Cortical Granule Distribution and Expression Pattern of Genes Regulating Cellular Component Size, Morphogenesis, and Potential to Differentiation are Related to Oocyte Developmental Competence and Maturational Capacity In Vivo and In Vitro
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals, Oocyte Collection, and Selection
2.2. Oocytes Maturation and Examination
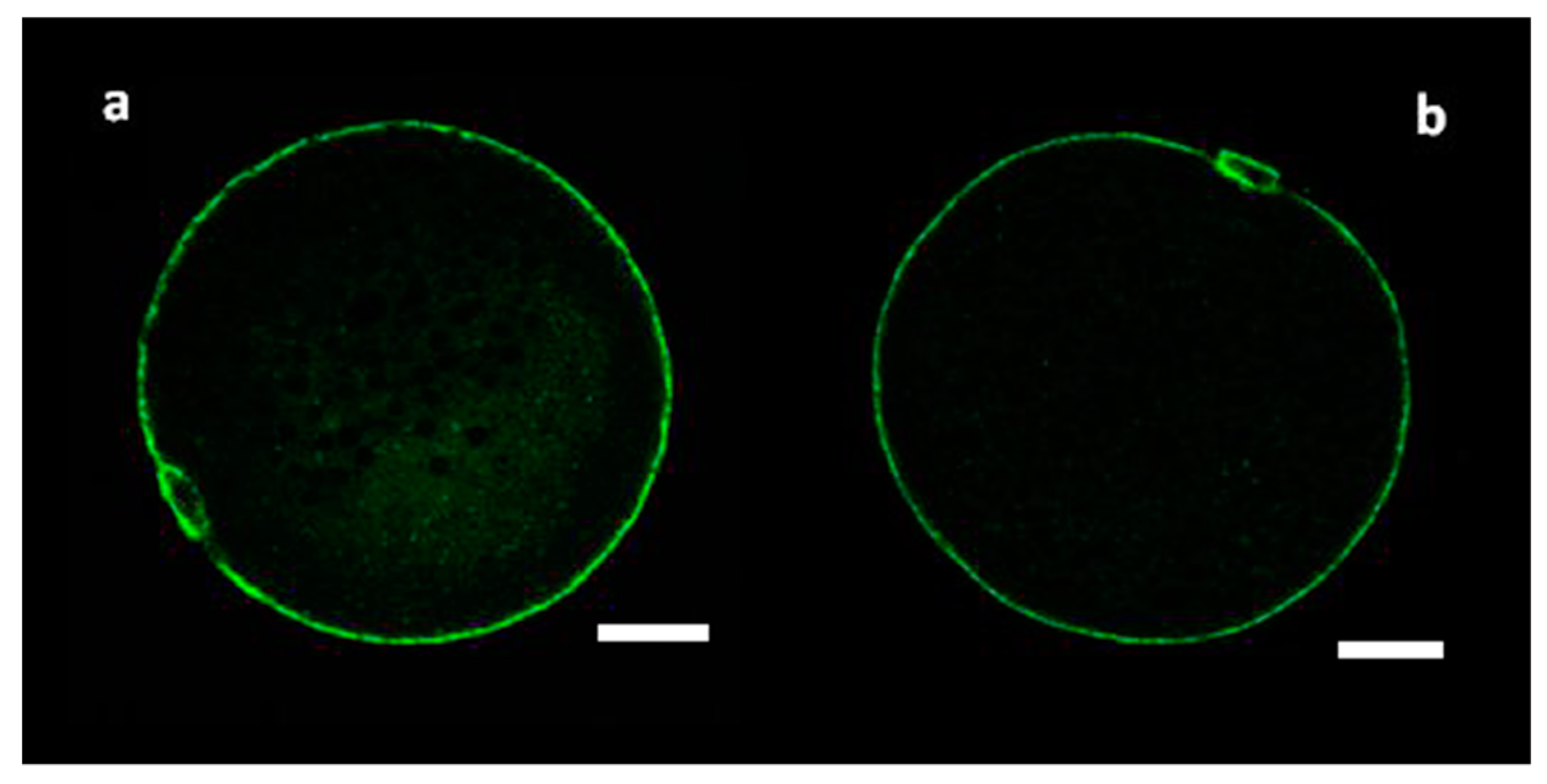
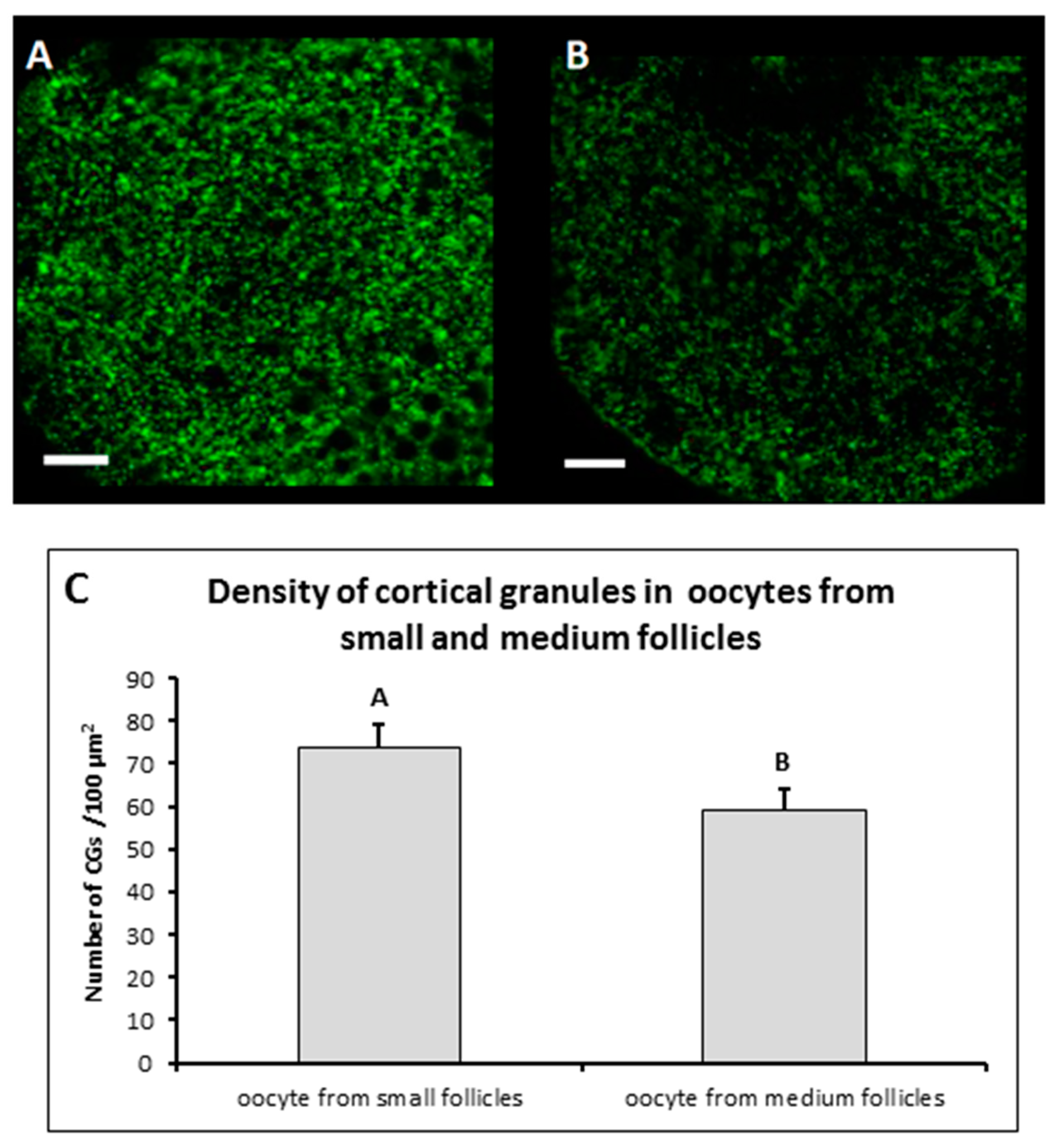
2.3. Cortical Granule Staining and Evaluation of Their Distribution
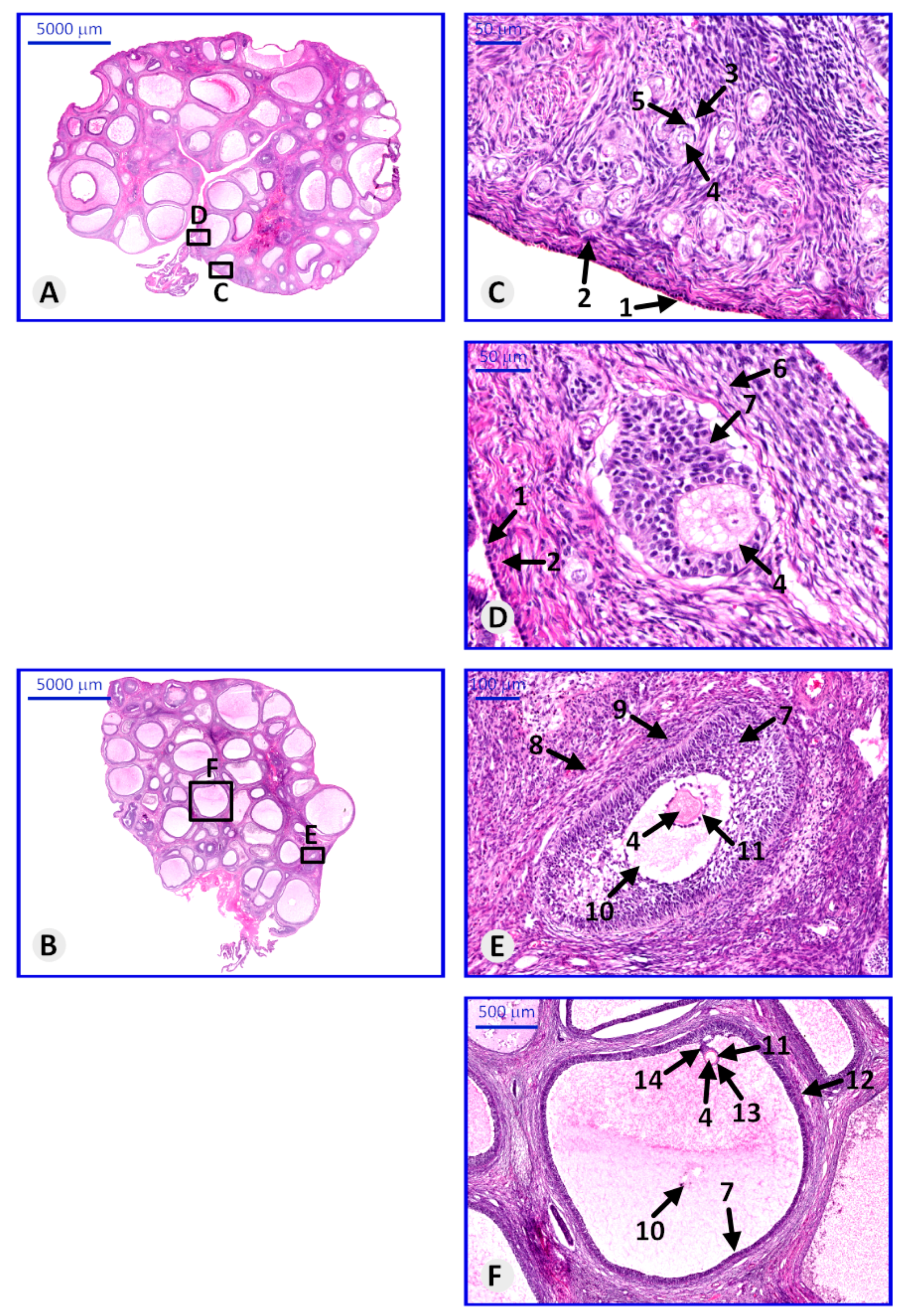
2.4. Histological Examination
2.5. RNA Isolation
2.6. Microarray Expression Analysis and Statistics
2.7. Quantitative Analysis of Polymerase Chain Reaction in Real Time (RT-qPCR) Analysis
3. Results
3.1. Distribution of CGs in Immature MF and SF Oocytes
3.2. Peripheral Density of CGs in Mature MF and SF Oocytes
3.3. Histological Structure of Porcine Ovary and Follicles
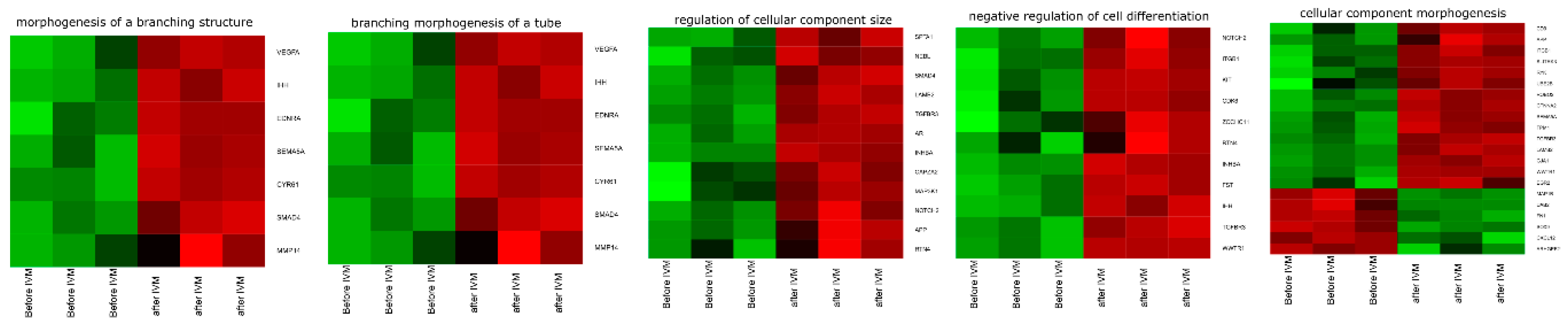
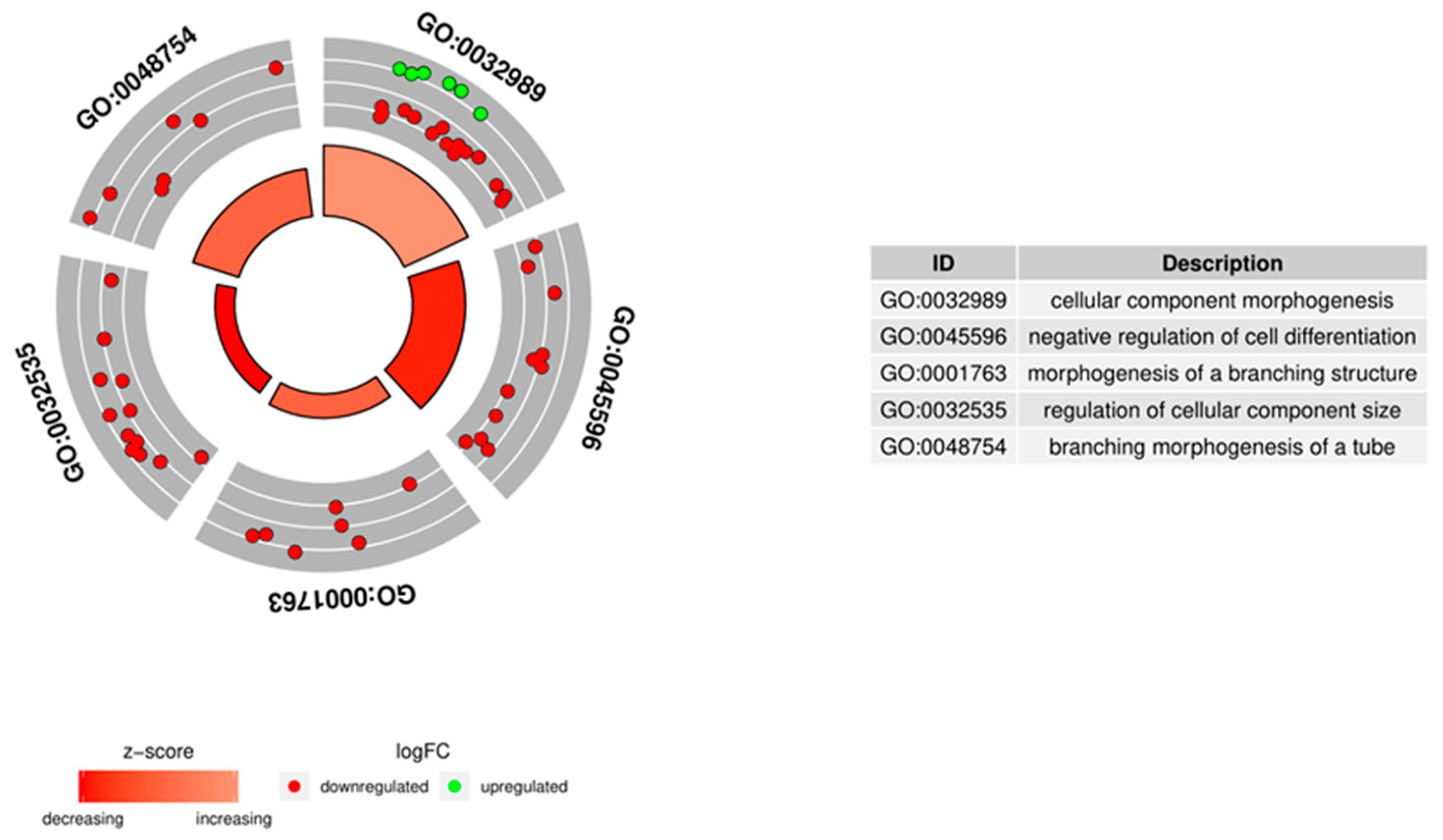
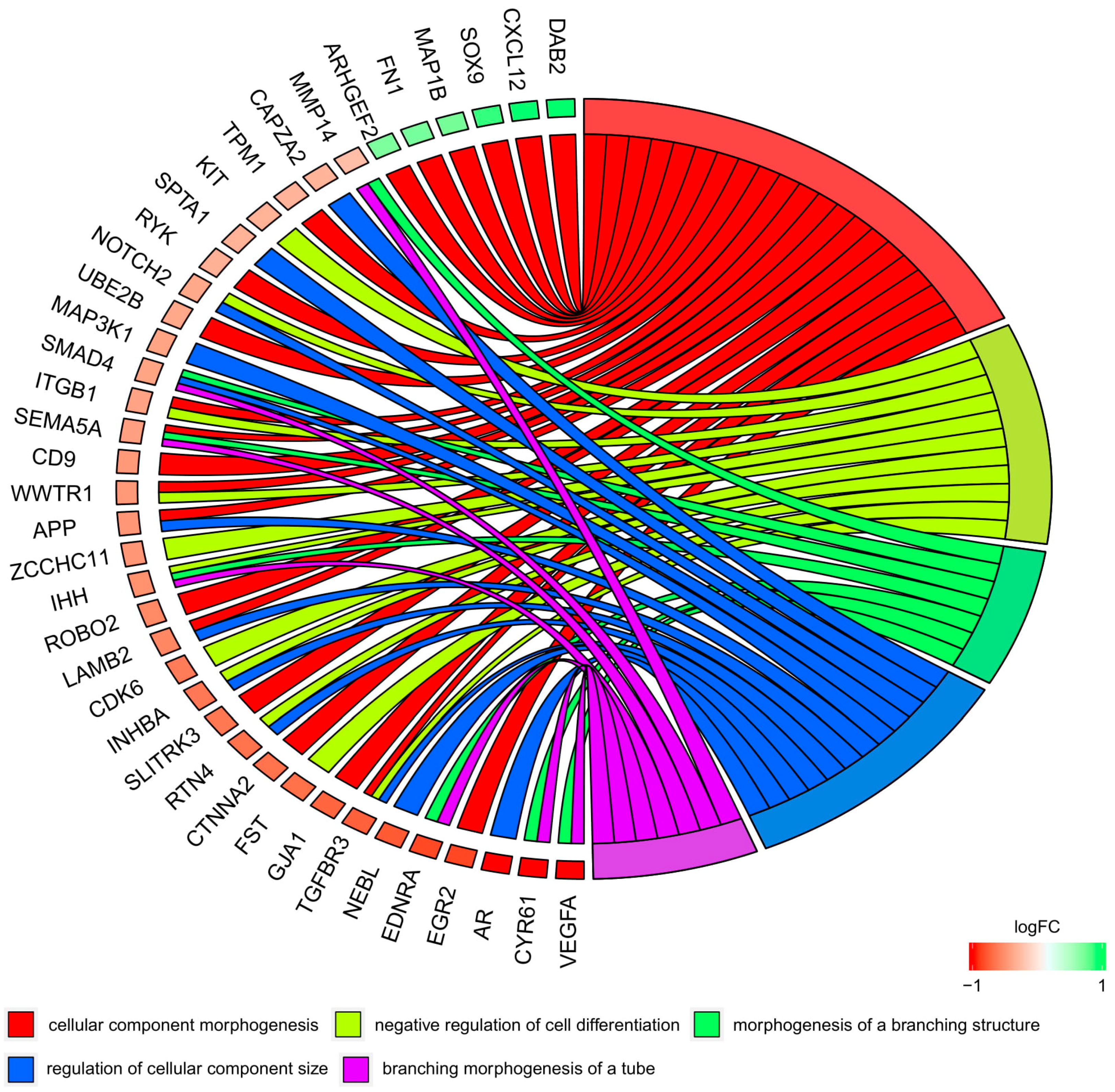
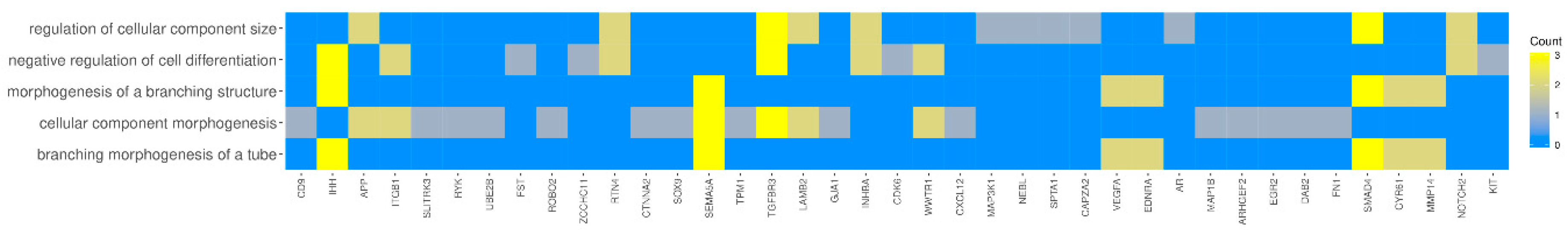
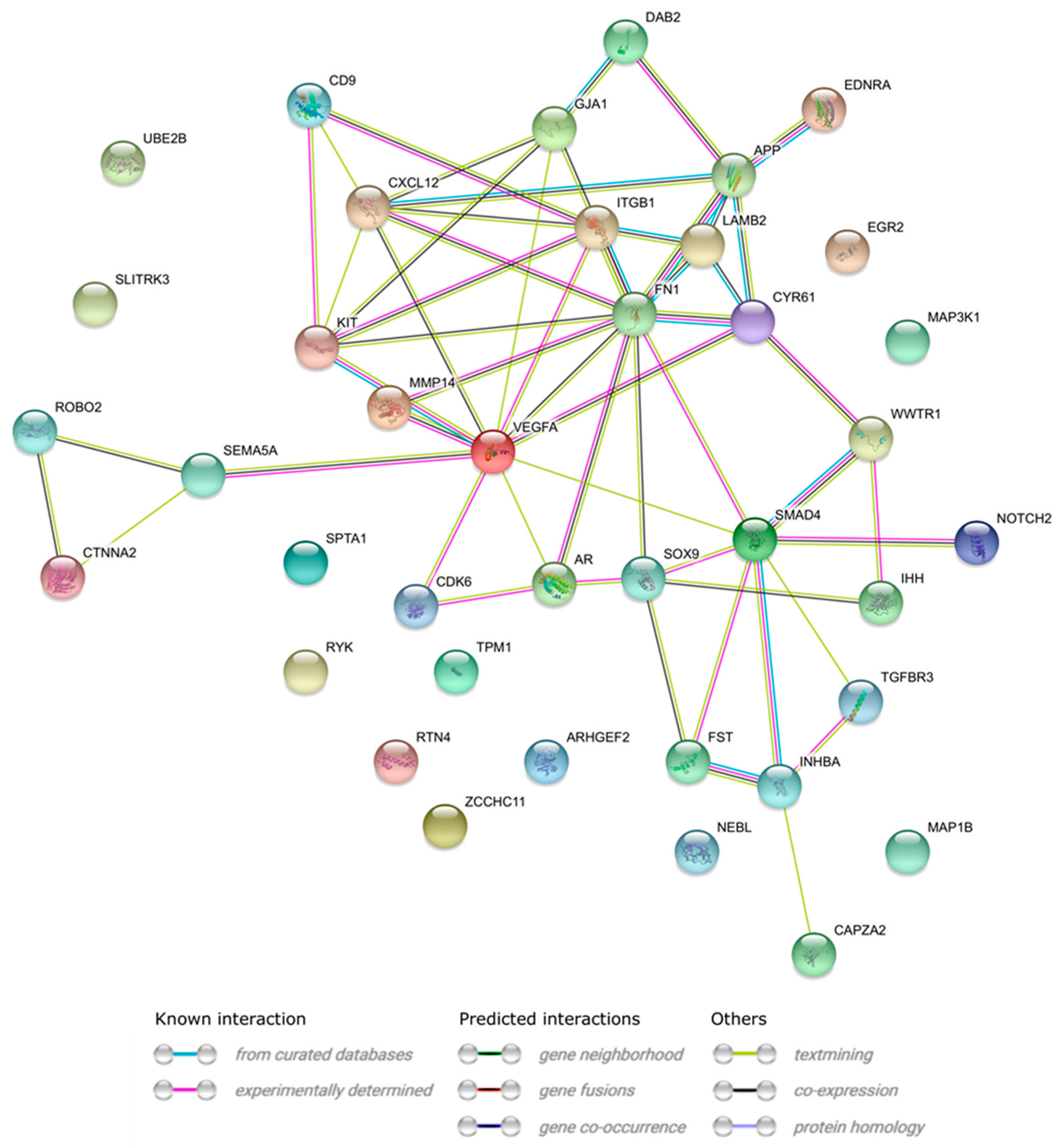
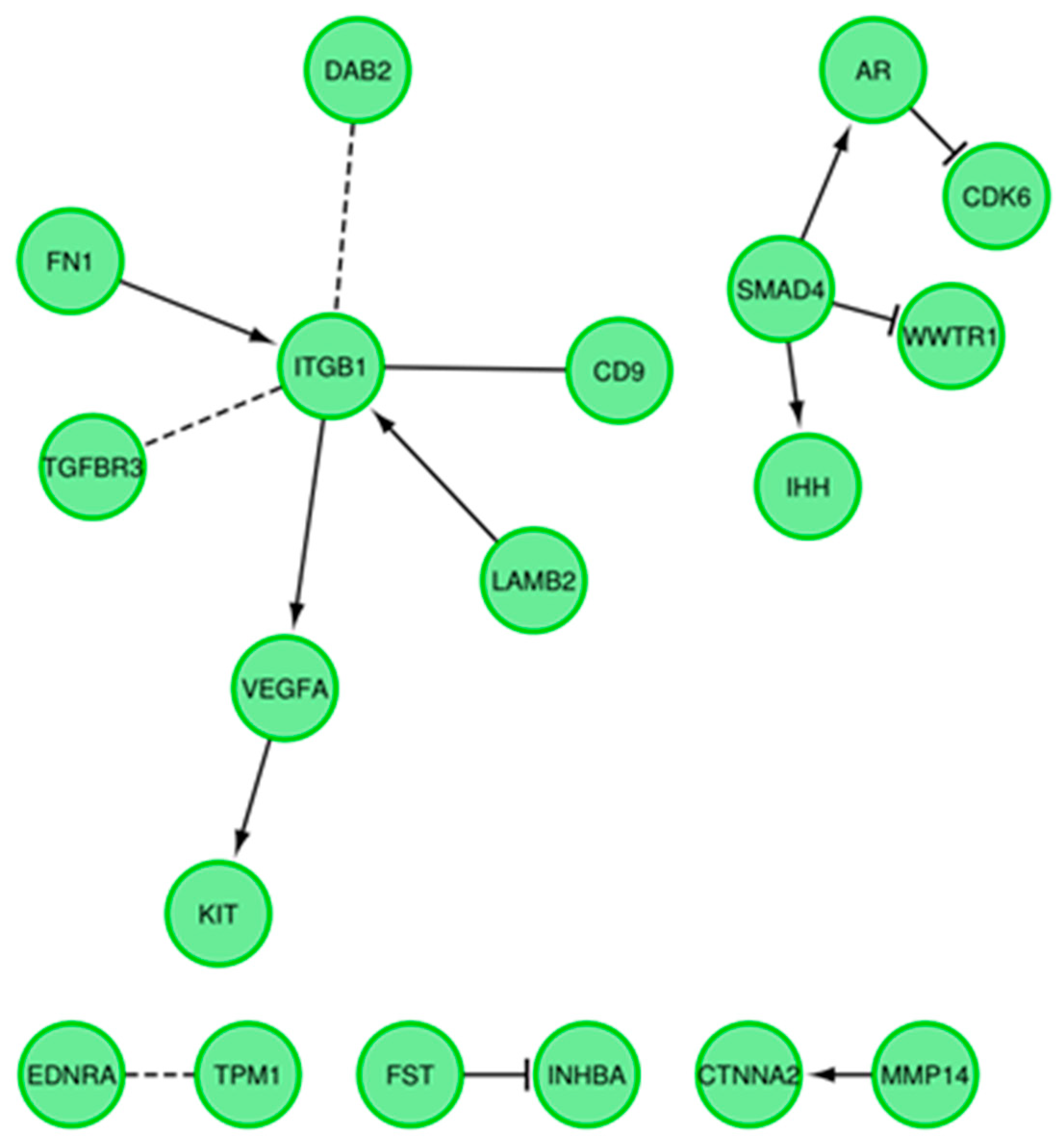
3.4. Microarrays Analysis
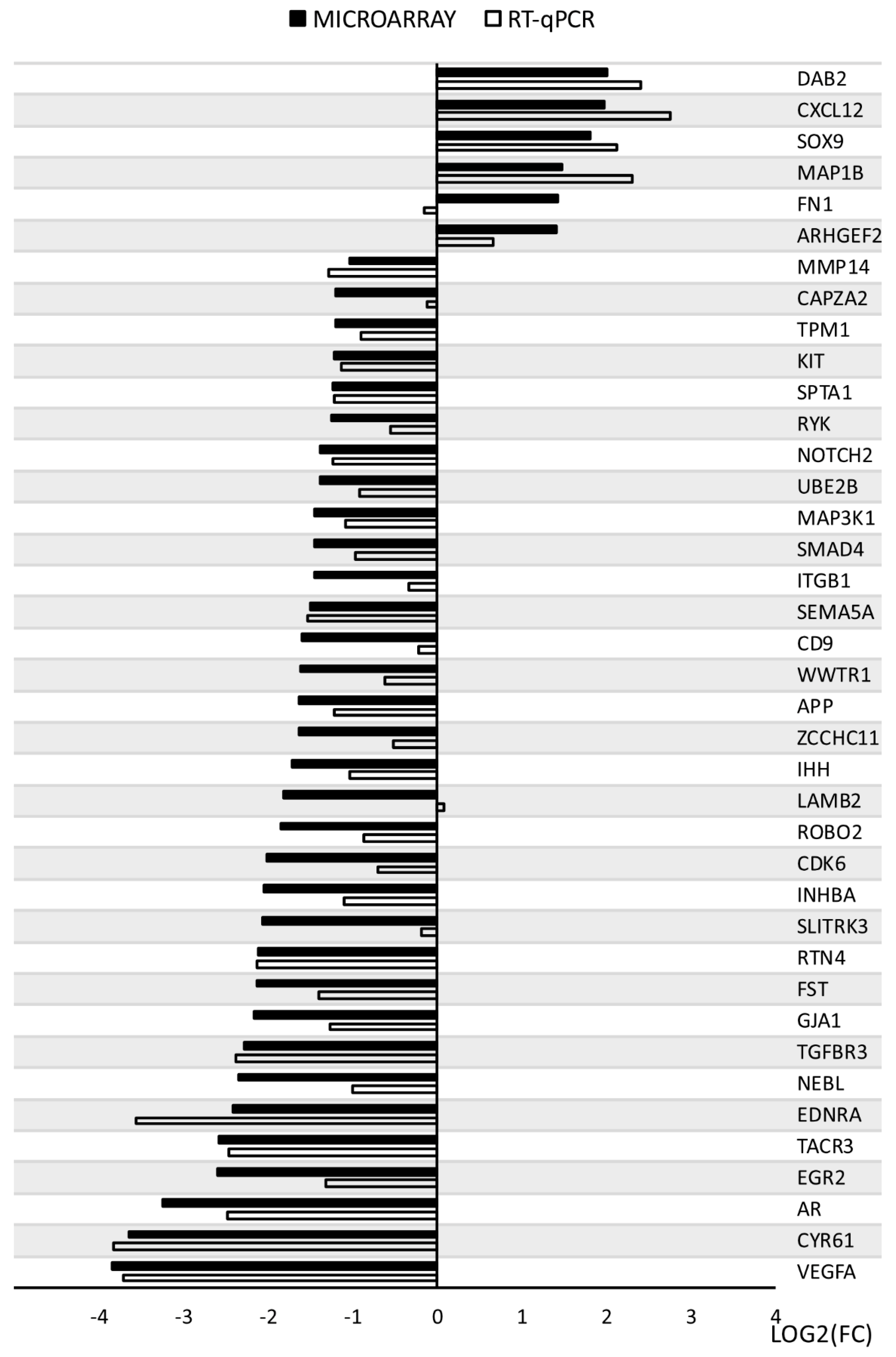
3.5. Microarray Validation—RT-qPCR
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| APP | Amyloid beta precursor protein gene |
| ARHGEF2 | Rho/Rac guanine nucleotide exchange factor 2 gene |
| BCB | The Brilliant Cresyl Blue staining |
| BFA | Brefeldin A—beta-lactam antibiotic |
| BFS | Bovine fetal serum |
| CAPZA2 | Capping actin protein of muscle Z-line subunit Alpha 2 gene |
| CCs | Cumulus cells |
| CDK6 | Cyclin dependent kinase 6 gene |
| CGs | Cortical granules |
| CTNNA2 | Catenin Alpha 2 gene |
| CXCL12 | C-X-C motif chemokine ligand 12 gene |
| CYR61 | Cellular communication network factor 1 gene |
| DAB2 | DAB adaptor protein 2 gene |
| DAVID | Database for Annotation, Visualization and Integrated Discovery |
| EDNRA | Endothelin receptor type A gene |
| EGR2 | Early growth response 2 gene |
| FI | Functional interaction |
| FITC | fluorescein isothiocyanate |
| FN1 | Fibronectin gene |
| Folr4 | Folate receptor 4, Juno |
| FST | Follistatin gene |
| GCs | Granulosa cells |
| GJA1 | Gap junction protein Alpha 1 |
| GJCs | Gap junctions connections |
| GO BP | Gene Ontology Biological Process term |
| GV | Germinal vesicle stage |
| H&E | Haematoxylin and eosin staining |
| hCG | Human chorionic gonadotropin |
| Icdg | Index of cortical granules distribution |
| IHH | Indian hedgehog signalling molecule gene |
| INHBA | Inhibit subunit beta A gene |
| ITGB1 | Integrin subunit beta 1 gene |
| IVF | In vitro fertilization |
| IVM | In vitro maturation |
| IVP | In vitro production |
| KEGG | Kyoto Encyclopedia of Genes and Genomes |
| KIT | Tyrosine kinase receptor gene |
| LAMB2 | Laminin subunit beta 2 gene |
| MAP1B | Microtubule associated protein 1B |
| MAP3K1 | Mitogen-activated protein kinase kinase kinase 1 gene |
| MF | Medium follicles |
| MMP14 | Matrix metalloproteinase-14 gene |
| NEBL | Nebulette gene |
| NOTCH2 | Notch receptor 2 gene |
| OCs | Oocytes |
| OECs | Oviduct epithelial cells |
| Rab27a | Ras superfamily of GTPases |
| RMA | Robust Multiarray Averaging algorithm |
| ROBO2 | Roundabout guidance receptor 2 gene |
| RTN4 | Reticulon 4 gene |
| RYK | Receptor like tyrosine kinase gene |
| SDF-1 | Stromal cell-derived factor gene |
| SEMA5A | Semaphorin 5A gene |
| SF | Small follicles |
| SLITRK3 | SLIT and NTRK like family member 3 gene |
| SMAD4 | Mothers against decapentaplegic homolog 4 gene |
| SOX9 | SRY-box transcription factor 9 gene |
| SPTA1 | Spectrin Alpha, erythrocytic 1 gene |
| TGFBR3 | Transforming growth factor beta receptor 3 gene |
| TPM1 | Tropomyosin 1 gene |
| TUT4 | Terminal uridylyl transferase 4 gene |
| UBE2B | Ubiquitin conjugating enzyme E2 B gene |
| VEGFA | Vascular endothelial growth factor A gene |
| WWTR1 | WW domain containing transcription regulator 1 gene |
Appendix A
| Gene | Gene ID | Primer Sequence (5’–3’) | Product Size (bp) |
|---|---|---|---|
| VEGFA | 397157 | GCGGATCAAACCTCACCAAG CGTCTGCGGATCTTGTACAA | 233 |
| CYR61 | 100153791 | CTAGATGCTGCTCGGGTTTC AGCTCCAAAATGAAGCAGGA | 246 |
| AR | 397582 | GGCAAAAGCAACGAAGAGAC CGACTCGGATAGGCTGCTAC | 188 |
| EGR2 | 100038004 | ACCCAGAAGGCATCATCAAC GAGGGGTCCTGGTAGAGGTC | 232 |
| TACR3 | 397457 | TCATTTATGCGCTTCACAGC GTCTGGGTTTCAAGGGATCA | 156 |
| EDNRA | 100522395 | GGCCCTTTGAGAACCATGAC GCTGTGACCAATGGAATCCC | 223 |
| NEBL | 397512 | CAAACCCTTCAAGGCTACCA CTGAGAACACGCTTCCATCA | 177 |
| TGFBR3 | 100518636 | TTTGTTTTAGCTGGGGGTTG TGGCCACAGGGATTTTTAAG | 177 |
| GJA1 | 445002 | ATGAGCAGTCTGCCTTTCGT TTGGGCAACTTTGAGTTCCT | 205 |
| FST | 100525337 | CTGAGCACCTCCGACGAG ACGTTTCTTTACATGGGATGC | 100 |
| RTN4 | 100170118 | TGGTGCCTTGTTCAATGGTC TTTTCGCTTCAATCCAGGGA | 181 |
| SLITRK3 | 106504067 | CGTATTGAGAGTGGGGCATT TCTGCCAATGTGGTCTAGCA | 183 |
| INHBA | 397093 | AAGCTTCATGTGGGCAAAGT TCACTGCCCTCTTTGGAGAT | 214 |
| CDK6 | 100518921 | GGTACAGAGCGCCAGAAGTC GAGGCCAGTCCTCTTCTCCT | 170 |
