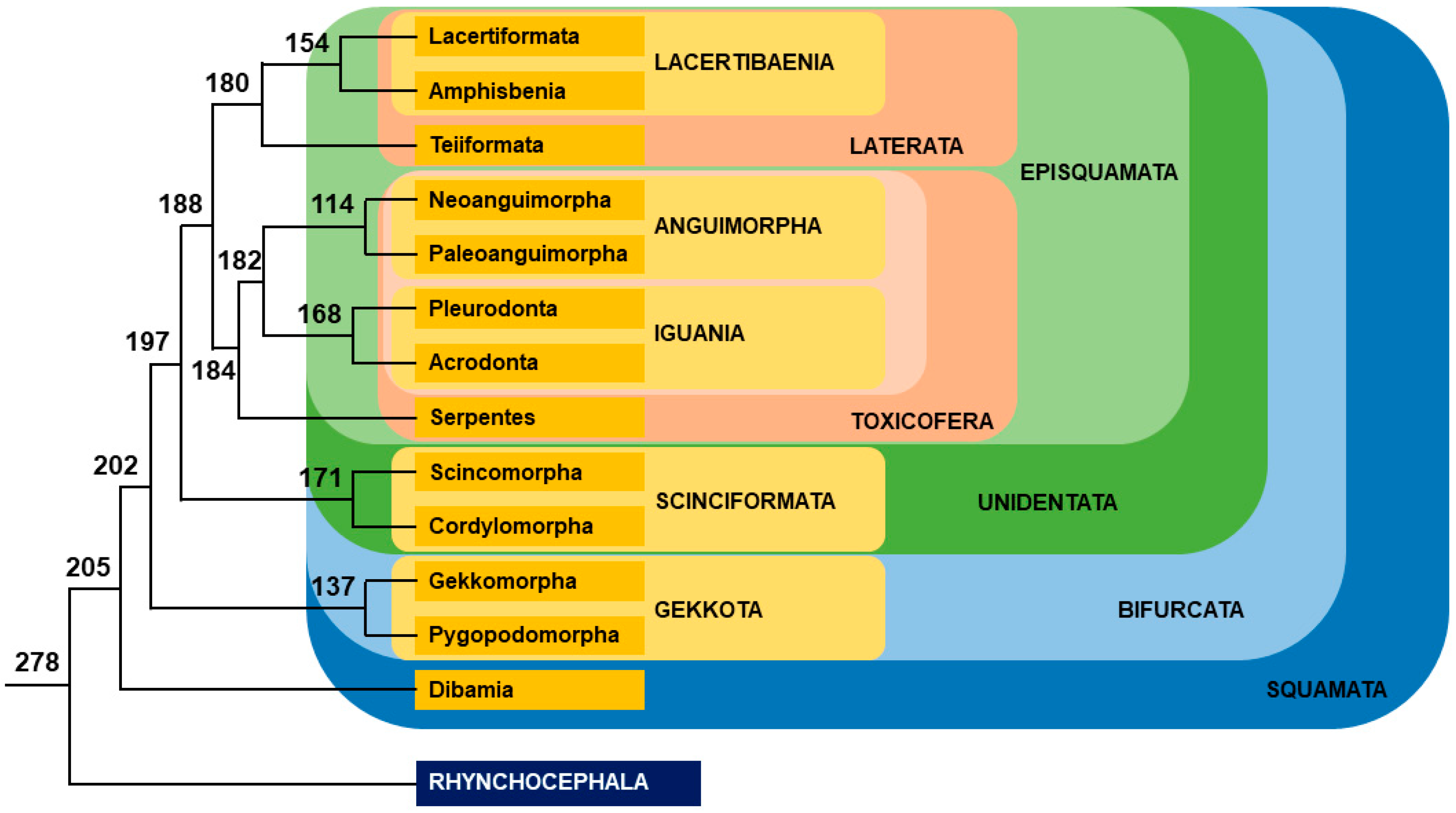

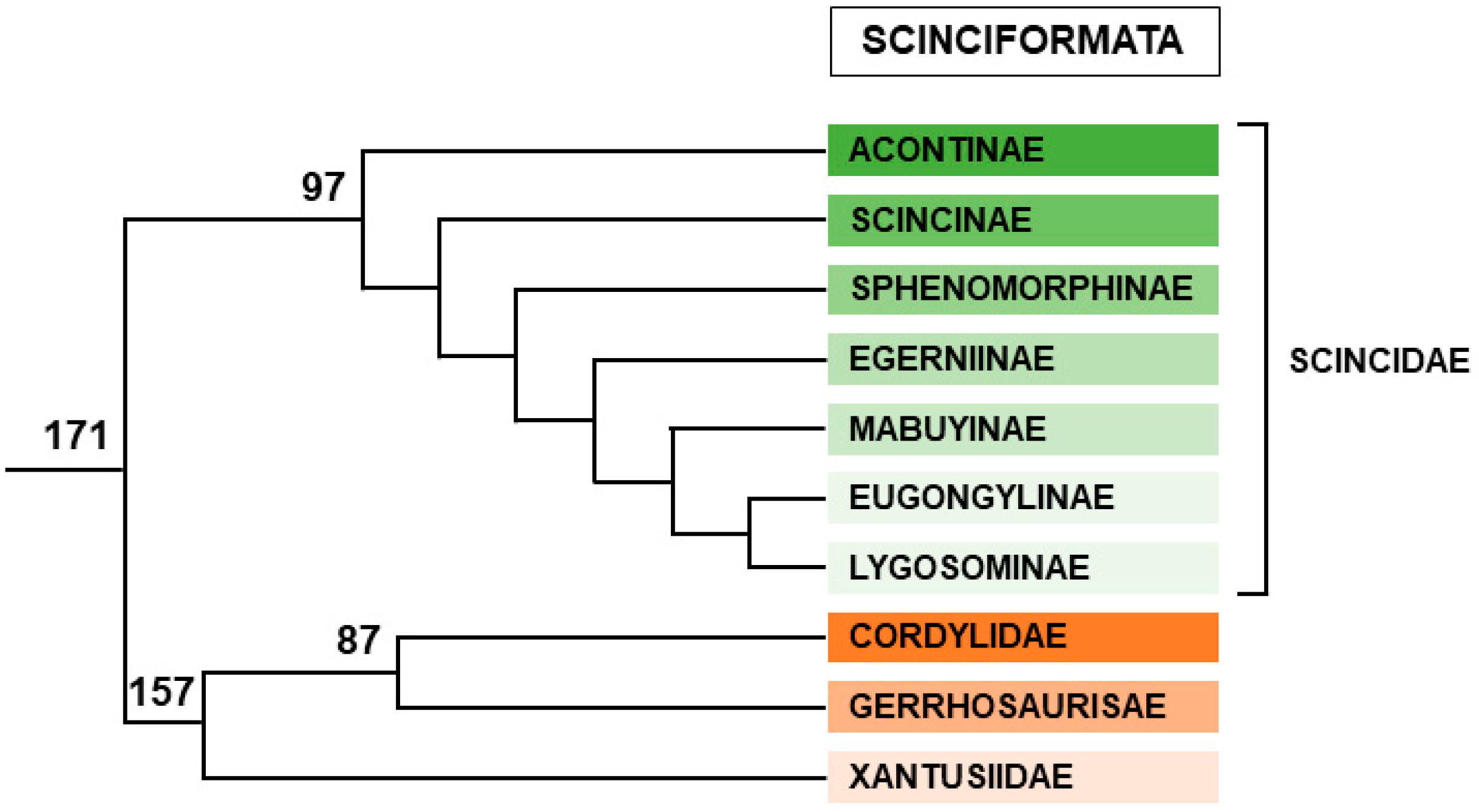
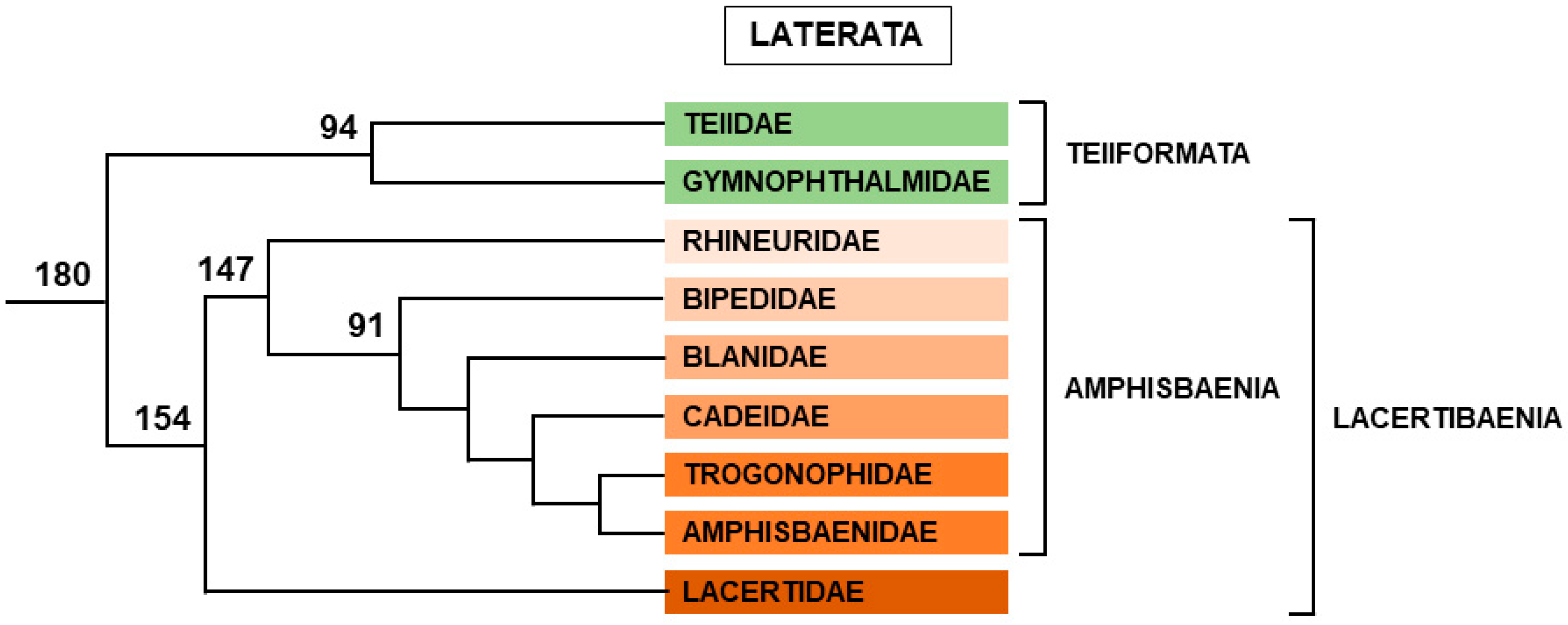
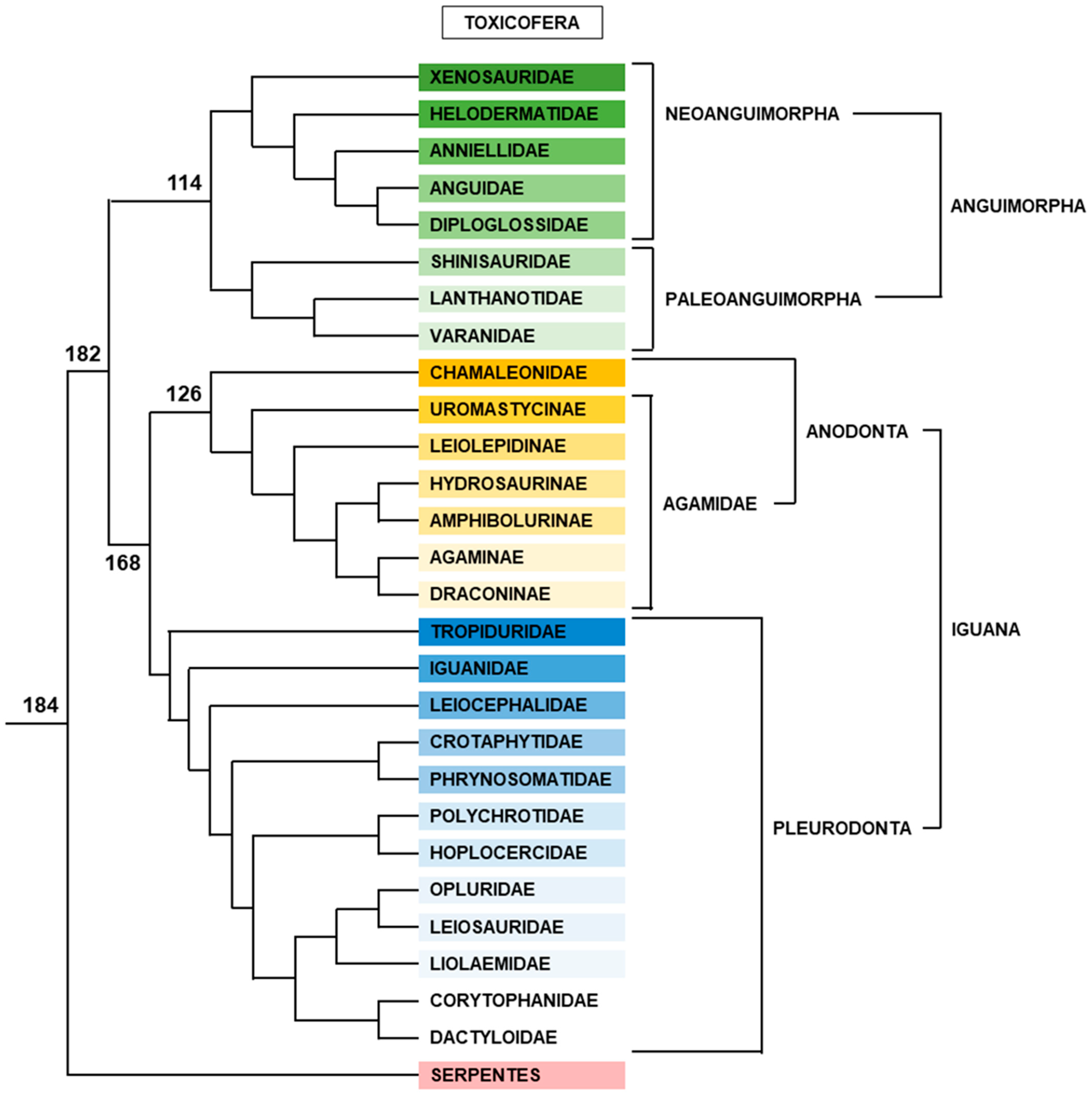

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Thépot, D. Sex Chromosomes and Master Sex-Determining Genes in Turtles and Other Reptiles. Genes 2021, 12, 1822. https://doi.org/10.3390/genes12111822
Thépot D. Sex Chromosomes and Master Sex-Determining Genes in Turtles and Other Reptiles. Genes. 2021; 12(11):1822. https://doi.org/10.3390/genes12111822
Chicago/Turabian StyleThépot, Dominique. 2021. "Sex Chromosomes and Master Sex-Determining Genes in Turtles and Other Reptiles" Genes 12, no. 11: 1822. https://doi.org/10.3390/genes12111822
APA StyleThépot, D. (2021). Sex Chromosomes and Master Sex-Determining Genes in Turtles and Other Reptiles. Genes, 12(11), 1822. https://doi.org/10.3390/genes12111822