| ROBO2 | 101101688 | TCGTGGTGGAAAACTGATGA CCTGGACCTGACAACGAAAT | 157 |
| LAMB2 | 100517681 | ATTGAGCCATCCACTGAAGG GTTGTCCCCCAGTGTGTGTA | 153 |
| IHH | 397174 | CTCCACTGCCCTCTCAGAAC AGCTCGCAGCTGTGTCACTA | 182 |
| ZCCHC11 | 100516979 | TGACACCTGAGCAGAGGCTA ATGTGCTCCCTGGATGTTTT | 167 |
| APP | 397663 | CAAGGAAGGCATCTTGCAGT CTAAGCAGCGGTACGGAATC | 151 |
| WWTR1 | 100522573 | GCCACTGGCCAGAGATACTT TCTGCTGCTGGTGATGATTC | 160 |
| CD9 | 397067 | GGCTCCGATTTGACTCTCAG CACCTGATCCTTGTGGGAAT | 154 |
| SEMA5A | 100737194 | CCTGGTTACGGGAGTTCAGA TTCATCACACTCCCACTCCA | 176 |
| ITGB1 | 397019 | AAATTCAAGCGAGCTGAGGA TCCACAAAAGAGCCAAATCC | 154 |
| SMAD4 | 397142 | CACAAGTCAGCCTGCCAGTA ATAGGTGGGTGGTGCTGAAG | 228 |
| MAP3K1 | 396617 | AGCCCAGACAACAAATACCG TGCGTGAAACAAACTTCTGG | 161 |
| UBE2B | 100513527 | TGGCATCTCCACTATGAGCA ACCTGCCTTGTTCAACCAAC | 164 |
| NOTCH2 | 100153369 | CCAGGTTTCGAAGGCATCAC AGCGGCAGTTGTAAGTGTTG | 160 |
| RYK | 100523513 | ATTTCTGCTCAGGGGGAAGT GCTGCATGTACGGGATCATA | 185 |
| SPTA1 | 100152068 | TGCTGACATCTTGGAGTGGA TCACTCCATTCACTCGTCCC | 153 |
| KIT | 396810 | ACCATCCATCCATCCAGCAA TGCTTTCTCCACGATCCACT | 228 |
| TPM1 | 100037999 | CGCAAGTATGAAGAGGTGGC CTCTTCTTCAAGCTCGGCAC | 201 |
| CAPZA2 | 100037958 | ATGGTCGTTGGAGGTCAGAG CTTTTGCTGTTTGCACCTCA | 175 |
| MMP14 | 397471 | TCCAGAACTACACCCCCAAG GCTGTCACCATGGAAACCTT | 185 |
| ARHGEF2 | 100145887 | CCACAACCGCTGTAAAGACA AAGCTGTCGGAGGGGTAGAT | 205 |
| FN1 | 397620 | TGAGCCTGAAGAGACCTGCT CAGCTCCAATGCAGGTACAG | 113 |
| MAP1B | 100519062 | TGGGACACAAACCTGATTGA TCTGAGGGGTTGATCAGGAC | 224 |
| SOX9 | 396840 | GACTCTGGGCAAACTCTGGA AAGATGGCGTTGGGAGAGAT | 183 |
| CXCL12 | 494460 | CCTTGCCGATTCTTTGAGAG AGAGAGTGGGACTGGGTTTG | 214 |
| DAB2 | 100519746 | CCGTGATGTGACAGACAACC CTGCTTTGTTGGCTTCTTCC | 170 |
References
- Rybska, M.; Knap, S.; Jankowski, M.; Jeseta, M.; Bukowska, D.; Antosik, P.; Nowicki, M.; Zabel, M.; Kempisty, B.; Jaśkowski, J.M. Cytoplasmic and nuclear maturation of oocytes in mammals—Living in the shadow of cells developmental capability. Med. J. Cell Biol. 2018, 6, 13–17. [Google Scholar] [CrossRef]
- Bianchi, E.; Doe, B.; Goulding, D.; Wright, G.J. Juno is the egg Izumo receptor and is essential for mammalian fertilization. Nature 2014, 508, 483–487. [Google Scholar] [CrossRef] [PubMed]
- Burkart, A.D.; Xiong, B.; Baibakov, B.; Jiménez-Movilla, M.; Dean, J. Ovastacin, a cortical granule protease, cleaves ZP2 in the zona pellucida to prevent polyspermy. J. Cell Biol. 2012, 197, 37–44. [Google Scholar] [CrossRef] [PubMed]
- Cheeseman, L.P.; Boulanger, J.; Bond, L.M.; Schuh, M. Two pathways regulate cortical granule translocation to prevent polyspermy in mouse oocytes. Nat. Commun. 2016, 7. [Google Scholar] [CrossRef]
- Wessel, G.M.; Conner, S.D.; Berg, L. Cortical granule translocation is microfilament mediated and linked to meiotic maturation in the sea urchin oocyte. Development 2002, 126, 567–576. [Google Scholar]
- Connors, S.A.; Kanatsu-Shinohara, M.; Schultz, R.M.; Kopf, G.S. Involvement of the Cytoskeleton in the Movement of Cortical Granules during Oocyte Maturation, and Cortical Granule Anchoring in Mouse Eggs. Dev. Biol. 1998, 200, 103–115. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.H.; Cui, Q.; Zhang, T.; Wang, Z.B.; Ouyang, Y.C.; Shen, W.; Ma, J.Y.; Schatten, H.; Sun, Q.Y. Rab3A, Rab27A, and Rab35 regulate different events during mouse oocyte meiotic maturation and activation. Histochem. Cell Biol. 2016, 145, 647–657. [Google Scholar] [CrossRef]
- de Paola, M.; Bello, O.D.; Michaut, M.A. Cortical Granule Exocytosis Is Mediated by α-SNAP and N-Ethilmaleimide Sensitive Factor in Mouse Oocytes. PLoS ONE 2015, 10. [Google Scholar] [CrossRef]
- Kimura, K.; Kimura, A. Rab6 is required for the exocytosis of cortical granules and the recruitment of separase to the granules during the oocyte-to-embryo transition in Caenorhabditis elegans. J. Cell Sci. 2012, 125, 5897–5905. [Google Scholar] [CrossRef]
- Hulinska, P.; Martecikova, S.; Jeseta, M.; Machatkova, M. Efficiency of in vitro fertilization is influenced by the meiotic competence of porcine oocytes and time of their maturation. Anim. Reprod. Sci. 2011, 124, 112–117. [Google Scholar] [CrossRef]
- Schoevers, E.J.; Colenbrander, B.; Roelen, B.A.J. Developmental stage of the oocyte during antral follicle growth and cumulus investment determines in vitro embryo development of sow oocytes. Theriogenology 2007, 67, 1108–1122. [Google Scholar] [CrossRef] [PubMed]
- Machatkova, M.; Horakova, J.; Hulinska, P.; Reckova, Z.; Hanzalova, K. Early oocyte penetration can predict the efficiency of bovine embryo production in vitro. Zygote 2008, 16, 203–209. [Google Scholar] [CrossRef] [PubMed]
- Marchal, R.; Feugang, J.M.; Perreau, C.; Venturi, E.; Terqui, M.; Mermillod, P. Meiotic and developmental competence of prepubertal and adult swine oocytes. Theriogenology 2001, 56, 17–29. [Google Scholar] [CrossRef]
- Bagg, M.A.; Nottle, M.B.; Armstrong, D.T.; Grupen, C.G. Relationship between follicle size and oocyte developmental competence in prepubertal and adult pigs. Reprod. Fertil. Dev. 2007, 19, 797–803. [Google Scholar] [CrossRef] [PubMed]
- Sun, Q.Y.; Lai, L.; Bonk, A.; Prather, R.S.; Schatten, H. Cytoplasmic changes in relation to nuclear maturation and early embryo developmental potential of porcine oocytes: Effects of gonadotropins, cumulus cells, follicular size, and protein synthesis inhibition. Mol. Reprod. Dev. 2001, 59, 192–198. [Google Scholar] [CrossRef] [PubMed]
- Funahashi, H. Polyspermic penetration in porcine IVM-IVF systems. Reprod. Fertil. Dev. 2003, 15, 167–177. [Google Scholar] [CrossRef]
- Liu, M. The biology and dynamics of mammalian cortical granules. Reprod. Biol. Endocrinol. 2011, 9, 149. [Google Scholar] [CrossRef]
- Zhou, C.; Zhang, X.; ShiYang, X.; Wang, H.; Xiong, B. Tea polyphenol protects against cisplatin-induced meiotic defects in porcine oocytes. Aging (Albany N. Y.) 2019, 11, 4706–4719. [Google Scholar] [CrossRef]
- Gardner, A.J.; Knott, J.G.; Jones, K.T.; Evans, J.P. CaMKII can participate in but is not sufficient for the establishment of the membrane block to polyspermy in mouse eggs. J. Cell. Physiol. 2007, 212, 275–280. [Google Scholar] [CrossRef]
- Ducibella, T. The cortical reaction and development of activation competence in mammalian oocytes. Hum. Reprod. Update 1996, 2, 29–42. [Google Scholar] [CrossRef]
- Yoshida, M.; Cran, D.G.; Pursel, V.G. Confocal and fluorescence microscopic study using lectins of the distribution of cortical granules during the maturation and fertilization of pig oocytes. Mol. Reprod. Dev. 1993, 36, 462–468. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.-H.; Sun, Q.-Y.; Hosoe, M.; Shioya, Y.; Day, B.N. Quantified Analysis of Cortical Granule Distribution and Exocytosis of Porcine Oocytes during Meiotic Maturation and Activation1. Biol. Reprod. 1997, 56, 1376–1382. [Google Scholar] [CrossRef]
- Wiesława, K.; Maurycy, J.; Joanna, B.; Piotr, C.; Ronza, K.; Artur, B.; Sylwia, B.; Marta, D.; Michal, J.; Magdalena, M.; et al. Amino acids metabolism and degradation is regulated during porcine oviductal epithelial cells (OECs) primary culture in vitro–signaling pathway activation approach. Med. J. Cell Biol. 2018, 6. [Google Scholar] [CrossRef]
- Budna, J.; Celichowski, P.; Knap, S.; Jankowski, M.; Magas, M.; Nawrocki, M.J.; Ramlau, P.; Nowicki, A.; Rojewska, M.; Chermuła, B.; et al. Fatty Acids Related Genes Expression Undergo Substantial Changes in Porcine Oviductal Epithelial Cells During Long-Term Primary Culture. Med. J. Cell Biol. 2018, 6, 39–47. [Google Scholar] [CrossRef]
- Kulus, M.; Sujka-kordowska, P.; Konwerska, A.; Celichowski, P.; Kranc, W.; Kulus, J.; Piotrowska-kempisty, H.; Antosik, P. New Molecular Markers Involved in Regulation of Ovarian Granulosa Cell Morphogenesis, Development and Di ff erentiation during Short-Term Primary In Vitro Culture—Transcriptomic and Histochemical Study Based on Ovaries and Individual Separated Follicle. Int. J. Mol. Sci. 2019, 20, 3966. [Google Scholar] [CrossRef] [PubMed]
- Stefańska, K.; Kocherova, I.; Knap, S.; Kulus, M.; Celichowski, P.; Jeseta, M. The genes regulating maintenance of cellular protein location are differentially expressed in porcine epithelial oviductal cells during longterm in vitro cultivation. Med. J. Cell Biol. 2019, 7, 77–85. [Google Scholar] [CrossRef]
- Chamier-Gliszczyńska, A.; Kałuzna, S.; Stefańska, K.; Celichowski, P.; Antosik, P.; Bukowska, D.; Bruska, M.; Zakova, J.; Machatkova, M.; Jeseta, M.; et al. Analysis of expression of genes responsible for regulation of cellular proliferation and migration-Microarray approach based on porcine oocyte model. Med. J. Cell Biol. 2019, 7, 48–57. [Google Scholar] [CrossRef]
- Walter, W.; Sánchez-Cabo, F.; Ricote, M. GOplot: An R package for visually combining expression data with functional analysis: Fig. 1. Bioinformatics 2015, 31, 2912–2914. [Google Scholar] [CrossRef]
- Kocherova, I.; Kulus, M.; Dompe, C.; Antosik, P.; Bukowska, D. Biochemical properties of cofactor and coenzyme metabolism in porcine oviductal epithelial cells—A microarray study. Med. J. Cell Biol. 2019, 7, 125–133. [Google Scholar] [CrossRef]
- Yanez, L.Z.; Han, J.; Behr, B.B.; Pera, R.A.R.; Camarillo, D.B. Human oocyte developmental potential is predicted by mechanical properties within hours after fertilization. Nat. Commun. 2016, 7. [Google Scholar] [CrossRef]
- Krendel, M.; Zenke, F.T.; Bokoch, G.M. Nucleotide exchange factor GEF-H1 mediates cross-talk between microtubules and the actin cytoskeleton. Nat. Cell Biol. 2002, 4, 294–301. [Google Scholar] [CrossRef]
- Aznar, S.; Fernández-Valerón, P.; Espina, C.; Lacal, J.C. Rho GTPases: Potential candidates for anticancer therapy. Cancer Lett. 2004, 206, 181–191. [Google Scholar] [CrossRef]
- Zhong, Z.-S.; Huo, L.-J.; Liang, C.-G.; Chen, D.-Y.; Sun, Q.-Y. Small GTPase RhoA is required for ooplasmic segregation and spindle rotation, but not for spindle organization and chromosome separation during mouse oocyte maturation, fertilization, and early cleavage. Mol. Reprod. Dev. 2005, 71, 256–261. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Duan, X.; Cao, R.; Liu, H.-L.; Cui, X.-S.; Kim, N.-H.; Rui, R.; Sun, S.-C. Small GTPase RhoA regulates cytoskeleton dynamics during porcine oocyte maturation and early embryo development. Cell Cycle 2014, 13, 3390–3403. [Google Scholar] [CrossRef] [PubMed]
- Tan, K.; An, L.; Wang, S.-M.; Wang, X.-D.; Zhang, Z.-N.; Miao, K.; Sui, L.-L.; He, S.-Z.; Nie, J.-Z.; Wu, Z.-H.; et al. Actin Disorganization Plays a Vital Role in Impaired Embryonic Development of In Vitro-Produced Mouse Preimplantation Embryos. PLoS ONE 2015, 10, e0130382. [Google Scholar] [CrossRef] [PubMed]
- Hoelker, M.; Salilew-Wondim, D.; Drillich, M.; Christine, G.-B.; Ghanem, N.; Goetze, L.; Tesfaye, D.; Schellander, K.; Heuwieser, W. Transcriptional response of the bovine endometrium and embryo to endometrial polymorphonuclear neutrophil infiltration as an indicator of subclinical inflammation of the uterine environment. Reprod. Fertil. Dev. 2012, 24, 778. [Google Scholar] [CrossRef] [PubMed]
- Villarroel-Campos, D.; Gonzalez-Billault, C. The MAP1B case: An old MAP that is new again. Dev. Neurobiol. 2014, 74, 953–971. [Google Scholar] [CrossRef] [PubMed]
- Gödel, M.; Temerinac, D.; Grahammer, F.; Hartleben, B.; Kretz, O.; Riederer, B.M.; Propst, F.; Kohl, S.; Huber, T.B. Microtubule Associated Protein 1b (MAP1B) Is a Marker of the Microtubular Cytoskeleton in Podocytes but Is Not Essential for the Function of the Kidney Filtration Barrier in Mice. PLoS ONE 2015, 10, e0140116. [Google Scholar] [CrossRef]
- Kim, J.-M.; Park, J.-E.; Yoo, I.; Han, J.; Kim, N.; Lim, W.-J.; Cho, E.-S.; Choi, B.; Choi, S.; Kim, T.-H.; et al. Integrated transcriptomes throughout swine oestrous cycle reveal dynamic changes in reproductive tissues interacting networks. Sci. Rep. 2018, 8, 5436. [Google Scholar] [CrossRef]
- Zhao, W.; Shahzad, K.; Jiang, M.; Graugnard, D.E.; Rodriguez-Zas, S.L.; Luo, J.; Loor, J.J.; Hurley, W.L. Bioinformatics and Gene Network Analyses of the Swine Mammary Gland Transcriptome during Late Gestation. Bioinform. Biol. Insights 2013, 7, 193–216. [Google Scholar] [CrossRef]
- Terenina, E.; Fabre, S.; Bonnet, A.; Monniaux, D.; Robert-Granié, C.; SanCristobal, M.; Sarry, J.; Vignoles, F.; Gondret, F.; Monget, P.; et al. Differentially expressed genes and gene networks involved in pig ovarian follicular atresia. Physiol. Genom. 2017, 49, 67–80. [Google Scholar] [CrossRef]
- Hatzirodos, N.; Hummitzsch, K.; Irving-Rodgers, H.F.; Rodgers, R.J. Transcriptome comparisons identify new cell markers for theca interna and granulosa cells from small and large antral ovarian follicles. PLoS ONE 2015, 10. [Google Scholar] [CrossRef]
- Budna, J.; Bryja, A.; Celichowski, P.; Kahan, R.; Kranc, W.; Ciesiółka, S.; Rybska, M.; Borys, S.; Jeseta, M.; Bukowska, D.; et al. Genes of cellular components of morphogenesis in porcine oocytes before and after IVM. Reproduction 2017, 154, 535–545. [Google Scholar] [CrossRef]
- Schuh, M. An actin-dependent mechanism for long range vesicle transport. Nat. Cell Biol. 2011, 13, 1431. [Google Scholar] [CrossRef]
- Cui, Z.; Yu, L.; Shi Yang, X.; Zhang, Y.; Shi, X.; Li, Y.; Chen, Q.; Xiong, B. Brefeldin A impairs porcine oocyte meiotic maturation via interruption of organelle dynamics. J. Cell. Physiol. 2019, 234, 20111–20117. [Google Scholar] [CrossRef] [PubMed]
- Miao, Y.; Zhou, C.; Cui, Z.; Tang, L.; ShiYang, X.; Lu, Y.; Zhang, M.; Dai, X.; Xiong, B. Dynein promotes porcine oocyte meiotic progression by maintaining cytoskeletal structures and cortical granule arrangement. Cell Cycle 2017, 16, 2139–2145. [Google Scholar] [CrossRef] [PubMed]
- Gieryng, A.; Bogunia-Kubik, K. Znaczenie interakcji między SDF-1 i CXCR4 w hematopoezie i mobilizacji macierzystych komórek hematopoetycznych do krwi obwodowej. Postepy Hig. Med. Dosw. 2007, 61, 369–383. [Google Scholar]
- Kulus, M.; Kulus, J.; Jankowski, M.; Borowiec, B.; Jeseta, M.; Bukowska, D.; Brüssow, K.P.; Kempisty, B.; Antosik, P. The use of mesenchymal stem cells in veterinary medicine. Med. J. Cell Biol. 2018, 6, 101–107. [Google Scholar] [CrossRef]
- Lorenz, M.; Mozdziak, P.; Kempisty, B.; Dyszkiewicz-konwińska, M. Application potential and plasticity of human stem cells. Med J. Cell Boil. 2019, 7, 140–145. [Google Scholar] [CrossRef]
- Zuccarello, D.; Ferlin, A.; Garolla, A.; Menegazzo, M.; Perilli, L.; Ambrosini, G.; Foresta, C. How the human spermatozoa sense the oocyte: A new role of SDF1-CXCR4 signalling. Int. J. Androl. 2011, 34, e554–e565. [Google Scholar] [CrossRef]
- Dyszkiewicz-Konwińska, M.; Bryja, A.; Jopek, K.; Budna, J.; Khozmi, R.; Jeseta, M.; Bukowska, D.; Antosik, P.; Bruska, M.; Nowicki, M.; et al. Expression of genes responsible for cell morphogenesis involved in differentiation in porcine buccal pouch mucosal cells during long-term primary culture and real-time proliferation in vitro. J. Biol. Regul. Homeost. Agents 2017, 31, 855–864. [Google Scholar]
- Kryczek, I.; Lange, A.; Mottram, P.; Alvarez, X.; Cheng, P.; Hogan, M.; Moons, L.; Wei, S.; Zou, L.; Machelon, V.; et al. CXCL12 and Vascular Endothelial Growth Factor Synergistically Induce Neoangiogenesis in Human Ovarian Cancers. Cancer Res. 2005, 65, 465–472. [Google Scholar] [PubMed]
- Nishigaki, A.; Okada, H.; Okamoto, R.; Shimoi, K.; Miyashiro, H.; Yasuda, K.; Kanzaki, H. The concentration of human follicular fluid stromal cell-derived factor-1 is correlated with luteinization in follicles. Gynecol. Endocrinol. 2013, 29, 230–234. [Google Scholar] [CrossRef] [PubMed]
- Chao, W.-T.; Kunz, J. Focal adhesion disassembly requires clathrin-dependent endocytosis of integrins. FEBS Lett. 2009, 583, 1337–1343. [Google Scholar] [CrossRef] [PubMed]
- Moncrieff, L.; Mozdziak, P.; Jeseta, M.; Machatkova, M.; Kranc, W. Ovarian follicular cells–living in the shadow of stemness cellular competence. Med J. Cell Boil. 2019, 7, 134–140. [Google Scholar] [CrossRef]
- Stefańska, K.; Chamier-Gliszczyńska, A.; Jankowski, M.; Celichowski, P.; Kulus, M.; Rojewska, M.; Antosik, P.; Bukowska, D.; Bruska, M.; Nowicki, M.; et al. Epithelium morphogenesis and oviduct development are regulated by significant increase of expression of genes after long-term in vitro primary culture—A microarray assays. Med. J. Cell Biol. 2018, 6, 195–204. [Google Scholar] [CrossRef]
- Mok, S.C.; Wong, K.-K.; Chan, R.K.W.; Lau, C.C.; Tsao, S.-W.; Knapp, R.C.; Berkowitz, R.S. Molecular Cloning of Differentially Expressed Genes in Human Epithelial Ovarian Cancer. Gynecol. Oncol. 1994, 52, 247–252. [Google Scholar] [CrossRef]
- Hocevar, B.A.; Smine, A.; Xu, X.X.; Howe, P.H. The adaptor molecule Disabled-2 links the transforming growth factor β receptors to the Smad pathway. Embo J. 2001, 20, 2789–2801. [Google Scholar] [CrossRef]
- Benito-Jardón, M.; Klapproth, S.; Gimeno-LLuch, I.; Petzold, T.; Bharadwaj, M.; Müller, D.J.; Zuchtriegel, G.; Reichel, C.A.; Costell, M. The fibronectin synergy site re-enforces cell adhesion and mediates a crosstalk between integrin classes. Elife 2017, 6. [Google Scholar] [CrossRef]
- Chermuła, B.; Brązert, M.; Iżycki, D.; Ciesiółka, S.; Kranc, W.; Celichowski, P.; Ożegowska, K.; Nawrocki, M.J.; Jankowski, M.; Jeseta, M.; et al. New Gene Markers of Angiogenesis and Blood Vessels Development in Porcine Ovarian Granulosa Cells during Short-Term Primary Culture In Vitro. Biomed Res. Int. 2019, 2019, 6545210. [Google Scholar] [CrossRef]
- Gonen, N.; Quinn, A.; O’Neill, H.C.; Koopman, P.; Lovell-Badge, R. Normal Levels of Sox9 Expression in the Developing Mouse Testis Depend on the TES/TESCO Enhancer, but This Does Not Act Alone. PLoS Genet. 2017, 13, e1006520. [Google Scholar]
- Uhlenhaut, N.H.; Jakob, S.; Anlag, K.; Eisenberger, T.; Sekido, R.; Kress, J.; Treier, A.-C.; Klugmann, C.; Klasen, C.; Holter, N.I.; et al. Somatic Sex Reprogramming of Adult Ovaries to Testes by FOXL2 Ablation. Cell 2009, 139, 1130–1142. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Dong, S. The Signaling Pathways Involved in Chondrocyte Differentiation and Hypertrophic Differentiation. Stem Cells Int. 2016, 2016, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Montero, J.A.; Lorda-Diez, C.I.; Francisco-Morcillo, J.; Chimal-Monroy, J.; Garcia-Porrero, J.A.; Hurle, J.M. Sox9 Expression in Amniotes: Species-Specific Differences in the Formation of Digits. Front. Cell Dev. Biol. 2017, 5, 23. [Google Scholar] [CrossRef] [PubMed]
- Di Giancamillo, A.; Deponti, D.; Modina, S.; Tessaro, I.; Domeneghini, C.; Peretti, G.M. Age-related modulation of angiogenesis-regulating factors in the swine meniscus. J. Cell. Mol. Med. 2017, 21, 3066–3075. [Google Scholar] [CrossRef]
- Kempisty, B.; Ziółkowska, A.; Ciesiółka, S.; Piotrowska, H.; Antosik, P.; Bukowska, D.; Nowicki, M.; Brüssow, K.P.; Zabel, M. Study on connexin gene and protein expression and cellular distribution in relation to real-time proliferation of porcine granulosa cells. J. Biol. Regul. Homeost. Agents 2014, 28, 625–635. [Google Scholar]
- Rybska, M.; Knap, S.; Jankowski, M.; Jeseta, M.; Bukowska, D.; Antosik, P.; Nowicki, M.; Zabel, M.; Kempisty, B.; Jaśkowski, J.M. Characteristic of factors influencing the proper course of folliculogenesis in mammals. Med. J. Cell Biol. 2018, 6, 33–38. [Google Scholar] [CrossRef]
- Kempisty, B.; Ziółkowska, A.; Piotrowska, H.; Ciesiółka, S.; Antosik, P.; Bukowska, D.; Zawierucha, P.; Woźna, M.; Jaśkowski, J.M.; Brüssow, K.P.; et al. Short-term cultivation of porcine cumulus cells influences the cyclin-dependent kinase 4 (Cdk4) and connexin 43 (Cx43) protein expression--a real-time cell proliferation approach. J. Reprod. Dev. 2013, 59, 339–345. [Google Scholar] [CrossRef]
- Simon, A.M.; Goodenough, D.A.; Li, E.; Paul, D.L. Female infertility in mice lacking connexin 37. Nature 1997, 385, 525–529. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.X.; Tong, D.; El-Gehani, F.; Tekpetey, F.R.; Kidder, G.M. Connexin expression and gap junctional coupling in human cumulus cells: Contribution to embryo quality. J. Cell. Mol. Med. 2009, 13, 972–984. [Google Scholar] [CrossRef] [PubMed]
- Li, S.-H.; Lin, M.-H.; Hwu, Y.-M.; Lu, C.-H.; Yeh, L.-Y.; Chen, Y.-J.; Lee, R.K.-K. Correlation of cumulus gene expression of GJA1, PRSS35, PTX3, and SERPINE2 with oocyte maturation, fertilization, and embryo development. Reprod. Biol. Endocrinol. 2015, 13, 93. [Google Scholar] [CrossRef] [PubMed]
- Shao, L.; Chian, R.-C.; Xu, Y.; Yan, Z.; Zhang, Y.; Gao, C.; Gao, L.; Liu, J.; Cui, Y. Genomic expression profiles in cumulus cells derived from germinal vesicle and MII mouse oocytes. Reprod. Fertil. Dev. 2016, 28, 1798. [Google Scholar] [CrossRef]
- Cao, R.; Wu, W.; Zhou, X.; Liu, K.; Li, B.; Huang, X.; Zhang, Y.; Liu, H. Let-7g induces granulosa cell apoptosis by targeting MAP3K1 in the porcine ovary. Int. J. Biochem. Cell Biol. 2015, 68, 148–157. [Google Scholar] [CrossRef] [PubMed]
- Fan, H.-Y.; Liu, Z.; Shimada, M.; Sterneck, E.; Johnson, P.F.; Hedrick, S.M.; Richards, J.S. MAPK3/1 (ERK1/2) in Ovarian Granulosa Cells Are Essential for Female Fertility. Science 2009, 324, 938–941. [Google Scholar] [CrossRef] [PubMed]
- Borys-Wójcik, S.; Kocherova, I.; Celichowski, P.; Popis, M.; Jeseta, M.; Bukowska, D.; Antosik, P.; Nowicki, M.; Kempisty, B. Protein oligomerization is the biochemical process highly up-regulated in porcine oocytes before in vitro maturation (IVM). Med. J. Cell Biol. 2018, 6, 155–162. [Google Scholar] [CrossRef]
- Kranc, W.; Budna, J.; Chachuła, A.; Borys, S.; Bryja, A.; Rybska, M.; Ciesiółka, S.; Sumelka, E.; Jeseta, M.; Brüssow, K.P.; et al. Cell Migration Is the Ontology Group Differentially Expressed in Porcine Oocytes before and after in Vitro Maturation: A Microarray Approach. DNA Cell Biol. 2017, 36, 273–282. [Google Scholar] [CrossRef]
- Lee, R.; Lee, W.-Y.; Park, H.-J.; Ha, W.-T.; Woo, J.-S.; Chung, H.-J.; Lee, J.-H.; Hong, K.; Song, H. Stage-specific expression of DDX4 and c-kit at different developmental stages of the porcine testis. Anim. Reprod. Sci. 2018, 190, 18–26. [Google Scholar] [CrossRef]
- Saatcioglu, H.D.; Cuevas, I.; Castrillon, D.H. Control of Oocyte Reawakening by Kit. PLoS Genet. 2016, 12, e1006215. [Google Scholar] [CrossRef]
- Moniruzzaman, M.; Miyano, T. KIT-KIT Ligand in the Growth of Porcine Oocytes in Primordial Follicles. J. Reprod. Dev. 2007, 53, 1273–1281. [Google Scholar] [CrossRef][Green Version]
- Sternlicht, M.; Werb, Z. How Matrix metalloproteinases regulate cell behavior. Annu. Rev. Cell. Biol. 2009, 463–516. [Google Scholar] [CrossRef]
- Hägglund, A.C.; Ny, A.; Leonardsson, G.; Ny, T. Regulation and localization of matrix metalloproteinases and tissue inhibitors of metalloproteinases in the mouse ovary during gonadotropin-induced ovulation. Endocrinology 1999, 140, 4351–4358. [Google Scholar] [CrossRef] [PubMed]
- Puttabyatappa, M.; Jacot, T.A.; Al-Alem, L.F.; Rosewell, K.L.; Duffy, D.M.; Brännström, M.; Curry, T.E. Ovarian Membrane-Type Matrix Metalloproteinases: Induction of MMP14 and MMP16 During the Periovulatory Period in the Rat, Macaque, and Human1. Biol. Reprod. 2014, 91. [Google Scholar] [CrossRef]
- Bakke, L.J.; Dow, M.P.; Cassar, C.A.; Peters, M.W.; Pursley, J.R.; Smith, G.W. Effect of the preovulatory gonadotropin surge on matrix metalloproteinase (MMP)-14, MMP-2, and tissue inhibitor of metalloproteinases-2 expression within bovine periovulatory follicular and luteal tissue. Biol. Reprod. 2002, 66, 1627–1634. [Google Scholar] [CrossRef] [PubMed]
- Kempisty, B.; Piotrowska, H.; Rybska, M.; Woźna, M.; Antosik, P.; Bukowska, D.; Zawierucha, P.; Ciesiółka, S.; Jaśkowski, J.M.; Nowicki, M.; et al. Expression of INHβA and INHβB proteins in porcine oocytes cultured in vitro is dependent on the follicle size. Zygote 2015, 23, 205–211. [Google Scholar] [CrossRef] [PubMed]
- Matzuk, M.M.; Brown, C.W.; Houston-Hawkins, D.E.; Woodruff, T.K. Insertion of Inhbb into the Inhba locus rescues the Inhba-null phenotype and reveals new activin functions. Nat. Genet. 2000, 25, 453–457. [Google Scholar]
- Richani, D.; Constance, K.; Lien, S.; Agapiou, D.; Stocker, W.A.; Hedger, M.P.; Ledger, W.L.; Thompson, J.G.; Robertson, D.M.; Mottershead, D.G.; et al. Cumulin and FSH Cooperate to Regulate Inhibin B and Activin B Production by Human Granulosa-Lutein Cells In Vitro. Endocrinology 2019, 160, 853–862. [Google Scholar] [CrossRef]
- Kempisty, B.; Jackowska, M.; Woźna, M.; Antosik, P.; Piotrowska, H.; Zawierucha, P.; Bukowska, D.; Jaśkowski, J.M.; Nowicki, M.; Brüssow, K.P. Expression and cellular distribution of INHA and INHB before and after in vitro cultivation of porcine oocytes isolated from follicles of different size. J. Biomed. Biotechnol. 2012, 2012, 742829. [Google Scholar] [CrossRef]
- Ohnuma, K.; Kaneko, H.; Noguchi, J.; Kikuchi, K.; Ozawa, M.; Hasegawa, Y. Production of inhibin A and inhibin B in boars: Changes in testicular and circulating levels of dimeric inhibins and characterization of inhibin forms during testis growth. Domest. Anim. Endocrinol. 2007, 33, 410–421. [Google Scholar] [CrossRef]
- Nguyen, T.; Lee, S.; Hatzirodos, N.; Hummitzsch, K.; Sullivan, T.R.; Rodgers, R.J.; Irving-Rodgers, H.F. Spatial differences within the membrana granulosa in the expression of focimatrix and steroidogenic capacity. Mol. Cell. Endocrinol. 2012, 363, 62–73. [Google Scholar] [CrossRef]
- Du, X.; Pan, Z.; Li, Q.; Liu, H.; Li, Q. SMAD4 feedback regulates the canonical TGF-β signaling pathway to control granulosa cell apoptosis. Cell Death Dis. 2018, 9, 151. [Google Scholar] [CrossRef]
- Li, Q.; Du, X.; Pan, Z.; Zhang, L.; Li, Q. The transcription factor SMAD4 and miR-10b contribute to E2 release and cell apoptosis in ovarian granulosa cells by targeting CYP19A1. Mol. Cell. Endocrinol. 2018, 476, 84–95. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Chen, X.; Li, X.; Wang, L.; Zhang, H.; He, Y.; Wang, J.; Zhao, Y.; Zhang, B.; Xu, Y. Interference RNA-based silencing of endogenous SMAD4 in porcine granulosa cells resulted in decreased FSH-mediated granulosa cells proliferation and steroidogenesis. Reproduction 2011, 141, 643–651. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, H.; Zhao, X.; Das, S.K.; Hogan, B.L.M.; Dey, S.K. Indian Hedgehog as a Progesterone-Responsive Factor Mediating Epithelial–Mesenchymal Interactions in the Mouse Uterus. Dev. Biol. 2002, 245, 280–290. [Google Scholar] [CrossRef] [PubMed]
- Goossens, K.; Van Soom, A.; Van Zeveren, A.; Favoreel, H.; Peelman, L.J. Quantification of Fibronectin 1 (FN1) splice variants, including two novel ones, and analysis of integrins as candidate FN1 receptors in bovine preimplantation embryos. Bmc Dev. Biol. 2009, 9, 1. [Google Scholar] [CrossRef]
- Jankovicova, J.; Secova, P.; Manaskova-Postlerova, P.; Simonik, O.; Frolikova, M.; Chmelikova, E.; Horovska, L.; Michalkova, K.; Dvorakova-Hortova, K.; Antalikova, J. Detection of CD9 and CD81 tetraspanins in bovine and porcine oocytes and embryos. Int. J. Biol. Macromol. 2019, 123, 931–938. [Google Scholar] [CrossRef]
- Runge, K.E.; Evans, J.E.; He, Z.-Y.; Gupta, S.; McDonald, K.L.; Stahlberg, H.; Primakoff, P.; Myles, D.G. Oocyte CD9 is enriched on the microvillar membrane and required for normal microvillar shape and distribution. Dev. Biol. 2007, 304, 317–325. [Google Scholar] [CrossRef]
- Schneyer, A.; Sidis, Y.; Xia, Y.; Saito, S.; Del Re, E.; Lin, H.Y.; Keutmann, H. Differential actions of follistatin and follistatin-like 3. Mol. Cell. Endocrinol. 2004, 225, 25–28. [Google Scholar]
- Tortoriello, D.V.; Sidis, Y.; Holtzman, D.A.; Holmes, W.E.; Schneyer, A.L. Human follistatin-related protein: A structural homologue of follistatin with nuclear localization. Endocrinology 2001, 142, 3426–3434. [Google Scholar] [CrossRef]
- Yao, H.H.C.; Matzuk, M.M.; Jorgez, C.J.; Menke, D.B.; Page, D.C.; Swain, A.; Capel, B. Follistatin operates downstream ofWnt4 in mammalian ovary organogenesis. Dev. Dyn. 2004, 230, 210–215. [Google Scholar] [CrossRef]
- Wang, Y.; Ge, W. Developmental profiles of activin betaA, betaB, and follistatin expression in the zebrafish ovary: Evidence for their differential roles during sexual maturation and ovulatory cycle. Biol. Reprod. 2004, 71, 2056–2064. [Google Scholar] [CrossRef]
- Zand-Vakili, M.; Golkar-Narenji, A.; Mozdziak, P.E.; Eimani, H. An in vitro study on oocyte and follicles of transplanted ovaries treated with vascular endothelial growth factor. J. Turk. Ger. Gynecol. Assoc. 2017, 18, 167–173. [Google Scholar] [CrossRef] [PubMed]
- Hanna, M.; Liu, H.; Amir, J.; Sun, Y.; Morris, S.W.; Siddiqui, M.A.Q.; Lau, L.F.; Chaqour, B. Mechanical regulation of the proangiogenic factor CCN1/CYR61 gene requires the combined activities of MRTF-A and CREB-binding protein histone acetyltransferase. J. Biol. Chem. 2009, 284, 23125–23136. [Google Scholar] [CrossRef] [PubMed]
- Lau, L.F. CCN1/CYR61: The very model of a modern matricellular protein. Cell. Mol. Life Sci. 2011, 68, 3149–3163. [Google Scholar] [CrossRef] [PubMed]
- Bigham, A.W.; Julian, C.G.; Wilson, M.J.; Vargas, E.; Browne, V.A.; Shriver, M.D.; Moore, L.G. Maternal PRKAA1 and EDNRA genotypes are associated with birth weight, and PRKAA1 with uterine artery diameter and metabolic homeostasis at high altitude. Physiol. Genom. 2014, 46, 687–697. [Google Scholar] [CrossRef] [PubMed]
- DeLaughter, D.M.; Clark, C.R.; Christodoulou, D.C.; Seidman, C.E.; Baldwin, H.S.; Seidman, J.G.; Barnett, J.V. Transcriptional Profiling of Cultured, Embryonic Epicardial Cells Identifies Novel Genes and Signaling Pathways Regulated by TGFβR3 In Vitro. PLoS ONE 2016, 11, e0159710. [Google Scholar] [CrossRef]
- Sevilla, T.; Sivera, R.; Martínez-Rubio, D.; Lupo, V.; Chumillas, M.J.; Calpena, E.; Dopazo, J.; Vílchez, J.J.; Palau, F.; Espinós, C. The EGR2 gene is involved in axonal Charcot−Marie−Tooth disease. Eur. J. Neurol. 2015, 22, 1548–1555. [Google Scholar] [CrossRef]
- Vite, A.; Li, J.; Radice, G.L. New functions for α-catenins in health and disease: From cancer to heart regeneration. Cell Tissue Res. 2015, 360, 773–783. [Google Scholar] [CrossRef]
- Watari, A.; Yutsudo, M. Multi-functional gene ASY/Nogo/RTN-X/RTN4: Apoptosis, tumor suppression, and inhibition of neuronal regeneration. Apoptosis 2003, 8, 5–9. [Google Scholar] [CrossRef]
- Wang, C.-J.; Zhang, Z.-Z.; Xu, J.; Wang, M.; Zhao, W.-Y.; Tu, L.; Zhuang, C.; Liu, Q.; Shen, Y.-Y.; Cao, H.; et al. SLITRK3 expression correlation to gastrointestinal stromal tumor risk rating and prognosis. World. J. Gastroenterol. 2015, 21, 8398. [Google Scholar] [CrossRef]
- Kidd, T.; Brose, K.; Mitchell, K.J.; Fetter, R.D.; Tessier-Lavigne, M.; Goodman, C.S.; Tear, G. Roundabout Controls Axon Crossing of the CNS Midline and Defines a Novel Subfamily of Evolutionarily Conserved Guidance Receptors. Cell 1998, 92, 205–215. [Google Scholar] [CrossRef]
- Wu, Y.; Zhang, S.; Xu, Q.; Zou, H.; Zhou, W.; Cai, F.; Li, T.; Song, W. Regulation of global gene expression and cell proliferation by APP. Sci. Rep. 2016, 6, 22460. [Google Scholar] [CrossRef]
- Purohit, A.; Sadanandam, A.; Myneni, P.; Singh, R.K. Semaphorin 5A mediated cellular navigation: Connecting nervous system and cancer. Biochim. Biophys. Acta Rev. Cancer 2014, 1846, 485–493. [Google Scholar] [CrossRef] [PubMed]
- Onishi, K.; Hollis, E.; Zou, Y. Axon guidance and injury—Lessons from Wnts and Wnt signaling. Curr. Opin. Neurobiol. 2014, 27, 232–240. [Google Scholar] [CrossRef] [PubMed]
| Follicle Size | Oocytes | |
|---|---|---|
| No. of Oocytes Examined | Index of Cortical Granules Distribution Icgd * (Mean ± SD) | |
| Small | 134 | 1.48 a ± 0.31 |
| Medium | 51 | 2.22 b ± 0.49 |
| Gene | Gene ID | Fold Change | p Value |
|---|---|---|---|
| VEGFA | 397157 | 0.069689389 | 0.001912689 |
| CYR61 | 100153791 | 0.080657036 | 0.0000754 |
| AR | 397582 | 0.1059863 | 0.000138367 |
| EGR2 | 100038004 | 0.165503832 | 0.007949861 |
| EDNRA | 397457 | 0.166939028 | 0.00185422 |
| NEBL | 100522395 | 0.187213468 | 0.005937926 |
| TGFBR3 | 397512 | 0.196522244 | 0.000405979 |
| GJA1 | 100518636 | 0.206907347 | 0.000107676 |
| FST | 445002 | 0.224696558 | 0.000364693 |
| CTNNA2 | 100525337 | 0.229925246 | 0.000512181 |
| RTN4 | 100170118 | 0.23137773 | 0.027495815 |
| SLITRK3 | 106504067 | 0.239141911 | 0.004260951 |
| INHBA | 397093 | 0.241259771 | 0.000148036 |
| CDK6 | 100518921 | 0.248001413 | 0.006042481 |
| LAMB2 | 101101688 | 0.278857947 | 0.000187911 |
| ROBO2 | 100517681 | 0.283748864 | 0.001183495 |
| IHH | 397174 | 0.304995843 | 0.000551261 |
| ZCCHC11 | 100516979 | 0.3216223 | 0.019809962 |
| APP | 397663 | 0.324138605 | 0.005602323 |
| WWTR1 | 100522573 | 0.327202092 | 0.000254025 |
| CD9 | 397067 | 0.329283105 | 0.006332387 |
| SEMA5A | 100737194 | 0.353391715 | 0.001092396 |
| ITGB1 | 397019 | 0.366233017 | 0.003705215 |
| SMAD4 | 397142 | 0.367802201 | 0.001238681 |
| MAP3K1 | 396617 | 0.36876538 | 0.024748462 |
| UBE2B | 100513527 | 0.382779667 | 0.041104659 |
| NOTCH2 | 100153369 | 0.3848262 | 0.002523723 |
| RYK | 100523513 | 0.421903006 | 0.00439989 |
| SPTA1 | 100152068 | 0.427042121 | 0.002476662 |
| KIT | 396810 | 0.430444215 | 0.00255635 |
| TPM1 | 100037999 | 0.433963109 | 0.001632742 |
| CAPZA2 | 100037958 | 0.434375309 | 0.017755958 |
| MMP14 | 397471 | 0.488721147 | 0.038060423 |
| ARHGEF2 | 100145887 | 2.656199858 | 0.006431804 |
| FN1 | 397620 | 2.679580656 | 0.001210303 |
| MAP1B | 100519062 | 2.788554105 | 0.00105385 |
| SOX9 | 396840 | 3.503340174 | 0.000620008 |
| CXCL12 | 494460 | 3.934705791 | 0.003163632 |
| DAB2 | 100519746 | 4.008208178 | 0.001912689 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kulus, M.; Kranc, W.; Jeseta, M.; Sujka-Kordowska, P.; Konwerska, A.; Ciesiółka, S.; Celichowski, P.; Moncrieff, L.; Kocherova, I.; Józkowiak, M.; et al. Cortical Granule Distribution and Expression Pattern of Genes Regulating Cellular Component Size, Morphogenesis, and Potential to Differentiation are Related to Oocyte Developmental Competence and Maturational Capacity In Vivo and In Vitro. Genes 2020, 11, 815. https://doi.org/10.3390/genes11070815
Kulus M, Kranc W, Jeseta M, Sujka-Kordowska P, Konwerska A, Ciesiółka S, Celichowski P, Moncrieff L, Kocherova I, Józkowiak M, et al. Cortical Granule Distribution and Expression Pattern of Genes Regulating Cellular Component Size, Morphogenesis, and Potential to Differentiation are Related to Oocyte Developmental Competence and Maturational Capacity In Vivo and In Vitro. Genes. 2020; 11(7):815. https://doi.org/10.3390/genes11070815
Chicago/Turabian StyleKulus, Magdalena, Wiesława Kranc, Michal Jeseta, Patrycja Sujka-Kordowska, Aneta Konwerska, Sylwia Ciesiółka, Piotr Celichowski, Lisa Moncrieff, Ievgeniia Kocherova, Małgorzata Józkowiak, and et al. 2020. "Cortical Granule Distribution and Expression Pattern of Genes Regulating Cellular Component Size, Morphogenesis, and Potential to Differentiation are Related to Oocyte Developmental Competence and Maturational Capacity In Vivo and In Vitro" Genes 11, no. 7: 815. https://doi.org/10.3390/genes11070815
APA StyleKulus, M., Kranc, W., Jeseta, M., Sujka-Kordowska, P., Konwerska, A., Ciesiółka, S., Celichowski, P., Moncrieff, L., Kocherova, I., Józkowiak, M., Kulus, J., Wieczorkiewicz, M., Piotrowska-Kempisty, H., Skowroński, M. T., Bukowska, D., Machatkova, M., Hanulakova, S., Mozdziak, P., Jaśkowski, J. M., ... Antosik, P. (2020). Cortical Granule Distribution and Expression Pattern of Genes Regulating Cellular Component Size, Morphogenesis, and Potential to Differentiation are Related to Oocyte Developmental Competence and Maturational Capacity In Vivo and In Vitro. Genes, 11(7), 815. https://doi.org/10.3390/genes11070815








